Sea Bass Primary Cultures versus RTgill-W1 Cell Line: Influence of Cell Model on the Sensitivity to Nanoparticles
Abstract
:1. Introduction
2. Materials and Methods
2.1. Preparation of Nanoparticle Stock Dispersions
2.2. Characterization
2.3. Protein Corona Quantification
2.4. Cell Cultures
2.4.1. Sea Bass Dicentrarchus labrax Gill Primary Cells
2.4.2. Rainbow Trout Oncorhynchus mykiss Gill Cell Line
2.5. Scanning Electron Microscopy (SEM)
2.6. Cytotoxicity Screening
2.7. Data Treatment
2.7.1. Toxicity Assays: Endpoints and Decision Trees
2.7.2. Integrative Biological Response Index
3. Results
3.1. Nanoparticle Characterization
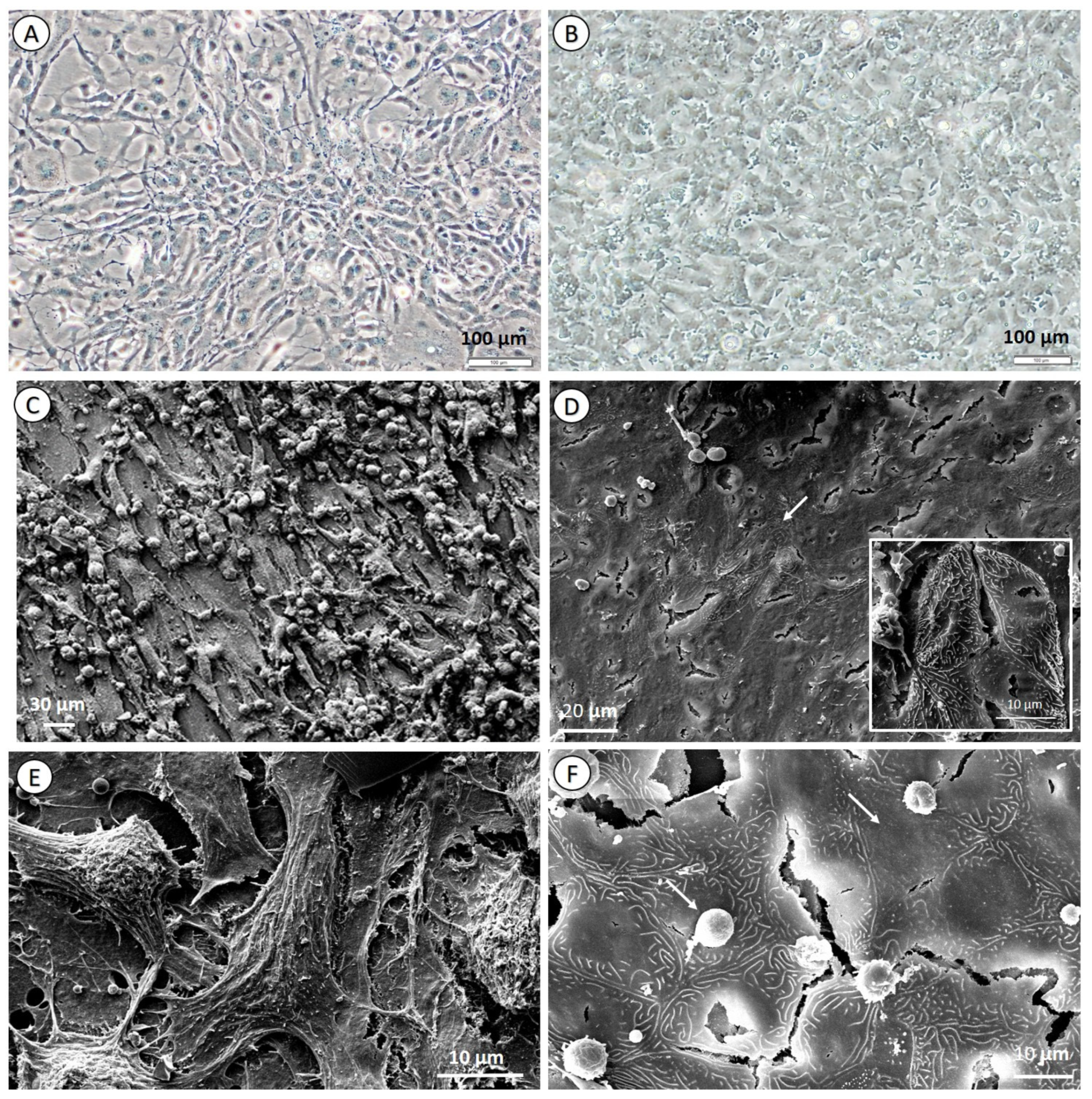
3.2. Cell Culture Morphology and Characterization
3.3. Toxicity Assays
3.3.1. Decision Trees and Individual Biomarkers
3.3.2. Exposure to TiO2 Nanoparticles
3.3.3. Exposure to Polystyrene (PS) Nanoparticles
3.3.4. Exposure to Ag Nanoparticles
3.3.5. Integrated Biological Response (IBR) Index
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Acknowledgments
Conflicts of Interest
References
- European Commission. Regulation (EC) No 1907/2006 of the European Parliament and of the Council of 18 December 2006 concerning the Registration, Evaluation, Authorisation and Restriction of Chemicals (REACH), Establishing a European Chemicals Agency, Amending Directive 1999/45/EC and Repealing Council Regulation (EEC) No 793/93 and Commission Regulation (EC) No 1488/94 as well as Council Directive 76/769/EEC and Commission Directives 91/155/EEC, 93/67/EEC, 93/105/EC and 2000/21/EC; European Union: Luxembourg, 2006.
- OECD. Test No. 203: Fish, Acute Toxicity Test, OECD Guidelines for the Testing of Chemicals, Section 2; OECD Publishing: Paris, France, 2019. [Google Scholar] [CrossRef]
- European Commission. Seventh Report on the Statistics on the Number of Animals Used for Experimental and Other Scientific Purposes in the Member States of the European Union; European Union: Luxembourg, 2013.
- European Commission. 2018 Statistical Report, SWD 204: Executive Summary Parts A and B and Part C. Data of 2018 from 28 Member States and Norway; European Union: Luxembourg, 2021.
- OECD. Test No. 210: Fish, Early-Life Stage Toxicity Test, OECD Guidelines for the Testing of Chemicals, Section 2; OECD Publishing: Paris, France, 2013. [Google Scholar] [CrossRef]
- OECD. Test No. 212: Fish, Short-Term Toxicity Test on Embryo and Sac-Fry Stages, OECD Guidelines for the Testing of Chemicals, Section 2; OECD Publishing: Paris, France, 1998. [Google Scholar] [CrossRef]
- Castaño, A.; Bols, N.; Braunbeck, T.; Dierickx, P.; Halder, M.; Isomaa, B.; Segner, H. The use of fish cells in ecotoxicology: The report and recommendations of ECVAM workshop 47. Altern. Lab. Anim. 2003, 31, 317–351. [Google Scholar] [CrossRef] [PubMed]
- Schirmer, K. Proposal to improve vertebrate cell cultures to establish them as substitutes for the regulatory testing of chemicals and effluents using fish. Toxicology 2006, 224, 163–183. [Google Scholar] [CrossRef]
- Tanneberger, K.; Knöbel, M.; Busser, F.; Sinnige, T.; Hermens, J.; Schirmer, K. Predicting fish acute toxicity using a fish gill cell line-based toxicity assay. Environ. Sci. Technol. 2013, 47, 1110–1119. [Google Scholar] [CrossRef] [PubMed]
- Natsch, A.; Laue, H.; Haupt, T.; von Niederhäusern, V.; Sanders, G. Accurate prediction of acute fish toxicity of fragrance chemicals with the RTgill-W1 cell assay. Environ. Toxicol. Chem. 2018, 37, 931–941. [Google Scholar] [CrossRef] [PubMed]
- Fischer, A.; Belanger, S.E.; Berckmans, P.; Bernhard, M.J.; Bláha, L.; Coman Schmid, D.E.; Dyer, S.D.; Haupt, T.; Hermens, J.L.M.; Hultman, M.T.; et al. Repeatability and Reproducibility of the RTgill-W1 Cell Line Assay for Predicting Fish Acute Toxicity. Toxicol. Sci. 2019, 169, 353–364. [Google Scholar] [CrossRef] [Green Version]
- Russell, W.M.S.; Burch, R.L. The Principles of Humane Experimental Technique; Universities Federation for Animal Welfare: Wheathampstead, UK, 1992. [Google Scholar]
- ISO.org. ISO 21115:2019. Water Quality—Determination of Acute Toxicity of Water Samples and Chemicals to a Fish Gill Cell Line (RTgill-W1). Available online: https://www.iso.org/standard/69933.html (accessed on 18 November 2021).
- Katsumiti, A.; Thorley, A.; Arostegui, I.; Reip, P.; Valsami-Jones, E.; Tetley, T.; Cajaraville, M. Cytotoxicity and cellular mechanisms of toxicity of CuO NPs in mussel cells in vitro and comparative sensitivity with human cells. Toxicol. In Vitro 2018, 48, 146–158. [Google Scholar] [CrossRef]
- Duroudier, N.; Katsumiti, A.; Mikolaczyk, M.; Schäfer, J.; Bilbao, E.; Cajaraville, M. Dietary exposure of mussels to PVP/PEI coated Ag nanoparticles causes Ag accumulation in adults and abnormal embryo development in their offspring. Sci. Total Environ. 2019, 655, 48–60. [Google Scholar] [CrossRef] [PubMed]
- Rosenkranz, P.; Fernández-Cruz, M.; Conde, E.; Ramírez-Fernández, M.; Flores, J.; Fernández, M.; Navas, J. Effects of cerium oxide nanoparticles to fish and mammalian cell lines: An assessment of cytotoxicity and methodology. Toxicol. In Vitro 2012, 26, 888–896. [Google Scholar] [CrossRef]
- García-Gómez, C.; García, S.; Obrador, A.; Almendros, P.; González, D.; Fernández, M. Effect of ageing of bare and coated nanoparticles of zinc oxide applied to soil on the Zn behaviour and toxicity to fish cells due to transfer from soil to water bodies. Sci. Total Environ. 2019, 706, 135713. [Google Scholar] [CrossRef]
- Book, F.; Ekvall, M.; Persson, M.; Lönnerud, S.; Lammel, T.; Sturve, J.; Backhaus, T. Ecotoxicity screening of seven different types of commercial silica nanoparticles using cellular and organismic assays: Importance of surface and size. NanoImpact 2019, 13, 100–111. [Google Scholar] [CrossRef]
- Farkas, J.; Christian, P.; Urrea, J.A.; Roos, N.; Hassellöv, M.; Tollefsen, K.E.; Thomas, K.V. Effects of silver and gold nanoparticles on rainbow trout (Oncorhynchus mykiss) hepatocytes. Aquat. Toxicol. 2010, 96, 44–52. [Google Scholar] [CrossRef]
- Minghetti, M.; Schirmer, K. Interference of silver nanoparticles with essential metal homeostasis in a novel enterohepatic fish in vitro system. Environ. Sci. Nano 2019, 6, 1777–1790. [Google Scholar] [CrossRef]
- Bermejo-Nogales, A.; Connolly, M.; Rosenkranz, P.; Fernández-Cruz, M.L.; Navas, J.M. Negligible cytotoxicity induced by different titanium dioxide nanoparticles in fish cell lines. Ecotoxicol. Environ. Saf. 2019, 138, 309–319. [Google Scholar] [CrossRef] [PubMed]
- Lammel, T.; Tsoukatou, G.; Jellinek, J.; Sturve, J. Development of three-dimensional (3D) spheroid cultures of the continuous rainbow trout liver cell line RTL-W1. Ecotoxicol. Environ. Saf. 2019, 167, 250–258. [Google Scholar] [CrossRef]
- Gillet, J.P.; Varma, S.; Gottesman, M.M. The Clinical Relevance of Cancer Cell Lines. J. Natl. Cancer Inst. 2013, 105, 452–458. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Scown, T.; van Aerle, R.; Tyler, C. Review: Do engineered nanoparticles pose a significant threat to the aquatic environment? Crit. Rev. Toxicol. 2009, 40, 653–670. [Google Scholar] [CrossRef]
- Federici, G.; Shaw, B.; Handy, R. Toxicity of titanium dioxide nanoparticles to rainbow trout (Oncorhynchus mykiss): Gill injury, oxidative stress, and other physiological effects. Aquat. Toxicol. 2007, 84, 415–430. [Google Scholar] [CrossRef]
- Griffitt, R.; Luo, J.; Gao, J.; Bonzongo, J.-C.; Barber, D. Effects of particle composition and species on toxicity of metallic nanomaterials in aquatic organisms. Environ. Toxicol. Chem. 2008, 27, 1972–1978. [Google Scholar] [CrossRef]
- Alvarado, N.; Quesada, I.; Hylland, K.; Marigómez, I.; Soto, M. Quantitative changes in metallothionein expression in target cell-types in the gills of turbot (Scophthalmus maximus) exposed to Cd, Cu, Zn and after a depuration treatment. Aquat. Toxicol. 2006, 77, 64–77. [Google Scholar] [CrossRef]
- Cedervall, T.; Lynch, I.; Lindman, S.; Berggård, T.; Thulin, E.; Nilsson, H.; Linse, S. Understanding the nanoparticle—Protein corona using methods to quantify exchange rates and affinities of proteins for nanoparticles. Proc. Natl. Acad. Sci. USA 2007, 104, 2050–2055. [Google Scholar] [CrossRef] [Green Version]
- Tenzer, S.; Docter, D.; Kuharev, J.; Musyanovych, A.; Fetz, V.; Hecht, R.; Stauber, R. Rapid formation of plasma protein corona critically affects nanoparticle pathophysiology. Nat. Nanotechnol. 2013, 8, 772–781. [Google Scholar] [CrossRef]
- Docter, D.; Distler, U.; Storck, W.; Kuharev, J.; Wünsch, D.; Hahlbrock, A.; Stauber, R. Quantitative profiling of the protein coronas that form around nanoparticles. Nat. Protoc. 2014, 9, 2030–2044. [Google Scholar] [CrossRef]
- Szekeres, G.; Fernandez-Iglesias, N.; Kneipp, J.; Montes-Bayón, M.; Bettmer, J. Mass spectrometric approach for the analysis of the hard protein corona of nanoparticles in living cells. J. Proteom. 2020, 2, 103582. [Google Scholar] [CrossRef] [PubMed]
- Collet, B.; Collins, C.; Lester, K. Engineered cell lines for fish health research. Dev. Comp. Immunol. 2018, 80, 34–40. [Google Scholar] [CrossRef]
- Roma, J.; Matos, A.R.; Vinagre, C.; Duarte, B. Engineered metal nanoparticles in the marine environment: A review of the effects on marine fauna. Mar. Environ. Res. 2020, 161, 105110. [Google Scholar] [CrossRef]
- Barnabé, G.; Boulineau-Coatanea, F.; Rene, F. Chronology of morphogenesis in Dicentrarchus labrax (L.) (Pisces, Serranidae) obtained by artificial reproduction. Aquaculture 1976, 8, 351–363. [Google Scholar] [CrossRef]
- Kelley, D.F. The importance of estuaries for sea bass, Dicentrarchus labrax (L.). J. Fish Biol. 1988, 33, 25–33. [Google Scholar] [CrossRef]
- Jensen, M.K.; Madsen, S.S.; Kristiansen, K. Osmoregulation and salinity effects on the expression and activity of Na+/K+-ATPase in the gills of European sea bass, Dicentrarchus labrax (L.). J. Exp. Zool. 1998, 282, 290–300. [Google Scholar] [CrossRef]
- Bols, N.; Barlian, A.; Chirino-Trejo, M.; Caldwell, S.; Goegan, P.; Lee, L. Development of a cell line from primary cultures of rainbow trout, Oncorhynchus mykiss (Walbaum), gills. J. Fish Dis. 1994, 17, 601–611. [Google Scholar] [CrossRef]
- Aengenheister, L.; Dugershaw, B.B.; Manser, P.; Wichser, A.; Schoenenberger, R.; Wick, P.; Hesler, M.; Kohl, Y.; Straskraba, S.; Suter, M.J.F.; et al. Investigating the accumulation and translocation of titanium dioxide nanoparticles with different surface modifications in static and dynamic human placental transfer models. Eur. J. Pharm. Biopharm. 2019, 142, 488–497. [Google Scholar] [CrossRef]
- Awet, T.T.; Kohl, Y.; Meier, F.; Straskraba, S.; Grün, A.L.; Ruf, T.; Jost, C.; Drexel, R.; Tunc, E.; Emmerling, C. Effects of polystyrene nanoparticles on the microbiota and functional diversity of enzymes in soil. Environ. Sci. Eur. 2018, 30, 11. [Google Scholar] [CrossRef] [PubMed]
- Grün, A.L.; Straskraba, S.; Schulz, S.; Schloter, M.; Emmerling, C. Long-term effects of environmentally relevant concentrations of silver nanoparticles on microbial biomass, enzyme activity, and functional genes involved in the nitrogen cycle of loamy soil. J. Environ. Sci. 2018, 69, 12–22. [Google Scholar] [CrossRef] [PubMed]
- Fröhlich, E.; Bonstingl, G.; Höfler, A.; Meindl, C.; Leitinger, G.; Pieber, T.; Roblegg, E. Comparison of two in vitro systems to assess cellular effects of nanoparticles-containing aerosols. Toxicol. In Vitro 2013, 27, 409–417. [Google Scholar] [CrossRef] [Green Version]
- Di Silvio, D.; Rigby, N.; Bajka, B.; Mayes, A.; Mackieb, A.; Baldelli Bombelli, F. Technical tip: High-resolution isolation of nanoparticle-protein corona complexes from physiological fluid. Nanoscale 2015, 7, 11980. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Schnell, S.; Stott, L.; Hogstrand, C.; Wood, C.; Kelly, S.; Pärt, P.; Bury, N. Procedures for the reconstruction, primary culture and experimental use of rainbow trout gill epithelia. Nat. Protoc. 2016, 11, 490–498. [Google Scholar] [CrossRef]
- Katsen-Globa, A.; Puetz, N.; Gepp, M.; Neubauer, J.; Zimmermann, H. Study of SEM preparation artefacts with correlative microscopy: Cell shrinkage of adherent cells by HMDS-drying. Scanning 2016, 38, 625–633. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rampersad, S.N. Multiple applications of Alamar Blue as an indicator of metabolic function and cellular health in cell viability bioassays. Sensors 2012, 12, 12347–12360. [Google Scholar] [CrossRef]
- Beliaeff, B.; Burgeot, T. Integrated biomarker response: A useful tool for ecological risk assessment. Environ. Toxicol. Chem. 2002, 21, 1316–1322. [Google Scholar] [CrossRef] [PubMed]
- Broeg, K.; Lehtonen, K. Indices for the assessment of environmental pollution of the Baltic Sea coasts: Integrated assessment of a multi-biomarker approach. Mar. Pollut. Bull. 2006, 53, 508–522. [Google Scholar] [CrossRef] [Green Version]
- Benito, D.; Ahvo, A.; Nuutinen, J.; Bilbao, D.; Saenz, J.; Etxebarria, N.; Lekube, X.; Izagirre, U.; Lehtonen, K.K.; Marigómez, I.; et al. Influence of season-depending ecological variables on biomarker baseline levels in mussels (Mytilus trossulus) from two Baltic Sea subregions. Sci. Total Environ. 2019, 689, 1087–1103. [Google Scholar] [CrossRef]
- Oh, N.; Park, J. Endocytosis and exocytosis of nanoparticles in mammalian cells. Int. J. Nanomed. 2014, 9 (Suppl. S1), 51–63. [Google Scholar] [CrossRef] [Green Version]
- He, C.; Hu, Y.; Yin, L.; Tang, C.; Yin, C. Effects of particle size and surface charge on cellular uptake and biodistribution of polymeric nanoparticles. Biomaterials 2010, 31, 3657–3666. [Google Scholar] [CrossRef]
- Walkey, C.D.; Olsen, J.B.; Guo, H.; Emili, A.; Chan, W.C. Nanoparticle size and surface chemistry determine serum protein adsorption and macrophage uptake. J. Am. Chem. Soc. 2012, 134, 2139–2147. [Google Scholar] [CrossRef] [PubMed]
- Lunov, O.; Zablotskii, V.; Syrovets, T.; Röcker, C.; Tron, K.; Nienhaus, G.; Simmet, T. Modeling receptor-mediated endocytosis of polymer-functionalized iron oxide nanoparticles by human macrophages. Biomaterials 2011, 32, 547–555. [Google Scholar] [CrossRef]
- Böhme, U.; Scheler, U. Effective charge of bovine serum albumin determined by electrophoresis NMR. Chem. Phys. Lett. 2007, 435, 342–345. [Google Scholar] [CrossRef]
- Lai, Y.-S.; John, J.; Lin, C.-H.; Guo, I.-C.; Chen, S.-C.; Fang, K.; Chang, C.-Y. Establishment of cell lines from a tropical grouper, Epinephelus awoara (Temminck & Schlegel), and their susceptibility to grouper irido- and nodaviruses. J. Fish Dis. 2003, 26, 31–42. [Google Scholar] [CrossRef]
- Lee, L.E.J.; Dayeh, V.R.; Schirmer, K.; Bols, N.C. Applications and potential uses of fish gill cell lines: Examples with RTgill-W1. Cell. Dev. Biol. Anim. 2009, 45, 127–134. [Google Scholar] [CrossRef]
- Carmona, R.; García-Gallego, M.; Sanz, A.; Domezaín, A.; Ostos-Garrido, M.V. Chloride cells and pavement cells in gill epithelia of Acipenser naccarii: Ultrastructural modifications in seawater-acclimated specimens. J. Fish Biol. 2004, 64, 553–566. [Google Scholar] [CrossRef]
- Avella, M.; Berhaut, J.; Payan, P. Primary culture of gill epithelial cells from the sea bass Dicentrarchus labrax. Cell. Dev. Biol. Anim. 1994, 30, 41–49. [Google Scholar] [CrossRef]
- Varsamos, S.; Diaz, J.; Charmantier, G.; Flik, G.; Blasco, C.; Connes, R. Branchial chloride cells in sea bass (Dicentrarchus labrax) adapted to fresh water, seawater, and doubly concentrated seawater. J. Exp. Zool. 2002, 293, 12–26. [Google Scholar] [CrossRef]
- Mallick, E.; Scutt, N.; Scutt, A.; Rolf, C. Passage and concentration-dependent effects of indomethacin on tendon derived cells. J. Orthop. Surg. Res. 2009, 4, 9. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Klein, R.; Nogueira, L.; Domingos-Moreira, F.; Gomes Costa, P.; Bianchini, A.; Wood, C. Effects of sublethal Cd, Zn, and mixture exposures on antioxidant defense and oxidative stress parameters in early life stages of the purple sea urchin Strongylocentrotus purpuratus. Aquat. Toxicol. 2019, 217, 105338. [Google Scholar] [CrossRef] [PubMed]
- Wood, C.M.; Farrell, A.P.; Brauner, C.J. (Eds.) Homeostasis and Toxicology of Non-Essential Metals, Fish Physiology; Elsevier: Amsterdam, The Netherlands, 2012; pp. 1–65. [Google Scholar] [CrossRef]
- Khosravi-Katuli, K.; Shabani, A.; Paknejad, H.; Imanpoor, M. Comparative toxicity of silver nanoparticle and ionic silver in juvenile common carp (Cyprinus carpio): Accumulation, physiology and histopathology. J. Hazard. Mater. 2018, 359, 373–381. [Google Scholar] [CrossRef]
- Ferreira, P.; Fonte, E.; Soares, M.; Carvalho, F.; Guilhermino, L. Effects of multi-stressors on juveniles of the marine fish Pomatoschistus microps: Gold nanoparticles, microplastics and temperature. Aquat. Toxicol. 2016, 170, 89–103. [Google Scholar] [CrossRef]
- Kühnel, D.; Busch, W.; Meißner, T.; Springer, A.; Potthoff, A.; Richter, V.; Gelinsky, M.; Scholz, M.; Schirmer, K. Agglomeration of tungsten carbide nanoparticles in exposure medium does not prevent uptake and toxicity toward a rainbow trout gill cell line. Aquat. Toxicol. 2009, 93, 91–99. [Google Scholar] [CrossRef]
- Calow, P. Physiological costs of combating chemical toxicants: Ecological implications. Comp. Biochem. Physiol. Part C Comp. Pharmacol. 1991, 100, 3–6. [Google Scholar] [CrossRef]
- Temmink, J.; Bouwmeister, P.; De Jong, P.; van den Berg, J. An ultrastructural study of chromate-induced hyperplasia in the gill of rainbow trout (Salmo gairdneri). Aquat. Toxicol. 1983, 4, 165–179. [Google Scholar] [CrossRef]
- Cengiz, E. Gill and kidney histopathology in the freshwater fish Cyprinus carpio after acute exposure to deltamethrin. Environ. Toxicol. Pharmacol. 2006, 22, 200–204. [Google Scholar] [CrossRef]
- Sayed, A.-D.; Mekkawy, I.; Mahmoud, U.; Nagiub, M. Histopathological and histochemical effects of silver nanoparticles on the gills and muscles of African catfish (Clarias garepinus). Sci. Afr. 2020, 7, e00230. [Google Scholar] [CrossRef]





| Material | Coating | Media | Z-Ave (nm) | St.Dev | Z-Pot (mV) | St.Dev |
|---|---|---|---|---|---|---|
| TiO2 | ||||||
| NH2 | H2O | 73.86 | 1.78 | 58.6 | 5.41 | |
| NH2 | L15 | 137.33 | 10.21 | −16.53 | 2.35 | |
| NH2 | SW | 1567.3 | 60.8 | −11.9 | 1.529 | |
| COOH | H2O | 24.82 | 5.37 | −29.2 | 3.62 | |
| COOH | L15 | 178.7 | 16.3 | −14.9 | 1.627 | |
| COOH | SW | 2103.3 | 45.6 | −9.6 | 1.816 | |
| Polystyrene | ||||||
| NH2 | H2O | 141.7 | 2.04 | 54.6 | 1.92 | |
| NH2 | L15 | 918.0 | 61.6 | −9.3 | 1.519 | |
| NH2 | SW | 5542.5 | 928.6 | −8.1 | 1.627 | |
| COOH | H2O | 61.02 | 1.3 | −60.04 | 0.987 | |
| COOH | L15 | 7640.7 | 609.6 | −12.5 | 2.003 | |
| COOH | SW | 9643.3 | 1682.1 | −10.9 | 0.123 | |
| Ag | ||||||
| NH2 | H2O | 93.33 | 2.04 | 18.4 | 0.1 | |
| NH2 | L15 | 5561.3 | 1576.1 | −13.9 | 0.354 | |
| NH2 | SW | 1.90 × 107 | 5.985 | −12 | 0.168 | |
| COOH | H2O | 89.61 | 0.285 | −61.1 | 1.4 | |
| COOH | L15 | 7.15 × 106 | 6.18 × 106 | −5.6 | 1.106 | |
| COOH | SW | 8417.0 | 666.3 | −4.7 | 0.026 |
| Scheme 1. | RTgill W1 | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| TiO2 | TiO2 | ||||||||||
| Assay | Factor 1 | Sig f1 | Factor 2 | Sig f2 | Factor 3 | Sig f3 | Assay | Factor 1 | Sig f1 | Factor 2 | Sig f2 |
| ROS | Time | 0.003 | Coating (48 h) | 0.046 | ROS | ||||||
| NRU | ExpMedia | <0.001 | Time (L15) | <0.001 | Weathering (48 h) | 0.011 | NRU | Time | 0.027 | ||
| WST | Time | 0.001 | Protein (48 h) | 0.01 | WST | Concentration | 0.004 | ||||
| AlaB | Weathering | <0.001 | Time (Y) | 0.003 | ExpMedia (48 h) | 0.008 | AlaB | Weathering | <0.001 | Coating (Y) | 0.032 |
| PS | PS | ||||||||||
| Assay | Factor 1 | Sig f1 | Factor 2 | Sig f2 | Factor 3 | Sig f3 | Assay | Factor 1 | Sig f1 | Factor 2 | Sig f2 |
| ROS | ExpMedia | <0.001 | Weathering (SW) | <0.001 | ROS | Time | <0.001 | ||||
| Time (L15) | <0.001 | ||||||||||
| NRU | Coating | 0.007 | ExpMedia (a) | 0.01 | Time (a, L15) | 0.023 | NRU | Conc | 0.003 | ||
| WST | n.a | WST | Weathering | 0.012 | |||||||
| AlaB | Coating | <0.001 | AlaB | ||||||||
| Ag | Ag | ||||||||||
| Assay | Factor 1 | Sig f1 | Factor 2 | Sig f2 | Factor 3 | Sig f3 | Assay | Factor 1 | Sig f1 | Factor 2 | Sig f2 |
| ROS | Time | 0.001 | Weathering/ ExpMedia | 0.044/0.009 | ExpMedia/Weathering | <0.001/0.015) | ROS | Time | <0.001 | Conc | 0.032 |
| NRU | Weathering | <0.001 | Time (non w) | 0.011 | ExpMedia (non w) | NRU | Time | <0.001 | |||
| WST | Time | 0.001 | WST | Conc | 0.001 | ||||||
| AlaB | Time | 0.002 | Weathering (24 h) | <0.001 | AlaB | Conc | 0.002 | Time (conc<10) | 0.002 | ||
| Coating (48 h) | <0.001 | ||||||||||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Jimeno-Romero, A.; Gwinner, F.; Müller, M.; Mariussen, E.; Soto, M.; Kohl, Y. Sea Bass Primary Cultures versus RTgill-W1 Cell Line: Influence of Cell Model on the Sensitivity to Nanoparticles. Nanomaterials 2021, 11, 3136. https://doi.org/10.3390/nano11113136
Jimeno-Romero A, Gwinner F, Müller M, Mariussen E, Soto M, Kohl Y. Sea Bass Primary Cultures versus RTgill-W1 Cell Line: Influence of Cell Model on the Sensitivity to Nanoparticles. Nanomaterials. 2021; 11(11):3136. https://doi.org/10.3390/nano11113136
Chicago/Turabian StyleJimeno-Romero, Alba, Frederik Gwinner, Michelle Müller, Espen Mariussen, Manu Soto, and Yvonne Kohl. 2021. "Sea Bass Primary Cultures versus RTgill-W1 Cell Line: Influence of Cell Model on the Sensitivity to Nanoparticles" Nanomaterials 11, no. 11: 3136. https://doi.org/10.3390/nano11113136
APA StyleJimeno-Romero, A., Gwinner, F., Müller, M., Mariussen, E., Soto, M., & Kohl, Y. (2021). Sea Bass Primary Cultures versus RTgill-W1 Cell Line: Influence of Cell Model on the Sensitivity to Nanoparticles. Nanomaterials, 11(11), 3136. https://doi.org/10.3390/nano11113136






