Thin Films Based on Cobalt Phthalocyanine:C60 Fullerene:ZnO Hybrid Nanocomposite Obtained by Laser Evaporation
Abstract
1. Introduction
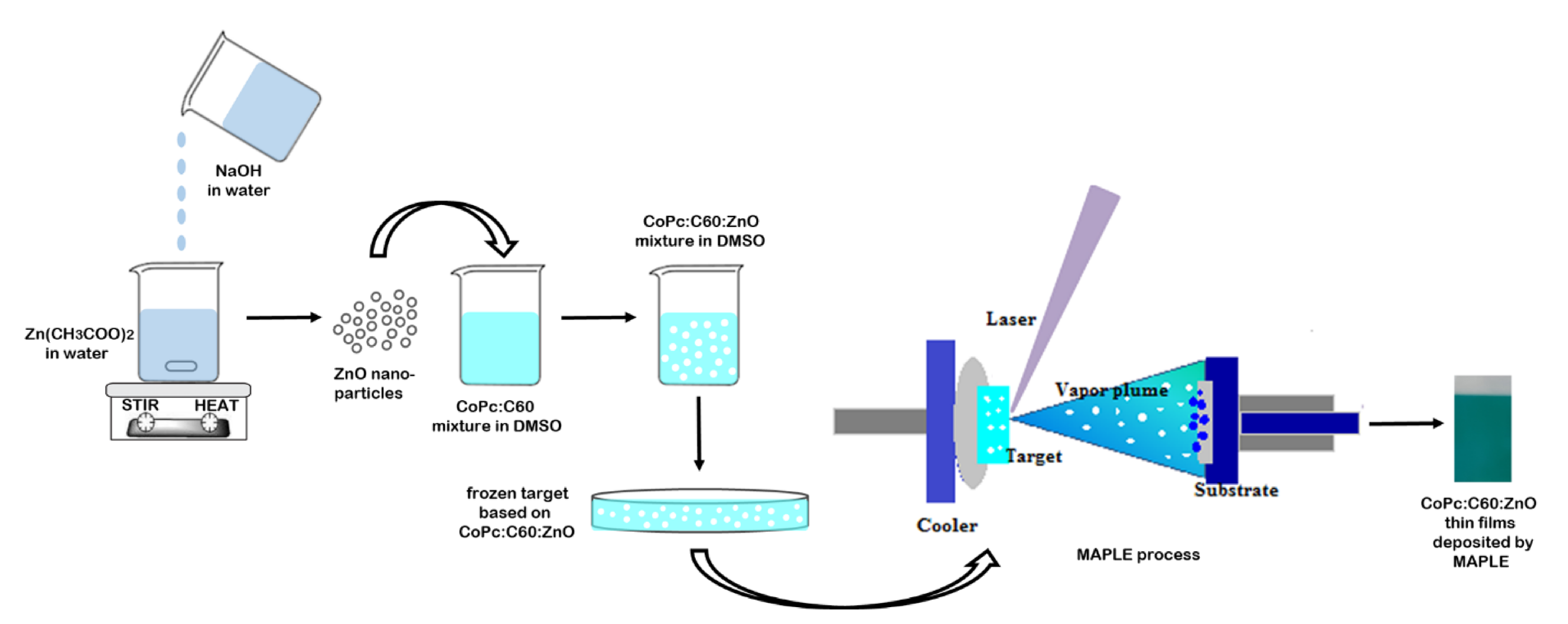
2. Experimental
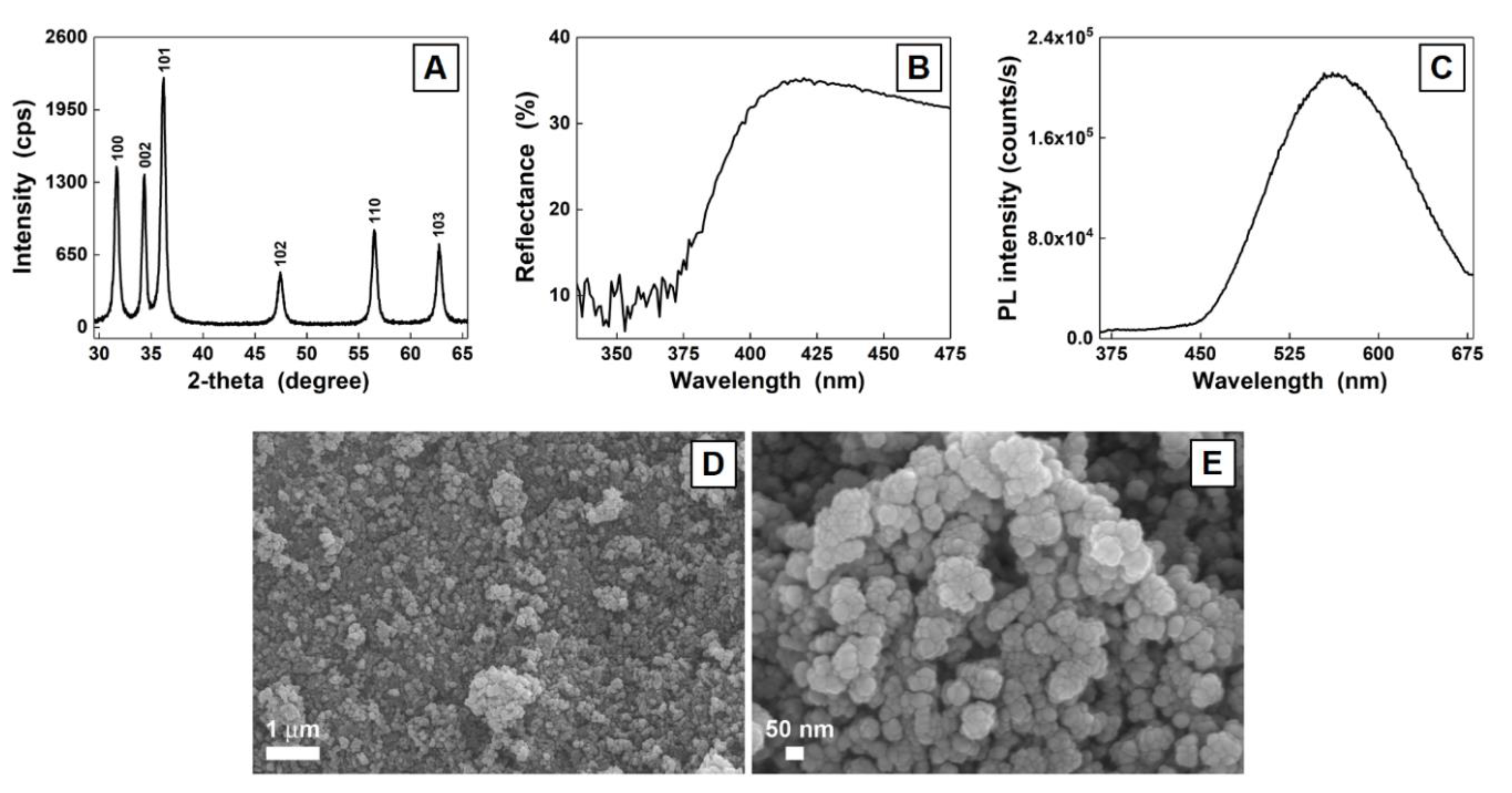
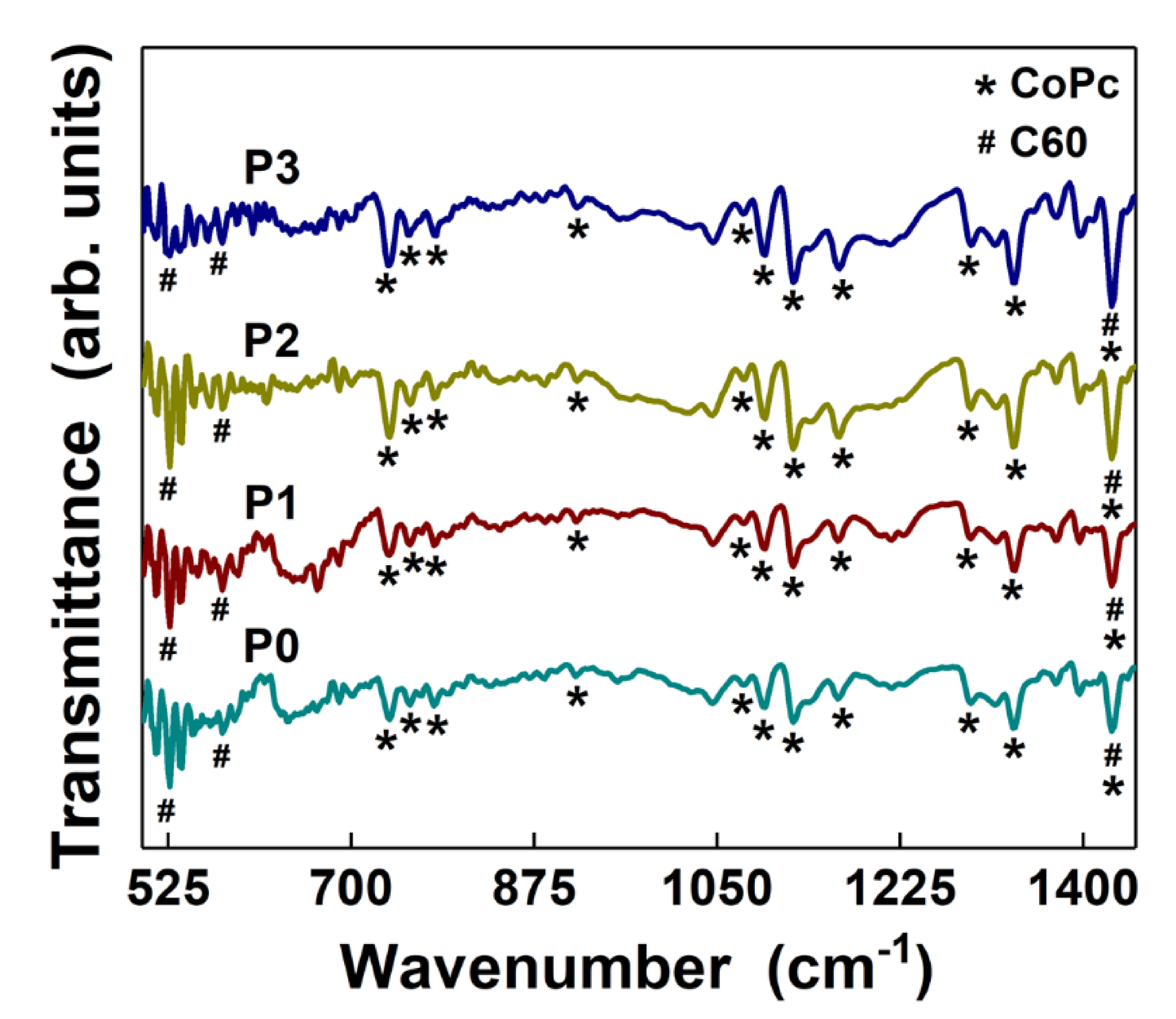
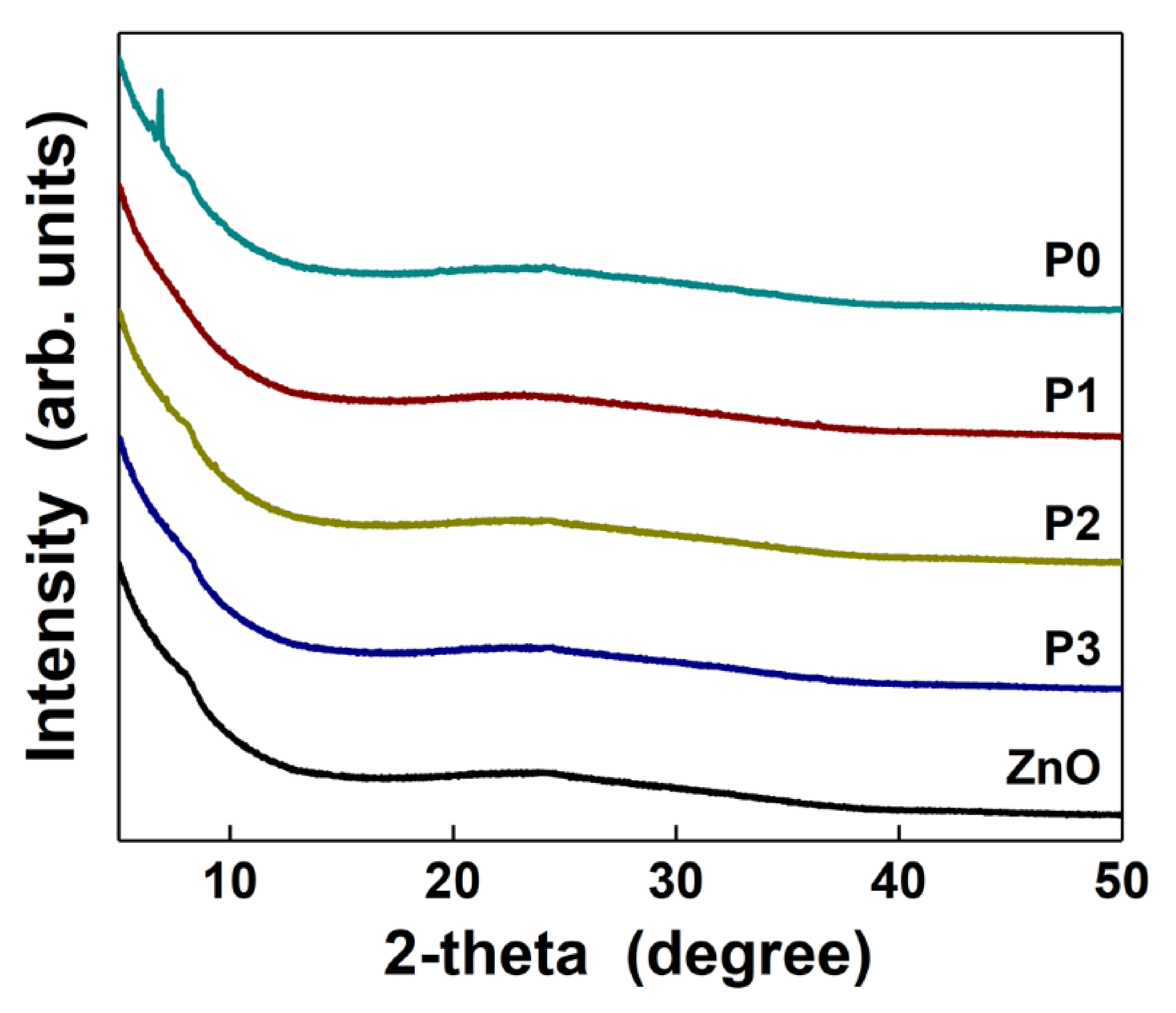
3. Results and Discussion
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Mir, S.H.; Nagahara, L.A.; Thundat, T.; Mokarian-Tabari, P.; Furukawa, H.; Khosla, A. Review-organic-inorganic hybrid functional materials: An integrated platform for applied technologies. J. Electrochem. Soc. 2018, 165, B3137–B3156. [Google Scholar] [CrossRef]
- Saveleva, M.S.; Eftekhari, K.; Abalymov, A.; Douglas, T.E.L.; Volodkin, D.; Parakhonskiy, B.V.; Skirtach, A.G. Hierarchy of hybrid materials-the place of inorganics-in-organics in it, their composition and applications. Front. Chem. 2019, 7, 179. [Google Scholar] [CrossRef]
- Nilofar, A.; Masita, M.; Marzieh, B. Novel nanomaterials for solar cell devices. In Nanomaterials for Green Energy; Bhanvase, B.A., Pawade, V.B., Dhoble, S.J., Sonawane, S.H., Ashokkumar, M., Eds.; Elsevier: Amsterdam, The Netherlands, 2018; Chapter 8; pp. 227–277. [Google Scholar] [CrossRef]
- Chen, F.C. Emerging organic and organic/inorganic hybrid photovoltaic devices for specialty applications: Low-level-lighting energy conversion and biomedical treatment. Adv. Opt. Mater. 2019, 7, 180066. [Google Scholar] [CrossRef]
- Liu, L. Hybrid organic/inorganic nanocomposites for photovoltaic cells. Materials 2017, 7, 2747. [Google Scholar] [CrossRef] [PubMed]
- Meng, L.; Zhang, Y.; Wan, X.; Li, C.; Zhang, X.; Wang, Y.; Ke, X.; Xiao, Z.; Ding, L.; Xia, R.; et al. Organic and solution-processed tandem solar cells with 17.3% efficiency. Science 2018, 361, 1094–1098. [Google Scholar] [CrossRef]
- Li, S.S.; Chen, C.W. Polymer–metal-oxide hybrid solar cells. J. Mater. Chem. A 2013, 1, 10574–10591. [Google Scholar] [CrossRef]
- Sevim, A.M.; Çakar, S.; Özacar, M.; Gül, A. Electrochemical and photovoltaic properties of highly efficient solar cells with cobalt/zinc phthalocyanine sensitizers. Sol. Energy 2018, 160, 18–24. [Google Scholar] [CrossRef]
- de la Torre, G.; Bottari, G.; Torres, T. Phthalocyanines and subphthalocyanines: Perfect partners for fullerenes and carbon nanotubes in molecular photovoltaics. Adv. Energy Mater. 2017, 7, 1601700. [Google Scholar] [CrossRef]
- Xiao, Y.; Zhang, L.; Peng, F.; Pan, G.-B. Fabrication of a cobalt phthalocyanine freestanding film on an ionic liquid surface for memory device applications. RSC Adv. 2018, 8, 5344–5349. [Google Scholar] [CrossRef]
- Florica, C.; Preda, N.; Enculescu, M.; Zgura, I.; Socol, M.; Enculescu, I. Superhydrophobic ZnO networks with high water adhesion. Nanoscale Res. Lett. 2014, 9, 385. [Google Scholar] [CrossRef]
- Preda, N.; Enculescu, M.; Florica, C.; Costas, A.; Evanghelidis, A.; Matei, E.; Enculescu, I. Morphology-controlled synthesis of ZnO structures by a simple wet chemical method. Dig. J. Nanomater. Biostruct. 2013, 8, 1591–1600. [Google Scholar]
- Florica, C.; Costas, A.; Kuncser, A.; Preda, N.; Enculescu, I. High performance FETs based on ZnO nanowires synthesized by low cost methods. Nanotechnology 2016, 27, 475303. [Google Scholar] [CrossRef] [PubMed]
- Haro, M.; Solis, C.; Blas-Ferrando, V.M.; Margeat, O.; Ben Dhkil, S.; Videlot-Ackermann, C.; Ackermann, J.; Di Fonzo, F.; Guerrero, A.; Gimenez, S. Direct hydrogen evolution from saline water reduction at neutral pH using organic photocathodes. ChemSusChem 2016, 9, 3062–3066. [Google Scholar] [CrossRef] [PubMed]
- Víctor-Román, S.; García-Bordejé, E.; Hernández-Ferrer, J.; González-Domínguez, J.M.; Ansón-Casaos, A.; Silva, A.M.T.; Maser, W.K.; Benito, A.M. Controlling the surface chemistry of graphene oxide: Key towards efficient ZnO-GO photocatalysts. Catal. Today 2020. [Google Scholar] [CrossRef]
- Luceño-Sánchez, J.A.; Díez-Pascual, A.M.; Peña Capilla, R. Materials for photovoltaics: State of art and recent developments. Int. J. Mol. Sci. 2019, 20, 976. [Google Scholar] [CrossRef]
- Socol, M.; Preda, N.; Costas, A.; Breazu, C.; Stanculescu, A.; Rasoga, O.; Popescu-Pelin, G.; Mihailescu, A.; Socol, G. Hybrid organic-inorganic thin films based on zinc phthalocyanine and zinc oxide deposited by MAPLE. Appl. Surf. Sci. 2020, 503, 144317. [Google Scholar] [CrossRef]
- Yang, S.; Zhang, J. Matrix-Assisted Pulsed Laser Evaporation (MAPLE) technique for deposition of hybrid nanostructures. Front. Nanosci. Nanotechnol. 2017, 3, 1–9. [Google Scholar] [CrossRef]
- Caricato, A.P.; Cesaria, M.; Gigli, G.; Loiudice, A.; Luches, A.; Martino, M.; Resta, V.; Rizzo, A.; Taurino, A. Poly-(3-hexylthiophene)/[6,6]-phenyl-C61-butyric-acid-methyl-ester bilayer deposition by matrix-assisted pulsed laser evaporation for organic photovoltaic applications. Appl. Phys. Lett. 2012, 100, 073306. [Google Scholar] [CrossRef]
- Socol, M.; Preda, N.; Rasoga, O.; Breazu, C.; Stavarache, I.; Stanculescu, F.; Socol, G.; Gherendi, F.; Grumezescu, V.; Stefan, N.; et al. Flexible heterostructures based on metal phthalocyanines thin films obtained by MAPLE. Appl. Surf. Sci. 2016, 374, 403–410. [Google Scholar] [CrossRef]
- Dunlap-Shohl, W.A.; Barraza, E.T.; Barrette, A.; Gundogdu, K.; Stiff-Roberts, A.D.; Mitzi, D.B. MAPbI3 solar cells with absorber deposited by Resonant Infrared Matrix-Assisted Pulsed Laser Evaporation. ACS Energy Lett. 2018, 3, 270–275. [Google Scholar] [CrossRef]
- Bian, S.W.; Mudunkotuwa, I.A.; Rupasinghe, T.; Grassian, V.H. Aggregation and dissolution of 4 nm ZnO nanoparticles in aqueous environments: Influence of pH, ionic strength, size, and adsorption of humic acid. Langmuir 2011, 27, 6059–6068. [Google Scholar] [CrossRef]
- Stanculescu, A.; Rasoga, O.; Mihut, L.; Socol, M.; Stanculescu, F.; Ionita, I.; Albu, A.-M.; Socol, G. Preparation and characterization of polar aniline functionalized copolymers thin films for optical non-linear applications. Ferroelectrics 2009, 389, 159–173. [Google Scholar] [CrossRef]
- Stanculescu, F.; Stanculescu, A.; Socol, M. Effect of the metallic contact on the electrical properties of organic semiconductor film. J. Optoelectron. Adv. Mater. 2007, 9, 1352–1357. [Google Scholar]
- Djurisic, A.B.; Leung, Y.H. Optical properties of ZnO nanostructures. Small 2006, 2, 944–961. [Google Scholar] [CrossRef] [PubMed]
- Reshchikov, M.A.; Morkoc, H.; Nemeth, B.; Nause, J.; Xie, J.; Hertog, B.; Osinsky, A. Luminescence properties of defects in ZnO. Phys. B 2007, 401, 358–361. [Google Scholar] [CrossRef]
- Tam, K.H.; Cheung, C.K.; Leung, Y.H.; Djurisic, A.B.; Ling, C.C.; Beling, C.D.; Fung, S.; Kwok, W.M.; Chan, W.K.; Phillips, D.L.; et al. Defects in ZnO nanorods prepared by a hydrothermal method. J. Phys. Chem. B 2006, 110, 20865–20871. [Google Scholar] [CrossRef] [PubMed]
- Ji, X.; Zou, T.; Gong, H.; Wu, Q.; Qiao, Z.; Wu, W.; Wang, H. Cobalt phthalocyanine nanowires: Growth, crystal structure, and optical properties. Cryst. Res. Technol. 2016, 51, 154–159. [Google Scholar] [CrossRef]
- Pu, J.; Mo, Y.; Wan, S.; Wang, L. Fabrication of novel graphene–fullerene hybrid lubricating films based on self-assembly for MEMS applications. Chem. Commun. 2014, 50, 469–471. [Google Scholar] [CrossRef]
- Soliman, H.S.; El-Barry, A.M.A.; Khosifan, N.M.; El Nahass, M.M. Structural and electrical properties of thermally evaporated cobalt phthalocyanine (CoPc) thin films. Eur. Phys. J. Appl. Phys. 2007, 37, 1–9. [Google Scholar] [CrossRef]
- Tahir, D.; Jae, K.H. Effect of growth temperature on structural and electronic properties of ZnO thin films. AIP Conf. Proc. 2017, 1801, 020007. [Google Scholar] [CrossRef]
- Ikram, M.; Imran, M.; Nunzi, J.M.; Bobbara, S.R.; Islah-u-din, S.A. Efficient and low cost inverted hybrid bulk heterojunction solar cells. J. Renew. Sustain. Energy 2015, 7, 4929603. [Google Scholar] [CrossRef]
- Djurisic, A.B.; Kwong, C.Y.; Lau, T.W.; Liu, Z.T.; Kwok, H.S.; Lam, L.S.M.; Chan, W.K. Spectroscopic ellipsometry of metal phthalocyanine thin films. Appl. Opt. 2003, 42, 6382–6387. [Google Scholar] [CrossRef] [PubMed]
- El-Nahass, M.M.; Abd-El-Rahman, K.F.; Darwish, A.A.A. Fourier-transform infrared and UV-vis spectroscopes of nickel phthalocyanine thin films. Mater. Chem. Phys. 2005, 92, 185–189. [Google Scholar] [CrossRef]
- Karan, S.; Mallik, B. Nanoflowers Grown from phthalocyanine seeds: organic nanorectifiers. J. Phys. Chem. C 2008, 112, 2436–2447. [Google Scholar] [CrossRef]
- Pfuetzner, S.; Meiss, J.; Petrich, A.; Riede, M.; Leo, K. Improved bulk heterojunction organic solar cells employing C70 fullerenes. Appl. Phys. Lett. 2009, 94, 145. [Google Scholar] [CrossRef]
- Aguilar, G.V.; Fonseca, M.R.J.; Ramírez, Á.M.; Gracia, A.G.J. Photoluminescence studies on ZnO thin films obtained by sol-gel method. In Recent Applications in Sol-Gel Synthesis; Chandra, U., Ed.; IntechOpen: Rijeka, Croatia, 2017. [Google Scholar] [CrossRef]
- Djurišić, A.B.; Ching Ng, A.M.; Chen, X. ZnO Nanostructures for optoelectronics: Material properties and device applications. Prog. Quant. Electron. 2010, 34, 191–259. [Google Scholar] [CrossRef]
- Bała, W.; Wojdyła, M.; Rębarz, M.; Szybowic, M.; Drozdowski, M.; Grodzicki, A.; Piszczek, P. Influence of central metal atom in MPc (M = Cu, Zn, Mg, Co) on Raman, FT-IR, absorbance, reflectance, and photoluminescence spectra. J. Optoelectron. Adv. Mater. 2009, 11, 264–269. [Google Scholar]
- Koeppe, R.; Sariciftci, N.S. Photoinduced charge and energy transfer involving fullerene derivatives. Photochem. Photobiol. Sci. 2006, 5, 1122–1131. [Google Scholar] [CrossRef]
- Kegel, J.; Laffir, F.; Povey, I.M.; Pemble, M.E. Defect-promoted photo-electrochemical performance enhancement of orange-luminescent ZnO nanorod-arrays. Phys. Chem. Chem. Phys. 2017, 19, 12255–12268. [Google Scholar] [CrossRef]
- Huang, J.; Yinb, Z.; Zheng, Q. Applications of ZnO in organic and hybrid solar cells. Energy Environ. Sci. 2011, 4, 3861–3877. [Google Scholar] [CrossRef]
- Ompong, D.; Singh, J. Charge carrier mobility dependent open-circuit voltage in organic and hybrid solar cells. Front. Nanosci. Nanotechnol. 2016, 21, 43–47. [Google Scholar] [CrossRef]
- Roest, A.L.; Kelly, J.J.; Vanmaekelbergh, D.; Meulenkamp, E.A. Staircase in the electron mobility of a ZnO quantum dot assembly due to shell filling. Phys. Rev. Lett. 2002, 89, 036801. [Google Scholar] [CrossRef] [PubMed]
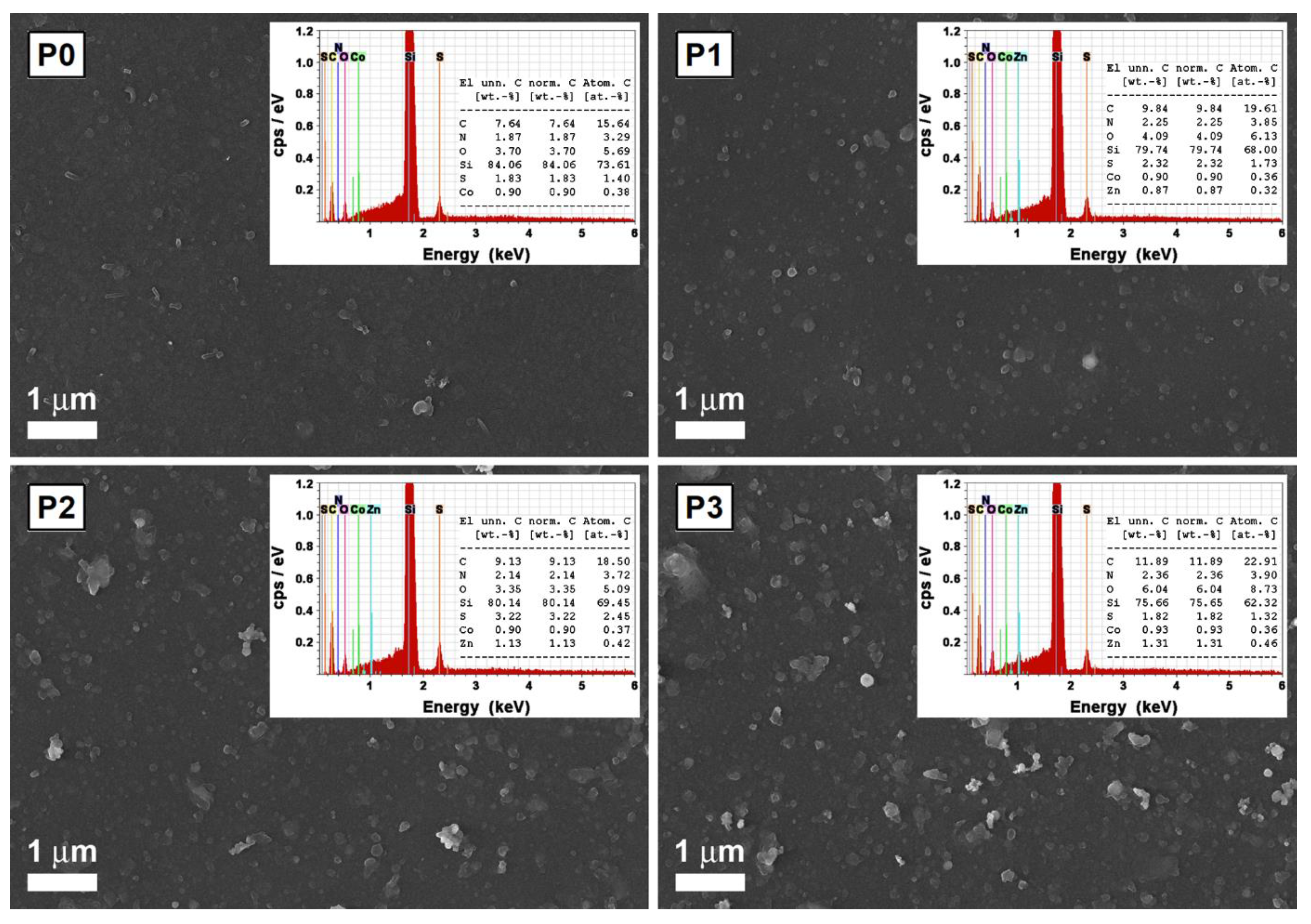

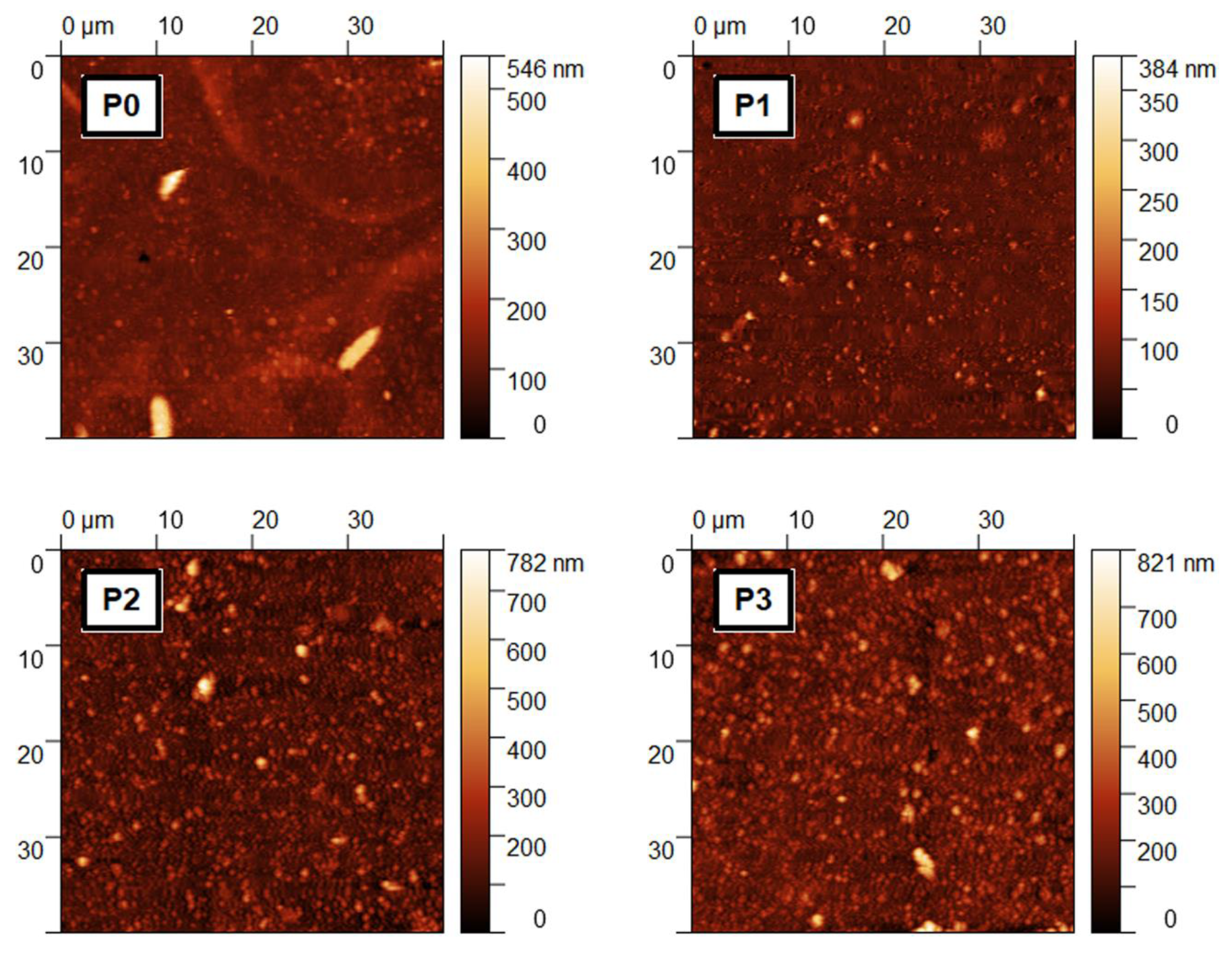
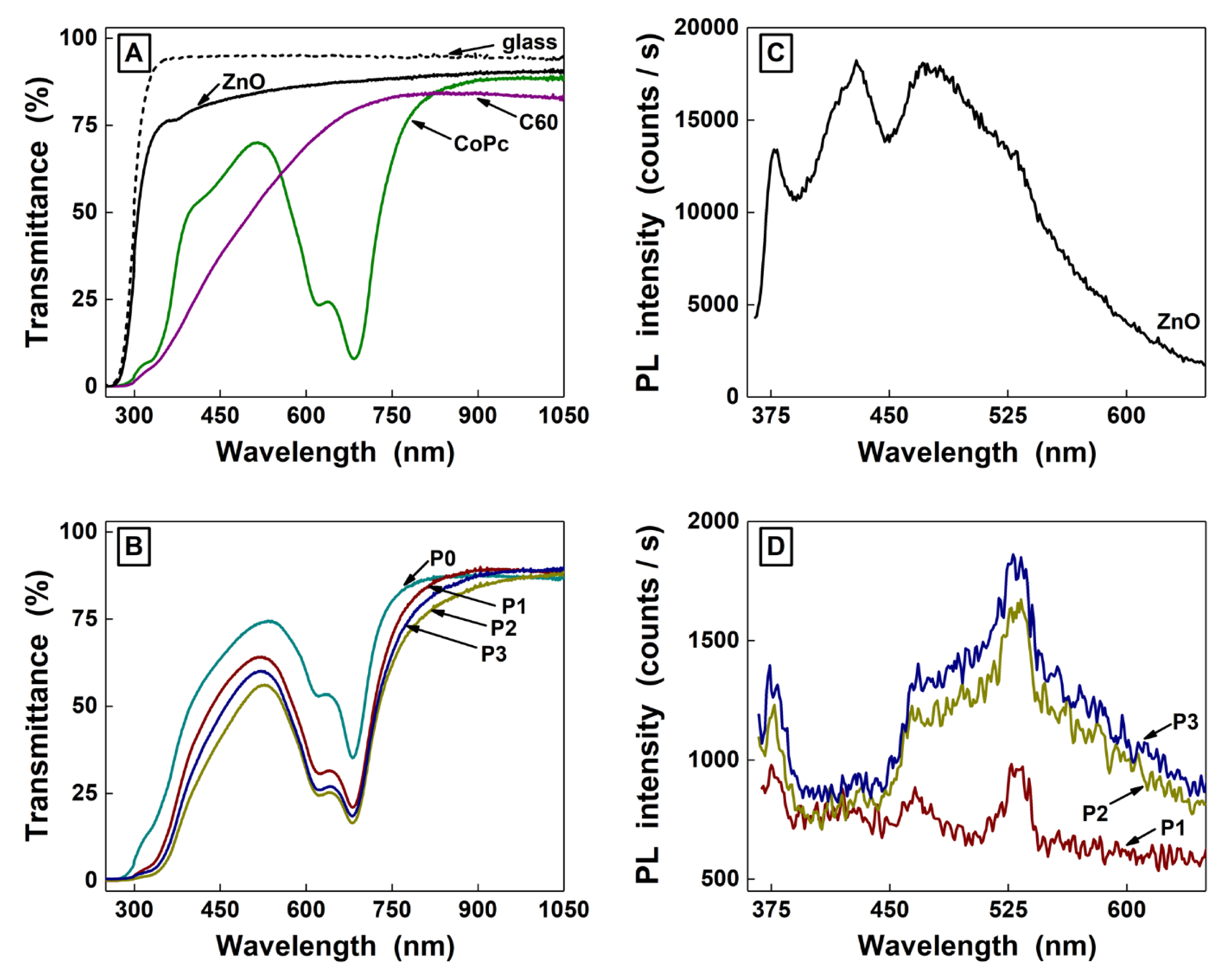
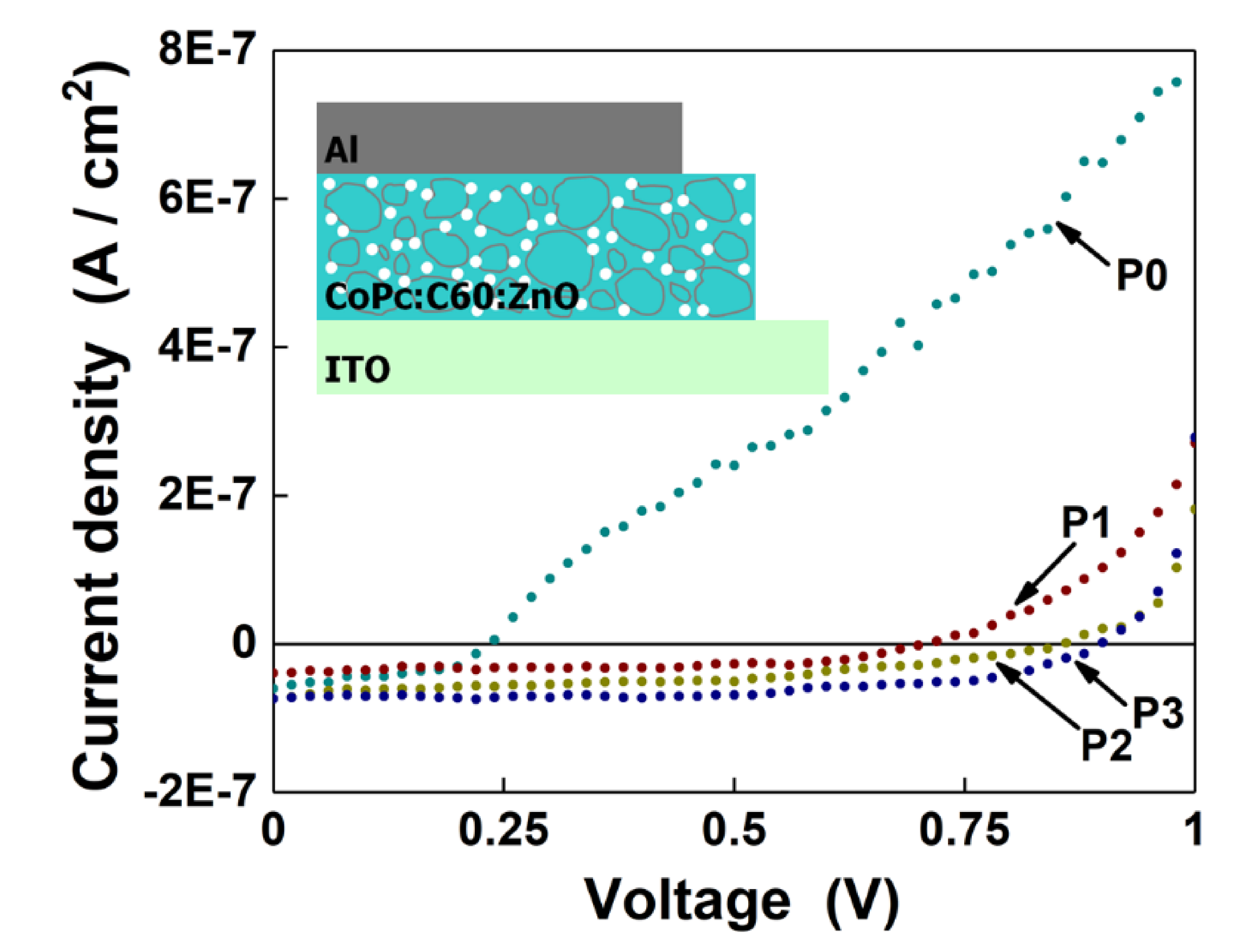
| Sample | Voc (V) | Jsc (A/cm2) | Pmax (W) |
|---|---|---|---|
| P0 | 0.23 | 5.9 × 10−8 | 5.8 × 10−9 |
| P1 | 0.71 | 4 × 10−8 | 1.5 × 10−8 |
| P2 | 0.86 | 7.2 × 10−8 | 2.5 × 10−8 |
| P3 | 0.9 | 7.5 × 10−8 | 3.6 × 10−8 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Socol, M.; Preda, N.; Costas, A.; Borca, B.; Popescu-Pelin, G.; Mihailescu, A.; Socol, G.; Stanculescu, A. Thin Films Based on Cobalt Phthalocyanine:C60 Fullerene:ZnO Hybrid Nanocomposite Obtained by Laser Evaporation. Nanomaterials 2020, 10, 468. https://doi.org/10.3390/nano10030468
Socol M, Preda N, Costas A, Borca B, Popescu-Pelin G, Mihailescu A, Socol G, Stanculescu A. Thin Films Based on Cobalt Phthalocyanine:C60 Fullerene:ZnO Hybrid Nanocomposite Obtained by Laser Evaporation. Nanomaterials. 2020; 10(3):468. https://doi.org/10.3390/nano10030468
Chicago/Turabian StyleSocol, Marcela, Nicoleta Preda, Andreea Costas, Bogdana Borca, Gianina Popescu-Pelin, Andreea Mihailescu, Gabriel Socol, and Anca Stanculescu. 2020. "Thin Films Based on Cobalt Phthalocyanine:C60 Fullerene:ZnO Hybrid Nanocomposite Obtained by Laser Evaporation" Nanomaterials 10, no. 3: 468. https://doi.org/10.3390/nano10030468
APA StyleSocol, M., Preda, N., Costas, A., Borca, B., Popescu-Pelin, G., Mihailescu, A., Socol, G., & Stanculescu, A. (2020). Thin Films Based on Cobalt Phthalocyanine:C60 Fullerene:ZnO Hybrid Nanocomposite Obtained by Laser Evaporation. Nanomaterials, 10(3), 468. https://doi.org/10.3390/nano10030468








