A High-Throughput NanoBiT-Based Serological Assay Detects SARS-CoV-2 Seroconversion
Abstract
1. Introduction
2. Experimental Section
2.1. Cell Culture
2.2. Immunoblotting
2.3. Plasmid Design and Transfection
2.4. Temperature and pH Sensitivity Assay
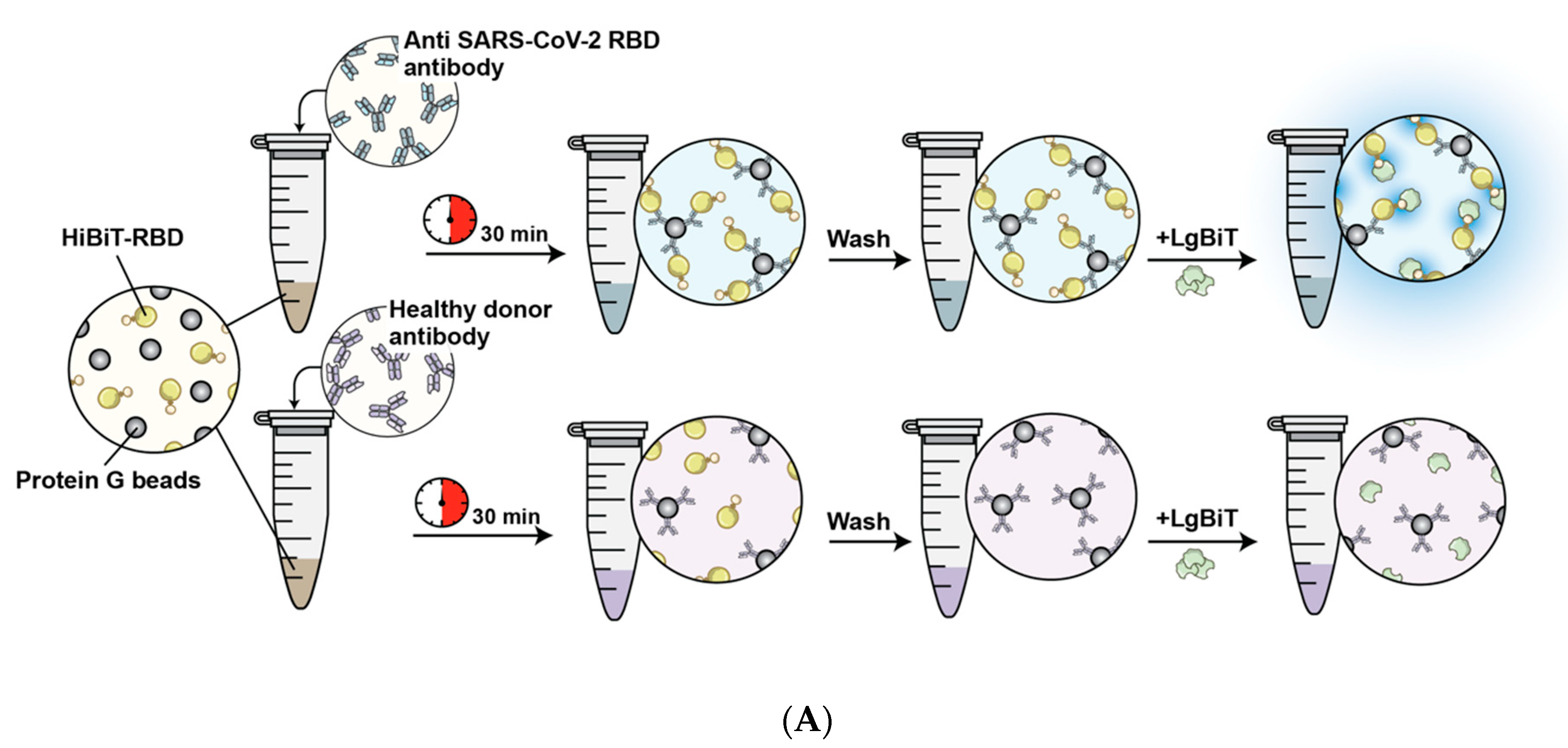
2.5. HiBiT-RBD Serological Assay
3. Results and Discussion
3.1. Establishment of a Reporter System to Detect Antibody-RBD Interaction
3.2. High Stability of the HiBiT-RBD System in a Wide Range of Temperatures and pH Levels
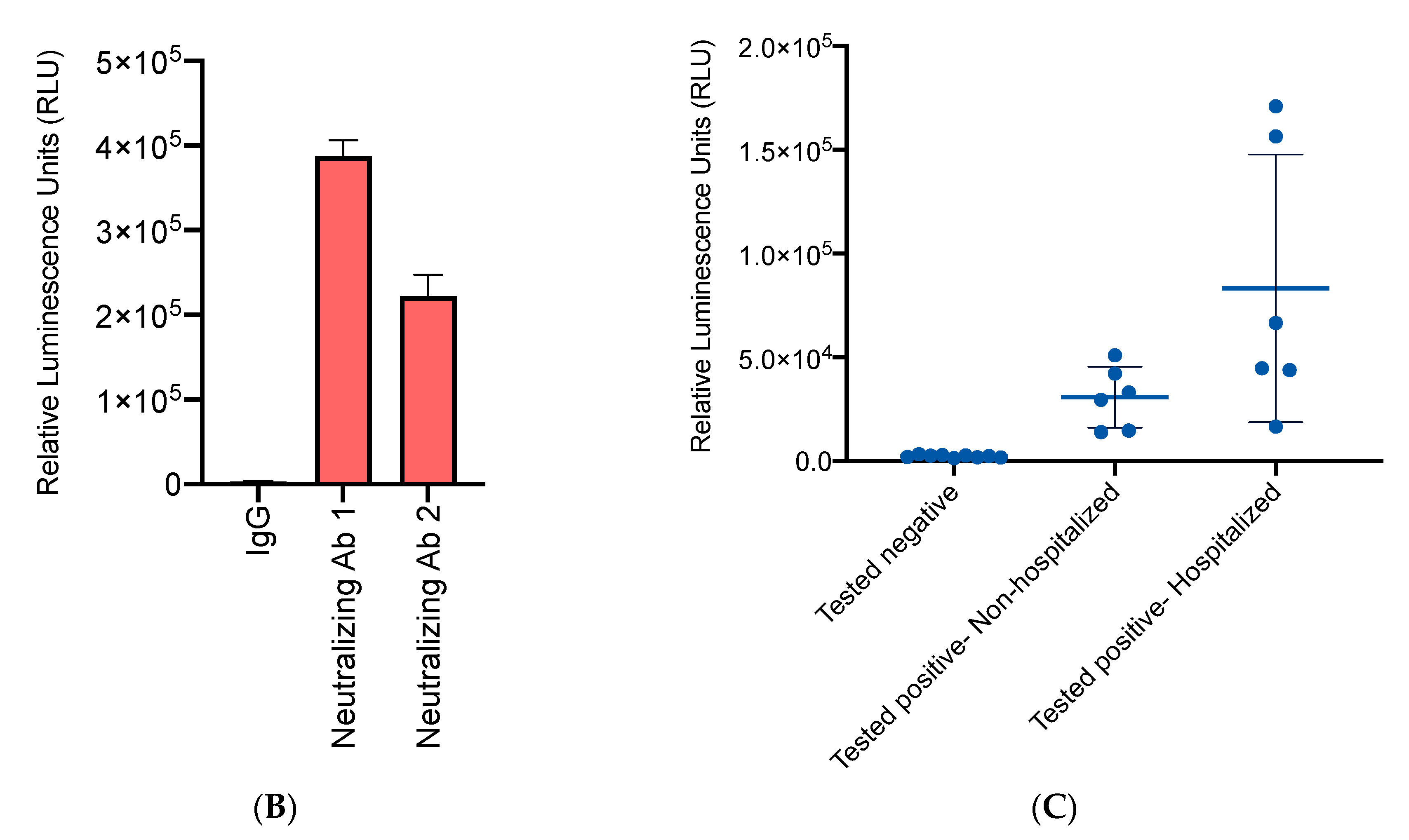
3.3. High Accuracy of the HiBiT-RBD Reporter for SARS-CoV-2 Neutralizing Antibodies with Considerable Affinity
3.4. High-Throughput NanoBiT Assay Detects Antibody Seroconversion in Recovered COVID-19 Individuals
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- COVID-19 Outbreak Live Update. Available online: https://www.worldometers.info/coronavirus/ (accessed on 14 March 2020).
- Corum, J.; Wee, S.; Zimmer, C. Coronavirus Vaccine Tracker. 2020. Available online: https://www.nytimes.com/interactive/2020/science/coronavirus-vaccine-tracker.html (accessed on 20 May 2020).
- Hueston, L.; Kok, J.; Guibone, A.; McDonald, D.; Hone, G.; Goodwin, J.; Carter, I.; Basile, K.; Sandaradura, I.; Maddocks, S.; et al. The Antibody Response to SARS-CoV-2 Infection. Open Forum Infect. Dis. 2020, 7. [Google Scholar] [CrossRef] [PubMed]
- Bastos, M.L.; Tavaziva, G.; Abidi, S.K.; Campbell, J.R.; Haraoui, L.-P.; Johnston, J.C.; Lan, Z.; Law, S.; MacLean, E.; Trajman, A.; et al. Diagnostic accuracy of serological tests for covid-19: Systematic review and meta-analysis. BMJ 2020, 370, m2516. [Google Scholar] [CrossRef]
- Lan, J.; Ge, J.; Yu, J.; Shan, S.; Zhou, H.; Fan, S.; Zhang, Q.; Shi, X.; Wang, Q.; Zhang, L.; et al. Structure of the SARS-CoV-2 spike receptor-binding domain bound to the ACE2 receptor. Nature 2020, 581, 215–220. [Google Scholar] [CrossRef]
- Azad, T.; Singaravelu, R.; Crupi, M.J.; Jamieson, T.; Dave, J.; Brown, E.E.; Rezaei, R.; Taha, Z.; Boulton, S.; Martin, N.T.; et al. Implications for SARS-CoV-2 Vaccine Design: Fusion of Spike Glycoprotein Transmembrane Domain to Receptor-Binding Domain Induces Trimerization. Membranes 2020, 10, 215. [Google Scholar] [CrossRef] [PubMed]
- Premkumar, L.; Segovia-Chumbez, B.; Jadi, R.; Martinez, D.R.; Raut, R.; Markmann, A.J.; Cornaby, C.; Bartelt, L.; Weiss, S.; Park, Y.; et al. The receptor binding domain of the viral spike protein is an immunodominant and highly specific target of antibodies in SARS-CoV-2 patients. Sci. Immunol. 2020, 5, eabc8413. [Google Scholar] [CrossRef] [PubMed]
- Piccoli, L.; Park, Y.-J.; Tortorici, M.A.; Czudnochowski, N.; Walls, A.C.; Beltramello, M.; Silacci-Fregni, C.; Pinto, D.; Rosen, L.E.; Bowen, J.E.; et al. Mapping Neutralizing and Immunodominant Sites on the SARS-CoV-2 Spike Receptor-Binding Domain by Structure-Guided High-Resolution Serology. Cell 2020, 183, 1024–1042.e21. [Google Scholar] [CrossRef]
- Fleiss, A.; Sarkisyan, K.S. A brief review of bioluminescent systems (2019). Curr. Genet. 2019, 65, 877–882. [Google Scholar] [CrossRef]
- Bioluminescent Reporters. Reporter Gene Applications An Introduction to Reporter Genes. Available online: https://www.promega.ca/resources/guides/cell-biology/bioluminescent-reporters/#references-6d127eb8-eeae-40b7-86e9-fe300545e8fa (accessed on 13 December 2020).
- Boute, N.; Lowe, P.; Berger, S.; Malissard, M.; Robert, A.; Tesar, M. NanoLuc Luciferase—A Multifunctional Tool for High Throughput Antibody Screening. Front. Pharmacol. 2016, 7, 7. [Google Scholar] [CrossRef]
- Dixon, A.S.; Schwinn, M.K.; Hall, M.P.; Zimmerman, K.; Otto, P.; Lubben, T.H.; Butler, B.L.; Binkowski, B.F.; Machleidt, T.; Kirkland, T.A.; et al. NanoLuc Complementation Reporter Optimized for Accurate Measurement of Protein Interactions in Cells. ACS Chem. Biol. 2016, 11, 400–408. [Google Scholar] [CrossRef] [PubMed]
- Azad, T.; Tashakor, A.; Hosseinkhani, S. Split-luciferase complementary assay: Applications, recent developments, and future perspectives. Anal. Bioanal. Chem. 2014, 406, 5541–5560. [Google Scholar] [CrossRef] [PubMed]
- Azad, T.; Van Rensburg, H.J.J.; Lightbody, E.D.; Neveu, B.; Champagne, A.; Ghaffari, A.; Kay, V.R.; Hao, Y.; Shen, H.; Yeung, B.; et al. A LATS biosensor screen identifies VEGFR as a regulator of the Hippo pathway in angiogenesis. Nat. Commun. 2018, 9, 1–15. [Google Scholar] [CrossRef] [PubMed]
- Nouri, K.; Azad, T.; Lightbody, E.; Khanal, P.; Nicol, C.J.; Yang, X. A kinome-wide screen using a NanoLuc LATS luminescent biosensor identifies ALK as a novel regulator of the Hippo pathway in tumorigenesis and immune evasion. FASEB J. 2019, 33, 12487–12499. [Google Scholar] [CrossRef] [PubMed]
- Nouri, K.; Azad, T.; Ling, M.; Van Rensburg, H.J.J.; Pipchuk, A.; Shen, H.; Hao, Y.; Zhang, J.; Yang, X. Identification of Celastrol as a Novel YAP-TEAD Inhibitor for Cancer Therapy by High Throughput Screening with Ultrasensitive YAP/TAZ–TEAD Biosensors. Cancers 2019, 11, 1596. [Google Scholar] [CrossRef] [PubMed]
- Azad, T.; Nouri, K.; Van Rensburg, H.J.J.; Maritan, S.M.; Wu, L.; Hao, Y.; Montminy, T.; Yu, J.; Khanal, P.; Mulligan, L.M.; et al. A gain-of-functional screen identifies the Hippo pathway as a central mediator of receptor tyrosine kinases during tumorigenesis. Oncogene 2020, 39, 334–355. [Google Scholar] [CrossRef] [PubMed]
- Azad, T.; Tashakor, A.; Rahmati, F.; Hemmati, R.; Hosseinkhani, S. Oscillation of apoptosome formation through assembly of truncated Apaf-1. Eur. J. Pharmacol. 2015, 760, 64–71. [Google Scholar] [CrossRef] [PubMed]
- Peng, H.-P.; Lee, K.H.; Jian, J.-W.; Yang, A.-S. Origins of specificity and affinity in antibody–protein interactions. Proc. Natl. Acad. Sci. USA 2014, 111, E2656–E2665. [Google Scholar] [CrossRef] [PubMed]
- Pan, R.; Qin, Y.; Banasik, M.; Lees, W.; Shepherd, A.J.; Cho, M.W.; Kong, X.-P. Increased Epitope Complexity Correlated with Antibody Affinity Maturation and a Novel Binding Mode Revealed by Structures of Rabbit Antibodies against the Third Variable Loop (V3) of HIV-1 gp120. J. Virol. 2018, 92, e01894-17. [Google Scholar] [CrossRef] [PubMed]
- Brown, E.; Rezaei, R.; Jamieson, T.; Dave, J.; Martin, N.; Singaravelu, R.; Crupi, M.; Boulton, S.; Tucker, S.; Duong, J.; et al. Characterization of Critical Determinants of ACE2–SARS CoV-2 RBD Interaction. Int. J. Mol. Sci. 2021, 22, 2268. [Google Scholar] [CrossRef] [PubMed]
- Azad, T.; Singaravelu, R.; Taha, Z.; Jamieson, T.R.; Boulton, S.; Crupi, M.J.; Martin, N.T.; Brown, E.E.; Poutou, J.; Ghahremani, M.; et al. Nanoluciferase complementation-based bioreporter reveals the importance of N-linked glycosylation of SARS-CoV-2 S for viral entry. Mol. Ther. 2021. [Google Scholar] [CrossRef] [PubMed]
- Azad, T.; Singaravelu, R.; Brown, E.E.; Taha, Z.; Rezaei, R.; Arulanandam, R.; Boulton, S.; Diallo, J.-S.; Ilkow, C.S.; Bell, J.C. SARS-CoV-2 S1 NanoBiT: A nanoluciferase complementation-based biosensor to rapidly probe SARS-CoV-2 receptor recognition. Biosens. Bioelectron. 2021, 180, 113122. [Google Scholar] [CrossRef] [PubMed]




Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Azad, T.; Rezaei, R.; Singaravelu, R.; Jamieson, T.R.; Crupi, M.J.F.; Surendran, A.; Poutou, J.; Taklifi, P.; Cowan, J.; Cameron, D.W.; et al. A High-Throughput NanoBiT-Based Serological Assay Detects SARS-CoV-2 Seroconversion. Nanomaterials 2021, 11, 807. https://doi.org/10.3390/nano11030807
Azad T, Rezaei R, Singaravelu R, Jamieson TR, Crupi MJF, Surendran A, Poutou J, Taklifi P, Cowan J, Cameron DW, et al. A High-Throughput NanoBiT-Based Serological Assay Detects SARS-CoV-2 Seroconversion. Nanomaterials. 2021; 11(3):807. https://doi.org/10.3390/nano11030807
Chicago/Turabian StyleAzad, Taha, Reza Rezaei, Ragunath Singaravelu, Taylor R. Jamieson, Mathieu J. F. Crupi, Abera Surendran, Joanna Poutou, Parisa Taklifi, Juthaporn Cowan, Donald William Cameron, and et al. 2021. "A High-Throughput NanoBiT-Based Serological Assay Detects SARS-CoV-2 Seroconversion" Nanomaterials 11, no. 3: 807. https://doi.org/10.3390/nano11030807
APA StyleAzad, T., Rezaei, R., Singaravelu, R., Jamieson, T. R., Crupi, M. J. F., Surendran, A., Poutou, J., Taklifi, P., Cowan, J., Cameron, D. W., & Ilkow, C. S. (2021). A High-Throughput NanoBiT-Based Serological Assay Detects SARS-CoV-2 Seroconversion. Nanomaterials, 11(3), 807. https://doi.org/10.3390/nano11030807






