Experimental Investigation of Polymer-Coated Silica Nanoparticles for EOR under Harsh Reservoir Conditions of High Temperature and Salinity
Abstract
1. Introduction
2. Experimental Materials and Methods
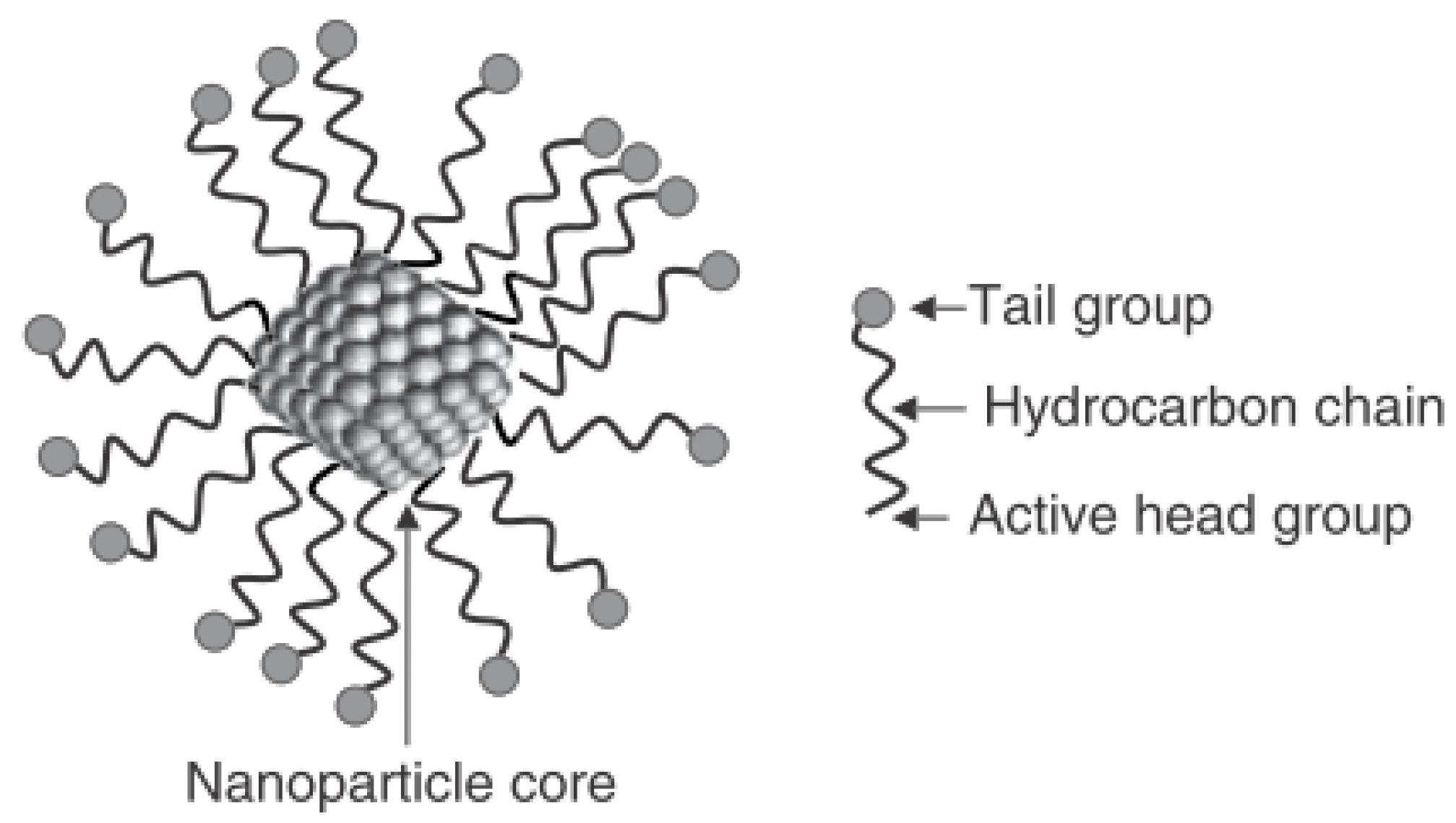
2.1. Nanoparticles and “Simulated” Seawater
2.2. Non-Wetting Phase
2.3. Preparation of Porous Medium
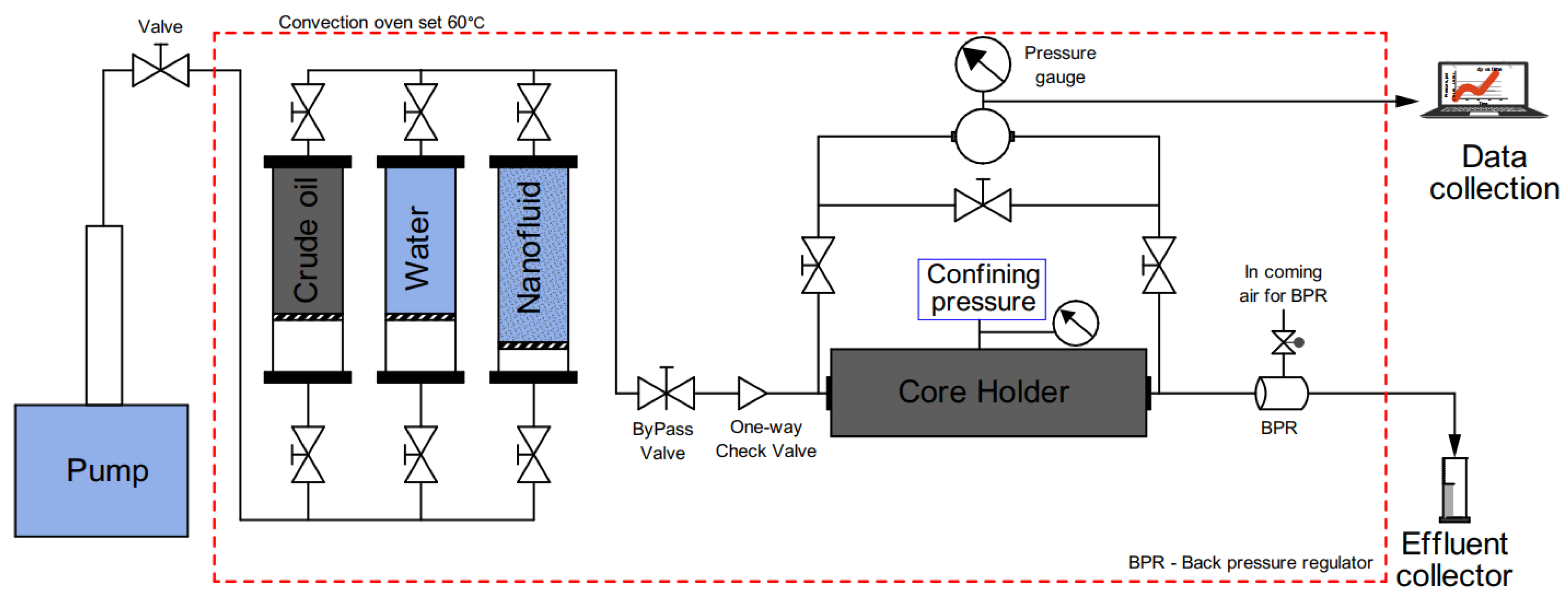
2.4. Displacement Tests
2.5. Interfacial Tension Measurement
2.6. Spontaneous Imbibition Tests
3. Results and Discussion
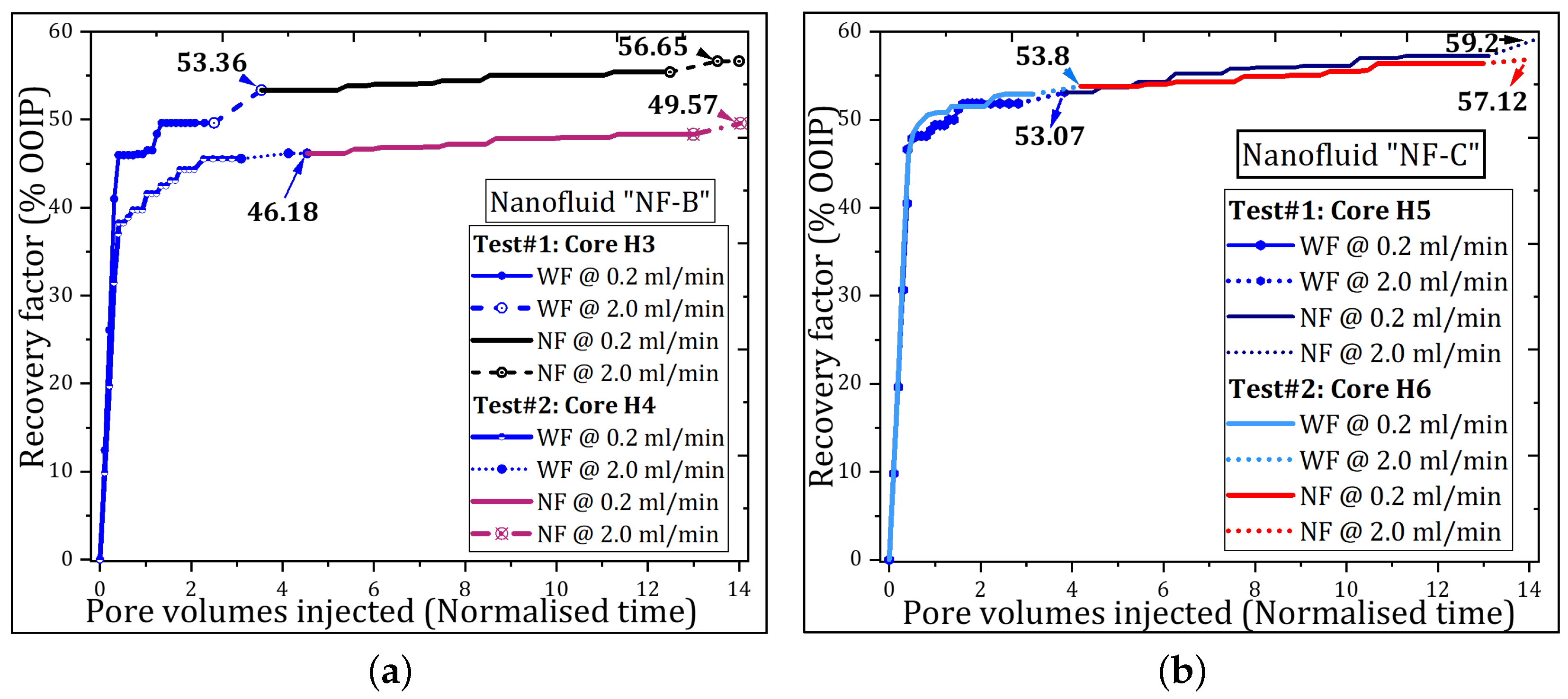
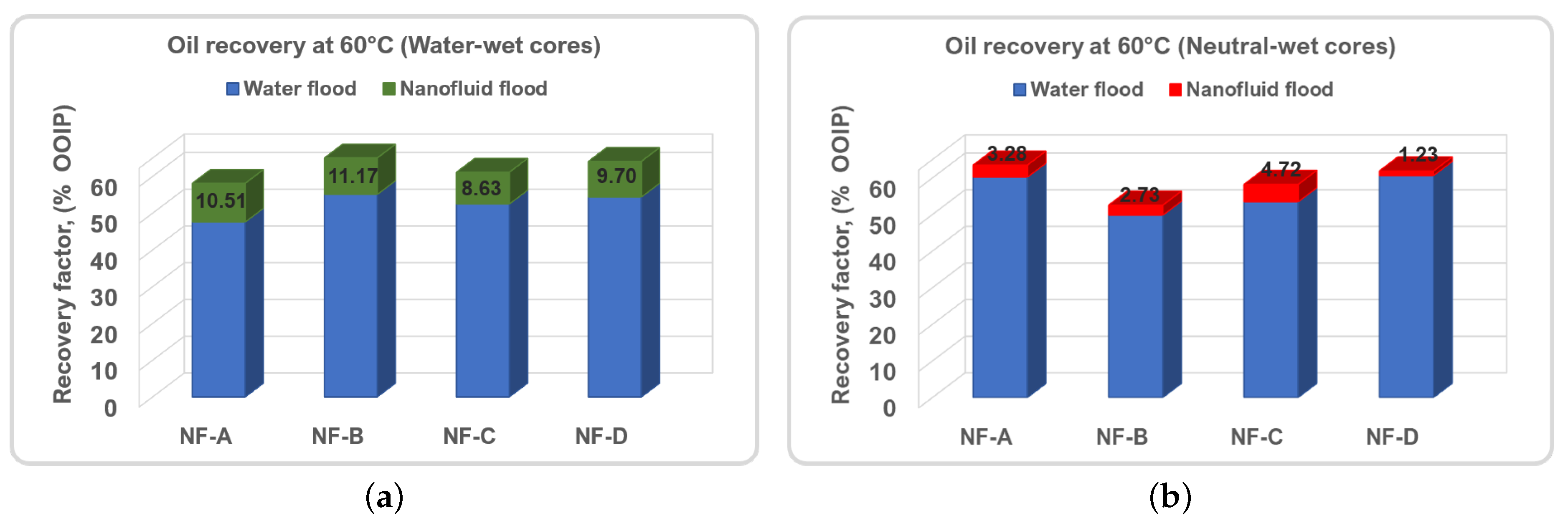
3.1. Nanoparticle’s Oil Recovery
3.2. Mechanisms behind EOR by Nanoparticles
3.2.1. Effect of Nanoparticles on Viscosity of Injection Water
3.2.2. Reduction in Interfacial Tension (IFT)
3.2.3. Formation of Emulsions Stabilized by Nanoparticles
3.2.4. Change in Rock Wettability and Surface Roughness
3.2.5. Nanofluid Displacement Pressure
4. Conclusions
- Polymer-coated silica nanoparticles have shown a strong ability to increase oil recovery after water flooding. The increment recovery was up to 6% of OOIP;
- The nanoparticles can reduce the oil/water interfacial tension at a concentration as low as 0.1 wt.%. The smallest nanoparticle size were more efficient in reducing the tension due to the large contact area;
- The flooding experiments indicated that oil was produced as oil-in-water emulsion droplets; these emulsion droplets were stabilized by the nanoparticles.
- The adsorption of nanoparticles in oil-wet pores can reverse the negative capillary pressure to positive values and change the wettability to water-wet condition;
- The size of nanoparticles and the formation of large aggregates within the pores were observed to increase displacement pressure, resulting in poor oil recovery efficiency;
- Different oil displacement mechanisms, such as reduced IFT, change in wettability, generation of in-situ emulsions and change log-jamming effect can explain the oil recovery phenomenon of polymer-coated silica NPs in intermediate reservoirs. However, the wettability alteration to a more water-wet seemed to govern the oil displacement process.
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Sydansk, R.D.; Romero-Zeron, L. Reservoir Conformance Improvement; Society of Petroleum Engineers: Richardson, TX, USA, 2011. [Google Scholar]
- Fletcher, A.; Davis, J. How EOR can be transformed by nanotechnology. In Proceedings of the SPE Improved Oil Recovery Symposium, Tulsa, OK, USA, 24–28 April 2010. [Google Scholar]
- Bera, A.; Belhaj, H. Application of nanotechnology by means of nanoparticles and nanodispersions in oil recovery—A comprehensive review. J. Nat. Gas Sci. Eng. 2016, 34, 1284–1309. [Google Scholar] [CrossRef]
- Das, S.K.; Choi, S.U.; Patel, H.E. Heat transfer in nanofluids—A review. Heat Transf. Eng. 2006, 27, 3–19. [Google Scholar] [CrossRef]
- Peng, B.; Zhang, L.; Luo, J.; Wang, P.; Ding, B.; Zeng, M.; Cheng, Z. A review of nanomaterials for nanofluid enhanced oil recovery. RSC Adv. 2017, 7, 32246–32254. [Google Scholar] [CrossRef]
- Sandeep, R.; Jain, S.; Agrawal, A. Application of Nanoparticles-Based Technologies in the Oil and Gas Industry. In Nanotechnology for Energy and Environmental Engineering; Springer: Berlin/Heidelberg, Germany, 2020; pp. 257–277. [Google Scholar]
- Yu, H.; Kotsmar, C.; Yoon, K.Y.; Ingram, D.R.; Johnston, K.P.; Bryant, S.L.; Huh, C. Transport and retention of aqueous dispersions of paramagnetic nanoparticles in reservoir rocks. In Proceedings of the SPE Improved Oil Recovery Symposium, Tulsa, OK, USA, 24–28 April 2010. [Google Scholar]
- ShamsiJazeyi, H.; Miller, C.A.; Wong, M.S.; Tour, J.M.; Verduzco, R. Polymer-coated nanoparticles for enhanced oil recovery. J. Appl. Polym. Sci. 2014, 131. [Google Scholar] [CrossRef]
- Gbadamosi, A.O.; Junin, R.; Manan, M.A.; Yekeen, N.; Agi, A.; Oseh, J.O. Recent advances and prospects in polymeric nanofluids application for enhanced oil recovery. J. Ind. Eng. Chem. 2018, 66, 1–19. [Google Scholar] [CrossRef]
- Rodriguez, E.; Roberts, M.; Yu, H.; Huh, C.; Bryant, S.L. Enhanced migration of surface-treated nanoparticles in sedimentary rocks. In Proceedings of the SPE Annual Technical Conference and Exhibition, New Orleans, LA, USA, 4–7 October 2009. [Google Scholar]
- Zhang, T.; Murphy, M.J.; Yu, H.; Bagaria, H.G.; Yoon, K.Y.; Nielson, B.M.; Bielawski, C.W.; Johnston, K.P.; Huh, C.; Bryant, S.L. Investigation of nanoparticle adsorption during transport in porous media. SPE J. 2015, 20, 667–677. [Google Scholar] [CrossRef]
- Ponnapati, R.; Karazincir, O.; Dao, E.; Ng, R.; Mohanty, K.; Krishnamoorti, R. Polymer-functionalized nanoparticles for improving waterflood sweep efficiency: Characterization and transport properties. Ind. Eng. Chem. Res. 2011, 50, 13030–13036. [Google Scholar] [CrossRef]
- Behzadi, A.; Mohammadi, A. Environmentally responsive surface-modified silica nanoparticles for enhanced oil recovery. J. Nanopart. Res. 2016, 18, 266. [Google Scholar] [CrossRef]
- Choi, S.K.; Son, H.A.; Kim, H.T.; Kim, J.W. Nanofluid Enhanced Oil Recovery Using Hydrophobically Associative Zwitterionic Polymer-Coated Silica Nanoparticles. Energy Fuels 2017, 31, 7777–7782. [Google Scholar] [CrossRef]
- Bila, A.; Stensen, J.Å.; Torsæter, O. Experimental Investigation of Polymer-Coated Silica Nanoparticles for Enhanced Oil Recovery. Nanomaterials 2019, 9, 822. [Google Scholar] [CrossRef] [PubMed]
- Bila, A.; Åge Stensen, J.; Torsæter, O. Polymer-functionalized silica nanoparticles for improving water flood sweep efficiency in Berea sandstones. E3S Web Conf. 2020, 146, 02001. [Google Scholar] [CrossRef]
- Bila, A.; Torsæter, O. Enhancing oil recovery with hydrophilic polymer-coated silica nanoparticles. Energies 2020, 13, 5720. [Google Scholar] [CrossRef]
- Roustaei, A.; Moghadasi, J.; Bagherzadeh, H.; Shahrabadi, A. An experimental investigation of polysilicon nanoparticles’ recovery efficiencies through changes in interfacial tension and wettability alteration. In Proceedings of the SPE International Oilfield Nanotechnology Conference and Exhibition, Noordwijk, The Netherlands, 12–14 June 2012. [Google Scholar]
- Roustaei, A.; Saffarzadeh, S.; Mohammadi, M. An evaluation of modified silica nanoparticles’ efficiency in enhancing oil recovery of light and intermediate oil reservoirs. Egypt. J. Pet. 2013, 22, 427–433. [Google Scholar] [CrossRef]
- Ding, H.; Zhang, N.; Zhang, Y.; Wei, M.; Bai, B. Experimental Data Analysis of Nanoparticles for Enhanced Oil Recovery. Ind. Eng. Chem. Res. 2019, 58, 12438–12450. [Google Scholar] [CrossRef]
- Aziz, H.; Tunio, S.Q. Enhancing oil recovery using nanoparticles—A review. Adv. Nat. Sci. Nanosci. Nanotechnol. 2019, 10, 033001. [Google Scholar] [CrossRef]
- Karimi, A.; Fakhroueian, Z.; Bahramian, A.; Pour Khiabani, N.; Darabad, J.B.; Azin, R.; Arya, S. Wettability Alteration in Carbonates using Zirconium Oxide Nanofluids: EOR Implications. Energy Fuels 2012, 26, 1028–1036. [Google Scholar] [CrossRef]
- Dai, C.; Wang, X.; Li, Y.; Lv, W.; Zou, C.; Gao, M.; Zhao, M. Spontaneous Imbibition Investigation of Self-Dispersing Silica Nanofluids for Enhanced Oil Recovery in Low-Permeability Cores. Energy Fuels 2017, 31, 2663–2668. [Google Scholar] [CrossRef]
- Wasan, D.; Nikolov, A. Spreading of Nanofluids on Solids. Nature 2003, 423, 156–159. [Google Scholar] [CrossRef]
- Afolabi, R.O.; Yusuf, E.O. Nanotechnology and global energy demand: Challenges and prospects for a paradigm shift in the oil and gas industry. J. Pet. Explor. Prod. Technol. 2019, 9, 1423–1441. [Google Scholar] [CrossRef]
- Yakasai, F.; Jaafar, M.Z.; Bandyopadhyay, S.; Agi, A. Current Developments and Future Outlook in Nanofluid Flooding: A Comprehensive Review of Various Parameters Influencing Oil Recovery Mechanisms. J. Ind. Eng. Chem. 2020, 93, 138–162. [Google Scholar] [CrossRef]
- Kumar, N.; Gaur, T.; Mandal, A. Characterization of SPN Pickering emulsions for application in enhanced oil recovery. J. Ind. Eng. Chem. 2017, 54, 304–315. [Google Scholar] [CrossRef]
- Skauge, T.; Spildo, K.; Skauge, A. Nano-sized particles for EOR. In Proceedings of the SPE Improved Oil Recovery Symposium, Tulsa, OK, USA, 24–28 April 2010. [Google Scholar]
- Kokubun, M.E.; Radu, F.A.; Keilegavlen, E.; Kumar, K.; Spildo, K. Transport of Polymer Particles in Oil–Water Flow in Porous Media: Enhancing Oil Recovery. Transp. Porous Media 2019, 126, 501–519. [Google Scholar] [CrossRef]
- Mohammed, M.; Babadagli, T. Wettability alteration: A comprehensive review of materials/methods and testing the selected ones on heavy-oil containing oil-wet systems. Adv. Colloid Interface Sci. 2015, 220, 54–77. [Google Scholar] [CrossRef] [PubMed]
- Miranda, C.R.; Lara, L.S.d.; Tonetto, B.C. Stability and mobility of functionalized silica nanoparticles for enhanced oil recovery applications. In Proceedings of the SPE International Oilfield Nanotechnology Conference and Exhibition, Noordwijk, The Netherlands, 12–14 June 2012. [Google Scholar] [CrossRef]
- Khalil, M.; Jan, B.M.; Tong, C.W.; Berawi, M.A. Advanced nanomaterials in oil and gas industry: Design, application and challenges. Appl. Energy 2017, 191, 287–310. [Google Scholar] [CrossRef]
- Anderson, W.G. Wettability Literature Survey Part 2: Wettability Measurement. J. Pet. Technol. 1986, 38, 1246–1262. [Google Scholar] [CrossRef]
- Ziauddin, M.; Montaron, B.; Hussain, H.; Habashy, T.; Seleznev, N.; Signer, C.; Abdallah, W. Fundamentals of wettability. Schlumberger Oilfield Rev. 2007, 19, 40–67. [Google Scholar]
- Eide, Ø.; Fernø, M.; Nybø, S.; Graue, A. Waterflood Stability in SCAL Analysis. In Proceedings of the International Symposium of the Society of Core Analysts, Avignon, France, 8–11 September 2014. [Google Scholar]
- Bila, A.L. Experimental Investigation of Surface-Functionalised Silica Nanoparticles for Enhanced Oil Recovery. Ph.D. Thesis, Norwegian University of Science and Technology (NTNU), Trondheim, Norway, 2020. [Google Scholar]
- Metin, C.; Bonnecaze, R.; Nguyen, Q. The viscosity of silica nanoparticle dispersions in permeable media. SPE Reserv. Eval. Eng. 2013, 16, 327–332. [Google Scholar] [CrossRef]
- Behera, U.S.; Sangwai, J.S. Interaction of Nanoparticles with Reservoir Fluids and Rocks for Enhanced Oil Recovery. In Nanotechnology for Energy and Environmental Engineering; Springer: Berlin/Heidelberg, Germany, 2020; pp. 299–328. [Google Scholar]
- Sheng, J.J. Status of surfactant EOR technology. Petroleum 2015, 1, 97–105. [Google Scholar] [CrossRef]
- Chatzis, I.; Kuntamukkula, M.; Morrow, N. Effect of capillary number on the microstructure of residual oil in strongly water-wet sandstones. SPE Reserv. Eng. 1988, 3, 902–912. [Google Scholar] [CrossRef]
- Kim, I.; Worthen, A.J.; Johnston, K.P.; DiCarlo, D.A.; Huh, C. Size-dependent properties of silica nanoparticles for Pickering stabilization of emulsions and foams. J. Nanoparticle Res. 2016, 18, 82. [Google Scholar] [CrossRef]
- Arab, D.; Kantzas, A.; Bryant, S.L. Nanoparticle stabilized oil in water emulsions: A critical review. J. Pet. Sci. Eng. 2018, 163, 217–242. [Google Scholar] [CrossRef]
- Suleimanov, B.A.; Ismailov, F.; Veliyev, E. Nanofluid for enhanced oil recovery. J. Pet. Sci. Eng. 2011, 78, 431–437. [Google Scholar] [CrossRef]
- Binks, B.P.; Whitby, C.P. Nanoparticle silica-stabilised oil-in-water emulsions: Improving emulsion stability. Colloids Surfaces Physicochem. Eng. Asp. 2005, 253, 105–115. [Google Scholar] [CrossRef]
- Hu, Z.; Azmi, S.M.; Raza, G.; Glover, P.W.J.; Wen, D. Nanoparticle-Assisted Water-Flooding in Berea Sandstones. Energy Fuels 2016, 30, 2791–2804. [Google Scholar] [CrossRef]
- Aurand, K.; Dahle, S.; Torsæter, O. Comparison of oil recovery for six nanofluids in Berea sandstone cores. In Proceedings of the International Symposium of the Society of Core Analysts, Avignon, France, 8–11 September 2014; pp. 1–12. [Google Scholar]


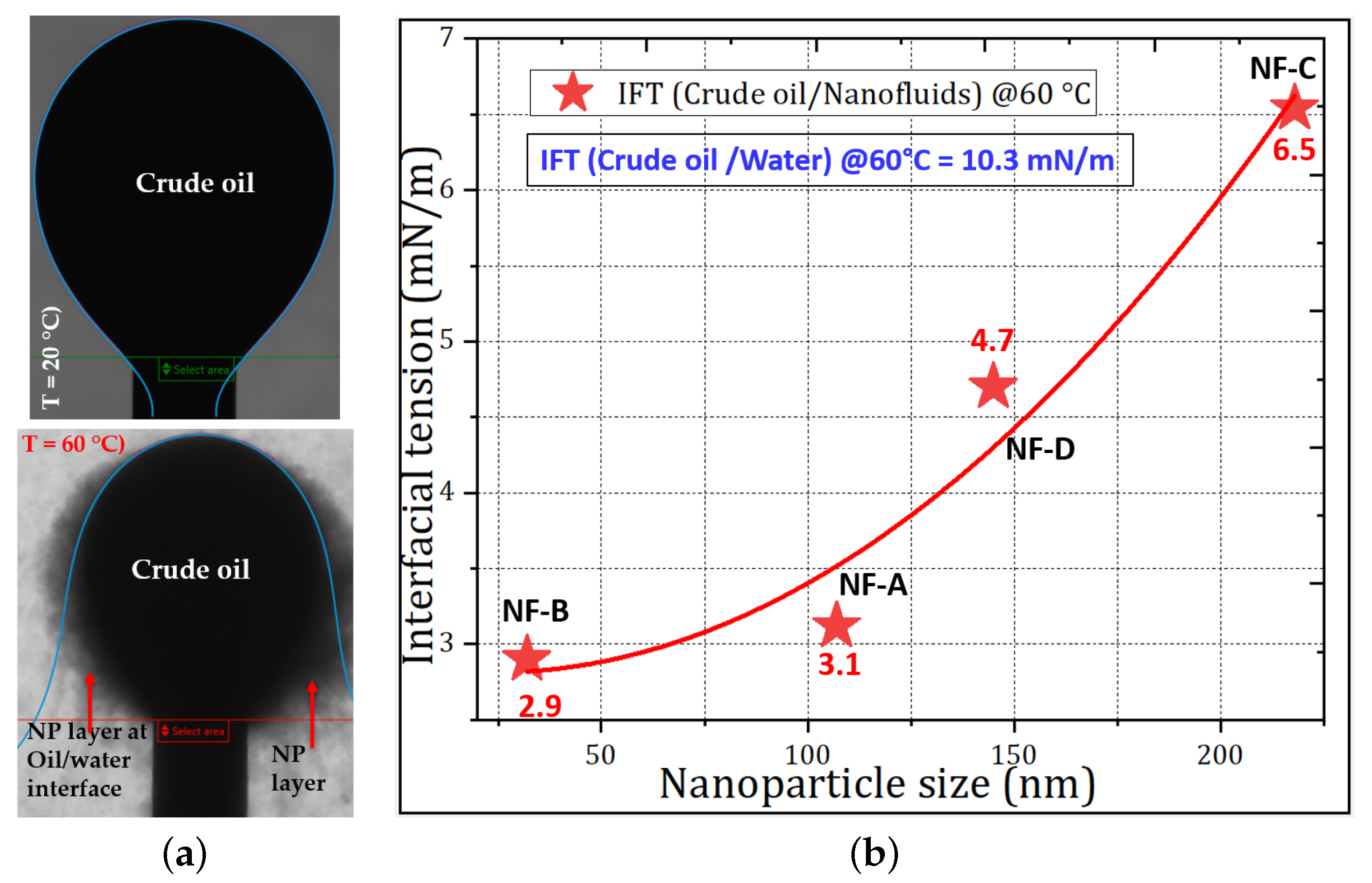

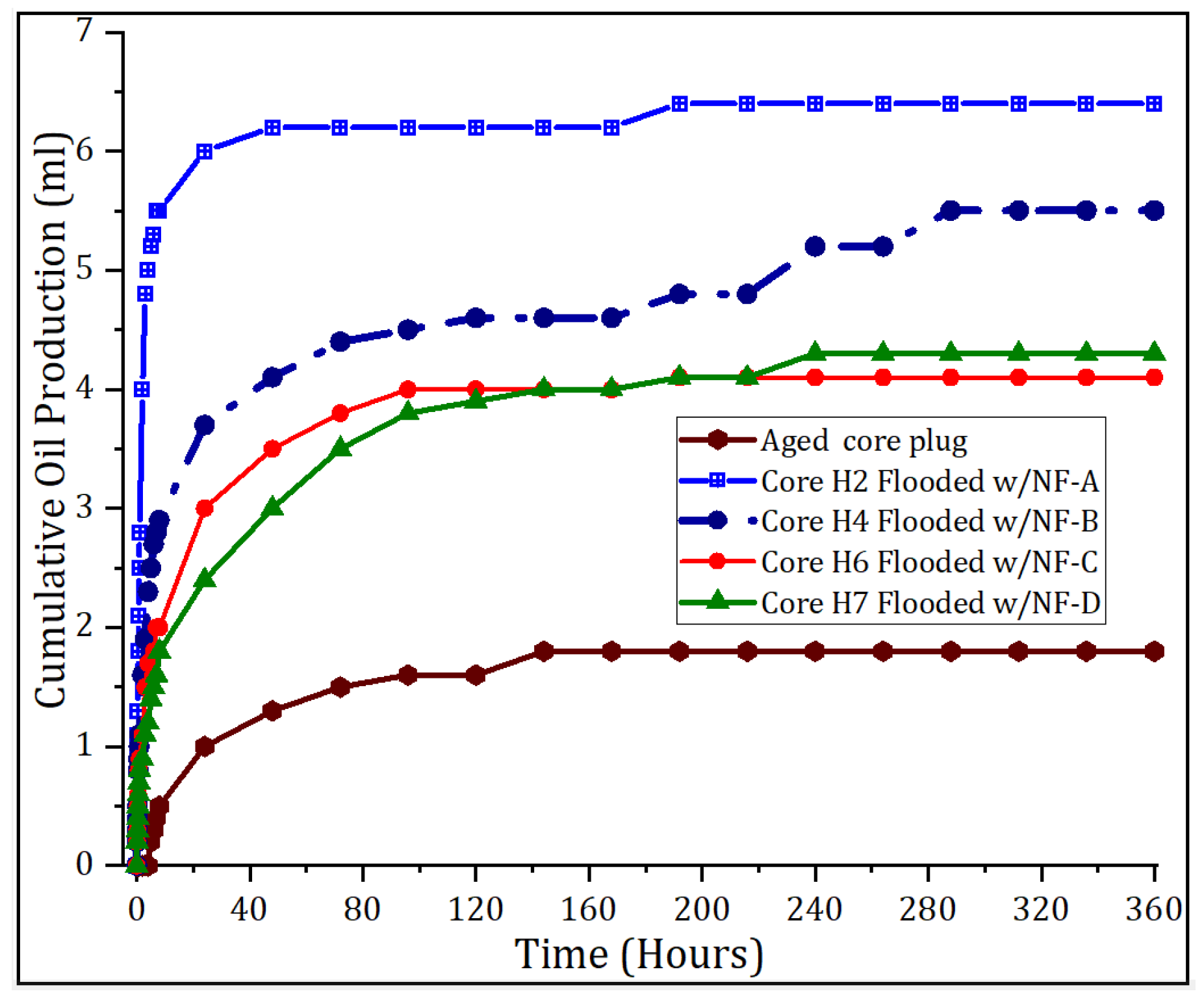
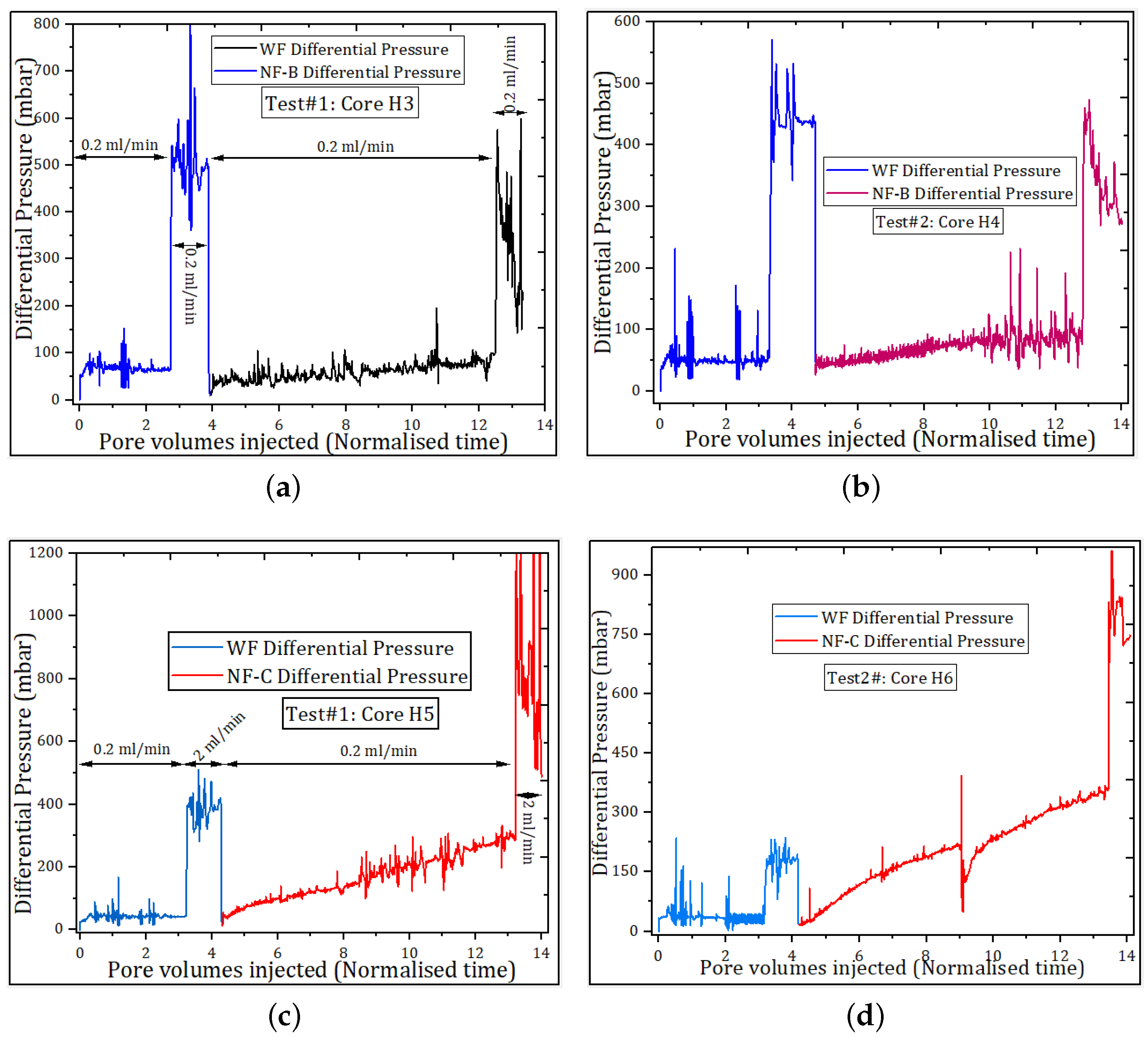
| Sample | Basis | Modification | Conc., wt.% | Size (nm) a | Size (nm) b |
|---|---|---|---|---|---|
| NF-A | SiO (sol-gel-cationic) | Polymer | 38.6 | 107 | 63 |
| NF-B | SiO (sol-gel-anionic) | Polymer | 26.0 | 32 | 38 |
| NF-C | SiO/AlO/MOX | Polymer | 21.6 | 218 | 155 |
| NF-D | SiO/AlO/MOX | Polymer | 25.5 | 145 | 135 |
| Fluid | Density (g/cm) | Viscosity (cP) | pH |
|---|---|---|---|
| Seawater | 1.008 | 0.53 | 7.97 |
| Nanofluids | 1.007–1.009 | 0.51–0.67 | 7.74–8.05 |
| Property | Value | Unit |
|---|---|---|
| Saturates | 71.57 | wt.% |
| Aromates | 20.81 | wt.% |
| Resins | 7.44 | wt.% |
| Asphaltenes | 0.18 | wt.% |
| Density at 60 C | 0.87 | g/cm |
| Viscosity at 60 C | 6.0 | cP |
| API gravity | 33 | deg |
| Core | Porosity, (%) | Permeability, mD | Pore Volume, mL | S (%) |
|---|---|---|---|---|
| H1 | 17.61 | 332 | 19.25 | 24.66 |
| H2 | 19.55 | 384 | 19.06 | 21.32 |
| H3 | 17.40 | 361 | 19.22 | 16.24 |
| H4 | 17.56 | 434 | 19.39 | 16.45 |
| H5 | 16.70 | 460 | 19.83 | 17.81 |
| H6 | 17.16 | 411 | 19.10 | 15.00 |
| H7 | 17.61 | 425 | 19.25 | 14.00 |
| H8 | 19.42 | 377 | 18.78 | 19.60 |
| Water Flood | Nanofluid Flood | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Core | Fluid | RF1 | RF2 | RF | S | RF1 | RF2 | RF | S | RF | E (%) |
| H1 | NF-A | 59.45 | 2.90 | 62.35 | 28.37 | 2.76 | 2.07 | 4.83 | 24.47 | 67.18 | 13.74 |
| H2 | 57.33 | 0.67 | 58.00 | 33.05 | 1.73 | 0.00 | 1.73 | 31.68 | 59.73 | 4.13 | |
| H3 | NF-B | 49.63 | 3.73 | 53.36 | 39.07 | 2.05 | 1.24 | 3.29 | 36.32 | 56.65 | 7.06 |
| H4 | 45.56 | 0.62 | 46.18 | 44.97 | 2.16 | 1.23 | 3.39 | 42.14 | 49.57 | 6.31 | |
| H5 | NF-C | 51.84 | 1.23 | 53.07 | 38.57 | 4.29 | 1.84 | 6.13 | 33.63 | 59.20 | 12.81 |
| H6 | 52.88 | 0.92 | 53.80 | 39.43 | 2.82 | 0.50 | 3.32 | 36.61 | 57.12 | 7.17 | |
| H7 | NF-D | 66.45 | 0.31 | 66.75 | 28.67 | 1.20 | 0.30 | 1.51 | 27.37 | 68.25 | 4.53 |
| H8 | 53.64 | 0.80 | 54.44 | 37.27 | 3.05 | 0.93 | 3.98 | 34.08 | 58.41 | 8.57 | |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bila, A.; Torsæter, O. Experimental Investigation of Polymer-Coated Silica Nanoparticles for EOR under Harsh Reservoir Conditions of High Temperature and Salinity. Nanomaterials 2021, 11, 765. https://doi.org/10.3390/nano11030765
Bila A, Torsæter O. Experimental Investigation of Polymer-Coated Silica Nanoparticles for EOR under Harsh Reservoir Conditions of High Temperature and Salinity. Nanomaterials. 2021; 11(3):765. https://doi.org/10.3390/nano11030765
Chicago/Turabian StyleBila, Alberto, and Ole Torsæter. 2021. "Experimental Investigation of Polymer-Coated Silica Nanoparticles for EOR under Harsh Reservoir Conditions of High Temperature and Salinity" Nanomaterials 11, no. 3: 765. https://doi.org/10.3390/nano11030765
APA StyleBila, A., & Torsæter, O. (2021). Experimental Investigation of Polymer-Coated Silica Nanoparticles for EOR under Harsh Reservoir Conditions of High Temperature and Salinity. Nanomaterials, 11(3), 765. https://doi.org/10.3390/nano11030765






