A Novel Experimental Study on the Rheological Properties and Thermal Conductivity of Halloysite Nanofluids
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Preparation of Nanofluids
2.3. Characterization Techniques
3. Results and Discussion
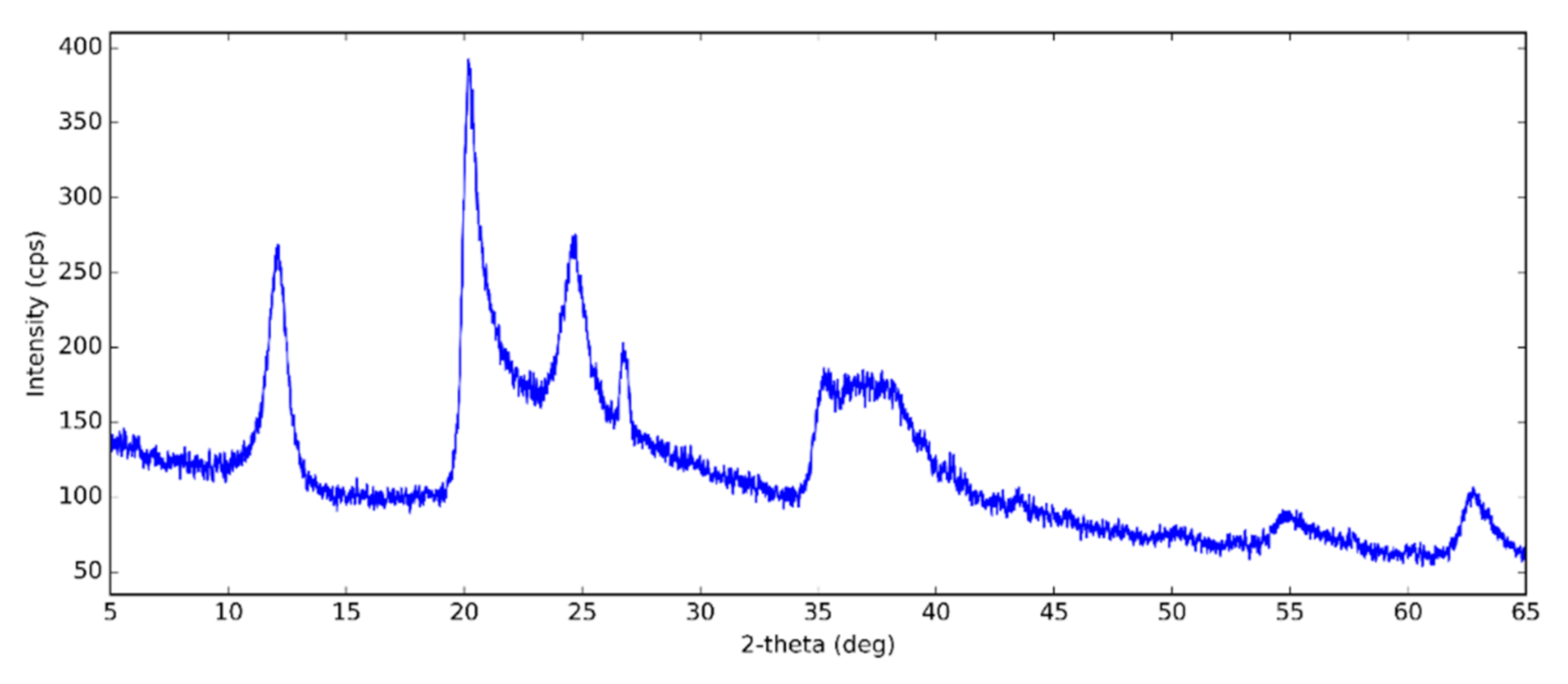
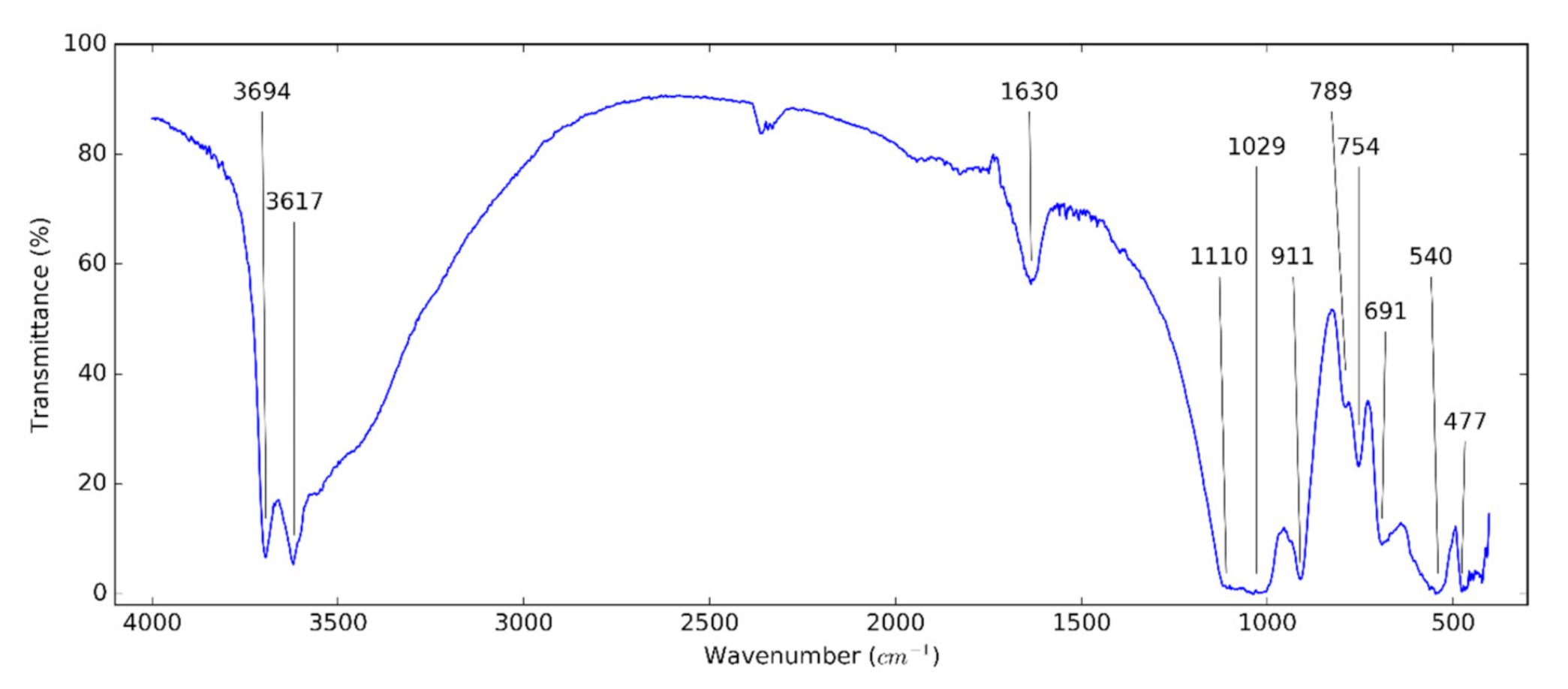
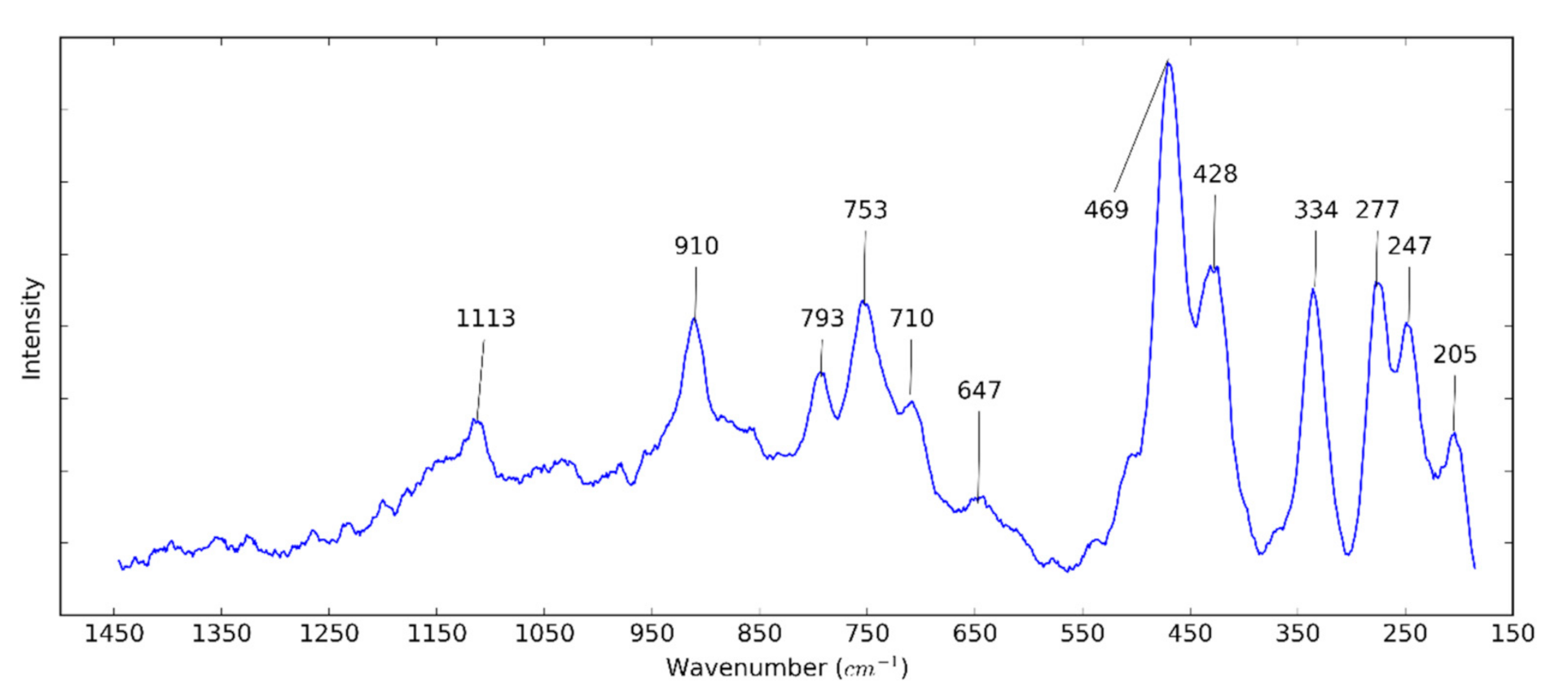
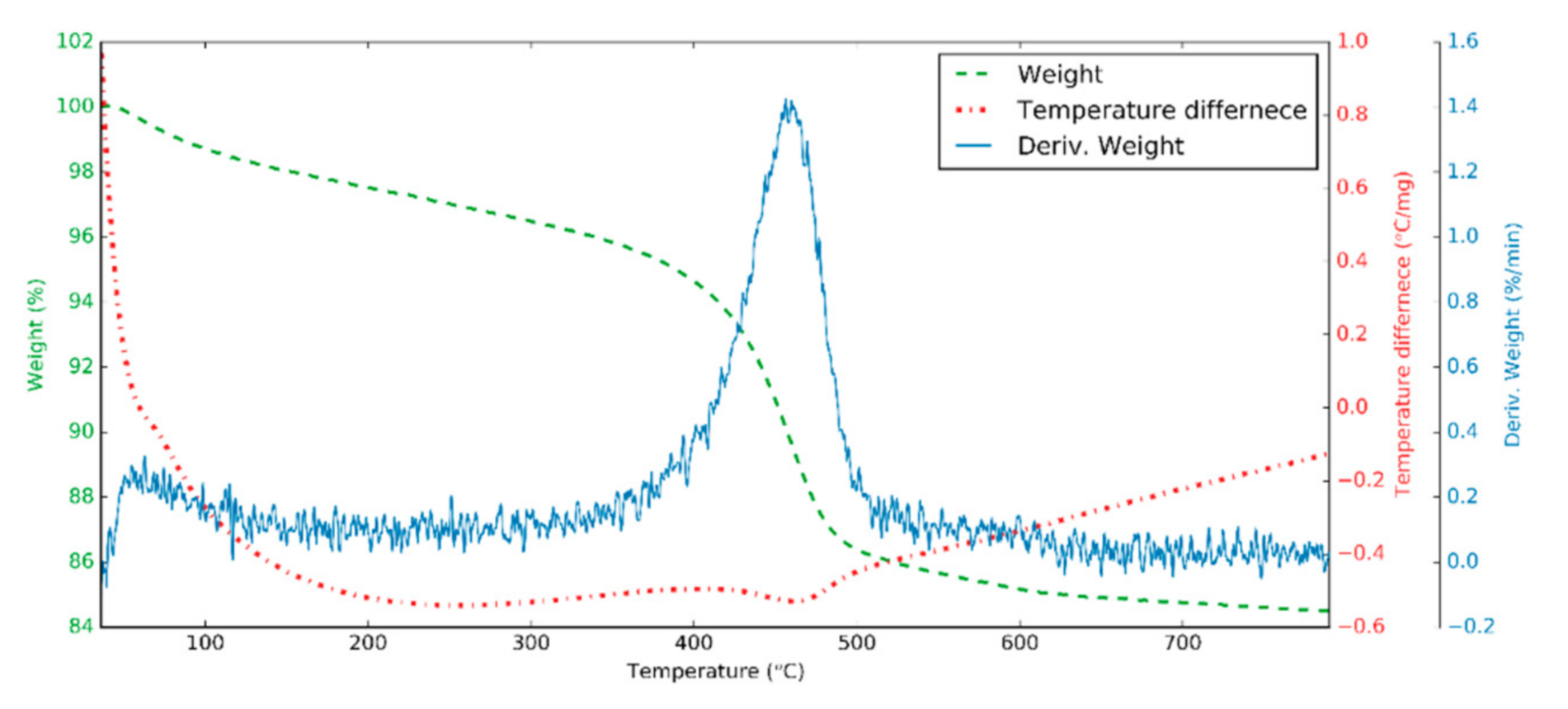
3.1. Halloysite Structure
3.2. Zeta Potential Measurement
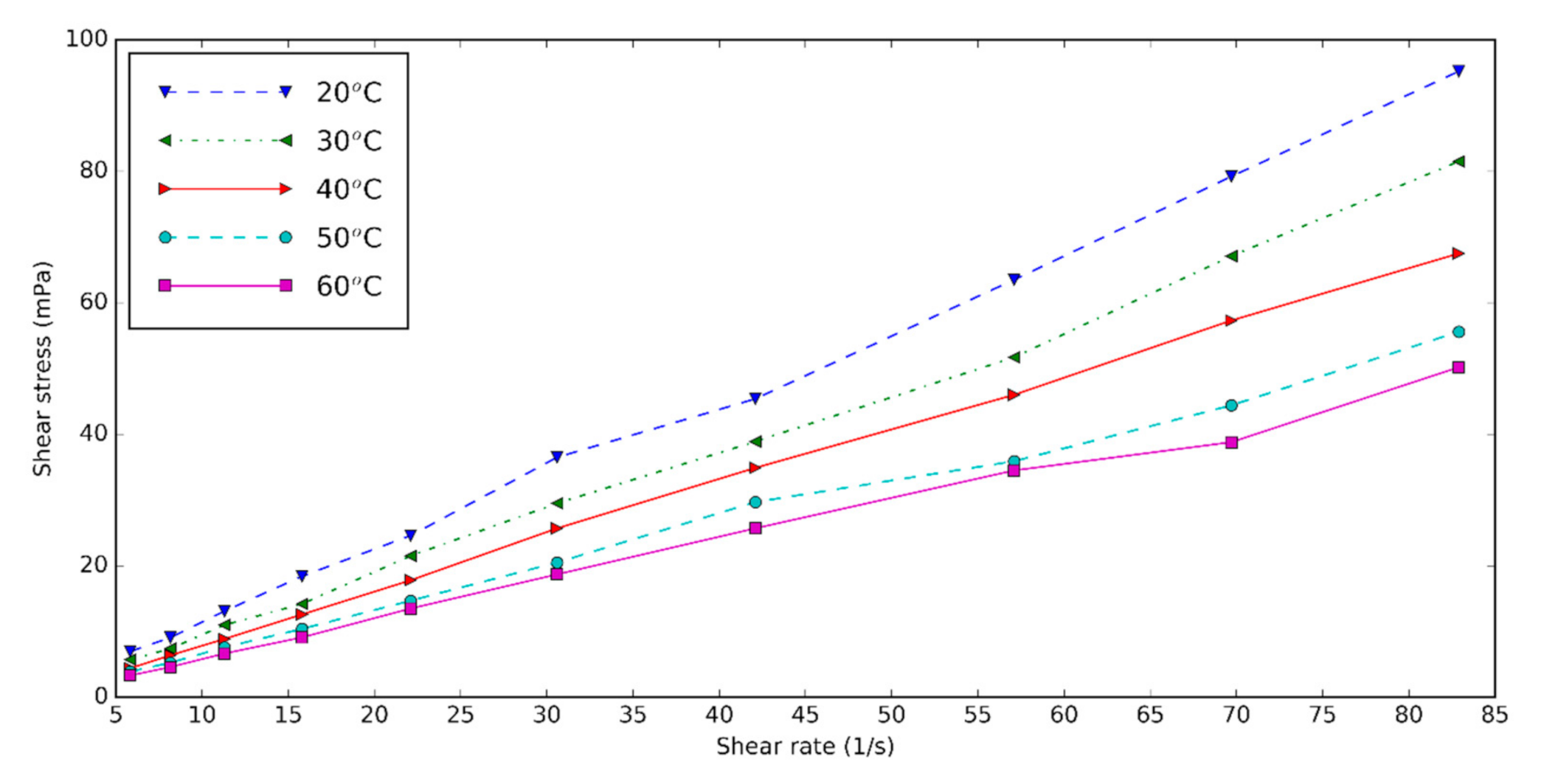
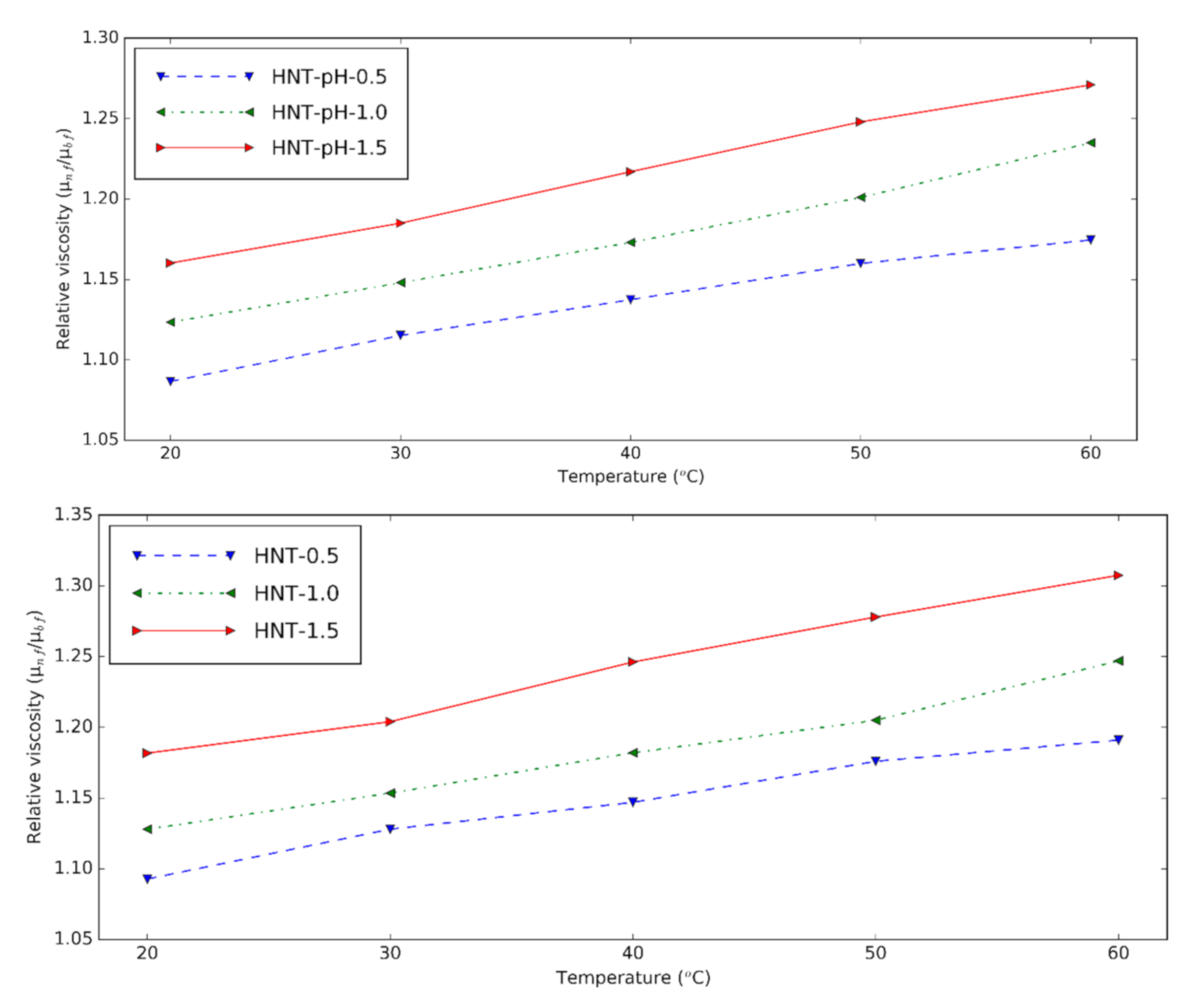
3.3. Rheological Properties of Halloysite Nanofluid
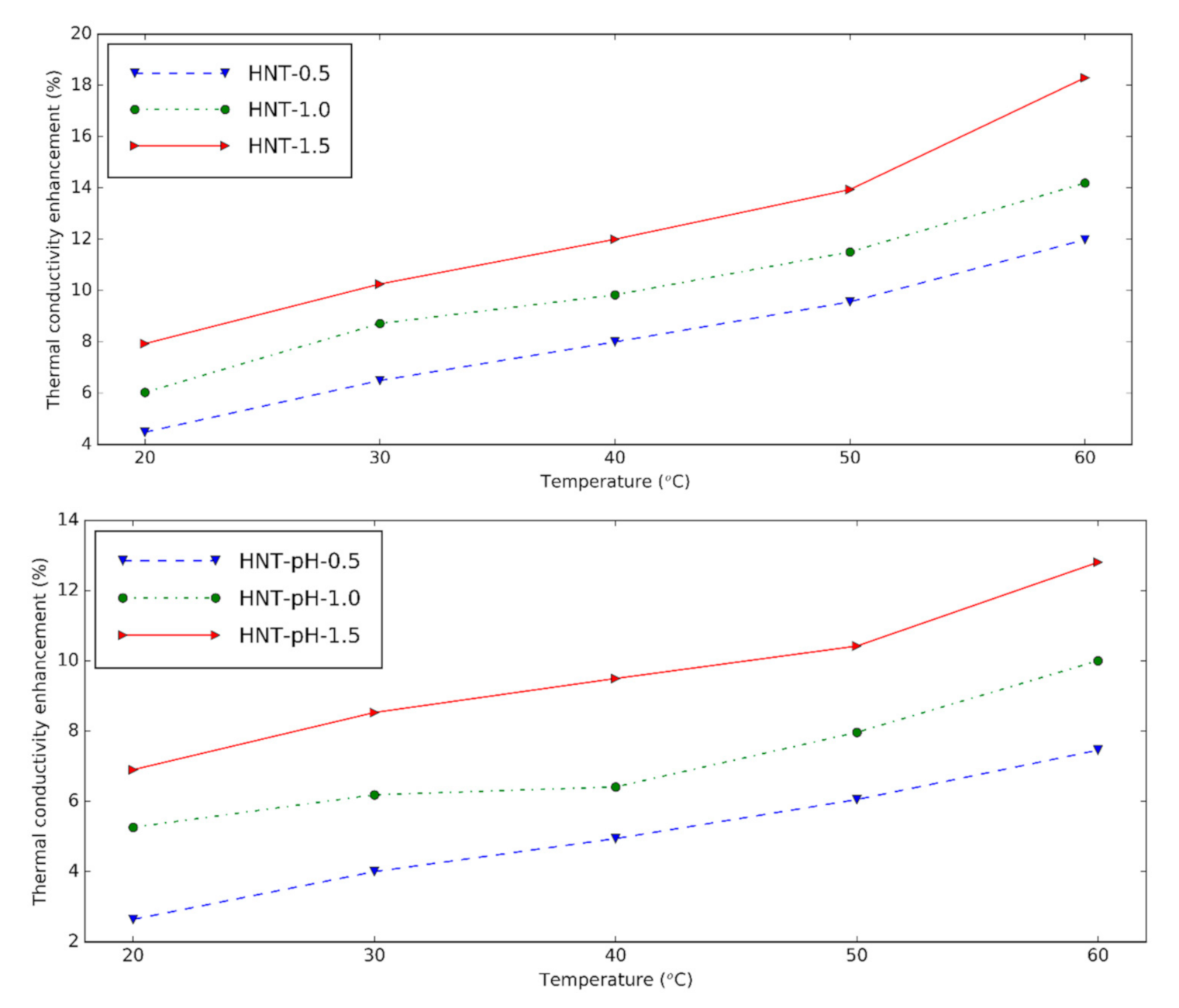
3.4. Thermal Conductivity of Halloysite Nanofluid
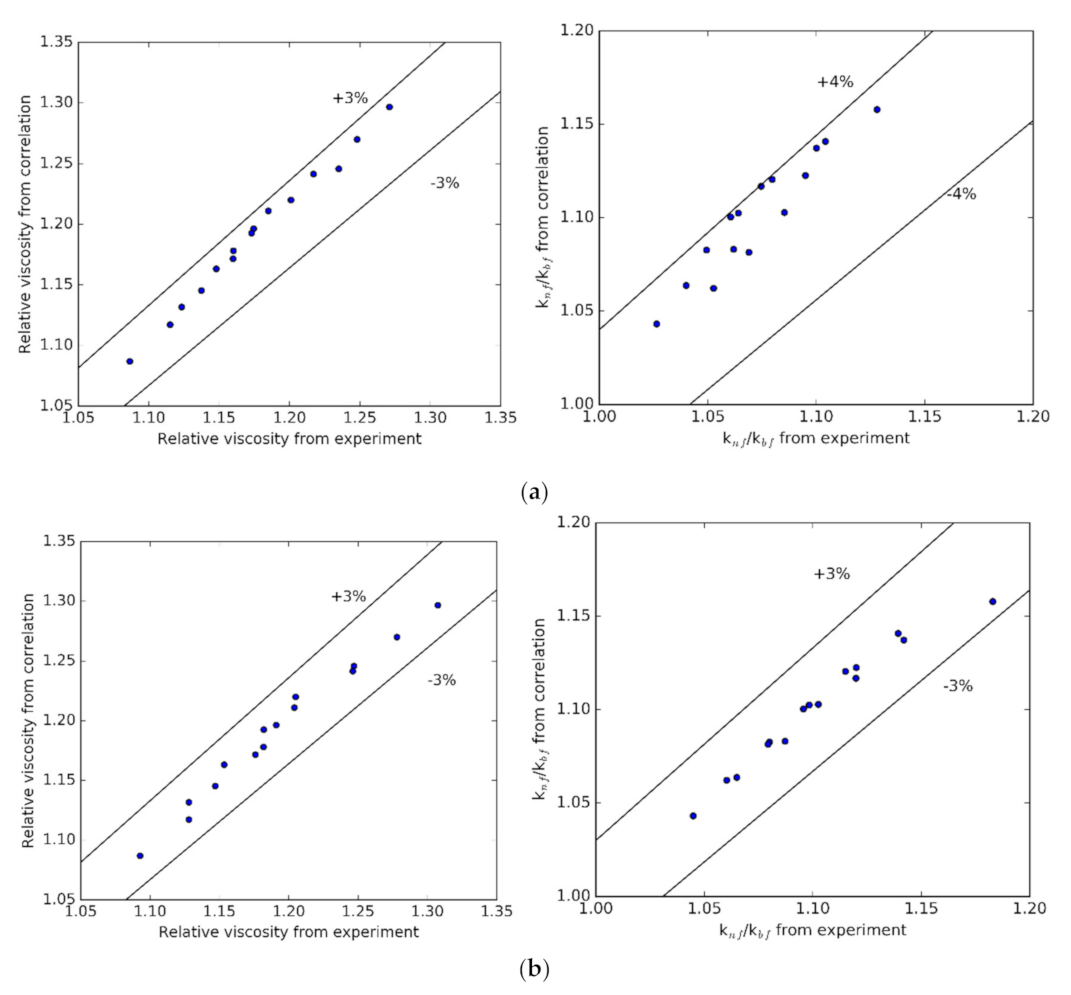
3.5. Regression Correlations
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Fan, J.; Wang, L. Review of Heat Conduction in Nanofluids. J. Heat Transf. 2011, 133. [Google Scholar] [CrossRef]
- Godson, L.; Raja, B.; Lal, D.M.; Wongwises, S. Enhancement of heat transfer using nanofluids—An overview. Renew. Sustain. Energy Rev. 2010, 14, 629–641. [Google Scholar] [CrossRef]
- Buongiorno, J.; Venerus, D.C.; Prabhat, N.; McKrell, T.; Townsend, J.; Christianson, R.; Tolmachev, Y.V.; Keblinski, P.; Hu, L.-W.; Alvarado, J.L.; et al. A benchmark study on the thermal conductivity of nanofluids. J. Appl. Phys. 2009, 106, 094312. [Google Scholar] [CrossRef]
- Masuda, N.H.H.; Ebata, A.; Teramae, K. Alteration of thermal conductivity and viscosity of Liquid by dispersing ultra fine particles. Netsu Bussei 1993, 7, 227–233. [Google Scholar] [CrossRef]
- Xuan, Y.; Li, Q. Investigation on Convective Heat Transfer and Flow Features of Nanofluids. J. Heat Transf. 2003, 125, 151–155. [Google Scholar] [CrossRef]
- Le Ba, T.; Mahian, O.; Wongwises, S.; Szilágyi, I.M. Review on the recent progress in the preparation and stability of graphene-based nanofluids. J. Therm. Anal. Calorim. 2020, 1–28. [Google Scholar] [CrossRef]
- Jang, S.P.; Choi, S.U.S. Role of Brownian motion in the enhanced thermal conductivity of nanofluids. Appl. Phys. Lett. 2004, 84, 4316. [Google Scholar] [CrossRef]
- Shukla, R.K.; Dhir, V.K. Effect of Brownian Motion on Thermal Conductivity of Nanofluids. J. Heat Transf. 2008, 130, 042406. [Google Scholar] [CrossRef]
- Lin, Y.; Jiang, Y. Effects of Brownian motion and thermophoresis on nanofluids in a rotating circular groove: A numerical simulation. Int. J. Heat Mass Transf. 2018, 123, 569–582. [Google Scholar] [CrossRef]
- Koo, J.; Kleinstreuer, C. Impact analysis of nanoparticle motion mechanisms on the thermal conductivity of nanofluids. Int. Commun. Heat Mass Transf. 2005, 32, 1111–1118. [Google Scholar] [CrossRef]
- Lee, S.; Choi, S.U.-S.; Li, S.; Eastman, J.A. Measuring Thermal Conductivity of Fluids Containing Oxide Nanoparticles. J. Heat Transf. 1999, 121, 280–289. [Google Scholar] [CrossRef]
- Eastman, J.A.; Choi, S.; Li, S.; Yu, W.; Thompson, L.J. Anomalously increased effective thermal conductivities of ethylene glycol-based nanofluids containing copper nanoparticles. Appl. Phys. Lett. 2001, 78, 718–720. [Google Scholar] [CrossRef]
- Murshed, S.; Leong, K.; Yang, C. Investigations of thermal conductivity and viscosity of nanofluids. Int. J. Therm. Sci. 2008, 47, 560–568. [Google Scholar] [CrossRef]
- Sekhar, Y.R.; Sharma, K.; Karupparaj, R.T.; Chiranjeevi, C. Heat Transfer Enhancement with Al2O3 Nanofluids and Twisted Tapes in a Pipe for Solar Thermal Applications. Procedia Eng. 2013, 64, 1474–1484. [Google Scholar] [CrossRef]
- Suresh, S.; Venkitaraj, K.; Selvakumar, P.; Chandrasekar, M. Effect of Al2O3–Cu/water hybrid nanofluid in heat transfer. Exp. Therm. Fluid Sci. 2012, 38, 54–60. [Google Scholar] [CrossRef]
- Putnam, S.A.; Cahill, D.G.; Braun, P.V.; Ge, Z.; Shimmin, R.G. Thermal conductivity of nanoparticle suspensions. J. Appl. Phys. 2006, 99, 084308. [Google Scholar] [CrossRef]
- Farbod, M.; Asl, R.K.; Abadi, A.R.N. Morphology dependence of thermal and rheological properties of oil-based nanofluids of CuO nanostructures. Colloids Surf. A Physicochem. Eng. Asp. 2015, 474, 71–75. [Google Scholar] [CrossRef]
- Biercuk, M.J.; Llaguno, M.C.; Radosavljevic, M.; Hyun, J.K.; Johnson, A.T.; Fischer, J.E. Carbon nanotube composites for thermal management. Appl. Phys. Lett. 2002, 80, 2767–2769. [Google Scholar] [CrossRef]
- Xie, H.; Lee, H.; Youn, W.; Choi, M. Nanofluids containing multiwalled carbon nanotubes and their enhanced thermal conductivities. J. Appl. Phys. 2003, 94, 4967. [Google Scholar] [CrossRef]
- Le Ba, T.; Várady, Z.I.; Lukács, I.E.; Molnár, J.; Balczár, I.A.; Wongwises, S.; Szilágyi, I.M. Experimental investigation of rheological properties and thermal conductivity of SiO2–P25 TiO2 hybrid nanofluids. J. Therm. Anal. Calorim. 2020, 1–15. [Google Scholar] [CrossRef]
- Shima, P.D.; Philip, J.; Raj, B. Influence of aggregation on thermal conductivity in stable and unstable nanofluids. Appl. Phys. Lett. 2010, 97, 153113. [Google Scholar] [CrossRef]
- Gao, J.; Zheng, R.T.; Ohtani, H.; Zhu, D.S.; Chen, G. Experimental Investigation of Heat Conduction Mechanisms in Nanofluids. Clue on Clustering. Nano Lett. 2009, 9, 4128–4132. [Google Scholar] [CrossRef] [PubMed]
- Wu, C.; Cho, T.-J.; Xu, J.; Lee, D.; Yang, B.; Zachariah, M.R. Effect of nanoparticle clustering on the effective thermal conductivity of concentrated silica colloids. Phys. Rev. E 2010, 81, 011406. [Google Scholar] [CrossRef] [PubMed]
- Askari, S.; Lotfi, R.; Seifkordi, A.; Rashidi, A.; Koolivand, H. A novel approach for energy and water conservation in wet cooling towers by using MWNTs and nanoporous graphene nanofluids. Energy Convers. Manag. 2016, 109, 10–18. [Google Scholar] [CrossRef]
- Verma, S.K.; Tiwari, A.K.; Chauhan, D.S. Experimental evaluation of flat plate solar collector using nanofluids. Energy Convers. Manag. 2017, 134, 103–115. [Google Scholar] [CrossRef]
- Haque, A.K.M.M.; Kim, T.; Oh, G.S.; Kim, J.; Noh, J.; Choi, B.; Chung, H.; Jeong, H.; Huh, S.; Mahmudul, H.A.K.M.; et al. Synthesis of Graphene/Multi-Walled Carbon Nanotube Composite and Its Nanofluid Preparation. Nanosci. Nanotechnol. Lett. 2016, 8, 316–323. [Google Scholar] [CrossRef]
- Ding, Y.; Alias, H.; Wen, D.; Williams, R. Heat transfer of aqueous suspensions of carbon nanotubes (CNT nanofluids). Int. J. Heat Mass Transf. 2006, 49, 240–250. [Google Scholar] [CrossRef]
- Shao, X.; Chen, Y.; Mo, S.; Cheng, Z.; Yin, T. Dispersion Stability of TiO2-H2O Nanofluids Containing Mixed Nanotubes and Nanosheets. Energy Procedia 2015, 75, 2049–2054. [Google Scholar] [CrossRef][Green Version]
- Chen, H.; Ding, Y.; Lapkin, A.; Fan, X. Rheological behaviour of ethylene glycol-titanate nanotube nanofluids. J. Nanopart. Res. 2009, 11, 1513–1520. [Google Scholar] [CrossRef]
- Alberola-Borràs, J.-A.; Mondragon, R.; Julia, J.E.; Hernández, L.; Cabedo, L. Characterization of halloysite-water nanofluid for heat transfer applications. Appl. Clay Sci. 2014, 99, 54–61. [Google Scholar] [CrossRef]
- Venerus, D.C.; Buongiorno, J.; Christianson, R.; Townsend, J.; Bang, I.C.; Chen, G.; Chung, S.J.; Chyu, M.; Chen, H.; Ding, Y.; et al. Viscosity measurements on colloidal dispersions (nanofluids) for heat transfer applications. Appl. Rheol. 2010, 20, 2. [Google Scholar] [CrossRef]
- Hamid, K.A.; Azmi, W.; Nabil, M.; Mamat, R.; Sharma, K. Experimental investigation of thermal conductivity and dynamic viscosity on nanoparticle mixture ratios of TiO2-SiO2 nanofluids. Int. J. Heat Mass Transf. 2018, 116, 1143–1152. [Google Scholar] [CrossRef]
- Turgut, A.; Tavman, I.; Chirtoc, M.; Karbstein, H.; Sauter, C.; Tavman, S. Thermal Conductivity and Viscosity Measurements of Water-Based TiO2 Nanofluids. Int. J. Thermophys. 2009, 30, 1213–1226. [Google Scholar] [CrossRef]
- Nabil, M.; Azmi, W.; Hamid, K.A.; Mamat, R.; Hagos, F.Y.; Mohamad, M.N.F. An experimental study on the thermal conductivity and dynamic viscosity of TiO2 -SiO2 nanofluids in water: Ethylene glycol mixture. Int. Commun. Heat Mass Transf. 2017, 86, 181–189. [Google Scholar] [CrossRef]
- Kluger, M.O.; Moon, V.; Kreiter, S.; Lowe, D.J.; Churchman, G.; Hepp, D.A.; Seibel, D.; Jorat, M.E.; Mörz, T. A new attraction-detachment model for explaining flow sliding in clay-rich tephras. Geology 2016, 45, 131–134. [Google Scholar] [CrossRef]
- Yuan, P.; Tan, D.; Annabi-Bergaya, F. Properties and applications of halloysite nanotubes: Recent research advances and future prospects. Appl. Clay Sci. 2015, 112, 75–93. [Google Scholar] [CrossRef]
- Yuan, P.; Southon, P.D.; Liu, Z.; Green, M.E.R.; Hook, J.M.; Antill, S.J.; Kepert, C.J. Functionalization of Halloysite Clay Nanotubes by Grafting with γ-Aminopropyltriethoxysilane. J. Phys. Chem. C 2008, 112, 15742–15751. [Google Scholar] [CrossRef]
- Tan, D.; Yuan, P.; Liu, D.; Du, P. Surface Modifications of Halloysite; Elsevier: Amsterdam, The Netherlands, 2016. [Google Scholar]
- Yang, S.; Li, S.; Yin, X.; Wang, L.; Chen, D.; Zhou, Y.; Wang, H. Preparation and characterization of non-solvent halloysite nanotubes nanofluids. Appl. Clay Sci. 2016, 126, 215–222. [Google Scholar] [CrossRef]
- Du, P.; Liu, D.; Yuan, P.; Deng, L.; Wang, S.; Zhou, J.; Zhong, X. Controlling the macroscopic liquid-like behaviour of halloysite-based solvent-free nanofluids via a facile core pretreatment. Appl. Clay Sci. 2018, 156, 126–133. [Google Scholar] [CrossRef]
- Shchukin, D.; Sukhorukov, G.; Price, R.; Lvov, Y. Halloysite Nanotubes as Biomimetic Nanoreactors. Small 2005, 1, 510–513. [Google Scholar] [CrossRef]
- Li, X.; Yang, Q.; Ouyang, J.; Yang, H.; Chang, S. Chitosan modified halloysite nanotubes as emerging porous microspheres for drug carrier. Appl. Clay Sci. 2016, 126, 306–312. [Google Scholar] [CrossRef]
- Deen, I.; Pang, X.; Zhitomirsky, I. Electrophoretic deposition of composite chitosan–halloysite nanotube–hydroxyapatite films. Colloids Surfaces A Physicochem. Eng. Asp. 2012, 410, 38–44. [Google Scholar] [CrossRef]
- Esfahani, M.R.; Stretz, H.A.; Wells, M.J. Abiotic reversible self-assembly of fulvic and humic acid aggregates in low electrolytic conductivity solutions by dynamic light scattering and zeta potential investigation. Sci. Total. Environ. 2015, 537, 81–92. [Google Scholar] [CrossRef] [PubMed]
- Yan, H.; Zhang, P.; Li, J.; Zhao, X.-L.; Zhang, K.; Zhang, B. PEDOT/PSS-Halloysite Nanotubes (HNTs) Hybrid Films: Insulating HNTs Enhance Conductivity of the PEDOT/PSS Films. Sci. Rep. 2015, 5, 18641. [Google Scholar] [CrossRef]
- Tazaki, K. Microbial formation of a halloysite-like mineral. Clays Clay Miner. 2005, 53, 224–233. [Google Scholar] [CrossRef]
- Abdullayev, E.; Joshi, A.; Wei, W.; Zhao, Y.; Lvov, Y.M. Enlargement of Halloysite Clay Nanotube Lumen by Selective Etching of Aluminum Oxide. ACS Nano 2012, 6, 7216–7226. [Google Scholar] [CrossRef]
- Nicolini, J.; Fukamachi, C.R.B.; Wypych, F.; Mangrich, A.S. Dehydrated halloysite intercalated mechanochemically with urea: Thermal behavior and structural aspects. J. Colloid Interface Sci. 2009, 338, 474–479. [Google Scholar] [CrossRef]
- Yuan, P.; Tan, D.; Aannabi-Bergaya, F.; Yan, W.; Fan, M.; Liu, D.; He, H. Changes in Structure, Morphology, Porosity, and Surface Activity of Mesoporous Halloysite Nanotubes Under Heating. Clays Clay Miner. 2012, 60, 561–573. [Google Scholar] [CrossRef]
- Cheng, H.; Frost, R.L.; Yang, J.; Liu, Q.; He, J. Infrared and infrared emission spectroscopic study of typical Chinese kaolinite and halloysite. Spectrochim. Acta Part Mol. Biomol. Spectrosc. 2010, 77, 1014–1020. [Google Scholar] [CrossRef]
- Kadi, S.; Lellou, S.; Khelifa, A.; Schott, J.; Gener-Batonneau, I.; Khelifa, A. Preparation, characterisation and application of thermally treated Algerian halloysite. Microporous Mesoporous Mater. 2012, 158, 47–54. [Google Scholar] [CrossRef]
- Tarì, G.; Bobos, I.; Gomes, C.; Ferreira, J.M.F. Modification of Surface Charge Properties during Kaolinite to Halloysite-7Å Transformation. J. Colloid Interface Sci. 1999, 210, 360–366. [Google Scholar] [CrossRef]
- Gaaz, T.S.; Sulong, A.; Kadhum, A.A.H.; Nassir, M.H.; Al-Amiery, A.A. Impact of Sulfuric Acid Treatment of Halloysite on Physico-Chemic Property Modification. Materials 2016, 9, 620. [Google Scholar] [CrossRef] [PubMed]
- Frost, R.L. Intercalation of Halloysite: A Raman Spectroscopic Study. Clays Clay Miner. 1997, 45, 551–563. [Google Scholar] [CrossRef]
- Frost, R.L.; Fredericks, P.M.; Bartlett, J.R. Fourier transform Raman spectroscopy of kandite clays. Spectrochim. Acta Part A Mol. Spectrosc. 1993, 49, 667–674. [Google Scholar] [CrossRef]
- Costanzo, P.M. Ordered Halloysite: Dimethylsulfoxide Intercalate. Clays Clay Miner. 1986, 34, 105–107. [Google Scholar] [CrossRef]
- Zhang, A.; Zhang, Y.; Zhu, Z. Thermal properties of Halloysite nanotubes (HNTs) intercalation complexes-A review. E3S Web Conf. 2019, 131, 01055. [Google Scholar] [CrossRef]
- Yu, W.; Xie, H. A Review on Nanofluids: Preparation, Stability Mechanisms, and Applications. J. Nanomater. 2012, 2012, 1–17. [Google Scholar] [CrossRef]
- Afrand, M.; Toghraie, D.; Ruhani, B. Effects of temperature and nanoparticles concentration on rheological behavior of Fe3O4–Ag/EG hybrid nanofluid: An experimental study. Exp. Therm. Fluid Sci. 2016, 77, 38–44. [Google Scholar] [CrossRef]
- Nguyen, C.; Desgranges, F.; Roy, G.; Galanis, N.; Mare, T.; Boucher, S.; Mintsa, H.A. Temperature and particle-size dependent viscosity data for water-based nanofluids–Hysteresis phenomenon. Int. J. Heat Fluid Flow. 2007, 28, 1492–1506. [Google Scholar] [CrossRef]
- Azmi, W.; Sharma, K.; Mamat, R.; Alias, A.B.S.; Misnon, I.I. Correlations for thermal conductivity and viscosity of water based nanofluids. IOP Conf. Series Mater. Sci. Eng. 2012, 36, 012029. [Google Scholar] [CrossRef]

| Sample Names | Halloysite (vol%) | DI (vol%) | 1M NaOH Solution (vol%) |
|---|---|---|---|
| HNT-0.5 | 0.50 | 99.50 | 0.00 |
| HNT-1.0 | 1.00 | 99.00 | 0.00 |
| HNT-1.5 | 1.50 | 98.50 | 0.00 |
| HNT-pH-0.5 | 0.50 | 98.50 | 1.00 |
| HNT-pH-1.0 | 1.00 | 98.00 | 1.00 |
| HNT-pH-1.5 | 1.50 | 97.50 | 1.00 |
| Element | Atomic% | ||
|---|---|---|---|
| Al | Si | O | |
| Present work | 15.59 | 16.13 | 68.28 |
| Tayser et al. [53] | 13.24 | 15.00 | 71.76 |
| Surfactant | Zeta Potential of 0.5% HNT Nanofluid (mV) |
|---|---|
| N/A | −11.83 |
| Tween | 7.91 |
| Oleylamine | 24.24 |
| CTAB | 20.42 |
| SDBS | −26.76 |
| GA | −16.99 |
| SCMC | −30.54 |
| Nanofluids | Zeta Potential (mV) |
|---|---|
| SCMC-0.5 | −30.54 |
| SCMC-1.0 | −32.18 |
| SCMC-1.5 | −31.22 |
| pH12-0.5 | −33.40 |
| pH12-1.0 | −39.72 |
| pH12-1.5 | −32.39 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Le Ba, T.; Alkurdi, A.Q.; Lukács, I.E.; Molnár, J.; Wongwises, S.; Gróf, G.; Szilágyi, I.M. A Novel Experimental Study on the Rheological Properties and Thermal Conductivity of Halloysite Nanofluids. Nanomaterials 2020, 10, 1834. https://doi.org/10.3390/nano10091834
Le Ba T, Alkurdi AQ, Lukács IE, Molnár J, Wongwises S, Gróf G, Szilágyi IM. A Novel Experimental Study on the Rheological Properties and Thermal Conductivity of Halloysite Nanofluids. Nanomaterials. 2020; 10(9):1834. https://doi.org/10.3390/nano10091834
Chicago/Turabian StyleLe Ba, Thong, Ahmed Qani Alkurdi, István Endre Lukács, János Molnár, Somchai Wongwises, Gyula Gróf, and Imre Miklós Szilágyi. 2020. "A Novel Experimental Study on the Rheological Properties and Thermal Conductivity of Halloysite Nanofluids" Nanomaterials 10, no. 9: 1834. https://doi.org/10.3390/nano10091834
APA StyleLe Ba, T., Alkurdi, A. Q., Lukács, I. E., Molnár, J., Wongwises, S., Gróf, G., & Szilágyi, I. M. (2020). A Novel Experimental Study on the Rheological Properties and Thermal Conductivity of Halloysite Nanofluids. Nanomaterials, 10(9), 1834. https://doi.org/10.3390/nano10091834








