A Novel Biocompatible Titanium–Gadolinium Quantum Dot as a Bacterial Detecting Agent with High Antibacterial Activity
Abstract
1. Introduction
2. Materials and Methods
2.1. Chemicals
2.2. Chemicals and Synthesis of Titanium–Gadolinium Quantum Dots (TGQDs)
2.3. Characterization of TGQD
2.4. Application of TGQD on Bacteria for Detection and Killing
2.5. In Vitro Cytotoxicity Testing Assay
3. Results and Discussion
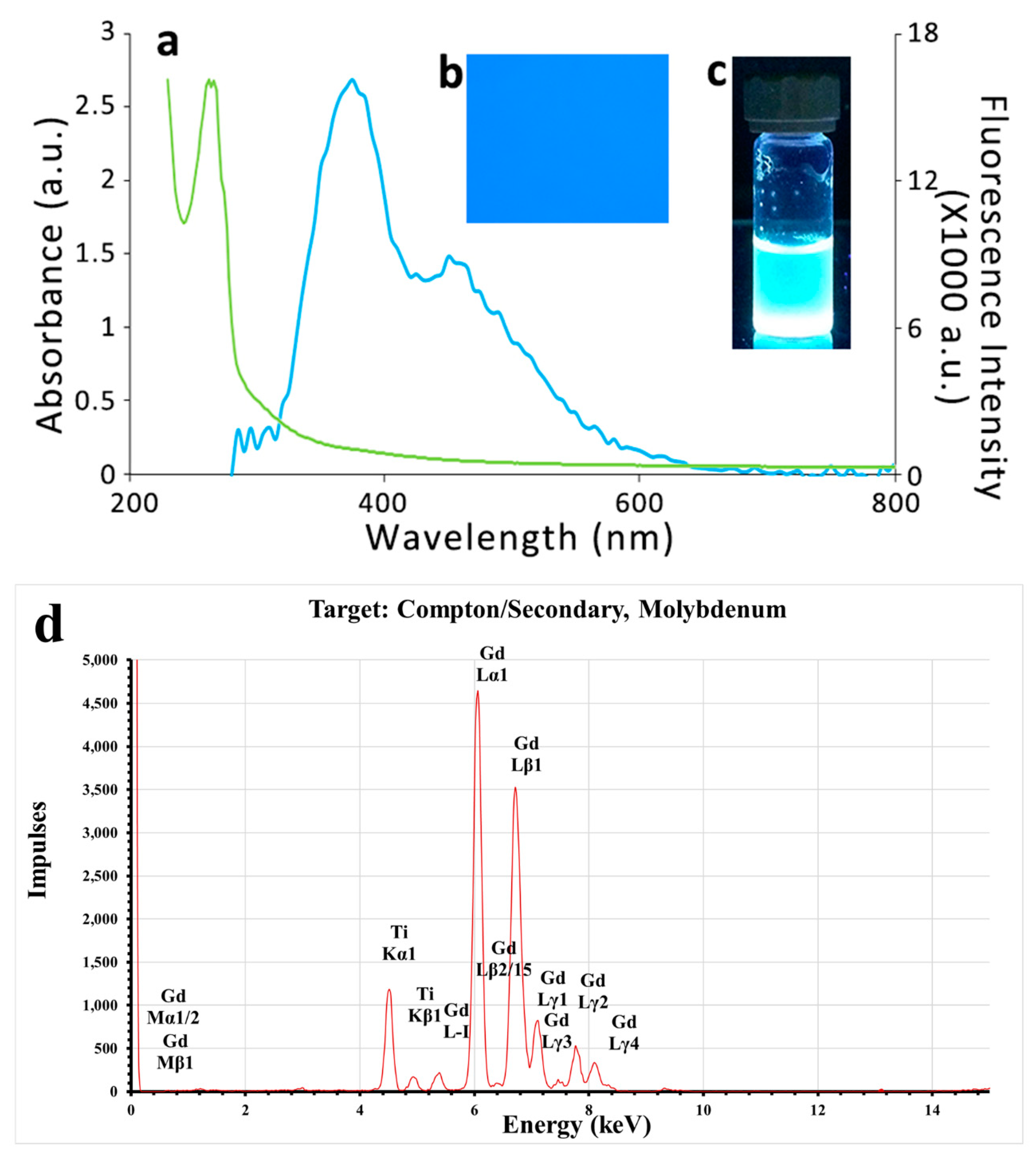
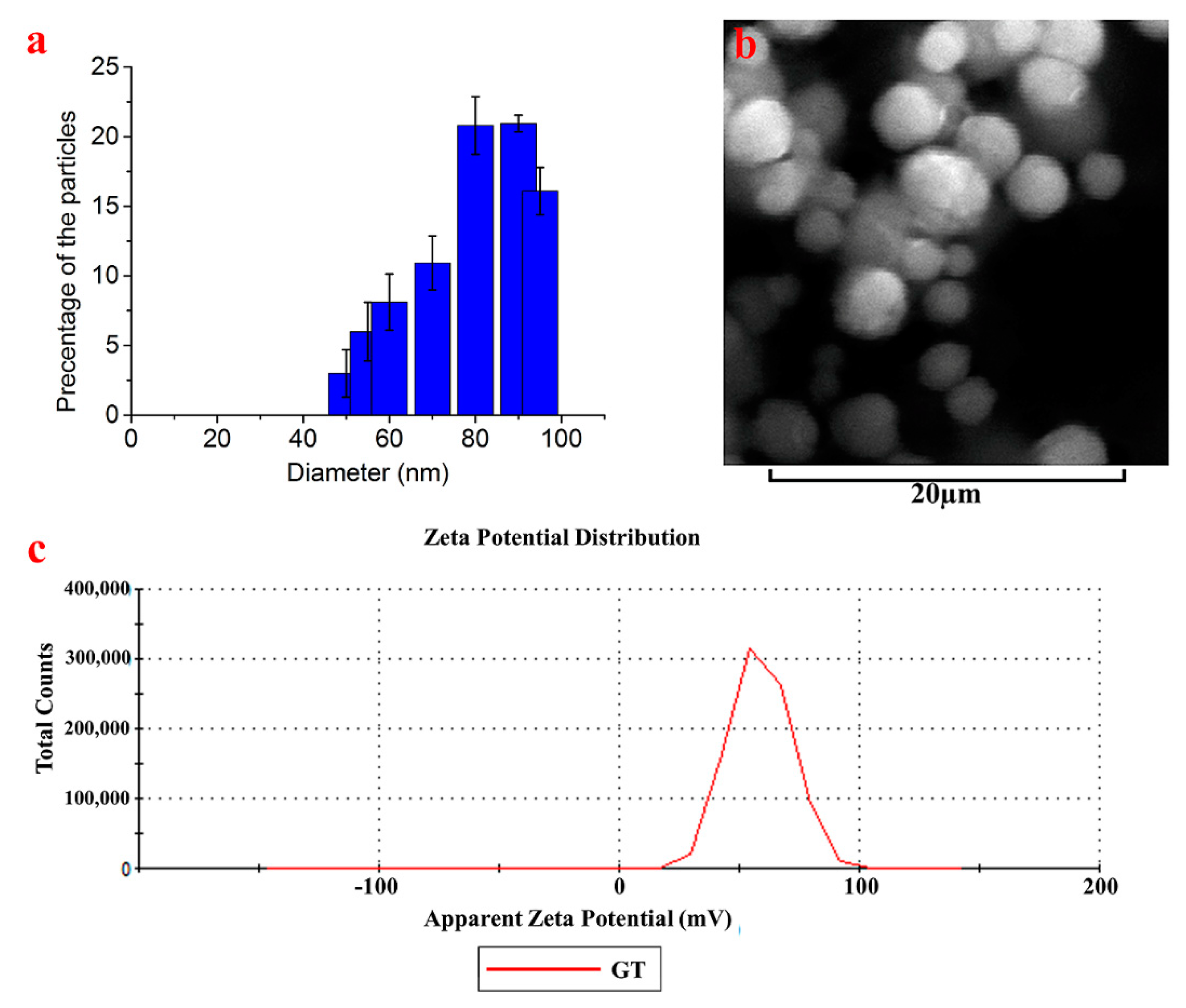
3.1. Synthesis and Characterization of TGQDs
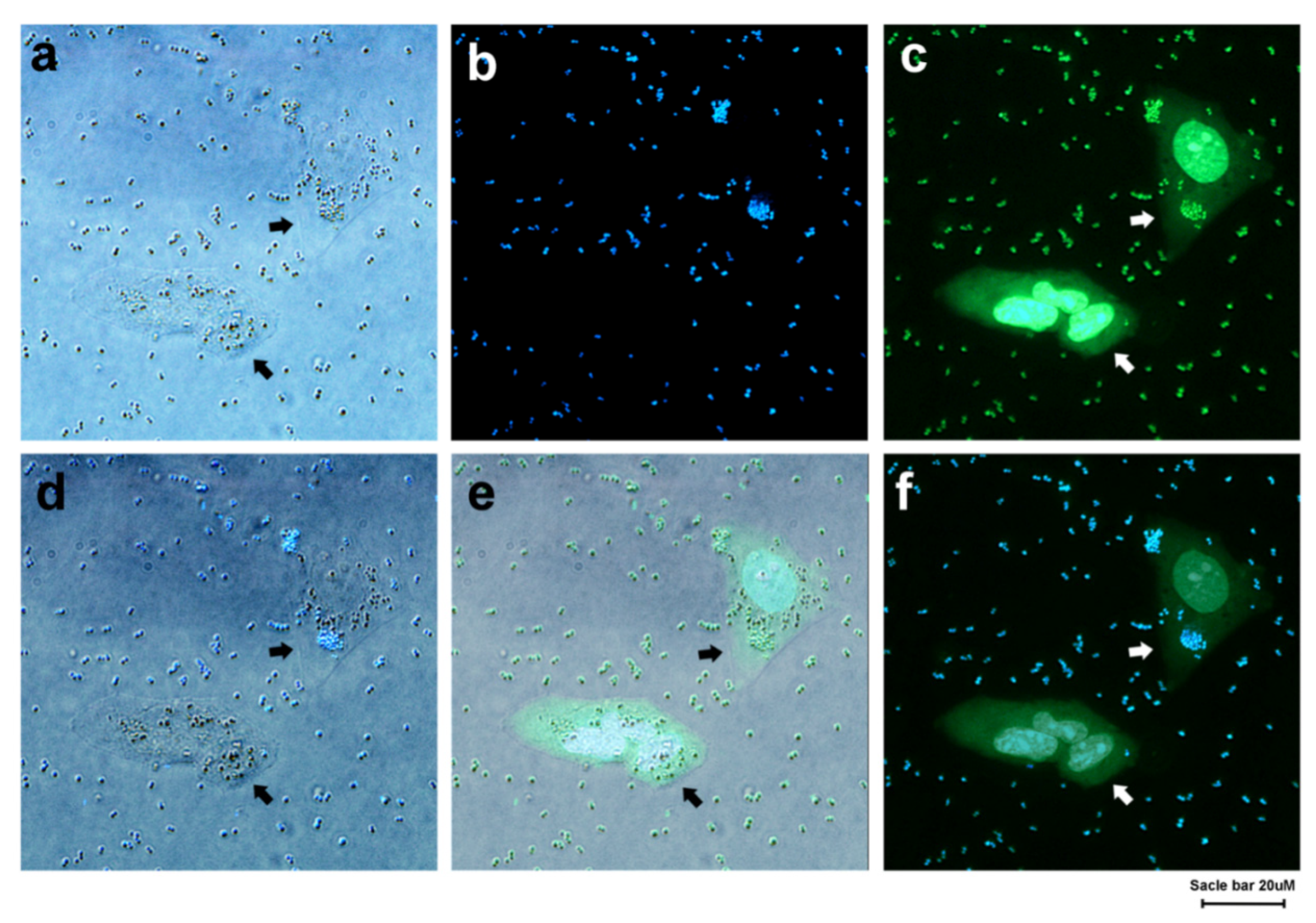
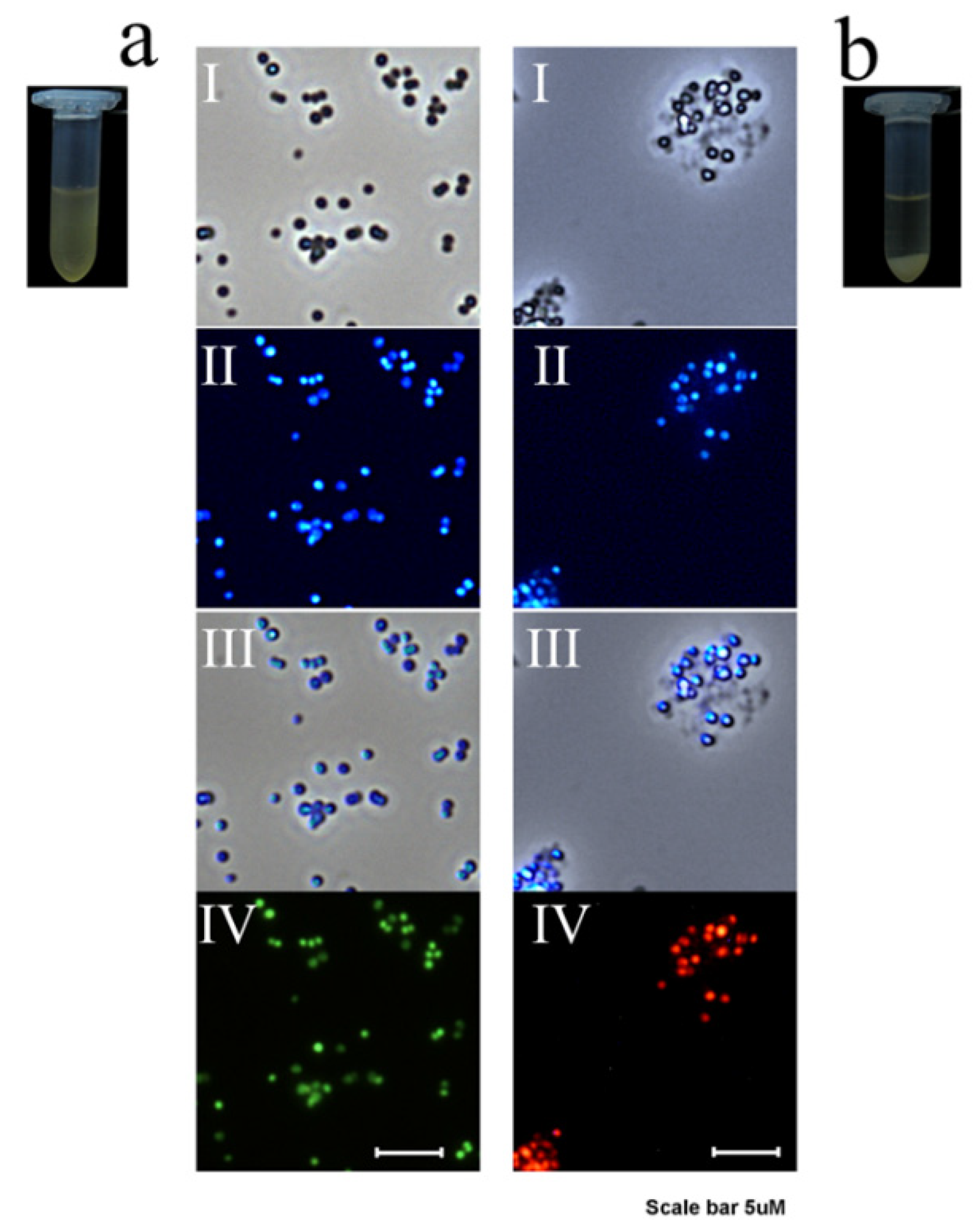
3.2. Detection of Bacteria
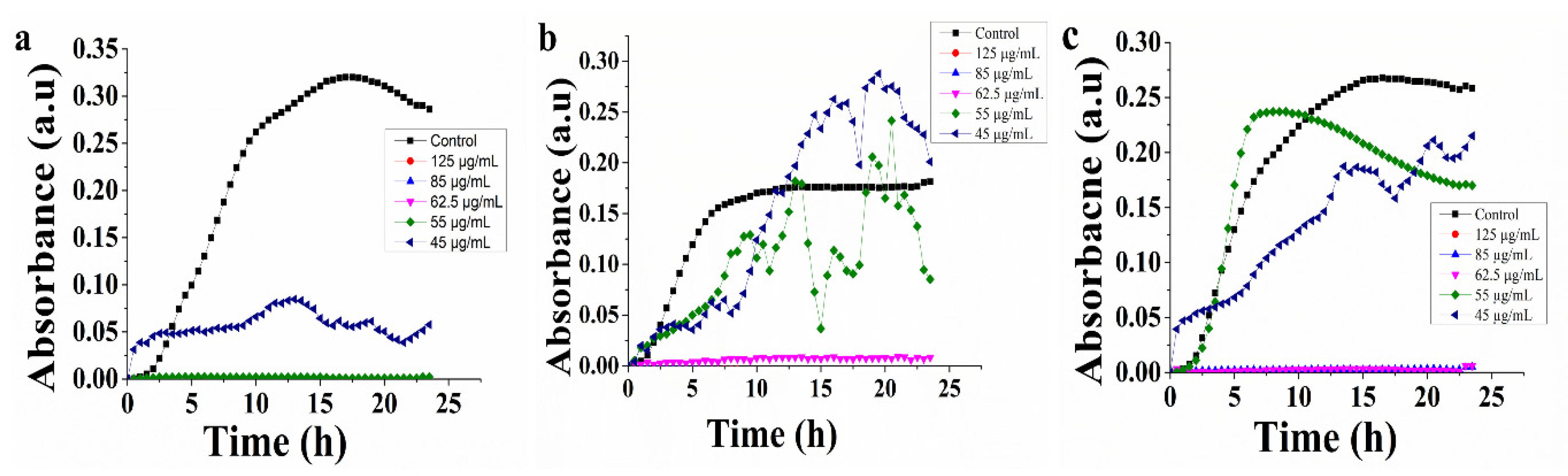
3.3. Antibacterial Activities
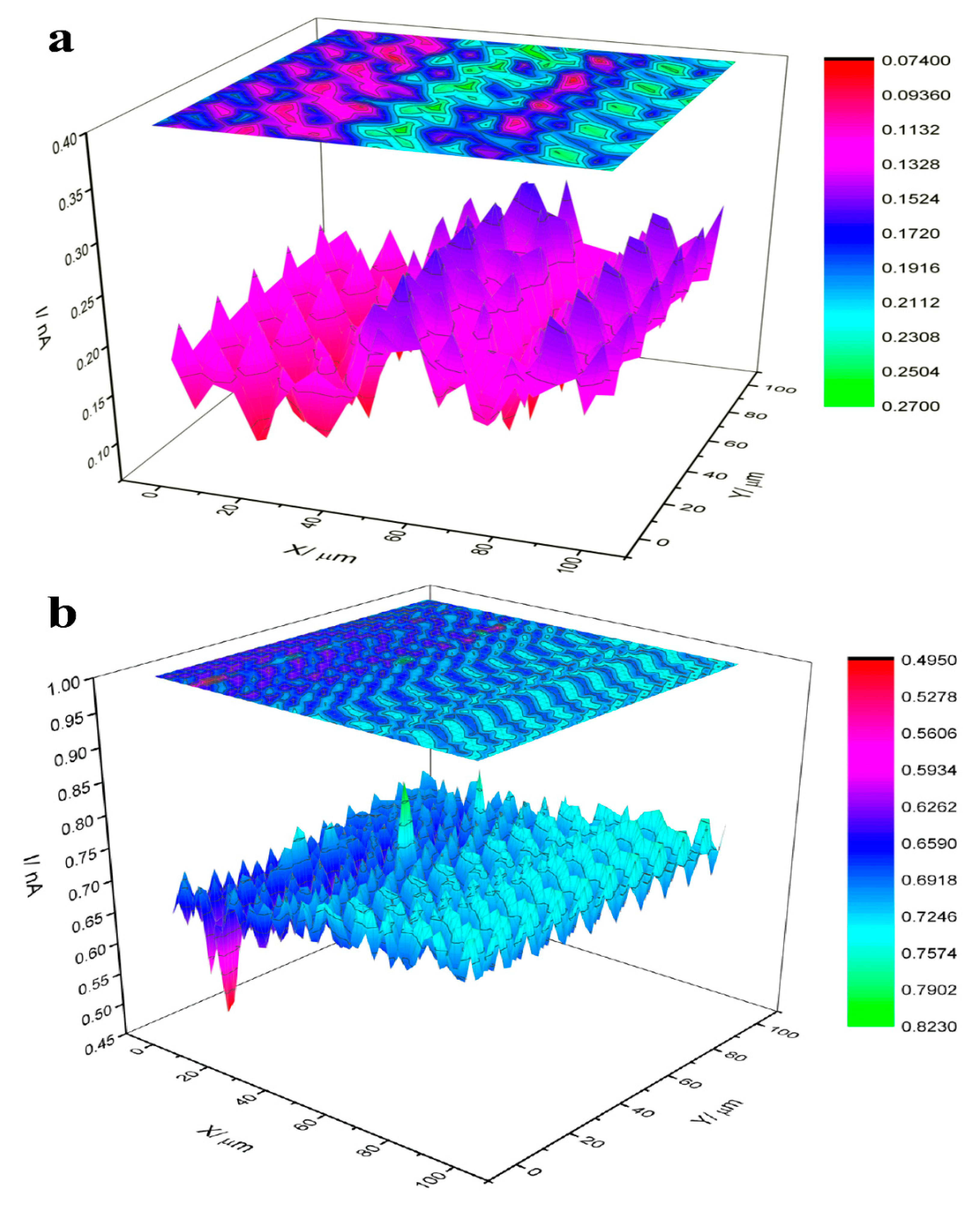
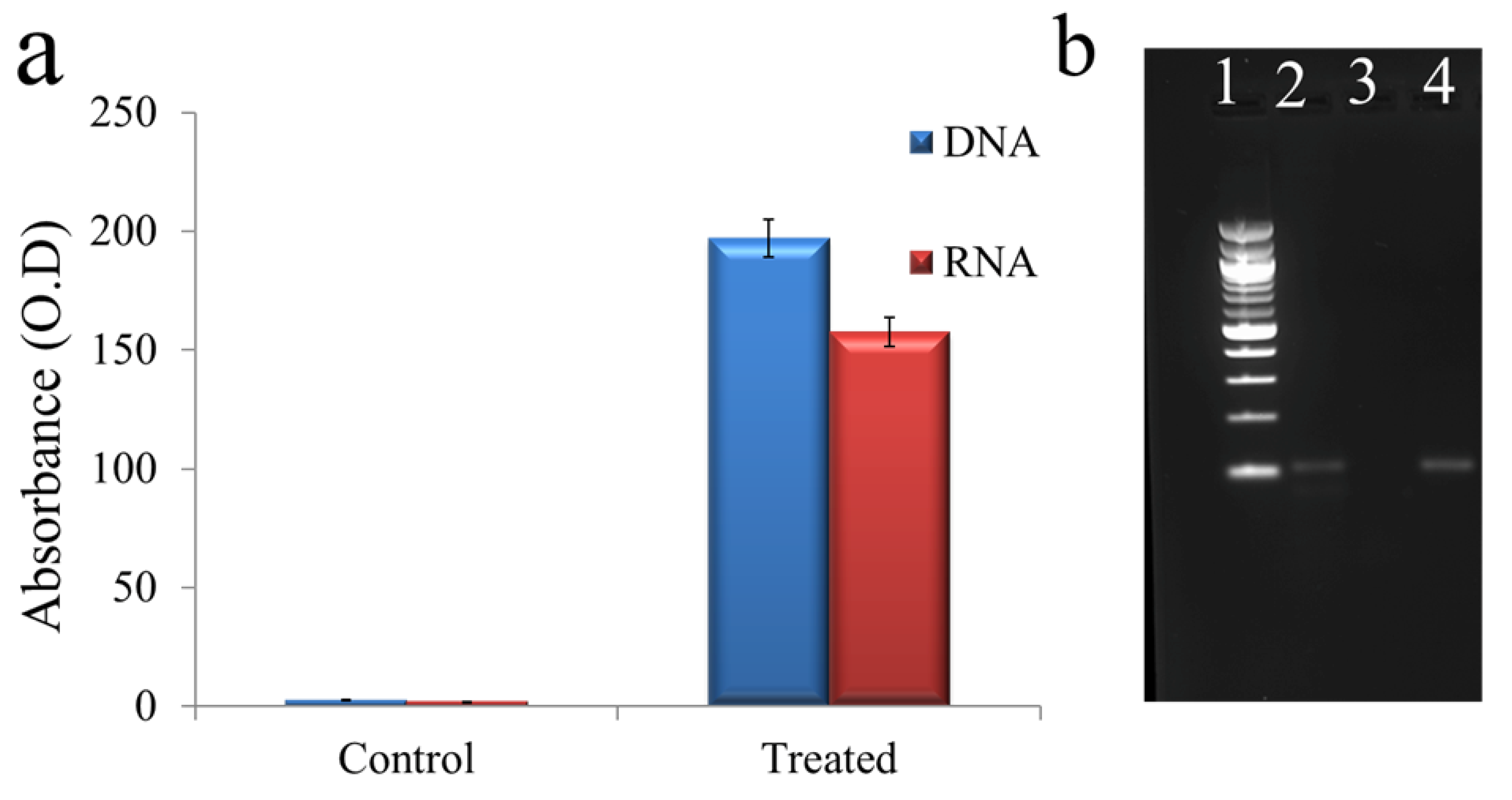
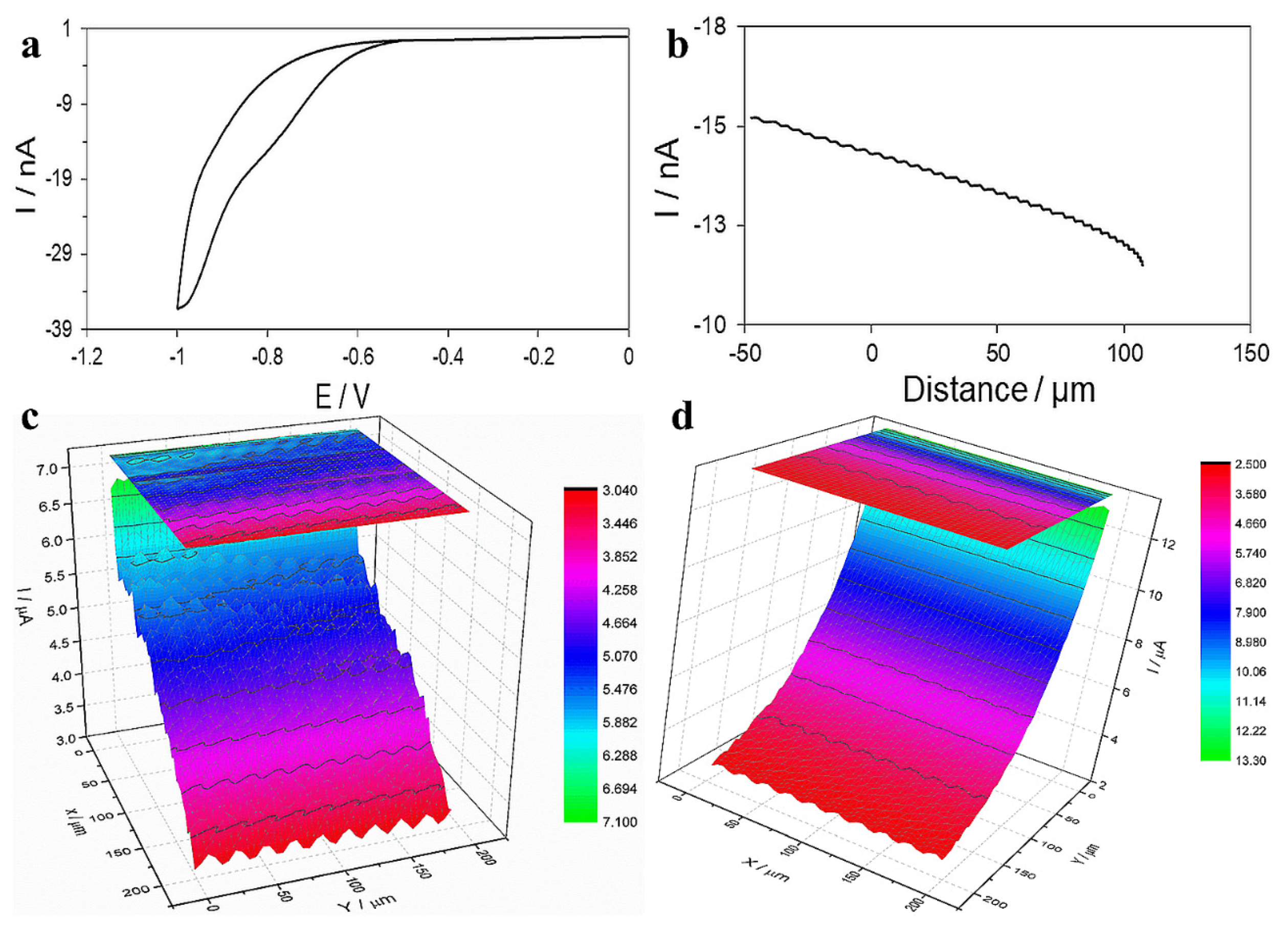
3.4. Mechanism of Actions
3.5. In Vitro Cytotoxicity Analysis
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Tacconelli, E.; Carrara, E.; Savoldi, A.; Harbarth, S.; Mendelson, M.; Monnet, D.L.; Pulcini, C.; Kahlmeter, G.; Kluytmans, J.; Carmeli, Y.; et al. Discovery, research, and development of new antibiotics: The WHO priority list of antibiotic-resistant bacteria and tuberculosis. Lancet Infect. Dis. 2018, 18, 318–327. [Google Scholar] [CrossRef]
- Foster, T.J. Antibiotic resistance in Staphylococcus aureus. Current status and future prospects. FEMS Microbiol. Rev. 2017, 41, 430–449. [Google Scholar] [CrossRef] [PubMed]
- Barker, A.K.; Brown, K.; Ahsan, M.; Sengupta, S.; Safdar, N. Social determinants of antibiotic misuse: A qualitative study of community members in Haryana, India. BMC Public Health 2017, 17, 333. [Google Scholar] [CrossRef] [PubMed]
- Antibiotics: Are You Misusing Them? Available online: https://www.mayoclinic.org/healthy-lifestyle/consumer-health/in-depth/antibiotics/art-20045720 (accessed on 16 April 2020).
- NHS U. Side Effects of Antibiotics. Available online: https://www.nhs.uk/conditions/antibiotics/side-effects/ (accessed on 16 April 2020).
- Westphal, J.F.; Vetter, D.; Brogard, J.M. Hepatic side-effects of antibiotics. J. Antimicrob. Chemother. 1994, 33, 387–401. [Google Scholar] [CrossRef]
- Dang, C.N.; Prasad, Y.D.M.; Boulton, A.J.M.; Jude, E.B. Methicillin-resistant Staphylococcus aureus in the diabetic foot clinic: A worsening problem. Diabet. Med. 2003, 20, 159–161. [Google Scholar] [CrossRef]
- Baptista, P.V.; McCusker, M.P.; Carvalho, A.; Ferreira, D.A.; Mohan, N.M.; Martins, M.; Fernandes, A.R. Nano-Strategies to Fight Multidrug Resistant Bacteria-“A Battle of the Titans”. Front. Microbiol. 2018, 9, 1441. [Google Scholar] [CrossRef]
- Habiba, K.; Bracho-Rincon, D.P.; Gonzalez-Feliciano, J.A.; Villalobos-Santos, J.C.; Makarov, V.I.; Ortiz, D.; Avalos, J.A.; Gonzalez, C.I.; Weiner, B.R.; Morell, G. Synergistic antibacterial activity of PEGylated silver–graphene quantum dots nanocomposites. Appl. Mater. Today 2015, 1, 80–87. [Google Scholar] [CrossRef]
- Alanis, A.J. Resistance to Antibiotics: Are We in the Post-Antibiotic Era? Arch. Med. Res. 2005, 36, 697–705. [Google Scholar] [CrossRef]
- Sang, Y.; Blecha, F. Antimicrobial peptides and bacteriocins: Alternatives to traditional antibiotics. Anim. Health Res. Rev. 2008, 9, 227–235. [Google Scholar] [CrossRef]
- Edwards-Jones, V. Chapter 1—Alternative Antimicrobial Approaches to Fighting Multidrug-Resistant Infections. In Fighting Multidrug Resistance with Herbal Extracts, Essential Oils and Their Components; Rai, M.K., Kon, K.V., Eds.; Academic Press: San Diego, CA, USA, 2013; pp. 1–9. [Google Scholar] [CrossRef]
- Kuete, V. Chapter 3—Bioactivity of Plant Constituents against Vancomycin-Resistant Enterococci. In Fighting Multidrug Resistance with Herbal Extracts, Essential Oils and Their Components; Rai, M.K., Kon, K.V., Eds.; Academic Press: San Diego, CA, USA, 2013; pp. 23–30. [Google Scholar] [CrossRef]
- Ramírez Rueda, R.Y. Chapter 2—Natural Plant Products Used against Methicillin-Resistant Staphylococcus aureus. In Fighting Multidrug Resistance with Herbal Extracts, Essential Oils and Their Components; Rai, M.K., Kon, K.V., Eds.; Academic Press: San Diego, CA, USA, 2013; pp. 11–22. [Google Scholar] [CrossRef]
- Barros, C.H.N.; Fulaz, S.; Stanisic, D.; Tasic, L. Biogenic Nanosilver against Multidrug-Resistant Bacteria (MDRB). Antibiotics 2018, 7, 69. [Google Scholar] [CrossRef]
- Pal, I.; Bhattacharyya, D.; Kar, R.K.; Zarena, D.; Bhunia, A.; Atreya, H.S. A Peptide-Nanoparticle System with Improved Efficacy against Multidrug Resistant Bacteria. Sci. Rep. 2019, 9, 4485. [Google Scholar] [CrossRef] [PubMed]
- Hemeg, H.A. Nanomaterials for alternative antibacterial therapy. Int. J. Nanomed. 2017, 12, 8211–8225. [Google Scholar] [CrossRef] [PubMed]
- Patra, J.K.; Das, G.; Fraceto, L.F.; Campos, E.V.R.; Rodriguez-Torres, M.D.P.; Acosta-Torres, L.S.; Diaz-Torres, L.A.; Grillo, R.; Swamy, M.K.; Sharma, S.; et al. Nano based drug delivery systems: Recent developments and future prospects. J. Nanobiotechnol. 2018, 16, 71. [Google Scholar] [CrossRef] [PubMed]
- Sur, V.P.; Kominkova, M.; Buchtova, Z.; Dolezelikova, K.; Zitka, O.; Moulick, A. CdSe QD Biosynthesis in Yeast Using Tryptone-Enriched Media and Their Conjugation with a Peptide Hecate for Bacterial Detection and Killing. Nanomaterials 2019, 9, 1463. [Google Scholar] [CrossRef]
- Thurn, K.T.; Brown, E.; Wu, A.; Vogt, S.; Lai, B.; Maser, J.; Paunesku, T.; Woloschak, G.E. Nanoparticles for applications in cellular imaging. Nanoscale Res. Lett. 2007, 2, 430–441. [Google Scholar] [CrossRef]
- Bahadar, H.; Maqbool, F.; Niaz, K.; Abdollahi, M. Toxicity of Nanoparticles and an Overview of Current Experimental Models. Iran. Biomed. J. 2016, 20, 1–11. [Google Scholar] [CrossRef]
- Crisponi, G.; Nurchi, V.M.; Lachowicz, J.I.; Peana, M.; Medici, S.; Zoroddu, M.A. Chapter 18—Toxicity of Nanoparticles: Etiology and Mechanisms. In Antimicrobial Nanoarchitectonics; Grumezescu, A.M., Ed.; Elsevier: Amsterdam, The Netherlands, 2017; pp. 511–546. [Google Scholar] [CrossRef]
- Stocks, S.M. Mechanism and use of the commercially available viability stain, BacLight. Cytom. Part A 2004, 61, 189–195. [Google Scholar] [CrossRef]
- Datta, S.; Sherman, J.M.; Bravard, M.A.; Valencia, T.; Gilman, R.H.; Evans, C.A. Clinical Evaluation of Tuberculosis Viability Microscopy for Assessing Treatment Response. Clin. Infect. Dis. 2014, 60, 1186–1195. [Google Scholar] [CrossRef]
- Kanade, S.; Nataraj, G.; Ubale, M.; Mehta, P. Fluorescein diacetate vital staining for detecting viability of acid-fast bacilli in patients on antituberculosis treatment. Int. J. Mycobacteriol. 2016, 5, 294–298. [Google Scholar] [CrossRef]
- Azzopardi, E.A.; Ferguson, E.L.; Thomas, D.W. The enhanced permeability retention effect: A new paradigm for drug targeting in infection. J. Antimicrob. Chemother. 2012, 68, 257–274. [Google Scholar] [CrossRef]
- Anderson, D.; Mj, S. Nanotechnology: The Risks and Benefits for Medical Diagnosis and Treatment. J. Nanomed. Nanotechnol. 2016, 7. [Google Scholar] [CrossRef]
- Moulick, A.; Heger, Z.; Milosavljevic, V.; Richtera, L.; Barroso-Flores, J.; Merlos Rodrigo, M.A.; Buchtelova, H.; Podgajny, R.; Hynek, D.; Kopel, P.; et al. Real-Time Visualization of Cell Membrane Damage Using Gadolinium–Schiff Base Complex-Doped Quantum Dots. ACS Appl. Mater. Interfaces 2018, 10, 35859–35868. [Google Scholar] [CrossRef] [PubMed]
- Ristic, B.Z.; Milenkovic, M.M.; Dakic, I.R.; Todorovic-Markovic, B.M.; Milosavljevic, M.S.; Budimir, M.D.; Paunovic, V.G.; Dramicanin, M.D.; Markovic, Z.M.; Trajkovic, V.S. Photodynamic antibacterial effect of graphene quantum dots. Biomaterials 2014, 35, 4428–4435. [Google Scholar] [CrossRef] [PubMed]
- Li, H.; Huang, J.; Song, Y.; Zhang, M.; Wang, H.; Lu, F.; Huang, H.; Liu, Y.; Dai, X.; Gu, Z.; et al. Degradable Carbon Dots with Broad-Spectrum Antibacterial Activity. ACS Appl. Mater. Interfaces 2018, 10, 26936–26946. [Google Scholar] [CrossRef] [PubMed]
- Garcia, I.M.; Leitune, V.C.B.; Visioli, F.; Samuel, S.M.W.; Collares, F.M. Influence of zinc oxide quantum dots in the antibacterial activity and cytotoxicity of an experimental adhesive resin. J. Dent. 2018, 73, 57–60. [Google Scholar] [CrossRef]
- Meikhail, M.S.; Abdelghany, A.M.; Awad, W.M. Role of CdSe quantum dots in the structure and antibacterial activity of chitosan/poly ɛ-caprolactone thin films. Egypt. J. Basic Appl. Sci. 2018, 5, 138–144. [Google Scholar] [CrossRef]
- Belletti, D.; Riva, G.; Luppi, M.; Tosi, G.; Forni, F.; Vandelli, M.A.; Ruozi, B.; Pederzoli, F. Anticancer drug-loaded quantum dots engineered polymeric nanoparticles: Diagnosis/therapy combined approach. Eur. J. Pharm. Sci. 2017, 107, 230–239. [Google Scholar] [CrossRef]
- Liu, Z.; Lin, Q.; Huang, Q.; Liu, H.; Bao, C.; Zhang, W.; Zhong, X.; Zhu, L. Semiconductor quantum dots photosensitizing release of anticancer drug. Chem. Commun. 2011, 47, 1482–1484. [Google Scholar] [CrossRef]
- Han, C.; Lalley, J.; Namboodiri, D.; Cromer, K.; Nadagouda, M.N. Titanium dioxide-based antibacterial surfaces for water treatment. Curr. Opin. Chem. Eng. 2016, 11, 46–51. [Google Scholar] [CrossRef]
- Haugen, H.; Lyngstadaas, S. Antibacterial effects of titanium dioxide in wounds. In Wound Healing Biomaterials; Elsevier: Amsterdam, The Netherlands, 2016; pp. 439–450. [Google Scholar]
- Kubacka, A.; Diez, M.S.; Rojo, D.; Bargiela, R.; Ciordia, S.; Zapico, I.; Albar, J.P.; Barbas, C.; Dos Santos, V.A.M.; Fernández-García, M. Understanding the antimicrobial mechanism of TiO 2-based nanocomposite films in a pathogenic bacterium. Sci. Rep. 2014, 4, 4134. [Google Scholar] [CrossRef]
- Besinis, A.; De Peralta, T.; Handy, R.D. The antibacterial effects of silver, titanium dioxide and silica dioxide nanoparticles compared to the dental disinfectant chlorhexidine on Streptococcus mutans using a suite of bioassays. Nanotoxicology 2014, 8, 1–16. [Google Scholar] [CrossRef] [PubMed]
- Karthikeyan, G.; Mohanraj, K.; Elango, K.P.; Girishkumar, K. Synthesis, spectroscopic characterization and antibacterial activity of lanthanide–tetracycline complexes. Transit. Met. Chem. 2004, 29, 86–90. [Google Scholar] [CrossRef]
- Peng, P.; Jiang, W.-P.; Li, S.-M.; Chen, C.-Z.; Liu, G.-H. Synthesis, characterization and antibacterial activity of complex for gadolinium iodide with thiourea. Appl. Chem. Ind. 2011, 1, 100–103. [Google Scholar]
- Pradhan, N.; Peng, X. Efficient and Color-Tunable Mn-Doped ZnSe Nanocrystal Emitters: Control of Optical Performance via Greener Synthetic Chemistry. J. Am. Chem. Soc. 2007, 129, 3339–3347. [Google Scholar] [CrossRef] [PubMed]
- Moulick, A.; Blazkova, I.; Milosavljevic, V.; Fohlerova, Z.; Hubalek, J.; Kopel, P.; Vaculovicova, M.; Adam, V.; Kizek, R. Application of CdTe/ZnSe Quantum Dots in In Vitro Imaging of Chicken Tissue and Embryo. Photochem. Photobiol. 2015, 91, 417–423. [Google Scholar] [CrossRef] [PubMed]
- Jelinkova, P.; Koudelkova, Z.; Milosavljevic, V.; Horky, P.; Kopel, P.; Adam, V. Utilization of Selenium Nanoparticles with Schiff Base Chitosan as Antibacterial Agents. MendelNet 2016, 989–993. Available online: https://pdfs.semanticscholar.org/6ba5/800ec37f37ab26508fefcafd3ac3056f3201.pdf (accessed on 16 April 2020).
- Herathge, N.D.S.; George, J.T.; Rowley, D.A. Differential antimicrobial activities of Human Beta-Defensins against Methicillin Resistant (MRSA) and Methicillin sensitive (MSSA) Staphylococcus aureus. In Science and Technology Against Microbial Pathogens; World Scientific: Singapore, 2011; pp. 9–12. [Google Scholar] [CrossRef]
- Mazumdar, A.; Haddad, Y.; Milosavljevic, V.; Michalkova, H.; Guran, R.; Bhowmick, S.; Moulick, A. Peptide-Carbon Quantum Dots conjugate, Derived from Human Retinoic Acid Receptor Responder Protein 2, against Antibiotic-Resistant Gram Positive and Gram Negative Pathogenic Bacteria. Nanomaterials 2020, 10, 325. [Google Scholar] [CrossRef]
- Jelinkova, P.; Splichal, Z.; Jimenez, A.M.J.; Haddad, Y.; Mazumdar, A.; Sur, V.P.; Milosavljevic, V.; Kopel, P.; Buchtelova, H.; Guran, R.; et al. Novel vancomycin-peptide conjugate as potent antibacterial agent against vancomycin-resistant Staphylococcus aureus. Infect. Drug Resist. 2018, 11, 1807–1817. [Google Scholar] [CrossRef]
- Chudobova, D.; Cihalova, K.; Dostalova, S.; Ruttkay-Nedecky, B.; Merlos Rodrigo, M.A.; Tmejova, K.; Kopel, P.; Nejdl, L.; Kudr, J.; Gumulec, J.; et al. Comparison of the effects of silver phosphate and selenium nanoparticles on Staphylococcus aureus growth reveals potential for selenium particles to prevent infection. FEMS Microbiol. Lett. 2014, 351, 195–201. [Google Scholar] [CrossRef]
- Richter, S.G.; Elli, D.; Kim, H.K.; Hendrickx, A.P.A.; Sorg, J.A.; Schneewind, O.; Missiakas, D. Small molecule inhibitor of lipoteichoic acid synthesis is an antibiotic for Gram-positive bacteria. Proc. Natl. Acad. Sci. USA 2013, 110, 3531–3536. [Google Scholar] [CrossRef]
- Vukomanovic, M.; Torrents, E. High time resolution and high signal-to-noise monitoring of the bacterial growth kinetics in the presence of plasmonic nanoparticles. J. Nanobiotechnol. 2019, 17, 21. [Google Scholar] [CrossRef]
- Stevenson, K.; McVey, A.F.; Clark, I.B.N.; Swain, P.S.; Pilizota, T. General calibration of microbial growth in microplate readers. Sci. Rep. 2016, 6, 38828. [Google Scholar] [CrossRef] [PubMed]
- van Sorge, N.M.; Beasley, F.C.; Gusarov, I.; Gonzalez, D.J.; von Köckritz-Blickwede, M.; Anik, S.; Borkowski, A.W.; Dorrestein, P.C.; Nudler, E.; Nizet, V. Methicillin-resistant Staphylococcus aureus Bacterial Nitric-oxide Synthase Affects Antibiotic Sensitivity and Skin Abscess Development. J. Biol. Chem. 2013, 288, 6417–6426. [Google Scholar] [CrossRef] [PubMed]
- Schumacher, A.; Vranken, T.; Malhotra, A.; Arts, J.J.C.; Habibovic, P. In vitro antimicrobial susceptibility testing methods: Agar dilution to 3D tissue-engineered models. Eur. J. Clin. Microbiol. Infect. Dis. 2018, 37, 187–208. [Google Scholar] [CrossRef] [PubMed]
- Stasiak-Różańska, L.; Błażejak, S.; Gientka, I. Effect of glycerol and dihydroxyacetone concentrations in the culture medium on the growth of acetic acid bacteria Gluconobacter oxydans ATCC 621. Eur. Food Res. Technol. 2014, 239, 453–461. [Google Scholar] [CrossRef]
- Hogenkamp, A.; Herías, M.V.; Tooten, P.C.J.; Veldhuizen, E.J.A.; Haagsman, H.P. Effects of surfactant protein D on growth, adhesion and epithelial invasion of intestinal Gram-negative bacteria. Mol. Immunol. 2007, 44, 3517–3527. [Google Scholar] [CrossRef] [PubMed]
- Li, R.C.; Nix, D.E.; Schentag, J.J. New turbidimetric assay for quantitation of viable bacterial densities. Antimicrob. Agents Chemother. 1993, 37, 371–374. [Google Scholar] [CrossRef]
- Sacar, M.; Sacar, S.; Cevahir, N.; Onem, G.; Teke, Z.; Asan, A.; Turgut, H.; Adali, F.; Kaleli, I.; Susam, I.; et al. Comparison of antimicrobial agents as therapy for experimental endocarditis: Caused by methicillin-resistant Staphylococcus aureus. Tex Heart Inst. J. 2010, 37, 400–404. [Google Scholar]
- Popova, M.; Martin, C.; Morgavi, D.P. Improved protocol for high-quality Co-extraction of DNA and RNA from rumen digesta. Folia Microbiol. 2010, 55, 368–372. [Google Scholar] [CrossRef]
- Welsh, S.; Peakman, T.; Sheard, S.; Almond, R. Comparison of DNA quantification methodology used in the DNA extraction protocol for the UK Biobank cohort. BMC Genom. 2017, 18, 26. [Google Scholar] [CrossRef]
- Quantification of DNA and RNA: A Spectrophotometric Method. In eLS; Wiley: New York, NY, USA, 2003. [CrossRef]
- Jorgez, C.J.; Dang, D.D.; Simpson, J.L.; Lewis, D.E.; Bischoff, F.Z. Quantity versus quality: Optimal methods for cell-free DNA isolation from plasma of pregnant women. Genet. Med. 2006, 8, 615–619. [Google Scholar] [CrossRef]
- Wilfinger, W.W.; Mackey, K.; Chomczynski, P. Effect of pH and Ionic Strength on the Spectrophotometric Assessment of Nucleic Acid Purity. BioTechniques 1997, 22, 474–481. [Google Scholar] [CrossRef]
- Heger, Z.; Merlos Rodrigo, M.A.; Michalek, P.; Polanska, H.; Masarik, M.; Vit, V.; Plevova, M.; Pacik, D.; Eckschlager, T.; Stiborova, M.; et al. Sarcosine Up-Regulates Expression of Genes Involved in Cell Cycle Progression of Metastatic Models of Prostate Cancer. PLoS ONE 2016, 11, e0165830. [Google Scholar] [CrossRef]
- Trivedi, M.K.; Branton, A.; Trivedi, D.; Nayak, G.; Bairwa, K.; Jana, S. Spectroscopic Characterization of Disodium Hydrogen Orthophosphate and Sodium Nitrate after Biofield Treatment. J. Chromatogr. Sep. Tech. 2015, 6, 5. [Google Scholar] [CrossRef]
- Perrin, F.X.; Nguyen, V.; Vernet, J.L. FT-IR Spectroscopy of Acid-Modified Titanium Alkoxides: Investigations on the Nature of Carboxylate Coordination and Degree of Complexation. J. Sol-Gel Sci. Technol. 2003, 28, 205–215. [Google Scholar] [CrossRef]
- Sondi, I.; Salopek-Sondi, B. Silver nanoparticles as antimicrobial agent: A case study on E. coli as a model for Gram-negative bacteria. J. Colloid Interface Sci. 2004, 275, 177–182. [Google Scholar] [CrossRef] [PubMed]
- Diana, P.; Sigifredo, O.; Jaime, R.; María Claudia, V.; Julieta, P.; Orville, H.; Jinnethe, R.; César, A.A. First Characterization of a Cluster of VanA-Type Glycopeptide-Resistant Enterococcus faecium Colombia. Emerg. Infect. Dis. J. 2002, 8, 961. [Google Scholar] [CrossRef]
- Qureshi, N.K.; Yin, S.; Boyle-Vavra, S. The role of the Staphylococcal VraTSR regulatory system on vancomycin resistance and vanA operon expression in vancomycin-resistant Staphylococcus aureus. PLoS ONE 2014, 9, e85873. [Google Scholar] [CrossRef]
- Liu, B.; Cheng, W.; Rotenberg, S.A.; Mirkin, M.V. Scanning electrochemical microscopy of living cells: Part 2. Imaging redox and acid/basic reactivities. J. Electroanal. Chem. 2001, 500, 590–597. [Google Scholar] [CrossRef]
- Bard, A.J.; Mirkin, M.V. Scanning Electrochemical Microscopy; CRC Press: Florida, FL, USA, 2012. [Google Scholar]
- Kim, S.R.; Park, M.J.; Lee, M.K.; Sung, S.H.; Park, E.J.; Kim, J.; Kim, S.Y.; Oh, T.H.; Markelonis, G.J.; Kim, Y.C. Flavonoids of Inula britannica protect cultured cortical cells from necrotic cell death induced by glutamate. Free Radic. Biol. Med. 2002, 32, 596–604. [Google Scholar] [CrossRef]
- Shoemaker, M.; Cohen, I.; Campbell, M. Reduction of MTT by aqueous herbal extracts in the absence of cells. J. Ethnopharmacol. 2004, 93, 381–384. [Google Scholar] [CrossRef] [PubMed]
| Bacterial Strains | TGQD (µg/mL) |
|---|---|
| S. aureus | 55 |
| MRSA | 62.5 |
| VRSA | 62.5 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sur, V.P.; Mazumdar, A.; Ashrafi, A.; Mukherjee, A.; Milosavljevic, V.; Michalkova, H.; Kopel, P.; Richtera, L.; Moulick, A. A Novel Biocompatible Titanium–Gadolinium Quantum Dot as a Bacterial Detecting Agent with High Antibacterial Activity. Nanomaterials 2020, 10, 778. https://doi.org/10.3390/nano10040778
Sur VP, Mazumdar A, Ashrafi A, Mukherjee A, Milosavljevic V, Michalkova H, Kopel P, Richtera L, Moulick A. A Novel Biocompatible Titanium–Gadolinium Quantum Dot as a Bacterial Detecting Agent with High Antibacterial Activity. Nanomaterials. 2020; 10(4):778. https://doi.org/10.3390/nano10040778
Chicago/Turabian StyleSur, Vishma Pratap, Aninda Mazumdar, Amirmansoor Ashrafi, Atripan Mukherjee, Vedran Milosavljevic, Hana Michalkova, Pavel Kopel, Lukáš Richtera, and Amitava Moulick. 2020. "A Novel Biocompatible Titanium–Gadolinium Quantum Dot as a Bacterial Detecting Agent with High Antibacterial Activity" Nanomaterials 10, no. 4: 778. https://doi.org/10.3390/nano10040778
APA StyleSur, V. P., Mazumdar, A., Ashrafi, A., Mukherjee, A., Milosavljevic, V., Michalkova, H., Kopel, P., Richtera, L., & Moulick, A. (2020). A Novel Biocompatible Titanium–Gadolinium Quantum Dot as a Bacterial Detecting Agent with High Antibacterial Activity. Nanomaterials, 10(4), 778. https://doi.org/10.3390/nano10040778









