In Situ DRIFTS-MS Methanol Adsorption Study onto Supported NiSn Nanoparticles: Mechanistic Implications in Methanol Steam Reforming
Abstract
:1. Introduction
2. Materials and Methods
2.1. Chemical Reagents
2.2. Catalysts Synthesis
2.3. Characterization Methods
2.4. Catalytic Activity Tests
2.5. In Situ Drifts Measurements
3. Results
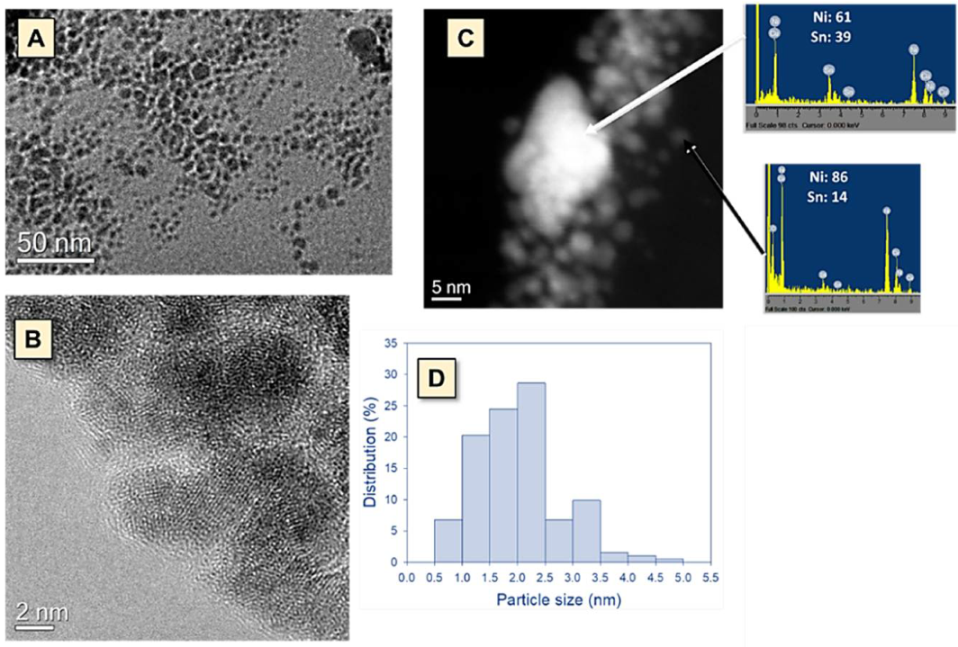
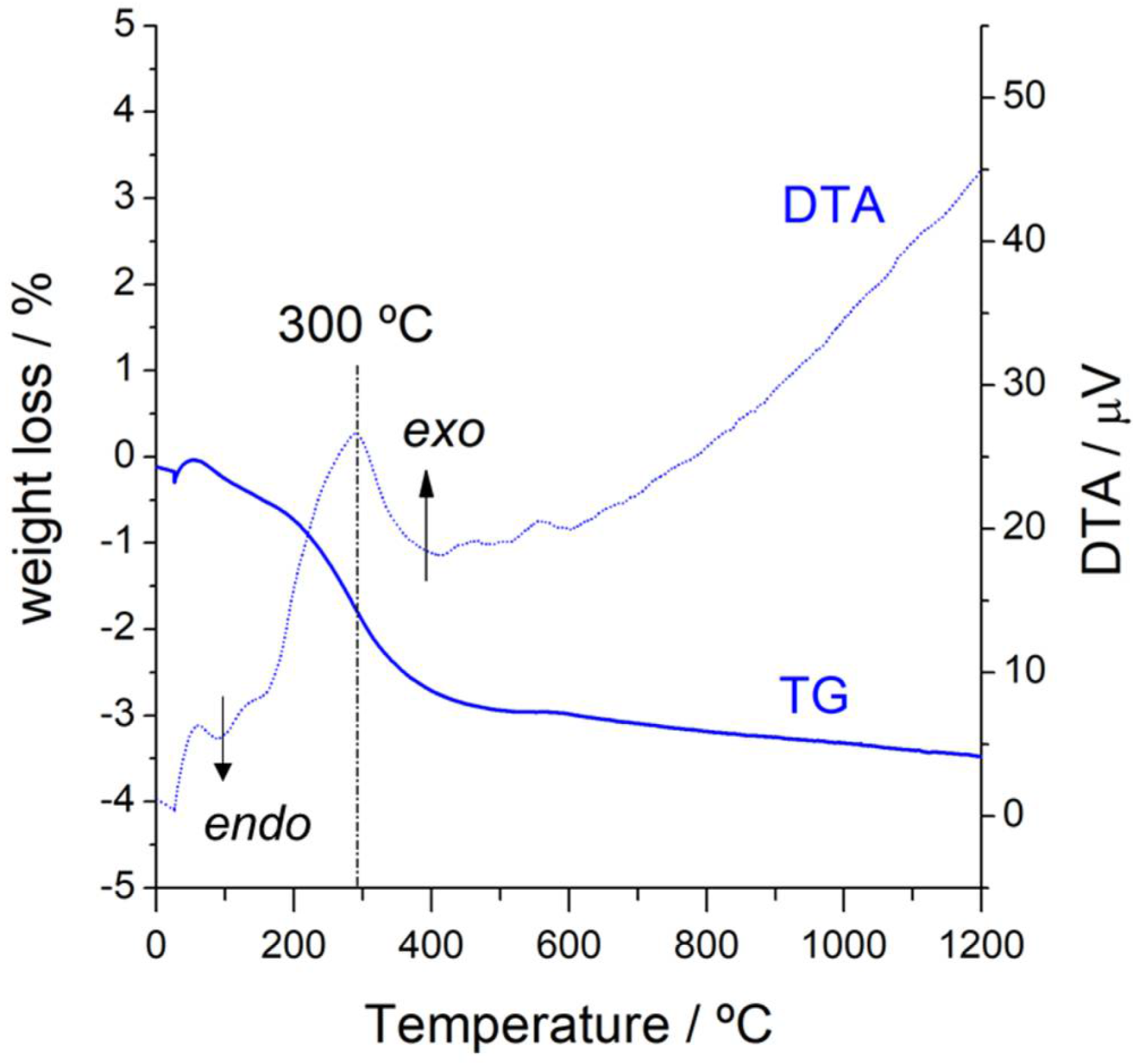
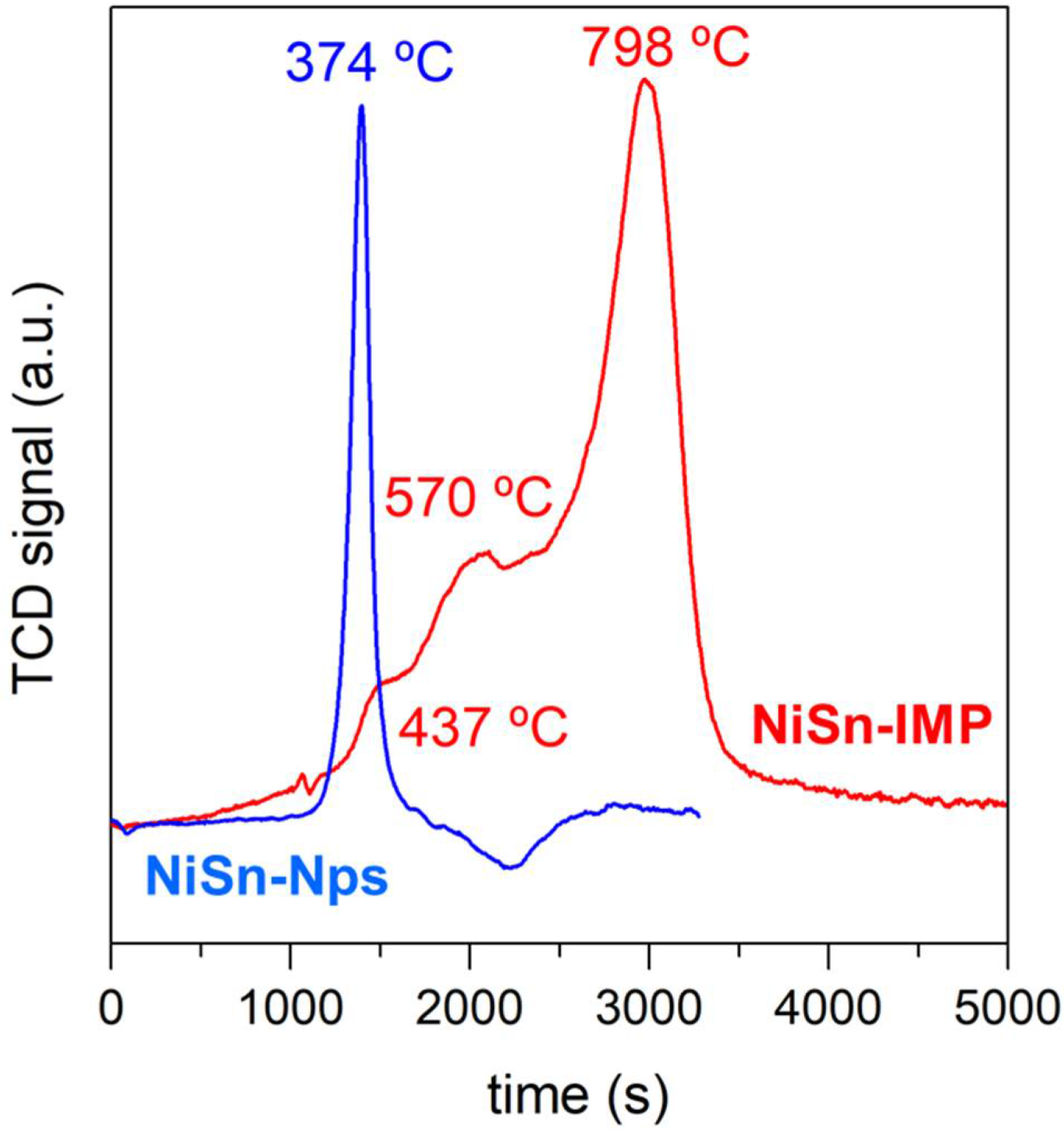
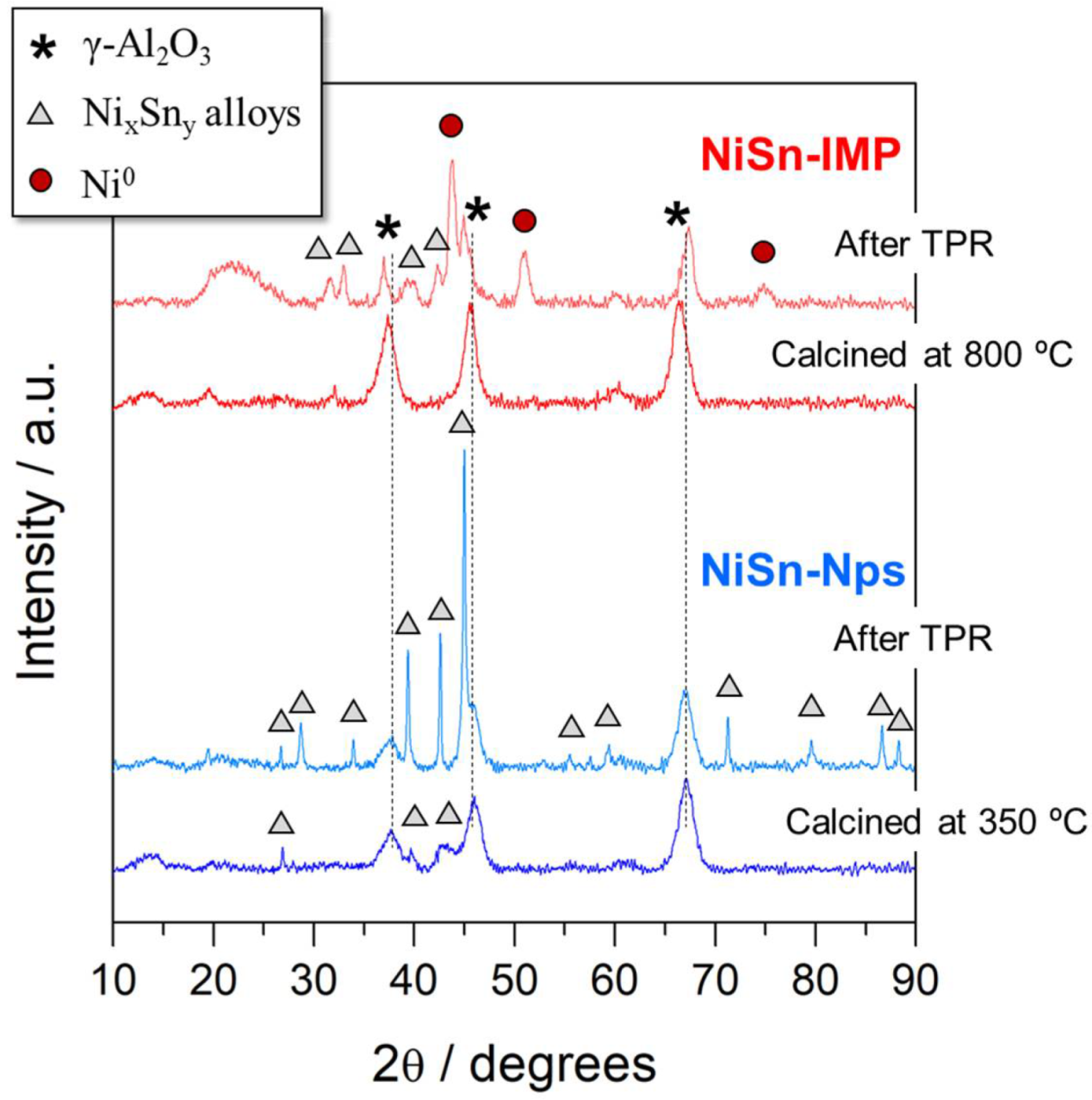
3.1. General Characterization for Both Catalysts
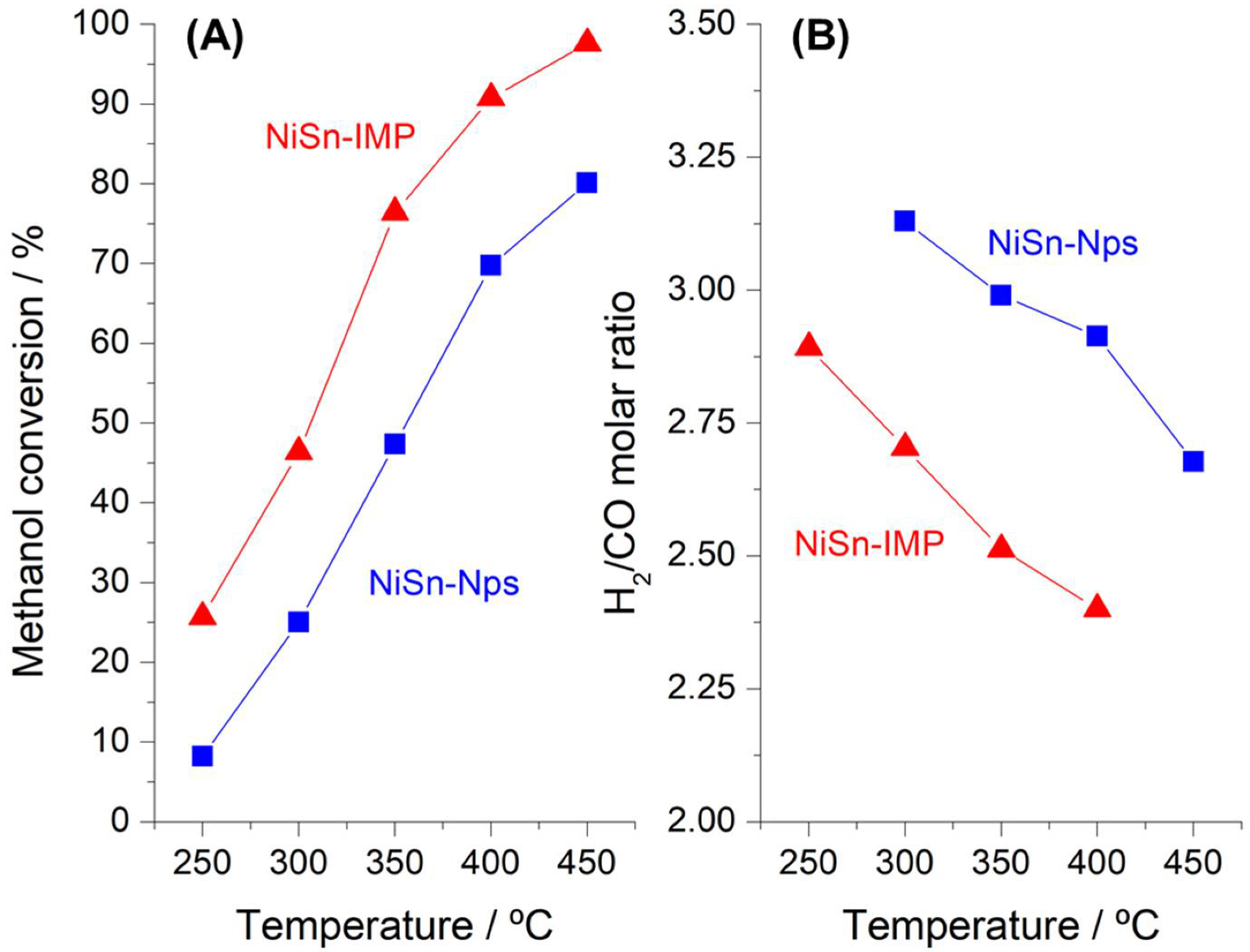
3.2. Catalytic Performance Testing on Methanol Steam Reforming
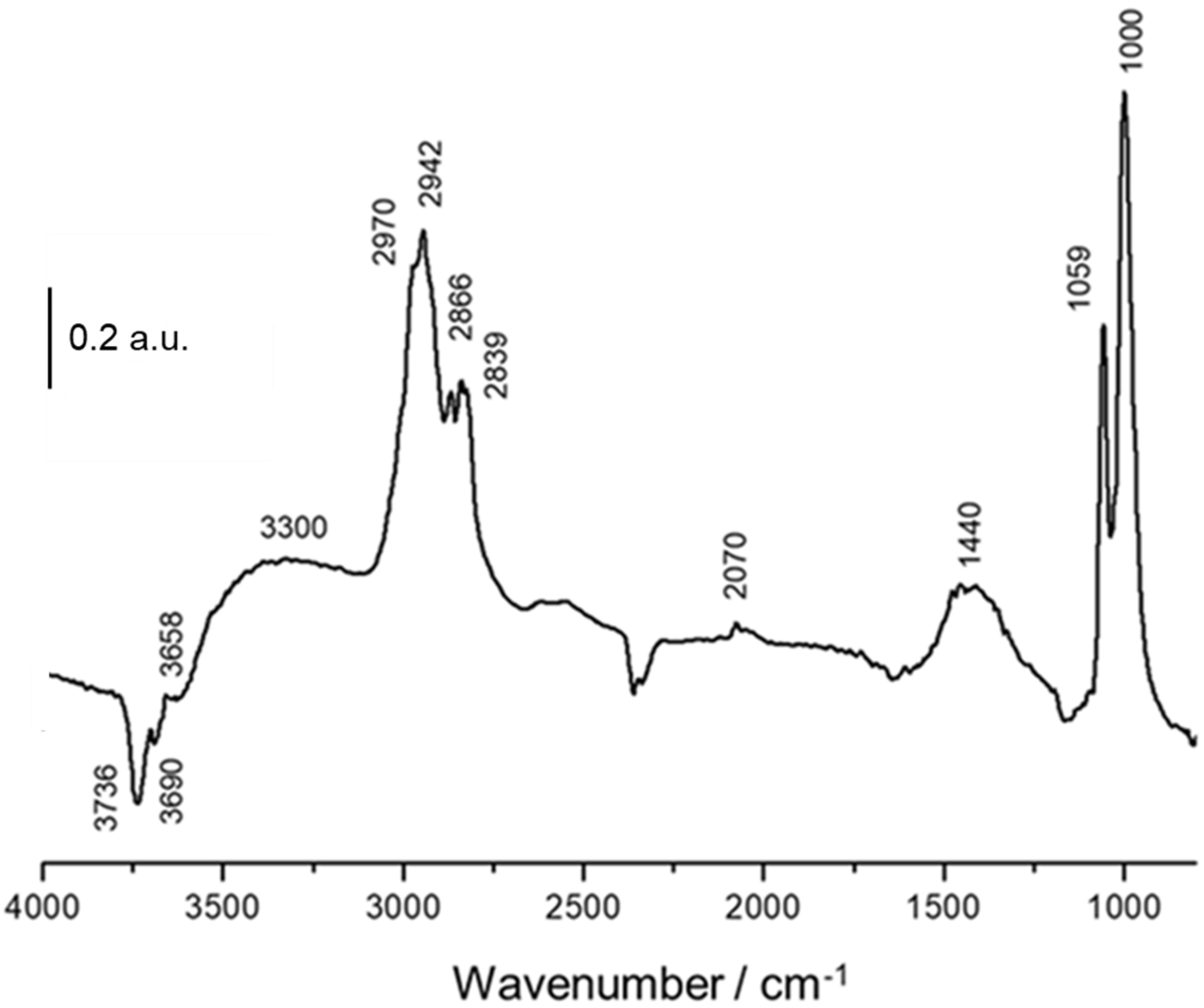
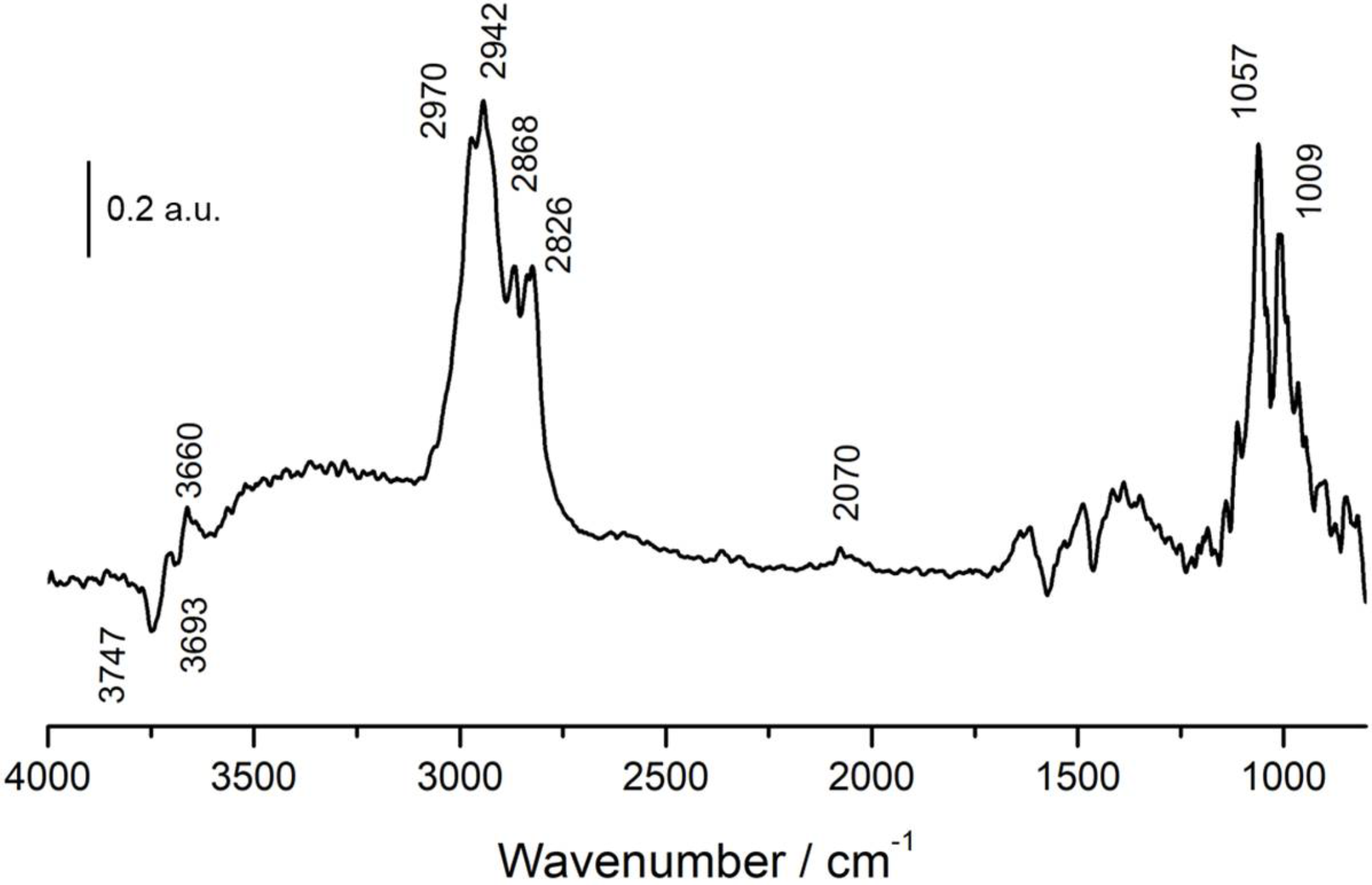
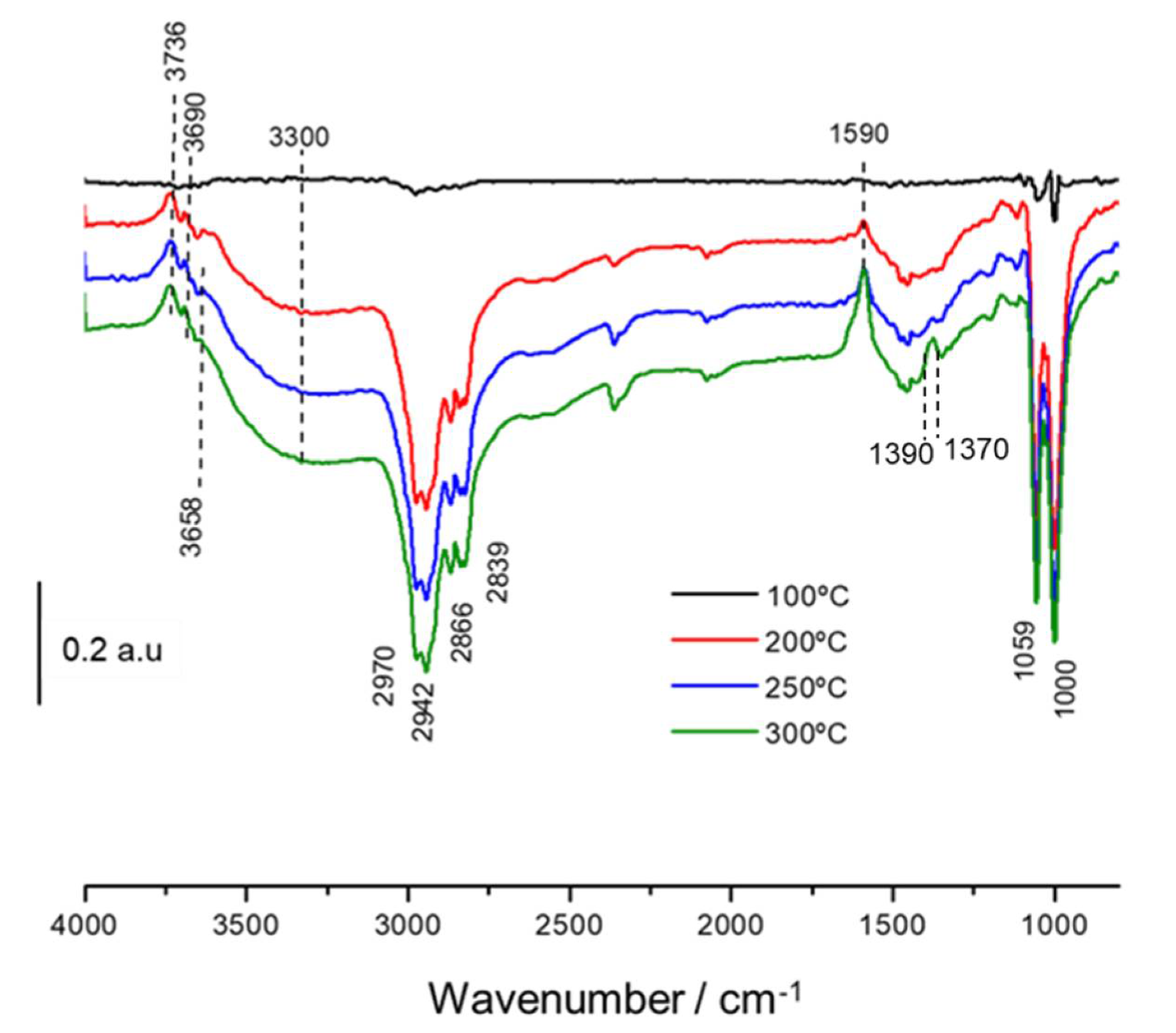
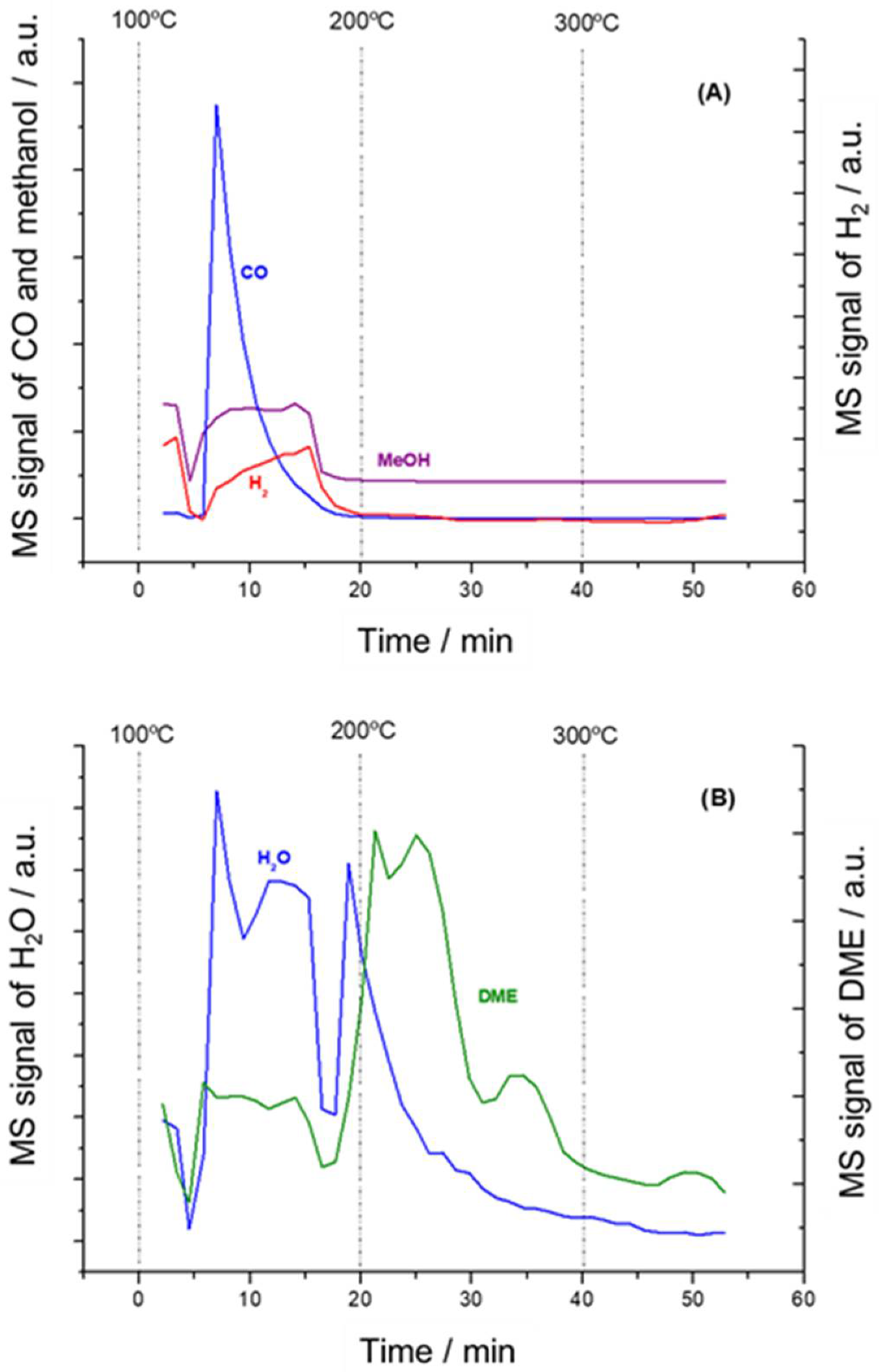
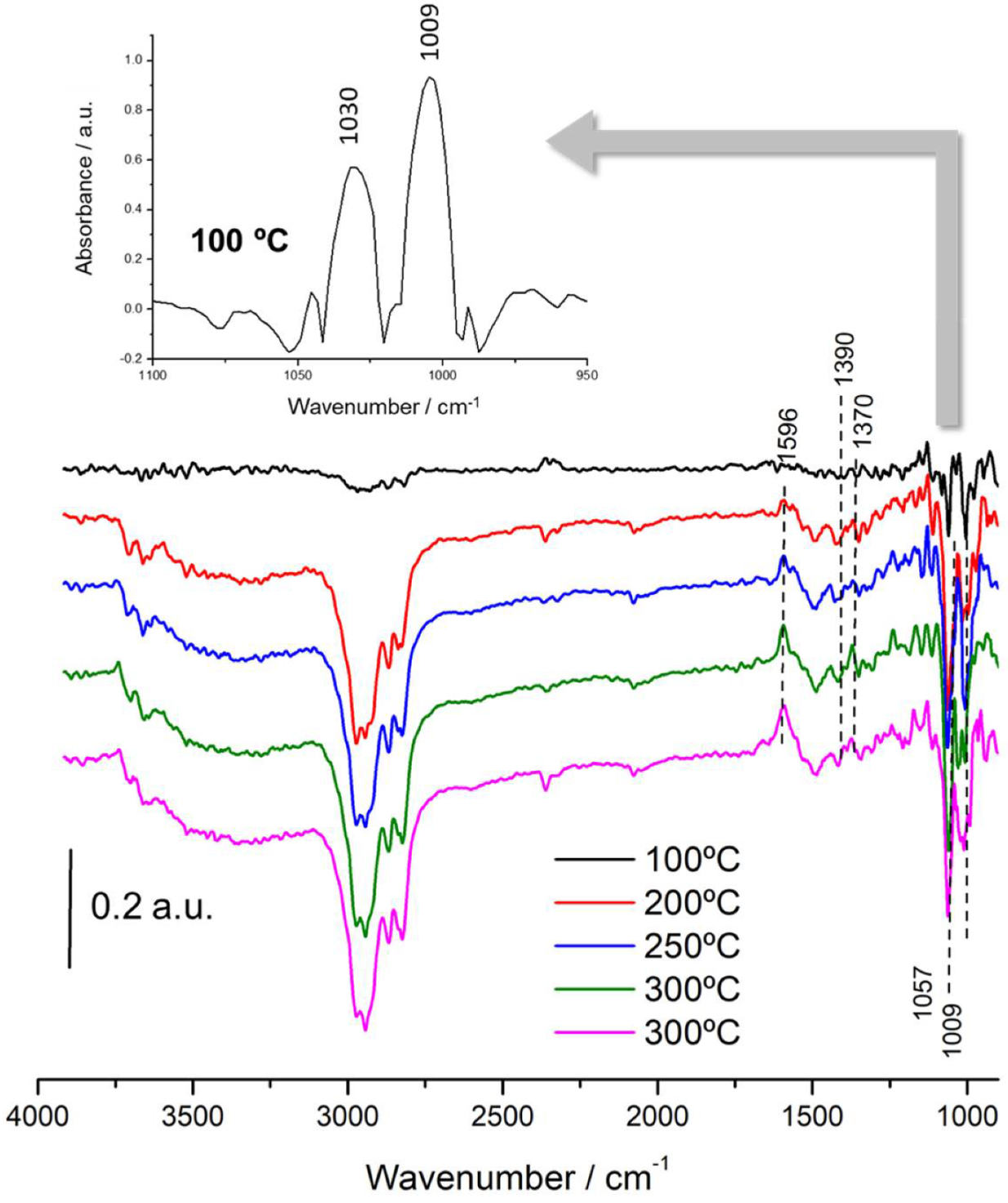
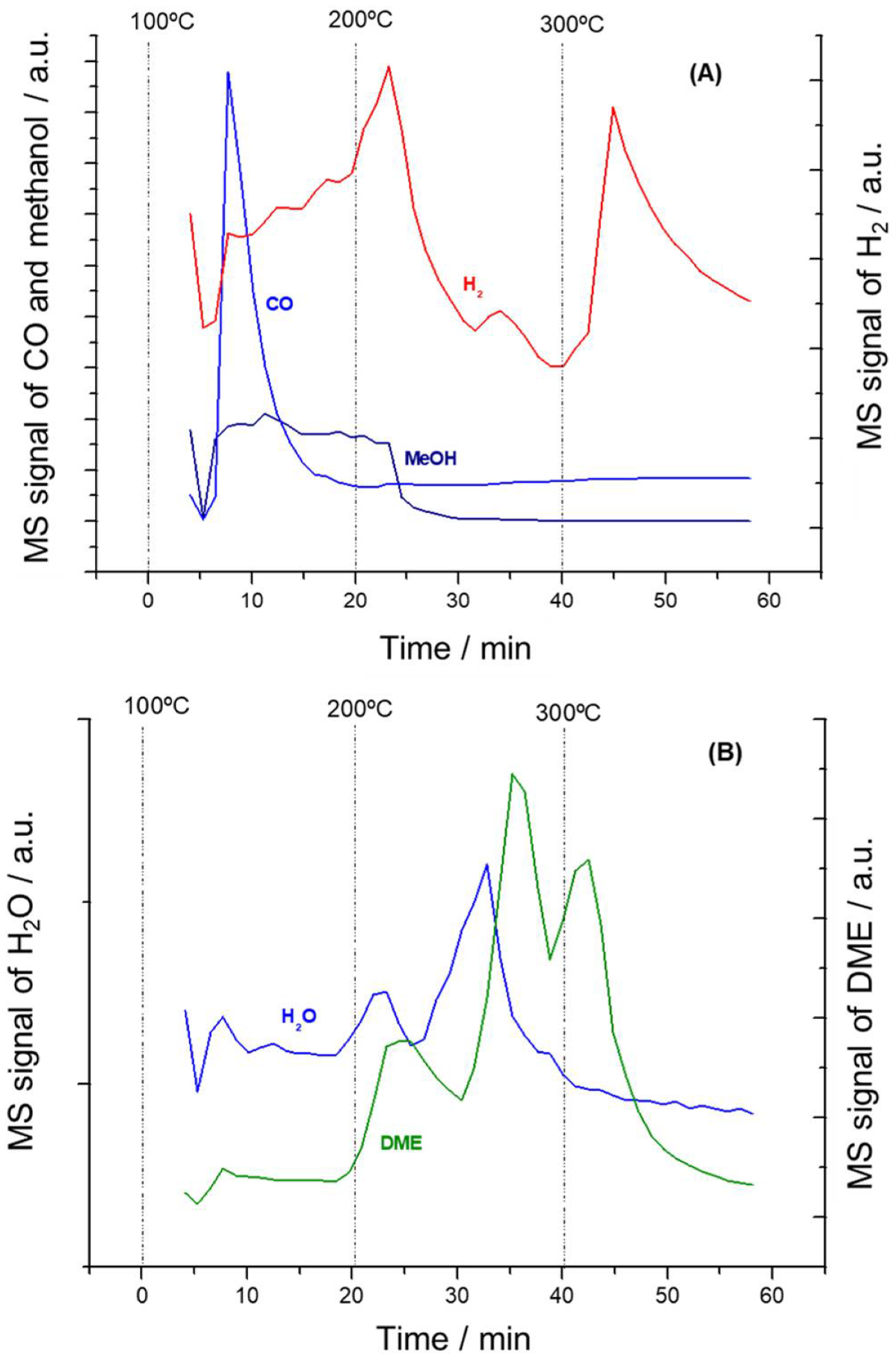
3.3. In Situ DRIFTS-MS Methanol Adsorption Studies
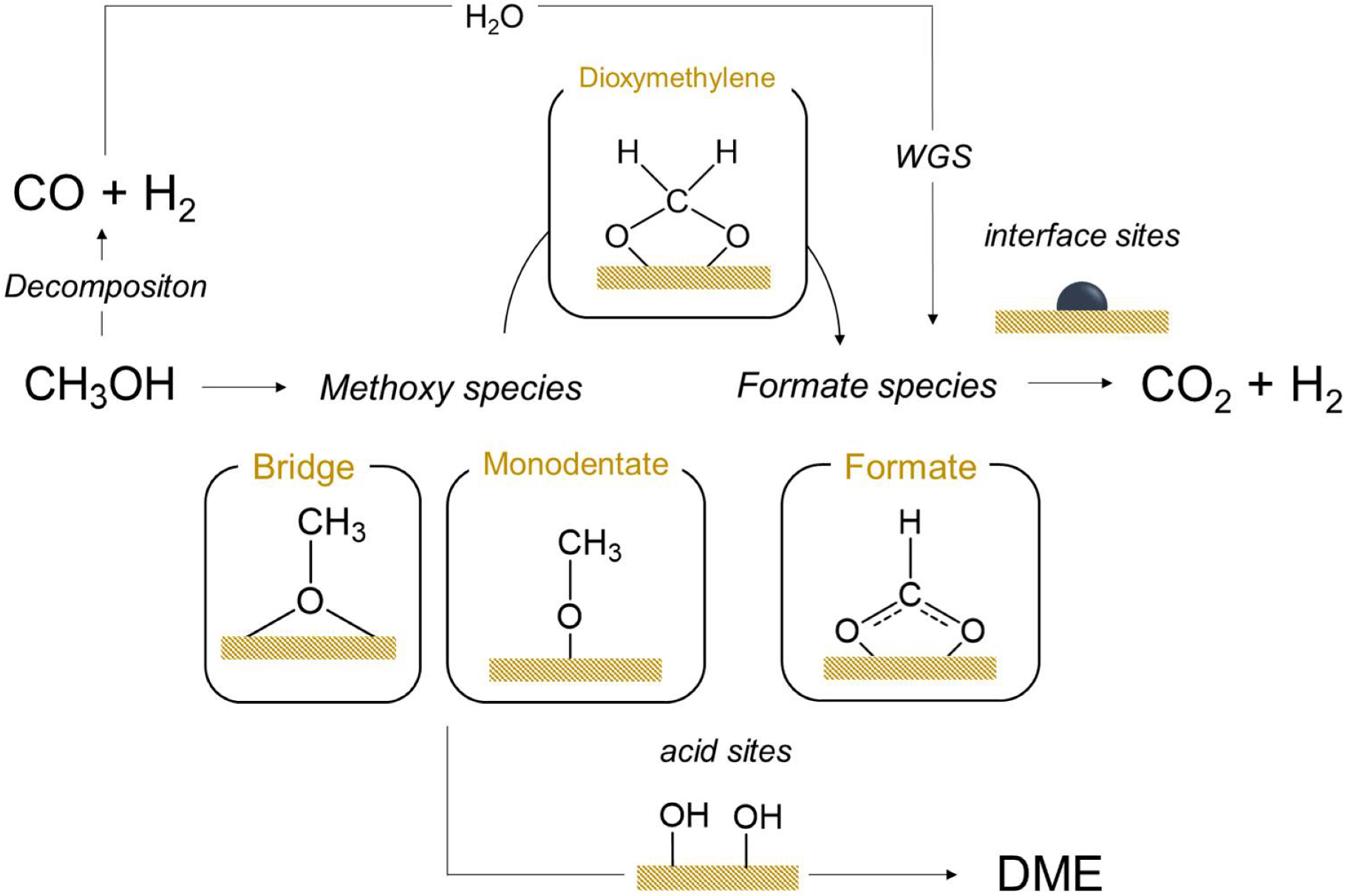
3.4. Mechanistic Insights
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Liobikiene, G.; Butkus, M. The European Union possibilities to achieve targets of Europe 2020 and Paris agreement climate policy. Renew. Energy 2017, 106, 298–309. [Google Scholar] [CrossRef]
- Gholami, P.; Khataee, A.; Bhatnagar, A.; Vahid, B. Synthesis of N-doped magnetic WO3–x@Mesoporous carbon using a diatom template and plasma modification: Visible-light-driven photocatalytic activities. ACS Appl. Mater. Interfaces 2021, 13, 13072–13086. [Google Scholar] [CrossRef] [PubMed]
- Gholami, P.; Khataee, A.; Bhatnagar, A. Photocatalytic degradation of antibiotic and hydrogen production using diatom-templated 3D WO3-x@mesoporous carbon nanohybrid under visible light irradiation. J. Clean. Prod. 2020, 275, 124157. [Google Scholar] [CrossRef]
- Garcia, G.; Arriola, E.; Chen, W.-H.; De Luna, M.D. A comprehensive review of hydrogen production from methanol thermochemical conversion for sustainability. Energy 2020, 217, 119384. [Google Scholar] [CrossRef]
- Abbandanak, M.H.; Taghizadeh, M.; Fallah, N. High-purity hydrogen production by sorption-enhanced methanol steam reforming over a combination of Cu–Zn–CeO2–ZrO2/MCM-41 catalyst and (Li–Na–K) NO3·MgO adsorbent. Int. J. Hydrog. Energy 2021, 46, 7099–7112. [Google Scholar] [CrossRef]
- Sá, S.; Silva, H.; Brandão, L.; Sousa, J.; Mendes, A. Catalysts for methanol steam reforming—A review. Appl. Catal. B Environ. 2010, 99, 43–57. [Google Scholar] [CrossRef]
- Yao, C.-Z.; Wang, L.-C.; Liu, Y.-M.; Wu, G.-S.; Cao, Y.; Dai, W.-L.; He, H.-Y.; Fan, K.-N. Effect of preparation method on the hydrogen production from methanol steam reforming over binary Cu/ZrO2 catalysts. Appl. Catal. A Gen. 2006, 297, 151–158. [Google Scholar] [CrossRef]
- Shishido, T.; Yamamoto, Y.; Morioka, H.; Takaki, K.; Takehira, K. Active Cu/ZnO and Cu/ZnO/Al2O3 catalysts prepared by homogeneous precipitation method in steam reforming of methanol. Appl. Catal. A Gen. 2004, 263, 249–253. [Google Scholar] [CrossRef]
- Águila, G.; Jiménez, J.; Guerrero, S.; Gracia, F.; Chornik, B.; Quinteros, S.; Araya, P. A novel method for preparing high surface area copper zirconia catalysts: Influence of the preparation variables. Appl. Catal. A Gen. 2009, 360, 98–105. [Google Scholar] [CrossRef]
- Iulianelli, A.; Ribeirinha, P.; Mendes, A.; Basile, A. Methanol steam reforming for hydrogen generation via conventional and membrane reactors: A review. Renew. Sustain. Energy Rev. 2014, 29, 355–368. [Google Scholar] [CrossRef] [Green Version]
- Jeong, H.; Kim, K.I.; Kim, T.H.; Ko, C.H.; Park, H.C.; Song, I.K. Hydrogen production by steam reforming of methanol in a micro-channel reactor coated with Cu/ZnO/ZrO2/Al2O3 catalyst. J. Power Sources 2006, 159, 1296–1299. [Google Scholar] [CrossRef]
- Papavasiliou, J.; Avgouropoulos, G.; Ioannides, T. Steam reforming of methanol over copper–manganese spinel oxide catalysts. Catal. Commun. 2005, 6, 497–501. [Google Scholar] [CrossRef]
- Dasireddy, V.D.; Likozar, B. Cu–Mn–O nano-particle/nano-sheet spinel-type materials as catalysts in methanol steam reforming (MSR) and preferential oxidation (PROX) reaction for purified hydrogen production. Renew. Energy 2021, 182, 713–724. [Google Scholar] [CrossRef]
- Bossola, F.; Roongcharoen, T.; Coduri, M.; Evangelisti, C.; Somodi, F.; Sementa, L.; Fortunelli, A.; Santo, V.D. Discovering indium as hydrogen production booster for a Cu/SiO2 catalyst in steam reforming of methanol. Appl. Catal. B Environ. 2021, 297, 120398. [Google Scholar] [CrossRef]
- Pérez-Hernández, R. Reactivity of Pt/Ni supported on CeO2-nanorods on methanol steam reforming for H2 production: Steady state and DRIFTS studies. Int. J. Hydrog. Energy 2021, 46, 25954–25964. [Google Scholar] [CrossRef]
- Conant, T.; Karim, A.M.; Lebarbier, V.; Wang, Y.; Girgsdies, F.; Schlögl, R.; Datye, A. Stability of bimetallic Pd–Zn catalysts for the steam reforming of methanol. J. Catal. 2008, 257, 64–70. [Google Scholar] [CrossRef]
- Iwasa, N.; Mayanagi, T.; Nomura, W.; Arai, M.; Takezawa, N. Effect of Zn addition to supported Pd catalysts in the steam reforming of methanol. Appl. Catal. A Gen. 2003, 248, 153–160. [Google Scholar] [CrossRef]
- Bobadilla, L.; Palma, S.; Ivanova, S.; Domínguez, M.; Romero-Sarria, F.; Centeno, M.; Odriozola, J. Steam reforming of methanol over supported Ni and Ni–Sn nanoparticles. Int. J. Hydrog. Energy 2013, 38, 6646–6656. [Google Scholar] [CrossRef]
- Zhou, Y.; Jin, C.; Li, Y.; Shen, W. Dynamic behavior of metal nanoparticles for catalysis. Nano Today 2018, 20, 101–120. [Google Scholar] [CrossRef]
- Huttunen, P.K.; Labadini, D.; Hafiz, S.S.; Gokalp, S.; Wolff, E.P.; Martell, S.M.; Foster, M. DRIFTS investigation of methanol oxidation on CeO2 nanoparticles. Appl. Surf. Sci. 2021, 554, 149518. [Google Scholar] [CrossRef]
- Bobadilla, L.F.; Garcia, C.; Delgado, J.J.; Sanz, O.; Romero-Sarria, F.; Centeno, M.A.; Odriozola, J.A. Influence of PVP in magnetic properties of NiSn nanoparticles prepared by polyol method. J. Magn. Magn. Mater. 2012, 324, 4011–4018. [Google Scholar] [CrossRef]
- Richardson, J.T.; Twigg, M.V. Reduction of impregnated NiO/α-Al2O3 association of Al3+ ions with NiO. Appl. Catal. A Gen. 1998, 167, 57–64. [Google Scholar] [CrossRef]
- Jacob, K.T.; Alcock, C.B. Activities and their relation to cation distribution in NiAl2O4/MgAl2O4 spinel solid solutions. J. Solid State Chem. 1977, 20, 79–88. [Google Scholar] [CrossRef]
- Penkova, A.D.; Bobadilla, L.; Ivanova, S.; Domínguez, M.; Romero-Sarria, F.; Roger, A.-C.; Centeno, M.; Odriozola, J.A. Hydrogen production by methanol steam reforming on NiSn/MgO–Al2O3 catalysts: The role of MgO addition. Appl. Catal. A Gen. 2011, 392, 184–191. [Google Scholar] [CrossRef]
- Richardson, J.T.; Scates, R.; Twigg, M.V. X-ray diffraction study of nickel oxide reduction by hydrogen. Appl. Catal. A Gen. 2003, 246, 137–150. [Google Scholar] [CrossRef]
- Gao, S.; Huang, H.; Wu, A.; Yu, J.; Gao, J.; Dong, X.; Liu, C.; Cao, G. Formation of Sn–M (M = Fe, Al, Ni) alloy nanoparticles by DC arc-discharge and their electrochemical properties as anodes for Li-ion batteries. J. Solid State Chem. 2016, 242, 127–135. [Google Scholar] [CrossRef]
- Adcock, P.A.; Pacheco, S.V.; Norman, K.M.; Uribe, F.A. Transition metal oxides as reconfigured fuel cell anode catalysts for improved CO tolerance: Polarization data. J. Electrochem. Soc. 2005, 152, A459. [Google Scholar] [CrossRef]
- Khzouz, M.; Gkanas, E.I.; Du, S.; Wood, J. Catalytic performance of Ni-Cu/Al2O3 for effective syngas production by methanol steam reforming. Fuel 2018, 232, 672–683. [Google Scholar] [CrossRef]
- Knözinger, H. Dehydration of alcohols on aluminum oxide. Angew. Chem. Int. Ed. 1968, 7, 791–805. [Google Scholar] [CrossRef]
- Vigué, H.; Quintard, P.; Merle-Méjeana, T.; Lorenzelli, V. FT-IR study of CH3OH adsorption on chlorinated γ-aluminas. J. Eur. Ceram. Soc. 1998, 18, 1753–1757. [Google Scholar] [CrossRef]
- Busca, G. Infrared studies of the reactive adsorption of organic molecules over metal oxides and of the mechanisms of their heterogeneously-catalyzed oxidation. Catal. Today 1996, 27, 457–496. [Google Scholar] [CrossRef]
- LaMotte, J.; Moravek, V.; Bensitel, M.; LaValley, J.C. FT-IR study of the structure and reactivity of methoxy species on ThO2 and CeO2. React. Kinet. Catal. Lett. 1988, 36, 113–118. [Google Scholar] [CrossRef]
- Hadjivanov, K.I.; Vayssilov, G.N. Characterization of oxide surfaces and zeolites by carbon monoxide as an IR probe molecule. ChemInform 2003, 34, 17296. [Google Scholar] [CrossRef]
- Greenler, R.G. Infrared study of the adsorption of methanol and ethanol on aluminum oxide. J. Chem. Phys. 1962, 37, 2094–2100. [Google Scholar] [CrossRef]
- Carrizosa, I.; Munuera, G.; Castañar, S. Study of the interaction of aliphatic alcohols with TiO2: III. Formation of alkyl-titanium species during methanol decomposition. J. Catal. 1977, 49, 265–277. [Google Scholar] [CrossRef]
- Turco, M.; Bagnasco, G.; Costantino, U.; Marmottini, F.; Montanari, T.; Ramis, G.; Busca, G. Production of hydrogen from oxidative steam reforming of methanolII. Catalytic activity and reaction mechanism on Cu/ZnO/Al2O3 hydrotalcite-derived catalysts. J. Catal. 2004, 228, 56–65. [Google Scholar] [CrossRef]
- Yaripour, F.; Baghaei, F.; Schmidt, I.; Perregaard, J. Catalytic dehydration of methanol to dimethyl ether (DME) over solid-acid catalysts. Catal. Commun. 2005, 6, 147–152. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bobadilla, L.F.; Azancot, L.; Ivanova, S.; Delgado, J.J.; Romero-Sarria, F.; Centeno, M.A.; Roger, A.-C.; Odriozola, J.A. In Situ DRIFTS-MS Methanol Adsorption Study onto Supported NiSn Nanoparticles: Mechanistic Implications in Methanol Steam Reforming. Nanomaterials 2021, 11, 3234. https://doi.org/10.3390/nano11123234
Bobadilla LF, Azancot L, Ivanova S, Delgado JJ, Romero-Sarria F, Centeno MA, Roger A-C, Odriozola JA. In Situ DRIFTS-MS Methanol Adsorption Study onto Supported NiSn Nanoparticles: Mechanistic Implications in Methanol Steam Reforming. Nanomaterials. 2021; 11(12):3234. https://doi.org/10.3390/nano11123234
Chicago/Turabian StyleBobadilla, Luis F., Lola Azancot, Svetlana Ivanova, Juan J. Delgado, Francisca Romero-Sarria, Miguel A. Centeno, Anne-Cécile Roger, and José A. Odriozola. 2021. "In Situ DRIFTS-MS Methanol Adsorption Study onto Supported NiSn Nanoparticles: Mechanistic Implications in Methanol Steam Reforming" Nanomaterials 11, no. 12: 3234. https://doi.org/10.3390/nano11123234
APA StyleBobadilla, L. F., Azancot, L., Ivanova, S., Delgado, J. J., Romero-Sarria, F., Centeno, M. A., Roger, A.-C., & Odriozola, J. A. (2021). In Situ DRIFTS-MS Methanol Adsorption Study onto Supported NiSn Nanoparticles: Mechanistic Implications in Methanol Steam Reforming. Nanomaterials, 11(12), 3234. https://doi.org/10.3390/nano11123234









