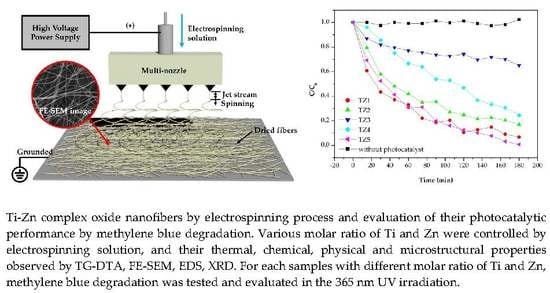
Photocatalytic Methylene Blue Degradation of Electrospun Ti–Zn Complex Oxide Nanofibers
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
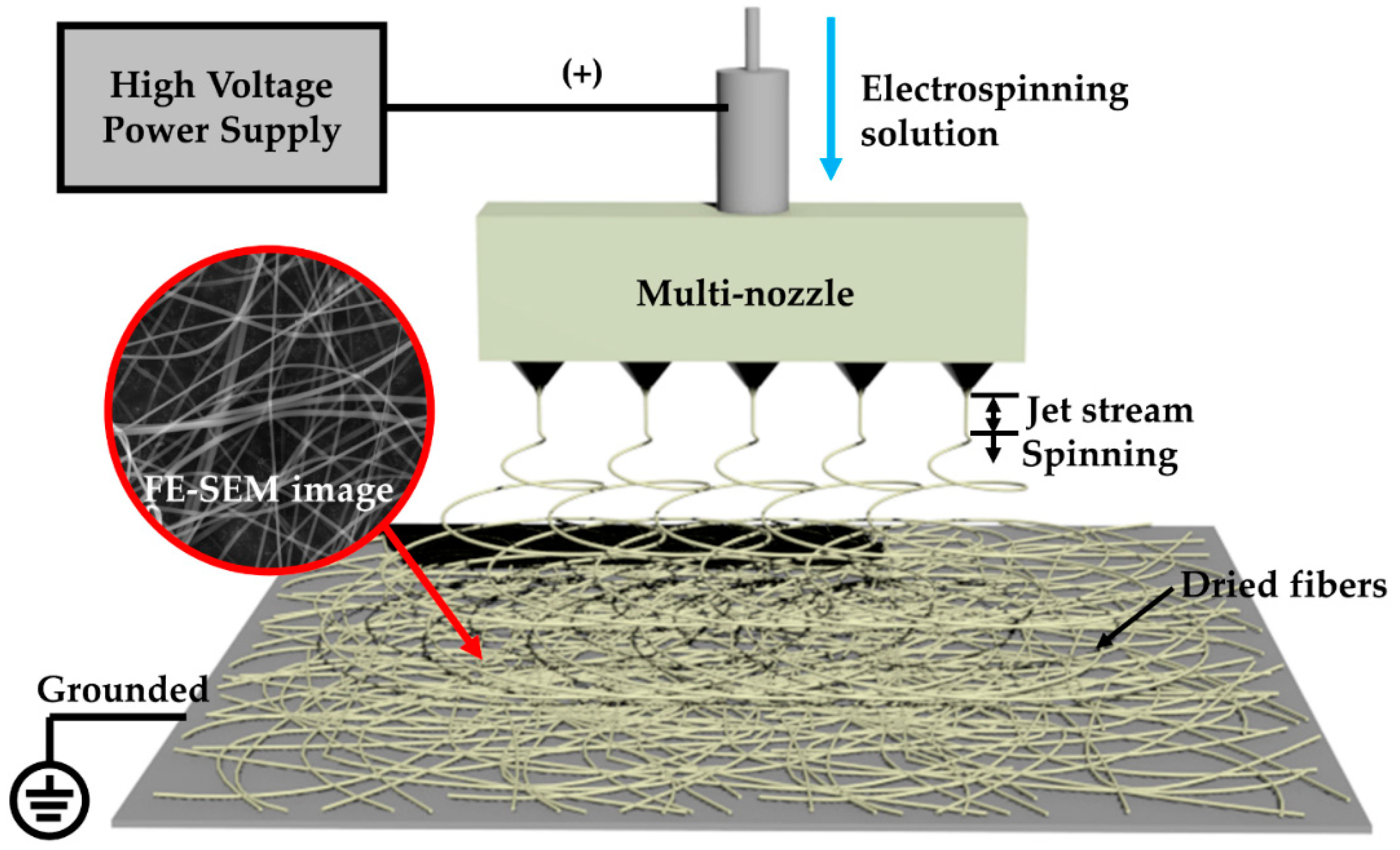
2.2. Fabrication of Ti–Zn Complex Oxide Nanofibers by an Electrospinning Process
2.3. Microstructural, Thermal and Crystalline Characterization
2.4. Photocatalytic Methylene Blue Degradation
3. Results and Discussion
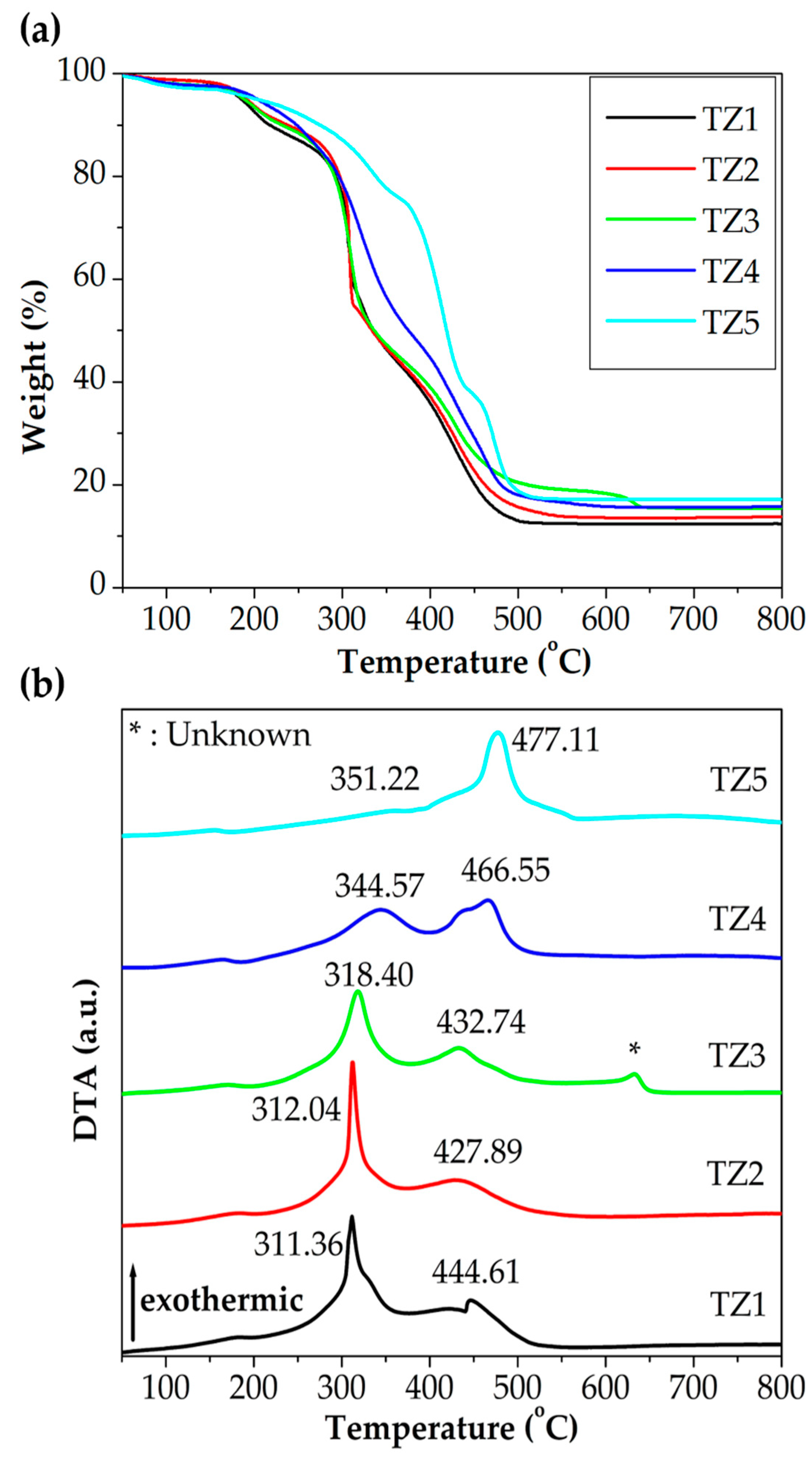
3.1. Thermal Properties of Ti–Zn Complex Oxide Nanofibers
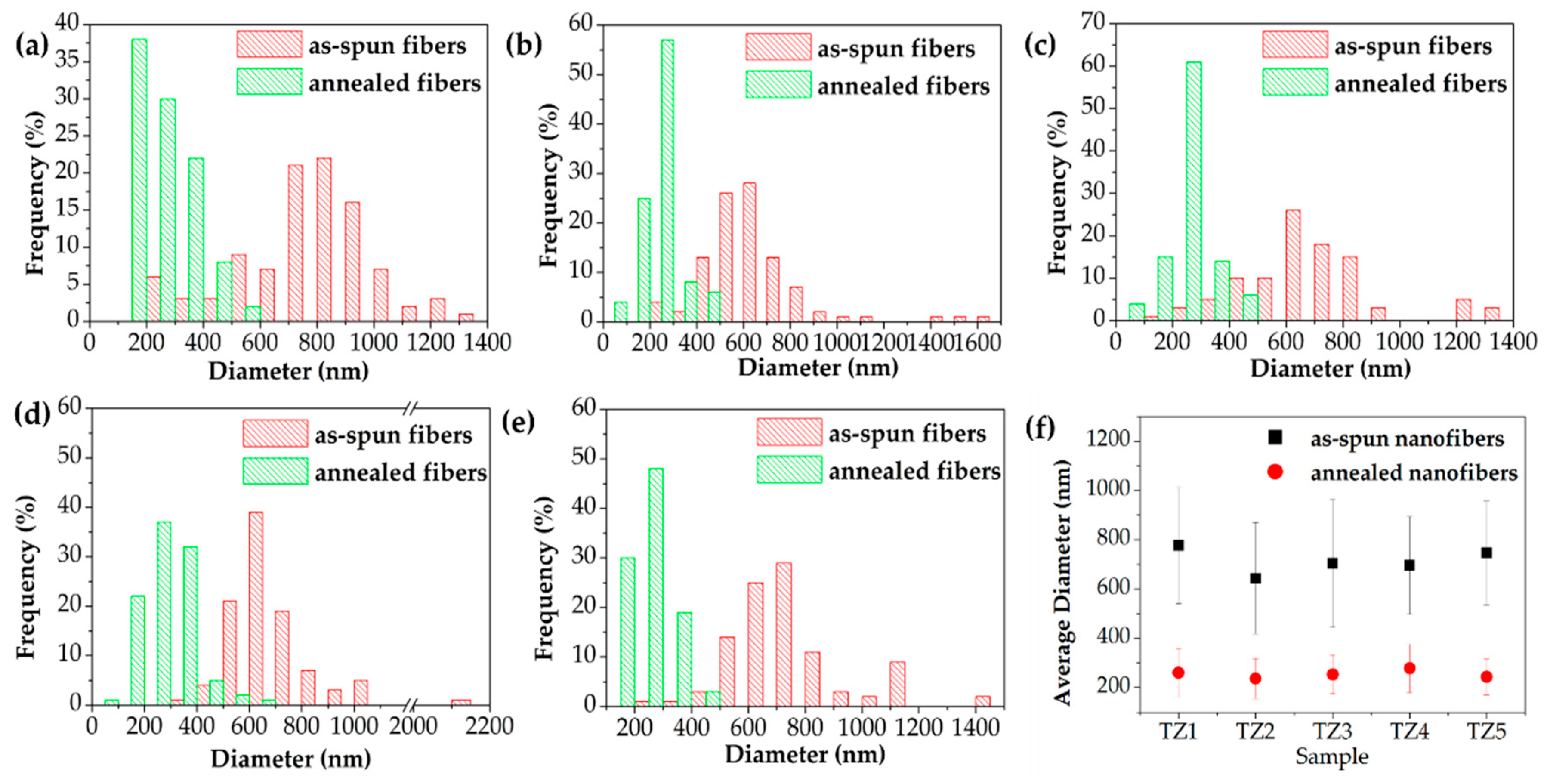
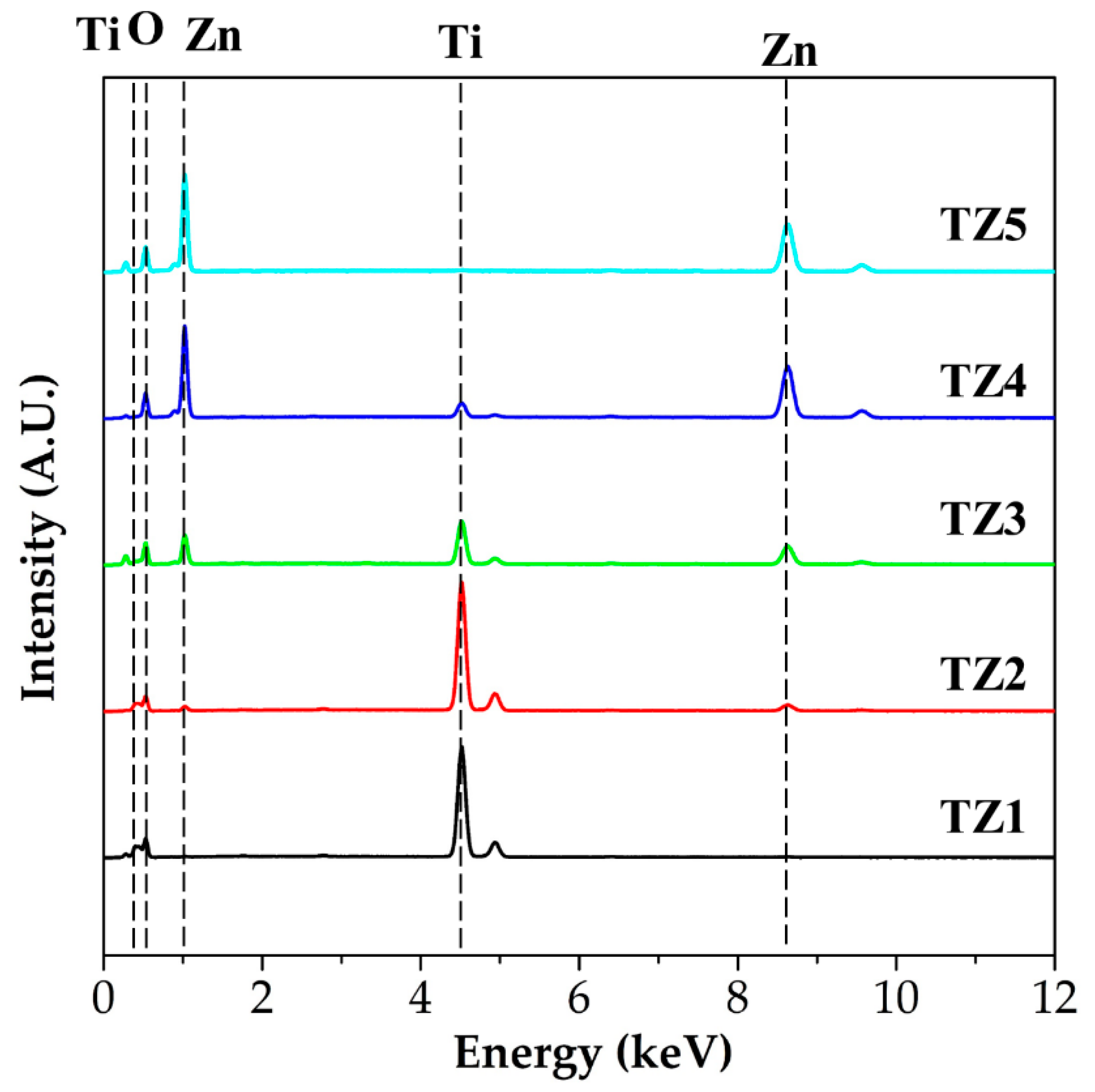
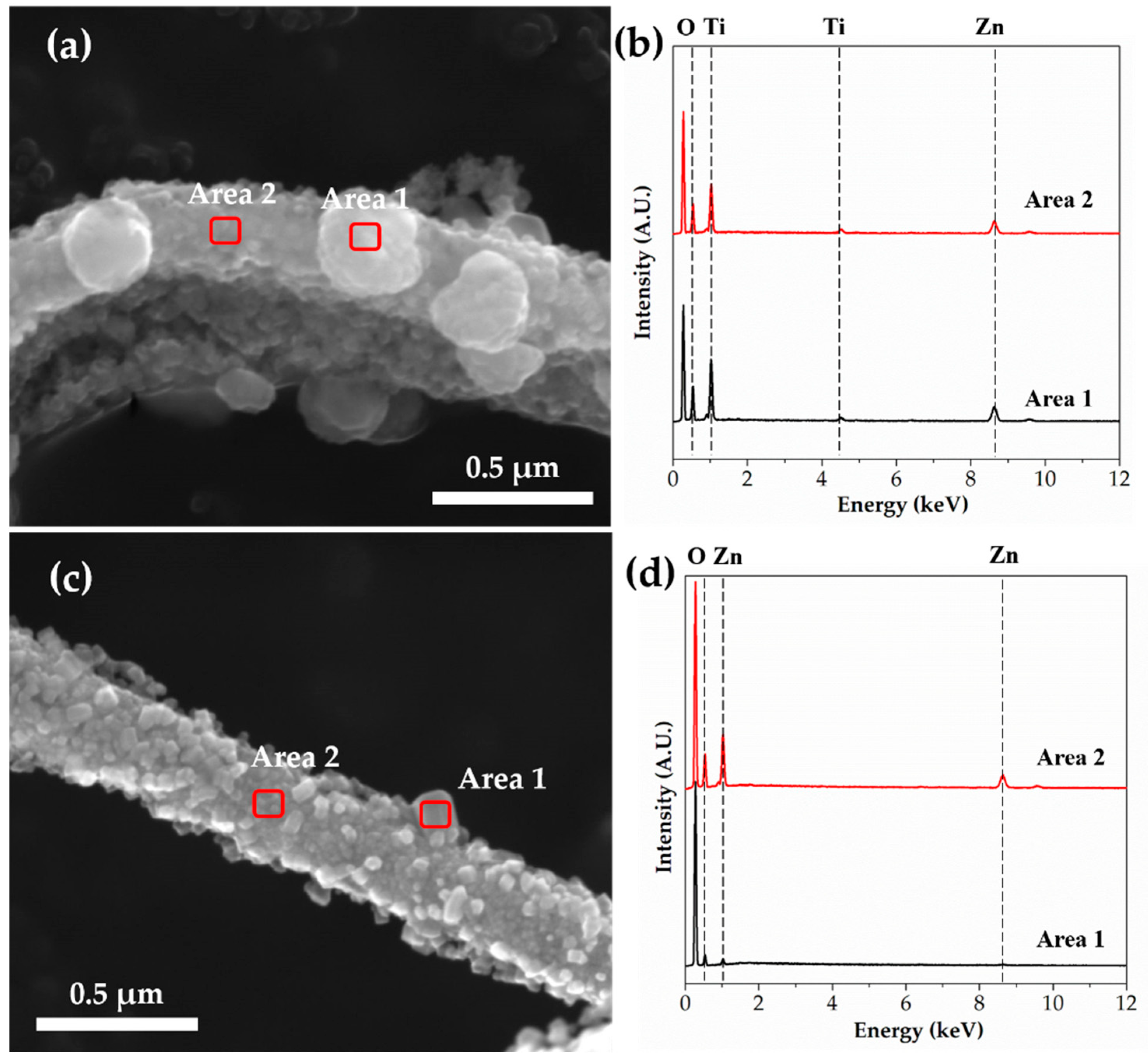
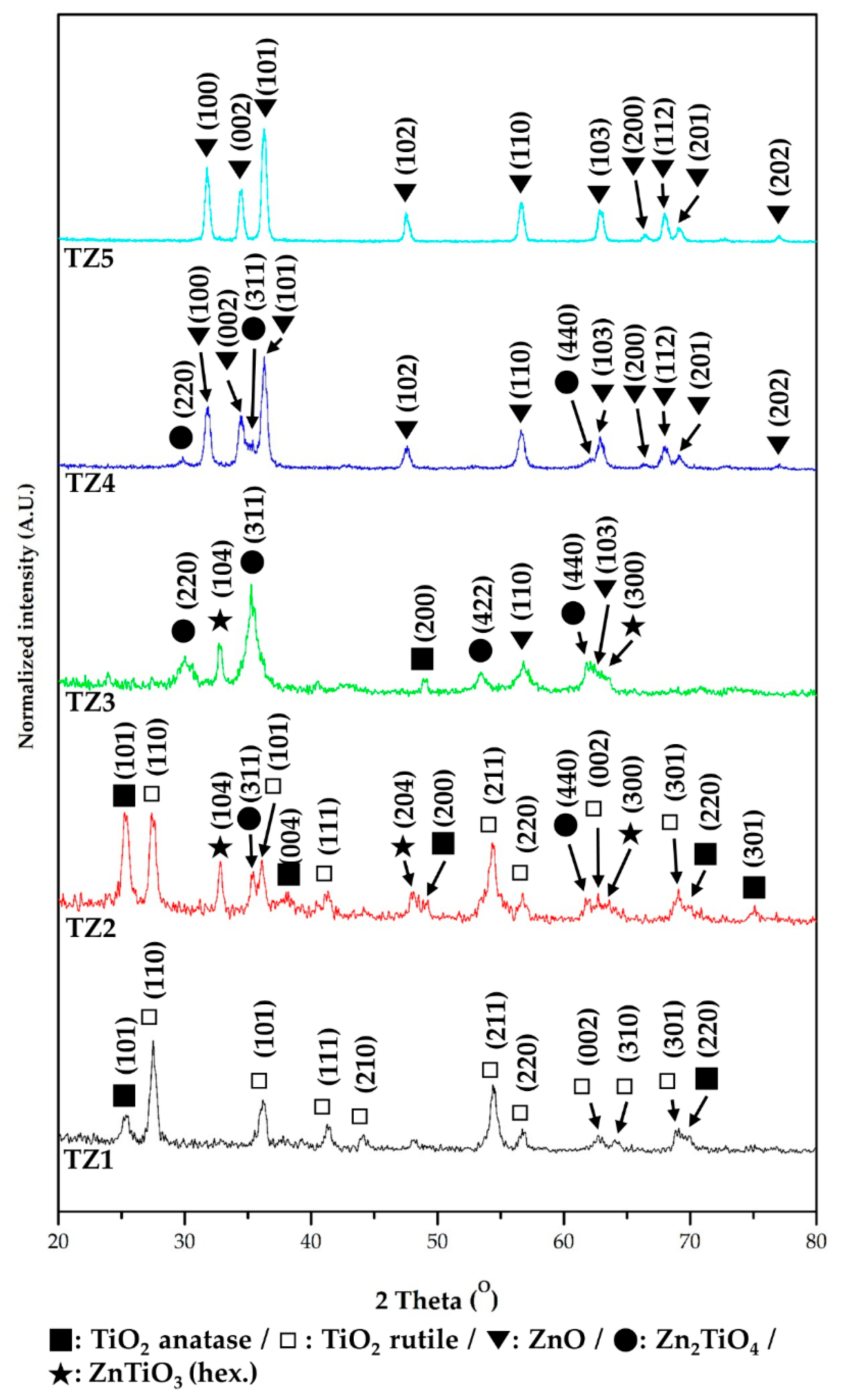
3.2. Microstructural, Chemical and Crystalline Properies of Ti–Zn Complex Oxide Nanofibers
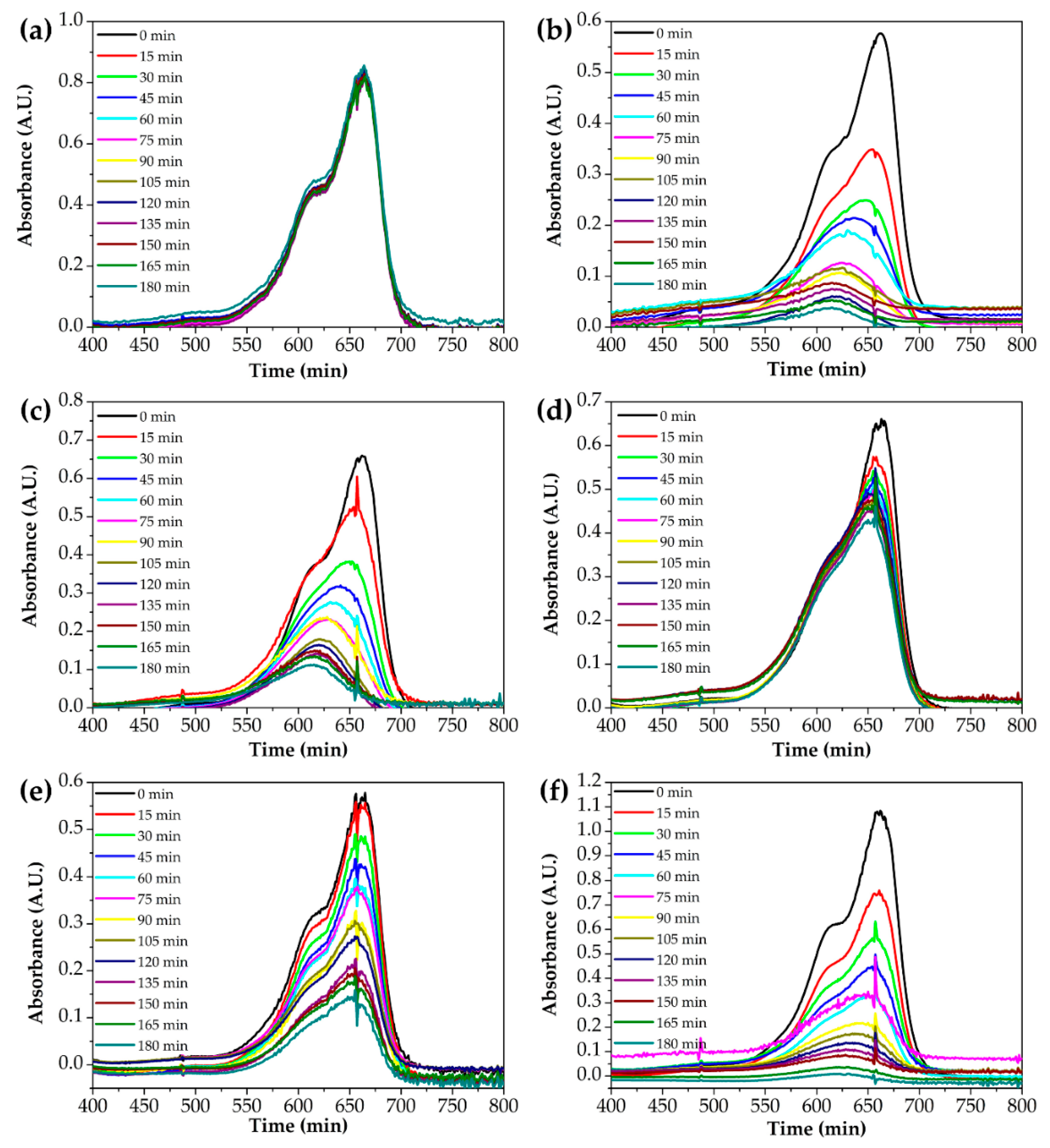
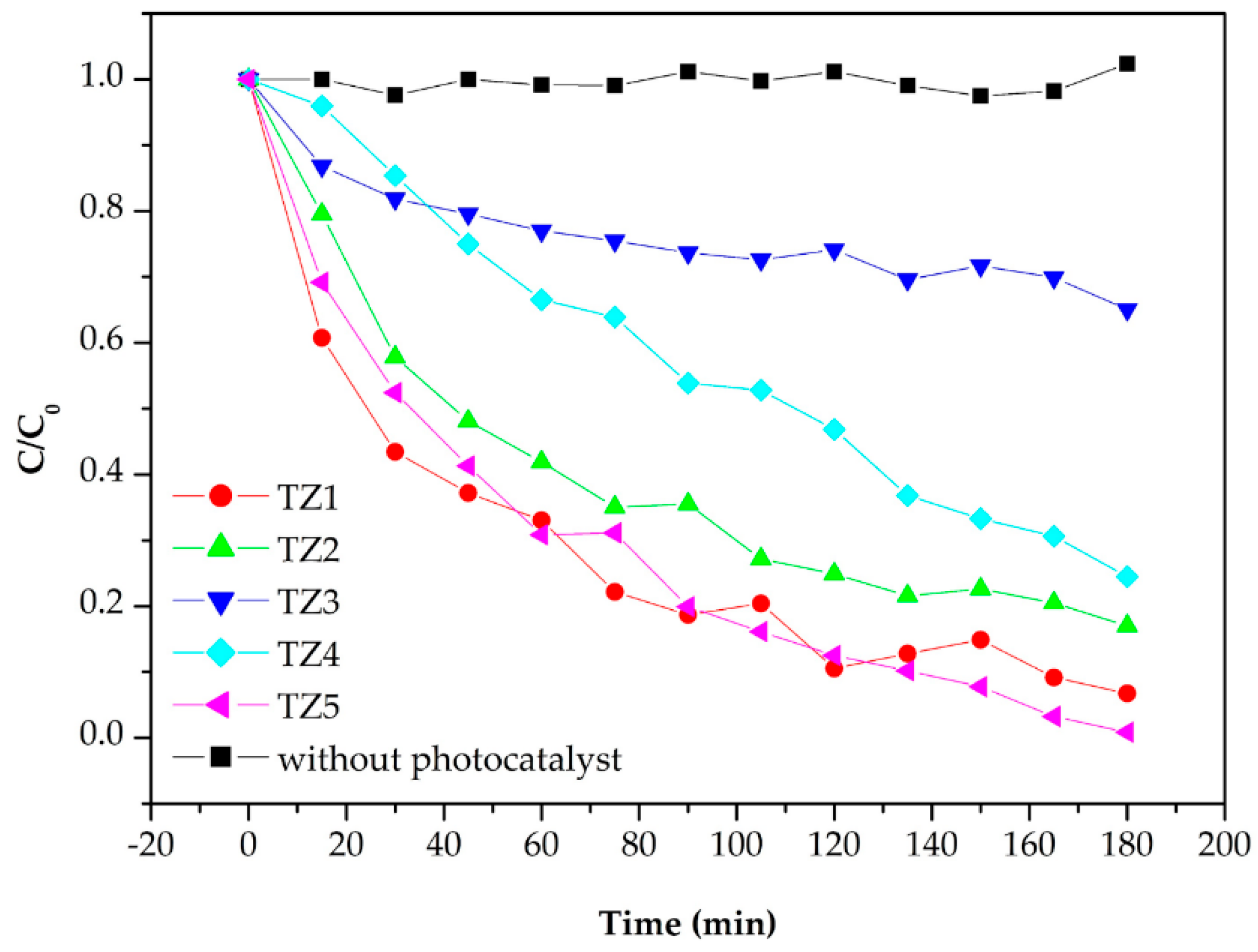
3.3. Photocatalytic Properties of Complex Oxide Nanofibers with Different Ti and Zn Ratios
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Ohno, T.; Sarukawa, K.; Tokieda, K.; Matsumura, M. Morphology of a TiO2 photocatalyst (Degussa, P-25) consisting of anatase and rutile crystalline phases. J. Catal. 2001, 203, 82–86. [Google Scholar] [CrossRef]
- Grätzel, M. Dye-sensitized solar cells. J. Photochem. Photobiol. C 2003, 4, 145–153. [Google Scholar] [CrossRef]
- Jeon, N.J.; Noh, J.H.; Yang, W.S.; Kim, Y.C.; Ryu, S.; Seo, J.; Seok, S.I. Compositional engineering of perovskite materials for high-performance solar cells. Nature 2015, 517, 476–480. [Google Scholar] [CrossRef] [PubMed]
- Mustafa, K.; Wroblewski, J.; Lopez, B.S.; Wennerberg, A.; Hultenby, K.; Arvidson, K. Determining optimal surface roughness of TiO2 blasted titanium implant material for attachment, proliferation and differentiation of cells derived from human mandibular alveolar bone. Clin. Oral Implant. Res. 2001, 12, 515–525. [Google Scholar] [CrossRef]
- Hwang, T.-H.; Kim, W.-T.; Choi, W.-Y. Mixed dimensionality with a TiO2 nanostructure and carbon nanotubes for the photoelectrode in dye-sensitized solar cells. J. Nanosci. Nanotechnol. 2017, 17, 4812–4816. [Google Scholar] [CrossRef]
- Shayegan, Z.; Lee, C.-S.; Haghighat, F. TiO2 photocatalyst for removal of volatile organic compounds in gas phase–a review. Chem. Eng. J. 2018, 334, 2408–2439. [Google Scholar] [CrossRef]
- Kim, W.-T.; Choi, W.-Y. Anodic Growth Behavior of TiO2 Nanotube Arrays with Process Parameter Control. J. Nanomater. 2019, 2019, 1–11. [Google Scholar] [CrossRef]
- Kim, W.-T.; Choi, W.-Y. Fabrication of TiO2 photonic crystal by anodic oxidation and their optical sensing properties. Sens. Actuators A Phys. 2017, 260, 178–184. [Google Scholar] [CrossRef]
- Kim, W.-T.; Hwang, T.-H.; Choi, W.-Y. Composite photoelectrode with TiO2 nanofibers and nanoparticles in dye-sensitized solar cells. Sci. Adv. Mater. 2018, 10, 210–214. [Google Scholar] [CrossRef]
- Kim, W.-T.; Kim, I.-H.; Choi, W.-Y. Fabrication of TiO2 nanotube arrays and their application to a gas sensor. J. Nanosci. Nanotechnol. 2015, 15, 8161–8165. [Google Scholar] [CrossRef]
- Kim, W.-T.; Na, K.-H.; Lee, J.-K.; Jang, I.; Choi, D.-S.; Choi, W.-Y. Porous TiO2 Nanotube Arrays for Drug Loading and Their Elution Sensing. J. Nanosci. Nanotechnol. 2019, 19, 1743–1748. [Google Scholar] [CrossRef] [PubMed]
- Ji, B.C.; Bae, S.S.; Rabbani, M.M.; Yeum, J.H. Photocatalytic Activity of Electrospun PAN/TiO2 Nanofibers in Dye Photodecomposition. Text. Color. Finish. 2013, 25, 94–101. [Google Scholar] [CrossRef]
- Bavykin, D.V.; Friedrich, J.M.; Walsh, F.C. Protonated titanates and TiO2 nanostructured materials: Synthesis, properties, and applications. Adv. Mater. 2006, 18, 2807–2824. [Google Scholar] [CrossRef]
- Tang, H.; Prasad, K.; Sanjines, R.; Schmid, P.; Levy, F. Electrical and optical properties of TiO2 anatase thin films. J. Appl. Phys. 1994, 75, 2042–2047. [Google Scholar] [CrossRef]
- Mor, G.K.; Varghese, O.K.; Paulose, M.; Shankar, K.; Grimes, C.A. A review on highly ordered, vertically oriented TiO2 nanotube arrays: Fabrication, material properties, and solar energy applications. Sol. Energy Mater. Sol. Cells 2006, 90, 2011–2075. [Google Scholar] [CrossRef]
- Choi, W.-Y.; Chung, J.; Cho, C.-H.; Kim, J.-O. Fabrication and photocatalytic activity of a novel nanostructured TiO2 metal membrane. Desalination 2011, 279, 359–366. [Google Scholar] [CrossRef]
- Lee, C.-G.; Kim, W.-T.; Na, K.-H.; Park, D.-C.; Yang, W.-H.; Choi, W.-Y. Control of Microstructure on TiO2 Nanofibers for Photocatalytic Application. J. Korean Inst. Electr. Electron. Mater. Eng. 2018, 31, 417–421. [Google Scholar]
- Lee, C.-G.; Na, K.-H.; Kim, W.-T.; Park, D.-C.; Yang, W.-H.; Choi, W.-Y. TiO2/ZnO Nanofibers Prepared by Electrospinning and Their Photocatalytic Degradation of Methylene Blue Compared with TiO2 Nanofibers. Appl. Sci. 2019, 9, 3404. [Google Scholar] [CrossRef]
- Wang, L.; Fu, X.; Han, Y.; Chang, E.; Wu, H.; Wang, H.; Li, K.; Qi, X. Preparation, characterization, and photocatalytic activity of TiO2/ZnO nanocomposites. J. Nanomater. 2013, 2013, 1–6. [Google Scholar] [CrossRef]
- Kim, W.-T.; Choi, W.-Y. Optical interference of TiO2 nanotube arrays for drug elution sensing. Sci. Adv. Mater. 2018, 10, 283–287. [Google Scholar] [CrossRef]
- Li, D.; Xia, Y. Fabrication of titania nanofibers by electrospinning. Nano Lett. 2003, 3, 555–560. [Google Scholar] [CrossRef]
- Antonelli, D.M.; Ying, J.Y. Synthesis of hexagonally packed mesoporous TiO2 by a modified sol–gel method. Angew. Chem. Int. Ed. 1995, 34, 2014–2017. [Google Scholar] [CrossRef]
- Yin, H.; Wada, Y.; Kitamura, T.; Kambe, S.; Murasawa, S.; Mori, H.; Sakata, T.; Yanagida, S. Hydrothermal synthesis of nanosized anatase and rutile TiO2 using amorphous phase TiO2. J. Mater. Chem. A 2001, 11, 1694–1703. [Google Scholar] [CrossRef]
- Bhardwaj, N.; Kundu, S.C. Electrospinning: A fascinating fiber fabrication technique. Biotechnol. Adv. 2010, 28, 325–347. [Google Scholar] [CrossRef] [PubMed]
- Taylor, G.I. Electrically driven jets. Proc. R. Soc. A 1969, 313, 453–475. [Google Scholar]
- Bognitzki, M.; Czado, W.; Frese, T.; Schaper, A.; Hellwig, M.; Steinhart, M.; Greiner, A.; Wendorff, J.H. Nanostructured fibers via electrospinning. Adv. Mater. 2001, 13, 70–72. [Google Scholar] [CrossRef]
- Spivak, A.; Dzenis, Y.; Reneker, D. Model of steady state jet in the electrospinning process. Mech. Res. Commun. 2000, 27, 37–42. [Google Scholar] [CrossRef]
- Blachowicz, T.; Ehrmann, A. Recent developments in electrospun ZnO nanofibers: A short review. J. Eng. Fiber. Fabr. 2020, 15, 1–6. [Google Scholar] [CrossRef]
- Maensiri, S.; Laokul, P.; Promarak, V. Synthesis and optical properties of nanocrystalline ZnO powders by a simple method using zinc acetate dihydrate and poly (vinyl pyrrolidone). J. Cryst. Growth 2006, 289, 102–106. [Google Scholar] [CrossRef]
- Singh, P.; Mondal, K.; Sharma, A. Reusable electrospun mesoporous ZnO nanofiber mats for photocatalytic degradation of polycyclic aromatic hydrocarbon dyes in wastewater. J. Colloid Interface Sci. 2013, 394, 208–215. [Google Scholar] [CrossRef]
- Sasikala, S.P.; Nibila, T.; Babitha, K.B.; Mohamed, A.A.P.; Solaiappan, A. Competitive photo-degradation performance of ZnO modified bentonite clay in water containing both organic and inorganic contaminants. Sustain. Environ. Res. 2019, 29, 1. [Google Scholar] [CrossRef]
- Du, P.; Song, L.; Xiong, J.; Xi, Z.; Chen, J.; Gao, L.; Wang, N. High-Efficiency Photocatalytic Degradation of Methylene Blue Using Electrospun ZnO Nanofibers as Catalyst. J. Nanosci. Nanotechnol. 2011, 11, 7723–7728. [Google Scholar] [CrossRef] [PubMed]
- Liao, D.; Badour, C.; Liao, B. Preparation of nanosized TiO2/ZnO composite catalyst and its photocatalytic activity for degradation of methyl orange. J. Photochem. Photobiol. A 2008, 194, 11–19. [Google Scholar] [CrossRef]
- Jiang, Y.; Sun, Y.; Liu, H.; Zhu, F.; Yin, H. Solar photocatalytic decolorization of CI Basic Blue 41 in an aqueous suspension of TiO2–ZnO. Dyes Pigment. 2008, 78, 77–83. [Google Scholar] [CrossRef]
- Siwińska-Stefańska, K.; Kubiak, A.; Piasecki, A.; Goscianska, J.; Nowaczyk, G.; Jurga, S.; Jesionowski, T. TiO2-ZnO binary oxide systems: Comprehensive characterization and tests of photocatalytic activity. Materials 2018, 11, 841. [Google Scholar] [CrossRef]
- Kubiak, A.; Siwińska-Ciesielczyk, K.; Bielan, Z.; Zielińska-Jurek, A.; Jesionowski, T. Synthesis of highly crystalline photocatalysts based on TiO2 and ZnO for the degradation of organic impurities under visible-light irradiation. Adsorption 2019, 25, 309–325. [Google Scholar] [CrossRef]
- Caratão, B.; Carneiro, E.; Sá, P.; Almeida, B.; Carvalho, S. Properties of electrospun TiO2 nanofibers. J. Nanotechnol. 2014, 2014, 1–5. [Google Scholar] [CrossRef]
- Cai, Z.; Song, J.; Li, J.; Zhao, F.; Luo, X.; Tang, X. Synthesis and characterization of zinc titanate fibers by sol-electrospinning method. J. Sol.-Gel Sci. Technol. 2012, 61, 49–55. [Google Scholar] [CrossRef]
- Mondal, K. Recent advances in the synthesis of metal oxide nanofibers and their environmental remediation applications. Inventions 2017, 2, 9. [Google Scholar] [CrossRef]
- Popov, A.P.; Zvyagin, A.V.; Lademann, J.; Roberts, M.S.; Sanchez, W.; Priezzhev, A.V.; Myllylä, R. Designing inorganic light-protective skin nanotechnology products. J. Biomed. Nanotechnol. 2010, 6, 432–451. [Google Scholar] [CrossRef]

| Sample Name | TZ1 | TZ2 | TZ3 | TZ4 | TZ5 |
|---|---|---|---|---|---|
| Ti/Zn molar ratio | 10:0 | 9:1 | 5:5 | 1:9 | 0:10 |
| Sample Elements | TZ1 | TZ2 | TZ3 | TZ4 | TZ5 |
|---|---|---|---|---|---|
| Ti | 26.04 | 31.57 | 14.16 | 4.72 | - |
| Zn | - | 3.28 | 13.83 | 39.67 | 44.91 |
| O | 73.96 | 65.15 | 72.01 | 55.61 | 55.09 |
| TZ4 | TZ5 | |||
|---|---|---|---|---|
| Area 1 | Area 2 | Area 1 | Area 2 | |
| Zn | 16.90 | 16.48 | 7.23 | 16.98 |
| Ti | 1.27 | 2.00 | - | - |
| O | 81.82 | 81.52 | 92.77 | 83.02 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kim, W.-T.; Na, K.-H.; Park, D.-C.; Yang, W.-H.; Choi, W.-Y. Photocatalytic Methylene Blue Degradation of Electrospun Ti–Zn Complex Oxide Nanofibers. Nanomaterials 2020, 10, 1311. https://doi.org/10.3390/nano10071311
Kim W-T, Na K-H, Park D-C, Yang W-H, Choi W-Y. Photocatalytic Methylene Blue Degradation of Electrospun Ti–Zn Complex Oxide Nanofibers. Nanomaterials. 2020; 10(7):1311. https://doi.org/10.3390/nano10071311
Chicago/Turabian StyleKim, Wan-Tae, Kyeong-Han Na, Dong-Cheol Park, Wan-Hee Yang, and Won-Youl Choi. 2020. "Photocatalytic Methylene Blue Degradation of Electrospun Ti–Zn Complex Oxide Nanofibers" Nanomaterials 10, no. 7: 1311. https://doi.org/10.3390/nano10071311
APA StyleKim, W.-T., Na, K.-H., Park, D.-C., Yang, W.-H., & Choi, W.-Y. (2020). Photocatalytic Methylene Blue Degradation of Electrospun Ti–Zn Complex Oxide Nanofibers. Nanomaterials, 10(7), 1311. https://doi.org/10.3390/nano10071311