Silver Nanoparticles from Annona muricata Peel and Leaf Extracts as a Potential Potent, Biocompatible and Low Cost Antitumor Tool
Abstract
1. Introduction
2. Materials and Methods
2.1. Collection and Conservation of Leaves and Peel of A. muricata
2.2. Biosynthesis of Silver Nanoparticles with Extracts of A. muricata
2.2.1. Preparation of Aqueous Leaf Extract (LE) and Fruit Peel (PE) from A. muricata
2.2.2. Synthesis of Silver Nanoparticles
2.3. Characterization
2.3.1. Characterization of Optical Properties of AgNPs
2.3.2. Morphological and Structural Characterization of AgNPs
2.3.3. Characterization of the Main Functional Groups Present in the Extracts of Annona muricata and AgNPs
2.3.4. Characterization of the Zeta Potential
2.4. Cell Viability Assay
2.4.1. Cell Lines and Cytotoxicity Assays
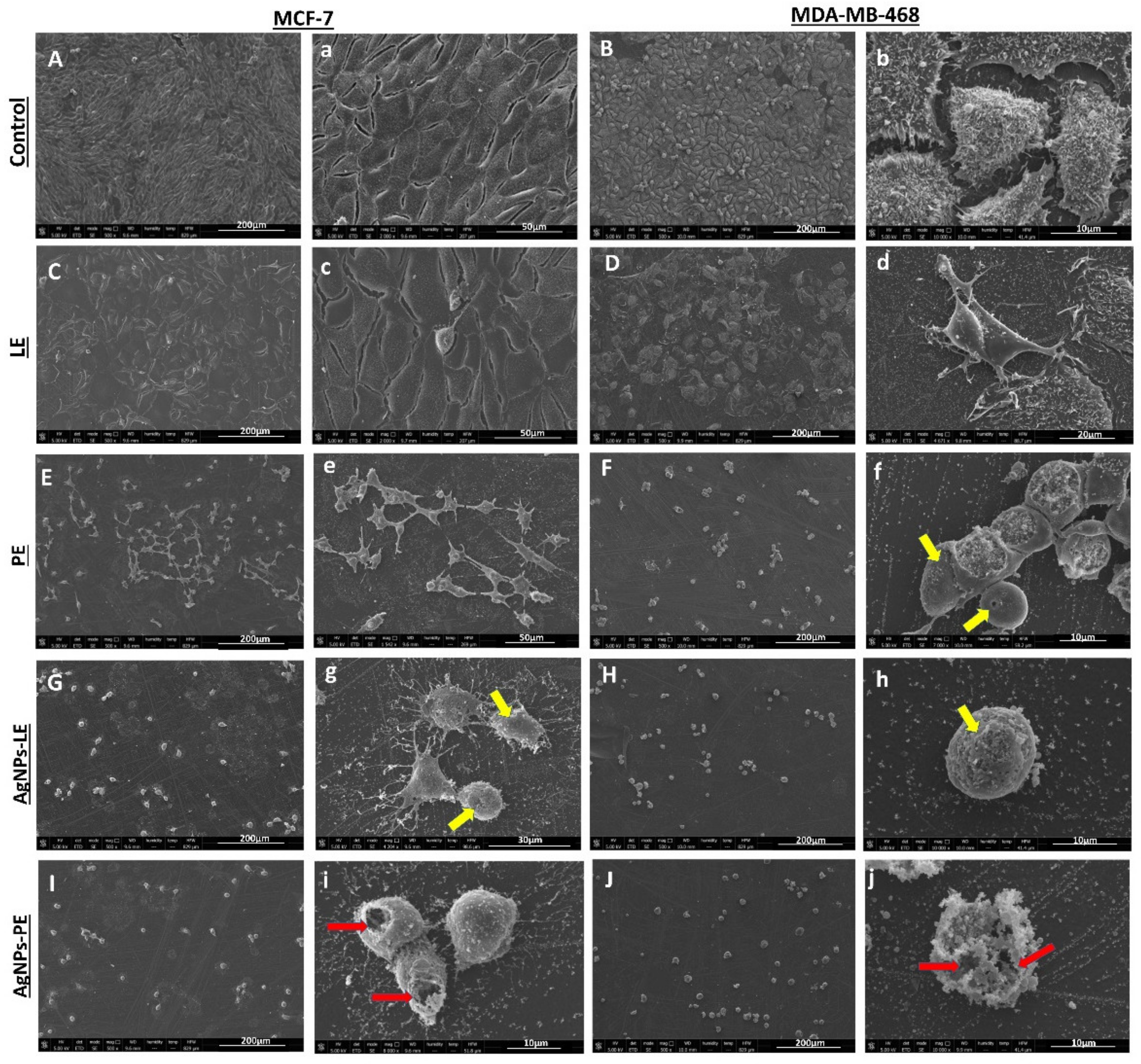
2.4.2. Morphological Findings
2.4.3. Statistical Analysis
3. Results and Discussion
3.1. Synthesis of Silver Nanoparticles (AgNPs)
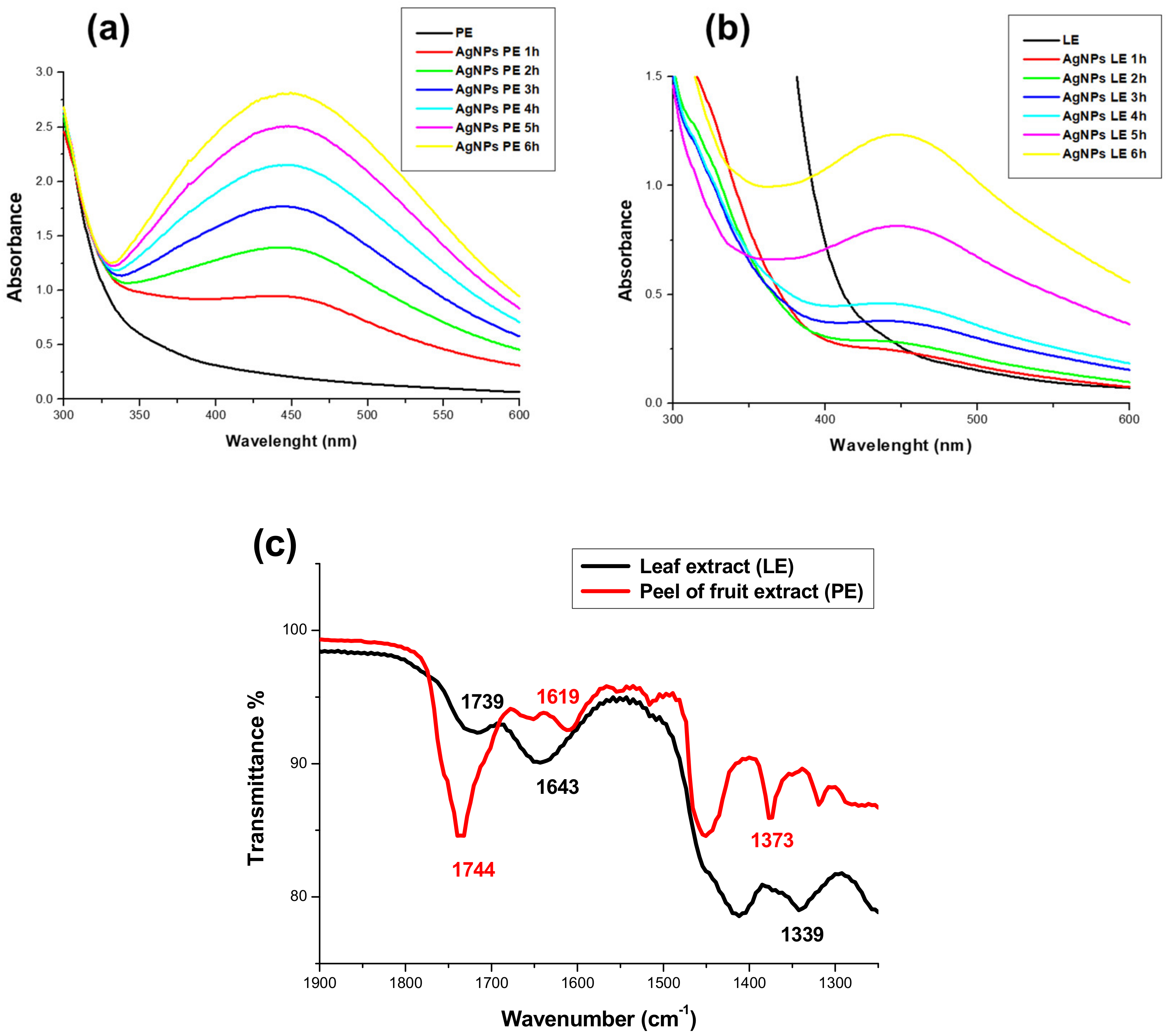
3.2. Spectroscopic Characterization
3.2.1. Spectroscopic Characterization (UV-Vis)
3.2.2. Spectroscopic Characterization (FTIR) and Z Potential
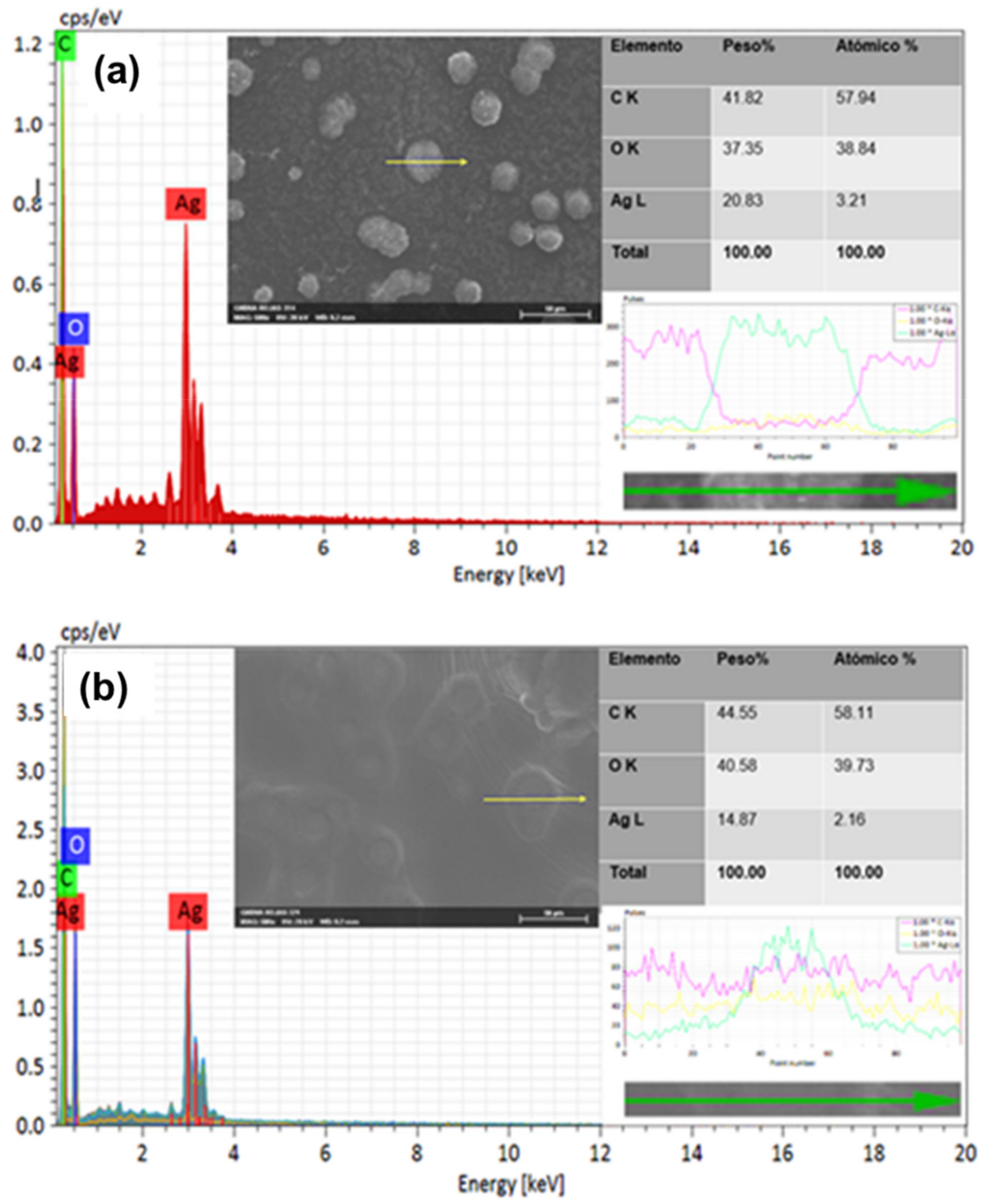
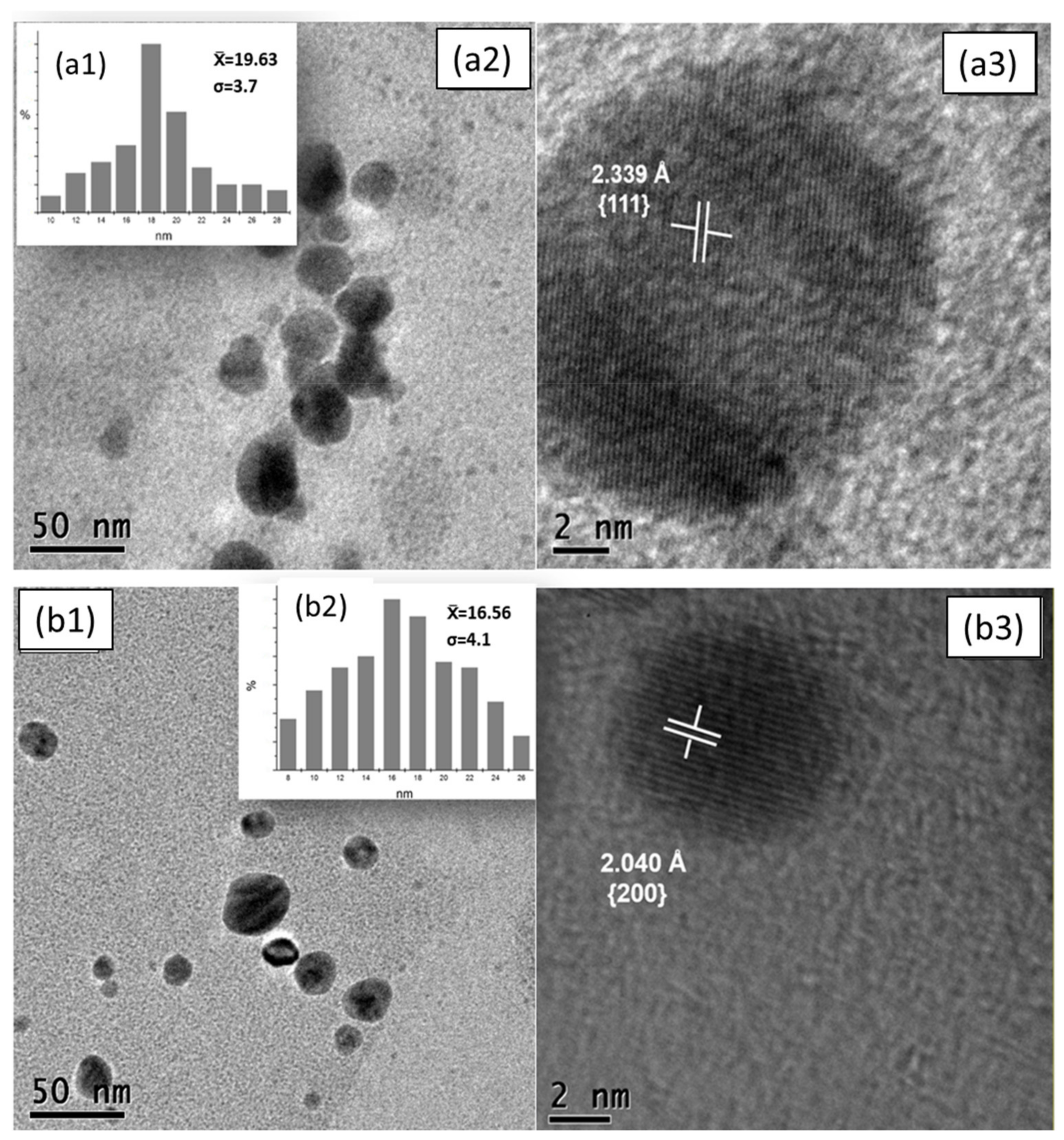
3.3. Morphological and Structural Characterization
3.3.1. Elemental Analysis (SEM-EDS)
3.3.2. Morphological Analysis (TEM)
3.4. Cell Viability Test
4. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Siegel, R.L.; Miller, K.D.; Fuchs, H.E.; Jemal, A. Cancer Statistics, 2021. CA Cancer J. Clin. 2021, 71, 7–33. [Google Scholar] [CrossRef]
- Sung, H.; Ferlay, J.; Siegel, R.L.; Laversanne, M.; Soerjomataram, I.; Jemal, A.; Bray, F. Global cancer statistics 2020: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J. Clin. 2021, 71, 209–249. [Google Scholar] [CrossRef]
- Vasan, N.; Baselga, J.; Hyman, D.M. A view on drug resistance in cancer. Nat. Cell Biol. 2019, 575, 299–309. [Google Scholar] [CrossRef] [PubMed]
- Boulaiz, H.; Alvarez, P.J.; Ramirez, A.; Marchal, J.A.; Prados, J.; Rodríguez-Serrano, F.; Perán, M.; Melguizo, C.; Aranega, A. Nanomedicine: Application Areas and Development Prospects. Int. J. Mol. Sci. 2011, 12, 3303–3321. [Google Scholar] [CrossRef]
- Chernousova, S.; Epple, M. Silver as Antibacterial Agent: Ion, Nanoparticle, and Metal. Angew. Chem. Int. Ed. 2013, 52, 1636–1653. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.-F.; Liu, Z.-G.; Shen, W.; Gurunathan, S. Silver nanoparticles: Synthesis, characterization, proper-ties, applications, and therapeutic approaches. Int. J. Mol. Sci. 2016, 17, 1534. [Google Scholar] [CrossRef] [PubMed]
- Elsupikhe, R.F.; Shameli, K.; Ahmad, M.B.; Ibrahim, N.A.; Zainudin, N. Green sonochemical synthesis of silver nanoparticles at varying concentrations of κ-carrageenan. Nanoscale Res. Lett. 2015, 10, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Shameli, K.; Ahmad, M.B.; Yunus, W.M.Z.W.; Ibrahim, N.A.; Gharayebi, Y.; Sedaghat, S. Synthesis of silver/montmorillonite nanocomposites using γ-irradiation. Int. J. Nanomed. 2010, 5, 1067. [Google Scholar] [CrossRef]
- Deepak, V.; Umamaheshwaran, P.S.; Guhan, K.; Nanthini, R.A.; Krithiga, B.; Jaithoon, N.M.H.; Gurunathan, S. Synthesis of gold and silver nanoparticles using purified URAK. Colloids Surf. B Biointerfaces 2011, 86, 353–358. [Google Scholar] [CrossRef]
- Gurunathan, S.; Han, J.W.; Park, J.-H.; Kim, E.S.; Choi, Y.-J.; Kwon, D.-N.; Kim, J.-H. Reduced graphene oxide–silver nanoparticle nanocomposite: A potential anticancer nanotherapy. Int. J. Nanomed. 2015, 10, 6257–6276. [Google Scholar] [CrossRef]
- Gurunathan, S.; Han, J.; Park, J.H.; Kim, J.-H. A green chemistry approach for synthesizing biocompatible gold nanoparticles. Nanoscale Res. Lett. 2014, 9, 248. [Google Scholar] [CrossRef] [PubMed]
- Kalimuthu, K.; Babu, R.S.; Venkataraman, D.; Bilal, M.; Gurunathan, S. Biosynthesis of silver nanocrystals by Bacillus licheniformis. Colloids Surf. B Biointerfaces 2008, 65, 150–153. [Google Scholar] [CrossRef] [PubMed]
- Javed, B.; Ikram, M.; Farooq, F.; Sultana, T.; Mashwani, Z.-U.-R.; Raja, N.I. Biogenesis of silver nanoparticles to treat cancer, diabetes, and microbial infections: A mechanistic overview. Appl. Microbiol. Biotechnol. 2021, 105, 2261–2275. [Google Scholar] [CrossRef]
- Alonso-Castro, A.J.; Villarreal, M.L.; Salazar-Olivo, L.A.; Gomez-Sanchez, M.; Dominguez, F.; Garcia-Carranca, A. Mexican medicinal plants used for cancer treatment: Pharmacological, phytochemical and ethnobotanical studies. J. Ethnopharmacol. 2011, 133, 945–972. [Google Scholar] [CrossRef] [PubMed]
- Kitamura, M.C.; Lemos, E.E.P. Enxertia precoce da gravioleira (Annona muricata L.). Rev. Bras. Frutic. 2004, 26, 186–188. [Google Scholar] [CrossRef]
- Moghadamtousi, S.; Fadaeinasab, M.; Nikzad, S.; Mohan, G.; Ali, H.; Kadir, H. Annona muricata (An-nonaceae): A review of its traditional uses, isolated acetogenins and biological activities. Int. J. Mol. Sci. 2015, 16, 15625–15658. [Google Scholar] [CrossRef] [PubMed]
- Fuentes, L.M.H.; Vildózola, A.C.; Santos, M.O.; Carrasco, J.V. El Chacuatete, Idiarthron subquadratum, en Guanábana, Annona muricata. Southwest. Ãntomol. 2016, 41, 855–864. [Google Scholar] [CrossRef]
- Correa-Gordillo, J.; Ortiz, D.; Larrahondo, J.; Sánchez-Mejía, M.; Pachón, H. Antioxidant activity in soursop (Annona muricata L.): A literature review. BLACPMA 2012, 11, 111–126. [Google Scholar]
- George, V.C.; Kumar, D.N.; Suresh, P.; Kumar, R.A. Antioxidant, DNA protective efficacy and HPLC analysis of Annona muricata (soursop) extracts. J. Food Sci. Technol. 2015, 52, 2328–2335. [Google Scholar] [CrossRef]
- Mittal, A.K.; Chisti, Y.; Banerjee, U.C. Synthesis of metallic nanoparticles using plant extracts. Biotechnol. Adv. 2013, 31, 346–356. [Google Scholar] [CrossRef]
- Schlie-Guzmán, M.A.; González-Esquinca, A.R.; Luna-Cazáres, L.M. Acetogenins from Annonaceae: Anti-proliferative effect in neoplastic cell lines. Lat. Am. Caribb. Bull. Med. Aromat. Plants 2009, 8, 245–257. [Google Scholar]
- Moghadamtousi, S.Z.; Karimian, H.; Rouhollahi, E.; Paydar, M.; Fadaeinasab, M.; Kadir, H.A. Annona muricata leaves induce G 1 cell cycle arrest and apoptosis through mitochondria-mediated pathway in human HCT-116 and HT-29 colon cancer cells. J. Ethnopharmacol. 2014, 156, 277–289. [Google Scholar] [CrossRef]
- Gajalakshmi, S.; Vijayalakshmi, S.; Devi, R.V. Phytochemical and pharmacological properties of Annona muricata: A review. Int. J. Pharm. Pharm. Sci. 2012, 4, 3–6. [Google Scholar]
- Gavamukulya, Y.; Abou-Elella, F.; Wamunyokoli, F.; Ael-Shemy, H. Phytochemical screening, antioxidant activity and in vitro anticancer potential of ethanolic and water leaves extracts of Annona muricata (Graviola). Asian Pac. J. Trop. Med. 2014, 7, S355–S363. [Google Scholar] [CrossRef]
- Gajendran, B.; Chinnasamy, A.; Durai, P.; Raman, J.; Ramar, M. Biosynthesis and characterization of silver nanoparticles from Datura inoxia and its apoptotic effect on human breast cancer cell line MCF7. Mater. Lett. 2014, 122, 98–102. [Google Scholar] [CrossRef]
- Cáceres, B.; Ramirez, A.; Carrillo, E.; Jimenez, G.; Griñán-Lisón, C.; López-Ruiz, E.; Jiménez-Martínez, Y.; Marchal, J.A.; Boulaiz, H. Deciphering the Mechanism of Action Involved in Enhanced Suicide Gene Colon Cancer Cell Killer Effect Mediated by Gef and Apoptin. Cancers 2019, 11, 264. [Google Scholar] [CrossRef]
- Rajan, R.; Chandran, K.; Harper, S.L.; Yun, S.-I.; Kalaichelvan, P.T. Plant extract synthesized silver nanoparticles: An ongoing source of novel biocompatible materials. Ind. Crop. Prod. 2015, 70, 356–373. [Google Scholar] [CrossRef]
- Santhosh, S.; Yuvarajan, R.; Natarajan, D. Annona muricata leaf extract-mediated silver nanoparticles synthesis and its larvicidal potential against dengue, malaria and filariasis vector. Parasitol. Res. 2015, 114, 3087–3096. [Google Scholar] [CrossRef] [PubMed]
- Jain, P.K.; El-Sayed, I.H.; El-Sayed, M.A. Au nanoparticles target cancer. Nano Today 2007, 2, 18–29. [Google Scholar] [CrossRef]
- Morales-Luckie, R.A.; Sánchez-Mendieta, V.; Olea-Mejia, O.; Vilchis-Nestor, A.R.; López-Téllez, G.; Varela-Guerrero, V.; Huerta, L.; Arenas-Alatorre, J. Facile solventless synthesis of a nylon-6, 6/silver nano-particles composite and its XPS study. Int. J. Polym. Sci. 2013, 2013. [Google Scholar] [CrossRef]
- Mallikarjuna, K.; Narasimha, G.; Dillip, G.; Praveen, B.; Shreedhar, B.; Lakshmi, C.S.; Reddy, B.; Raju, B.D.P. Green synthesis of silver nanoparticles using Ocimum leaf extract and their characterization. Dig. J. Nanomater. Biostruct. 2011, 6, 181–186. [Google Scholar]
- Kumar, R.; Roopan, S.M.; Prabhakarn, A.; Khanna, V.G.; Chakroborty, S. Agricultural waste Annona squa-mosa peel extract: Biosynthesis of silver nanoparticles. Spectrochim. Acta Part A Mol. Biomol. Spectrosc. 2012, 90, 173–176. [Google Scholar] [CrossRef]
- Vivek, R.; Thangam, R.; Muthuchelian, K.; Gunasekaran, P.; Kaveri, K.; Kannan, S. Green biosynthesis of silver nanoparticles from Annona squamosa leaf extract and its in vitro cytotoxic effect on MCF-7 cells. Process. Biochem. 2012, 47, 2405–2410. [Google Scholar] [CrossRef]
- Hosseini-Koupaei, M.; Shareghi, B.; Saboury, A.A.; Davar, F.; Sirotkin, V.A.; Hosseini-Koupaei, M.H.; Enteshari, Z. Catalytic activity, structure and stability of proteinase K in the presence of biosynthesized CuO nanoparticles. Int. J. Biol. Macromol. 2019, 122, 732–744. [Google Scholar] [CrossRef]
- Nasrollahzadeh, M.; Sajjadi, M.; Dadashi, J.; Ghafuri, H. Pd-based nanoparticles: Plant-assisted biosynthesis, characterization, mechanism, stability, catalytic and antimicrobial activities. Adv. Colloid Interface Sci. 2020, 276, 102103. [Google Scholar] [CrossRef]
- Jadhav, K.; Dhamecha, D.; Bhattacharya, D.; Patil, M. Green and ecofriendly synthesis of silver nanoparticles: Characterization, biocompatibility studies and gel formulation for treatment of infections in burns. J. Photochem. Photobiol. B Biol. 2016, 155, 109–115. [Google Scholar] [CrossRef] [PubMed]
- Garcia, K. Isolation and Structural Characterization of Acetogenins Obtained from Annona cherimolia and Annona muricata Seeds. Genotoxic Evaluation and Chemotherapeutic Potential. Ph.D. Thesis, National School Biological Deficiencies Mexico, National Polytechnic Institute, Mexico City, Mexico, 2009. [Google Scholar]
- Rieser, M.J.; Gu, Z.-M.; Fang, X.-P.; Zeng, L.; Wood, K.V.; McLaughlin, J.L. Five Novel Mono-tetrahydrofuran Ring Acetogenins from the Seeds of Annona muricata. J. Nat. Prod. 1996, 59, 100–108. [Google Scholar] [CrossRef] [PubMed]
- Ivanov, M.R.; Bednar, H.R.; Haes, A.J. Investigations of the Mechanism of Gold Nanoparticle Stability and Surface Functionalization in Capillary Electrophoresis. ACS Nano 2009, 3, 386–394. [Google Scholar] [CrossRef] [PubMed]
- Durán, N.; Marcato, P.D.; de Souza, G.I.H.; Alves, O.L.; Esposito, E. Antibacterial Effect of Silver Nanoparticles Produced by Fungal Process on Textile Fabrics and Their Effluent Treatment. J. Biomed. Nanotechnol. 2007, 3, 203–208. [Google Scholar] [CrossRef]
- Kubo, A.M.; Gorup, L.F.; Amaral, L.S.; Rodrigues-Filho, E.; de Camargo, E.R. Heterogeneous Microtubules of Self-assembled Silver and Gold Nanoparticles Using Alive Biotemplates. Mater. Res. 2018, 21, 21. [Google Scholar] [CrossRef]
- Auffan, M.; Rose, J.; Bottero, J.-Y.; Lowry, G.V.; Jolivet, J.-P.; Wiesner, M.R. Towards a definition of inorganic nanoparticles from an environmental, health and safety perspective. Nat. Nanotechnol. 2009, 4, 634–641. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.; Yang, Y.; Mao, L.; Li, Z.; Zhou, C.; Liu, X.; Zheng, S.; Hu, Y. SPR quantitative analysis of direct detection of atrazine traces on Au-nanoparticles: Nanoparticles size effect. Sens. Actuators B Chem. 2015, 218, 1–7. [Google Scholar] [CrossRef]
- El-Sayed, I.H.; Huang, X.; El-Sayed, M.A. Selective laser photo-thermal therapy of epithelial carcinoma us-ing anti-EGFR antibody conjugated gold nanoparticles. Cancer Lett. 2006, 239, 129–135. [Google Scholar] [CrossRef]
- Ahmed, S.; Ahmad, M.; Swami, B.L.; Ikram, S. A review on plants extract mediated synthesis of silver nanoparticles for antimicrobial applications: A green expertise. J. Adv. Res. 2016, 7, 17–28. [Google Scholar] [CrossRef]
- Wahab, S.M.A.; Jantan, I.; Haque, A.; Arshad, L. Exploring the Leaves of Annona muricata L. as a Source of Potential Anti-inflammatory and Anticancer Agents. Front. Pharmacol. 2018, 9, 661. [Google Scholar] [CrossRef] [PubMed]
- Fattahi, S.; Ardekani, A.M.; Zabihi, E.; Abedian, Z.; Mostafazadeh, A.; Pourbagher, R.; Akhavan-Niaki, H. Antioxidant and Apoptotic Effects of an Aqueous Extract of Urtica dioica on the MCF-7 Human Breast Cancer Cell Line. Asian Pac. J. Cancer Prev. 2013, 14, 5317–5323. [Google Scholar] [CrossRef] [PubMed]
- Raghunandan, D.; Ravishankar, B.; Sharanbasava, G.; Mahesh, D.B.; Harsoor, V.; Yalagatti, M.S.; Bhaga-Wanraju, M.; Venkataraman, A. Anti-cancer studies of noble metal nanoparticles synthesized using different plant extracts. Cancer Nanotechnol. 2011, 2, 57–65. [Google Scholar] [CrossRef] [PubMed]
- Kuppusamy, P.; Ichwan, S.J.A.; Al-Zikri, P.N.H.; Suriyah, W.H.; Soundharrajan, I.; Govindan, N.; Maniam, G.P.; Yusoff, M.M. In Vitro Anticancer Activity of Au, Ag Nanoparticles Synthesized Using Commelina nudiflora L. Aqueous Extract Against HCT-116 Colon Cancer Cells. Biol. Trace Element Res. 2016, 173, 297–305. [Google Scholar] [CrossRef]
- Saratale, R.G.; Benelli, G.; Kumar, G.; Kim, D.S.; Saratale, G.D. Bio-fabrication of silver nanoparticles using the leaf extract of an ancient herbal medicine, dandelion (Taraxacum officinale), evaluation of their antioxidant, anticancer potential, and antimicrobial activity against phytopathogens. Environ. Sci. Pollut. Res. 2017, 25, 10392–10406. [Google Scholar] [CrossRef]
- El-Naggar, N.E.-A.; Hussein, M.H.; El-Sawah, A.A. Bio-fabrication of silver nanoparticles by phycocyanin, characterization, in vitro anticancer activity against breast cancer cell line and in vivo cytotxicity. Sci. Rep. 2017, 7, 10844. [Google Scholar] [CrossRef]
- Chavez, K.J.; Garimella, S.V.; Lipkowitz, S. Triple negative breast cancer cell lines: One tool in the search for better treatment of triple negative breast cancer. Breast Dis. 2011, 32, 35–48. [Google Scholar] [CrossRef]
- Jabir, M.; Saleh, Y.; Sulaiman, G.; Yaseen, N.; Sahib, U.; Dewir, Y.; Alwahibi, M.; Soliman, D. Green Synthesis of Silver Nanoparticles Using Annona muricata Extract as an Inducer of Apoptosis in Cancer Cells and Inhibitor for NLRP3 Inflammasome via Enhanced Autophagy. Nanomaterials 2021, 11, 384. [Google Scholar] [CrossRef]
- Valares, M.C. Variación del Metabolismo Secundario en Plantas Debida al Genotipo y al Ambiente; Universidad de Extremadura: Badajoz, Spain, 2011. [Google Scholar]
- Flores, C.L.; Garró, V.; Yrei, V.; Gallardo, T. Acción antimicrobiana Caesalpinea Tintoria (Molina) Kuntze o tara de diferentes regiones del Perú. Cienc. Investig. 1998, 1, 27–31. [Google Scholar] [CrossRef]
- Jarma Orozco, A.; Cardona, A.C.; Araméndiz, T.H. Effect of climate change on the physiology of cultivated plants: A review. Rev. UDCA Act. Div. Cient. 2012, 15, 63–76. [Google Scholar]
- Omoifo, C. Biochemical composition of soursop fruit, Annona muricata L., as affected by two harvest seasons. Trop. Agric. Res. Ext. 2004, 7, 1–8. [Google Scholar]
- Ramírez, A.; Conejo-García, A.; Griñán-Lisón, C.; López-Cara, L.C.; Jiménez, G.; Campos, J.M.; Marchal, J.A.; Boulaiz, H. Enhancement of Tumor Cell Death by Combining gef Gene Mediated Therapy and New 1,4-Benzoxazepin-2,6-Dichloropurine Derivatives in Breast Cancer Cells. Front. Pharmacol. 2018, 9, 798. [Google Scholar] [CrossRef] [PubMed]
- Arpa, T.L. Molecular Mechanisms that Regulate Classical and Alternative Activation in Macrophages. Ph.D. Thesis, Universitat de Barcelona, Barcelona, Spain, 2008. [Google Scholar]
- Raynaud, S.; Némati, F.; Miccoli, L.; Michel, P.; Poupon, M.-F.; Fourneau, C.; Laurens, A.; Hocquemiller, R. Antitumoral effects of squamocin on parental and multidrug resistant MCF7 (human breast adenocarci-noma) cell lines. Life Sci. 1999, 65, 525–533. [Google Scholar] [CrossRef]
- Choi, C.Y.; Kim, J.Y.; Kim, Y.S.; Chung, Y.C.; Seo, J.K.; Jeong, H.G. Aqueous extract isolated from Platycodon grandiflorum elicits the release of nitric oxide and tumor necrosis factor-α from murine macrophages. Int. Immunopharmacol. 2001, 1, 1141–1151. [Google Scholar] [CrossRef]
- Yuan, S.-S.F.; Chang, H.-L.; Chen, H.-W.; Yeh, Y.-T.; Kao, Y.-H.; Lin, K.-H.; Wu, Y.-C.; Su, J.-H. Annonacin, a mono-tetrahydrofuran acetogenin, arrests cancer cells at the G1 phase and causes cytotoxicity in a Bax- and caspase-3-related pathway. Life Sci. 2003, 72, 2853–2861. [Google Scholar] [CrossRef]
- AshaRani, P.; Low, K.M.G.; Hande, M.P.; Valiyaveettil, S. Cytotoxicity and genotoxicity of silver nano-particles in human cells. ACS Nano 2009, 3, 279–290. [Google Scholar] [CrossRef]
- Sonawane, A.; Jena, P.; Mohanty, S.; Mallick, R.; Jacob, B. Toxicity and antibacterial assessment of chitosan-coated silver nanoparticles on human pathogens and macrophage cells. Int. J. Nanomed. 2012, 7, 1805–1818. [Google Scholar] [CrossRef] [PubMed]
- Moutin, M.-J.; Abramson, J.J.; Salama, G.; Dupont, Y. Rapid Ag+-induced release of Ca2+ from sarcoplasmic reticulum vesicles of skeletal muscle: A rapid filtration study. Biochim. Biophys. Acta Biomembr. 1989, 984, 289–292. [Google Scholar] [CrossRef]
- Orrenius, S.; McCabe, M.J., Jr.; Nicotera, P. Ca2+-dependent mechanisms of cytotoxicity and programmed cell death. Toxicol. Lett. 1992, 64, 357–364. [Google Scholar] [CrossRef]
- Sathishkumar, G.; Gobinath, C.; Wilson, A.; Sivaramakrishnan, S. Dendrophthoe falcata (L.f) Ettingsh (Neem mistletoe): A potent bioresource to fabricate silver nanoparticles for anticancer effect against human breast cancer cells (MCF-7). Spectrochim. Acta Part A Mol. Biomol. Spectrosc. 2014, 128, 285–290. [Google Scholar] [CrossRef]

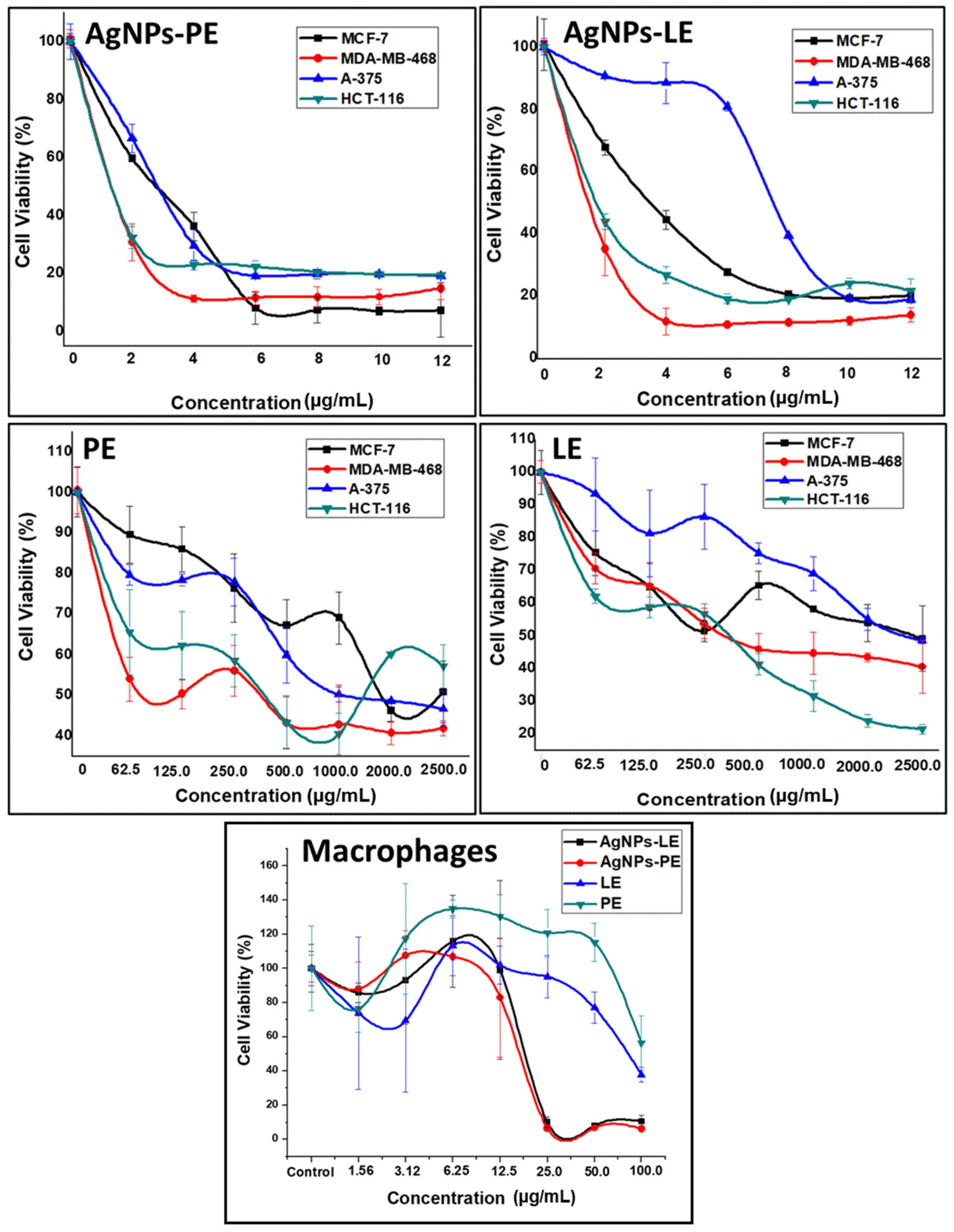
| Treatment | MCF-7 | MDA-MB-468 | A-375 | HCT-116 | Macrophages | |||||
|---|---|---|---|---|---|---|---|---|---|---|
| IC50 | R2 | IC50 | R2 | IC50 | R2 | IC50 | R2 | IC50 | R2 | |
| AgNPs-PE | 2.996 | 0.9309 | 1.685 | 0.9662 | 2.943 | 0.9728 | 1.285 | 0.9623 | 13.7 | 0.9980 |
| AgNPs-LE | 3.109 | 0.9806 | 1.910 | 0.9825 | 8.404 | 0.9672 | 2.004 | 0.9986 | 10.7 | 0.9792 |
| PE | 1278 | 0.9592 | 264.9 | 0.9676 | 1880 | 0.9934 | 309.3 | 0.9731 | - | - |
| LE | 2280 | 0.9803 | 776.4 | 0.9879 | 2478 | 0.9726 | 404.8 | 0.9708 | 68.97 | 0.9674 |
| Treatment | MCF-7 | MDA-MB-468 | A375 | HCT-116 |
|---|---|---|---|---|
| AgNPS-PE | 4.34 | 8.13 | 4.655 | 10.66 |
| AgNPS-LE | 3.44 | 5.602 | 1.273 | 5.245 |
| PE | ND * | ND * | ND * | ND * |
| LE | 0.030 | 0.088 | 0.027 | 0.170 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
González-Pedroza, M.G.; Argueta-Figueroa, L.; García-Contreras, R.; Jiménez-Martínez, Y.; Martínez-Martínez, E.; Navarro-Marchal, S.A.; Marchal, J.A.; Morales-Luckie, R.A.; Boulaiz, H. Silver Nanoparticles from Annona muricata Peel and Leaf Extracts as a Potential Potent, Biocompatible and Low Cost Antitumor Tool. Nanomaterials 2021, 11, 1273. https://doi.org/10.3390/nano11051273
González-Pedroza MG, Argueta-Figueroa L, García-Contreras R, Jiménez-Martínez Y, Martínez-Martínez E, Navarro-Marchal SA, Marchal JA, Morales-Luckie RA, Boulaiz H. Silver Nanoparticles from Annona muricata Peel and Leaf Extracts as a Potential Potent, Biocompatible and Low Cost Antitumor Tool. Nanomaterials. 2021; 11(5):1273. https://doi.org/10.3390/nano11051273
Chicago/Turabian StyleGonzález-Pedroza, María G., Liliana Argueta-Figueroa, René García-Contreras, Yaiza Jiménez-Martínez, Eduardo Martínez-Martínez, Saúl A. Navarro-Marchal, Juan A. Marchal, Raúl A. Morales-Luckie, and Houria Boulaiz. 2021. "Silver Nanoparticles from Annona muricata Peel and Leaf Extracts as a Potential Potent, Biocompatible and Low Cost Antitumor Tool" Nanomaterials 11, no. 5: 1273. https://doi.org/10.3390/nano11051273
APA StyleGonzález-Pedroza, M. G., Argueta-Figueroa, L., García-Contreras, R., Jiménez-Martínez, Y., Martínez-Martínez, E., Navarro-Marchal, S. A., Marchal, J. A., Morales-Luckie, R. A., & Boulaiz, H. (2021). Silver Nanoparticles from Annona muricata Peel and Leaf Extracts as a Potential Potent, Biocompatible and Low Cost Antitumor Tool. Nanomaterials, 11(5), 1273. https://doi.org/10.3390/nano11051273









