Hybrid Nanocomposite Thin Films for Photovoltaic Applications: A Review
Abstract
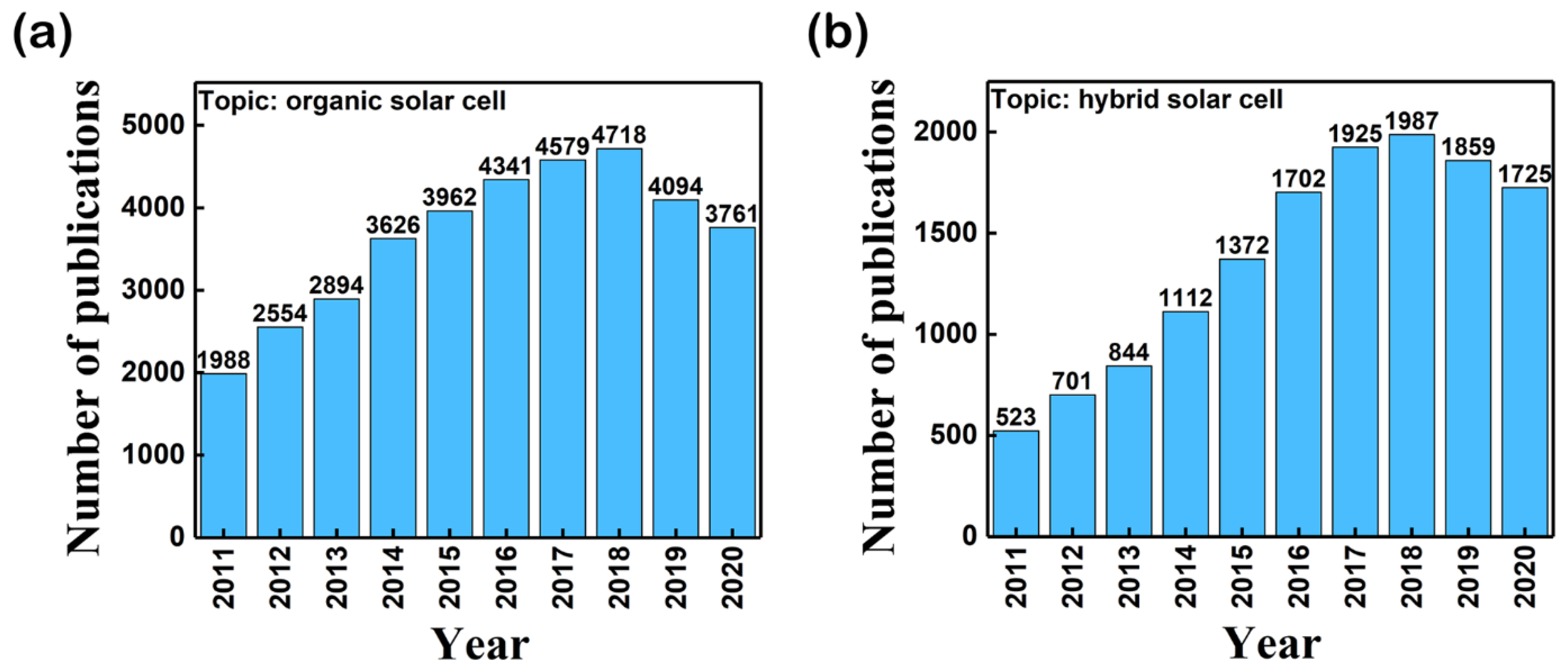
1. Introduction
2. Organic Compounds and Inorganic Nanostructures—Materials for Designing PV Cells
2.1. Organic Compounds for PV Applications
2.1.1. Conducting Polymers and Metal Phthalocyanines as p-Type Materials in the PV Cells
2.1.2. Fullerene Derivatives and Non-Fullerene Compounds as n-Type Materials in the PV Cells
2.2. Inorganic Semiconductor Nanostructures for PV Applications
3. Spin-Coating and Matrix-Assisted Pulsed Laser Evaporation (MAPLE)—Techniques Used in the Deposition of the Hybrid Nanocomposites Thin Films for Developing PV Cells
4. Hybrid Nanocomposite Films Deposited by Spin-Coating for PV Cells
4.1. Metal Oxides Based Nanocomposites
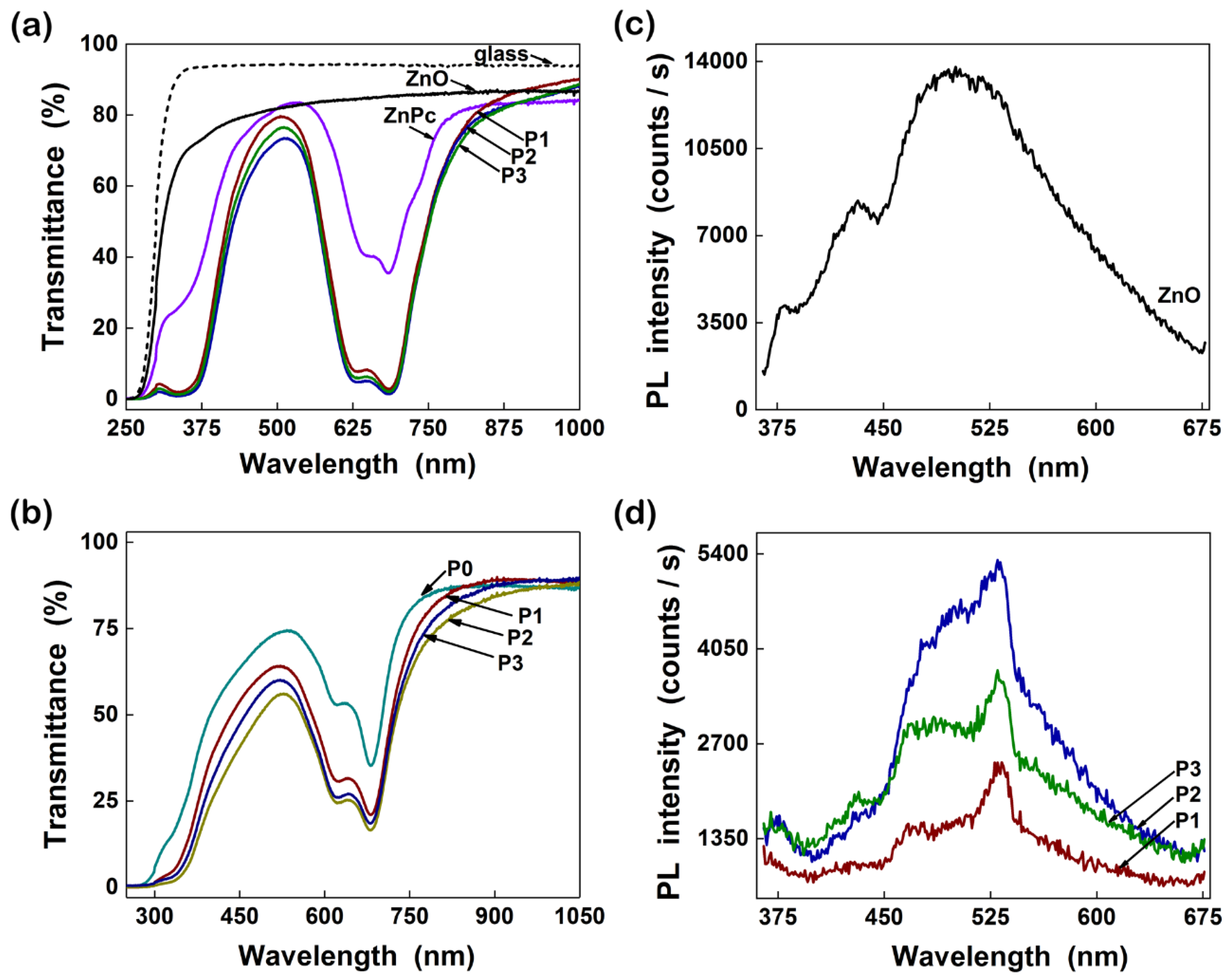
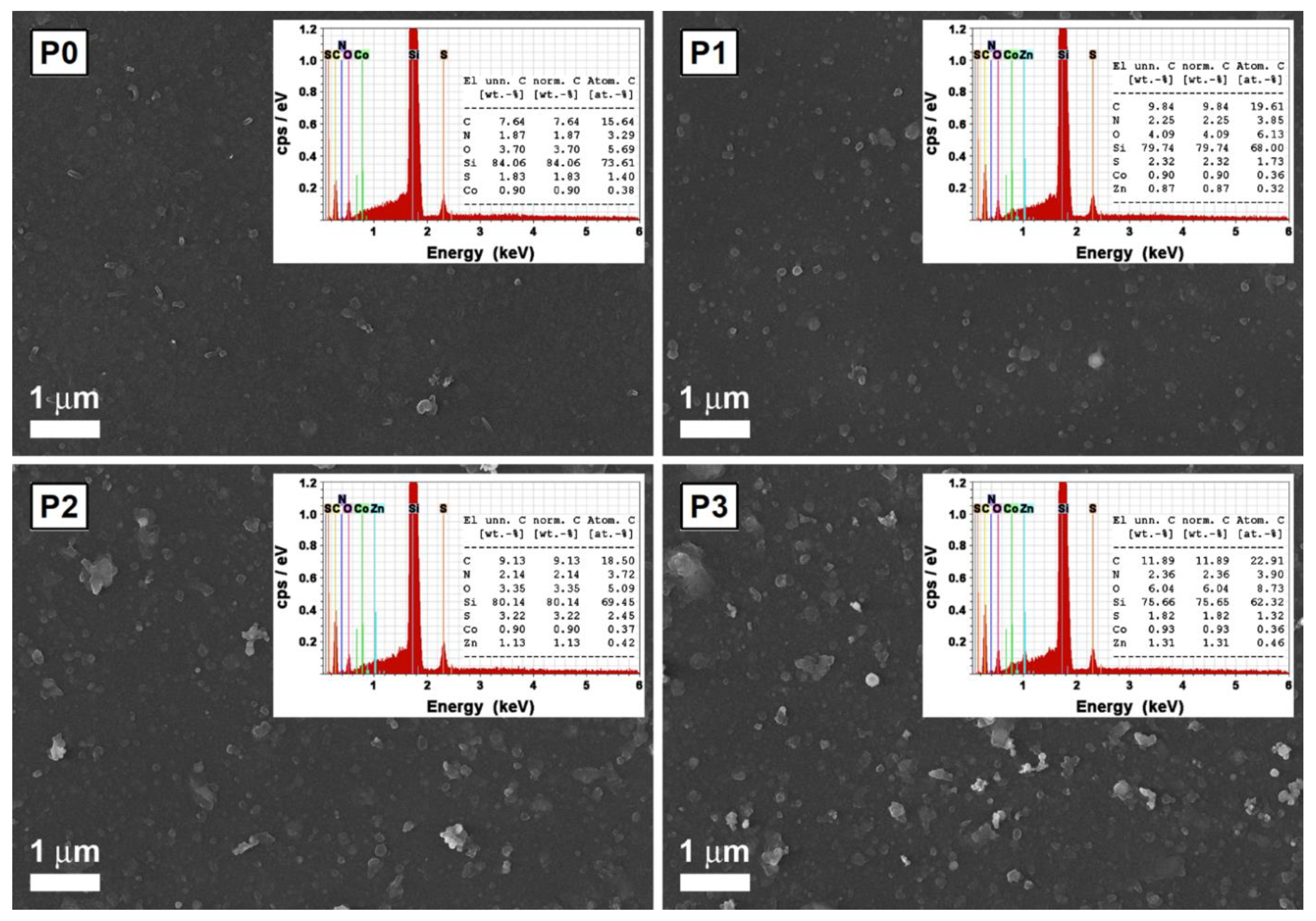
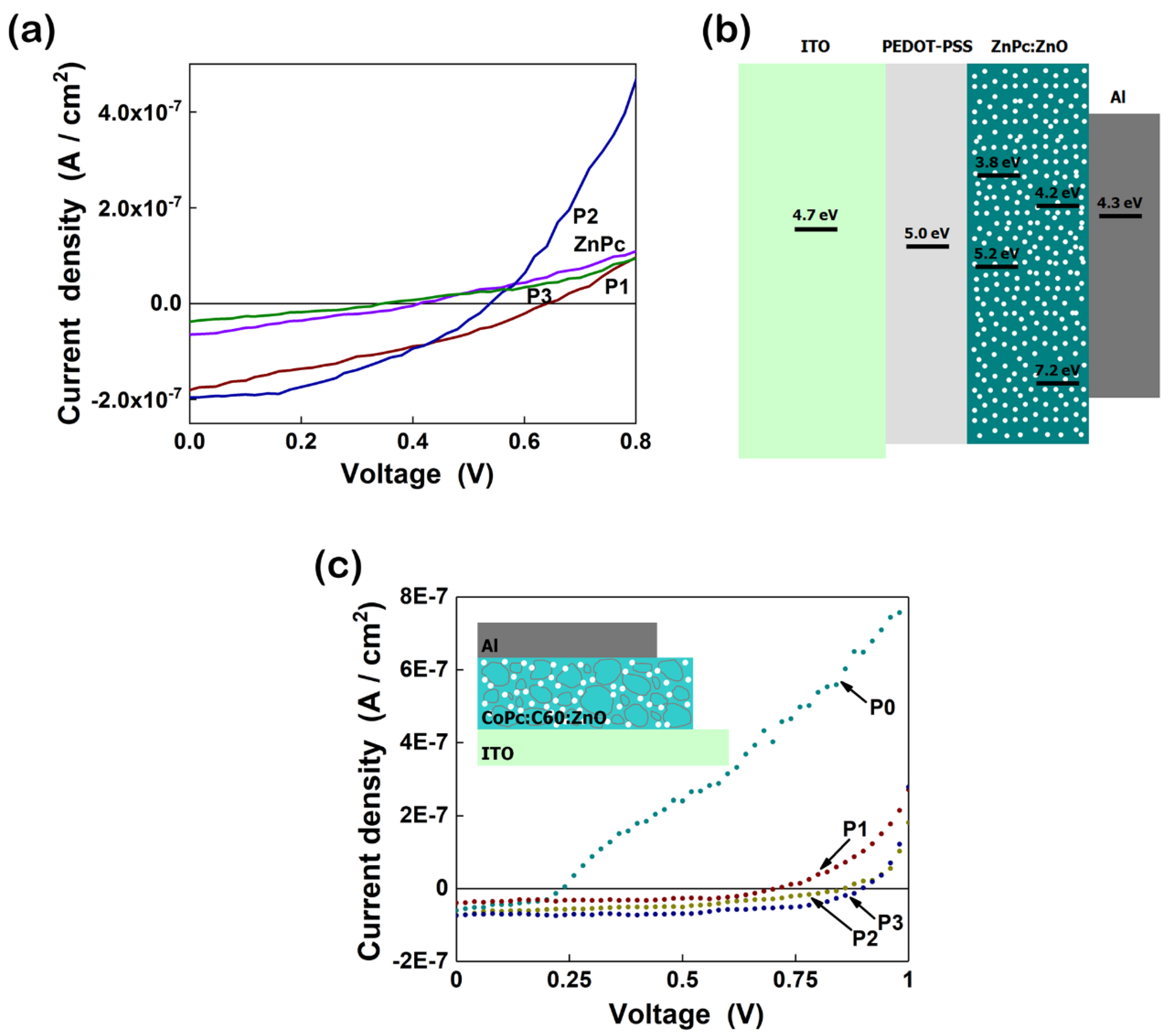
4.1.1. ZnO
4.1.2. CuO
4.1.3. TiO2
4.1.4. Cr2O3
4.2. Chalcogenides Based Nanocomposites
4.2.1. CdSe
4.2.2. CdTe
4.2.3. CdS
4.2.4. PbSe
4.2.5. PbS
4.2.6. SnS2
4.2.7. FeS2
4.2.8. ZnSe
4.2.9. ZnS
4.2.10. Cu2S
4.2.11. CuS
4.2.12. CuInS2
4.2.13. PbSxSe1-x
4.3. Silicon-Based Nanocomposites
4.4. Power Conversion Efficiency of the Analyzed PV Cells Fabricated with Hybrid Composite Layers–Summary
5. Hybrid Nanocomposite Films Deposited by MAPLE for PV Cells
6. Conclusions and Future Perspectives
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Grätzel, M. Powering the planet. Nature 2000, 403, 363. [Google Scholar] [CrossRef]
- Solar Power, Origami-Style. Available online: https://www.nasa.gov/jpl/news/origami-style-solar-power-20140814 (accessed on 11 January 2021).
- Ohl, R.S. Light-Sensitive Electric Device Including Silicon. U.S. Patent 2443542, 15 June 1948. [Google Scholar]
- Chapin, D.M.; Fuller, C.S.; Pearson, G.L. A new Silicon p-n junction photocell for converting solar radiation into electrical power. J. Appl. Phys. 1954, 25, 676–677. [Google Scholar] [CrossRef]
- Chapin, D.M.; Fuller, C.; Pearson, G. Solar Energy Converting Apparatus. U.S. Patent 2780765, 5 May 1957. [Google Scholar]
- Most Efficient Solar Panels: Solar Panel Cell Efficiency Explained. Available online: https://news.energysage.com/what-are-the-most-efficient-solar-panels-on-the-market/ (accessed on 11 January 2021).
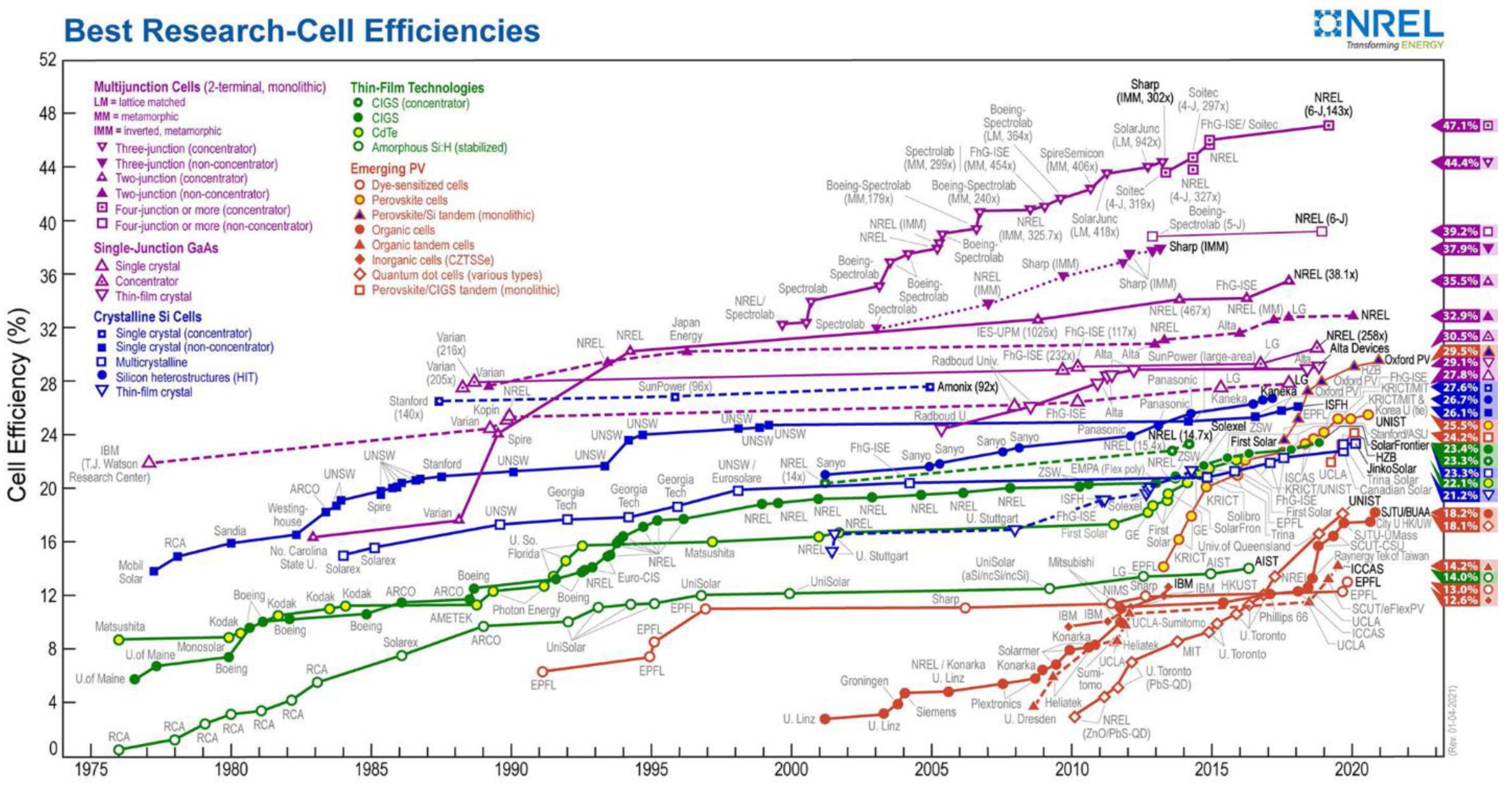
- Research Cell Efficiency Records. Available online: https://www.nrel.gov/pv/assets/pdfs/best-research-cell-efficiencies.20200104.pdf (accessed on 11 January 2021).
- Luceño-Sánchez, J.A.; Díez-Pascual, A.M.; Capilla, R.P. Materials for photovoltaics: State of art and recent developments. Int. J. Mol. Sci. 2019, 20, 976. [Google Scholar] [CrossRef]
- Simya, O.K.; Nair, P.R.; Ashok, A.M. Engineered Nanomaterials for Energy Applications. In Handbook of Nanomaterials for Industrial Applications; Hussain, C.M., Ed.; Elsevier: Amsterdam, The Netherlands, 2018; pp. 751–767. ISBN 978-0-12-813351-4. [Google Scholar] [CrossRef]
- Zhang, T.; Wang, M.; Yang, H. A review of the energy performance and life-cycle assessment of building-integrated photovoltaic (BIPV) systems. Energies 2018, 11, 3157. [Google Scholar] [CrossRef]
- Muhammad, J.Y.; Waziri, A.B.; Shitu, A.M.; Ahmad, U.M.; Muhammad, M.H.; Alhaji, Y.; Olaniyi, A.T.; Bala, A.A. Recent progressive status of materials for solar photovoltaic cell: A comprehensive review. Sci. J. Energy Eng. 2019, 7, 77–89. [Google Scholar] [CrossRef]
- Kuhlmann, A.M. The second most abundant element in the earth’s crust. J. Met. 1963, 15, 502–505. [Google Scholar] [CrossRef]
- Green, M.A.; Dunlop, E.D.; Hohl-Ebinger, J.; Yoshita, M.; Kopidakis, N.; Hao, X. Solar cell efficiency tables (version 56). Prog. Photovolt. Res. Appl. 2020, 28, 629–638. [Google Scholar] [CrossRef]
- Shockley, W.; Queisser, H.J. Detailed balance limit of efficiency of p-n junction solar cells. J. Appl. Phys. 1961, 32, 510–519. [Google Scholar] [CrossRef]
- Irvine, S. Solar Cells and Photovoltaics. In Handbook of Electronic and Photonic Materials; Kasap, S., Capper, P., Eds.; Springer: Cham, Switzerland, 2017; pp. 1097–1109. [Google Scholar] [CrossRef]
- Deibel, C.; Dyakonov, V. Polymer–fullerene bulk heterojunction solar cells. Rep. Prog. Phys. 2010, 73, 096401. [Google Scholar] [CrossRef]
- Lim, I.; Bui, H.T.; Shrestha, N.K.; Lee, J.K.; Han, S.-H. Interfacial engineering for enhanced light absorption and charge transfer of a solution-processed bulk heterojunction based on heptazole as a small molecule type of donor. ACS Appl. Mater. Interfaces 2016, 8, 8637–8643. [Google Scholar] [CrossRef]
- Gao, Y.; Jin, F.; Li, W.; Su, Z.; Chu, B.; Wang, J.; Zhao, H.; Wu, H.; Liu, C.; Hou, F.; et al. Highly efficient organic tandem solar cell with a SubPc interlayer based on TAPC:C70 bulk heterojunction. Sci. Rep. 2016, 6, 23916. [Google Scholar] [CrossRef]
- Chuang, H.-Y.; Lin, S.-W.; Hsu, S.L.-C. Efficiency enhancement of P3HT:PC61BM bulk heterojunction solar cells by doping with a small molecule dye. Thin Solid Films 2016, 603, 317–322. [Google Scholar] [CrossRef]
- Emmott, C.J.M.; Röhr, J.A.; Campoy-Quiles, M.; Kirchartz, T.; Urbina, A.; Ekins-Daukes, N.J.; Nelson, J. Organic Photovoltaic greenhouses: A unique application for semi-transparent PV? Energy Environ. Sci. 2015, 8, 1317–1328. [Google Scholar] [CrossRef]
- Iwase, M.; Suzuki, A.; Akiyama, T.; Oku, T. Fabrication and characterization of phthalocyanine-based organic solar cells. Mater. Sci. Appl. 2014, 5, 278–284. [Google Scholar] [CrossRef][Green Version]
- Chen, Q.; De Marco, N.; Yang, Y.; Song, T.-B.; Chen, C.-C.; Zhao, H.; Hong, Z.; Zhou, H.; Yang, Y. Under the spotlight: The organic–inorganic hybrid halide perovskite for optoelectronic applications. Nanotoday 2015, 10, 355–396. [Google Scholar] [CrossRef]
- Kearns, D.; Calvin, M. Photovoltaic effect and photoconductivity in laminated organic systems. J. Chem. Phys. 1958, 29, 950–951. [Google Scholar] [CrossRef]
- Tang, C.W. Two-layer organic photovoltaic cell. Appl. Phys. Lett. 1986, 48, 183–185. [Google Scholar] [CrossRef]
- Gevorgyan, S.A.; Heckler, I.M.; Bundgaard, E.; Corazza, M.; Hösel, M.; Søndergaard, R.R.; dos Reis Benatto, G.A.; Jørgensen, M.; Krebs, F.C. Improving, characterizing and predicting the lifetime of organic photovoltaics. J. Phys. D Appl. Phys. 2017, 50, 103001. [Google Scholar] [CrossRef]
- Fan, B.; Zhong, W.; Ying, L.; Zhang, D.; Li, M.; Lin, Y.; Xia, R.; Liu, F.; Yip, H.-L.; Li, N.; et al. Surpassing the 10% efficiency milestone for 1-cm2 all-polymer solar cells. Nat. Commun. 2019, 10, 4100. [Google Scholar] [CrossRef] [PubMed]
- Meng, L.; Zhang, Y.; Wan, X.; Li, C.; Zhang, X.; Wang, Y.; Ke, X.; Xiao, Z.; Ding, L.; Xia, R.; et al. Organic and solution-processed tandem solar cells with 17.3% efficiency. Science 2018, 361, 1094–1098. [Google Scholar] [CrossRef]
- Liu, Q.; Jiang, Y.; Jin, K.; Qin, J.; Xu, J.; Li, W.; Xiong, J.; Liu, J.; Xiao, Z.; Sun, K.; et al. 18% Efficiency organic solar cells. Sci. Bull. 2020, 65, 272–275. [Google Scholar] [CrossRef]
- Yu, G.; Gao, J.; Hummelen, J.C.; Wudi, F.; Heeger, A.J. Polymer photovoltaic cells: Enhanced efficiencies via a network of internal donor-acceptor heterojunctions. Science 1995, 270, 1789–1791. [Google Scholar] [CrossRef]
- Laquai, F.; Andrienko, D.; Mauer, R.; Blom, P.W.M. Charge carrier transport and photogeneration in P3HT:PCBM photovoltaic blends. Macromol. Rapid Commun. 2015, 36, 1001–1025. [Google Scholar] [CrossRef] [PubMed]
- Glowacki, I.; Jung, J.; Wiosna–Salyga, G.; Chapran, M.; Luczak, A.; Dupont, B.G.R.; Luszczynska, B.; Ulanski, J. Role of charge–carrier trapping in organic optoelectronic devices. Disp. Imaging 2017, 2, 279–319. [Google Scholar] [CrossRef]
- Cheng, L.; Zhang, C.; Liu, Y. Why two-dimensional semiconductors generally have low electron mobility. Phys. Rev. Lett. 2020, 125, 177701. [Google Scholar] [CrossRef] [PubMed]
- Haneef, H.F.; Zeidell, A.M.; Jurchescu, O.D. Charge carrier traps in organic semiconductors: A review on the underlying physics and impact on electronic devices. J. Mater. Chem. C 2020, 8, 759–787. [Google Scholar] [CrossRef]
- Kumar, G.N.M.; Chidambaram, S.; Park Jinsub, J.K.; Jayavel, R. Hybrid Nanostructures for Photovoltaics. In Nanostructure, Nanosystems and Nanostructured Materials, 1st ed.; Sivakumar, P.M., Kodolov, V.I., Zaikov, G.E., Haghi, A.K., Eds.; Apple Academic Press: Pal Bay, FL, USA, 2013; pp. 462–486. [Google Scholar] [CrossRef]
- Liu, R. Hybrid organic/inorganic nanocomposites for photovoltaic cells. Materials 2014, 7, 2747–2771. [Google Scholar] [CrossRef] [PubMed]
- Xie, S.; Li, X.; Jiang, Y.; Yang, R.; Fu, M.; Li, W.; Pan, Y.; Qin, D.; Xu, W.; Hou, L. Recent progress in hybrid solar cells based on solution-processed organic and semiconductor nanocrystal: Perspectives on device design. Appl. Sci. 2020, 10, 4285. [Google Scholar] [CrossRef]
- Available online: https://apps.webofknowledge.com/ (accessed on 11 January 2021).
- Ahmad, M.; Zhu, J. ZnO based advanced functional nanostructures: Synthesis, properties and applications. J. Mater. Chem. 2011, 21, 599–614. [Google Scholar] [CrossRef]
- Misra, S.K.; Nuseibeh, S.; Dybowska, A.; Berhanu, D.; Tetley, T.D.; Valsami-Jones, E. Comparative study using spheres, rods and spindle-shaped nanoplatelets on dispersion stability, dissolution and toxicity of CuO nanomaterials. Nanotoxicology 2013, 8, 422–432. [Google Scholar] [CrossRef]
- Zhang, Q.; Zhang, K.; Xu, D.; Yang, G.; Huang, H.; Nie, F.; Liu, C.; Yang, S. CuO nanostructures: Synthesis, characterization, growth mechanisms, fundamental properties, and applications. Prog. Mater. Sci. 2014, 60, 208–337. [Google Scholar] [CrossRef]
- Ali, I.; Suhail, M.; Alothman, Z.A.; Alwarthan, A. Recent advances in syntheses, properties and applications of TiO2 nanostructures. RSC Adv. 2018, 8, 30125–30147. [Google Scholar] [CrossRef]
- Hussain, A.A.; Pal, A.R.; Kar, R.; Bailung, H.; Chutia, J.; Patil, D.S. Comparative study of nanocomposites prepared by pulsed and dc sputtering combined with plasma polymerization suitable for photovoltaic device applications. Mater. Chem. Phys. 2014, 148, 540–547. [Google Scholar] [CrossRef]
- Florica, C.; Preda, N.; Enculescu, M.; Zgura, I.; Socol, M.; Enculescu, I. Superhydrophobic ZnO networks with high water adhesion. Nanoscale Res. Lett. 2014, 9, 385. [Google Scholar] [CrossRef]
- Socol, M.; Preda, N.; Costas, A.; Breazu, C.; Stanculescu, A.; Rasoga, O.; Popescu-Pelin, G.; Mihailescu, A.; Socol, G. Hybrid organic-inorganic thin films based on zinc phthalocyanine and zinc oxide deposited by MAPLE. Appl. Surf. Sci. 2020, 503, 144317. [Google Scholar] [CrossRef]
- Preda, N.; Enculescu, M.; Florica, C.; Costas, A.; Evanghelidis, A.; Matei, E.; Enculescu, I. Morphology-controlled synthesis of ZnO structures by a simple wet chemical method. Dig. J. Nanomater. Biostruct. 2013, 8, 159–600. [Google Scholar]
- Preda, N.; Enculescu, M.; Zgura, I.; Socol, M.; Matei, E.; Vasilache, V.; Enculescu, I. Superhydrophobic properties of cotton fabrics functionalized with ZnO by electroless deposition. Mater. Chem. Phys. 2013, 138, 253–261. [Google Scholar] [CrossRef]
- Preda, N.; Costas, A.; Enculescu, M.; Enculescu, I. Biomorphic 3D fibrous networks based on ZnO, CuO and ZnO–CuO composite nanostructures prepared from eggshell membranes. Mater. Chem. Phys. 2020, 240, 122205. [Google Scholar] [CrossRef]
- Florica, C.; Preda, N.; Costas, A.; Zgura, I.; Enculescu, I. ZnO nanowires grown directly on zinc foils by thermal oxidation in air: Wetting and water adhesion properties. Mater. Lett. 2016, 170, 156–159. [Google Scholar] [CrossRef]
- Florica, C.; Costas, A.; Boni, A.G.; Negrea, R.; Ion, L.; Preda, N.; Pintilie, L.; Enculescu, I. Electrical properties of single CuO nanowires for device fabrication: Diodes and field effect transistors. Appl. Phys. Lett. 2015, 106, 223501. [Google Scholar] [CrossRef]
- Freitas, J.N.; Gonçalves, A.S.; Nogueira, A.F. A comprehensive review of the application of chalcogenide nanoparticles in polymer solar cells. Nanoscale 2014, 6, 6371–6397. [Google Scholar] [CrossRef]
- Feng, L.; Niu, M.; Wen, Z.; Hao, X. Recent advances of plasmonic organic solar cells: Photophysical investigations. Polymers 2018, 10, 123. [Google Scholar] [CrossRef]
- Kozanoglu, D.; Apaydin, D.H.; Cirpan, A.; Esenturk, E.N. Power conversion efficiency enhancement of organic solar cells by addition of gold nanostars, nanorods, and nanospheres. Org. Electron. 2013, 14, 1720–1727. [Google Scholar] [CrossRef]
- Ahn, S.; Rourke, D.; Park, W. Plasmonic nanostructures for organic photovoltaic devices. J. Opt. 2016, 18, 033001. [Google Scholar] [CrossRef]
- Uddin, A.; Yang, X. Surface plasmonic effects on organic solar cells. J. Nanosci. Nanotechnol. 2014, 14, 1099–1119. [Google Scholar] [CrossRef] [PubMed]
- Qiao, L.; Wang, D.; Zuo, L.; Ye, Y.; Qian, J.; Chen, H.; He, S. Localized surface plasmon resonance enhanced organic solar cell with gold nanospheres. Appl. Energy 2011, 88, 848–852. [Google Scholar] [CrossRef]
- Teichler, A.; Perelaer, J.; Schubert, U.S. Inkjet printing of organic electronics—Comparison of deposition techniques and state-of-the-art developments. J. Mater. Chem. C 2013, 1, 1910. [Google Scholar] [CrossRef]
- Cui, Y.; Yao, H.; Hong, L.; Zhang, T.; Tang, Y.; Lin, B.; Xian, K.; Gao, B.; An, C.; Bi, P.; et al. Organic photovoltaic cell with 17% efficiency and superior processability. Natl. Sci. Rev. 2020, 7, 1239–1246. [Google Scholar] [CrossRef]
- Sahu, N.; Parija, B.; Panigrahi, S. Fundamental understanding and modelling of spin coating process: A review. Indian J. Phys. 2009, 83, 493–502. [Google Scholar] [CrossRef]
- McGill, R.A.; Chrisey, D.B. Method of Producing a film coating by matrix assisted pulsed laser deposition. U.S. Patent 6025036, 15 February 2000. [Google Scholar]
- Socol, M.; Preda, N.; Rasoga, O.; Breazu, C.; Stavarache, I.; Stanculescu, F.; Socol, G.; Gherendi, F.; Grumezescu, V.; Stefan, N.; et al. Flexible heterostructures based on metal phthalocyanines thin films obtained by MAPLE. Appl. Surf. Sci. 2016, 374, 403–410. [Google Scholar] [CrossRef]
- Caricato, A.P.; Cesaria, M.; Gigli, G.; Loiudice, A.; Luches, A.; Martino, M.; Resta, V.; Rizzo, A.; Taurino, A. Poly-(3-hexylthiophene)/[6,6]-phenyl-C61-butyric-acid-methyl-ester bilayer deposition by matrix-assisted pulsed laser evaporation for organic photovoltaic applications. Appl. Phys. Lett. 2012, 100, 073306. [Google Scholar] [CrossRef]
- Ge, W.; Li, N.K.; McCormick, R.D.; Lichtenberg, E.; Yingling, Y.G.; Stiff-Roberts, A.D. Emulsion-based RIR-MAPLE deposition of conjugated polymers: Primary solvent effect and its implications on organic solar cell performance. ACS Appl. Mater. Interfaces 2016, 8, 19494–19506. [Google Scholar] [CrossRef]
- Stanculescu, F.; Rasoga, O.; Catargiu, A.M.; Vacareanu, L.; Socol, M.; Breazu, C.; Preda, N.; Socol, G.; Stanculescu, A. MAPLE prepared heterostructures with arylene based polymer active layer for photovoltaic applications. Appl. Surf. Sci. 2015, 336, 240–248. [Google Scholar] [CrossRef]
- Socol, M.; Preda, N.; Costas, A.; Borca, B.; Popescu-Pelin, G.; Mihailescu, A.; Socol, G.; Stanculescu, A. Thin films based on cobalt phthalocyanine:C60 fullerene:ZnO hybrid nanocomposite obtained by laser evaporation. Nanomaterials 2020, 10, 468. [Google Scholar] [CrossRef]
- Ge, W.; Atewologun, A.; Stiff-Roberts, A.D. Hybrid nanocomposite thin films deposited by emulsion-based resonant infrared matrix-assisted pulsed laser evaporation for photovoltaic applications. Org. Electron. 2015, 22, 98–107. [Google Scholar] [CrossRef]
- Kim, H.; Piqué, A.; Horwitz, J.S.; Murat, H.; Kafafi, Z.H.; Gilmore, C.M.; Chrisey, D. Effect of aluminum doping on zinc oxide thin films grown by pulsed laser deposition for organic light-emitting devices. Thin Solid Films 2000, 377–378, 798–802. [Google Scholar] [CrossRef]
- Caricato, A.P. MAPLE and MALDI: Theory and Experiments. In Lasers in Materials Science, 1st ed.; Castillejo, M., Ossi, P., Zhigilei, L., Eds.; Springer International Publishing: Cham, Switzerland, 2014; pp. 295–323. [Google Scholar] [CrossRef]
- Axente, E.; Sima, L.E.; Sima, F. Biomimetic coatings obtained by combinatorial laser technologies. Coatings 2020, 10, 463. [Google Scholar] [CrossRef]
- Stiff-Roberts, A.D.; Ge, W. Organic/hybrid thin films deposited by matrix-assisted pulsed laser evaporation (MAPLE). Appl. Phys. Rev. 2017, 4, 041303. [Google Scholar] [CrossRef]
- Socol, M.; Preda, N.; Breazu, C.; Stanculescu, A.; Costas, A.; Stanculescu, F.; Girtan, M.; Gherendi, F.; Popescu-Pelin, G.; Socol, G. Flexible organic heterostructures obtained by MAPLE. Appl. Phys. A 2018, 124, 602. [Google Scholar] [CrossRef]
- Chen, C.; Hu, P.; Yang, J.; Liu, Z. Synthesis of PVDF/SBT composite thin films by spin coating technology and their ferroelectric properties. Mater. Sci. Poland 2016, 34, 650–654. [Google Scholar] [CrossRef][Green Version]
- Nguyen, A.N.; Solard, J.; Nong, H.T.T.; Ben Osman, C.; Gomez, A.; Bockelée, V.; Tencé-Girault, S.; Schoenstein, F.; Simón-Sorbed, M.; Carrillo, A.S.; et al. Spin Coating and Micro-Patterning Optimization of Composite Thin Films Based on PVDF. Materials 2020, 13, 1342. [Google Scholar] [CrossRef] [PubMed]
- Kwon, Y.H.; Huie, M.M.; Choi, D.; Chang, M.; Marschilok, A.C.; Takeuchi, K.J.; Takeuchi, A.S.; Reichmanis, E. Toward Uniformly Dispersed Battery Electrode Composite Materials: Characteristics and Performance. ACS Appl. Mater. Interfaces 2016, 8, 3452–3463. [Google Scholar] [CrossRef]
- Szydowska, B.M.; Graf, A.; Kelly, A.; Blau, W.J.; Gather, M.C.; Zaumseil, J.; Backes, C. Preparation of WS2-PMMA composite films for optical applications. J. Mater. Chem. C 2020, 8, 10805–10815. [Google Scholar] [CrossRef]
- Afifi, M.; Ahmed, M.K.; Fathi, A.M.; Uskoković, V. Physical, electrochemical and biological evaluations of spin-coated ε-polycaprolactone thin films containing alumina/graphene/carbonated hydroxyapatite/titania for tissue engineering applications. Int. J. Pharm. 2020, 585, 119502. [Google Scholar] [CrossRef]
- Holban, A.M.; Grumezescu, V.; Grumezescu, A.M.; Vasile, B.Ş.; Truşcă, R.; Cristescu, R.; Socol, G.; Iordache, F. Antimicrobial nanospheres thin coatings prepared by advanced pulsed laser technique. Beilstein J Nanotechnol. 2014, 5, 872–880. [Google Scholar] [CrossRef]
- Oprea, A.; Pandel, L.; Dumitrescu, A.; Andronescu, E.; Grumezescu, V.; Chifiriuc, M.; Mogoanta, L.; Balseanu, T.A.; Mogosanu, G.D.; Socol, G.; et al. Bioactive ZnO Coatings Deposited by MAPLE—An Appropriate Strategy to Produce Efficient Anti-Biofilm Surfaces. Molecules 2016, 21, 220. [Google Scholar] [CrossRef]
- Huang, G.; Chen, Y.; Zhang, J. Nanocomposited coatings produced by laser-assisted process to prevent silicone hydrogels from protein fouling and bacterial contamination. Appl. Surf. Sci. 2016, 360, 383–388. [Google Scholar] [CrossRef]
- Yang, S.; Tse, W.H.; Zhang, J. Deposition of antibody modified upconversion nanoparticles on glass by a laser-assisted method to improve the performance of cell culture. Nanoscale Res. Lett. 2019, 14, 101. [Google Scholar] [CrossRef]
- Icriverzi, M.; Rusen, L.; Brajnicov, S.; Bonciu, A.; Dinescu, M.; Cimpean, A.; Evans, R.W.; Dinca, V.; Roseanu, A. Macrophage in vitro response on hybrid coatings obtained by matrix assisted pulsed laser evaporation. Coatings 2019, 9, 236. [Google Scholar] [CrossRef]
- Ajayan, J.; Nirmal, D.; Mohankumar, P.; Saravanan, M.; Jagadesh, M.; Arivazhagan, L. A review of photovoltaic performance of organic/inorganic solar cells for future renewable and sustainable energy technologies. Superlattices Microstruct. 2020, 143, 106549. [Google Scholar] [CrossRef]
- Abdullah, M.F.; Hashim, A.M. Review and assessment of photovoltaic performance of graphene/Si heterojunction solar cells. J. Mater. Sci. 2018, 54, 911–948. [Google Scholar] [CrossRef]
- Antohe, S.; Iftimie, S.; Hrostea, L.; Antohe, V.A.; Girtan, M. A critical review of photovoltaic cells based on organic monomeric and polymeric thin film heterojunctions. Thin Solid Films 2017, 642, 219–231. [Google Scholar] [CrossRef]
- Yu, J.; Zheng, Y.; Huang, J. Towards high performance organic photovoltaic cells: A review of recent development in organic photovoltaics. Polymers 2014, 6, 2473–2509. [Google Scholar] [CrossRef]
- Sun, Z.; He, Y.; Xiong, B.; Chen, S.; Li, M.; Zhou, Y.; Zheng, Y.; Sun, K.; Yang, C. Performance-enhancing approaches for PEDOT:PSS-Si hybrid solar cells. Angew. Chem. Int. Ed. 2021, 60, 5036–5055. [Google Scholar] [CrossRef] [PubMed]
- Chen, Z.; Li, W.; Li, R.; Zhang, Y.; Xu, G.; Cheng, H. Fabrication of highly transparent and conductive indium–tin oxide thin films with a high figure of merit via solution processing. Langmuir 2013, 29, 13836–13842. [Google Scholar] [CrossRef]
- Dang, M.T.; Hirsch, L.; Wantz, G. P3HT:PCBM, Best seller in polymer photovoltaic research. Adv. Mater. 2011, 23, 3597–3602. [Google Scholar] [CrossRef]
- Way, A.; Luke, J.; Evans, A.D.; Li, Z.; Kim, J.-S.; Durrant, J.R.; Lee, H.K.H.; Tsoi, W.C. Fluorine doped tin oxide as an alternative of indium tin oxide for bottom electrode of semi-transparent organic photovoltaic devices. AIP Adv. 2019, 9, 085220. [Google Scholar] [CrossRef]
- Socol, M.; Preda, N.; Stanculescu, A.; Breazu, C.; Florica, C.; Stanculescu, F.; Iftimie, S.; Girtan, M.; Popescu-Pelin, G.; Socol, G. Organic heterostructures deposited by MAPLE on AZO substrate. Appl. Surf. Sci. 2017, 417, 196–203. [Google Scholar] [CrossRef]
- Liang, Z.; Zhang, Q.; Jiang, L.; Cao, G. ZnO cathode buffer layers for inverted polymer solar cells. Energy Environ. Sci. 2015, 8, 3442–3476. [Google Scholar] [CrossRef]
- Yin, Z.; Zheng, Q.; Chen, S.-C.; Li, J.; Cai, D.; Ma, Y.; Wei, J. Solution-derived poly(ethylene glycol)-TiOx nanocomposite film as a universal cathode buffer layer for enhancing efficiency and stability of polymer solar cells. Nano Res. 2015, 8, 456–468. [Google Scholar] [CrossRef]
- Sun, K.; Zhang, S.; Li, P.; Xia, Y.; Zhang, X.; Du, D.; Isikgor, F.H.; Ouyang, J. Review on application of PEDOTs and PEDOT:PSS in energy conversion and storage devices. J. Mater. Sci. Mater. Electron. 2015, 26, 4438–4462. [Google Scholar] [CrossRef]
- Ameen, Y.M.; Pradhan, S.; Remyth Suresh, M.; Reddy, V.S. MoO3 anode buffer layer for efficient and stable small molecular organic solar cells. Opt. Mater. 2015, 39, 134–139. [Google Scholar] [CrossRef]
- Ikram, M.; Imran, M.; Nunzi, J.M.; Bobbara, S.R.; Ali, S.; Islah-u-din. Efficient and low cost inverted hybrid bulk heterojunction solar cells. J. Renew. Sustain. Energy 2015, 7, 043148. [Google Scholar] [CrossRef]
- Qiu, M.; Zhu, D.; Bao, X.; Wang, J.; Wang, X.; Yang, R. WO3 with surface oxygen vacancies as an anode buffer layer for high performance polymer solar cells. J. Mater. Chem. A 2016, 4, 894–900. [Google Scholar] [CrossRef]
- Li, M.; Li, J.; Yu, L.; Zhang, Y.; Dai, Y.; Chen, R.; Huang, W. Trap-filling of ZnO buffer layer for improved efficiencies of organic solar cells. Front. Chem. 2020, 8, 399. [Google Scholar] [CrossRef] [PubMed]
- Yan, Q.; Gu, Z.; Li, Q.; Fu, W.; Chen, X.; Liu, W.; Pan, H.; Wang, M.; Chen, H. Water soluble amino grafted silicon nanoparticles and their use in polymer solar cells. Chinese J. Polym. Sci. 2014, 32, 395–401. [Google Scholar] [CrossRef]
- Hamed, M.S.G.; Mola, G.T. Copper sulphide as a mechanism to improve energy harvesting in thin film solar cells. J. Alloys Compd. 2019, 802, 252–258. [Google Scholar] [CrossRef]
- Lee, K.-S.; Kim, I.; Gullapalli, S.; Wong, M.S.; Jabbour, G.E. Enhanced performance of hybrid solar cells using longer arms of quantum cadmium selenide tetrapods. Appl. Phys. Lett. 2011, 99, 223515. [Google Scholar] [CrossRef]
- Oosterhout, S.D.; Koster, L.J.A.; van Bavel, S.S.; Loos, J.; Stenzel, O.; Thiedmann, R.; Schmidt, V.; Campo, B.; Cleij, T.J.; Lutzen, L.; et al. Controlling the morphology and efficiency of hybrid ZnO:polythiophene solar cells via side chain functionalization. Adv. Energy Mater. 2011, 1, 90–96. [Google Scholar] [CrossRef]
- Kim, S.; Jeon, K.; Lee, J.C.; Swihart, M.T.; Yang, M. Enhanced performance of a polymer solar cell upon addition of free-standing, freshly etched, photoluminescent silicon nanocrystals. Appl. Phys. Express 2012, 5, 022302. [Google Scholar] [CrossRef]
- Venkataprasad Bhat, S.; Govindaraj, A.; Rao, C.N.R. Hybrid solar cell based on P3HT–ZnO nanoparticle blend in the inverted device configuration. Sol. Energy Mater. Sol. Cells 2011, 95, 2318–2321. [Google Scholar] [CrossRef]
- Inpor, K.; Meeyoo, V.; Thanachayanont, C. Enhancement of photovoltaic performance using hybrid CdS nanorods and MEH-PPV active layer in ITO/TiO2/MEH-PPV:CdS/Au devices. Curr. Appl. Phys. 2011, 11, S171–S174. [Google Scholar] [CrossRef]
- Hemaprabha, E.; Pandey, U.K.; Chattopadhyay, K.; Ramamurthy, P.C. Doped silicon nanoparticles for enhanced charge transportation in organic-inorganic hybrid solar cells. Sol. Energy 2018, 173, 744–751. [Google Scholar] [CrossRef]
- Mazzio, K.A.; Luscombe, C.K. The future of organic photovoltaics. Chem. Soc. Rev. 2015, 44, 78–90. [Google Scholar] [CrossRef] [PubMed]
- Raj, M.R.; Anandan, S. Donor conjugated polymers-based on alkyl chain substituted oligobenzo[c]thiophene derivatives with well-balanced energy levels for bulk heterojunction solar cells. RSC Adv. 2013, 3, 14595–14608. [Google Scholar] [CrossRef]
- Wakim, S.; Beaupré, S.; Blouin, N.; Aich, B.-R.; Rodman, S.; Gaudiana, R.; Tao, Y.; Leclerc, M. Highly efficient organic solar cells based on a poly(2,7-carbazole) derivative. J. Mater. Chem. 2009, 19, 5351–5358. [Google Scholar] [CrossRef]
- Li, G.; Chang, W.-H.; Yang, Y. Low-bandgap conjugated polymers enabling solution-processable tandem solar cells. Nat. Rev. Mater. 2017, 2, 17043. [Google Scholar] [CrossRef]
- Murad, A.R.; Iraqi, A.; Aziz, S.B.; Abdullah, S.N.; Brza, M.A. Conducting Polymers for Optoelectronic Devices and Organic Solar Cells: A Review. Polymers 2020, 12, 2627. [Google Scholar] [CrossRef] [PubMed]
- Kroon, R.; Lenes, M.; Hummelen, J.C.; Blom, P.W.M.; de Boer, B. Small bandgap polymers for organic solar cells (polymer material development in the last 5 years). Polym. Rev. 2008, 48, 531–582. [Google Scholar] [CrossRef]
- Yang, X.; Loos, J.; Veenstra, S.C.; Verhees, W.J.H.; Wienk, M.M.; Kroon, J.M.; Michels, M.A.J.; Janssen, R.A.J. Nanoscale morphology of high-performance polymer solar cells. Nano Lett. 2005, 5, 579–583. [Google Scholar] [CrossRef]
- Poelking, C.; Daoulas, K.; Troisi, A.; Andrienko, D. Morphology and charge transport in P3HT: A theorist’s perspective. Adv. Polym. Sci. 2014, 265, 139–180. [Google Scholar] [CrossRef]
- Mühlbacher, D.; Scharber, M.; Morana, M.; Zhu, Z.; Waller, D.; Gaudiana, R.; Brabec, C. High photovoltaic performance of a low-bandgap polymer. Adv. Mater. 2006, 18, 2884–2889. [Google Scholar] [CrossRef]
- Gusain, A.; Faria, R.M.; Miranda, P.B. Polymer solar cells—Interfacial processes related to performance issues. Front. Chem. 2019, 7, 61. [Google Scholar] [CrossRef] [PubMed]
- Benten, H.; Mori, D.; Ohkita, H.; Ito, S. Recent research progress of polymer donor/polymer acceptor blend solar cells. J. Mater. Chem. A 2016, 4, 5340–5365. [Google Scholar] [CrossRef]
- Kularatne, R.S.; Magurudeniya, H.D.; Sista, P.; Biewer, M.C.; Stefan, M.C. Donor-acceptor semiconducting polymers for organic solar cells. J. Polym. Sci. A Polym. Chem. 2012, 51, 743–768. [Google Scholar] [CrossRef]
- Facchetti, A. Polymer donor–polymer acceptor (all-polymer) solar cells. Mater. Today 2013, 16, 123–132. [Google Scholar] [CrossRef]
- Liang, Y.; Xu, Z.; Xia, J.; Tsai, S.-T.; Wu, Y.; Li, G.; Ray, C.; Yu, L. For the bright future-bulk heterojunction polymer solar cells with power conversion efficiency of 7.4%. Adv. Mater. 2010, 22, E135–E138. [Google Scholar] [CrossRef]
- Xiao, B.; Zhao, Y.; Tang, A.; Wang, H.; Yang, J.; Zhou, E. PTB7-Th based organic solar cell with a high VOC of 1.05 V by modulating the LUMO energy level of benzotriazole-containing non-fullerene acceptor. Sci. Bull. 2017, 62, 1275–1282. [Google Scholar] [CrossRef]
- He, Z.; Zhong, C.; Huang, X.; Wong, W.-Y.; Wu, H.; Chen, L.; Su, S.; Cao, Y. Simultaneous enhancement of open-circuit voltage, short-circuit current density, and fill factor in polymer solar cells. Adv. Mater. 2011, 23, 4636–4643. [Google Scholar] [CrossRef] [PubMed]
- Sharma, R.; Bhalerao, S.; Gupta, D. Effect of incorporation of CdS NPs on performance of PTB7: PCBM organic solar cells. Org. Electron. 2016, 33, 274–280. [Google Scholar] [CrossRef]
- Wan, Q.; Guo, X.; Wang, Z.; Li, W.; Guo, B.; Ma, W.; Zhang, M.; Li, Y. 10.8% efficiency polymer solar cells based on PTB7-Th and PC71BM via binary solvent additives treatment. Adv. Funct. Mater. 2016, 26, 6635–6640. [Google Scholar] [CrossRef]
- Sun, J.; Zhang, Z.; Yin, X.; Zhou, J.; Yang, L.; Geng, R.; Zhang, F.; Zhu, R.; Yu, J.; Tang, W. High performance non-fullerene polymer solar cells based on PTB7-Th as the electron donor with 10.42% efficiency. J. Mater. Chem. A 2018, 6, 2549–2554. [Google Scholar] [CrossRef]
- Pratyusha, T.; Sivakumar, G.; Yella, A.; Gupta, D. Novel ternary blend of PCDTBT, PCPDTBT and PC70BM for the fabrication of bulk heterojunction organic solar cells. Mater. Today Proc. 2017, 4, 5067–5073. [Google Scholar] [CrossRef]
- Lu, H.; Joy, J.; Gaspar, R.L.; Bradforth, S.E.; Brutchey, R.L. Iodide-passivated colloidal PbS nanocrystals leading to highly efficient polymer:nanocrystal hybrid solar cells. Chem. Mater. 2016, 28, 1897–1906. [Google Scholar] [CrossRef]
- Suryana, R.; Tyas, L.K.; Nurosyid, F. Variation of MEH-PPV layers with/without PEDOT:PSS layer in organic solar cells. IOP Conf. Ser. Mater. Sci. Eng. 2018, 333, 012022. [Google Scholar] [CrossRef]
- Giro, R.; Caldas, M.J.; Galvão, D.S. Band gap engineering for poly(p-phenylene) and poly(p-phenylene vinylene) copolymers using the tight-binding approach. Int. J. Quantum Chem. 2005, 103, 588–596. [Google Scholar] [CrossRef]
- Hardeli, H.; Sanjaya, H.; Resikarnila, R.; Nitami, H.R. Solar cell polymer based active ingredients PPV and PCBM. IOP Conf. Ser. Mater. Sci. Eng. 2018, 335, 012029. [Google Scholar] [CrossRef]
- Liang, Z.; Dzienis, K.L.; Xu, J.; Wang, Q. Covalent layer-by-layer assembly of conjugated polymers and CdSe nanoparticles: Multilayer structure and photovoltaic properties. Adv. Funct. Mater. 2006, 16, 542–548. [Google Scholar] [CrossRef]
- Yahya, N.Z.; Rusop, M. Investigation on the optical and surface morphology of conjugated polymer MEH-PPV:ZnO nanocomposite thin films. J. Nanomater. 2012, 793679. [Google Scholar] [CrossRef]
- Crone, B.K.; Campbell, I.H.; Davids, P.S.; Smith, D.L. Charge injection and transport in single-layer organic light-emitting diodes. Appl. Phys. Lett. 1998, 73, 3162–3164. [Google Scholar] [CrossRef]
- Sensfuss, S.; Blankenburg, L.; Schache, H.; Shokhovets, S.; Erb, T.; Konkin, A.; Herasimovich, A.; Scheinert, S.; Shahid, M.; Sell, S.; et al. Thienopyrazine-based low-bandgap polymers for flexible polymer solar cells. Eur. Phys. J. Appl. Phys. 2010, 51, 33204. [Google Scholar] [CrossRef]
- Liu, F.; Chen, Z.; Du, X.; Zeng, Q.; Ji, T.; Cheng, Z.; Jin, G.; Yang, B. High efficiency aqueous-processed MEH-PPV/CdTe hybrid solar cells with a PCE of 4.20%. J. Mater. Chem. A 2016, 4, 1105–1111. [Google Scholar] [CrossRef]
- Faure, M.; Grant, T.; Lessard, B. Silicon phthalocyanines as acceptor candidates in mixed solution/evaporation processed planar heterojunction organic photovoltaic devices. Coatings 2019, 9, 203. [Google Scholar] [CrossRef]
- Roy, D.; Das, N.M.; Shakti, N.; Gupta, P.S. Comparative study of optical, structural and electrical properties of zinc phthalocyanine Langmuir–Blodgett thin film on annealing. RSC Adv. 2014, 4, 42514–42522. [Google Scholar] [CrossRef]
- Grant, T.M.; Josey, D.S.; Sampson, K.L.; Mudigonda, T.; Bender, T.P.; Lessard, B.H. Boron subphthalocyanines and silicon phthalocyanines for use as active materials in organic photovoltaics. Chem. Rec. 2019, 19, 1093–1112. [Google Scholar] [CrossRef]
- Boileau, N.T.; Cranston, R.; Mirka, B.; Melville, O.A.; Lessard, B.H. Metal phthalocyanine organic thin-film transistors: Changes in electrical performance and stability in response to temperature and environment. RSC Adv. 2019, 9, 21478–21485. [Google Scholar] [CrossRef]
- Terao, Y.; Sasabe, H.; Adachi, C. Correlation of hole mobility, exciton diffusion length, and solar cell characteristics in phthalocyanine/fullerene organic solar cells. Appl. Phys. Lett. 2007, 90, 103515. [Google Scholar] [CrossRef]
- Ahn, H.; Liou, W.-H.; Chen, H.-M.P.; Hsu, C.-H. Anisotropic exciton relaxation in nanostructured metal (Zn and F16Zn)-phthalocyanine. Opt. Express 2015, 23, 3230–3325. [Google Scholar] [CrossRef] [PubMed]
- Ghani, F.; Kristen, J.; Riegler, H. Solubility properties of unsubstituted metal phthalocyanines in different types of solvents. J. Chem. Eng. Data 2012, 57, 439–449. [Google Scholar] [CrossRef]
- Sharma, G.D.; Kumar, R.; Sharma, S.K.; Roy, M.S. Charge generation and photovoltaic properties of hybrid solar cells based on ZnO and copper phthalocyanines (CuPc). Sol. Energy Mater. Sol. Cells 2006, 90, 933–943. [Google Scholar] [CrossRef]
- Islam, Z.U.; Tahir, M.; Syed, W.A.; Aziz, F.; Wahab, F.; Said, S.M.; Sarker, M.R.; Ali, S.H.M.; Sabri, M.F.M. Fabrication and photovoltaic properties of organic solar cell based on zinc phthalocyanine. Energies 2020, 13, 962. [Google Scholar] [CrossRef]
- Maruhashi, H.; Oku, T.; Suzuki, A.; Akiyama, T.; Yamasaki, Y. Fabrication and characterization of PCBM: P3HT-based thin-film organic solar cells with zinc phthalocyanine and 1,8-diiodooctane. Chem. Mater. Eng. 2017, 5, 1–7. [Google Scholar] [CrossRef]
- Mingqing, W.; Xiaogong, W. P3HT/TiO2 Bulk heterojunction solar cell sensitized by copper phthalocyanine. In Proceedings of the ISES World Congress 2007 (Vol. I–Vol. V), Beijing, China, 18–21 September 2007; Goswami, D.Y., Zhao, Y., Eds.; Springer: Berlin/Heidelberg, Germany, 2007. [Google Scholar] [CrossRef]
- Liu, J.; Wang, S.; Bian, Z.; Shan, M.; Huang, C. Organic/inorganic hybrid solar cells with vertically oriented ZnO nanowires. Appl. Phys. Lett. 2009, 94, 173107. [Google Scholar] [CrossRef]
- Riedel, W.; Wiesner, S.; Greiner, D.; Hinrichs, V.; Rusu, M.; Lux-Steiner, M.C. Hybrid solar cells with ZnO-nanorods and dry processed small molecule absorber. Appl. Phys. Lett. 2014, 104, 173503. [Google Scholar] [CrossRef]
- Matsuo, Y.; Ogumi, K.; Jeon, I.; Wang, H.; Nakagawa, T. Recent progress in porphyrin- and phthalocyanine containing perovskite solar cells. RSC Adv. 2020, 10, 32678. [Google Scholar] [CrossRef]
- Mola, G.T.; Mbuyise, X.G.; Oseni, S.O.; Dlamini, W.M.; Tonui, P.; Arbab, E.A.A.; Kaviyarasu, K.; Maaza, M. Nanocomposite for solar energy application. In Nano Hybrids and Composites; Al-Ahmed, A., Kim, Y.H., Eds.; Trans Tech Publications, Ltd.: Stafa-Zurich, Switzerland, 2018; Volume 20, pp. 90–107. [Google Scholar] [CrossRef]
- Kažukauskas, V.; Arlauskas, A.; Pranaitis, M.; Lessmann, R.; Riede, M.; Leo, K. Conductivity, charge carrier mobility and ageing of ZnPc/C60 solar cells. Opt. Mater. 2010, 32, 1676–1680. [Google Scholar] [CrossRef]
- Jin, B.; Yu, Y.; Peng, R.; Fan, L.; Cai, L.; Fan, B.; Goi, X.; Chu, S. Functional group addition of [6,6]-phenyl-C61-butyric acid methyl ester as electron acceptor in polymer solar cells with high performance. Synth. Met. 2016, 220, 141–146. [Google Scholar] [CrossRef]
- Zhang, F.; Zhuo, Z.; Zhang, J.; Wang, X.; Xu, X.; Wang, Z.; Xin, Y.; Wang, J.; Wang, J.; Tang, W.; et al. Influence of PC60BM or PC70BM as electron acceptor on the performance of polymer solar cells. Sol. Energy Mater. Sol. Cells 2012, 97, 71–77. [Google Scholar] [CrossRef]
- Yamanari, T.; Taima, T.; Sakai, J.; Saito, K. Highly efficient organic thin-film solar cells based on poly(3-hexylthiophene) and soluble C70 fullerene derivative. Jpn. J. Appl. Phys. 2008, 47, 1230–1233. [Google Scholar] [CrossRef]
- Bin, H.; Gao, L.; Zhang, Z.-G.; Yang, Y.; Zhang, Y.; Zhang, C.; Chen, S.; Xue, L.; Yang, C.; Xiao, M.; et al. 11.4% Efficiency non-fullerene polymer solar cells with trialkylsilyl substituted 2D-conjugated polymer as donor. Nat. Commun. 2016, 7, 13651. [Google Scholar] [CrossRef] [PubMed]
- Lin, Y.; Wang, J.; Zhang, Z.-G.; Bai, H.; Li, Y.; Zhu, D.; Zhan, X. An Electron Acceptor Challenging Fullerenes for Efficient Polymer Solar Cells. Adv. Mater. 2015, 27, 1170–1174. [Google Scholar] [CrossRef]
- Zhou, Y.; Eck, M.; Kruger, M. Organic-inorganic hybrid solar cells: State of the art, challenges and perspectives. In Solar Cells-New Aspects and Solutions; Kosyachenko, L.A., Ed.; InTech Europe: Rijeka, Croatia, 2011; pp. 95–120. [Google Scholar] [CrossRef]
- Kumar, R.; Umar, A.; Kumar, G.; Nalwa, H.S.; Kumar, A.; Akhtar, M.S. Zinc oxide nanostructure-based dye-sensitized solar cells. J. Mater Sci. 2017, 52, 4743–4795. [Google Scholar] [CrossRef]
- Wojnarowicz, J.; Chudoba, T.; Lojkowski, W. A review of microwave synthesis of zinc oxide nanomaterials: Reactants, process parameters and morphologies. Nanomaterials 2020, 10, 1086. [Google Scholar] [CrossRef] [PubMed]
- Dubey, R.S.; Krishnamurthy, K.V.; Singh, S. Experimental studies of TiO2 nanoparticles synthesized by sol-gel and solvothermal routes for DSSCs application. Results Phys. 2019, 14, 102390. [Google Scholar] [CrossRef]
- Alfaro Cruz, M.R.; Sanchez-Martinez, D.; Torres-Martínez, L.M. CuO thin films deposited by DC sputtering and their photocatalytic performance under simulated sunlight. Mater. Res. Bull. 2019, 122, 110678. [Google Scholar] [CrossRef]
- Kukreti, K.; Rathod, A.P.S.; Kumar, B. TiO2 Nanoparticles in bulk heterojunction P3HT-PCBM organic solar cell. J. Graph. Era Univ. 2017, 5, 97–111. [Google Scholar]
- Liu, K.; Bi, Y.; Qu, S.; Tan, F.; Chi, D.; Lu, S.; Li, Y.; Kou, Y.; Wang, Z. Efficient hybrid plasmonic polymer solar cells with Ag nanoparticle decorated TiO2 nanorods embedded in the active layer. Nanoscale 2014, 6, 6180. [Google Scholar] [CrossRef]
- Lee, H.; Song, H.-J.; Shim, M.; Lee, C. Towards commercialization of colloidal quantum dot solar cells: Perspective on device structures and manufacturing. Energy Environ. Sci. 2020, 13, 404–431. [Google Scholar] [CrossRef]
- Chang, J.; Waclawik, E.R. Colloidal semiconductor nanocrystals: Controlled synthesis and surface chemistry in organic media. RSC Adv. 2014, 4, 23505–23527. [Google Scholar] [CrossRef]
- Sharma, S.N.; Vats, T.; Dhenadhayalan, N.; Ramamurthy, P.; Narula, A.K. Ligand-dependent transient absorption studies of hybrid polymer:CdSe quantum dot composites. Sol. Energy Mater. Sol. Cells 2012, 100, 6–15. [Google Scholar] [CrossRef]
- Ibrahim, I.; Lim, H.N.; Mohd Zawawi, R.; Ahmad Tajudin, A.; Ng, Y.H.; Guo, H.; Huang, N.M. A review on visible-light induced photoelectrochemical sensors based on CdS nanoparticles. J. Mater. Chem. B 2018, 6, 4551–4568. [Google Scholar] [CrossRef]
- Hullavarad, N.V.; Hullavarad, S.S.; Karulkar, P.C. Cadmium sulphide (CdS) nanotechnology: Synthesis and applications. J. Nanosci. Nanotechnol. 2008, 8, 3272–3299. [Google Scholar] [CrossRef] [PubMed]
- Dai, Q.; Song, Y.; Li, D.; Chen, H.; Kan, S.; Zou, B.; Wang, Y.; Deng, Y.; Hou, Y.; Yu, S.; et al. Temperature dependence of band gap in CdSe nanocrystals. Chem. Phys. Lett. 2007, 439, 65–68. [Google Scholar] [CrossRef]
- Isshiki, M.; Wang, J. II-IV Semiconductors for Optoelectronics: CdS, CdSe, CdTe. In Handbook of Electronic and Photonic Materials; Kasap, S., Capper, P., Eds.; Springer: Cham, Switzerland, 2017. [Google Scholar] [CrossRef]
- Li, Y.; Jing, L.; Qiao, R.; Gao, M. Aqueous synthesis of CdTe nanocrystals: Progresses and perspectives. Chem. Commun. 2011, 47, 9293–9311. [Google Scholar] [CrossRef]
- Shrestha, A.; Batmunkh, M.; Tricoli, A.; Dai, S.; Qiao, S.Z. Recent advance in near-infrared active lead chalcogenide quantum dots: Preparation, post-synthesis ligand exchange and applications in solar cells. Angew. Chem. Int. Ed. 2018, 58, 5202–5224. [Google Scholar] [CrossRef]
- Fu, H.; Tsang, S.-W. Infrared colloidal lead chalcogenide nanocrystals: Synthesis, properties, and photovoltaic applications. Nanoscale 2012, 4, 2187. [Google Scholar] [CrossRef]
- Kolny-Olesiak, J. Colloidal synthesis of CuInS2 and CuInSe2 nanocrystals for photovoltaic applications. In Solar Cell Nanotechnology; Tiwari, A., Boukherroub, R., Sharon, M., Eds.; John Wiley & Sons, Inc.: Hoboken, NJ, USA, 2013; pp. 97–115. [Google Scholar] [CrossRef]
- Yu, P.; Qu, S.; Jia, C.; Liu, K.; Tan, F. Modified synthesis of FeS2 quantum dots for hybrid bulk-heterojunction solar cells. Mater. Lett. 2015, 157, 235–238. [Google Scholar] [CrossRef]
- Dkhil, S.B.; Ebdelli, R.; Dachraoui, W.; Faltakh, H.; Bourguiga, R.; Davenas, J. Improved photovoltaic performance of hybrid solar cells based on silicon nanowire and P3HT. Synth. Met. 2014, 192, 74–81. [Google Scholar] [CrossRef]
- Rakshit, T.; Mondal, S.P.; Manna, I.; Ray, S.K. CdS-decorated ZnO nanorod heterostructures for improved hybrid photovoltaic devices. ACS Appl. Mater. Interfaces 2012, 4, 6085–6095. [Google Scholar] [CrossRef] [PubMed]
- Hu, T.; Li, F.; Yuan, K.; Chen, Y. Efficiency and air-stability improvement of flexible inverted polymer solar cells using ZnO/poly(ethylene glycol) hybrids as cathode buffer layers. ACS Appl. Mater. Interfaces 2013, 5, 5763–5770. [Google Scholar] [CrossRef] [PubMed]
- Zhong, M.; Sheng, D.; Li, C.; Xu, S.; Wei, X. Hybrid bulk heterojunction solar cells based on poly (3-hexylthiophene) and Z907-modified ZnO nanorods. Sol. Energy Mater. Sol. Cells 2014, 121, 22–27. [Google Scholar] [CrossRef]
- Anjum, S.; Ikram, M.; Ali, S. Hybrid organic solar cells based on polymer/metal oxide nanocrystals. J. Ovonic Res. 2015, 11, 85–89. [Google Scholar]
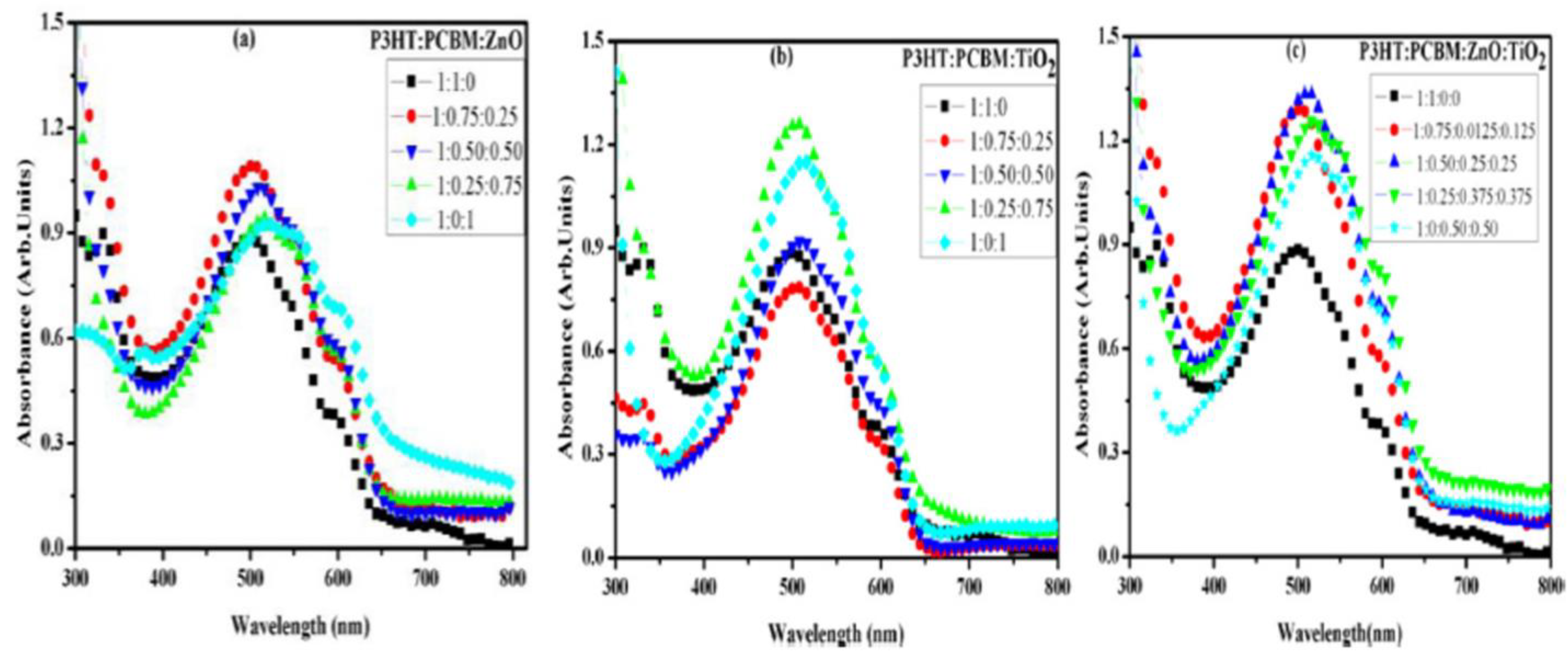
- Muller, T.; Ramashia, T.A.; Motaung, D.E.; Cummings, F.; Malgas, G.; Oliphant, C.; Arendse, C. Effect of additional electron acceptor in hybrid P3HT:PCBM:ZnO spin-coated films for photovoltaic application. Phys. Status Solidi A 2016, 213, 1915–1921. [Google Scholar] [CrossRef]
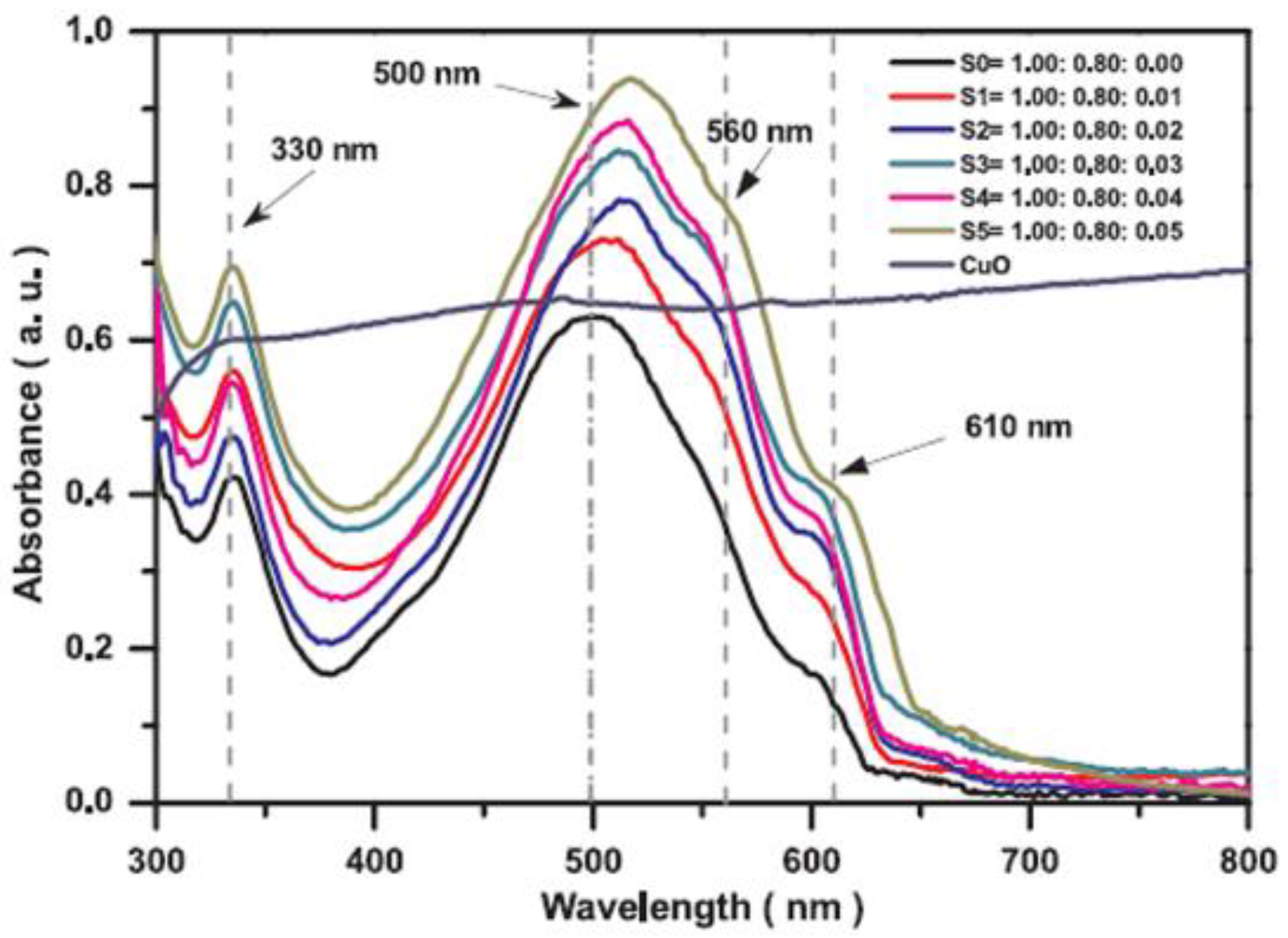
- Wanninayake, A.; Gunashekar, S.; Li, S.; Church, B.C.; Abu-Zahra, N. CuO nanoparticles based bulk heterojunction solar cells: Investigations on morphology and performance. J. Sol. Energy Eng. 2015, 137, 031016. [Google Scholar] [CrossRef]
- Ikram, M.; Imran, M.; Nunzi, J.M.; Ali, S. Efficient inverted hybrid solar cells using both CuO and P3HT as an electron donor material. J. Mater. Sci. Mater. Electron. 2015, 26, 6478–6483. [Google Scholar] [CrossRef]
- Ikram, M.; Imran, M.; Nunzi, J.M.; Islah-u-din; Ali, S. Replacement of P3HT and PCBM with metal oxides nanoparticles in inverted hybrid organic solar cells. Synth. Met. 2015, 210, 268–272. [Google Scholar] [CrossRef]
- Wanninayake, A.P.; Church, B.C.; Abu-Zahra, N. Effect of ZnO nanoparticles on the power conversion efficiency of organic photovoltaic devices synthesized with CuO nanoparticles. AIMS Mater. Sci. 2016, 3, 927–937. [Google Scholar] [CrossRef]
- Salim, E.; Bobbara, S.R.; Oraby, A.; Nunzi, J.M. Copper oxide nanoparticle doped bulk-heterojunction photovoltaic devices. Synth. Met. 2019, 252, 21–28. [Google Scholar] [CrossRef]
- Siddiqui, H.; Parra, M.R.; Pandey, P.; Qureshi, M.S.; Haque, F.Z. Utility of copper oxide nanoparticles (CuO-NPs) as efficient electron donor material in bulk-heterojunction solar cells with enhanced power conversion efficiency. J. Sci.-Adv. Mater. Dev. 2020, 5, 104–110. [Google Scholar] [CrossRef]
- Dong, Q.; Yu, W.; Li, Z.; Yao, S.; Zhang, X.; Yang, B.; Im, C.; Tian, W. All-water-solution processed solar cells based on PPV and TiO2 nanocrystals. Sol. Energy Mater. Sol. Cells 2012, 104, 75–80. [Google Scholar] [CrossRef]
- Thao, T.T.; Trung, T.Q.; Truong, V.-V.; Dinh, N.N. Enhancement of power efficiency and stability of P3HT-based organic solar cells under elevated operating-temperatures by using a nanocomposite photoactive layer. J. Nanomater. 2015, 463565. [Google Scholar] [CrossRef]
- Geethalakshmi, D.; Muthukumarasamy, N.; Balasundaraprabhu, R. CSA-doped PANI/TiO2 hybrid BHJ solar cells—Material synthesize and device fabrication. Mater. Sci. Semicond. Process. 2016, 51, 71–80. [Google Scholar] [CrossRef]
- Amber Yousaf, S.; Ikram, M.; Ali, S. Compositional engineering of acceptors for highly efficient bulk heterojunction hybrid organic solar cells. J. Colloid Interface Sci. 2018, 527, 172–179. [Google Scholar] [CrossRef]
- Huynh, W.U.; Dittmer, J.J.; Libby, W.C.; Whiting, G.L.; Alivisatos, A.P. Controlling the morphology of nanocrystal–polymer composites for solar cells. Adv. Funct. Mater. 2003, 13, 73–79. [Google Scholar] [CrossRef]
- Lek, J.Y.; Xi, L.; Kardynal, B.E.; Wong, L.H.; Lam, Y.M. Understanding the effect of surface chemistry on charge generation and transport in poly (3-hexylthiophene)/CdSe hybrid solar cells. ACS Appl. Mater. Interfaces 2011, 3, 287–292. [Google Scholar] [CrossRef] [PubMed]
- Olson, J.D.; Gray, G.P.; Carter, S.A. Optimizing hybrid photovoltaics through annealing and ligand choice. Sol. Energy Mater. Sol. Cells 2009, 93, 519–523. [Google Scholar] [CrossRef]
- Seo, J.; Kim, S.J.; Kim, W.J.; Singh, R.; Samoc, M.; Cartwright, A.N.; Prasad, P.N. Enhancement of the photovoltaic performance in PbS nanocrystals: P3HT hybrid composite devices by post-tratement-driven ligand exchance. Nanotechnology 2009, 20, 095202. [Google Scholar] [CrossRef] [PubMed]
- Brandenburg, J.E.; Jin, X.; Kruszynska, M.; Ohland, J.; Kolny-Olesiak, J.; Riedel, I.; Parisi, J. Influence of particle size in hybrid solar cells composed of CdSe nanocrystals and poly(3-hexylthiophene). J. Appl. Phys. 2011, 110, 064509. [Google Scholar] [CrossRef]
- Qian, L.; Yang, J.; Zhou, R.; Tang, A.; Zheng, Y.; Tseng, T.-K.; Bera, D.; Xue, J.; Holloway, P.H. Hybrid polymer-CdSe solar cells with a ZnO nanoparticle buffer layer for improved efficiency and lifetime. J. Mater. Chem. 2011, 21, 3814. [Google Scholar] [CrossRef]
- Radychev, N.; Lokteva, I.; Witt, F.; Kolny-Olesiak, J.; Borchert, H.; Parisi, J. Physical origin of the impact of different nanocrystal surface modifications on the performance of CdSe/P3HT hybrid solar cells. J. Phys. Chem. C. 2011, 115, 14111–14122. [Google Scholar] [CrossRef]
- Zhou, Y.; Eck, M.; Veit, C.; Zimmermann, B.; Rauscher, F.; Niyamakom, P.; Yilmaz, S.; Dumsch, I.; Allard, I.S.; Scherf, U.; et al. Efficiency enhancement for bulk-heterojunction hybrid solar cells based on acid treated CdSe quantum dots and low bandgap polymer PCPDTBT. Sol. Energy Mater. Sol. Cells 2011, 95, 1232–1237. [Google Scholar] [CrossRef]
- Greaney, M.J.; Das, S.; Webber, D.H.; Bradforth, S.E.; Brutchey, R.L. Improving open circuit potential in hybrid P3HT:CdSe bulk heterojunction solar cells via colloidal tert-butylthiol ligand exchange. ACS Nano 2012, 6, 4222–4230. [Google Scholar] [CrossRef]
- Lek, J.Y.; Lam, Y.M.; Niziol, J.; Marzec, M. Understanding polycarbazole-based polymer:CdSe hybrid solar cells. Nanotechnology 2012, 23, 315401. [Google Scholar] [CrossRef] [PubMed]
- Jeltsch, K.F.; Schädel, M.; Bonekamp, J.-B.; Niyamakom, P.; Rauscher, F.; Lademann, H.W.A.; Dumsch, I.; Allard, S.; Scherf, U.; Meerholz, K. Efficiency enhanced hybrid solar cells using a blend of quantum dots and nanorods. Adv. Funct. Mater. 2012, 22, 397–404. [Google Scholar] [CrossRef]
- Kim, S.W.; Kim, D.W.; Kim, H.J.; Lee, Y.J.; Wang, S.S.; Baek, K.Y. Hybrid nanocomposite of CdSe quantum dots and a P3HT-b-PDMAEMA block copolymer for photovoltaic applications. Mater. Sci. Forum 2012, 700, 120–124. [Google Scholar] [CrossRef]
- Kwon, S.; Moon, H.C.; Lim, K.-G.; Bae, D.; Jang, S.; Shin, J.; Park, J.; Lee, T.-W.; Kim, J.K. Improvement of power conversion efficiency of P3HT:CdSe hybrid solar cells by enhanced interconnection of CdSe nanorods via decomposable selenourea. J. Mater. Chem. A 2013, 1, 2401–2405. [Google Scholar] [CrossRef]
- Zhou, R.; Stalder, R.; Xie, D.; Cao, W.; Zheng, Y.; Yang, Y.; Plaisant, M.; Holloway, P.H.; Schanze, K.S.; Reynolds, J.R.; et al. Enhancing the efficiency of solution-processed polymer:colloidal nanocrystal hybrid photovoltaic cells using ethanedithiol treatment. ACS Nano 2013, 7, 4846–4854. [Google Scholar] [CrossRef]
- Fu, W.; Wang, L.; Zhang, Y.; Ma, R.; Zuo, L.; Mai, J.; Lau, T.-K.; Du, S.; Lu, X.; Shi, M.; et al. Improving polymer/nanocrystal hybrid solar cell performance via tuning ligand orientation at CdSe quantum dot surface. ACS Appl. Mater. Interfaces 2014, 6, 19154–19160. [Google Scholar] [CrossRef]
- Benchaabane, A.; Belhadi, J.; Ben hamed, Z.; Lejeune, M.; Lahmar, A.; Sanhoury, M.A.; Kouki, F.; Zellama, K.; Zeinert, A.; Bouchriha, H. Effect of CdSe nanoparticles incorporation on the performance of P3OT organic photovoltaic cells. Mater. Sci. Semicond. Process. 2016, 41, 343–349. [Google Scholar] [CrossRef]
- Dayneko, S.; Linkov, P.; Martynov, I.; Tameev, A.; Tedoradze, M.; Samokhvalov, P.; Nabiev, I.; Chistyakov, A. Photoconductivity of composites based on CdSe quantum dots and low-band-gap polymers. Physica E Low Dimens. Syst. Nanostruct. 2016, 79, 206–211. [Google Scholar] [CrossRef]
- Xu, W.; Tan, F.; Liu, X.; Zhang, W.; Qu, S.; Wang, Z.; Wang, Z. Efficient organic/inorganic hybrid solar cell integrating polymer nanowires and inorganic nanotetrapods. Nanoscale Res. Lett. 2017, 12, 11. [Google Scholar] [CrossRef]
- Madsuha, A.F.; Yuwono, A.H.; Sofyan, N.; Krueger, M. Enhanced device performance of bulk heterojunction (BHJ) hybrid solar cells based on colloidal CdSe quantum dots (QDs) via optimized hexanoic acid-assisted washing treatment. Adv. Mater. Sci. Eng. 2019, 7516890. [Google Scholar] [CrossRef]
- Nabil, M.; Mohamed, S.A.; Easawi, K.; Obayya, S.S.A.; Negm, S.; Talaat, H.; El-Mansy, M.K. Surface modification of CdSe nanocrystals: Application to polymer solar cell. Curr. Appl. Phys. 2020, 20, 470–476. [Google Scholar] [CrossRef]
- Kumar, A.; Kumar Jarwal, D.; Kumar Mishra, A.; Ratan, S.; Kumar, C.; Chandra Upadhyay, D.; Mukherjee, B.; Jit, S. Synergistic effect of CdSe quantum dots (QDs) and PC61BM on ambient-air processed ZnO QDs/PCDTBT: PC61BM:CdSe QDs/MoO3 based ternary organic solar cells. Nanotechnology 2020, 31, 465404. [Google Scholar] [CrossRef] [PubMed]
- Yang, X.-K.; Qiao, J.-W.; Chen, Z.-H.; Wen, Z.-C.; Yin, H.; Hao, X.-T. CdSe quantum dot organic solar cells with improved photovoltaic performance. J. Phys. D Appl. Phys. 2021, 54. [Google Scholar] [CrossRef]
- Fan, Z.; Zhang, H.; Yu, W.; Xing, Z.; Wei, H.; Dong, Q.; Tian, W.; Yang, B. Aqueous-solution-processed hybrid solar cells from poly(1,4-naphthalenevinylene) and CdTe nanocrystals. ACS Appl. Mater. Interfaces 2011, 3, 2919–2923. [Google Scholar] [CrossRef] [PubMed]
- Chen, Z.; Zhang, H.; Du, X.; Cheng, X.; Chen, X.; Jiang, Y.; Yang, B. From planar-heterojunction to n–i structure: An efficient strategy to improve short-circuit current and power conversion efficiency of aqueous-solution-processed hybrid solar cells. Energy Environ. Sci. 2013, 6, 1597. [Google Scholar] [CrossRef]
- Yao, S.; Chen, Z.; Li, F.; Xu, B.; Song, J.; Yan, L.; Jin, G.; Wen, S.; Wang, C.; Yang, B.; et al. High-efficiency aqueous-solution-processed hybrid solar cells based on P3HT dots and CdTe nanocrystals. ACS Appl. Mater. Interfaces 2015, 7, 7146–7152. [Google Scholar] [CrossRef]
- Jin, G.; Chen, N.; Zeng, Q.; Liu, F.; Yuan, W.; Xiang, S.; Feng, T.; Du, X.; Ji, T.; Wang, L.; et al. Aqueous-processed polymer/nanocrystal hybrid solar cells with double-side bulk heterojunction. Adv. Energy Mater. 2017, 8, 1701966. [Google Scholar] [CrossRef]
- Chen, N.; Jin, G.; Wang, L.; Sun, H.; Zeng, Q.; Yang, B.; Sun, H. Highly efficient aqueous-processed hybrid solar cells: Control depletion region and improve carrier extraction. Adv. Energy Mater. 2019, 9, 1803849. [Google Scholar] [CrossRef]
- Aruna-Devi, R.; Marasamy, L.; Cruz-Gómez, J.; Mayén-Hernández, S.A.; De Moure-Flores, F.; Santos-Cruz, J. Organic polymers/inorganic CdS nanostructures. Mater. Lett. 2020, 282, 128856. [Google Scholar] [CrossRef]
- Tan, Z.N.; Zhang, W.Q.; Qian, D.P.; Zheng, H.; Xiao, S.Q.; Yang, Y.P.; Zhu, T.; Xu, J. Efficient hybrid infrared solar cells based on P3HT and PbSe nanocrystal quantum dots. Mater. Sci. Forum 2011, 685, 38–43. [Google Scholar] [CrossRef]
- Thomas, A.; Vinayakan, R.; Ison, V.V. An inverted ZnO/P3HT:PbS bulk-heterojunction hybrid solar cell with a CdSe quantum dot interface buffer layer. RSC Adv. 2020, 10, 16693–16699. [Google Scholar] [CrossRef]
- Baek, S.-W.; Jun, S.; Kim, B.; Proppe, A.H.; Ouellette, O.; Voznyy, O.; Kim, C.; Kim, J.; Walters, G.; Song, J.H.; et al. Efficient hybrid colloidal quantum dot/organic solar cells mediated by near-infrared sensitizing small molecules. Nat. Energy 2019, 4, 969–976. [Google Scholar] [CrossRef]
- Tan, F.; Qu, S.; Wu, J.; Liu, K.; Zhou, S.; Wang, Z. Preparation of SnS2 colloidal quantum dots and their application in organic/inorganic hybrid solar cells. Nanoscale Res. Lett. 2011, 6, 298. [Google Scholar] [CrossRef]
- Benchaabane, A.; Hamed, Z.B.; Telfah, A.; Sanhoury, M.A.; Kouki, F.; Zellama, K.; Bouchriha, H. Effect of OA-ZnSe nanoparticles incorporation on the performance of PVK organic photovoltaic cells. Mater. Sci. Semicond. Process. 2017, 64, 115–123. [Google Scholar] [CrossRef]
- Jabeen, U.; Adhikari, T.; Shah, S.M.; Pathak, D.; Nunzi, J.-M. Synthesis, characterization and photovoltaic performance of Mn-doped ZnS quantum dots-P3HT hybrid bulk heterojunction solar cells. Opt. Mater. 2017, 73, 754–762. [Google Scholar] [CrossRef]
- Han, Y.W.; Jeon, S.J.; Choi, J.Y.; Kim, J.H.; Moon, D.K. Highly efficient Ternary Solar Cells of 10.2% with Core/Shell Quantum Dots via FRET Effect. Sol. RRL 2018, 2, 1800077. [Google Scholar] [CrossRef]
- Lakhotiya, G.; Belsare, N.; Arbuj, S.; Kale, B.; Rana, A. Enhanced performance of PTB7-Th:PCBM based active layers in ternary organic solar cells. RSC Adv. 2019, 9, 7457–7463. [Google Scholar] [CrossRef]
- Lakhotiya, G.; Belsare, N.; Rana, A.; Gupta, V. Cu2S nanocrystals incorporated highly efficient non-fullerene ternary organic solar cells. Curr. Appl. Phys. 2019, 19, 394–399. [Google Scholar] [CrossRef]
- Nam, M.; Lee, S.; Park, J.; Kim, S.-W.; Lee, K.-K. Development of hybrid photovoltaic cells by incorporating CuInS2 quantum dots into organic photoactive layers. Jpn. J. Appl. Phys. 2011, 50, 06GF02. [Google Scholar] [CrossRef]
- Kumari, A.; Singh, I.; Dixit, S.K. Effect of annealing on graphene incorporated poly-(3-hexylthiophene):CuInS2 photovoltaic device. AIP Conf. Proc. 2014, 1620, 35. [Google Scholar] [CrossRef]
- Liu, Z.; Sun, Y.; Yuan, J.; Wei, H.; Huang, X.; Han, L.; Wang, W.; Wang, H.; Ma, W. High-efficiency hybrid solar cells based on polymer/PbSxSe1-x nanocrystals benefiting from vertical phase segregation. Adv. Mater. 2013, 25, 5772–5778. [Google Scholar] [CrossRef] [PubMed]
- Sahoo, M.K.; Kale, P. Integration of silicon nanowires in solar cell structure for efficiency enhancement: A review. J. Mater. 2018, 5, 34–48. [Google Scholar] [CrossRef]
- Ding, Y.; Zhou, S.; Juangsa, F.B.; Sugaya, M.; Zhang, X.; Zhao, Y.; Nozaki, T. Double-parallel-junction hybrid solar cells based on silicon nanocrystals. Org. Electron. 2016, 30, 99–104. [Google Scholar] [CrossRef]
- Eisenhawer, B.; Sensfuss, S.; Sivakov, V.; Pietsch, M.; Andrä, G.; Falk, F. Increasing the efficiency of polymer solar cells by silicon nanowires. Nanotechnology 2011, 22, 315401. [Google Scholar] [CrossRef] [PubMed]
- Švrček, V.; Yamanari, T.; Shibata, Y.; Kondo, M. Tailoring of hybrid silicon nanocrystal-based bulk heterojunction photovoltaic properties upon nanocrystal laser processing in liquid medium. Acta Mater. 2011, 59, 764–773. [Google Scholar] [CrossRef]
- Švrček, V.; Yamanari, T.; Mariotti, D.; Matsubara, K.; Kondo, M. Enhancement of hybrid solar cell performance by polythieno [3,4-b]thiophenebenzodithiophene and microplasma-induced surface engineering of silicon nanocrystals. Appl. Phys. Lett. 2012, 100, 223904. [Google Scholar] [CrossRef]
- Dkhil, S.B.; Bourguiga, R.; Davenas, J.; Cornu, D. Influence of the polymer matrix on the efficiency of hybrid solar cells based on silicon nanowires. Mater. Sci. Eng. B 2012, 177, 173–179. [Google Scholar] [CrossRef]
- Liu, C.-Y.; Kortshagen, U.R. Hybrid solar cells from MDMO-PPV and silicon nanocrystals. Nanoscale 2012, 4, 3963. [Google Scholar] [CrossRef]
- Kim, J.; Lee, S.; Nam, S.; Lee, H.; Kim, H.; Kim, Y. A Pronounced dispersion effect of crystalline silicon nanoparticles on the performance and stability of polymer:fullerene solar cells. ACS Appl. Mater. Interfaces 2012, 4, 5300–5308. [Google Scholar] [CrossRef]
- Ding, Y.; Gresback, R.; Yamada, R.; Okazaki, K.; Nozaki, T. Hybrid silicon nanocrystal/poly(3-hexylthiophene-2,5-diyl) solar cells from a chlorinated silicon precursor. Jpn. J. Appl. Phys. 2013, 52, 11NM04. [Google Scholar] [CrossRef]
- Yan, Q.; Zuo, L.; Gu, Z.; Yang, L.; Wang, R.; Wang, M.; Chen, H. Improved photovoltaic performance of MEH-PPV/PCBM solar cells via incorporation of Si nanocrystals. Chin. J. Chem. 2013, 31, 1380–1384. [Google Scholar] [CrossRef]
- Chang, S.-C.; Hsiao, Y.-J.; Li, T.-S. P3HT:PCBM Incorporated with silicon nanoparticles as photoactive layer in efficient organic photovoltaic devices. J. Nanomater. 2013, 354035. [Google Scholar] [CrossRef]
- Chehata, N.; Ltaief, A.; Beyou, E.; Ilahi, B.; Salem, B.; Baron, T.; Gentile, P.; Maaref, H.; Bouazizi, A. Functionalized silicon nanowires/conjugated polymer hybrid solar cells: Optical, electrical and morphological characterizations. J. Lumin. 2015, 168, 315–324. [Google Scholar] [CrossRef]
- Arunmetha, S.; Vinoth, M.; Srither, S.R.; Karthik, A.; Sridharpanday, M.; Suriyaprabha, R.; Manivasakan, P.; Rajendran, V. Study on production of silicon nanoparticles from quartz sand for hybrid solar cell applications. J. Electron. Mater. 2018, 47, 493–502. [Google Scholar] [CrossRef]
- Vinoth, M.; Surendhiran, S.; Senthilmurugan, P.R.; Rajendran, V. Enhanced photovoltaic performance of hybrid solar cells with a calcium interfacial metal electrode. J. Electron. Mater. 2019, 48, 4589–4597. [Google Scholar] [CrossRef]
- De la Torre, G.; Bottari, G.; Torres, T. Phthalocyanines and subphthalocyanines: Perfect partners for fullerenes and carbon nanotubes in molecular photovoltaics. Adv. Energy Mater. 2016, 7, 1601700. [Google Scholar] [CrossRef]
- Preda, N.; Enculescu, M.; Enculescu, I. Polysaccharide-assisted crystallization of ZnO micro/nanostructures. Mater. Lett. 2014, 115, 256–260. [Google Scholar] [CrossRef]
- Glaser, M.; Peisert, H.; Adler, H.; Aygül, U.; Ivanovic, M.; Nagel, P.; Merz, M.; Schuppler, S.; Chassé, T. Electronic structure at transition metal phthalocyanine-transition metal oxide interfaces: Cobalt phthalocyanine on epitaxial MnO films. J. Chem. Phys. 2015, 142, 101918. [Google Scholar] [CrossRef] [PubMed]




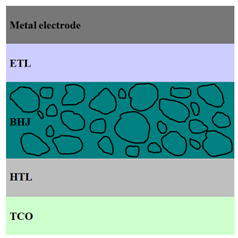
| Regular PV Cell Architectures | ||
 |  |  |
| ITO/PEDOT:PSS/ P3HT:PCBM/LiF/Al [98] | ITO/PEDOT:PSS/ P3HT:CdSe/LiF/Al [99] | ITO/PEDOT:PSS/ P3HT:PCBM:CuS/LiF/Al [98] |
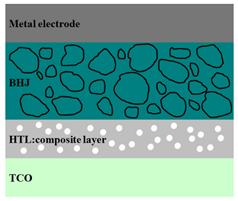 |  |  |
| ITO/PEDOT:PSS:Si/ MEH-PPV:PCBM/Al [97] | ITO/PEDOT:PSS/ P3HT:ZnO/Al [100] | ITO/PEDOT:PSS/ P3HT:PCBM:Si/Al [101] |
| Inverted PV cell architectures | ||
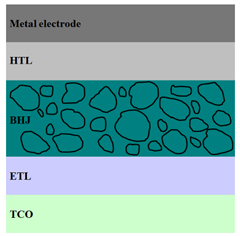 |  | 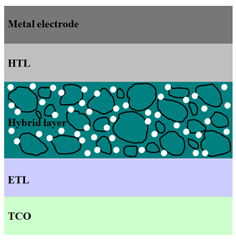 |
| ITO/ZnO/ P3HT:PCBM/MoO3/Ag [94] | ITO/ZnO/ P3HT:ZnO/PEDOT:PSS/Au [102] | ITO/ZnO/ P3HT:PCBM:ZnO/MoO3/Ag [94] |
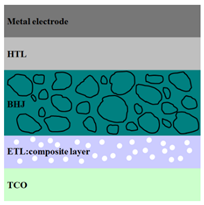 |  | 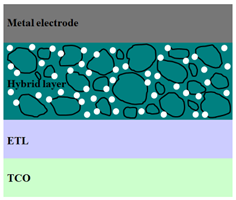 |
| ITO/F4-TCNQ:ZnO/ PTB7-Th:PC71BM/MoO3/Al [96] | ITO/TiO2/ MEH-PPV:CdS/Au [103] | ITO/ZnO/ P3HT:PCBM:Si/Al [104] |
| PV Cell Structure | Device Architecture | Composite Layer Role | PCE (%) | PCE (%) Reference PV Cell | Published Year | Ref. |
|---|---|---|---|---|---|---|
| Composites based on ZnO | ||||||
| ITO/PEDOT:PSS/P3HT:ZnO/Al | Regular | AL | 1.7 | - | 2011 | [100] |
| ITO/PEDOT:PSS/P3HT-E:ZnO/Al | 0.83 | - | ||||
| ITO/ZnO/P3HT:ZnO/PEDOT:PSS/Au | Inverted | AL | 1 | - | 2011 | [102] |
| ITO/PEDOT:PSS/P3HT:ZnO/Al | Regular | AL | 0.12 | - | 2012 | [175] |
| ITO/PEDOT:PSS/P3HT:CdS-ZnO/Al | 0.38 | - | ||||
| ITO/PEG:ZnO/P3HT:PCBM/PEDOT:PSS/Ag | Inverted | CBL | 3.3 | 2.3 | 2013 | [176] |
| ITO/PEDOT:PSS/P3HT:ZnO/Al | Regular | AL | 0.21 | - | 2014 | [177] |
| ITO/PEDOT:PSS/P3HT:PCBM:ZnO/Al | Regular | AL | 0.77 | 0.65 | 2015 | [178] |
| ITO/TiO2/P3HT:PCBM:ZnO/Al | Inverted | 0.63 | 0.61 | |||
| ITO/ZnO/P3HT:PCBM:ZnO/MoO3/Ag | Inverted | AL | 2.96 | 2.69 | 2015 | [94] |
| ITO/TiO2/P3HT:PCBM:ZnO/MoO3/Al | Inverted | AL | 1.7 | 1 | 2016 | [179] |
| ITO/F4-TCNQ-ZnO/PTB7-Th:PC71BM/MoO3/Al | Inverted | CBL | 8.14 | 7.17 | 2020 | [96] |
| ITO/F4-TCNQ-ZnO/PTB7-Th:ITIC/MoO3/Al | 6.96 | 6.13 | ||||
| Composites based on CuO | ||||||
| ITO/PEDOT:PSS/P3HT:PC70BM:CuO/Al | Regular | AL | 2.963 | 2.106 | 2015 | [180] |
| ITO/ZnO/P3HT:PCBM:CuO/MoO3/Ag | Inverted | AL | 3.7 | 3.4 | 2015 | [181] |
| ITO/ZnO/P3HT:PCBM:ZnO:CuO/MoO3/Ag | Inverted | AL | 4.09 | 3.45 | 2015 | [182] |
| ITO/PEDOT:PSS/P3HT:PC70BM:CuO/ZnO/Al | Regular | AL | 3.95 | 2.358 | 2016 | [183] |
| ITO/ZnO/P3HT:PCBM:CuO/MoO3/Ag | Inverted | AL | 4.1 | 3 | 2019 | [184] |
| ITO/PEDOT:PSS/P3HT:PC70BM:CuO/Al | Regular | AL | 3.82 | 2.85 | 2020 | [185] |
| Composites based on TiO2 or TiOx | ||||||
| ITO/PPV/PPV:TiO2/LiF/Al | Regular | AL | 0.11 | - | 2012 | [186] |
| ITO/PEDOT/P3HT:PCBM:TiO2/LiF/Al | Regular | AL | 2.1 | 1.6 | 2015 | [187] |
| ITO/ZnO/P3HT:PCBM:TiO2/MoO3/Ag | Inverted | AL | 3.10 | 2.69 | 2015 | [94] |
| ITO/ZnO/P3HT:PCBM:ZnO:TiO2/MoO3/Ag | Inverted | 3.03 | 2.69 | |||
| ITO/PEDOT:PSS/CSA-doped PANI-TiO2/Al | Regular | AL | 0.21 | - | 2016 | [188] |
| ITO/PEDOT:PSS/PEG-TiOx/ PDTTBT-3:PC71BM/MoO3/Ag | Inverted | CBL | 5.14 | 2.80 | 2015 | [91] |
| ITO/PEDOT:PSS/PEG-TiOx/ PTB7:PC71BM/MoO3/Ag | 8.72 | 6.82 | ||||
| ITO/PEDOT:PSS/PEG-TiOx/ PTB7-Th:PC71BM/MoO3/Ag | 9.05 | 7.36 | ||||
| Composites based on Cr2O3 | ||||||
| ITO/ZnO/P3HT:PCBM:Cr2O3/MoO3/Ag | Inverted | AL | 3.67 | 2.51 | 2018 | [189] |
| ITO/ZnO/PTB7:PC70BM:Cr2O3/MoO3/Ag | 6.72 | 5.61 | ||||
| Composites based on CdSe | ||||||
| ITO/PEDOT:PSS/P3HT:CdSe/Al | Regular | AL | 0.45 | - | 2011 | [194] |
| ITO/PEDOT:PSS/P3HT:CdSe/LiF/Al | Regular | AL | 1.12 | - | 2011 | [99] |
| ITO/PEDOT:PSS/P3HT:CdSe/ZnO/Al | Regular | AL | 2.2 | - | 2011 | [195] |
| ITO/PEDOT:PSS/P3HT:CdSe/Al | Regular | AL | 2.0 | - | 2011 | [196] |
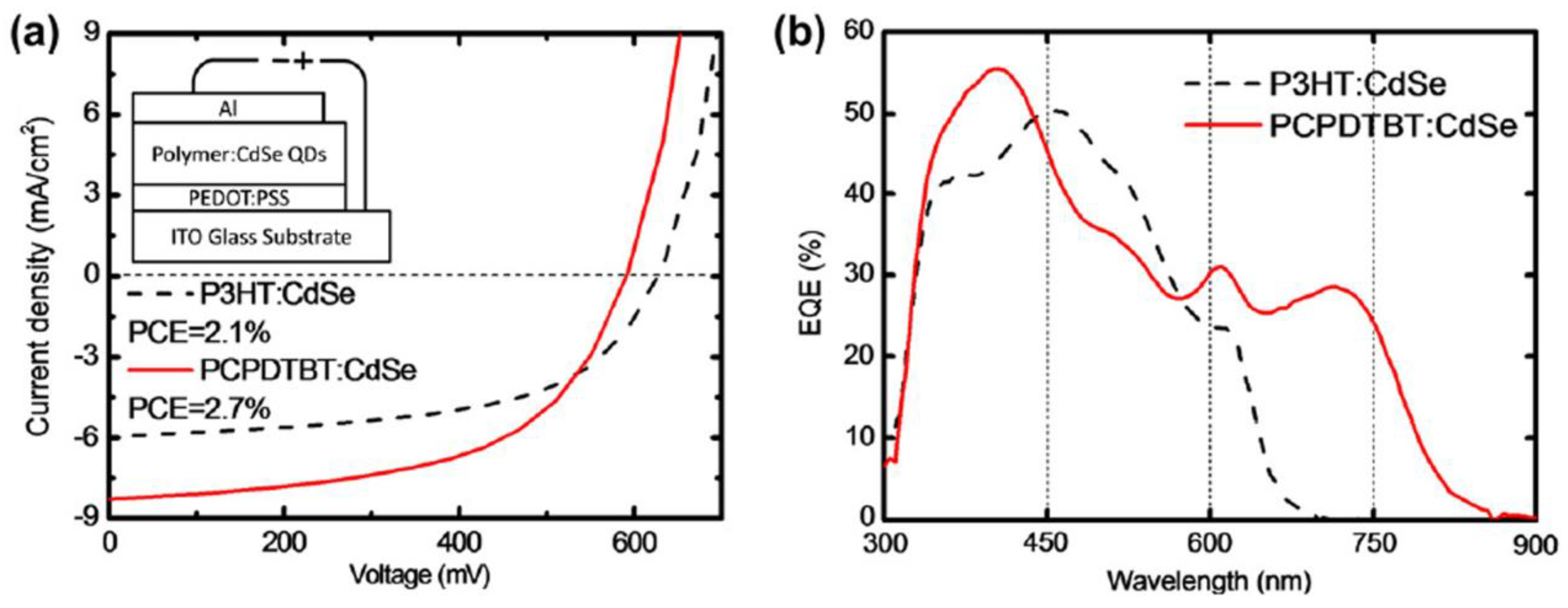
| ITO/PEDOT:PSS/PCPDTBT:CdSe/Al | Regular | AL | 2.7 | - | 2011 | [197] |
| ITO/PEDOT:PSS/P3HT:CdSe/Al | Regular | AL | 1.9 | - | 2012 | [198] |
| ITO/PEDOT:PSS/P3HT:CdSe/Al | Regular | AL | 2.17 | - | 2012 | [199] |
| ITO/PEDOT:PSS/PCDTBT:CdSe/Al | 2 | - | ||||
| ITO/PEDOT:PSS/PCPDTBT:CdSe/Ca/Ag | Regular | AL | 2.8 | - | 2012 | [200] |
| ITO/PEDOT:PSS/PCPDTBT:CdSe/Ca/Ag | 3.6 | - | ||||
| ITO/PEDOT:PSS/P3HT-b-PDMAEMA:CdSe/Al | Regular | AL | 0.1 | - | 2012 | [201] |
| ITO/PEDOT:PSS/P3HT:CdSe-SeU/Al | Regular | AL | 2.63 | - | 2013 | [202] |
| ITO/PEDOT:PSS/PCPDTBT:CdSe/Al | Regular | AL | 4.7 | - | 2013 | [203] |
| ITO/PEDOT:PSS/PCPDTBT:CdSe/CdSe/PFN/Al | Regular | AL | 4.18 | - | 2014 | [204] |
| ITO/P3OT:CdSe/Al | Regular | AL | 0.51 | 0.025 | 2016 | [205] |
| ITO/TiO2/P3HT:CdSe/MoO3/Au/Ag | Inverted | AL | 1.7 | - | 2017 | [207] |
| ITO/PEDOT:PSS/PCPDTBT:CdSe/Al | Regular | AL | 2.81 | - | 2019 | [208] |
| FTO/TiOx/P3HT:PCBM:CdSe/MoO3/Ag | Inverted | AL | 2.55 | 1.99 | 2020 | [209] |
| ITO/ZnO/PCDTBT:PCBM:CdSe/MoO3/Ag | Inverted | AL | 5.02 | 3.62 | 2020 | [210] |
| ITO/ZnO/PTB7-Th:PC71BM:CdSe/MoO3/Ag | Inverted | AL | 9.57 | 8.58 | 2021 | [211] |
| Composites based on CdTe | ||||||
| ITO/PEDOT:PSS/PNV:CdTe/Al | Regular | AL | 0.86 | 0.001 | 2011 | [212] |
| ITO/TiO2/CdTe/CdCl2/PPV:CdTe/MoO3/Au | Inverted | AL | 4.76 | - | 2013 | [213] |
| ITO/TiO2/CdTe/P3HT:CdTe/MoO3/Au | Inverted | AL | 4.32 | - | 2015 | [214] |
| ITO/TiO2/CdTe/MEHPPV:CdTe/MoO3/Au | Inverted | AL | 4.2 | - | 2016 | [133] |
| ITO/TiO2/TiO2:CdTe/CdTe:PPV/MoO3/Au | Inverted | AL | 6.01 | - | 2017 | [215] |
| ITO/ZnO/ZnO:CdTe/CdTe/CdTe:PPV/MoO3/Au | Inverted | AL | 6.51 | - | 2019 | [216] |
| ITO/TiO2/TiO2:CdTe/CdTe/CdTe:PPV/MoO3/Au | 5.87 | - | ||||
| Composites based on CdS | ||||||
| ITO/TiO2/MEH-PPV:CdS/Au | Inverted | AL | 0.23 | 0.1 | 2011 | [103] |
| ITO/ZnO/PTB7:CdS:PCBM/MoO3/Ag | Regular | AL | 7.01 | 6.3 | 2016 | [121] |
| FTO/TiO2/P3HT:PCBM:CdS/MoO3/Ag | Inverted | AL | 2.42 | 1.2 | 2021 | [217] |
| Composites based on PbSe or PbS | ||||||
| ITO/PEDOT:PSS/P3HT/P3HT:PbSe/Al | Regular | AL | 0.26 | 0.1 | 2011 | [218] |
| ITO/PEDOT:PSS/Si-PCPDTBT:PbS/ZnO/Al | Regular | AL | 4.8 | - | 2016 | [125] |
| ITO/ZnO/PBDTTT-E-T:IEICO:PbS/MoO3/Ag | Inverted | AL | 13.1 | - | 2019 | [220] |
| Composites based on SnS2 or FeS2 | ||||||
| ITO/PEDOT:PSS/MEHPPV:SnS2/ZnO/Al | Regular | AL | 0.26 | - | 2011 | [221] |
| ITO/PEDOT:PSS/P3HT:FeS2/ZnO/Al | Regular | AL | 0.61 | - | 2015 | [173] |
| Composites based on ZnSe or ZnS | ||||||
| ITO/PVK:OA-ZnSe/Al | Regular | AL | 0.25 | 0.02 | 2017 | [222] |
| ITO/ZnO/P3HT:ZnS/MoO3/Ag | Inverted | AL | 0.52 | - | 2017 | [223] |
| ITO/ZnO/P3HT:Mn-doped ZnS/MoO3/Ag | 0.83 | |||||
| ITO/ZnO/PTBT:PC70BM:InP-ZnS/MoO3/Ag | Inverted | AL | 10.2 | 9.1 | 2018 | [224] |
| Composites based on Cu2S or CuS | ||||||
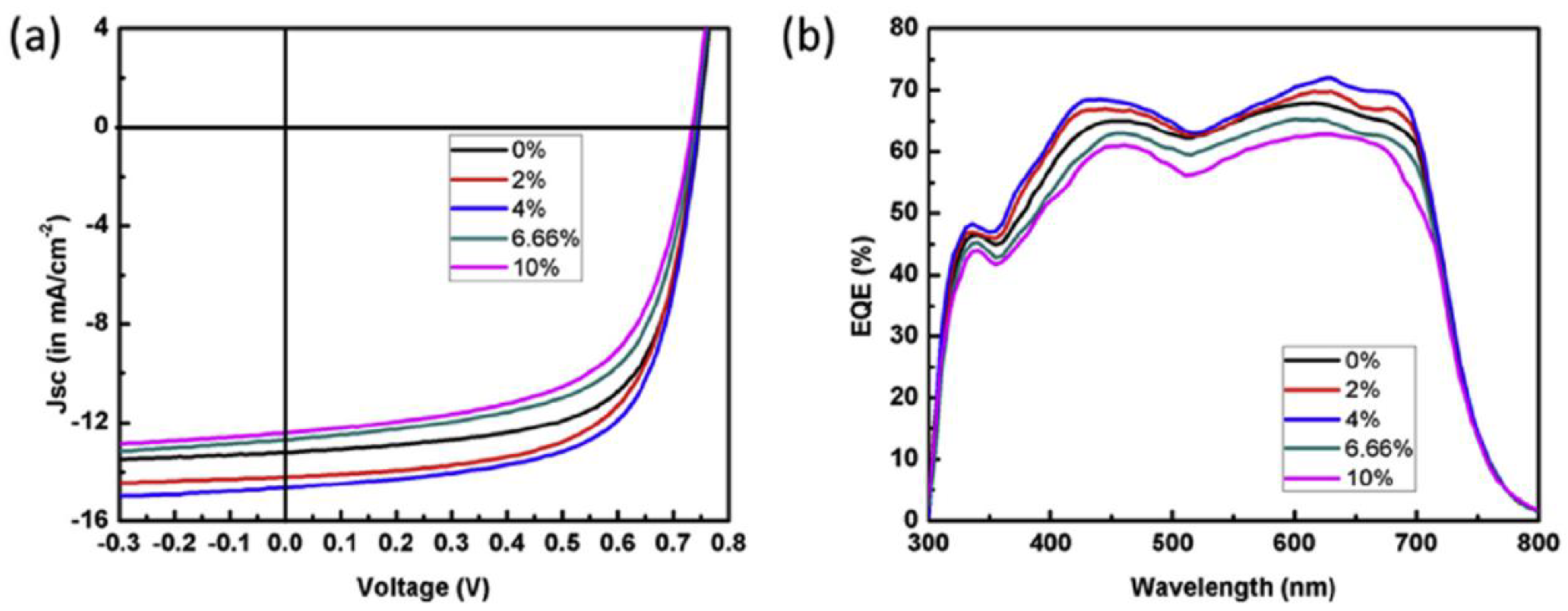
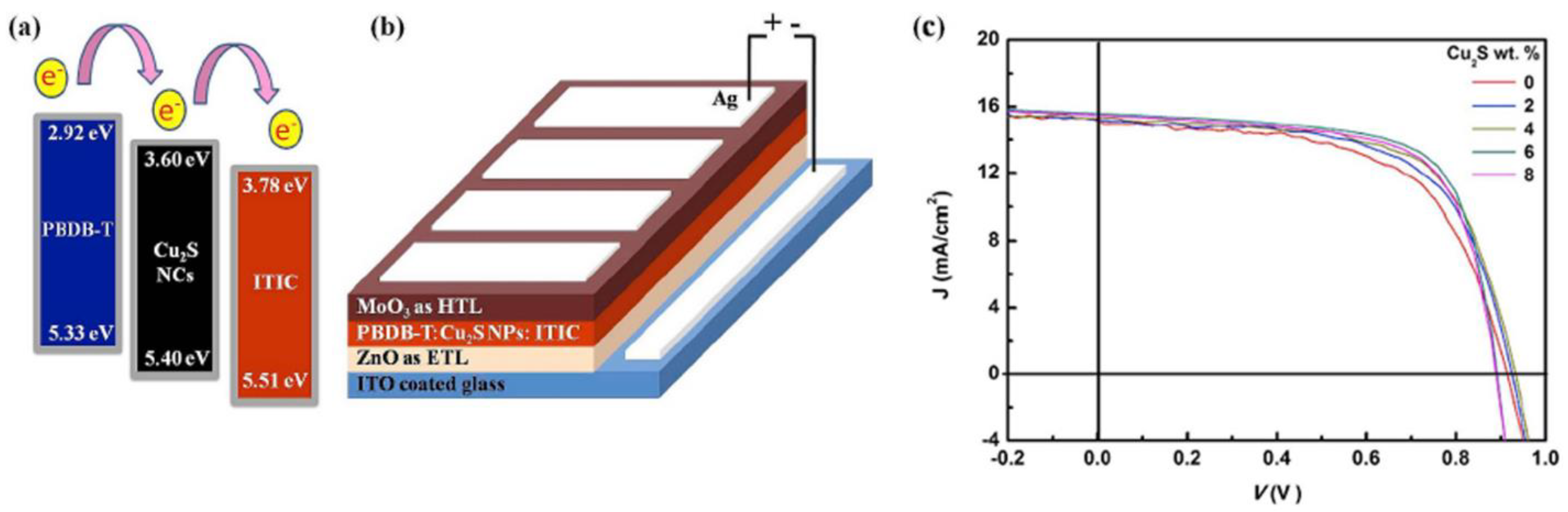
| ITO/ZnO/PTB7-Th:Cu2S:PCBM/MoO3/Ag | Inverted | AL | 8.2 | 6.96 | 2019 | [225] |
| ITO/ZnO/PBDB-T:Cu2S:ITIC/MoO3/Ag | Inverted | AL | 9.53 | 8.24 | 2019 | [226] |
| ITO/PEDOT:PSS/P3HT:PCBM:CuS/LiF/Al | Regular | AL | 5.04 | 2.98 | 2019 | [98] |
| Composites based on CuInS2 or PbSxSe1-x | ||||||
| ITO/PEDOT:PSS/P3HT:PCBM:CuInS2/Al | Regular | AL | 2.76 | 2.44 | 2011 | [227] |
| ITO/PEDOT:PSS/P3HT:CuInS2:graphene/LiF/Al | Regular | AL | 1.3 | - | 2014 | [228] |
| ITO/PEDOT:PSS/PDTPBT:PbSxSe1-x/PbSxSe1-x/LiF/Al | Regular | AL | 5.5 | - | 2013 | [229] |
| Composites based on Si | ||||||
| ITO/PEDOT:PSS/P3HT:PCBM:Si/Al | Regular | AL | 4.16 | - | 2011 | [232] |
| ITO/PEDOT:PSS/PTB7:Si/Al | Regular | AL | 0.03 | - | 2012 | [234] |
| ITO/PEDOT:PSS/MDMO-PPV:Si/Al | Regular | AL | 0.49 | - | 2012 | [236] |
| ITO/PEDOT:PSS/P3HT:PCBM:Si/Al | Regular | AL | 2.7 | 2.6 | 2012 | [237] |
| ITO/PEDOT:PSS/P3HT:PCBM:Si/Al | Regular | AL | 3.2 | 2.8 | 2012 | [101] |
| ITO/PEDOT:PSS/P3HT:Si/Al | Regular | AL | 0.15 | - | 2013 | [238] |
| ITO/PEDOT:PSS/MEH-PPV:PCBM:Si/Al | Regular | AL | 2.28 | 1.92 | 2013 | [239] |
| ITO/PEDOT:PSS/P3HT:PCBM:Si/Ca/Al | Regular | AL | 3.38 | 3.01 | 2013 | [240] |
| ITO/PEDOT:PSS/P3HT:Si/Al | Regular | AL | 0.08 | - | 2014 | [174] |
| ITO/PEDOT:PSS:Si/MEH-PPV:PCBM/Al | Regular | ABL | 2.46 | 2.24 | 2014 | [97] |
| ITO/PEDOT:PSS/MEH-PPV:PS-Si/Al | Regular | AL | 0.03073 | 0.00008 | 2015 | [241] |
| ITO/PEDOT:PSS/Si:PTB7/PC71BM/Al | Regular | AL | 2.46 | - | 2016 | [231] |
| FTO/PEDOT:PSS/P3HT:Si/Al | Regular | AL | 1.067 | 0.099 | 2018 | [242] |
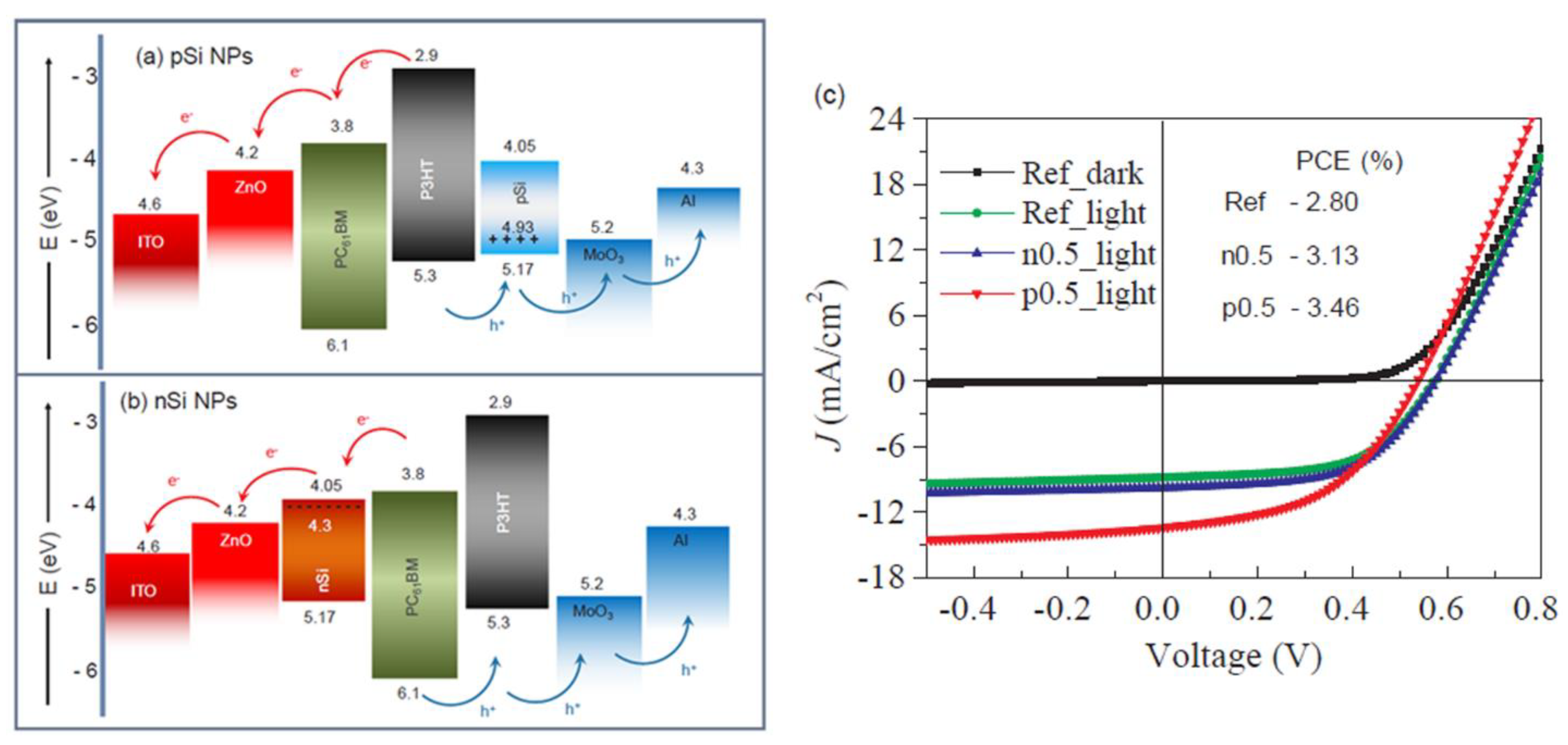
| ITO/ZnO/P3HT:PCBM:p-Si/MoO3/Al | Inverted | AL | 3.46 | 2.8 | 2018 | [104] |
| ITO/ZnO/P3HT:PCBM:n-Si/MoO3/Al | 3.13 | |||||
| ITO/PEDOT:PSS/P3HT:Si/Ca/Al | Regular | AL | 3.77 | - | 2019 | [243] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Socol, M.; Preda, N. Hybrid Nanocomposite Thin Films for Photovoltaic Applications: A Review. Nanomaterials 2021, 11, 1117. https://doi.org/10.3390/nano11051117
Socol M, Preda N. Hybrid Nanocomposite Thin Films for Photovoltaic Applications: A Review. Nanomaterials. 2021; 11(5):1117. https://doi.org/10.3390/nano11051117
Chicago/Turabian StyleSocol, Marcela, and Nicoleta Preda. 2021. "Hybrid Nanocomposite Thin Films for Photovoltaic Applications: A Review" Nanomaterials 11, no. 5: 1117. https://doi.org/10.3390/nano11051117
APA StyleSocol, M., & Preda, N. (2021). Hybrid Nanocomposite Thin Films for Photovoltaic Applications: A Review. Nanomaterials, 11(5), 1117. https://doi.org/10.3390/nano11051117






