Multi-Channel Exploration of O Adatom on TiO2(110) Surface by Scanning Probe Microscopy
Abstract
1. Introduction
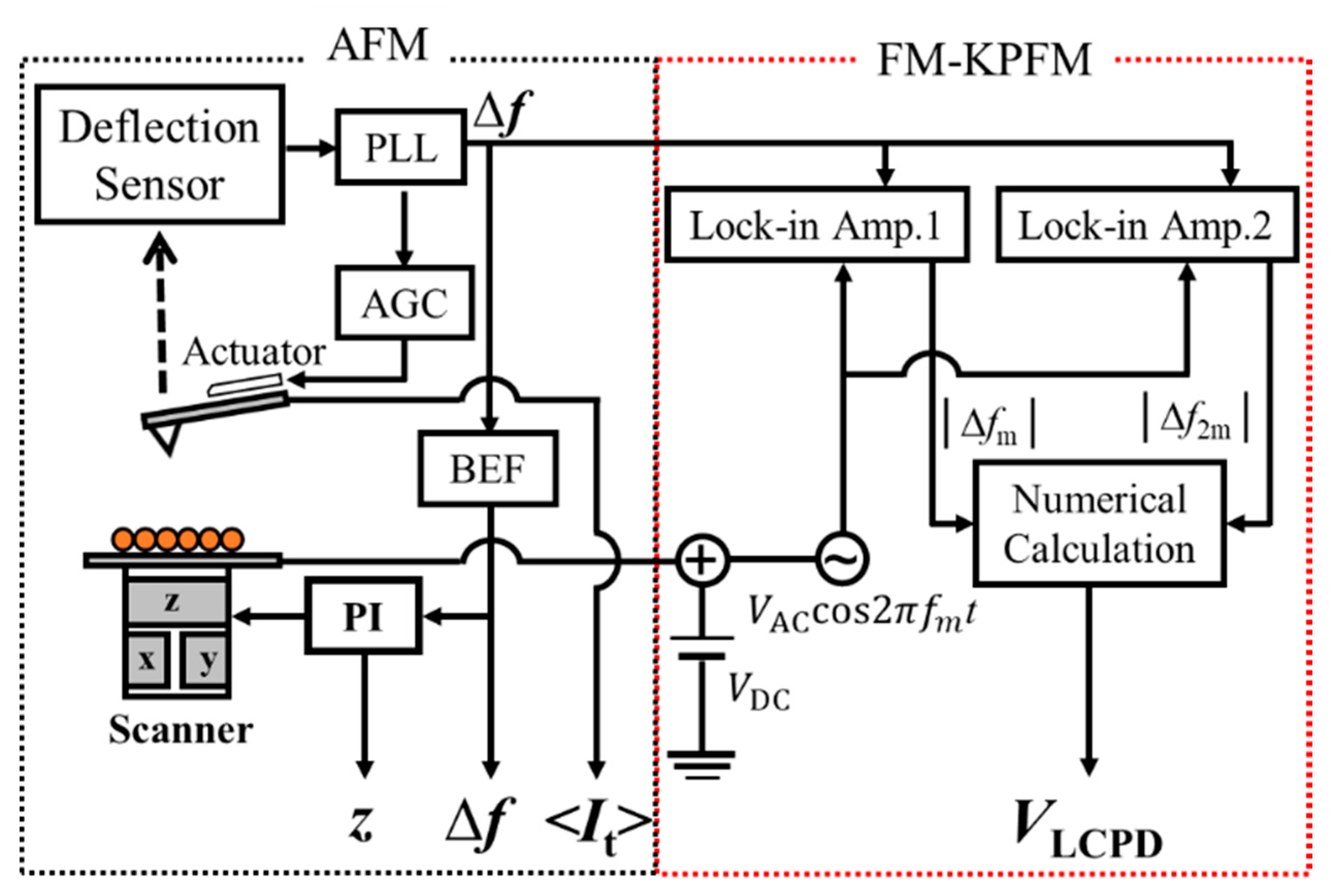
2. Experimental Details
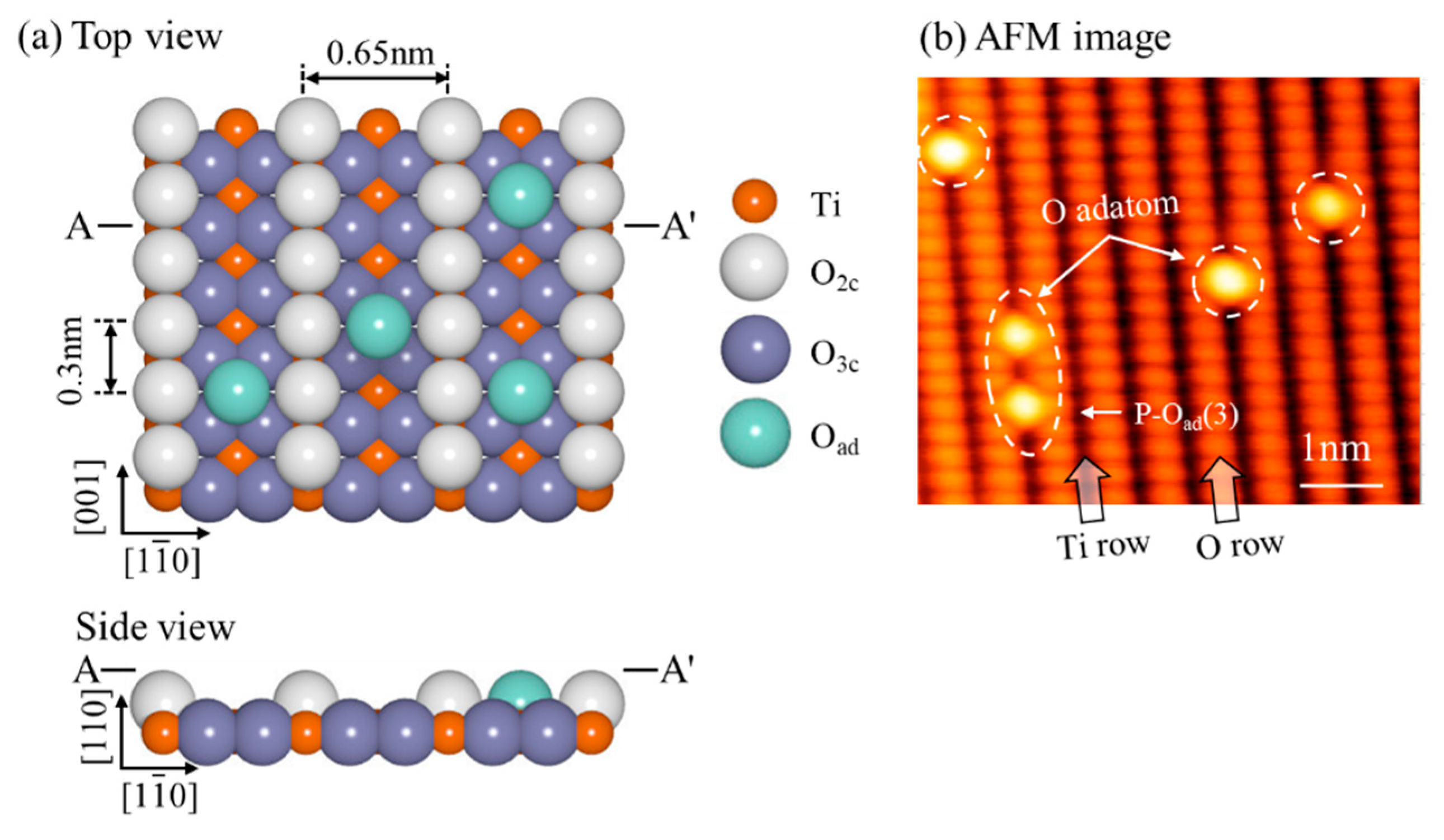
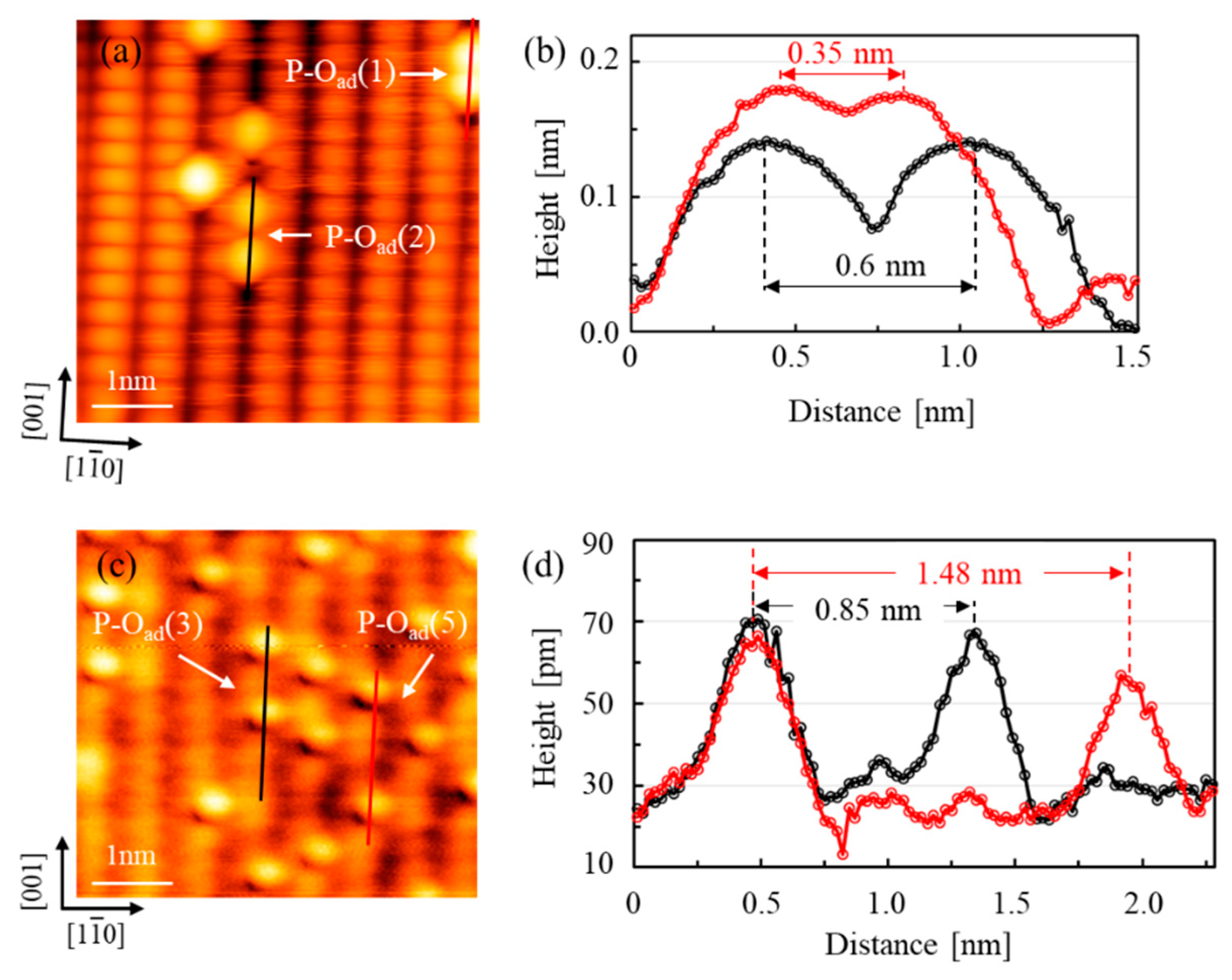
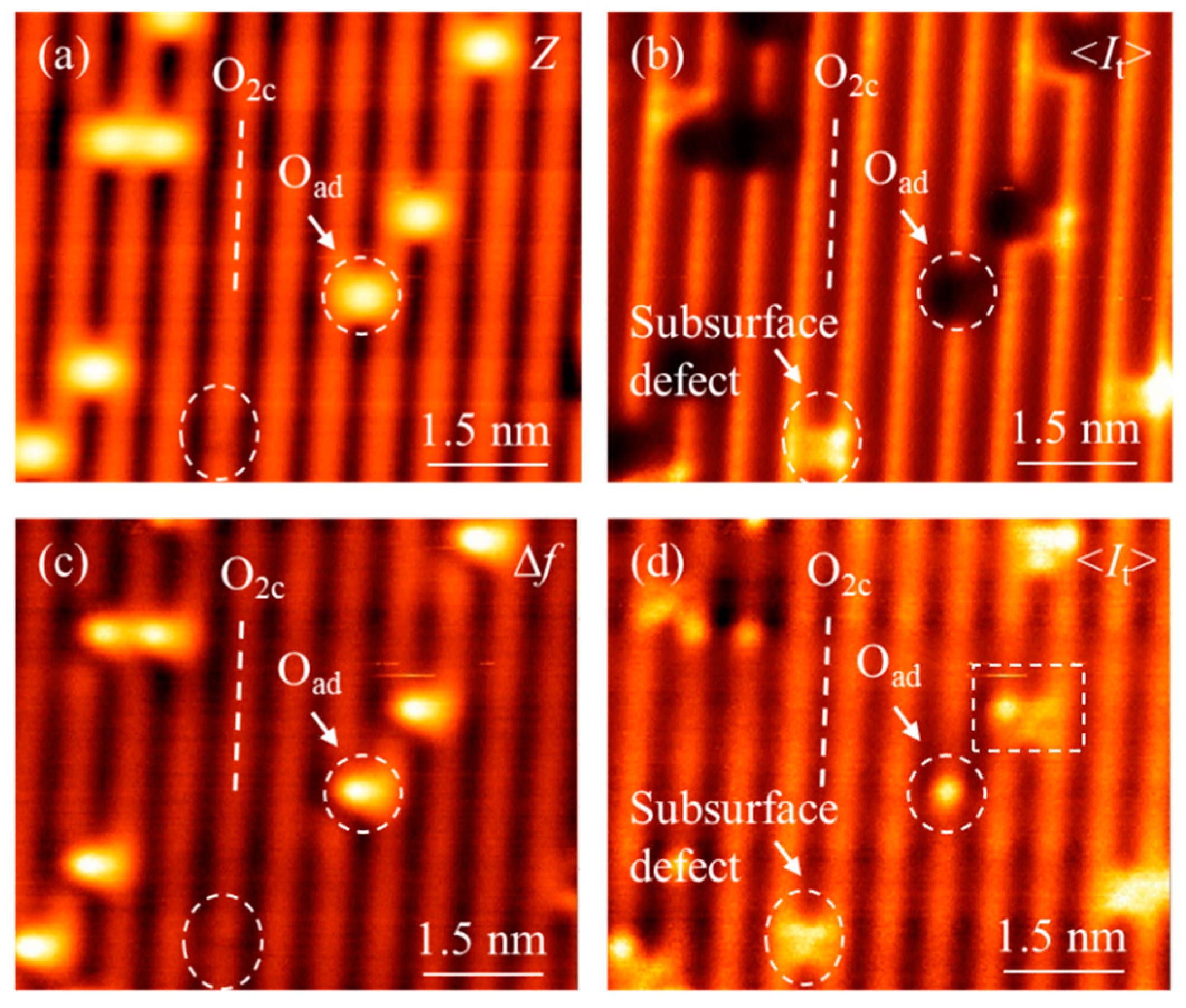
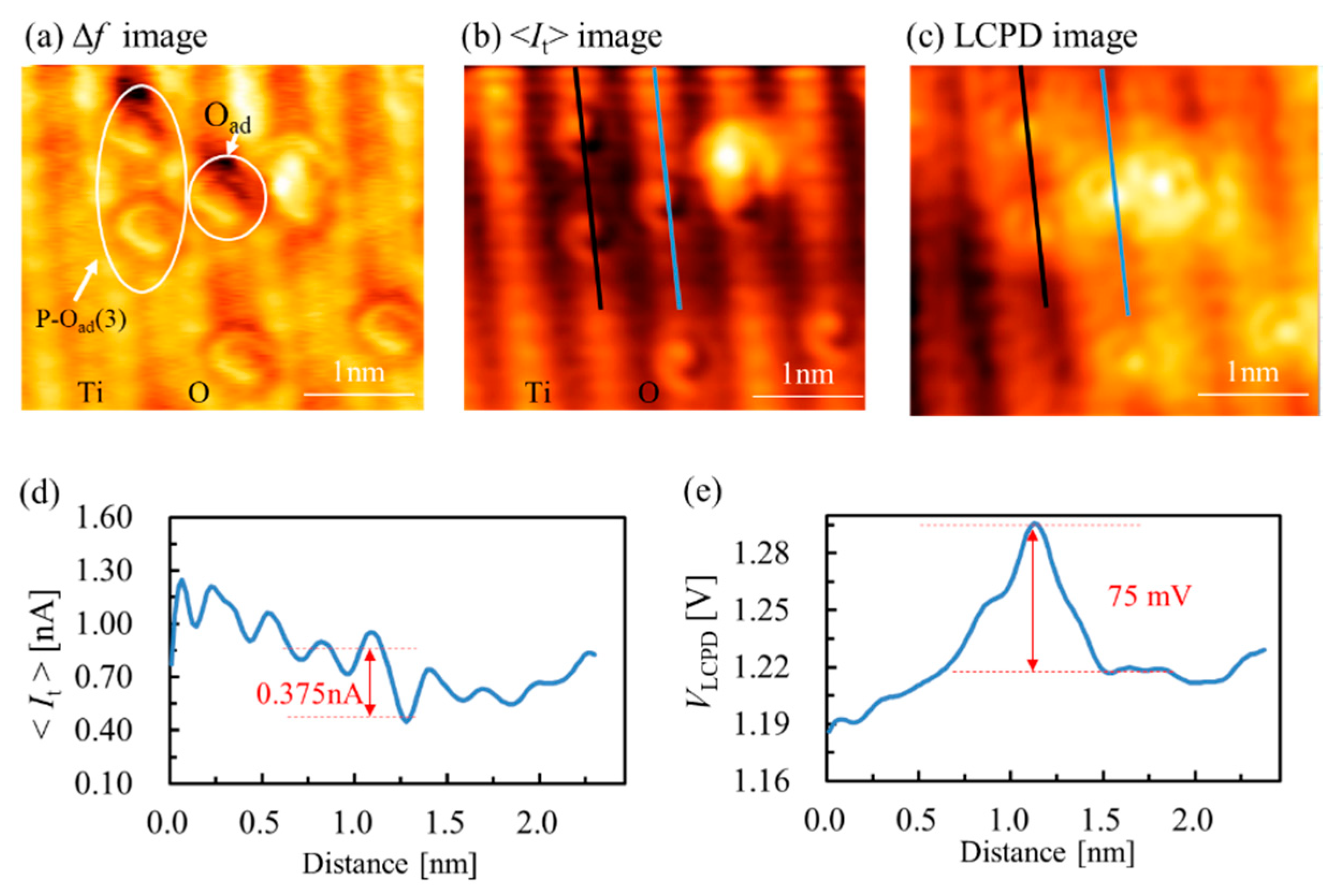
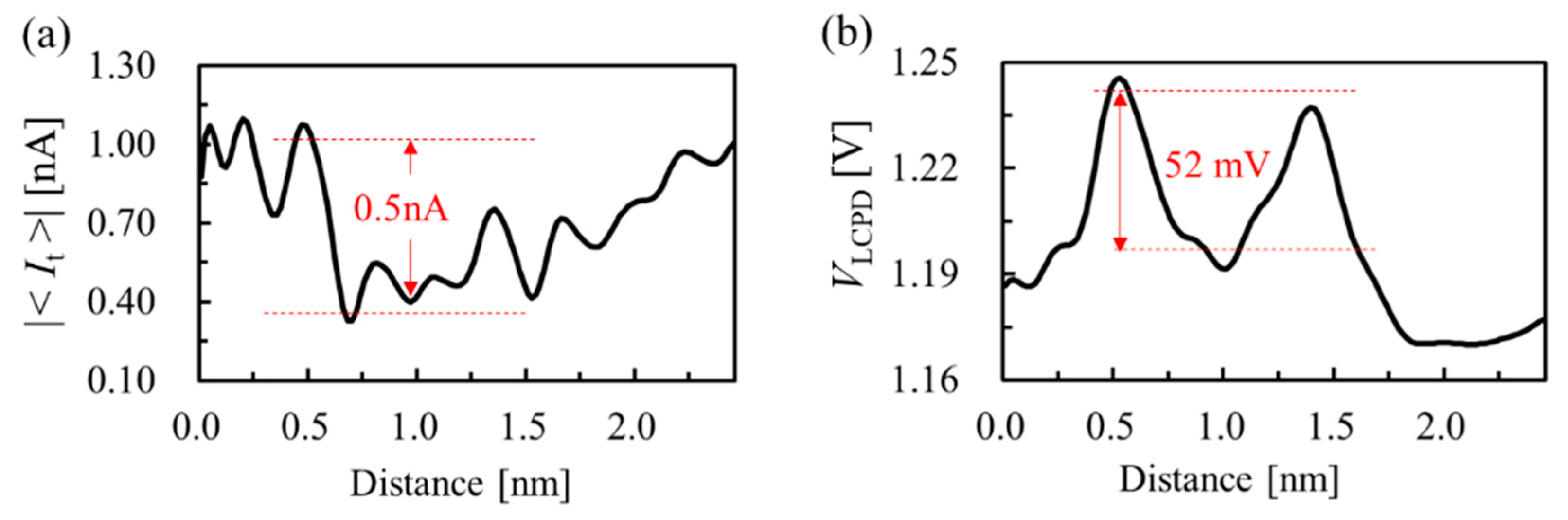
3. Results and Discussion
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Sugimoto, Y.; Pou, P.; Custance, O.; Jelinek, P.; Abe, M.; Perez, R.; Morita, S. Complex patterning by vertical interchange atom manipulation using atomic force microscopy. Science 2008, 322, 413–417. [Google Scholar] [CrossRef] [PubMed]
- Gross, L.; Mohn, F.; Moll, N.; Liljeroth, P.; Meyer, G. The chemical structure of a molecule resolved by atomic force microscopy. Science 2009, 325, 1110–1114. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Chen, P.C.; Yuan, B.K.; Ji, W.; Cheng, Z.; Qiu, X.H. Real-space identification of intermolecular bonding with atomic force microscopy. Science 2013, 342, 611–614. [Google Scholar] [CrossRef] [PubMed]
- Guo, J.; Meng, X.Z.; Chen, J.; Peng, J.B.; Sheng, J.M.; Li, X.Z.; Xu, L.M.; Shi, J.R.; Wang, E.G.; Jiang, Y. Real-space imaging of interfacial water with submolecular resolution. Nat. Mater. 2014, 13, 184–189. [Google Scholar] [CrossRef] [PubMed]
- Bamidele, J.; Lee, S.H.; Kinoshita, Y.; Turansky, R.; Naitoh, Y.; Li, Y.J.; Sugawara, Y.; Stich, I.; Kantorovich, L. Vertical atomic manipulation with dynamic atomic-force microscopy without tip change via a multi-step mechanism. Nat. Commun. 2014, 5, 4476. [Google Scholar] [CrossRef] [PubMed]
- Jiang, P.; Bao, X.; Salmeron, M. Catalytic reaction processes revealed by scanning probe microscopy. Acc. Chem. Res. 2015, 48, 1524–1531. [Google Scholar] [CrossRef] [PubMed]
- Kim, H.; Kyhm, K.; Taylor, R.A.; Kim, J.S.; Song, J.D.; Park, S. Optical shaping of the polarization anisotropy in a laterally coupled quantum dot dimer. Light Sci. Appl. 2020, 9, 100. [Google Scholar] [CrossRef]
- Jiang, S.; Zhang, X.; Zhang, Y.; Hu, C.; Zhang, R.; Zhang, Y.; Liao, Y.; Smith, Z.J.; Dong, Z.; Hou, J.G. Subnanometer-resolved chemical imaging via multivariate analysis of tip-enhanced Raman maps. Light Sci. Appl. 2017, 9, e17098. [Google Scholar] [CrossRef]
- Wen, H.F.; Zhang, Q.; Yuuki, A.; Miyazaki, M.; Naitoh, Y.; Li, Y.J.; Sugawara, Y. Direct visualization of oxygen reaction with paired hydroxyl on TiO2110 surface at 78 K by atomic force microscopy. J. Phys. Chem. C 2018, 122, 17395–17399. [Google Scholar] [CrossRef]
- Adachi, Y.; Wen, H.; Zhang, Q.; Miyazaki, M.; Sugawara, Y.; Sang, H.; Brndiar, J.; Kantorovich, L.; Štich, I.; Li, Y.J. Tip-induced control of charge and molecular bonding of oxygen atoms on the rutile TiO2110 surface with atomic force microscopy. ACS Nano 2019, 13, 6917–6924. [Google Scholar] [CrossRef]
- Melitz, W.; Shen, J.; Kummel, A.C.; Lee, S. Kelvin probe force microscopy and its application. Surf. Sci. Rep. 2011, 66, 1–27. [Google Scholar] [CrossRef]
- Wandelt, K. The local work function: Concept and implications. Appl. Surf. Sci. 1997, 111, 1–10. [Google Scholar] [CrossRef]
- Albrecht, T.R.; Grütter, P.; Horne, D.; Rugar, D. Frequency modulation detection using high-Q cantilevers for enhanced force microscope sensitivity. J. Appl. Phys. 1991, 69, 668–673. [Google Scholar] [CrossRef]
- Sugawara, Y.; Kou, L.; Ma, Z.; Kamijo, T.; Naitoh, Y.; Li, Y.J. High potential sensitivity in heterodyne amplitude-modulation Kelvin probe force microscopy. Appl. Phys. Lett. 2012, 100, 223104. [Google Scholar] [CrossRef]
- Ma, Z.M.; Kou, L.; Naitoh, Y.; Li, Y.J.; Sugawara, Y. The stray capacitance effect in Kelvin probe force microscopy using FM, AM and heterodyne. Nanotechnology 2013, 24, 225701. [Google Scholar] [CrossRef]
- Ziegler, D.; Stemmer, A. Force gradient sensitive detection in lift-mode Kelvin probe force microscopy. Nanotechnology 2011, 22, 075501. [Google Scholar] [CrossRef]
- Takeuchi, O.; Ohrai, Y.; Yoshida, S.; Shigekawa, H. Kelvin Probe Force Microscopy without Bias-Voltage Feedback. Jpn. J. Appl. Phys. 2007, 46, 5626–5630. [Google Scholar] [CrossRef]
- Kou, L.; Ma, Z.; Li, Y.J.; Naitoh, Y.; Komiyama, M.; Sugawara, Y. Surface potential imaging with atomic resolution by frequency-modulation Kelvin probe force microscopy without bias voltage feedback. Nanotechnology 2015, 26, 165701. [Google Scholar] [CrossRef]
- Wen, H.F.; Li, Y.J.; Arima, E.; Naitoh, Y.; Sugawara, Y.; Xu, R.; Cheng, Z.H. Investigation of tunneling current and local contact potential difference on the TiO2(110) surface by AFM/KPFM at 78 K. Nanotechnology 2017, 28, 105704. [Google Scholar] [CrossRef]
- Ashino, M.; Sugawara, Y.; Morita, S.; Ishikawa, M. Atomic resolution noncontact atomic force and scanning tunneling microscopy of TiO2(110)-(1 × 1) and (1 × 2): Simultaneous imaging of surface structures and electronic states. Phys. Rev. Lett. 2001, 86, 4334–4337. [Google Scholar] [CrossRef]
- Sugimoto, Y.; Nakajima, Y.; Sawada, D.; Morita, K.I.; Abe, M.; Morita, S. Simultaneous AFM and STM measurements on the Si(111)-(7 × 7) surface. Phys. Rev. B 2010, 81, 245322. [Google Scholar] [CrossRef]
- Baykara, M.Z.; Mönig, H.; Schwendemann, T.C.; Ünverdi, Ö.; Altman, E.I.; Schwarz, U.D. Three-dimensional interaction force and tunneling current spectroscopy of point defects on rutile TiO2(110). Appl. Phys. Lett. 2016, 108, 071601. [Google Scholar] [CrossRef]
- Wang, Z.T.; Deskins, N.A.; Lyubinetsky, I. Direct imaging of site-specific photocatalytical reactions of O2 on TiO2(110). J. Phys. Chem. Lett. 2012, 3, 102. [Google Scholar] [CrossRef]
- Du, Y.; Deskins, N.A.; Zhang, Z.; Dohnalek, Z.; Dupuis, M.; Lyubinetsky, I. Formation of O adatom pairs and charge transfer upon O2 dissociation on reduced TiO2(110). Phys. Chem. Chem. Phys. 2010, 12, 6337–6344. [Google Scholar] [CrossRef] [PubMed]
- Naitoh, Y.; Kinoshita, Y.; Li, Y.J.; Kageshima, M.; Sugawara, Y. The influence of a Si cantilever tip with/without tungsten coating on noncontact atomic force. Nanotechnology 2009, 20, 264011. [Google Scholar] [CrossRef]
- Li, Y.J.; Wen, H.F.; Naitoh, Y.; Sugawara, Y. Stable contrast mode on TiO2110 surface with metal-coated tip using AFM. Ultramicroscopy 2018, 191, 51–55. [Google Scholar] [CrossRef]
- Zhang, Q.; Li, Y.J.; Wen, H.F.; Adachi, Y.; Miyazaki, M.; Sugawara, Y.; Xu, R.; Cheng, Z.H.; Brndiar, J.; Kantorovich, L.; et al. Measurement and manipulation of the charge state of an adsorbed oxygen adatom on the rutile TiO2(110)-(1 × 1) surface by nc-AFM and KPFM. J. Am. Chem. Soc. 2018, 140, 15668–15674. [Google Scholar] [CrossRef]
- Wen, H.F.; Yuuki, A.; Zhang, Q.; Miyazaki, M.; Li, Y.J.; Sugawara, Y. Identification of atomic defects and adsorbate on rutile TiO2(110)-(1 × 1) surface by atomic force microscopy. J. Phys. Chem. C 2019, 123, 25756–25760. [Google Scholar] [CrossRef]
- Wen, H.F.; Zhang, Q.; Yuuki, A.; Miyazaki, M.; Sugawara, Y.; Li, Y.J. Contrast inversion of O adatom on rutile TiO2(110)-(1 × 1) surface by atomic force microscopy imaging. App. Surf. Sci. 2020, 505, 144623. [Google Scholar] [CrossRef]
- Lauritsen, J.V.; Foster, A.S.; Olesen, G.H.; Christensen, M.C.; Kühnle, A.; Helveg, S.; Rostrup-Niesen, J.R.; Clausen, B.S.; Reichling, M.; Besenbacher, F. Chemical identification of point defects and adsorbates on a metal oxide surface by atomic force microscopy. Nanotechnology 2006, 17, 3436. [Google Scholar] [CrossRef]
- Yurtsever, A.; Sugimoto, Y.; Abe, M.; Morita, S. NC-AFM imaging of the TiO2110-1×1 surface at low temperature. Nanotechnology 2010, 21, 165702. [Google Scholar] [CrossRef] [PubMed]
- Bechstein, R.; González, C.; Schütte, J.; Jelínek, P.; Pérez, R.; Kühnle, A. ‘All-inclusive’ imaging of the rutileTiO2(110) surface using NC-AFM. Nanotechnology 2009, 20, 505703. [Google Scholar] [CrossRef]
- Lira, E.; Hansen, J.Ø.; Huo, P.; Bechstein, R.; Galliker, P.; Lægsgaard, E.; Hammer, B.; Wendt, S.; Besenbacher, F. Dissociative and molecular oxygen chemisorption channels on reduced rutile TiO2(110): An STM and TPD study. Surf. Sci. 2010, 604, 1945–1960. [Google Scholar] [CrossRef]
- Wendt, S.; Sprunger, P.T.; Lira, E.; Madsen, G.K.; Li, Z.; Hansen, J.Ø.; Matthiesen, J.; Blekinge-Rasmussen, A.; Lægsgaard, E.; Hammer, B.; et al. The role of interstitial sites in the Ti3d defect state in the band gap of titania. Science 2008, 320, 1755. [Google Scholar] [CrossRef] [PubMed]
- Minato, T.; Sainoo, Y.; Kim, Y.; Kato, H.S.; Aika, K.I.; Kawai, M.; Zhao, J.; Petek, H.; Huang, T.; He, W.; et al. The electronic structure of oxygen atom vacancy and hydroxyl impurity defects on titanium dioxide 110 surface. J. Chem. Phys. 2009, 130, 124502. [Google Scholar] [CrossRef]
- Woolcot, T.; Teobaldi, G.; Pang, C.L.; Beglitis, N.S.; Fisher, A.J.; Hofer, W.A.; Thornton, G. Scanning tunneling microscopy contrast mechanisms for TiO2. Phys. Rev. Lett. 2012, 109, 156105. [Google Scholar] [CrossRef]
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wen, H.F.; Sugawara, Y.; Li, Y.J. Multi-Channel Exploration of O Adatom on TiO2(110) Surface by Scanning Probe Microscopy. Nanomaterials 2020, 10, 1506. https://doi.org/10.3390/nano10081506
Wen HF, Sugawara Y, Li YJ. Multi-Channel Exploration of O Adatom on TiO2(110) Surface by Scanning Probe Microscopy. Nanomaterials. 2020; 10(8):1506. https://doi.org/10.3390/nano10081506
Chicago/Turabian StyleWen, Huan Fei, Yasuhiro Sugawara, and Yan Jun Li. 2020. "Multi-Channel Exploration of O Adatom on TiO2(110) Surface by Scanning Probe Microscopy" Nanomaterials 10, no. 8: 1506. https://doi.org/10.3390/nano10081506
APA StyleWen, H. F., Sugawara, Y., & Li, Y. J. (2020). Multi-Channel Exploration of O Adatom on TiO2(110) Surface by Scanning Probe Microscopy. Nanomaterials, 10(8), 1506. https://doi.org/10.3390/nano10081506




