Designing pH-Dependent Systems Based on Nanoscale Calcium Carbonate for the Delivery of an Antitumor Drug
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Synthesis of CaNPs by Coprecipitation
2.3. Characterization of CaNPs
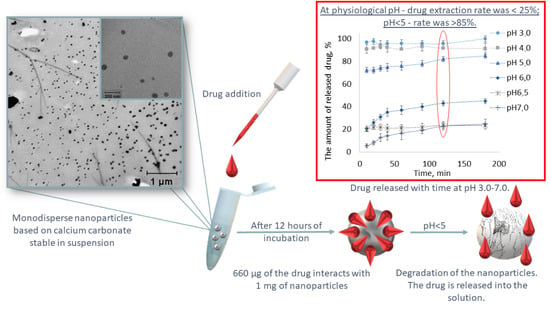
2.4. Reversible Binding of CaNPs to DOX
2.5. The Cytotoxicity of CaNPs, CaNP-DOX, and DOX
2.6. Statistical Evaluation of Experimental Error
3. Results and Discussion
3.1. Synthesis and Assessment of CaCO3 Properties
3.2. Interaction of DOX with CaNP7
3.3. Cytotoxicity Assays of CaNP7 and Its Composites with DOX
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Shan, D.; Zhu, M.; Xue, H.; Cosnier, S. Development of amperometric biosensor for glucose based on a novel attractive enzyme immobilization matrix: Calcium carbonate nanoparticles. Biosens. Bioelectron. 2007, 22, 1612–1617. [Google Scholar] [CrossRef]
- Bala, H.; Zhang, Y.; Ynag, H.; Wang, C.; Li, M.; Lv, X.; Wang, Z. Preparation and characteristics of calcium carbonate/silica nanoparticles with core-shell structure. Colloids Surf. A Physicochem. Eng. Asp. 2007, 294, 8–13. [Google Scholar] [CrossRef]
- Zhang, Y.; Ma, P.; Wang, Y.; Du, J.; Zhou, Q.; Zhu, Z.; Yang, X.; Yuan, J. Biocompatibility of Porous Spherical Calcium Carbonate Microparticles on Hela Cells. World J. Nano Sci. Eng. 2012, 2, 25–31. [Google Scholar] [CrossRef] [Green Version]
- Maleki Dizaj, S.; Barzegar-Jalali, M.; Zarrintan, M.H.; Adibkia, K.; Lotfipour, F. Calcium carbonate nanoparticles as cancer drug delivery system. Expert Opin. Drug Deliv. 2015, 12, 1649–1660. [Google Scholar] [CrossRef] [PubMed]
- Iafisco, M.; Delgado-Lopez, J.M.; Varoni, E.M.; Tampieri, A.; Rimondini, L.; Gomez-Morales, J.; Prat, M. Cell surface receptor targeted biomimetic apatite nanocrystals for cancer therapy. Small 2013, 9, 3834–3844. [Google Scholar] [CrossRef] [PubMed]
- Guo, Y.; Li, H.; Shi, W.; Zhang, J.; Feng, J.; Yang, X.; Wang, K.; Zhang, H.; Yang, L. Targeted delivery and pH-responsive release of doxorubicin to cancer cells using calcium car-bonate/hyaluronate/glutamate mesoporous hollow spheres. J. Colloid Interface Sci. 2017, 502, 59–66. [Google Scholar] [CrossRef]
- Li, L.; Yang, Y.; Lv, Y.; Yin, P.; Lei, T. Porous calcite CaCO3 microspheres: Preparation, characterization and release behavior as doxoru-bicin carrier. Colloids Surf. B Biointerfaces 2020, 186, 110720. [Google Scholar] [CrossRef] [PubMed]
- Dai, Y.; Xu, C.; Sun, X.; Chen, X. Nanoparticle design strategies for enhanced anticancer therapy by exploiting the tumour mi-croenvironment. Chem. Soc. Rev. 2017, 46, 3830–3852. [Google Scholar] [CrossRef]
- Zhao, Y.; Luo, Z.; Li, M.; Qu, Q.; Ma, X.; Yu, S.-H.; Zhao, Y. A Preloaded Amorphous Calcium Carbonate/Doxorubicin@Silica Nanoreactor for pH-Responsive Delivery of an Anticancer Drug. Angew. Chem. Int. Ed. 2015, 54, 919–922. [Google Scholar] [CrossRef]
- Hamidu, A.; Mokrish, A.; Mansor, R.; Razak, I.S.A.; Danmaigoro, A.; Jaji, A.Z.; Bakar, Z.A. Modified methods of nanoparticles synthesis in pH-sensitive nano-carriers produc-tion for doxorubicin delivery on MCF-7 breast cancer cell line. Int. J. Nanomed. 2019, 14, 3615. [Google Scholar]
- Fu, W.; Mohd Noor, M.H.; Yusof, L.M.P.; Ibrahim, T.A.T.; Keong, Y.S.; Jaji, A.Z.; Zakaria, M.Z.A.B. In vitro evaluation of a novel pH sensitive drug delivery system based cockle shell-derived aragonite nanoparticles against osteosarcoma. J. Exp. Nanosci. 2017, 12, 166–187. [Google Scholar] [CrossRef] [Green Version]
- Ibiyeye, K.M.; Idris, S.B.; Zuki, A.B.Z. Cockle shell-derived aragonite calcium carbonate nanoparticle for targeting cancer and breast cancer stem cells. Cancer Nanotechnol. 2020, 11, 1–15. [Google Scholar] [CrossRef]
- Danmaigoro, A.; Selvarajah, G.T.; Mohd Noor, M.H.; Mahmud, R.; Abu Bakar, M.Z. Toxicity and safety evaluation of doxorubicin-loaded cockle-shell-derived calcium carbonate nanoparticle in dogs. Adv. Pharmacol. Sci. 2018, 2018, 4848602. [Google Scholar]
- Wenliang, F.; Rameli, M.A.B.P.; Ibrahim, T.A.T.; Noor, M.H.M.; Yusof, L.M.; Zakaria, M.Z.A.B. In vivo evaluation of anticancer efficacy of drug loaded cockle shell-derived aragonite nanoparticles. J. Biomed. Mater. Res. Part B Appl. Biomater. 2019, 107, 1898–1907. [Google Scholar] [CrossRef] [PubMed]
- Ghaji, M.S.; Zakaria, Z.A.B.; Shameha, A.R.; Noor, M.H.M.; Hazilawati, H. Novel synthesis of nanoparticles from cockle shells via mechanical method for cytarabine drug release. J. Comput. Theor. Nanosci. 2018, 15, 1128–1136. [Google Scholar] [CrossRef]
- Som, A.; Raliya, R.; Tian, L.; Akers, W.; Ippolito, J.E.; Singamaneni, S.; Biswas, P.; Achilefu, S. Monodispersed calcium carbonate nanoparticles modulate local pH and inhibit tumor growth in vivo. Nanoscale 2016, 8, 12639–12647. [Google Scholar] [CrossRef] [Green Version]
- Kollenda, S.A.; Klose, J.; Knuschke, T.; Sokolova, V.; Schmitz, J.; Staniszewska, M.; Costa, P.F.; Herrmann, K.; Westendorf, A.M.; Fendler, W.P.; et al. In vivo biodistribution of calcium phosphate nanoparticles after intravascular, in-tramuscular, intratumoral, and soft tissue admin-istration in mice investigated by small animal PET/CT. Acta Biomater. 2020, 109, 244–253. [Google Scholar] [CrossRef]
- Ma, X.; Zhang, X.; Yang, L.; Wang, G.; Jiang, K.; Wu, G.; Cui, W.; Wei, Z. Tunable construction of multi-shelled hollow carbonate nanospheres and their potential ap-plications. Nanoscale 2016, 8, 8687–8695. [Google Scholar] [CrossRef] [PubMed]
- Guo, Y.; Zhang, J.; Jiang, L.; Shi, X.; Yang, L.; Fang, Q.; Fang, H.; Wang, K.; Jiang, K. Facile one-pot preparation of calcite mesoporous carrier for sustained and targeted drug re-lease for cancer cells. Chem. Commun. 2012, 48, 10636–10638. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Peer, D.; Karp, J.M.; Hong, S.; Farokhzad, O.C.; Margalit, R.; Langer, R. Nanocarriers as an emerging platform for cancer therapy. Nat. Nanotechnol. 2007, 2, 751–760. [Google Scholar] [CrossRef] [PubMed]
- Boyjoo, Y.; Pareek, V.K.; Liu, J. Synthesis of micro and nano-sized calcium carbonate particles and their applications. J. Mater. Chem. A 2014, 2, 14270–14288. [Google Scholar] [CrossRef]
- Krajewska, B. Urease-aided calcium carbonate mineralization for engineering applications: A review. J. Adv. Res. 2018, 13, 59–67. [Google Scholar] [CrossRef]
- Avaro, J.T.; Ruiz-Agudo, C.; Landwehr, E.; Hauser, K.; Gebauer, D. Impurity-free amorphous calcium carbonate, a preferential material for pharmaceutical and medical applications. Eur. J. Mineral. 2019, 31, 231–236. [Google Scholar] [CrossRef]
- Xu, H.; Peng, X.; Bai, S.; Ta, K.; Yang, S.; Liu, S.; Bin Jang, H.; Guo, Z. Precipitation of calcium carbonate mineral induced by viral lysis of cyanobacteria: Evidence from laboratory experiments. Biogeosciences 2019, 16, 949–960. [Google Scholar] [CrossRef] [Green Version]
- Martel, J.; Young, J.D.E. Purported nanobacteria in human blood as calcium carbonate nanoparticles. Proc. Natl. Acad. Sci. USA 2008, 105, 5549–5554. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Khouzani, M.F.; Chevrier, D.M.; Güttlein, P.; Hauser, K.; Zhang, P.; Hedin, N.; Gebauer, D. Disordered amorphous calcium carbonate from direct precipitation. CrystEngComm 2015, 17, 4842–4849. [Google Scholar] [CrossRef] [Green Version]
- Zelepukin, I.V.; Yaremenko, A.V.; Yuryev, M.V.; Mirkasymov, A.B.; Sokolov, I.L.; Deyev, S.M.; Nikitin, P.I.; Nikitin, M.P. Fast processes of nanoparticle blood clearance: Comprehensive study. J. Control. Release 2020, 326, 181–191. [Google Scholar] [CrossRef] [PubMed]
- Zyuzin, M.V.; Díez, P.; Goldsmith, M.; Carregal-Romero, S. Comprehensive and systematic analysis of the immunocompatibility of polyelectrolyte capsules. Bioconjug. Chem. 2017, 28, 556–564. [Google Scholar] [CrossRef]
- Qiu, N.; Yin, H.; Ji, B.; Klauke, N.; Glidle, A.; Zhang, Y.; Song, H.; Cai, L.; Ma, L.; Wang, G.; et al. Calcium carbonate microspheres as carriers for the anticancer drug camptothecin. Mater. Sci. Eng. C 2012, 32, 2634–2640. [Google Scholar] [CrossRef]
- Tang, H.; Guo, J.; Sun, Y.; Chang, B.; Ren, Q.; Yang, W. Facile synthesis of pH sensitive polymer-coated mesoporous silica nanoparticles and their application in drug delivery. Int. J. Pharm. 2011, 421, 388–396. [Google Scholar] [CrossRef] [PubMed]
- Matyszewska, D. Drug delivery systems in the transport of doxorubicin. Surf. Innov. 2014, 2, 201–210. [Google Scholar] [CrossRef]
- Wu, J.L.; Wang, C.Q.; Zhuo, R.X.; Cheng, S.X. Multi-drug delivery system based on alginate/calcium carbonate hybrid nanoparticles for combination chemotherapy. Colloids Surf. B 2014, 123, 498–505. [Google Scholar] [CrossRef]
- Sushko, A.D.; Yaminskii, I.V.; Gavryushina, E.S.; Drygin, Y.F. Release of ribonucleic acid from human common cold HRV 2 virus in acidic medium. Colloid J. 2010, 72, 559–564. [Google Scholar] [CrossRef]
- Swietach, P.; Vaughan-Jones, R.D.; Harris, A.L.; Hulikova, A. The chemistry, physiology and pathology of pH in cancer. Philos. Trans. R. Soc. B 2014, 369, 20130099. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Idris, S.B.; Arifah, A.K.; Jesse, F.F.; Ramanoon, S.Z.; Basit, M.A.; Zakaria, M.Z. In Vitro Cytotoxicity Evaluation of Oxytetracycline Loaded Cockle Shell Derived Calcium Carbonate Aragonite Nanoparticles. Nanosci. Nanotechnol.-Asia 2021, 11, 237–242. [Google Scholar] [CrossRef]
- Yamada, Y. Dimerization of Doxorubicin Causes Its Precipitation. ACS Omega 2020, 5, 33235–33241. [Google Scholar] [CrossRef] [PubMed]
- Thedrattanawong, C.; Manaspon, C.; Nasongkla, N. Controlling the burst release of doxorubicin from polymeric depots via ad-justing hydrophobic/hydrophilic properties. J. Drug Deliv. Sci. Technol. 2018, 46, 446–451. [Google Scholar] [CrossRef]
- King, H.D.; Dubowchik, G.M.; Mastalerz, H.; Willner, D.; Hofstead, S.J.; Firestone, R.A.; Lasch, S.J.; Trail, P.A. Monoclonal antibody conjugates of doxorubicin prepared with branched pep-tide linkers: Inhibition of aggregation by methoxytriethyleneglycol chains. J. Med. Chem. 2002, 45, 4336–4343. [Google Scholar] [CrossRef] [PubMed]











| # (Figure 4 Panel) | Sample | Special Conditions | D, nm | PDI |
|---|---|---|---|---|
| 1 (A) | CaNP1 | - | 339 ± 4 | 0.20 ± 0.01 |
| 2 (B) | CaNP2 | Solvent: isopropanol | 2637 ± 125 | 0.33 ± 0.05 |
| 3 (C) | CaNP3 | MgCl2, 0.01 M | 278 ± 4 | 0.15 ± 0.01 |
| 4 (D) | CaNP4 | DMEM, 2 w.% | 333 ± 2 | 0.26 ± 0.01 |
| 5 (E) | CaNP5 | MgCl2, 0.01 M; DMEM, 2 w.% | 326 ± 6 | 0.26 ± 0.01 |
| 6 (F) | CaNP6 | MgCl2, 0.05 M; DMEM, 2 w.% | 332 ± 1 | 0.19 ± 0.01 |
| 7 (G) | CaNP7 | MgCl2, 0.01 M; DMEM, 10 w.% | 249 ± 1 | 0.10 ± 0.01 |
| 8 (H) | CaNP8 | MgCl2, 0.05 M; DMEM, 10 w.% | 276 ± 4 | 0.10 ± 0.01 |
| Sample | IC50, µM |
|---|---|
| CaNP7–DOX | 0.97 ± 0.04 |
| DOX | 2.41 ± 0.02 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Popova, V.; Poletaeva, Y.; Pyshnaya, I.; Pyshnyi, D.; Dmitrienko, E. Designing pH-Dependent Systems Based on Nanoscale Calcium Carbonate for the Delivery of an Antitumor Drug. Nanomaterials 2021, 11, 2794. https://doi.org/10.3390/nano11112794
Popova V, Poletaeva Y, Pyshnaya I, Pyshnyi D, Dmitrienko E. Designing pH-Dependent Systems Based on Nanoscale Calcium Carbonate for the Delivery of an Antitumor Drug. Nanomaterials. 2021; 11(11):2794. https://doi.org/10.3390/nano11112794
Chicago/Turabian StylePopova, Victoriya, Yuliya Poletaeva, Inna Pyshnaya, Dmitrii Pyshnyi, and Elena Dmitrienko. 2021. "Designing pH-Dependent Systems Based on Nanoscale Calcium Carbonate for the Delivery of an Antitumor Drug" Nanomaterials 11, no. 11: 2794. https://doi.org/10.3390/nano11112794
APA StylePopova, V., Poletaeva, Y., Pyshnaya, I., Pyshnyi, D., & Dmitrienko, E. (2021). Designing pH-Dependent Systems Based on Nanoscale Calcium Carbonate for the Delivery of an Antitumor Drug. Nanomaterials, 11(11), 2794. https://doi.org/10.3390/nano11112794