A TiO2 Coated Carbon Aerogel Derived from Bamboo Pulp Fibers for Enhanced Visible Light Photo-Catalytic Degradation of Methylene Blue
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Synthesis of CA/TiO2
2.3. Characterization
2.4. The Adsorption Performance for MB
2.5. Photo-Catalytic Activity Test
3. Results and Discussion
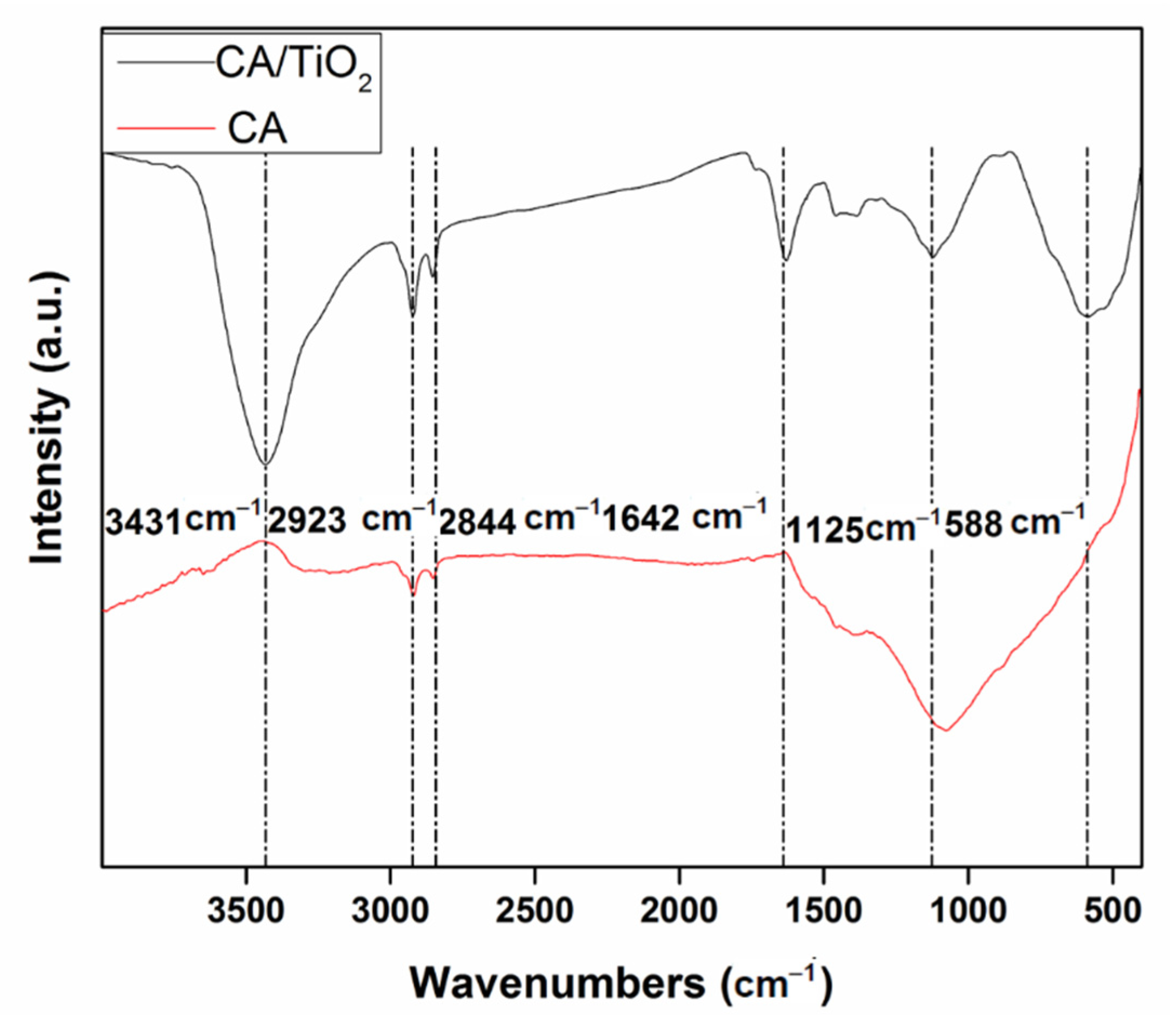
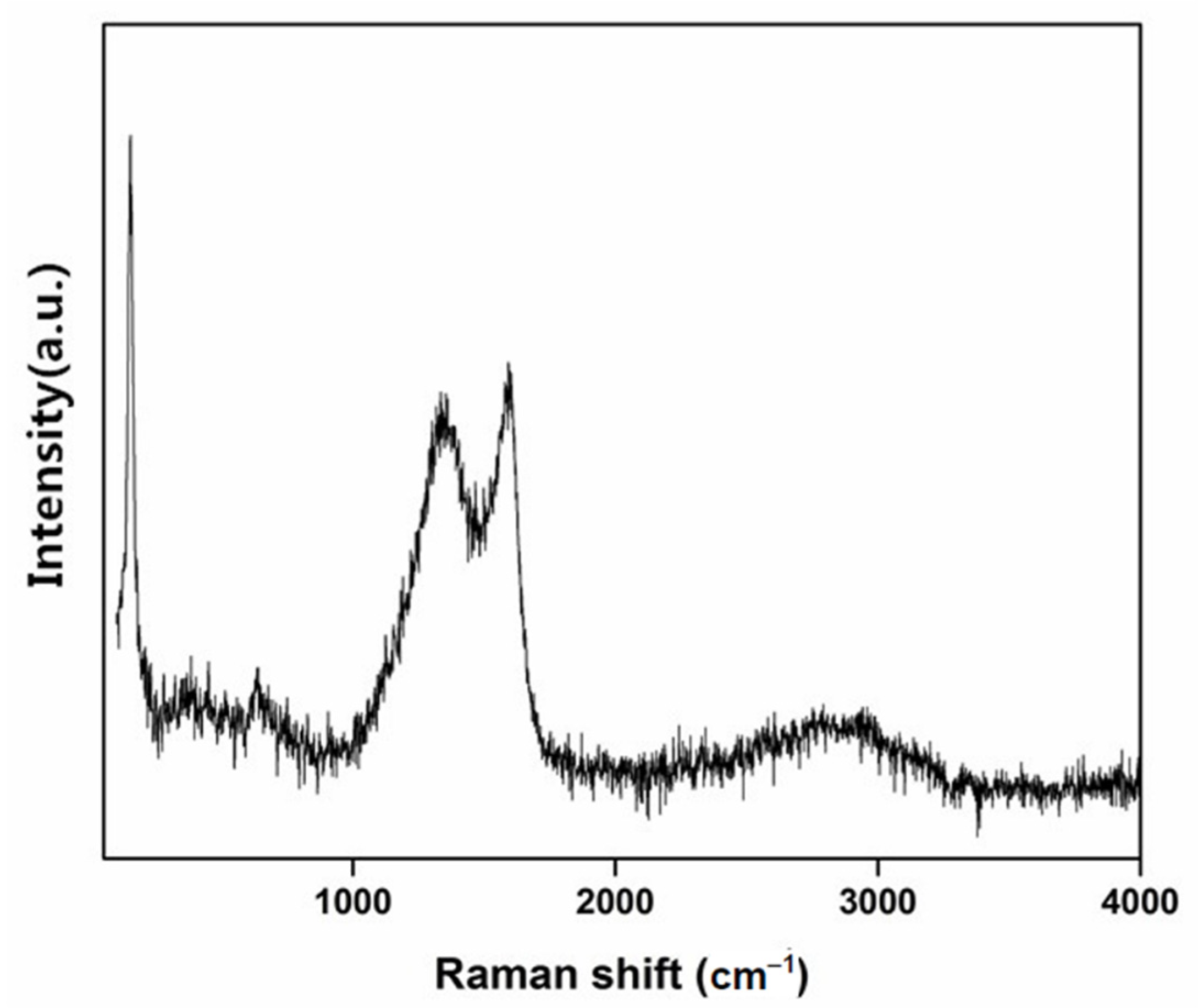
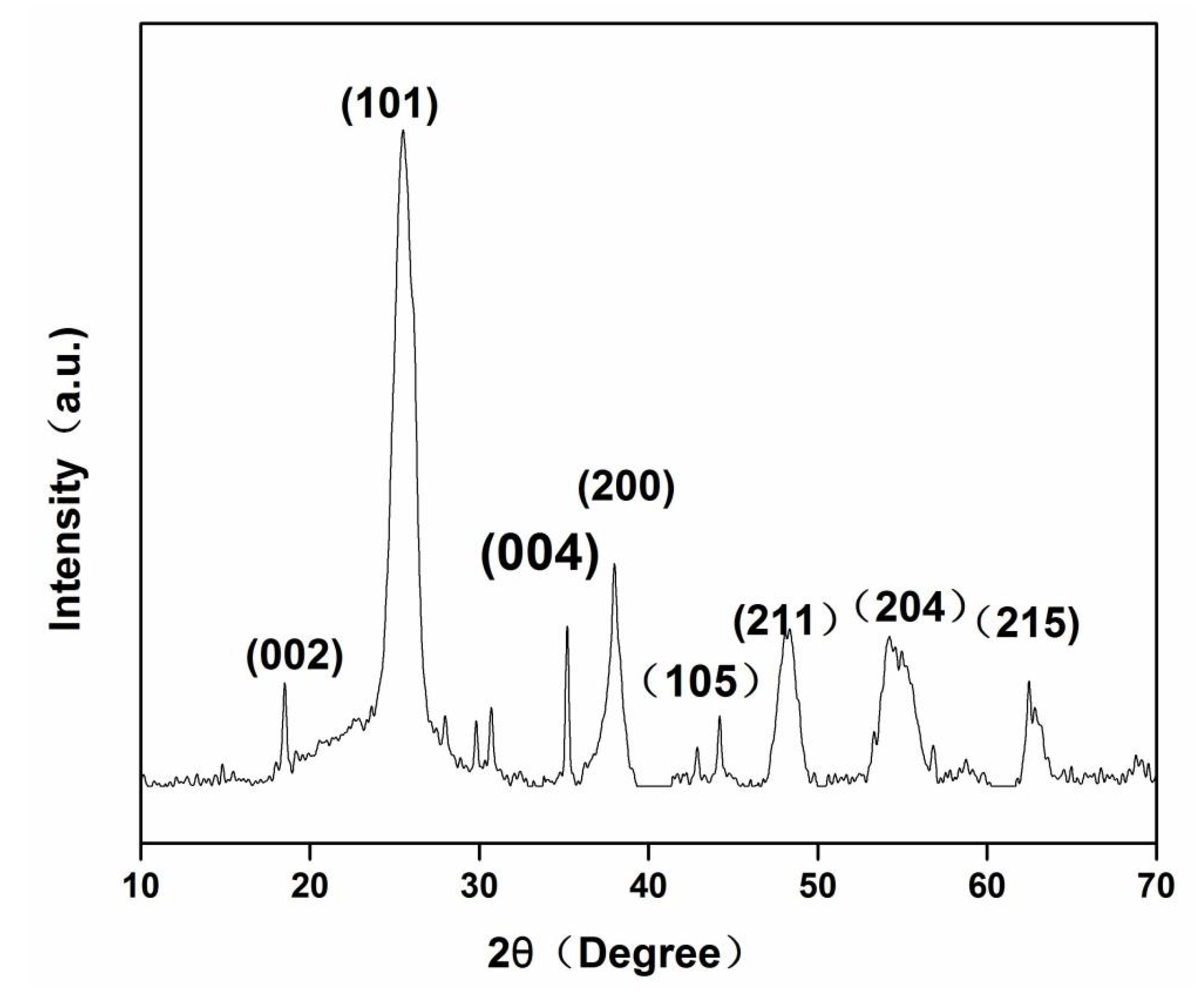
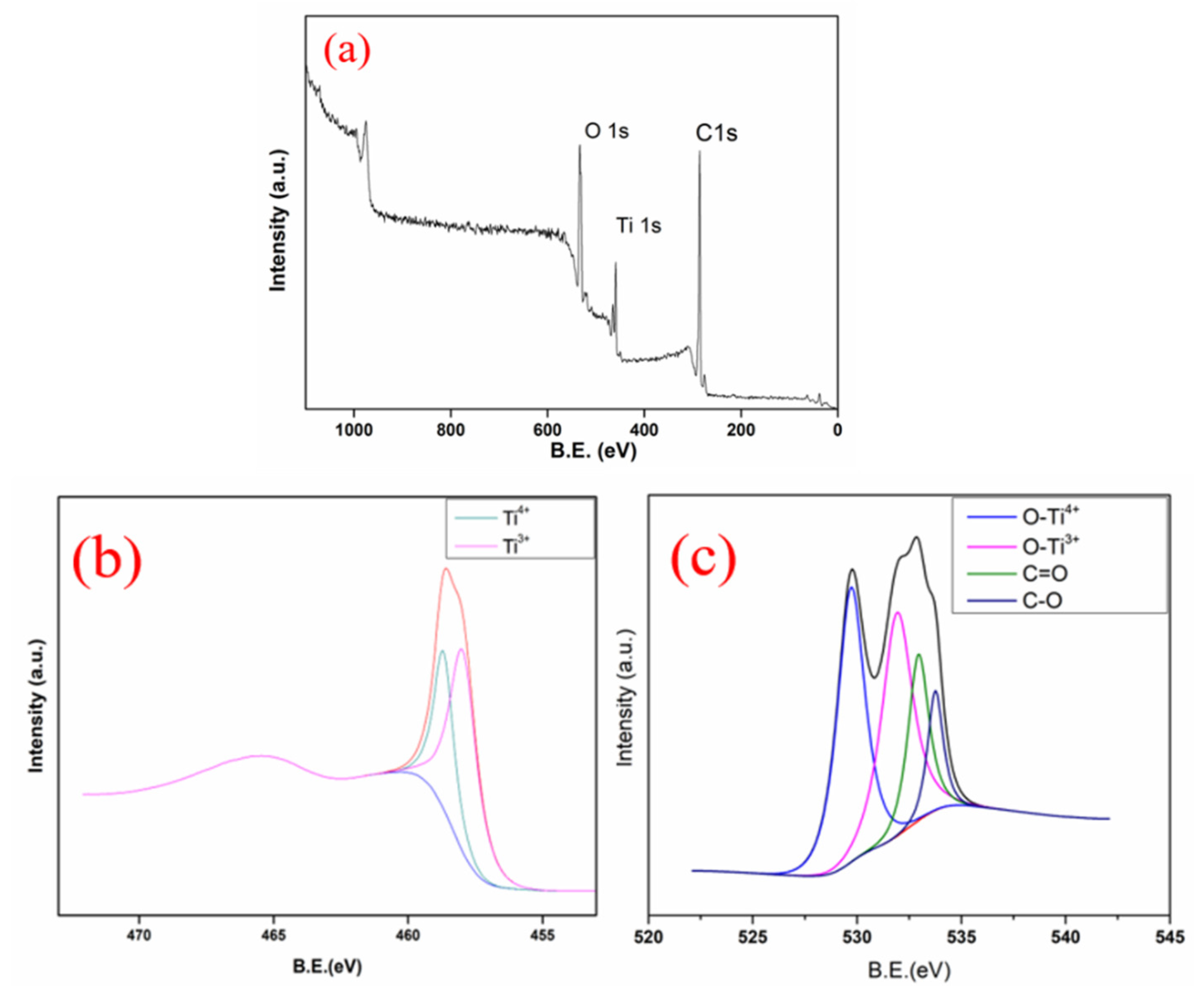
3.1. Characterization of Materials
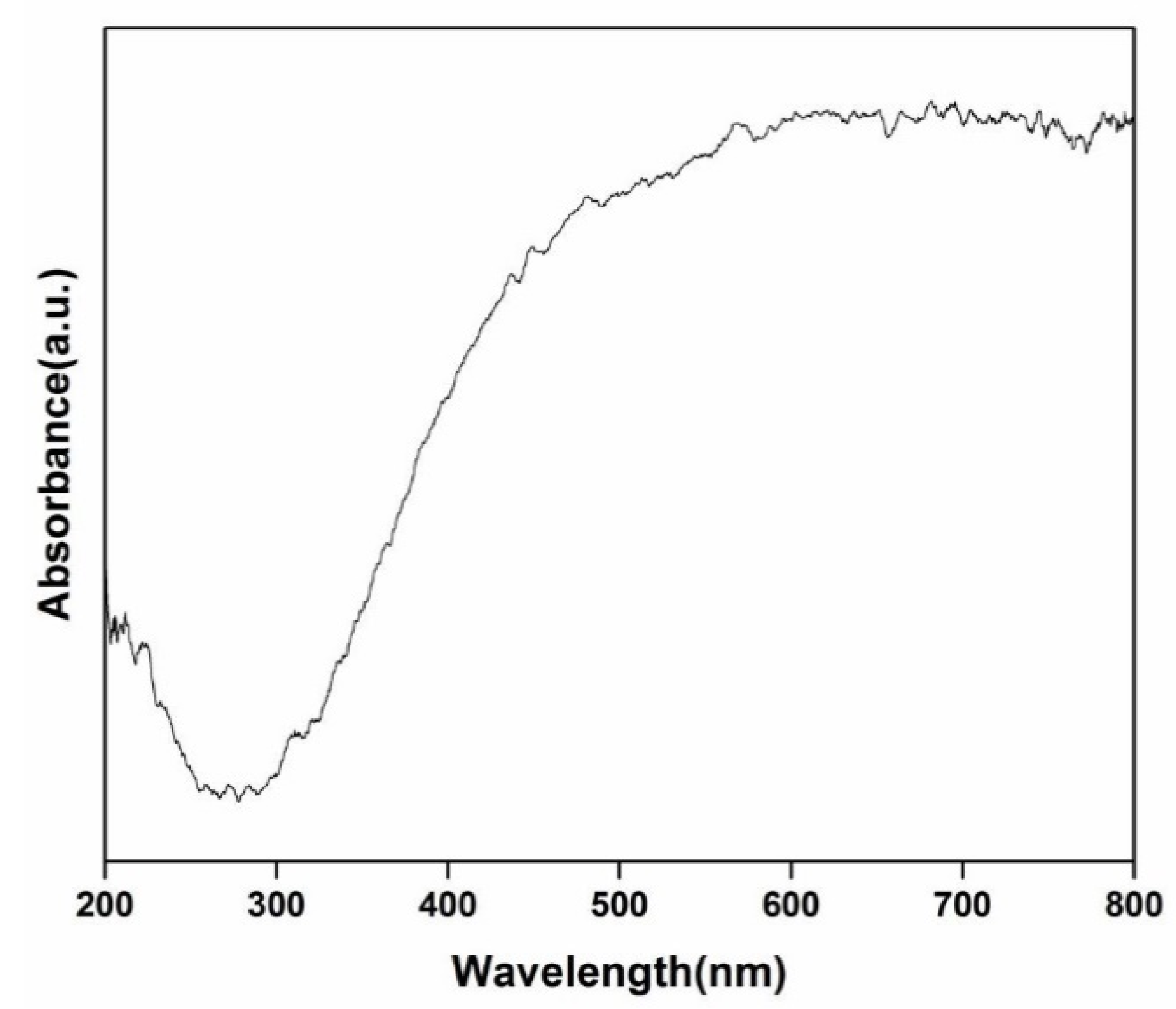
3.2. Optical Property
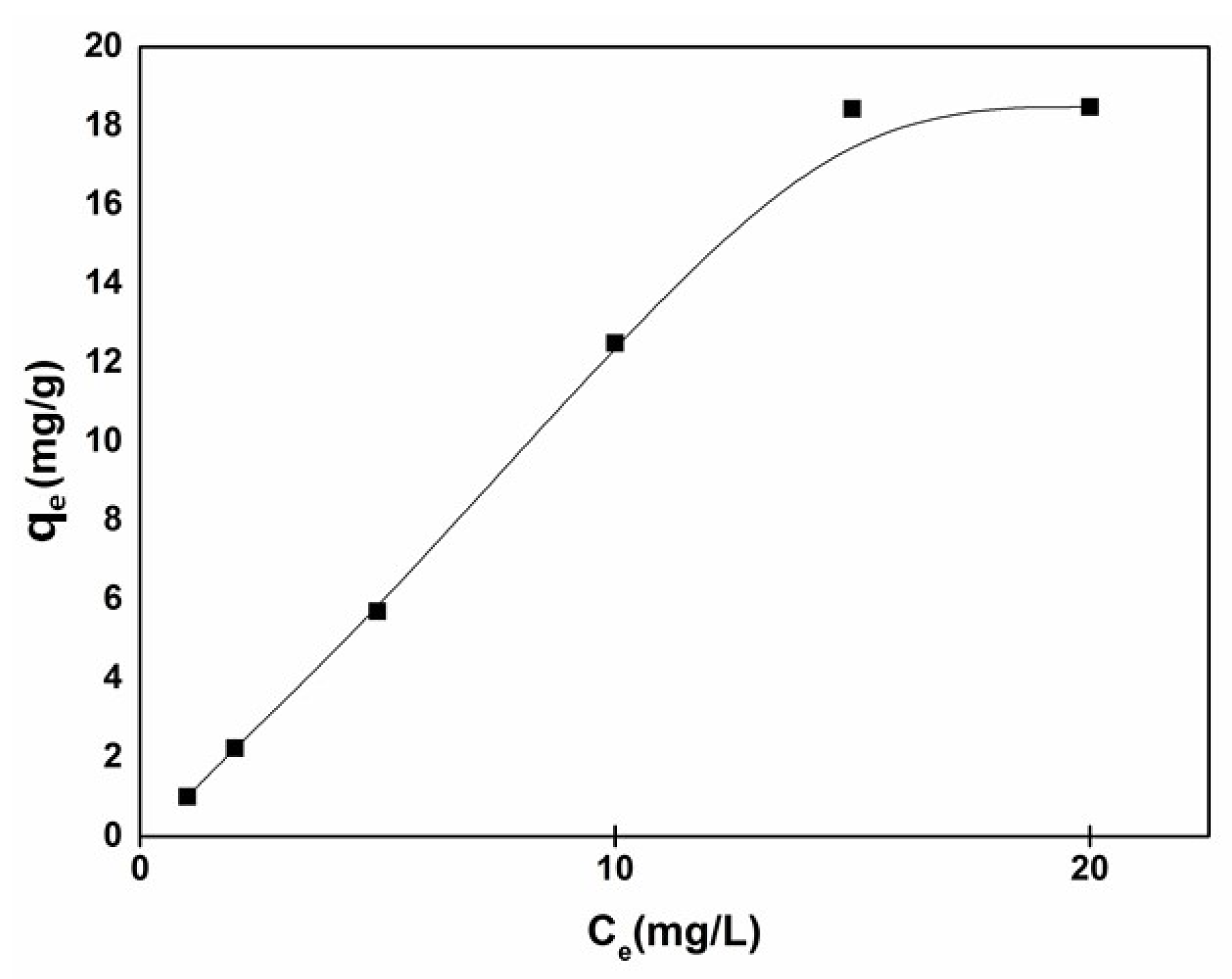
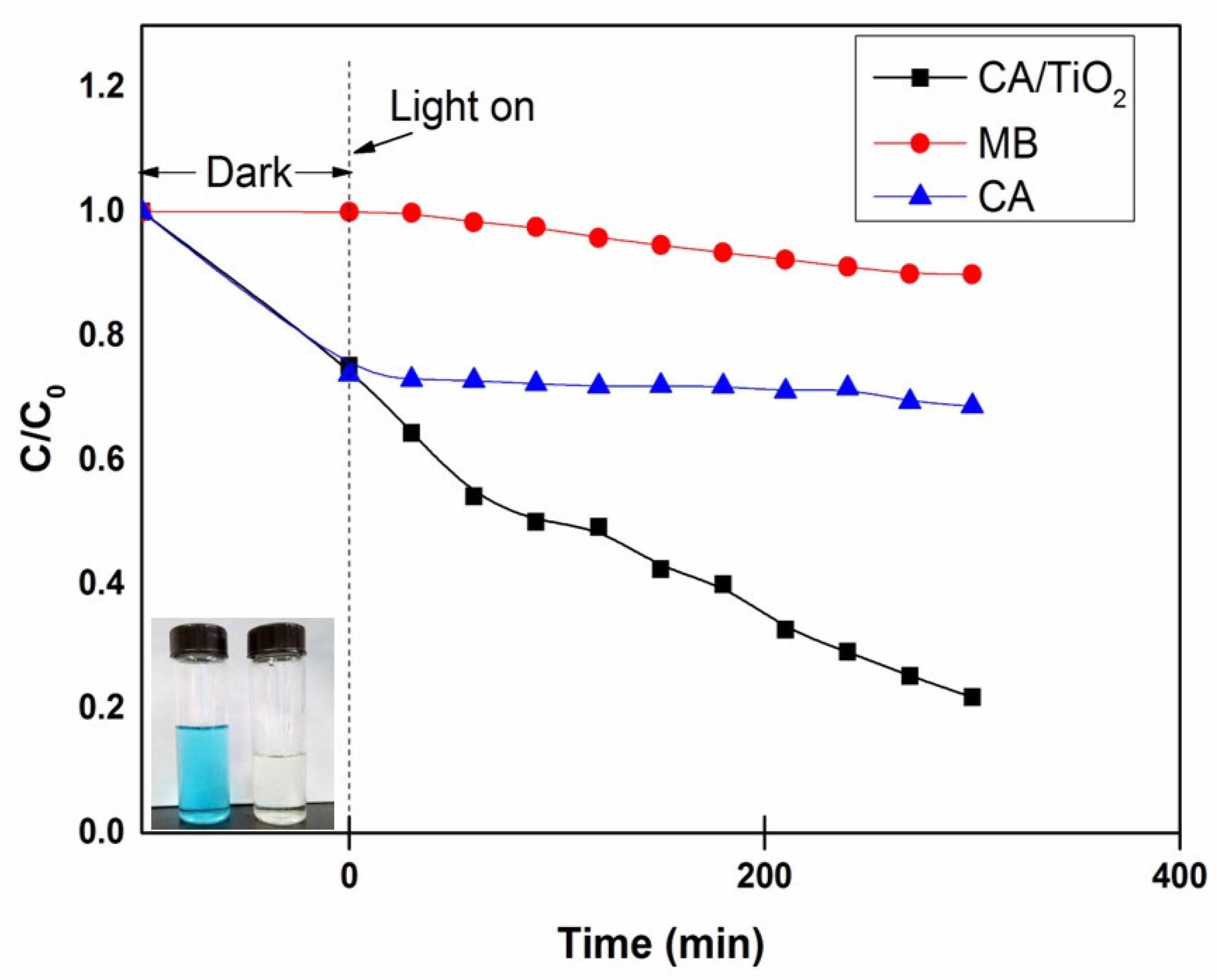
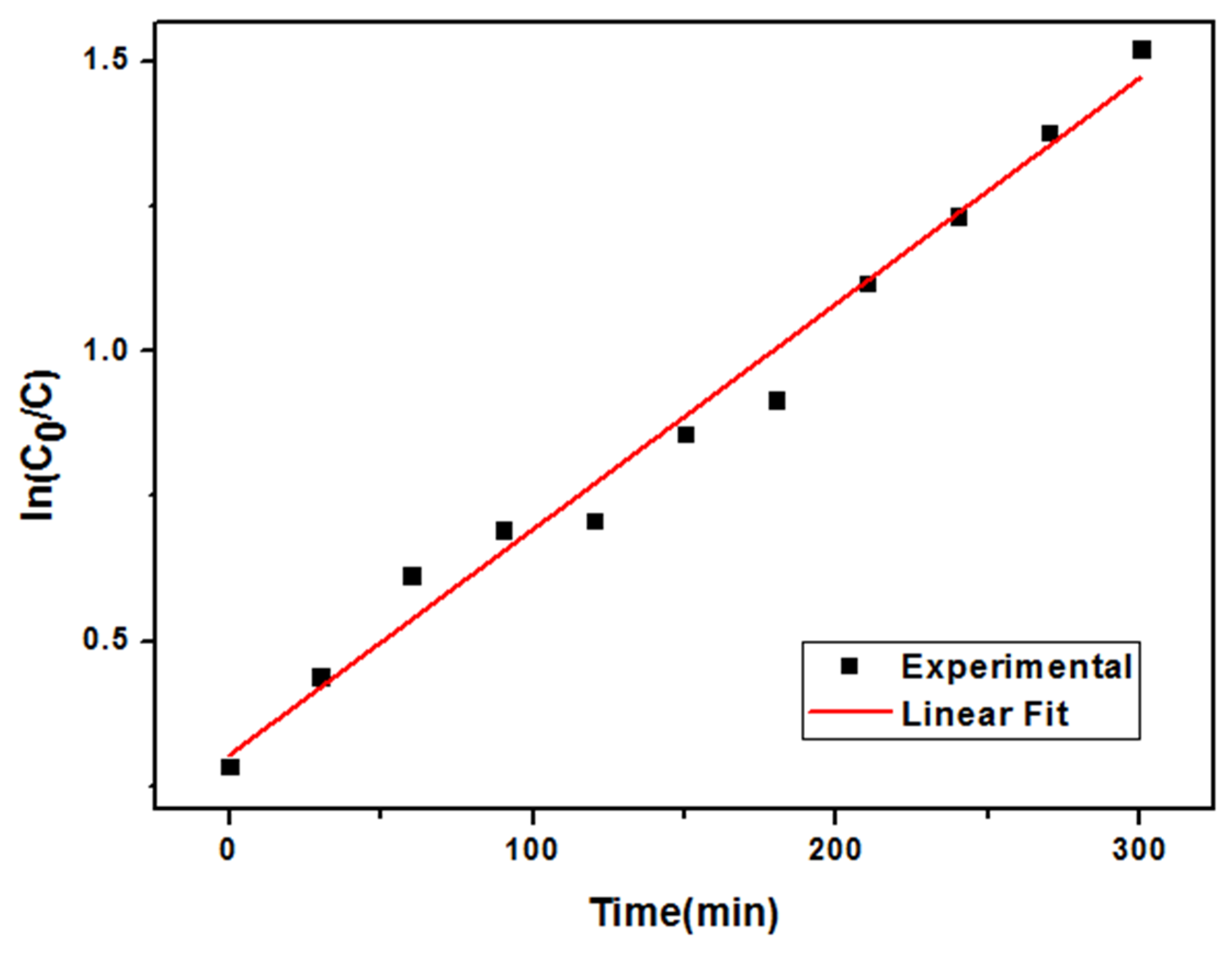
3.3. Adsorption and Photo-Catalytic Activity Study
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Rafatullah, M.; Sulaiman, O.; Hashim, R.; Ahmad, A. Adsorption of methylene blue on low-cost adsorbents: A review. J. Hazard. Mater. 2010, 177, 70–80. [Google Scholar] [CrossRef] [PubMed]
- Ghosh, D.; Bhattacharyya, K.G. Adsorption of methylene blue on kaolinite. Appl. Clay Sci. 2002, 20, 295–300. [Google Scholar] [CrossRef]
- Tan, I.A.W.; Ahmad, A.L.; Hameed, B.H. Adsorption of basic dye on high-surface-area activated carbon prepared from coconut husk: Equilibrium, kinetic and thermodynamic studies. J. Hazard. Mater. 2008, 154, 337–346. [Google Scholar] [CrossRef] [PubMed]
- Tan, I.A.W.; Ahmad, A.L.; Hameed, B.H. Adsorption of basic dye using activated carbon prepared from oil palm shell: Batch and fixed bed studies. Desalination 2008, 225, 13–28. [Google Scholar] [CrossRef]
- Abbasi, M.; Asl, N.R. Sonochemical degradation of Basic Blue 41 dye assisted by nanoTiO2 and H2O2. J. Hazard. Mater. 2008, 153, 942–947. [Google Scholar] [CrossRef] [PubMed]
- Wu, J.-S.; Liu, C.-H.; Chu, K.H.; Suen, S.-Y. Removal of cationic dye methyl violet 2B from water by cation exchange membranes. J. Membr. Sci. 2008, 309, 239–245. [Google Scholar] [CrossRef]
- Fan, L.; Zhou, Y.; Yang, W.; Chen, G.; Yang, F. Electrochemical degradation of aqueous solution of Amaranth azo dye on ACF under potentiostatic model. Dye. Pigment. 2008, 76, 440–446. [Google Scholar] [CrossRef]
- Zaghbani, N.; Hafiane, A.; Dhahbi, M. Removal of Safranin T from wastewater using micellar enhanced ultrafiltration. Desalination 2008, 222, 348–356. [Google Scholar] [CrossRef]
- Lodha, B.; Chaudhari, S. Optimization of Fenton-biological treatment scheme for the treatment of aqueous dye solutions. J. Hazard. Mater. 2007, 148, 459–466. [Google Scholar] [CrossRef]
- Zhu, M.-X.; Lee, L.; Wang, H.-H.; Wang, Z. Removal of an anionic dye by adsorption/precipitation processes using alkaline white mud. J. Hazard. Mater. 2007, 149, 735–741. [Google Scholar] [CrossRef]
- Lin, Y.; Li, D.; Hu, J.; Xiao, G.; Wang, J.; Li, W.; Fu, X. Highly Efficient Photocatalytic Degradation of Organic Pollutants by PANI-Modified TiO2 Composite. J. Phys. Chem. C 2012, 116, 5764–5772. [Google Scholar] [CrossRef]
- Huang, Y.; Ho, S.; Lu, Y.; Niu, R.; Xu, L.; Cao, J.; Lee, S. Removal of Indoor Volatile Organic Compounds via Photocatalytic Oxidation: A Short Review and Prospect. Molecules 2016, 21, 56. [Google Scholar] [CrossRef] [PubMed]
- Herney-Ramirez, J.; Vicente, M.A.; Madeira, L.M. Heterogeneous photo-Fenton oxidation with pillared clay-based catalysts for wastewater treatment: A review. Appl. Catal. B Environ. 2010, 98, 10–26. [Google Scholar] [CrossRef]
- Asahi, R.; Morikawa, T.; Irie, H.; Ohwaki, T. Nitrogen-Doped Titanium Dioxide as Visible-Light-Sensitive Photocatalyst: Designs, Developments, and Prospects. Chem. Rev. 2014, 114, 9824–9852. [Google Scholar] [CrossRef]
- Asahi, R.; Morikawa, T.; Ohwaki, T.; Aoki, K.; Taga, Y. Visible-Light Photocatalysis in Nitrogen-Doped Titanium Oxides. Science 2001, 293, 269–271. [Google Scholar] [CrossRef]
- Han, C.; Pelaez, M.; Likodimos, V.; Kontos, A.G.; Falaras, P.; O’Shea, K.; Dionysiou, D.D. Innovative visible light-activated sulfur doped TiO2 films for water treatment. Appl. Catal. B Environ. 2011, 107, 77–87. [Google Scholar] [CrossRef]
- Dong, F.; Guo, S.; Wang, H.; Li, X.; Wu, Z. Enhancement of the Visible Light Photocatalytic Activity of C-Doped TiO2 Nanomaterials Prepared by a Green Synthetic Approach. J. Phys. Chem. C 2011, 115, 13285–13292. [Google Scholar] [CrossRef]
- Su, W.; Zhang, Y.; Li, Z.; Wu, L.; Wang, X.; Li, J.; Fu, X. Multivalency Iodine Doped TiO2: Preparation, Characterization, Theoretical Studies, and Visible-Light Photocatalysis. Langmuir 2008, 24, 3422–3428. [Google Scholar] [CrossRef]
- Yu, J.C.; Yu, J.G.; Ho, W.; Jiang, Z.T.; Zhang, L.Z. Effects of F- Doping on the Photo-catalytic Activity and Microstructures of Nanocrystalline TiO2 Powders. Chem. Mater. 2002, 14, 3808–3816. [Google Scholar] [CrossRef]
- Qi, D.; Xing, M.; Zhang, J. Hydrophobic Carbon-Doped TiO2/MCF-F Composite as a High Performance Photocatalyst. J. Phys. Chem. C 2014, 118, 7329–7336. [Google Scholar] [CrossRef]
- Huang, J.F.; Liu, J.M.; Xiao, L.M. Facile synthesis of porous hybrid materials based on Calix-3 dye and TiO2 for high photocatalytic water splitting performance with excellent stability. J. Mater. Chem. A 2019, 7, 2993–2999. [Google Scholar] [CrossRef]
- Jiang, Z.; Zhu, C.; Wan, W.; Qian, K.; Xie, J. Constructing graphite-like carbon nitride modified hierarchical yolk-shell TiO2 spheres for water pollution treatment and hydrogen production. J. Mater. Chem. A 2016, 4, 1806–1818. [Google Scholar] [CrossRef]
- Zhang, Y.; Tang, Z.-R.; Fu, X.; Xu, Y.-J. Engineering the Unique 2D Mat of Graphene to Achieve Graphene-TiO2 Nanocomposite for Photocatalytic Selective Transformation: What Advantage does Graphene Have over Its Forebear Carbon Nanotube? ACS Nano 2011, 5, 7426–7435. [Google Scholar] [CrossRef] [PubMed]
- Cho, K.M.; Kim, K.H.; Choi, H.O.; Jung, H.-T. A highly photoactive, visible-light-driven graphene/2D mesoporous TiO2 photocatalyst. Green Chem. 2015, 17, 3972–3978. [Google Scholar] [CrossRef]
- Xing, M.; Li, X.; Zhang, J. Synergistic effect on the visible light activity of Ti3+ doped TiO2 nanorods/boron doped graphene composite. Sci. Rep. 2014, 4, 5493. [Google Scholar] [CrossRef] [PubMed]
- Tan, L.-L.; Ong, W.-J.; Chai, S.-P.; Mohamed, A.R. Reduced graphene oxide-TiO2 nanocomposite as a promising visible-light-active photocatalyst for the conversion of carbon dioxide. Nanoscale Res. Lett. 2013, 8, 465. [Google Scholar] [CrossRef]
- Cai, X.; Li, J.; Liu, Y. Titanium dioxide-coated biochar composites as adsorptive and photocatalytic degradation materials for the removal of aqueous organic pollutants. Chem. Technol. Biotechnol. 2018, 93, 783–791. [Google Scholar] [CrossRef]
- Lee, W.J.; Lee, J.M.; Kochuveedu, S.T.; Han, T.H.; Jeong, H.Y.; Park, M.; Yun, J.M.; Kwon, J.; No, K.; Kim, D.H.; et al. Biomineralized N-Doped CNT/TiO2 Core/Shell Nanowires for Visible Light Photocatalysis. ACS Nano 2012, 6, 935–943. [Google Scholar] [CrossRef]
- Reddy, K.R.; Hassan, M.; Gomes, V.G. Hybrid nanostructures based on titanium dioxide for enhanced photocatalysis. Appl. Catal. A Gen. 2015, 489, 1–16. [Google Scholar] [CrossRef]
- Zhao, L.; Chen, X.; Wang, X.; Zhang, Y.; Wei, W.; Sun, Y.; Antonietti, M.; Titirici, M.M. One-Step Solvothermal Synthesis of a Carbon@TiO2 Dyade Structure Effectively Promoting Visible-Light Photocatalysis. Adv. Mater. 2010, 22, 3317–3321. [Google Scholar] [CrossRef]
- Zhuang, J.; Tian, Q.; Zhou, H.; Liu, Q.; Liu, P.; Zhong, H. Hierarchical porous TiO2@C hollow microspheres: One-pot synthesis and enhanced visible-light photocatalysis. J. Mater. Chem. 2012, 22, 7036–7042. [Google Scholar] [CrossRef]
- Zhao, D.; Sheng, G.; Chen, C.; Wang, X. Enhanced photocatalytic degradation of methylene blue under visible irradiation on graphene@TiO2 dyade structure. Appl. Catal. B Environ. 2012, 111–112, 303–308. [Google Scholar] [CrossRef]
- Tan, L.-L.; Ong, W.-J.; Chai, S.-P.; Goh, B.T.; Mohamed, A.R. Visible-light-active oxygen-rich TiO2 decorated 2D graphene oxide with enhanced photocatalytic activity toward carbon dioxide reduction. Appl. Catal. B Environ. 2015, 179, 160–170. [Google Scholar] [CrossRef]
- Li, X.; Jiang, Y.; Cheng, W.; Li, Y.; Xu, X.; Lin, K. Mesoporous TiO2/Carbon Beads: One-Pot Preparation and Their Application in Visible-Light-Induced Photodegradation. Nano-Micro Lett. 2015, 7, 243–254. [Google Scholar] [CrossRef] [PubMed]
- Bailón-García, E.; Elmouwahidi, A.; Álvarez, M.A.; Carrasco-Marín, F.; Pérez-Cadenas, A.F.; Maldonado-Hódar, F.J. New carbon xerogel-TiO2 composites with high performance as visible-light photocatalysts for dye mineralization. Appl. Catal. B Environ. 2017, 201, 29–40. [Google Scholar] [CrossRef]
- Moreno-Castilla, C.; Maldonado-Hódar, F.J. Carbon aerogels for catalysis applications: An overview. Carbon 2005, 43, 455–465. [Google Scholar] [CrossRef]
- Yuan, W.; Zhang, X.; Zhao, J.; Li, Q.; Ao, C.; Xia, T.; Zhang, W.; Lu, C. Ultra-lightweight and highly porous carbon aerogels from bamboo pulp fibers as an effective sorbent for water treatment. Results Phys. 2017, 7, 2919–2924. [Google Scholar] [CrossRef]
- Li, Y.-Q.; Samad, Y.A.; Polychronopoulou, K.; Alhassan, S.M.; Liao, K. Carbon Aerogel from Winter Melon for Highly Efficient and Recyclable Oils and Organic Solvents Absorption. ACS Sustain. Chem. Eng. 2014, 2, 1492–1497. [Google Scholar] [CrossRef]
- Han, S.; Sun, Q.; Zheng, H.; Li, J.; Jin, C. Green and facile fabrication of carbon aerogels from cellulose-based waste newspaper for solving organic pollution. Carbohydr. Polym. 2016, 136, 95–100. [Google Scholar] [CrossRef]
- He, D.; Li, Y.; Wang, I.; Wu, J.; Yang, Y.; An, Q. Carbon wrapped and doped TiO2 mesoporous nanostructure with efficient visible-light photocatalysis for NO removal. Appl. Surf. Sci. 2017, 391, 318–325. [Google Scholar] [CrossRef]
- Kataoka, S.; Tejedor-Tejedor, M.I.; Coronado, J.M.; Anderson, M.A. Thin-film transmission IR spectroscopy as an in situ probe of the gas–solid interface in photocatalytic processes. J. Photochem. Photobiol. A Chem. 2004, 163, 323–329. [Google Scholar] [CrossRef]
- Sang, Y.; Zhao, Z.; Tian, J.; Hao, P.; Jiang, H.; Liu, H.; Claverie, J.P. Enhanced Photocatalytic Property of Reduced Graphene Oxide/TiO2 Nanobelt Surface Heterostructures Constructed by an In Situ Photochemical Reduction Method. Small 2014, 10, 3775–3782. [Google Scholar] [CrossRef] [PubMed]
- Yu, X.; Liu, J.; Yu, Y.; Zuo, S.; Li, B. Preparation and visible light photocatalytic activity of carbon quantum dots/TiO2 nanosheet composites. Carbon 2014, 68, 718–724. [Google Scholar] [CrossRef]
- Guo, S.; Zhang, G.; Guo, Y.; Yu, J.C. Graphene oxide–Fe2O3 hybrid material as highly efficient heterogeneous catalyst for degradation of organic contaminants. Carbon 2013, 60, 437–444. [Google Scholar] [CrossRef]
- Marie, J.; Berthon-Fabry, S.; Chatenet, M.; Chainet, E.; Pirard, R.; Cornet, N.; Achard, P. Platinum supported on resorcinol–formaldehyde based carbon aerogels for PEMFC electrodes: Influence of the carbon support on electrocatalytic properties. J. Appl. Electrochem. 2007, 37, 147–153. [Google Scholar] [CrossRef]
- Wiener, M.; Reichenauer, G.; Braxmeier, S.; Hemberger, F.; Ebert, H.-P. Carbon Aerogel-Based High-Temperature Thermal Insulation. Int. J. Thermophys. 2009, 30, 1372–1385. [Google Scholar] [CrossRef]
- Bock, V.; Nilsson, O.; Blumm, J.; Fricke, J. Thermal properties of carbon aerogels. J. Non-Cryst. Solids 1995, 185, 233–239. [Google Scholar] [CrossRef]
- Hanaor, D.A.H.; Sorrell, C.C. Review of the anatase to rutile phase transformation. J. Mater. Sci. 2011, 46, 855–874. [Google Scholar] [CrossRef]
- Maldonado-Hódar, F.J.; Moreno-Castilla, C.; Rivera-Utrilla, J. Synthesis, pore texture and surface acid–base character of TiO2/carbon composite xerogels and aerogels and their carbonized derivatives. Appl. Catal. A Gen. 2000, 203, 151–159. [Google Scholar] [CrossRef]
- Moreno-Castilla, C.; Maldonado-Hodar, F.J. Synthesis and surface characteristics of silica- and alumina-carbon composite xerogels. Phys. Chem. Chem. Phys. 2000, 2, 4818–4822. [Google Scholar] [CrossRef]
- Gonzalez-Elipe, A.R.; Malet, P.; Espinos, J.P.; Caballero, A.; Munuera, G. Effect of Water in the Encapsulation of the Metallic Phase During Smsi Generation in Pt/TiO2 Catalysts. Struct. React. Surf. 1989, 48, 427–436. [Google Scholar] [CrossRef]
- González-Elipe, A.R.; Fernández, A.; Espinós, J.P.; Munuera, G. Role of hydrogen in the mobility of phases in Ni TiOx systems. J. Catal. 1991, 131, 51–59. [Google Scholar] [CrossRef]
- Pastrana-Martínez, L.M.; Morales-Torres, S.; Likodimos, V.; Falaras, P.; Figueiredo, J.L.; Faria, J.L.; Silva, A.M.T. Role of oxygen functionalities on the synthesis of photocatalytically active graphene–TiO2 composites. Appl. Catal. B Environ. 2014, 158–159, 329–340. [Google Scholar] [CrossRef]
- Wang, B.; Karthikeyan, R.; Lu, X.-Y.; Xuan, J.; Leung, M.K.H. High photocatalytic activity of immobilized TiO2 nanorods on carbonized cotton fibers. J. Hazard. Mater. 2013, 263, 659–669. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.; Wu, Z.; Liu, Y. A Simple Two-Step Template Approach for Preparing Carbon-Doped Mesoporous TiO2 Hollow Microspheres. J. Phys. Chem. C 2009, 113, 13317–13324. [Google Scholar] [CrossRef]
- Zhang, Y.; Zhao, Z.; Chen, J.; Cheng, L.; Chang, J.; Sheng, W.; Hu, C.; Cao, S. C-doped hollow TiO2 spheres: In situ synthesis, controlled shell thickness, and superior visible-light photocatalytic activity. Appl. Catal. B Environ. 2015, 165, 715–722. [Google Scholar] [CrossRef]
- Monticone, S.; Tufeu, R.; Kanaev, A.V.; Scolan, E.; Sanchez, C. Quantum size effect in TiO2 nanoparticles: Does it exist? Appl. Surf. Sci. 2000, 162–163, 565–570. [Google Scholar] [CrossRef]
- Moya, A.; Cherevan, A.; Marchesan, S.; Gebhardt, P.; Prato, M.; Eder, D.; Vilatela, J.J. Oxygen vacancies and interfaces enhancing photocatalytic hydrogen production in mesoporous CNT/TiO2 hybrids. Appl. Catal. B Environ. 2015, 179, 574–582. [Google Scholar] [CrossRef]
- Zhang, G.; Teng, F.; Zhao, C.; Chen, L.; Zhang, P.; Wang, Y.; Gong, C.; Zhang, Z.; Xie, E. Enhanced photocatalytic activity of TiO2/carbon@TiO2 core–shell nanocomposite prepared by two-step hydrothermal method. Appl. Surf. Sci. 2014, 311, 384–390. [Google Scholar] [CrossRef]
- Chen, D.; Jiang, Z.; Geng, J.; Wang, A.Q.; Yang, D. Carbon and Nitrogen Co-doped TiO2 with Enhanced Visible-Light Photocatalytic Activity. Huazhong Norm. Univ. J. Postgrad. 2010, 46, 2741–2746. [Google Scholar] [CrossRef]

Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhang, J.; Yuan, W.; Xia, T.; Ao, C.; Zhao, J.; Huang, B.; Wang, Q.; Zhang, W.; Lu, C. A TiO2 Coated Carbon Aerogel Derived from Bamboo Pulp Fibers for Enhanced Visible Light Photo-Catalytic Degradation of Methylene Blue. Nanomaterials 2021, 11, 239. https://doi.org/10.3390/nano11010239
Zhang J, Yuan W, Xia T, Ao C, Zhao J, Huang B, Wang Q, Zhang W, Lu C. A TiO2 Coated Carbon Aerogel Derived from Bamboo Pulp Fibers for Enhanced Visible Light Photo-Catalytic Degradation of Methylene Blue. Nanomaterials. 2021; 11(1):239. https://doi.org/10.3390/nano11010239
Chicago/Turabian StyleZhang, Jian, Wei Yuan, Tian Xia, Chenghong Ao, Jiangqi Zhao, Bingxue Huang, Qunhao Wang, Wei Zhang, and Canhui Lu. 2021. "A TiO2 Coated Carbon Aerogel Derived from Bamboo Pulp Fibers for Enhanced Visible Light Photo-Catalytic Degradation of Methylene Blue" Nanomaterials 11, no. 1: 239. https://doi.org/10.3390/nano11010239
APA StyleZhang, J., Yuan, W., Xia, T., Ao, C., Zhao, J., Huang, B., Wang, Q., Zhang, W., & Lu, C. (2021). A TiO2 Coated Carbon Aerogel Derived from Bamboo Pulp Fibers for Enhanced Visible Light Photo-Catalytic Degradation of Methylene Blue. Nanomaterials, 11(1), 239. https://doi.org/10.3390/nano11010239






