Copper-Silver Nanohybrids: SARS-CoV-2 Inhibitory Surfaces
Abstract
:1. Introduction
2. Results and Discussion
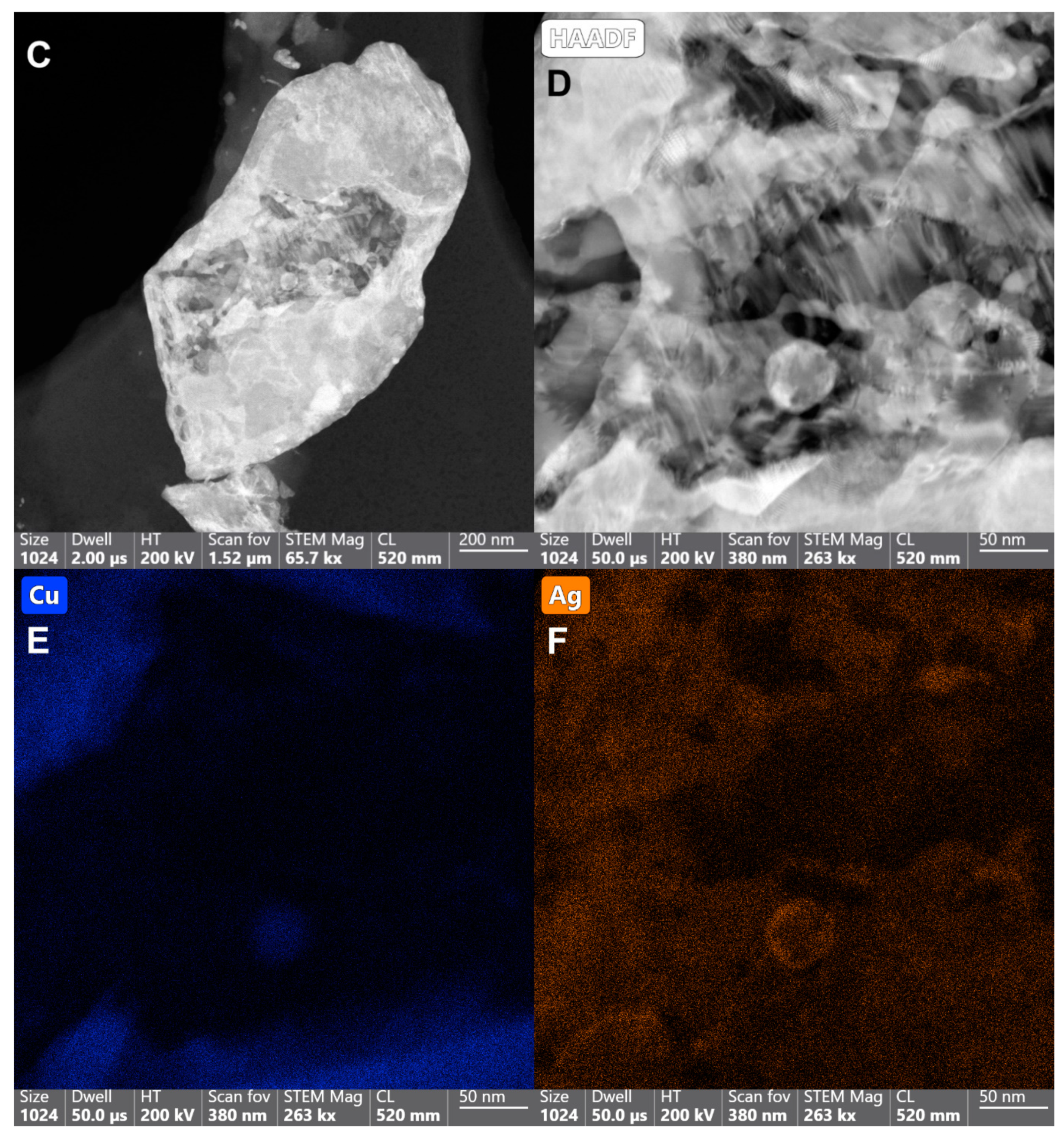
2.1. Characterization of Copper-Silver Nanohybrids’ Samples
2.2. Anti-SARS-CoV-2 Effects at Surface Exposure
3. Conclusions and Outlook
4. Methods
4.1. Copper-Silver Nanohybrid Powders and Plated Surfaces
4.2. Characterization of Copper-Silver Nanohybrids’ Samples
4.3. Viral Strain, Cell Culture, Anti-SARS-CoV-2 Surface Exposure Tests
4.3.1. Cytopathic Effects (CPE)
4.3.2. Real-Time Reverse-Transcription Polymerase Chain Reaction (RT-PCR)
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Acknowledgments
Conflicts of Interest
References
- World Health Organization. Coronavirus Disease (COVID-19): Weekly Epidemiological Update; WHO: Geneva, Switzerland, 2021. [Google Scholar] [CrossRef]
- OIE Events in Animals: World Organisation for Animal Health. Available online: https://www.oie.int/en/scientific-expertise/specific-information-and-recommendations/questions-and-answers-on-2019novel-coronavirus/events-in-animals/ (accessed on 2 March 2021).
- Oude Munnink, B.B.; Sikkema, R.S.; Nieuwenhuijse, D.F.; Jan Molenaar, R.; Munger, E.; Molenkamp, R.; van der Spek, A.; Tolsma, P.; Brouwer, M.; Bouwmeester-Vincken, N.; et al. Transmission of SARS-CoV-2 on Mink Farms between Humans and Mink and Back to Humans. Science 2020, 371, 172–177. [Google Scholar] [CrossRef]
- Koopmans, M. SARS-CoV-2 and the human-animal interface: Outbreaks on mink farms. Lancet Infect. Dis. 2021, 21, 18–19. [Google Scholar] [CrossRef]
- Editorial. Let’s talk about lipid nanoparticles. Nat. Rev. Mater. 2021, 6, 99. [Google Scholar] [CrossRef]
- Machhi, J.; Shahjin, F.; Das, S.; Paetl, M.; Abdelmoaty, M.M.; Cohen, J.D.; Singh, P.A.; Baldi, A.; Bajwa, N.; Kumar, R.; et al. Nanocarrier Vaccines for SARS-CoV-2. Adv. Drug Deliv. Rev. 2021. [Google Scholar] [CrossRef]
- Mosselhy, D.; Virtanen, J.; Kant, R.; He, W.; Elbahri, M.; Sironen, T. COVID-19 Pandemic: What about the Safety of Anti-Coronavirus Nanoparticles? Nanomaterials 2021, 11, 796. [Google Scholar] [CrossRef] [PubMed]
- Hoffmann, M.; Arora, P.; Groß, R.; Seidel, A.; Hörnich, B.F.; Hahn, A.S.; Krüger, N.; Graichen, L.; Hofmann-Winkler, H.; Kempf, A.; et al. SARS-CoV-2 variants B.1.351 and P.1 escape from neutralizing antibodies. Cell 2021, 184, 2384–2393. [Google Scholar] [CrossRef] [PubMed]
- Centers for Disease Control and Prevention (CDC). Science Brief: SARS-CoV-2 and Surface (Fomite) Transmission for Indoor Community Environments. Available online: https://www.cdc.gov/coronavirus/2019-ncov/more/science-and-research/surface-transmission.html (accessed on 27 May 2021).
- Chia, P.Y.; Coleman, K.K.; Tan, Y.K.; Ong, S.W.X.; Gum, M.; Lau, S.K.; Lim, X.F.; Lim, A.S.; Sutjipto, S.; Lee, P.H.; et al. Detection of air and surface contamination by SARS-CoV-2 in hospital rooms of infected patients. Nat. Commun. 2020, 11, 2800. [Google Scholar] [CrossRef] [PubMed]
- Greenhalgh, T.; Jimenez, J.L.; Prather, K.; Tufekci, Z.; Fisman, D.; Schooley, R. Ten scientific reasons in support of airborne transmission of SARS-CoV-2. Lancet 2021, 397, 1603–1605. [Google Scholar] [CrossRef]
- Bedrosian, N.; Mitchell, E.; Rohm, E.; Rothe, M.; Kelly, C.; String, G.; Lantagne, D. A Systematic Review of Surface Contamination, Stability, and Disinfection Data on SARS-CoV-2 (Through July 10, 2020). Environ. Sci. Technol. 2021, 55, 4162–4173. [Google Scholar] [CrossRef]
- Virtanen, J.; Aaltonen, K.; Kivistö, I.; Sironen, T. Survival of SARS-CoV-2 on Clothing Materials. Adv. Virol. 2021, 2021, 1–5. [Google Scholar] [CrossRef]
- Chin, A.W.H.; Chu, J.T.S.; Perera, M.R.; Hui, K.P.Y.; Yen, H.-L.; Chan, M.C.W.; Peiris, M.; Poon, L.L.M. Stability of SARS-CoV-2 in different environmental conditions. Lancet Microbe 2020, 1, e10. [Google Scholar] [CrossRef]
- Van Doremalen, N.; Bushmaker, T.; Morris, D.H.; Holbrook, M.G.; Gamble, A.; Williamson, B.N.; Tamin, A.; Harcourt, J.L.; Thornburg, N.J.; Gerber, S.I.; et al. Aerosol and Surface Stability of SARS-CoV-2 as Compared with SARS-CoV-1. N. Engl. J. Med. 2020, 382, 1564–1567. [Google Scholar] [CrossRef] [PubMed]
- Warnes, S.L.; Keevil, C. Inactivation of Norovirus on Dry Copper Alloy Surfaces. PLoS ONE 2013, 8, e75017. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hodek, J.; Zajícová, V.; Lovětinská-Šlamborová, I.; Stibor, I.; Müllerová, J.; Weber, J. Protective hybrid coating containing silver, copper and zinc cations effective against human immunodeficiency virus and other enveloped viruses. BMC Microbiol. 2016, 16, 1–12. [Google Scholar] [CrossRef] [Green Version]
- Zerbib, S.; Vallet, L.; Muggeo, A.; de Champs, C.; Lefebvre, A.; Jolly, D.; Kanagaratnam, L. Copper for the Prevention of Outbreaks of Health Care–Associated Infections in a Long-term Care Facility for Older Adults. J. Am. Med. Dir. Assoc. 2020, 21, 68–71. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Talebian, S.; Wallace, G.G.; Schroeder, A.; Stellacci, F.; Conde, J. Nanotechnology-based disinfectants and sensors for SARS-CoV-2. Nat. Nanotechnol. 2020, 15, 618–621. [Google Scholar] [CrossRef] [PubMed]
- Mantlo, E.K.; Paessler, S.; Seregin, A.; Mitchell, A. Luminore CopperTouch™ surface coating effectively inactivates SARS-CoV-2, Ebola, and Marburg viruses in vitro. MedRxiv 2020, 1–18. [Google Scholar] [CrossRef]
- Balagna, C.; Perero, S.; Percivalle, E.; Nepita, E.V.; Ferraris, M. Virucidal effect against coronavirus SARS-CoV-2 of a silver nanocluster/silica composite sputtered coating. Open Ceram. 2020, 1, 100006. [Google Scholar] [CrossRef]
- Cortes, A.A.; Zuñiga, J.M. The use of copper to help prevent transmission of SARS-coronavirus and influenza viruses. A general review. Diagn. Microbiol. Infect. Dis. 2020, 98, 115176. [Google Scholar] [CrossRef]
- Fujimori, Y.; Sato, T.; Hayata, T.; Nagao, T.; Nakayama, M.; Nakayama, T.; Sugamata, R.; Suzuki, K. Novel Antiviral Characteristics of Nanosized Copper(I) Iodide Particles Showing Inactivation Activity against 2009 Pandemic H1N1 Influenza Virus. Appl. Environ. Microbiol. 2012, 78, 951–955. [Google Scholar] [CrossRef] [Green Version]
- Minoshima, M.; Lu, Y.; Kimura, T.; Nakano, R.; Ishiguro, H.; Kubota, Y.; Hashimoto, K.; Sunada, K. Comparison of the antiviral effect of solid-state copper and silver compounds. J. Hazard. Mater. 2016, 312, 1–7. [Google Scholar] [CrossRef]
- Das Jana, I.; Kumbhakar, P.; Banerjee, S.; Gowda, C.C.; Kedia, N.; Kuila, S.K.; Banerjee, S.; Das, N.C.; Das, A.K.; Manna, I.; et al. Copper Nanoparticle–Graphene Composite-Based Transparent Surface Coating with Antiviral Activity against Influenza Virus. ACS Appl. Nano Mater. 2021, 4, 352–362. [Google Scholar] [CrossRef]
- Noyce, J.O.; Michels, H.; Keevil, C.W. Inactivation of Influenza A Virus on Copper versus Stainless Steel Surfaces. Appl. Environ. Microbiol. 2007, 73, 2748–2750. [Google Scholar] [CrossRef] [Green Version]
- United States Environmental Protection Agency Pesticides: Topical & Chemical Fact Sheets EPA Registers Copper-Containing Alloy Products. Pesticides Tropical Chemical Fact Sheets. 2008. Available online: https://www.trimcohardware.com/wp-content/uploads/2015/07/EPA-Copper-Registration.pdf (accessed on 7 May 2021).
- Hutasoit, N.; Kennedy, B.; Hamilton, S.; Luttick, A.; Rashid, R.A.R.; Palanisamy, S. SARS-CoV-2 (COVID-19) Inactivation Capability of Copper-coated Touch Surface Fabricated by Cold-spray Technology. Manuf. Lett. 2020, 25, 93–97. [Google Scholar] [CrossRef]
- Behzadinasab, S.; Chin, A.; Hosseini, M.; Poon, L.; Ducker, W.A. A Surface Coating that Rapidly Inactivates SARS-CoV-2. ACS Appl. Mater. Interfaces 2020, 12, 34723–34727. [Google Scholar] [CrossRef] [PubMed]
- Kheirabad, A.K.; Pan, X.; Long, S.; Kochovski, Z.; Zhou, S.; Lu, Y.; McInerney, G.; Yuan, J. Colloidal dispersion of poly(ionic liquid)/Cu composite particles for protective surface coating against SAR-CoV-2. Nano Sel. 2021. [Google Scholar] [CrossRef]
- Mosselhy, D.A.; Assad, M.A.; Sironen, T.; Elbahri, M. Could Nanotheranostics be the Answer to the Coronavirus Crisis? Glob. Chall. 2021, 5, 2000112. [Google Scholar] [CrossRef] [PubMed]
- Du, T.; Liang, J.; Dong, N.; Lu, J.; Fu, Y.; Fang, L.; Xiao, S.; Han, H. Glutathione-Capped Ag2S Nanoclusters Inhibit Coronavirus Proliferation through Blockage of Viral RNA Synthesis and Budding. ACS Appl. Mater. Interfaces 2018, 10, 4369–4378. [Google Scholar] [CrossRef]
- Selwyn, G.S.; Ye, C.; Bradfute, S.B. Anti-SARS-CoV-2 Activity of Surgical Masks Infused with Quaternary Ammonium Salts. Viruses 2021, 13, 960. [Google Scholar] [CrossRef]
- Corman, V.M.; Landt, O.; Kaiser, M.; Molenkamp, R.; Meijer, A.; Chu, D.K.; Bleicker, T.; Brünink, S.; Schneider, J.; Schmidt, M.L.; et al. Detection of 2019 novel coronavirus (2019-nCoV) by real-time RT-PCR. Eurosurveillance 2020, 25, 2000045. [Google Scholar] [CrossRef] [PubMed] [Green Version]



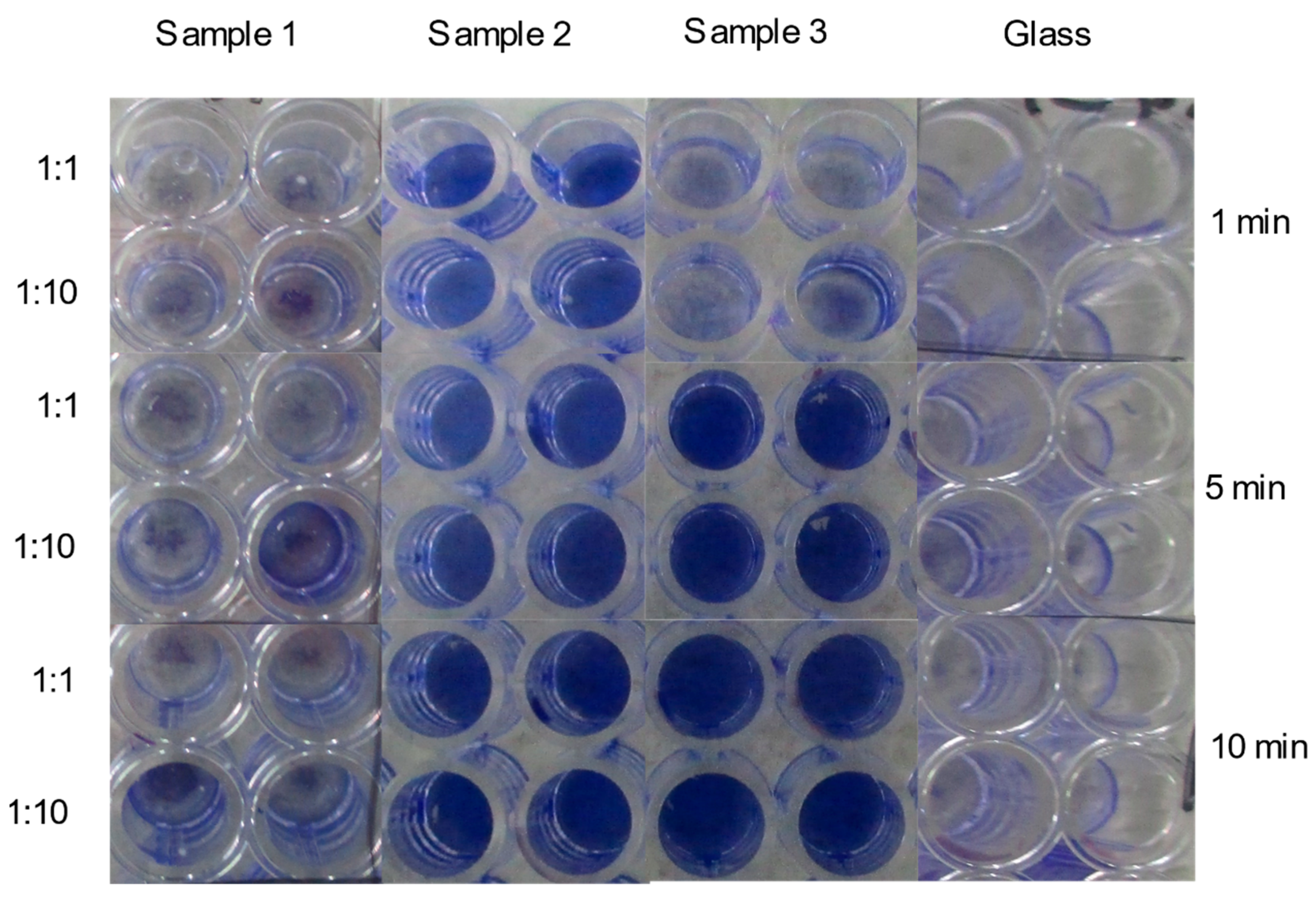
| 1 min | 5 min | 10 min | |||||||
|---|---|---|---|---|---|---|---|---|---|
| Mean | SD | Result | Mean | SD | Result | Mean | SD | Result | |
| Sample 1 | 16.08 | 0.58 | pos | 16.95 | 0.10 | pos | 16.40 | 1.29 | pos |
| Sample 2 | 38.07 | 1.79 | neg | NA | NA | neg | 36.71 | 1.10 | neg |
| Sample 3 | 16.60 | 0.45 | pos | 35.38 | 2.00 | neg | 36.03 | 2.76 | neg |
| Glass | 16.39 | 0.40 | pos | 21.06 | 9.36 | pos | 17.96 | 3.87 | pos |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mosselhy, D.A.; Kareinen, L.; Kivistö, I.; Aaltonen, K.; Virtanen, J.; Ge, Y.; Sironen, T. Copper-Silver Nanohybrids: SARS-CoV-2 Inhibitory Surfaces. Nanomaterials 2021, 11, 1820. https://doi.org/10.3390/nano11071820
Mosselhy DA, Kareinen L, Kivistö I, Aaltonen K, Virtanen J, Ge Y, Sironen T. Copper-Silver Nanohybrids: SARS-CoV-2 Inhibitory Surfaces. Nanomaterials. 2021; 11(7):1820. https://doi.org/10.3390/nano11071820
Chicago/Turabian StyleMosselhy, Dina A., Lauri Kareinen, Ilkka Kivistö, Kirsi Aaltonen, Jenni Virtanen, Yanling Ge, and Tarja Sironen. 2021. "Copper-Silver Nanohybrids: SARS-CoV-2 Inhibitory Surfaces" Nanomaterials 11, no. 7: 1820. https://doi.org/10.3390/nano11071820
APA StyleMosselhy, D. A., Kareinen, L., Kivistö, I., Aaltonen, K., Virtanen, J., Ge, Y., & Sironen, T. (2021). Copper-Silver Nanohybrids: SARS-CoV-2 Inhibitory Surfaces. Nanomaterials, 11(7), 1820. https://doi.org/10.3390/nano11071820







