Inorganic and Polymeric Nanoparticles for Human Viral and Bacterial Infections Prevention and Treatment
Abstract
1. Introduction
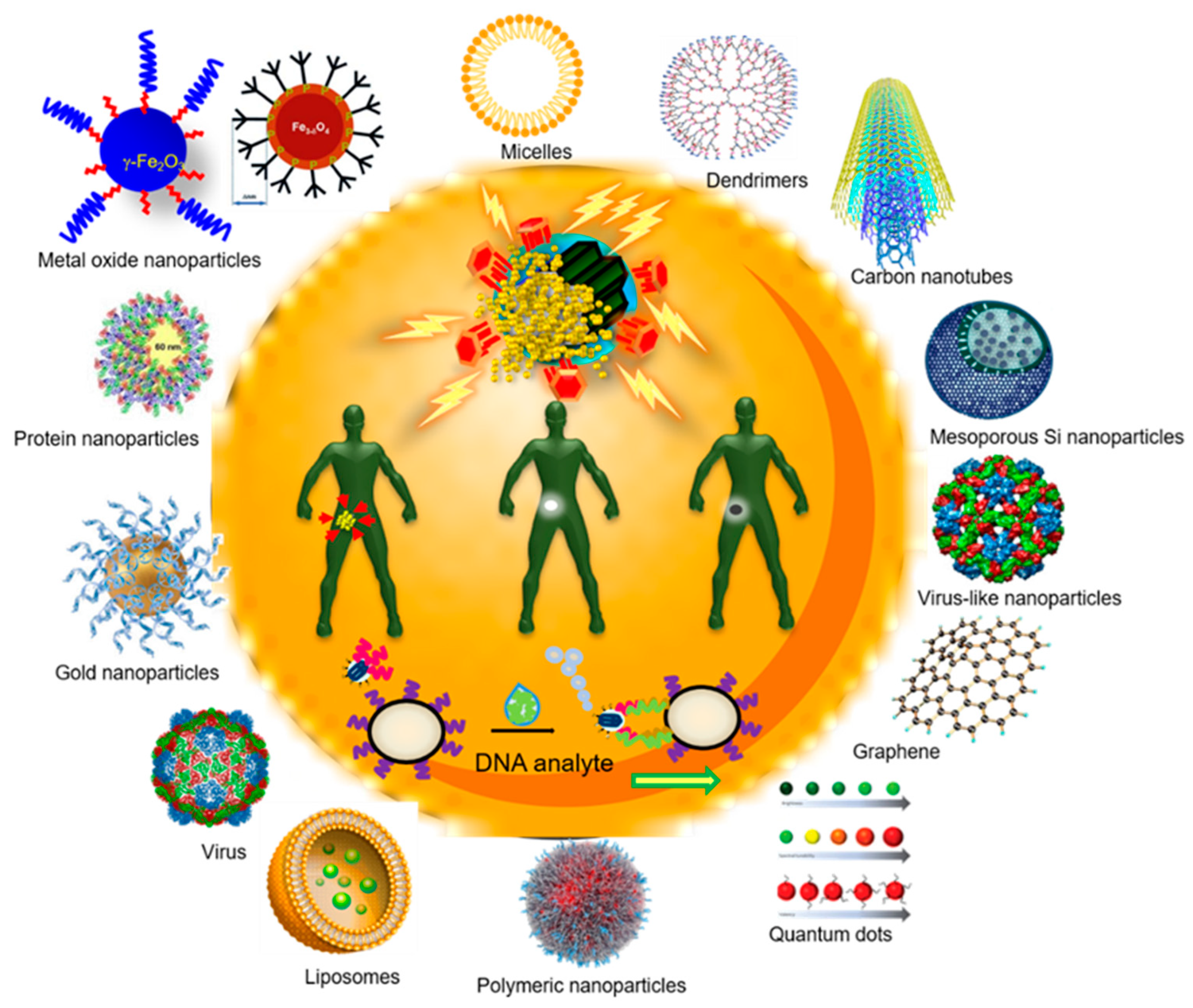
2. Nanoparticles Types and Their Uses
2.1. Inorganic Nanoparticles
2.2. Polymeric Nanoparticles
3. Nanoparticles and Human Infections
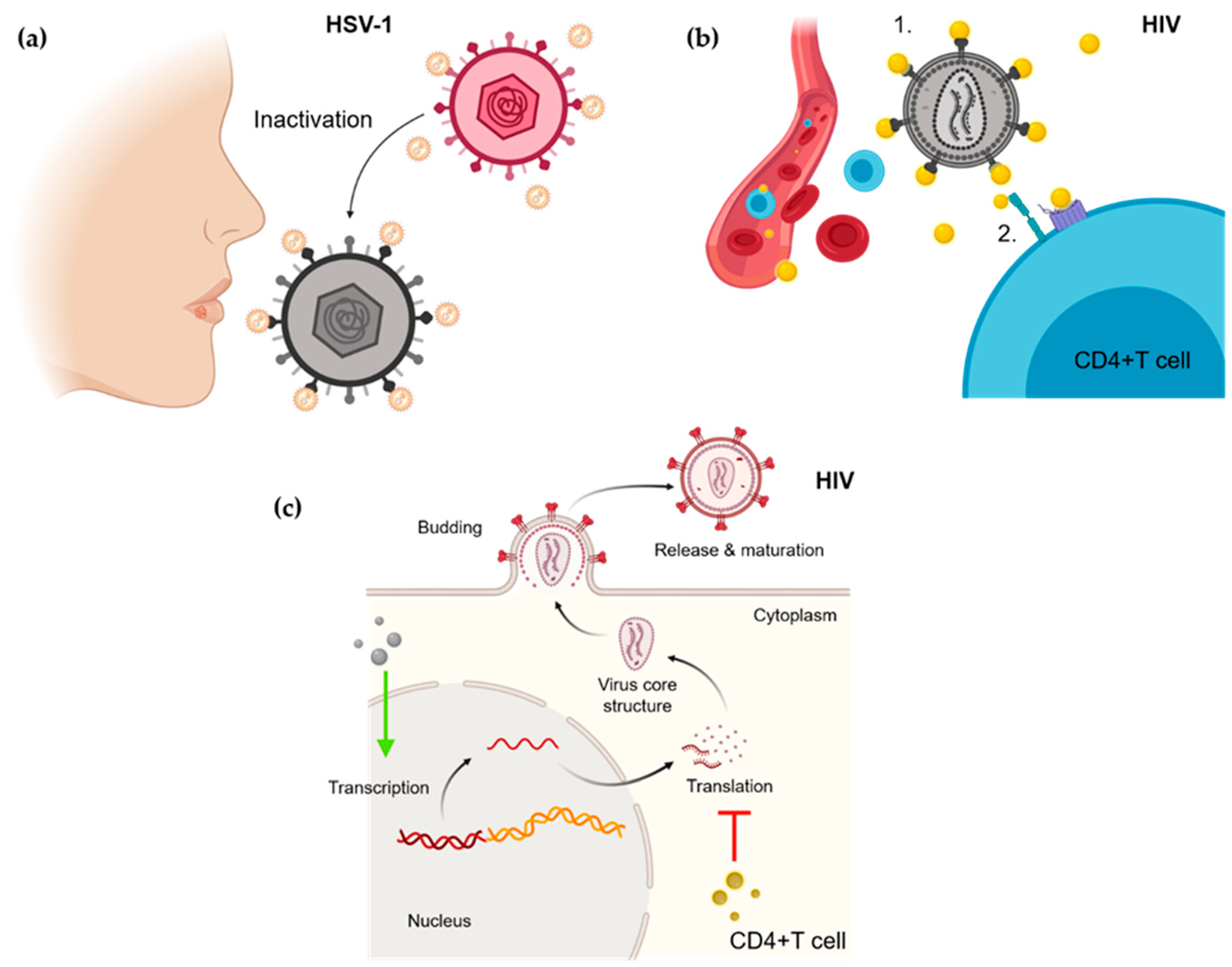
3.1. Nanoparticles and Human Viral Infections
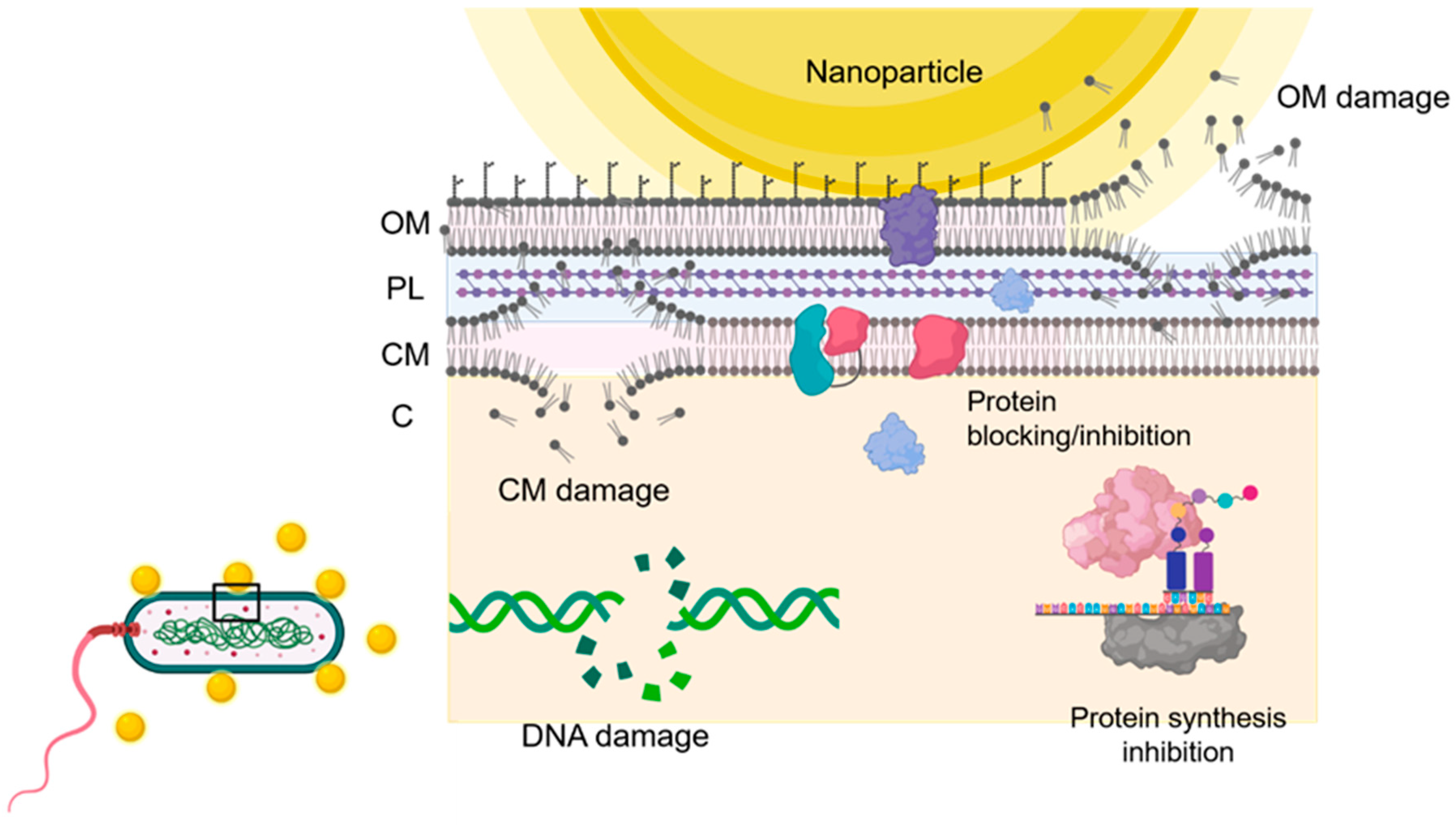
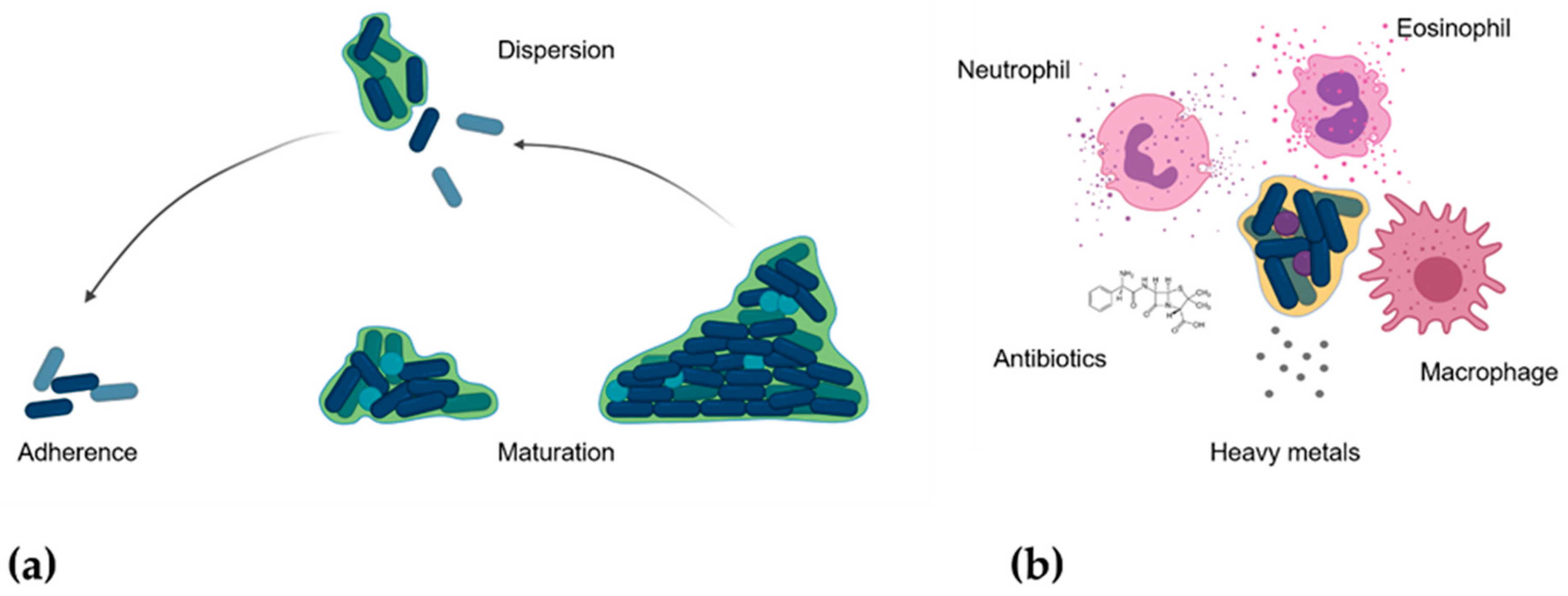
3.2. Nanoparticles and Human Bacterial Infections
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Bloom, D.E.; Cadarette, D. Infectious Disease Threats in the Twenty-First Century: Strengthening the Global Response. Front. Immunol. 2019, 10, 549. [Google Scholar] [CrossRef] [PubMed]
- The Top 10 Causes of Death. Available online: https://www.who.int/news-room/fact-sheets/detail/the-top-10-causes-of-death (accessed on 2 November 2020).
- Christiansen, J. Global Infections by the Numbers. Available online: https://www.scientificamerican.com/article/global-infections-by-the-numbers/ (accessed on 2 November 2020).
- Michael, C.A.; Dominey-Howes, D.; Labbate, M. The Antimicrobial Resistance Crisis: Causes, Consequences, and Management. Front. Public Health 2014, 2, 145. [Google Scholar] [CrossRef] [PubMed]
- Uchil, R.R.; Kohli, G.S.; Katekhaye, V.M.; Swami, O.C. Strategies to Combat Antimicrobial Resistance. J. Clin. Diagn. Res. 2014, 8, ME01-04. [Google Scholar] [CrossRef] [PubMed]
- Tinkle, S.; McNeil, S.E.; Mühlebach, S.; Bawa, R.; Borchard, G.; Barenholz, Y.C.; Tamarkin, L.; Desai, N. Nanomedicines: Addressing the Scientific and Regulatory Gap: Nanomedicines. Ann. N. Y. Acad. Sci. 2014, 1313, 35–56. [Google Scholar] [CrossRef] [PubMed]
- Soares, S.; Sousa, J.; Pais, A.; Vitorino, C. Nanomedicine: Principles, Properties, and Regulatory Issues. Front. Chem. 2018, 6, 360. [Google Scholar] [CrossRef] [PubMed]
- Hua, S.; Wu, S.Y. Editorial: Advances and Challenges in Nanomedicine. Front. Pharmacol. 2018, 9, 1397. [Google Scholar] [CrossRef] [PubMed]
- Manzano, M.; Vallet-Regí, M. Mesoporous Silica Nanoparticles for Drug Delivery. Adv. Funct. Mater. 2020, 30, 1902634. [Google Scholar] [CrossRef]
- Vallet-Regí, M.; Balas, F.; Arcos, D. Mesoporous Materials for Drug Delivery. Angew. Chem. Int. Ed. 2007, 46, 7548–7558. [Google Scholar] [CrossRef]
- Paris, J.L.; Vallet-Regí, M. Mesoporous Silica Nanoparticles for Co-Delivery of Drugs and Nucleic Acids in Oncology: A Review. Pharmaceutics 2020, 12, 526. [Google Scholar] [CrossRef]
- Martínez-Carmona, M.; Lozano, D.; Colilla, M.; Vallet-Regí, M. Lectin-Conjugated PH-Responsive Mesoporous Silica Nanoparticles for Targeted Bone Cancer Treatment. Acta Biomater. 2018, 65, 393–404. [Google Scholar] [CrossRef]
- Yohan, D.; Chithrani, B.D. Applications of Nanoparticles in Nanomedicine. J. Biomed. Nanotechnol. 2014, 10, 2371–2392. [Google Scholar] [CrossRef] [PubMed]
- Bawa, R. Regulating Nanomedicine—Can the FDA Handle It? Curr. Drug Deliv. 2011, 8, 227–234. [Google Scholar] [CrossRef] [PubMed]
- Vallet-Regi, M.; Rámila, A.; del Real, R.P.; Pérez-Pariente, J. A New Property of MCM-41: Drug Delivery System. Chem. Mater. 2001, 13, 308–311. [Google Scholar] [CrossRef]
- Ventola, C.L. The Nanomedicine Revolution: Part 1: Emerging Concepts. Pharm. Ther. 2012, 37, 512–525. [Google Scholar]
- Gisbert-Garzarán, M.; Vallet-Regí, M. Influence of the Surface Functionalization on the Fate and Performance of Mesoporous Silica Nanoparticles. Nanomaterials 2020, 10, 916. [Google Scholar] [CrossRef]
- Jeevanandam, J.; Barhoum, A.; Chan, Y.S.; Dufresne, A.; Danquah, M.K. Review on Nanoparticles and Nanostructured Materials: History, Sources, Toxicity and Regulations. Beilstein. J. Nanotechnol. 2018, 9, 1050–1074. [Google Scholar] [CrossRef]
- Boboc, M.; Curti, F.; Fleacă, A.M.; Jianu, M.L.; Roşu, A.-M.; Curutiu, C.; Lazar, V.; Chifiriuc, M.C.; Grumezescu, A.M. Preparation and Antimicrobial Activity of Inorganic Nanoparticles. In Nanostructures for Antimicrobial Therapy; Elsevier: Amsterdam, The Netherlands, 2017; pp. 325–340. ISBN 978-0-323-46152-8. [Google Scholar]
- Zazo, H.; Colino, C.I.; Lanao, J.M. Current Applications of Nanoparticles in Infectious Diseases. J. Control. Release 2016, 224, 86–102. [Google Scholar] [CrossRef]
- Lee, N.-Y.; Ko, W.-C.; Hsueh, P.-R. Nanoparticles in the Treatment of Infections Caused by Multidrug-Resistant Organisms. Front. Pharmacol. 2019, 10, 1153. [Google Scholar] [CrossRef]
- Djurišić, A.B.; Leung, Y.H.; Ng, A.M.C.; Xu, X.Y.; Lee, P.K.H.; Degger, N.; Wu, R.S.S. Toxicity of Metal Oxide Nanoparticles: Mechanisms, Characterization, and Avoiding Experimental Artefacts. Small 2015, 11, 26–44. [Google Scholar] [CrossRef]
- Vallet-Regí, M. Ordered Mesoporous Materials in the Context of Drug Delivery Systems and Bone Tissue Engineering. Chem.-Eur. J. 2006, 12, 5934–5943. [Google Scholar] [CrossRef]
- Gisbert-Garzarán, M.; Manzano, M.; Vallet-Regí, M. Mesoporous Silica Nanoparticles for the Treatment of Complex Bone Diseases: Bone Cancer, Bone Infection and Osteoporosis. Pharmaceutics 2020, 12, 83. [Google Scholar] [CrossRef] [PubMed]
- Vallet-Regí, M.; González, B.; Izquierdo-Barba, I. Nanomaterials as Promising Alternative in the Infection Treatment. Int. J. Mol. Sci. 2019, 20, 3806. [Google Scholar] [CrossRef] [PubMed]
- Vallet-Regí, M. Bioceramics: From Bone Substitutes to Nanoparticles for Drug Delivery. Pure Appl. Chem. 2019, 91, 687–706. [Google Scholar] [CrossRef] [PubMed]
- Mitragotri, S.; Stayton, P. Organic Nanoparticles for Drug Delivery and Imaging. MRS Bull. 2014, 39, 219–223. [Google Scholar] [CrossRef]
- Aparna, V.A.; Biswas, R.; Jayakumar, R. Targeted nanoparticles for treating infectious diseases. In Biomimetic Nanoengineered Materials for Advanced Drug Delivery; Elsevier: Amsterdam, The Netherlands, 2019; pp. 169–185. ISBN 978-0-12-814944-7. [Google Scholar]
- Schütz, C.A.; Juillerat-Jeanneret, L.; Mueller, H.; Lynch, I.; Riediker, M. Therapeutic Nanoparticles in Clinics and under Clinical Evaluation. Nanomedicine 2013, 8, 449–467. [Google Scholar] [CrossRef]
- Simonsen, J.B. A Liposome-Based Size Calibration Method for Measuring Microvesicles by Flow Cytometry. J. Thromb. Haemost. 2016, 14, 186–190. [Google Scholar] [CrossRef]
- Singh, L.; Kruger, H.G.; Maguire, G.E.M.; Govender, T.; Parboosing, R. The Role of Nanotechnology in the Treatment of Viral Infections. Ther. Adv. Infect. Dis. 2017, 4, 105–131. [Google Scholar] [CrossRef]
- Abbasi, E.; Aval, S.F.; Akbarzadeh, A.; Milani, M.; Nasrabadi, H.T.; Joo, S.W.; Hanifehpour, Y.; Nejati-Koshki, K.; Pashaei-Asl, R. Dendrimers: Synthesis, Applications, and Properties. Nanoscale Res. Lett. 2014, 9, 247. [Google Scholar] [CrossRef]
- Ghasemiyeh, P.; Mohammadi-Samani, S. Solid Lipid Nanoparticles and Nanostructured Lipid Carriers as Novel Drug Delivery Systems: Applications, Advantages and Disadvantages. Res. Pharm. Sci. 2018, 13, 288–303. [Google Scholar] [CrossRef]
- Moghimi, R.; Aliahmadi, A.; McClements, D.J.; Rafati, H. Nanoemulsification of Salvia officinalis Essential Oil; The Impact on the Antibacterial Activity in Liquid and Vapour Phase. J. Bionanosci. 2017, 11, 80–86. [Google Scholar] [CrossRef]
- Klepser, M.E. Socioeconomic Impact of Seasonal (Epidemic) Influenza and the Role of Over-the-Counter Medicines. Drugs 2014, 74, 1467–1479. [Google Scholar] [CrossRef] [PubMed]
- Troeger, C.; Forouzanfar, M.; Rao, P.C.; Khalil, I.; Brown, A.; Swartz, S.; Fullman, N.; Mosser, J.; Thompson, R.L.; Reiner, R.C.; et al. Estimates of the Global, Regional, and National Morbidity, Mortality, and Aetiologies of Lower Respiratory Tract Infections in 195 Countries: A Systematic Analysis for the Global Burden of Disease Study 2015. Lancet Infect. Dis. 2017, 17, 1133–1161. [Google Scholar] [CrossRef]
- Giraud-Gatineau, A.; Colson, P.; Jimeno, M.-T.; Zandotti, C.; Ninove, L.; Boschi, C.; Lagier, J.-C.; La Scola, B.; Chaudet, H.; Raoult, D. Comparison of Mortality Associated with Respiratory Viral Infections between December 2019 and March 2020 with That of the Previous Year in Southeastern France. Int. J. Infect. Dis. 2020, 96, 154–156. [Google Scholar] [CrossRef] [PubMed]
- Al-Halifa, S.; Gauthier, L.; Arpin, D.; Bourgault, S.; Archambault, D. Nanoparticle-Based Vaccines Against Respiratory Viruses. Front. Immunol. 2019, 10, 22. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Lin, Z.; Zhao, M.; Xu, T.; Wang, C.; Hua, L.; Wang, H.; Xia, H.; Zhu, B. Silver Nanoparticle Based Codelivery of Oseltamivir to Inhibit the Activity of the H1N1 Influenza Virus through ROS-Mediated Signaling Pathways. ACS Appl. Mater. Interfaces 2016, 8, 24385–24393. [Google Scholar] [CrossRef]
- Lin, Z.; Li, Y.; Guo, M.; Xu, T.; Wang, C.; Zhao, M.; Wang, H.; Chen, T.; Zhu, B. The Inhibition of H1N1 Influenza Virus-Induced Apoptosis by Silver Nanoparticles Functionalized with Zanamivir. RSC Adv. 2017, 7, 742–750. [Google Scholar] [CrossRef]
- Papp, I.; Sieben, C.; Ludwig, K.; Roskamp, M.; Böttcher, C.; Schlecht, S.; Herrmann, A.; Haag, R. Inhibition of Influenza Virus Infection by Multivalent Sialic-Acid-Functionalized Gold Nanoparticles. Small 2010, 6, 2900–2906. [Google Scholar] [CrossRef]
- Hendricks, G.L.; Weirich, K.L.; Viswanathan, K.; Li, J.; Shriver, Z.H.; Ashour, J.; Ploegh, H.L.; Kurt-Jones, E.A.; Fygenson, D.K.; Finberg, R.W.; et al. Sialylneolacto- N -Tetraose c (LSTc)-Bearing Liposomal Decoys Capture Influenza a Virus. J. Biol. Chem. 2013, 288, 8061–8073. [Google Scholar] [CrossRef]
- Lin, Z.; Li, Y.; Guo, M.; Xiao, M.; Wang, C.; Zhao, M.; Xu, T.; Xia, Y.; Zhu, B. Inhibition of H1N1 Influenza Virus by Selenium Nanoparticles Loaded with Zanamivir through P38 and JNK Signaling Pathways. RSC Adv. 2017, 7, 35290–35296. [Google Scholar] [CrossRef]
- Li, Y.; Lin, Z.; Guo, M.; Zhao, M.; Xia, Y.; Wang, C.; Xu, T.; Zhu, B. Inhibition of H1N1 Influenza Virus-Induced Apoptosis by Functionalized Selenium Nanoparticles with Amantadine through ROS-Mediated AKT Signaling Pathways. Int. J. Nanomed. 2018, 13, 2005–2016. [Google Scholar] [CrossRef]
- Lin, Z.; Li, Y.; Gong, G.; Xia, Y.; Wang, C.; Chen, Y.; Hua, L.; Zhong, J.; Tang, Y.; Liu, X.; et al. Restriction of H1N1 Influenza Virus Infection by Selenium Nanoparticles Loaded with Ribavirin via Resisting Caspase-3 Apoptotic Pathway. Int. J. Nanomed. 2018, 13, 5787–5797. [Google Scholar] [CrossRef]
- Li, Y.; Lin, Z.; Guo, M.; Xia, Y.; Zhao, M.; Wang, C.; Xu, T.; Chen, T.; Zhu, B. Inhibitory Activity of Selenium Nanoparticles Functionalized with Oseltamivir on H1N1 Influenza Virus. Int. J. Nanomed. 2017, 12, 5733–5743. [Google Scholar] [CrossRef]
- Kim, J.; Yeom, M.; Lee, T.; Kim, H.-O.; Na, W.; Kang, A.; Lim, J.-W.; Park, G.; Park, C.; Song, D.; et al. Porous Gold Nanoparticles for Attenuating Infectivity of Influenza a Virus. J. Nanobiotechnol. 2020, 18, 54. [Google Scholar] [CrossRef] [PubMed]
- Sametband, M.; Shukla, S.; Meningher, T.; Hirsh, S.; Mendelson, E.; Sarid, R.; Gedanken, A.; Mandelboim, M. Effective Multi-Strain Inhibition of Influenza Virus by Anionic Gold Nanoparticles. Med. Chem. Commun. 2011, 2, 421. [Google Scholar] [CrossRef]
- Levina, A.S.; Repkova, M.N.; Mazurkova, N.A.; Zarytova, V.F. Nanoparticle-Mediated Nonviral DNA Delivery for Effective Inhibition of Influenza a Viruses in Cells. IEEE Trans. Nanotechnol. 2016, 15, 248–254. [Google Scholar] [CrossRef]
- Ghaffari, H.; Tavakoli, A.; Moradi, A.; Tabarraei, A.; Bokharaei-Salim, F.; Zahmatkeshan, M.; Farahmand, M.; Javanmard, D.; Kiani, S.J.; Esghaei, M.; et al. Inhibition of H1N1 Influenza Virus Infection by Zinc Oxide Nanoparticles: Another Emerging Application of Nanomedicine. J. Biomed. Sci. 2019, 26, 70. [Google Scholar] [CrossRef]
- Wang, C.-Z.; Li, W.-J.; Tao, R.; Ye, J.-Z.; Zhang, H.-Y. Antiviral Activity of a Nanoemulsion of Polyprenols from Ginkgo Leaves against Influenza a H3N2 and Hepatitis B Virus in Vitro. Molecules 2015, 20, 5137–5151. [Google Scholar] [CrossRef]
- Morris, D.; Ansar, M.; Speshock, J.; Ivanciuc, T.; Qu, Y.; Casola, A.; Garofalo, R. Antiviral and Immunomodulatory Activity of Silver Nanoparticles in Experimental RSV Infection. Viruses 2019, 11, 732. [Google Scholar] [CrossRef]
- Yang, X.X.; Li, C.M.; Huang, C.Z. Curcumin Modified Silver Nanoparticles for Highly Efficient Inhibition of Respiratory Syncytial Virus Infection. Nanoscale 2016, 8, 3040–3048. [Google Scholar] [CrossRef]
- Cagno, V.; Andreozzi, P.; D’Alicarnasso, M.; Jacob Silva, P.; Mueller, M.; Galloux, M.; Le Goffic, R.; Jones, S.T.; Vallino, M.; Hodek, J.; et al. Broad-Spectrum Non-Toxic Antiviral Nanoparticles with a Virucidal Inhibition Mechanism. Nature Mater. 2018, 17, 195–203. [Google Scholar] [CrossRef]
- Osminkina, L.A.; Timoshenko, V.Y.; Shilovsky, I.P.; Kornilaeva, G.V.; Shevchenko, S.N.; Gongalsky, M.B.; Tamarov, K.P.; Abramchuk, S.S.; Nikiforov, V.N.; Khaitov, M.R.; et al. Porous Silicon Nanoparticles as Scavengers of Hazardous Viruses. J. Nanopart. Res. 2014, 16, 2430. [Google Scholar] [CrossRef]
- Huang, X.; Li, M.; Xu, Y.; Zhang, J.; Meng, X.; An, X.; Sun, L.; Guo, L.; Shan, X.; Ge, J.; et al. Novel Gold Nanorod-Based HR1 Peptide Inhibitor for Middle East Respiratory Syndrome Coronavirus. ACS Appl. Mater. Interfaces 2019, 11, 19799–19807. [Google Scholar] [CrossRef]
- Gurunathan, S.; Qasim, M.; Choi, Y.; Do, J.T.; Park, C.; Hong, K.; Kim, J.-H.; Song, H. Antiviral Potential of Nanoparticles—Can Nanoparticles Fight Against Coronaviruses? Nanomaterials 2020, 10, 1645. [Google Scholar] [CrossRef]
- COVID-19 Map. Available online: https://coronavirus.jhu.edu/map.html (accessed on 3 November 2020).
- Medhi, R.; Srinoi, P.; Ngo, N.; Tran, H.-V.; Lee, T.R. Nanoparticle-Based Strategies to Combat COVID-19. ACS Appl. Nano Mater. 2020, 3, 8557–8580. [Google Scholar] [CrossRef]
- Aydemir, D.; Ulusu, N.N. Correspondence: Angiotensin-Converting Enzyme 2 Coated Nanoparticles Containing Respiratory Masks, Chewing Gums and Nasal Filters May Be Used for Protection against COVID-19 Infection. Travel. Med. Infect. Dis. 2020, 37, 101697. [Google Scholar] [CrossRef] [PubMed]
- Bellido-Blasco, J.B.; Arnedo-Pena, A. Epidemiology of Infectious Diarrhea. In Encyclopedia of Environmental Health; Elsevier: Amsterdam, The Netherlands, 2011; pp. 659–671. ISBN 978-0-444-63952-3. [Google Scholar]
- Broglie, J.J.; Alston, B.; Yang, C.; Ma, L.; Adcock, A.F.; Chen, W.; Yang, L. Antiviral Activity of Gold/Copper Sulfide Core/Shell Nanoparticles against Human Norovirus Virus-Like Particles. PLoS ONE 2015, 10, e0141050. [Google Scholar] [CrossRef]
- Li, Z.; Cui, K.; Wang, H.; Liu, F.; Huang, K.; Duan, Z.; Wang, F.; Shi, D.; Liu, Q. A Milk-Based Self-Assemble Rotavirus VP6–Ferritin Nanoparticle Vaccine Elicited Protection against the Viral Infection. J. Nanobiotechnol. 2019, 17, 13. [Google Scholar] [CrossRef]
- Heinimäki, S.; Hankaniemi, M.M.; Sioofy-Khojine, A.-B.; Laitinen, O.H.; Hyöty, H.; Hytönen, V.P.; Vesikari, T.; Blazevic, V. Combination of Three Virus-Derived Nanoparticles as a Vaccine against Enteric Pathogens; Enterovirus, Norovirus and Rotavirus. Vaccine 2019, 37, 7509–7518. [Google Scholar] [CrossRef]
- Lysenko, V.; Lozovski, V.; Lokshyn, M.; Gomeniuk, Y.V.; Dorovskih, A.; Rusinchuk, N.; Pankivska, Y.; Povnitsa, O.; Zagorodnya, S.; Tertykh, V.; et al. Nanoparticles as Antiviral Agents against Adenoviruses. Adv. Nat. Sci. Nanosci. Nanotechnol. 2018, 9, 025021. [Google Scholar] [CrossRef]
- Baun, A.; Hansen, S.F. Environmental Challenges for Nanomedicine. Nanomedicine 2008, 3, 605–608. [Google Scholar] [CrossRef]
- Mahapatra, I.; Clark, J.R.A.; Dobson, P.J.; Owen, R.; Lynch, I.; Lead, J.R. Expert Perspectives on Potential Environmental Risks from Nanomedicines and Adequacy of the Current Guideline on Environmental Risk Assessment. Environ. Sci.: Nano 2018, 5, 1873–1889. [Google Scholar] [CrossRef]
- Sexually Transmitted Infections (STIs). Available online: https://www.who.int/news-room/fact-sheets/detail/sexually-transmitted-infections-(stis) (accessed on 4 November 2020).
- Nijmeijer, B.M.; Koopsen, J.; Schinkel, J.; Prins, M.; Geijtenbeek, T.B. Sexually Transmitted Hepatitis C Virus Infections: Current Trends, and Recent Advances in Understanding the Spread in Men Who Have Sex with Men. J. Int. AIDS Soc. 2019, 22 (Suppl. 6), e25348. [Google Scholar] [CrossRef]
- Global Statistics. Available online: https://www.hiv.gov/hiv-basics/overview/data-and-trends/global-statistics (accessed on 16 November 2020).
- Prokofjeva, M.M.; Kochetkov, S.N.; Prassolov, V.S. Therapy of HIV Infection: Current Approaches and Prospects. Acta Nat. 2016, 8, 23–32. [Google Scholar] [CrossRef]
- HIV Treatment: The Basics. Available online: https://hivinfo.nih.gov/understanding-hiv/fact-sheets/hiv-treatment-basics (accessed on 16 November 2020).
- Zdanowicz, M.M. The Pharmacology of HIV Drug Resistance. Am. J. Pharm. Educ. 2006, 70, 100. [Google Scholar] [CrossRef]
- Sun, R.W.-Y.; Chen, R.; Chung, N.P.-Y.; Ho, C.-M.; Lin, C.-L.S.; Che, C.-M. Silver Nanoparticles Fabricated in Hepes Buffer Exhibit Cytoprotective Activities toward HIV-1 Infected Cells. Chem. Commun. 2005, 5059. [Google Scholar] [CrossRef]
- Rosemary Bastian, A.; Nangarlia, A.; Bailey, L.D.; Holmes, A.; Kalyana Sundaram, R.V.; Ang, C.; Moreira, D.R.M.; Freedman, K.; Duffy, C.; Contarino, M.; et al. Mechanism of Multivalent Nanoparticle Encounter with HIV-1 for Potency Enhancement of Peptide Triazole Virus Inactivation. J. Biol. Chem. 2015, 290, 529–543. [Google Scholar] [CrossRef]
- Di Gianvincenzo, P.; Marradi, M.; Martínez-Ávila, O.M.; Bedoya, L.M.; Alcamí, J.; Penadés, S. Gold Nanoparticles Capped with Sulfate-Ended Ligands as Anti-HIV Agents. Bioorg. Med. Chem. Lett. 2010, 20, 2718–2721. [Google Scholar] [CrossRef]
- Jaramillo-Ruiz, D.; De La Mata, F.J.; Gómez, R.; Correa-Rocha, R.; Muñoz-Fernández, M.Á. Nanotechnology as a New Therapeutic Approach to Prevent the HIV-Infection of Treg Cells. PLoS ONE 2016, 11, e0145760. [Google Scholar] [CrossRef]
- Parboosing, R.; Chonco, L.; de la Mata, F.J.; Govender, T.; Maguire, G.E.; Kruger, H.G. Potential Inhibition of HIV-1 Encapsidation by Oligoribonucleotide-Dendrimer Nanoparticle Complexes. Int. J. Nanomed. 2017, 12, 317–325. [Google Scholar] [CrossRef]
- Jayant, R.; Atluri, V.; Agudelo, M.; Sagar, V.; Kaushik, A.; Nair, M. Sustained-Release NanoART Formulation for The Treatment of NeuroAIDS. Int. J. Nanomed. 2015, 10, 1077. [Google Scholar] [CrossRef]
- Smith, K.A. Activation of HIV-1 with Nanoparticle-Packaged Small Molecule Protein Phosphatase-1-Targeting Compound. Sci. Pharm. 2015, 83, 535–548. [Google Scholar] [CrossRef] [PubMed]
- Jiang, Y.; Cao, S.; Bright, D.K.; Bever, A.M.; Blakney, A.K.; Suydam, I.T.; Woodrow, K.A. Nanoparticle-Based ARV Drug Combinations for Synergistic Inhibition of Cell-Free and Cell–Cell HIV Transmission. Mol. Pharm. 2015, 12, 4363–4374. [Google Scholar] [CrossRef]
- Chaowanachan, T.; Krogstad, E.; Ball, C.; Woodrow, K.A. Drug Synergy of Tenofovir and Nanoparticle-Based Antiretrovirals for HIV Prophylaxis. PLoS ONE 2013, 8, e61416. [Google Scholar] [CrossRef]
- de Souza e Silva, J.M.; Hanchuk, T.D.M.; Santos, M.I.; Kobarg, J.; Bajgelman, M.C.; Cardoso, M.B. Viral Inhibition Mechanism Mediated by Surface-Modified Silica Nanoparticles. ACS Appl. Mater. Interfaces 2016, 8, 16564–16572. [Google Scholar] [CrossRef]
- Bromberg, L.; Bromberg, D.J.; Hatton, T.A.; Bandín, I.; Concheiro, A.; Alvarez-Lorenzo, C. Antiviral Properties of Polymeric Aziridine- and Biguanide-Modified Core–Shell Magnetic Nanoparticles. Langmuir 2012, 28, 4548–4558. [Google Scholar] [CrossRef]
- Baram-Pinto, D.; Shukla, S.; Perkas, N.; Gedanken, A.; Sarid, R. Inhibition of Herpes Simplex Virus Type 1 Infection by Silver Nanoparticles Capped with Mercaptoethane Sulfonate. Bioconjug. Chem. 2009, 20, 1497–1502. [Google Scholar] [CrossRef]
- Lima, T.; Feitosa, R.; dos Santos-Silva, E.; dos Santos-Silva, A.; Siqueira, E.; Machado, P.; Cornélio, A.; do Egito, E.; Fernandes-Pedrosa, M.; Farias, K.; et al. Improving Encapsulation of Hydrophilic Chloroquine Diphosphate into Biodegradable Nanoparticles: A Promising Approach against Herpes Virus Simplex-1 Infection. Pharmaceutics 2018, 10, 255. [Google Scholar] [CrossRef]
- Al-Dhubiab, B.E.; Nair, A.B.; Kumria, R.; Attimarad, M.; Harsha, S. Formulation and Evaluation of Nano Based Drug Delivery System for the Buccal Delivery of Acyclovir. Colloids Surf. B Biointerfaces 2015, 136, 878–884. [Google Scholar] [CrossRef]
- Palliser, D.; Chowdhury, D.; Wang, Q.-Y.; Lee, S.J.; Bronson, R.T.; Knipe, D.M.; Lieberman, J. An SiRNA-Based Microbicide Protects Mice from Lethal Herpes Simplex Virus 2 Infection. Nature 2006, 439, 89–94. [Google Scholar] [CrossRef]
- Woodrow, K.A.; Cu, Y.; Booth, C.J.; Saucier-Sawyer, J.K.; Wood, M.J.; Saltzman, W.M. Intravaginal Gene Silencing Using Biodegradable Polymer Nanoparticles Densely Loaded with Small-Interfering RNA. Nat. Mater. 2009, 8, 526–533. [Google Scholar] [CrossRef]
- Ramyadevi, D.; Rajan, K.S.; Vedhahari, B.N.; Ruckmani, K.; Subramanian, N. Heterogeneous Polymer Composite Nanoparticles Loaded in Situ Gel for Controlled Release Intra-Vaginal Therapy of Genital Herpes. Colloids Surf. B Biointerfaces 2016, 146, 260–270. [Google Scholar] [CrossRef] [PubMed]
- Lu, L.; Sun, R.W.-Y.; Chen, R.; Hui, C.-K.; Ho, C.-M.; Luk, J.M.; Lau, G.K.K.; Che, C.-M. Silver Nanoparticles Inhibit Hepatitis B Virus Replication. Antivir. Ther. 2008, 13, 253–262. [Google Scholar] [PubMed]
- Wang, J.; Feng, S.-S.; Wang, S.; Chen, Z. Evaluation of Cationic Nanoparticles of Biodegradable Copolymers as SiRNA Delivery System for Hepatitis B Treatment. Int. J. Pharm. 2010, 400, 194–200. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Mulvenon, A.; Makarov, E.; Wagoner, J.; Knibbe, J.; Kim, J.O.; Osna, N.; Bronich, T.K.; Poluektova, L.Y. Antiviral Peptide Nanocomplexes as a Potential Therapeutic Modality for HIV/HCV Co-Infection. Biomaterials 2013, 34, 3846–3857. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Garrison, J.C.; Poluektova, L.Y.; Bronich, T.K.; Osna, N.A. Liver-Targeted Antiviral Peptide Nanocomplexes as Potential Anti-HCV Therapeutics. Biomaterials 2015, 70, 37–47. [Google Scholar] [CrossRef]
- Katas, H.; Alpar, H.O. Development and Characterisation of Chitosan Nanoparticles for SiRNA Delivery. J. Control. Release 2006, 115, 216–225. [Google Scholar] [CrossRef]
- Torrecilla, J.; del Pozo-Rodríguez, A.; Apaolaza, P.S.; Solinís, M.Á.; Rodríguez-Gascón, A. Solid Lipid Nanoparticles as Non-Viral Vector for the Treatment of Chronic Hepatitis C by RNA Interference. Int. J. Pharm. 2015, 479, 181–188. [Google Scholar] [CrossRef]
- Hang, X.; Peng, H.; Song, H.; Qi, Z.; Miao, X.; Xu, W. Antiviral Activity of Cuprous Oxide Nanoparticles against Hepatitis C Virus in Vitro. J. Virol. Methods 2015, 222, 150–157. [Google Scholar] [CrossRef]
- Ryoo, S.-R.; Jang, H.; Kim, K.-S.; Lee, B.; Kim, K.B.; Kim, Y.-K.; Yeo, W.-S.; Lee, Y.; Kim, D.-E.; Min, D.-H. Functional Delivery of DNAzyme with Iron Oxide Nanoparticles for Hepatitis C Virus Gene Knockdown. Biomaterials 2012, 33, 2754–2761. [Google Scholar] [CrossRef]
- Ahmed–Belkacem, A.; Ahnou, N.; Barbotte, L.; Wychowski, C.; Pallier, C.; Brillet, R.; Pohl, R.; Pawlotsky, J. Silibinin and Related Compounds Are Direct Inhibitors of Hepatitis C Virus RNA-Dependent RNA Polymerase. Gastroenterology 2010, 138, 1112–1122. [Google Scholar] [CrossRef]
- Delaviz, N.; Gill, P.; Ajami, A.; Aarabi, M. Aptamer-Conjugated Magnetic Nanoparticles for the Efficient Removal of HCV Particles from Human Plasma Samples. RSC Adv. 2015, 5, 79433–79439. [Google Scholar] [CrossRef]
- Mathew, J., Jr.; Sapra, A. Herpes Simplex Type 2. In StatPearls; StatPearls Publishing: Treasure Island, FL, USA, 2020. [Google Scholar]
- Rupar, M.J.; Golusinski, P.; Golusinski, W.; Masternak, M.M. Human Papillomavirus and the Use of Nanoparticles for Immunotherapy in HPV-Related Cancer: A Review. Rep. Pract. Oncol. Radiother. 2019, 24, 544–550. [Google Scholar] [CrossRef]
- Gilson, R.; Brook, M.G. Hepatitis A, B, and C. Sexually Transmitted Infections 2006, 82, iv35–iv39. [Google Scholar] [CrossRef] [PubMed]
- Wang, B.; He, X.; Zhang, Z.; Zhao, Y.; Feng, W. Metabolism of Nanomaterials in Vivo: Blood Circulation and Organ Clearance. Acc. Chem. Res. 2013, 46, 761–769. [Google Scholar] [CrossRef] [PubMed]
- Harris, J.B.; LaRocque, R.C.; Qadri, F.; Ryan, E.T.; Calderwood, S.B. Cholera. Lancet 2012, 379, 2466–2476. [Google Scholar] [CrossRef]
- Sarwar, S.; Ali, A.; Pal, M.; Chakrabarti, P. Zinc Oxide Nanoparticles Provide Anti-Cholera Activity by Disrupting the Interaction of Cholera Toxin with the Human GM1 Receptor. J. Biol. Chem. 2017, 292, 18303–18311. [Google Scholar] [CrossRef]
- Rajput, V.D.; Minkina, T.M.; Behal, A.; Sushkova, S.N.; Mandzhieva, S.; Singh, R.; Gorovtsov, A.; Tsitsuashvili, V.S.; Purvis, W.O.; Ghazaryan, K.A.; et al. Effects of Zinc-Oxide Nanoparticles on Soil, Plants, Animals and Soil Organisms: A Review. Environ. Nanotechnol. Monit. Manag. 2018, 9, 76–84. [Google Scholar] [CrossRef]
- Das, S.; Angsantikul, P.; Le, C.; Bao, D.; Miyamoto, Y.; Gao, W.; Zhang, L.; Eckmann, L. Neutralization of Cholera Toxin with Nanoparticle Decoys for Treatment of Cholera. PLoS Negl. Trop. Dis. 2018, 12, e0006266. [Google Scholar] [CrossRef]
- Yang, R. Plague: Recognition, Treatment, and Prevention. J. Clin. Microbiol. 2018, 56. [Google Scholar] [CrossRef]
- Tao, P.; Mahalingam, M.; Rao, V.B. Highly Effective Soluble and Bacteriophage T4 Nanoparticle Plague Vaccines Against Yersinia pestis. In Vaccine Design; Thomas, S., Ed.; Methods in Molecular Biology; Springer: New York, NY, USA, 2016; Volume 1403, pp. 499–518. ISBN 978-1-4939-3385-3. [Google Scholar]
- Barberis, I.; Bragazzi, N.L.; Galluzzo, L.; Martini, M. The History of Tuberculosis: From the First Historical Records to the Isolation of Koch’s Bacillus. J. Prev. Med. Hyg. 2017, 58, E9–E12. [Google Scholar]
- MacNeil, A.; Glaziou, P.; Sismanidis, C.; Date, A.; Maloney, S.; Floyd, K. Global Epidemiology of Tuberculosis and Progress Toward Meeting Global Targets—Worldwide, 2018. MMWR Morb. Mortal. Wkly. Rep. 2020, 69, 281–285. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization. Global Tuberculosis Report 2019; World Health Organization: Geneva, Switzerland, 2019; ISBN 978-92-4-156571-4. [Google Scholar]
- Tăbăran, A.-F.; Matea, C.T.; Mocan, T.; Tăbăran, A.; Mihaiu, M.; Iancu, C.; Mocan, L. Silver Nanoparticles for the Therapy of Tuberculosis. Int. J. Nanomed. 2020, 15, 2231–2258. [Google Scholar] [CrossRef]
- Estevez, H.; Palacios, A.; Gil, D.; Anguita, J.; Vallet-Regi, M.; González, B.; Prados-Rosales, R.; Luque-Garcia, J.L. Antimycobacterial Effect of Selenium Nanoparticles on Mycobacterium Tuberculosis. Front. Microbiol. 2020, 11, 800. [Google Scholar] [CrossRef] [PubMed]
- Ramalingam, V.; Sundaramahalingam, S.; Rajaram, R. Size-Dependent Antimycobacterial Activity of Titanium Oxide Nanoparticles against Mycobacterium Tuberculosis. J. Mater. Chem. B 2019, 7, 4338–4346. [Google Scholar] [CrossRef]
- de Faria, T.J.; Roman, M.; de Souza, N.M.; De Vecchi, R.; de Assis, J.V.; dos Santos, A.L.G.; Bechtold, I.H.; Winter, N.; Soares, M.J.; Silva, L.P.; et al. An Isoniazid Analogue Promotes Mycobacterium Tuberculosis-Nanoparticle Interactions and Enhances Bacterial Killing by Macrophages. Antimicrob. Agents Chemother. 2012, 56, 2259–2267. [Google Scholar] [CrossRef]
- Centers for Disease Control and Prevention (USA). Antibiotic Resistance Threats in the United States; Department of Health and Human Services, CDC: Atlanta, GA, USA, 2019. [Google Scholar]
- Tacconelli, E. Global Priority List of Antibiotic-Resistant Bacteria to Guide Research, Discovery, and Development of New Antibiotics; World Health Organization: Geneva, Switzerland, 2017; Volume 27, pp. 318–327. [Google Scholar]
- Reygaert, W.C. An Overview of the Antimicrobial Resistance Mechanisms of Bacteria. AIMS Microbiol 2018, 4, 482–501. [Google Scholar] [CrossRef] [PubMed]
- Huh, A.J.; Kwon, Y.J. “Nanoantibiotics”: A New Paradigm for Treating Infectious Diseases Using Nanomaterials in the Antibiotics Resistant Era. J. Control. Release 2011, 156, 128–145. [Google Scholar] [CrossRef]
- Cavassin, E.D.; de Figueiredo, L.F.P.; Otoch, J.P.; Seckler, M.M.; de Oliveira, R.A.; Franco, F.F.; Marangoni, V.S.; Zucolotto, V.; Levin, A.S.S.; Costa, S.F. Comparison of Methods to Detect the in Vitro Activity of Silver Nanoparticles (AgNP) against Multidrug Resistant Bacteria. J. Nanobiotechnol. 2015, 13, 64. [Google Scholar] [CrossRef]
- Zaidi, S.; Misba, L.; Khan, A.U. Nano-Therapeutics: A Revolution in Infection Control in Post Antibiotic Era. Nanomed. Nanotechnol. Biol. Med. 2017, 13, 2281–2301. [Google Scholar] [CrossRef]
- Rudramurthy, G.; Swamy, M.; Sinniah, U.; Ghasemzadeh, A. Nanoparticles: Alternatives Against Drug-Resistant Pathogenic Microbes. Molecules 2016, 21, 836. [Google Scholar] [CrossRef]
- Hemeg, H. Nanomaterials for Alternative Antibacterial Therapy. Int. J. Nanomed. 2017, 12, 8211–8225. [Google Scholar] [CrossRef]
- Chen, C.-W.; Hsu, C.-Y.; Lai, S.-M.; Syu, W.-J.; Wang, T.-Y.; Lai, P.-S. Metal Nanobullets for Multidrug Resistant Bacteria and Biofilms. Adv. Drug Deliv. Rev. 2014, 78, 88–104. [Google Scholar] [CrossRef] [PubMed]
- Dizaj, S.M.; Lotfipour, F.; Barzegar-Jalali, M.; Zarrintan, M.H.; Adibkia, K. Antimicrobial Activity of the Metals and Metal Oxide Nanoparticles. Mater. Sci. Eng. C 2014, 44, 278–284. [Google Scholar] [CrossRef]
- Chatterjee, A.K.; Chakraborty, R.; Basu, T. Mechanism of Antibacterial Activity of Copper Nanoparticles. Nanotechnology 2014, 25, 135101. [Google Scholar] [CrossRef] [PubMed]
- Feng, Q.L.; Wu, J.; Chen, G.Q.; Cui, F.Z.; Kim, T.N.; Kim, J.O. A Mechanistic Study of the Antibacterial Effect of Silver Ions on Escherichia Coli and Staphylococcus Aureus. J. Biomed. Mater. Res. 2000, 52, 662–668. [Google Scholar] [CrossRef]
- Smetana, A.B.; Klabunde, K.J.; Marchin, G.R.; Sorensen, C.M. Biocidal Activity of Nanocrystalline Silver Powders and Particles. Langmuir 2008, 24, 7457–7464. [Google Scholar] [CrossRef] [PubMed]
- Panáček, A.; Kvítek, L.; Prucek, R.; Kolář, M.; Večeřová, R.; Pizúrová, N.; Sharma, V.K.; Nevěčná, T.; Zbořil, R. Silver Colloid Nanoparticles: Synthesis, Characterization, and Their Antibacterial Activity. J. Phys. Chem. B 2006, 110, 16248–16253. [Google Scholar] [CrossRef]
- Nanda, A.; Saravanan, M. Biosynthesis of Silver Nanoparticles from Staphylococcus Aureus and Its Antimicrobial Activity against MRSA and MRSE. Nanomed. Nanotechnol. Biol. Med. 2009, 5, 452–456. [Google Scholar] [CrossRef]
- Sondi, I.; Salopek-Sondi, B. Silver Nanoparticles as Antimicrobial Agent: A Case Study on E. Coli as a Model for Gram-Negative Bacteria. J. Colloid Interface Sci. 2004, 275, 177–182. [Google Scholar] [CrossRef]
- Kim, J.S.; Kuk, E.; Yu, K.N.; Kim, J.-H.; Park, S.J.; Lee, H.J.; Kim, S.H.; Park, Y.K.; Park, Y.H.; Hwang, C.-Y.; et al. Antimicrobial Effects of Silver Nanoparticles. Nanomed. Nanotechnol. Biol. Med. 2007, 3, 95–101. [Google Scholar] [CrossRef] [PubMed]
- Sharma, V.K.; Yngard, R.A.; Lin, Y. Silver Nanoparticles: Green Synthesis and Their Antimicrobial Activities. Adv. Colloid Interface Sci. 2009, 145, 83–96. [Google Scholar] [CrossRef] [PubMed]
- Gil-Tomás, J.; Tubby, S.; Parkin, I.P.; Narband, N.; Dekker, L.; Nair, S.P.; Wilson, M.; Street, C. Lethal Photosensitisation of Staphylococcus Aureus Using a Toluidine Blue O–Tiopronin–Gold Nanoparticle Conjugate. J. Mater. Chem. 2007, 17, 3739. [Google Scholar] [CrossRef]
- Perni, S.; Piccirillo, C.; Pratten, J.; Prokopovich, P.; Chrzanowski, W.; Parkin, I.P.; Wilson, M. The Antimicrobial Properties of Light-Activated Polymers Containing Methylene Blue and Gold Nanoparticles. Biomaterials 2009, 30, 89–93. [Google Scholar] [CrossRef] [PubMed]
- MubarakAli, D.; Thajuddin, N.; Jeganathan, K.; Gunasekaran, M. Plant Extract Mediated Synthesis of Silver and Gold Nanoparticles and Its Antibacterial Activity against Clinically Isolated Pathogens. Colloids Surf. B Biointerfaces 2011, 85, 360–365. [Google Scholar] [CrossRef]
- Li, X.; Robinson, S.M.; Gupta, A.; Saha, K.; Jiang, Z.; Moyano, D.F.; Sahar, A.; Riley, M.A.; Rotello, V.M. Functional Gold Nanoparticles as Potent Antimicrobial Agents against Multi-Drug-Resistant Bacteria. ACS Nano 2014, 8, 10682–10686. [Google Scholar] [CrossRef]
- Matsunaga, T.; Tomoda, R.; Nakajima, T.; Nakamura, N.; Komine, T. Continuous-Sterilization System That Uses Photosemiconductor Powders. Appl. Environ. Microbiol. 1988, 54, 1330–1333. [Google Scholar] [CrossRef]
- Kim, B.; Kim, D.; Cho, D.; Cho, S. Bactericidal Effect of TiO2 Photocatalyst on Selected Food-Borne Pathogenic Bacteria. Chemosphere 2003, 52, 277–281. [Google Scholar] [CrossRef]
- Chawengkijwanich, C.; Hayata, Y. Development of TiO2 Powder-Coated Food Packaging Film and Its Ability to Inactivate Escherichia Coli in Vitro and in Actual Tests. Int. J. Food Microbiol. 2008, 123, 288–292. [Google Scholar] [CrossRef]
- Jiang, W.; Mashayekhi, H.; Xing, B. Bacterial Toxicity Comparison between Nano- and Micro-Scaled Oxide Particles. Environ. Pollut. 2009, 157, 1619–1625. [Google Scholar] [CrossRef]
- Jones, N.; Ray, B.; Ranjit, K.T.; Manna, A.C. Antibacterial Activity of ZnO Nanoparticle Suspensions on a Broad Spectrum of Microorganisms. FEMS Microbiol. Lett. 2008, 279, 71–76. [Google Scholar] [CrossRef]
- Ghule, K.; Ghule, A.V.; Chen, B.-J.; Ling, Y.-C. Preparation and Characterization of ZnO Nanoparticles Coated Paper and Its Antibacterial Activity Study. Green Chem. 2006, 8, 1034. [Google Scholar] [CrossRef]
- Cioffi, N.; Torsi, L.; Ditaranto, N.; Tantillo, G.; Ghibelli, L.; Sabbatini, L.; Bleve-Zacheo, T.; D’Alessio, M.; Zambonin, P.G.; Traversa, E. Copper Nanoparticle/Polymer Composites with Antifungal and Bacteriostatic Properties. Chem. Mater. 2005, 17, 5255–5262. [Google Scholar] [CrossRef]
- Ruparelia, J.P.; Chatterjee, A.K.; Duttagupta, S.P.; Mukherji, S. Strain Specificity in Antimicrobial Activity of Silver and Copper Nanoparticles. Acta Biomater. 2008, 4, 707–716. [Google Scholar] [CrossRef]
- Koper, O.B.; Klabunde, J.S.; Marchin, G.L.; Klabunde, K.J.; Stoimenov, P.; Bohra, L. Nanoscale Powders and Formulations with Biocidal Activity toward Spores and Vegetative Cells of Bacillus Species, Viruses, and Toxins. Curr. Microbiol. 2002, 44, 49–55. [Google Scholar] [CrossRef] [PubMed]
- Krishnamoorthy, K.; Manivannan, G.; Kim, S.J.; Jeyasubramanian, K.; Premanathan, M. Antibacterial Activity of MgO Nanoparticles Based on Lipid Peroxidation by Oxygen Vacancy. J. Nanopart. Res. 2012, 14, 1063. [Google Scholar] [CrossRef]
- Roy, A.; Gauri, S.S.; Bhattacharya, M.; Bhattacharya, J. Antimicrobial Activity of CaO Nanoparticles. J. Biomed. Nanotechnol. 2013, 9, 1570–1578. [Google Scholar] [CrossRef]
- Sawai, J. Quantitative Evaluation of Antibacterial Activities of Metallic Oxide Powders (ZnO, MgO and CaO) by Conductimetric Assay. J. Microbiol. Methods 2003, 54, 177–182. [Google Scholar] [CrossRef]
- Catauro, M.; Raucci, M.G.; de Gaetano, F.; Marotta, A. Antibacterial and Bioactive Silver-Containing Na2O·CaO·2SiO2 Glass Prepared by Sol–Gel Method. J. Mater. Sci. Mater. Med. 2004, 15, 831–837. [Google Scholar] [CrossRef]
- Li, B.; Logan, B.E. Bacterial Adhesion to Glass and Metal-Oxide Surfaces. Colloids Surf. B Biointerfaces 2004, 36, 81–90. [Google Scholar] [CrossRef]
- Prashanth, P.A.; Raveendra, R.S.; Hari Krishna, R.; Ananda, S.; Bhagya, N.P.; Nagabhushana, B.M.; Lingaraju, K.; Raja Naika, H. Synthesis, Characterizations, Antibacterial and Photoluminescence Studies of Solution Combustion-Derived α-Al2O3 Nanoparticles. J. Asian Ceram. Soc. 2015, 3, 345–351. [Google Scholar] [CrossRef]
- Jastrzębska, A.M.; Radziun, E.; Rosłon, M.; Kunicki, A.R.; Olszyna, A.R.; Dudkiewicz-Wilczyńska, J.; Anuszewska, E.; Karwowska, E. In Vitro Assessment of Antibacterial Properties and Cytotoxicity of Al2O3–Ag Nanopowders. Adv. Appl. Ceram. 2011, 110, 353–359. [Google Scholar] [CrossRef]
- Adams, L.K.; Lyon, D.Y.; Alvarez, P.J.J. Comparative Eco-Toxicity of Nanoscale TiO2, SiO2, and ZnO Water Suspensions. Water Res. 2006, 40, 3527–3532. [Google Scholar] [CrossRef] [PubMed]
- Haydel, S.E.; Remenih, C.M.; Williams, L.B. Broad-Spectrum in Vitro Antibacterial Activities of Clay Minerals against Antibiotic-Susceptible and Antibiotic-Resistant Bacterial Pathogens. J. Antimicrob. Chemother. 2007, 61, 353–361. [Google Scholar] [CrossRef] [PubMed]
- Suganya, P.; Vaseeharan, B.; Vijayakumar, S.; Balan, B.; Govindarajan, M.; Alharbi, N.S.; Kadaikunnan, S.; Khaled, J.M.; Benelli, G. Biopolymer Zein-Coated Gold Nanoparticles: Synthesis, Antibacterial Potential, Toxicity and Histopathological Effects against the Zika Virus Vector Aedes Aegypti. J. Photochem. Photobiol. B Biol. 2017, 173, 404–411. [Google Scholar] [CrossRef] [PubMed]
- Yang, S.B.; Rabbani, M.M.; Ji, B.C.; Han, D.-W.; Lee, J.S.; Kim, J.W.; Yeum, J.H. Optimum Conditions for the Fabrication of Zein/Ag Composite Nanoparticles from Ethanol/H₂O Co-Solvents Using Electrospinning. Nanomaterials 2016, 6, 230. [Google Scholar] [CrossRef] [PubMed]
- Bianchetti, M.G.; Simonetti, G.D.; Bettinelli, A. Body Fluids and Salt Metabolism—Part I. Ital. J. Pediatr. 2009, 35, 36. [Google Scholar] [CrossRef]
- Lobo, V.; Patil, A.; Phatak, A.; Chandra, N. Free Radicals, Antioxidants and Functional Foods: Impact on Human Health. Pharm. Rev. 2010, 4, 118–126. [Google Scholar] [CrossRef]
- Aguilera-Correa, J.-J.; Conde, A.; Arenas, M.-A.; de-Damborenea, J.-J.; Marin, M.; Doadrio, A.L.; Esteban, J. Bactericidal Activity of the Ti–13Nb–13Zr Alloy against Different Species of Bacteria Related with Implant Infection. Biomed. Mater. 2017, 12, 045022. [Google Scholar] [CrossRef]
- Schrand, A.M.; Rahman, M.F.; Hussain, S.M.; Schlager, J.J.; Smith, D.A.; Syed, A.F. Metal-based Nanoparticles and Their Toxicity Assessment. WIREs Nanomed. Nanobiotechnol. 2010, 2, 544–568. [Google Scholar] [CrossRef]
- Ding, L.; Liu, Z.; Aggrey, M.; Li, C.; Chen, J.; Tong, L. Nanotoxicity: The Toxicity Research Progress of Metal and Metal- Containing Nanoparticles. MRMC 2015, 15, 529–542. [Google Scholar] [CrossRef]
- Ahghari, M.R.; Soltaninejad, V.; Maleki, A. Synthesis of Nickel Nanoparticles by a Green and Convenient Method as a Magnetic Mirror with Antibacterial Activities. Sci. Rep. 2020, 10, 12627. [Google Scholar] [CrossRef] [PubMed]
- Jeyaraj Pandian, C.; Palanivel, R.; Dhanasekaran, S. Screening Antimicrobial Activity of Nickel Nanoparticles Synthesized Using Ocimum Sanctum Leaf Extract. J. Nanopart. 2016, 2016, 1–13. [Google Scholar] [CrossRef]
- Bellio, P.; Luzi, C.; Mancini, A.; Cracchiolo, S.; Passacantando, M.; Di Pietro, L.; Perilli, M.; Amicosante, G.; Santucci, S.; Celenza, G. Cerium Oxide Nanoparticles as Potential Antibiotic Adjuvant. Effects of CeO2 Nanoparticles on Bacterial Outer Membrane Permeability. Biochim. Biophys. Acta (BBA)-Biomembr. 2018, 1860, 2428–2435. [Google Scholar] [CrossRef] [PubMed]
- Vahdati, M.; Tohidi Moghadam, T. Synthesis and Characterization of Selenium Nanoparticles-Lysozyme Nanohybrid System with Synergistic Antibacterial Properties. Sci. Rep. 2020, 10, 510. [Google Scholar] [CrossRef]
- Geoffrion, L.D.; Hesabizadeh, T.; Medina-Cruz, D.; Kusper, M.; Taylor, P.; Vernet-Crua, A.; Chen, J.; Ajo, A.; Webster, T.J.; Guisbiers, G. Naked Selenium Nanoparticles for Antibacterial and Anticancer Treatments. ACS Omega 2020, 5, 2660–2669. [Google Scholar] [CrossRef]
- Kang, S.-M.; Jang, S.-C.; Heo, N.S.; Oh, S.Y.; Cho, H.-J.; Rethinasabapathy, M.; Vilian, A.T.E.; Han, Y.-K.; Roh, C.; Huh, Y.S. Cesium-Induced Inhibition of Bacterial Growth of Pseudomonas Aeruginosa PAO1 and Their Possible Potential Applications for Bioremediation of Wastewater. J. Hazard. Mater. 2017, 338, 323–333. [Google Scholar] [CrossRef]
- Lellouche, J.; Friedman, A.; Gedanken, A.; Banin, E. Antibacterial and Antibiofilm Properties of Yttrium Fluoride Nanoparticles. Int. J. Nanomed. 2012, 7, 5611–5624. [Google Scholar] [CrossRef]
- Adams, C.P.; Walker, K.A.; Obare, S.O.; Docherty, K.M. Size-Dependent Antimicrobial Effects of Novel Palladium Nanoparticles. PLoS ONE 2014, 9, e85981. [Google Scholar] [CrossRef]
- Mohana, S.; Sumathi, S. Multi-Functional Biological Effects of Palladium Nanoparticles Synthesized Using Agaricus Bisporus. J. Clust. Sci. 2020, 31, 391–400. [Google Scholar] [CrossRef]
- Xu, C.; Akakuru, O.U.; Zheng, J.; Wu, A. Applications of Iron Oxide-Based Magnetic Nanoparticles in the Diagnosis and Treatment of Bacterial Infections. Front. Bioeng. Biotechnol. 2019, 7, 141. [Google Scholar] [CrossRef]
- Kim, T.; Zhang, Q.; Li, J.; Zhang, L.; Jokerst, J.V. A Gold/Silver Hybrid Nanoparticle for Treatment and Photoacoustic Imaging of Bacterial Infection. ACS Nano 2018, 12, 5615–5625. [Google Scholar] [CrossRef]
- Wu, W.; Mao, D.; Cai, X.; Duan, Y.; Hu, F.; Kong, D.; Liu, B. ONOO – and ClO – Responsive Organic Nanoparticles for Specific in Vivo Image-Guided Photodynamic Bacterial Ablation. Chem. Mater. 2018, 30, 3867–3873. [Google Scholar] [CrossRef]
- Qin, H.; Cao, H.; Zhao, Y.; Zhu, C.; Cheng, T.; Wang, Q.; Peng, X.; Cheng, M.; Wang, J.; Jin, G.; et al. In Vitro and in Vivo Anti-Biofilm Effects of Silver Nanoparticles Immobilized on Titanium. Biomaterials 2014, 35, 9114–9125. [Google Scholar] [CrossRef]
- Bai, X.; Li, L.; Liu, H.; Tan, L.; Liu, T.; Meng, X. Solvothermal Synthesis of ZnO Nanoparticles and Anti-Infection Application in Vivo. ACS Appl. Mater. Interfaces 2015, 7, 1308–1317. [Google Scholar] [CrossRef]
- Xu, T.; Zhang, J.; Zhu, Y.; Zhao, W.; Pan, C.; Ma, H.; Zhang, L. A Poly(Hydroxyethyl Methacrylate)–Ag Nanoparticle Porous Hydrogel for Simultaneous in Vivo Prevention of the Foreign-Body Reaction and Bacterial Infection. Nanotechnology 2018, 29, 395101. [Google Scholar] [CrossRef]
- Wang, C.; Zhao, W.; Cao, B.; Wang, Z.; Zhou, Q.; Lu, S.; Lu, L.; Zhan, M.; Hu, X. Biofilm-Responsive Polymeric Nanoparticles with Self-Adaptive Deep Penetration for In Vivo Photothermal Treatment of Implant Infection. Chem. Mater. 2020, 32, 7725–7738. [Google Scholar] [CrossRef]
- Cheng, H.; Li, Y.; Huo, K.; Gao, B.; Xiong, W. Long-Lasting in Vivo and in Vitro Antibacterial Ability of Nanostructured Titania Coating Incorporated with Silver Nanoparticles: Antibacterial Ability of Silver Nanoparticles. J. Biomed. Mater. Res. 2014, 102, 3488–3499. [Google Scholar] [CrossRef]
- González, B.; Colilla, M.; Díez, J.; Pedraza, D.; Guembe, M.; Izquierdo-Barba, I.; Vallet-Regí, M. Mesoporous Silica Nanoparticles Decorated with Polycationic Dendrimers for Infection Treatment. Acta Biomater. 2018, 68, 261–271. [Google Scholar] [CrossRef]
- Mas, N.; Galiana, I.; Mondragón, L.; Aznar, E.; Climent, E.; Cabedo, N.; Sancenón, F.; Murguía, J.R.; Martínez-Máñez, R.; Marcos, M.D.; et al. Enhanced Efficacy and Broadening of Antibacterial Action of Drugs via the Use of Capped Mesoporous Nanoparticles. Chem. Eur. J. 2013, 19, 11167–11171. [Google Scholar] [CrossRef]
- Ruehle, B.; Clemens, D.L.; Lee, B.-Y.; Horwitz, M.A.; Zink, J.I. A Pathogen-Specific Cargo Delivery Platform Based on Mesoporous Silica Nanoparticles. J. Am. Chem. Soc. 2017, 139, 6663–6668. [Google Scholar] [CrossRef]
- Xu, T.; Li, J.; Zhang, S.; Jin, Y.; Wang, R. Integration of Diagnosis and Treatment in the Detection and Kill of S.Aureus in the Whole Blood. Biosens. Bioelectron. 2019, 142, 111507. [Google Scholar] [CrossRef] [PubMed]
- Kavruk, M.; Celikbicak, O.; Ozalp, V.C.; Borsa, B.A.; Hernandez, F.J.; Bayramoglu, G.; Salih, B.; Arica, M.Y. Antibiotic Loaded Nanocapsules Functionalized with Aptamer Gates for Targeted Destruction of Pathogens. Chem. Commun. 2015, 51, 8492–8495. [Google Scholar] [CrossRef] [PubMed]
- Yang, S.; Han, X.; Yang, Y.; Qiao, H.; Yu, Z.; Liu, Y.; Wang, J.; Tang, T. Bacteria-Targeting Nanoparticles with Microenvironment-Responsive Antibiotic Release to Eliminate Intracellular Staphylococcus Aureus and Associated Infection. ACS Appl. Mater. Interfaces 2018, 10, 14299–14311. [Google Scholar] [CrossRef]
- Rathnayake, K.; Patel, U.; Pham, C.; McAlpin, A.; Budisalich, T.; Jayawardena, S.N. Targeted Delivery of Antibiotic Therapy to Inhibit Pseudomonas aeruginosa Using Lipid-Coated Mesoporous Silica Core–Shell Nanoassembly. ACS Appl. Bio Mater. 2020, 3, 6708–6721. [Google Scholar] [CrossRef]
- Zhou, J.; Jayawardana, K.W.; Kong, N.; Ren, Y.; Hao, N.; Yan, M.; Ramström, O. Trehalose-Conjugated, Photofunctionalized Mesoporous Silica Nanoparticles for Efficient Delivery of Isoniazid into Mycobacteria. ACS Biomater. Sci. Eng. 2015, 1, 1250–1255. [Google Scholar] [CrossRef]
- Mudakavi, R.J.; Vanamali, S.; Chakravortty, D.; Raichur, A.M. Development of Arginine Based Nanocarriers for Targeting and Treatment of Intracellular Salmonella. RSC Adv. 2017, 7, 7022–7032. [Google Scholar] [CrossRef]
- Chen, X.; Liu, Y.; Lin, A.; Huang, N.; Long, L.; Gang, Y.; Liu, J. Folic Acid-Modified Mesoporous Silica Nanoparticles with PH-Responsiveness Loaded with Amp for an Enhanced Effect against Anti-Drug-Resistant Bacteria by Overcoming Efflux Pump Systems. Biomater. Sci. 2018, 6, 1923–1935. [Google Scholar] [CrossRef]
- Qi, G.; Li, L.; Yu, F.; Wang, H. Vancomycin-Modified Mesoporous Silica Nanoparticles for Selective Recognition and Killing of Pathogenic Gram-Positive Bacteria Over Macrophage-Like Cells. ACS Appl. Mater. Interfaces 2013, 5, 10874–10881. [Google Scholar] [CrossRef]
- Pedraza, D.; Díez, J.; Isabel-Izquierdo-Barba; Colilla, M.; Vallet-Regí, M. Amine-Functionalized Mesoporous Silica Nanoparticles: A New Nanoantibiotic for Bone Infection Treatment. Biomed. Glasses 2018, 4, 1–12. [Google Scholar] [CrossRef]
- Martínez-Carmona, M.; Izquierdo-Barba, I.; Colilla, M.; Vallet-Regí, M. Concanavalin A-Targeted Mesoporous Silica Nanoparticles for Infection Treatment. Acta Biomater. 2019, 96, 547–556. [Google Scholar] [CrossRef]
- LiPuma, J.J.; Rathinavelu, S.; Foster, B.K.; Keoleian, J.C.; Makidon, P.E.; Kalikin, L.M.; Baker, J.R. In Vitro Activities of a Novel Nanoemulsion against Burkholderia and Other Multidrug-Resistant Cystic Fibrosis-Associated Bacterial Species. Antimicrob. Agents Chemother. 2009, 53, 249–255. [Google Scholar] [CrossRef] [PubMed]
- Hemmila, M.R.; Mattar, A.; Taddonio, M.A.; Arbabi, S.; Hamouda, T.; Ward, P.A.; Wang, S.C.; Baker, J.R. Topical Nanoemulsion Therapy Reduces Bacterial Wound Infection and Inflammation after Burn Injury. Surgery 2010, 148, 499–509. [Google Scholar] [CrossRef] [PubMed]
- Hwang, Y.Y.; Ramalingam, K.; Bienek, D.R.; Lee, V.; You, T.; Alvarez, R. Antimicrobial Activity of Nanoemulsion in Combination with Cetylpyridinium Chloride in Multidrug-Resistant Acinetobacter Baumannii. Antimicrob. Agents Chemother. 2013, 57, 3568–3575. [Google Scholar] [CrossRef]
- Ghaderi, L.; Moghimi, R.; Aliahmadi, A.; McClements, D.J.; Rafati, H. Development of Antimicrobial Nanoemulsion-Based Delivery Systems against Selected Pathogenic Bacteria Using a Thymol-Rich Thymus Daenensis Essential Oil. J. Appl. Microbiol. 2017, 123, 832–840. [Google Scholar] [CrossRef] [PubMed]
- Krishnamoorthy, R.; Athinarayanan, J.; Periasamy, V.S.; Adisa, A.R.; Al-Shuniaber, M.A.; Gassem, M.A.; Alshatwi, A.A. Antimicrobial Activity of Nanoemulsion on Drug-Resistant Bacterial Pathogens. Microb. Pathog. 2018, 120, 85–96. [Google Scholar] [CrossRef]
- Coune, A. Liposomes as Drug Delivery System in the Treatment of Infectious Diseases. Potential Applications and Clinical Experience. Infection 1988, 16, 141–147. [Google Scholar] [CrossRef]
- Henry, B.D.; Neill, D.R.; Becker, K.A.; Gore, S.; Bricio-Moreno, L.; Ziobro, R.; Edwards, M.J.; Mühlemann, K.; Steinmann, J.; Kleuser, B.; et al. Engineered Liposomes Sequester Bacterial Exotoxins and Protect from Severe Invasive Infections in Mice. Nat. Biotechnol. 2015, 33, 81–88. [Google Scholar] [CrossRef]
- Jiménez-Jiménez, C.; Manzano, M.; Vallet-Regí, M. Nanoparticles Coated with Cell Membranes for Biomedical Applications. Biology 2020, 9, 406. [Google Scholar] [CrossRef]
- Gao, F.; Xu, L.; Yang, B.; Fan, F.; Yang, L. Kill the Real with the Fake: Eliminate Intracellular Staphylococcus Aureus Using Nanoparticle Coated with Its Extracellular Vesicle Membrane as Active-Targeting Drug Carrier. ACS Infect. Dis. 2019, 5, 218–227. [Google Scholar] [CrossRef]
- Whitlow, C.B. Bacterial Sexually Transmitted Diseases. Clin. Colon Rectal. Surg. 2004, 17, 209–214. [Google Scholar] [CrossRef]
- Li, L.-H.; Yen, M.-Y.; Ho, C.-C.; Wu, P.; Wang, C.-C.; Maurya, P.K.; Chen, P.-S.; Chen, W.; Hsieh, W.-Y.; Chen, H.-W. Non-Cytotoxic Nanomaterials Enhance Antimicrobial Activities of Cefmetazole against Multidrug-Resistant Neisseria Gonorrhoeae. PLoS ONE 2013, 8, e64794. [Google Scholar] [CrossRef]
- Lucío, M.I.; Kyriazi, M.-E.; Hamilton, J.; Batista, D.; Sheppard, A.; Sams-Dodd, E.; Humbert, M.V.; Hussain, I.; Christodoulides, M.; Kanaras, A.G. Bactericidal Effect of 5-Mercapto-2-Nitrobenzoic Acid-Coated Silver Nanoclusters against Multidrug-Resistant Neisseria Gonorrhoeae. ACS Appl. Mater. Interfaces 2020, 12, 27994–28003. [Google Scholar] [CrossRef]
- Alqahtani, F.; Aleanizy, F.; El Tahir, E.; Alhabib, H.; Alsaif, R.; Shazly, G.; AlQahtani, H.; Alsarra, I.; Mahdavi, J. Antibacterial Activity of Chitosan Nanoparticles Against Pathogenic N. Gonorrhoea. Int. J. Nanomed. 2020, 15, 7877–7887. [Google Scholar] [CrossRef]
- Yang, S.; Traore, Y.; Jimenez, C.; Ho, E.A. Autophagy Induction and PDGFR-β Knockdown by SiRNA-Encapsulated Nanoparticles Reduce Chlamydia Trachomatis Infection. Sci. Rep. 2019, 9, 1306. [Google Scholar] [CrossRef]
- Costerton, J.W. Bacterial Biofilms: A Common Cause of Persistent Infections. Science 1999, 284, 1318–1322. [Google Scholar] [CrossRef]
- Parsek, M.R.; Greenberg, E.P. Sociomicrobiology: The Connections between Quorum Sensing and Biofilms. Trends in Microbiology 2005, 13, 27–33. [Google Scholar] [CrossRef]
- Deva, A.K.; Adams, W.P.; Vickery, K. The Role of Bacterial Biofilms in Device-Associated Infection. Plast. Reconstr. Surg. 2013, 132, 1319–1328. [Google Scholar] [CrossRef]
- Lewis, K. Riddle of Biofilm Resistance. Antimicrob. Agents Chemother. 2001, 45, 999–1007. [Google Scholar] [CrossRef]
- Jamal, M.; Ahmad, W.; Andleeb, S.; Jalil, F.; Imran, M.; Nawaz, M.A.; Hussain, T.; Ali, M.; Rafiq, M.; Kamil, M.A. Bacterial Biofilm and Associated Infections. J. Chin. Med Assoc. 2018, 81, 7–11. [Google Scholar] [CrossRef]
- Lebeaux, D.; Ghigo, J.-M.; Beloin, C. Biofilm-Related Infections: Bridging the Gap between Clinical Management and Fundamental Aspects of Recalcitrance toward Antibiotics. Microbiol. Mol. Biol. Rev. 2014, 78, 510–543. [Google Scholar] [CrossRef]
- Colilla, M.; Vallet-Regí, M. Targeted Stimuli-Responsive Mesoporous Silica Nanoparticles for Bacterial Infection Treatment. Int. J. Mol. Sci. 2020, 21, 8605. [Google Scholar] [CrossRef] [PubMed]
- Vallet-Regí, M.; Lozano, D.; González, B.; Izquierdo-Barba, I. Biomaterials against Bone Infection. Adv. Healthcare Mater. 2020, 9, 2000310. [Google Scholar] [CrossRef]
- Castillo, R.R.; Lozano, D.; Vallet-Regí, M. Mesoporous Silica Nanoparticles as Carriers for Therapeutic Biomolecules. Pharmaceutics 2020, 12, 432. [Google Scholar] [CrossRef] [PubMed]
- Villegas, M.F.; Garcia-Uriostegui, L.; Rodríguez, O.; Izquierdo-Barba, I.; Salinas, A.J.; Toriz, G.; Vallet-Regí, M.; Delgado, E. Lysine-Grafted MCM-41 Silica as an Antibacterial Biomaterial. Bioengineering 2017, 4, 80. [Google Scholar] [CrossRef]
- Zhao, Y.; Sun, X.; Zhang, G.; Trewyn, B.G.; Slowing, I.I.; Lin, V.S.-Y. Interaction of Mesoporous Silica Nanoparticles with Human Red Blood Cell Membranes: Size and Surface Effects. ACS Nano 2011, 5, 1366–1375. [Google Scholar] [CrossRef]
| Virus | Nanoparticles (NPs) w/o Conjugate | Size (nm) | Nanoparticle Role | Action Mechanism | Cytotoxicity (%) | Level Study | Cell Lines/Animal Used In Vivo | Reference |
|---|---|---|---|---|---|---|---|---|
| Influenza A virus (subtype H1N1) | Ag NPs conjugated with oseltamivir | 3 | Antimicrobial and nanocarrier | Viral deformation/viral entry blocking/apoptosis inhibition | <10 | in vitro | MDCK | [39] |
| Ag NPs conjugated with zanamivir | 3 | Antimicrobial and nanocarrier | Viral deformation/ viral entry blocking/apoptosis inhibition | <20 | in vitro | MDCK | [40] | |
| Au NPs with sialic acid | 14 | Antimicrobial | Viral entry blocking | <1 | in vitro | MDCK | [41] | |
| Liposome loaded with glycan sialylneolacto-N-tetraose | 1–1000 | Nanocarrier | Viral entry blocking | - | in vitro/in vivo | MDCK/C57BL/6 mice | [42] | |
| Se NPs conjugated with zanamivir | 82 | Antimicrobial and nanocarrier | Viral entry blocking/apoptosis inhibition | <50 | in vitro | MDCK | [43] | |
| Se NPs conjugated with amantadine | 70 | Antimicrobial and nanocarrier | Viral entry blocking/apoptosis inhibition | <20 | in vitro | MDCK | [44] | |
| Se NPs conjugated with ribavirin | 65 | Antimicrobial and nanocarrier | Viral entry blocking/apoptosis inhibition | <20 | in vitro | MDCK | [45] | |
| Se NPs conjugated with oseltamivir | 100 | Antimicrobial and nanocarrier | Viral entry blocking/apoptosis inhibition | <10 | in vitro | MDCK | [46] | |
| (H1N1, H3N2, and H9N1 subtypes) | Porous AuNPs | 154 ± 37 | Antimicrobial | Viral entry blocking | <5 | in vitro | MDCK | [47] |
| (subtypes H1N1, H3N2, PR8, B-Bris, and B-Shan) | Au NPs conjugated with MES | 4 ± 1 | Antimicrobial | Infection inhibition | - | in vitro | MDCK | [48] |
| TiO2 NPs conjugated polylysines w/o DNA | >5 | Antimicrobial | Virus replication inhibition | - | in vitro | MDCK | [49] | |
| ZnO NPs conjugated with PEG | 18 | Antimicrobial | Viral inactivation | <20 | in vitro | MDCK | [50] | |
| (H3N2 subtype) | Ginkgo biloba leaves polyphenols nanoemulsions | 389–988 | Nanocarrier | Virucidal and protective effect | 0 | in vitro | MDCK | [51] |
| Respiratory syncytial virus | Ag NPs | 10 | Antimicrobial | Viral replication inhibition | 0 | in vitro/in vivo | A549, a human alveolar type II-like epithelial cell, and HEp-2/BALB/c mice | [52] |
| Ag NPs with curcumin | 20 | Antimicrobial | Viral entry blocking | <5 | in vitro | HEp-2 | [53] | |
| Au NPs conjugated with MES/MUS-OT | 2.5 ± 0.7 | Antimicrobial | Viral inactivation/viral deformation | 0 | in vitro/in vivo | HeLa, HEK 293T, CHO-K1, Vero, Hep-2, and HT-1080/BALB/c mice | [54] | |
| Porous Si NPs | 5–50 | Antimicrobial | Viral entry blocking | 0 | in vitro | CEM SS and MA-104 | [55] | |
| MERS | Au nanorod conjugated with PH-petide+PEG | 54 ± 18 | Antimicrobial | Viral entry blocking | 0 | in vitro/in vivo | Huh-7, 293T, and L02/ICR mice | [56] |
| Virus | Nanoparticles (NPs) w/o Conjugate | Size (nm) | Nanoparticle Role | Action Mechanism | Cytotoxicity (%) | Level Study | Cell Lines/Animal Used In Vivo | Reference |
|---|---|---|---|---|---|---|---|---|
| HIV | PVP-coated Ag NPs | 1–10 | Antimicrobial | Cyto-protection | - | in vitro | Hut-CCR5 | [74] |
| Au NPs conjugated with peptide triazoles | 13–123 | Antimicrobial | Viral inactivation | - | in vitro | HOS.T4.R, 293T | [75] | |
| Au NPs coated with sulfate-ended ligand | 2 | Antimicrobial | Infection inhibition | 0 | in vitro | MT-2 | [76] | |
| Carboxilan dendrimers | - | Antimicrobial | Infection inhibition | <20 | in vitro | human CD4+, CD25+, CD127low | [77] | |
| Carboxilan dendrimers conjugated with RNA decoy | - | Nanocarrier | Cyto-protetion | <20 | in vitro | MT4 | [78] | |
| Fe3O4 NPs loaded with tenofovir+dextran sulphate] + vorinostat | 10 ± 3 | Nanocarrier | Latency-breaking | 0 | in vitro | MTT, primary human astrocytes | [79] | |
| PEG-MA NPs loaded with SMAPP1 | - | Nanocarrier | Latency-breaking | - | in vitro | CCRP-CEM | [80] | |
| PLGA NPs loaded with efavirenz or saquinavir | 340 | Nanocarrier | Infection inhibition/infection treatment | 0 | in vitro/ex vivo | TZM-bL, PM-1 and CEMx174/macaque cervicovaginal tissue | [81] | |
| PLGA NPs loaded with maraviroc, etravirine, and/or raltegravir | 200 | Nanocarrier | Infection inhibition/infection treatment | <20 | in vitro/ex vivo | TZM-bl/human ectocervical explants | [82] | |
| Porous Si NPs | 5–50 | Antimicrobial | Viral entry blocking | 0 | in vitro | CEM SS | [55] | |
| SiO2 NPs conjugated with GPTMS, APTES, and TMPES | 354 | Nanocarrier | Infection inhibition | 0 | in vitro | HEK 293T | [83] | |
| HSV-1 | Fe3O4 SiO2 NPs conjugated with biguanide, polymeric aziridine | 150–250 | Antimicrobial and nanocarrier | Viral inactivation | <20 | in vitro | Vero | |
| MES-coated Ag NPs | 4 | Antimicrobial | Infection inhibition | 0 | in vitro | Vero | [84] | |
| PLA NPs loaded with cloroquine | <300 | Nanocarrier | Viral entry blocking | <30 | in vitro | Vero | [85] | |
| PLGA nanosphere loaded with acyclovir | 190–700 | Nanocarrier | Treatment tolerance | - | in vivo | Rabbit | [86] | |
| HSV-2 | Liposomes with siRNA | - | Nanocarrier | Infection inhibition | 0 | in vitro/in vivo | NIH3T3, Vero/BALBc mice | [87] |
| PVC NPs loaded with acyclovir | 400 ± 6 | Nanocarrier | Infection treatment | - | in vivo | Wistar rats | [88,89] | |
| HSV-1/2 | Mycosinthetized Ag NPs | 4–46 | Antimicrobial | Infection inhibition | - | in vitro | Vero | [90] |
| HBV | Ag NPs | 10–50 | Antimicrobial | Viral inactivation/Infection inhibition | <50 | in vitro | HepAD38 | [1] |
| (mPEG)–PLA/PEI, mPEG–PLA–chitosan NPs loaded with siRNA | 500–800 | Antimicrobial and nanocarrier | Inhibition of the HBV surface antigen | <6 | in vitro | PLC/PRF/5 c | [91] | |
| Ginkgo biloba leaves polyphenols nanoemulsions | 389–988 | Nanocarrier | Inhibition of the HBV surface antigen | 0 | in vitro | HepG 2215 | [51] | |
| HCV | PEG-PLDn+PEG-PLEm NPs loaded with antiviral peptides | 20–40 | Nanocarrier | Cyto-protection | 0 | in vitro | Huh-7.5 | [92] |
| Anionic poly(amino acid)-based NPs loaded with antiviral peptides | 108 | Nanocarrier | Infection treatment | 0 | in vitro/ in vivo | Huh-7.5/BALBc mice | [93] | |
| Chitosan-TTP NPs loaded with siRNA | <500 | Nanocarrier | Infection treatment | <10 | in vitro | CHO K1 | [94] | |
| Solid lipid NPs loaded with RNAi | 240 | Nanocarrier | Infection treatment | 10 | in vitro | HepG2 | [95] | |
| Cu NPs | 45.4 ± 6.8 | Antimicrobial | Viral entry blocking | <5 | in vitro | Huh-7.5 | [96] | |
| Dextran-coated magnetic Fe oxide NP conjugated with DNAzyme | 75–80 | Nanocarrier | Inhibition of expression of the HCV NS3 gene | - | in vitro/in vivo | Huh-7 Luc-Neo/BALBc mice | [97] | |
| Silibinin-encapsulated liposome | 129 ± 3 | Nanocarrier | Viral entry blocking/viral inactivation | <20 | in vitro | Huh7.5, Huh7.5/Conl/FL-Neo | [98] | |
| Aptamer-functionalized Fe2O3 NPs | 100 | Antimicrobial | Viral inactivation | - | ex vivo | Human plasma | [99] | |
| Polyanionic carbosilane dendrimers w/o sofosbuvir | - | Nanocarrier | Viral entry blocking | 0 | in vitro | Huh-7, Huh-7.5.1, Huh-7.5.1-c2 | [100] |
| Nanoparticles (NPs) w/o Conjugate | Size (nm) | Nanoparticle Role | Action Mechanism | Bacterial Species | Application | Life Style | Cytotoxicity (%) | Level Study | Cell Lines/Animal Used In Vivo | Reference |
|---|---|---|---|---|---|---|---|---|---|---|
| Au/Ag nanorods | 12–14 × 50–55 | Antimicrobial | Ag+ release | E. coli, MRSA | S. aureus-related and E. coli-related infections | Planktonic | 0 | In vitro/in vivo | ‒/C57BL6 mice | [175] |
| TBD-PEG NPs loaded with IR786S, ONOO- and ClO- | 50 | Nanocarrier | ROS generation | E. coli | Wound infection and abscessus cauded by E. coli | Biofilm | ‒ | ‒/In vivo | ‒/BALBc mice | [176] |
| Ag NPs anchored on Ti surface | 18–24 | Antimicrobial | Ag+ release | S. epidermidis | Metallic implant infection | Planktonic/biofilm | <10 | In vitro/in vivo | MC3T3-E1/Sprague Dawley rats | [177] |
| ZnO NPs dispersed in a polyvinyl alcohol gel | 4–10 | Antimicrobial | ROS generation | E. coli | Vaginitis | Planktonic | <20 | In vitro/in vivo | HepG-2, A-431/ ICR mice | [178] |
| Ag NPs in a poly (hydroxyethyl methacrylate) gel | ‒ | Antimicrobial | Ag+ release | S. aureus/E. coli | Idwelling implant infection | Planktonic | <20 | In vitro/in vivo | NIH-3T3/BALBc mice | [179] |
| P(GEMADA-co-DMA)-b-PBMA NPs loaded with guanidine lighted with NIR laser | 50 | Nanocarrier | ROS generation | S. aureus | Catheter infection | Planktonic/biofilm | ‒ | In vitro/in vivo | ‒/BALBc mice | [180] |
| Ag NPs loaded into Ti nanotubes | 10–20 | Antimicrobial | Ag+ release | MRSA | Metallic implant infection | Biofilm | ‒ | In vitro/in vivo | MC3T3-E1/Sprague Dawley rats | [181] |
| Dendrimer-coated MSNPs load with levofloxacin | 150 | Nanocarrier | Antibiotic-bactericidal effect favored by dendrimer | E. coli | E. coli biofilm-related infection | Planktonic/biofilm | ‒ | In vitro | ‒ | [182] |
| ε-poly-lysine-coated MSNPs were loaded with histidine kinase autophosphorylation inhibitors (HKAI) | 100 | Nanocarrier | HKAI-inhibitory effect | E. coli, Serratia marcensens | Enterobacterial infection | Planktonic | 0 | In vitro/in vivo | Caco-2 BBE, RAW 264.7/Zebra fish | [183] |
| EDC/NHS or ICPTES-functionalized mesoporous SiO2 NPs | ‒ | Nanocarrier | Antibiotic-bactericidal effect of the possible loaded antibiotic | Francisella tularensis | Turalemia | Planktonic | ‒ | In vitro | ‒ | [184] |
| Anti-S. aureus antibody-coated Fe3O4/MSNPs loaded with vancomycin | 250 | Nanocarrier | Vacomycin-bactericidal effect | S. aureus | S. aureus-associated bacteremia | Planktonic | ‒ | In vitro | Erytrocyte | [185] |
| Aptemr-gated MSNPs loaded with vancomycin | 177.5 | Nanocarrier | Vacomycin-bactericidal effect | S. aureus/S. epidermidis | Staphylococcal infections | Planktonic | ‒ | ‒ | ‒ | [186] |
| Lipidids-coated MSNPs loaded with gentamicin and conjugated with UBI29–41 | 81.2–99.5 | Nanocarrier | Gentamicin-bacteriostatic effect | S. aureus | S. aureus-associated bone infections | Planktonic/Intracellular | <20 | In vitro | MC3T3-E1, RAW 264.7 | [187] |
| Lipidids-coated MSNPs loaded with colistin and conjugated with LL-37 | 80–99.6 | Nanocarrier | Colisitn-bactericidal effect | P. aeruginosa | P. aeruginosa-associated pulmonar infections | Planktonic/Intracellular | <20 | In vitro | A549 | [188] |
| Trehalose-coated PFPA-functionalized MSNPs loaded with isozianid | 154-188 | Nanocarrier | Isozianid-bactericidal effect | M. smegmatis | Mycobacterial infections | Planktonic | ‒ | ‒ | ‒ | [189] |
| Arginine-coated MSNPs loaded with ciprofloxacin | 75 | Nanocarrier | Ciprofloxacin-bactericidal effect | Salmonella enterica serovar typhimurium | Salmonellosis | Planktonic | Low | In vitro/in vivo | RAW 264.7/BALBc mice | [190] |
| FA-CP-FA-coated MSNPs loaded with ampicilin | 80 | Nanocarrier | Ampicilin-bactericidal effect | S. aureus/E. coli | S. aureus-related and E. coli-related infections | Planktonic | 0 | In vitro/in vivo | HEK 2931T/Kin Ming mice | [191] |
| Vancomycin-functionalized mesoporous SiO2 NPs | 90–127 | Nanocarrier | Vacomycin-bactericidal effect | S. aureus/E. coli | S. aureus-related infections | Planktonic | ‒ | In vitro/in vivo | ‒/BALBc mice | [192] |
| Concavalin-functionalized MSNPs loaded with levofloxacin | 120 | Nanocarrier | Levofloxacin-bactericidal effect | S. aureus | S. aureus-related infections | Planktonic/biofilm | ‒ | In vitro | ‒ | [193] |
| Amino-functionalized MSNPs loaded with levofloxacin | 150 | Nanocarrier | Levofloxacin-bactericidal effect | E. coli | E. coli-related infection | Planktonic/biofilm | <10 | In vitro | MC3T3-E1 | [194] |
| NB-401 nanoemulsions | 400 | Nanocarrier | NB-401-bactericidal effect | P. aeruginosa, A. xylosoxidans, S. maltophilia, Acinetobacter species, Pandoraea species), and Ralstonia species | Non-fermenting Gram-negative bacteria-related respiratory infections | Planktonic/biofilm | ‒ | In vitro | ‒ | [195] |
| NB-201 nanoemulsions | 350 | Nanocarrier | NB-201-bactericidal effect | P. aeruginosa | P. aeruginosa burn infections | Planktonic/biofilm | ‒ | In vivo | Sprague-Dawley rats | [196] |
| N5 and cetylpyridinium chloride nanoemulsions | 153 | Nanocarrier | Cetylpyridinium-bactericidal effect | Acinetobacter baumannii | Acinetobacter baumannii infections | Planktonic/biofilm | ‒ | In vitro | ‒ | [197] |
| Thymus daenensis oil nanoemulsions | 131 | Antimicrobial | Oil-bactericidal effect | Haemophilus influenzae, Pseudomonas aeruginosa, and Streptococcus pneumoniae | Pneumococcal infections | Planctonic | ‒ | In vitro/in vivo | ‒ | [198] |
| Cleome viscosa oil nanoemulsions | 86 | Antimicrobial | Oil-bactericidal effect | Methicillin-resistant Staphylococcus aureus, drug-resistant Streptococcus pyogenes, and extended spectrum beta-lactamase-producing Escherichia coli, Klebsiella pneumoniae, and Pseudomonas aeruginosa | Multidrug-resistance bacterial infections | Planctonic | ‒ | In vitro/in vivo | ‒ | [199] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Aguilera-Correa, J.J.; Esteban, J.; Vallet-Regí, M. Inorganic and Polymeric Nanoparticles for Human Viral and Bacterial Infections Prevention and Treatment. Nanomaterials 2021, 11, 137. https://doi.org/10.3390/nano11010137
Aguilera-Correa JJ, Esteban J, Vallet-Regí M. Inorganic and Polymeric Nanoparticles for Human Viral and Bacterial Infections Prevention and Treatment. Nanomaterials. 2021; 11(1):137. https://doi.org/10.3390/nano11010137
Chicago/Turabian StyleAguilera-Correa, John Jairo, Jaime Esteban, and María Vallet-Regí. 2021. "Inorganic and Polymeric Nanoparticles for Human Viral and Bacterial Infections Prevention and Treatment" Nanomaterials 11, no. 1: 137. https://doi.org/10.3390/nano11010137
APA StyleAguilera-Correa, J. J., Esteban, J., & Vallet-Regí, M. (2021). Inorganic and Polymeric Nanoparticles for Human Viral and Bacterial Infections Prevention and Treatment. Nanomaterials, 11(1), 137. https://doi.org/10.3390/nano11010137







