The Structure and Chemical Composition of the Cr and Fe Pyrolytic Coatings on the MWCNTs’ Surface According to NEXAFS and XPS Spectroscopy
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Experimental Details
2.2.1. Synthesis of Multi-Walled Carbon Nanotubes (MWCNTs)
2.2.2. Synthesis of Cr/MWCNTs Nanocomposites
2.2.3. Synthesis of Fe/MWCNTs Nanocomposites
2.3. Characterization
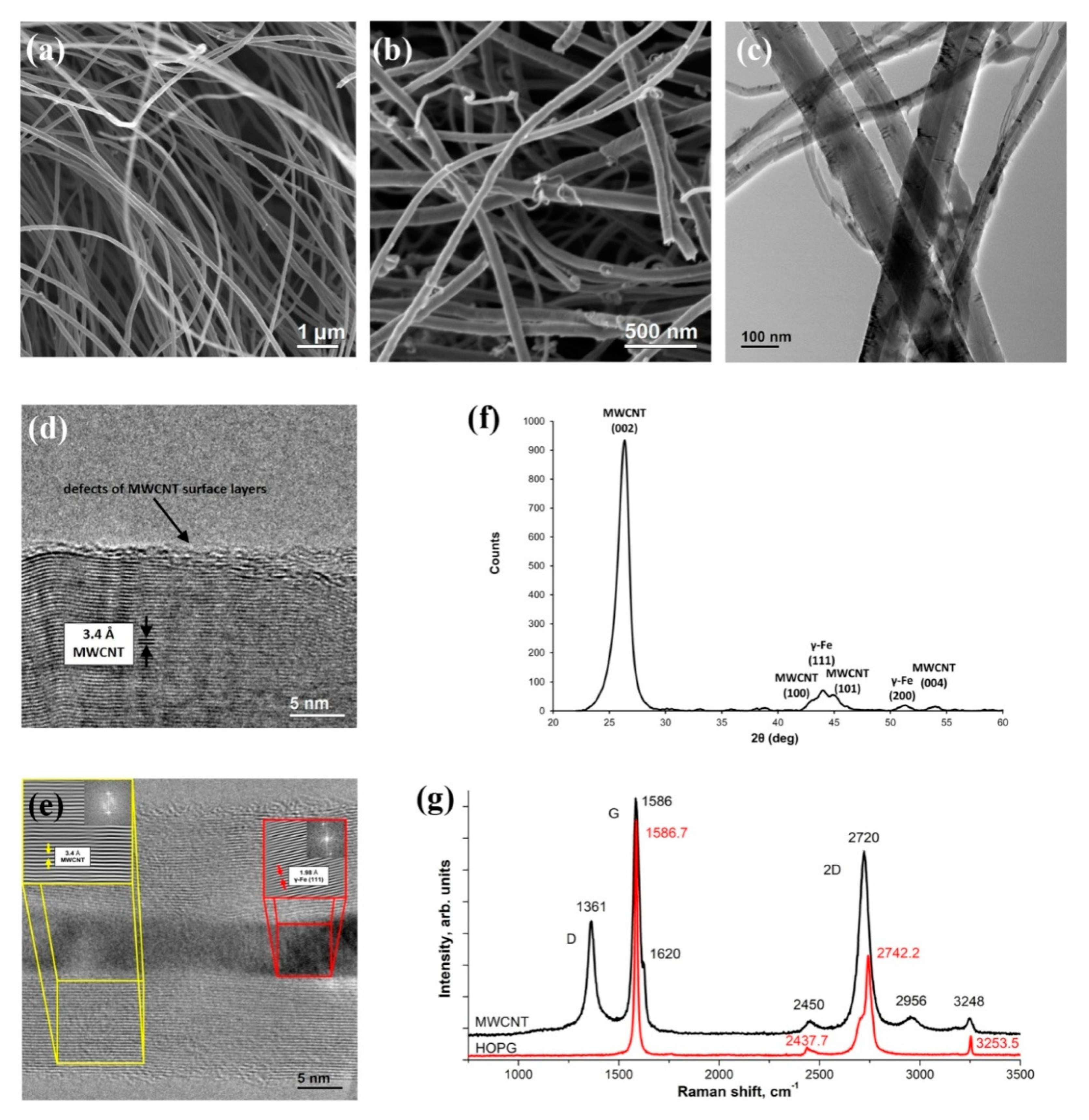
2.3.1. Raman Spectroscopy
2.3.2. X-ray Absorption Spectroscopy
2.3.3. X-ray Photoelectron Spectroscopy (XPS)
3. Results and Discussion
3.1. Initial MWCNTs Research
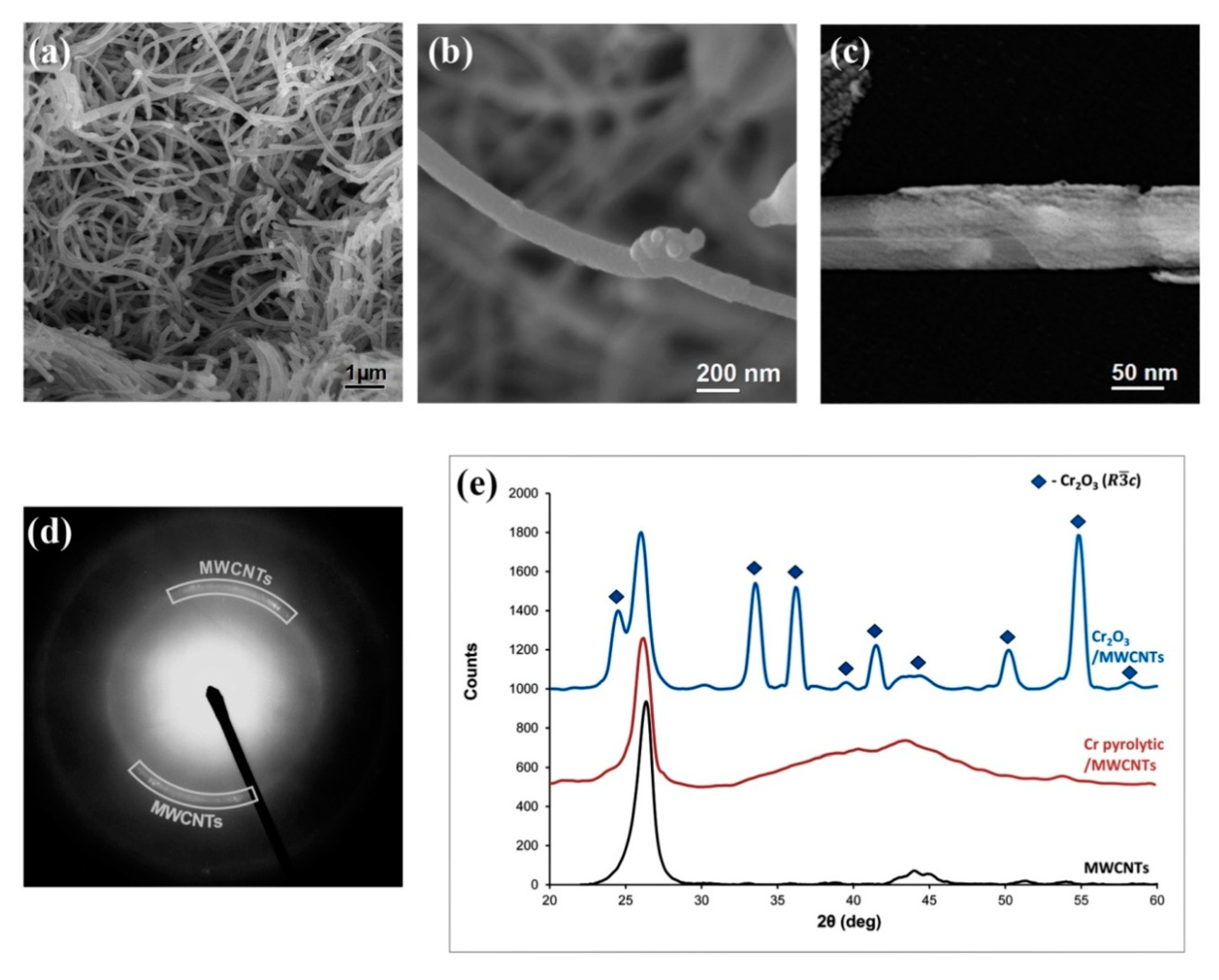
3.2. Cr/MWCNTs Nanocomposite Research
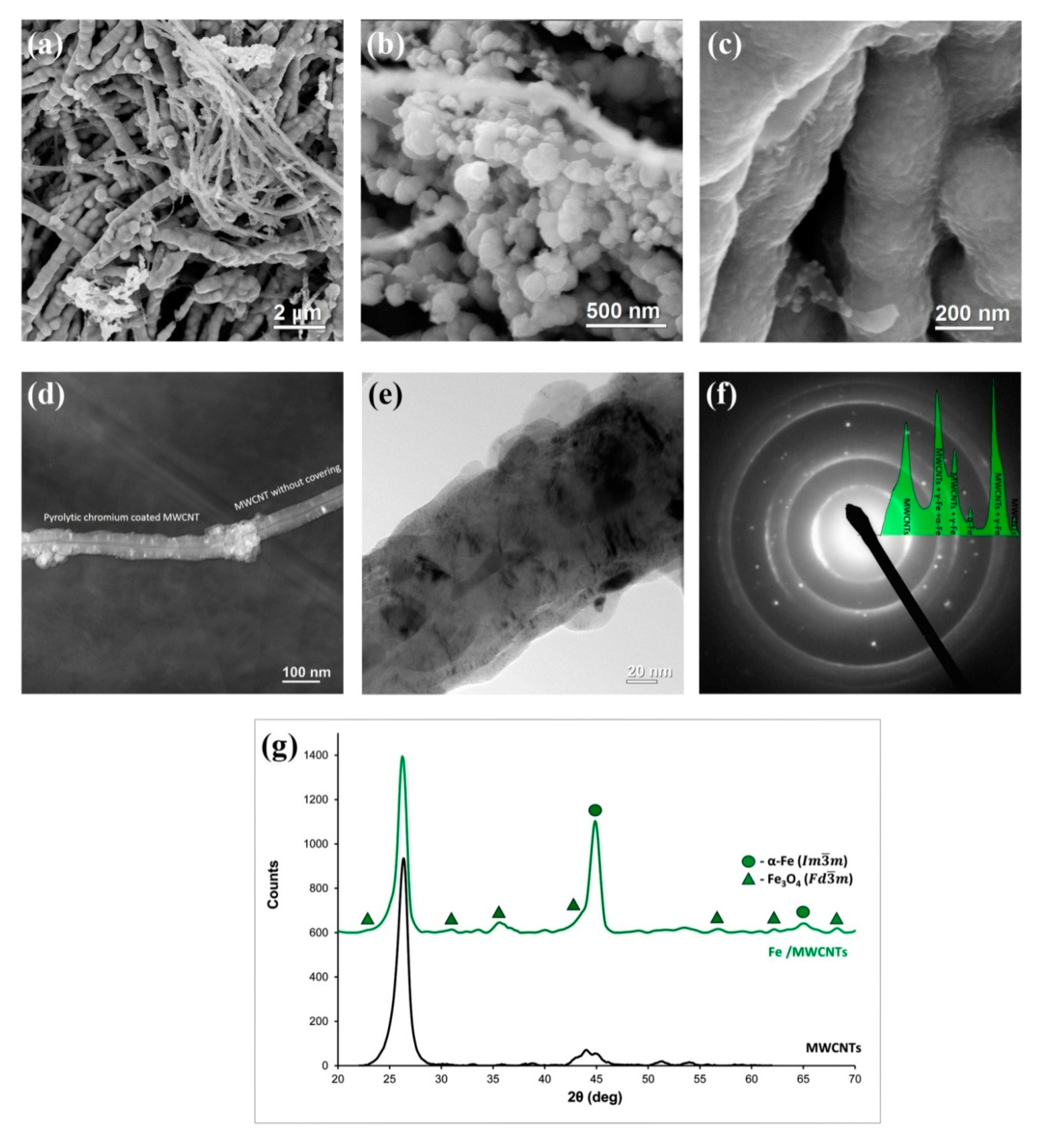
3.3. Fe/MWCNTs Nanocomposite Research
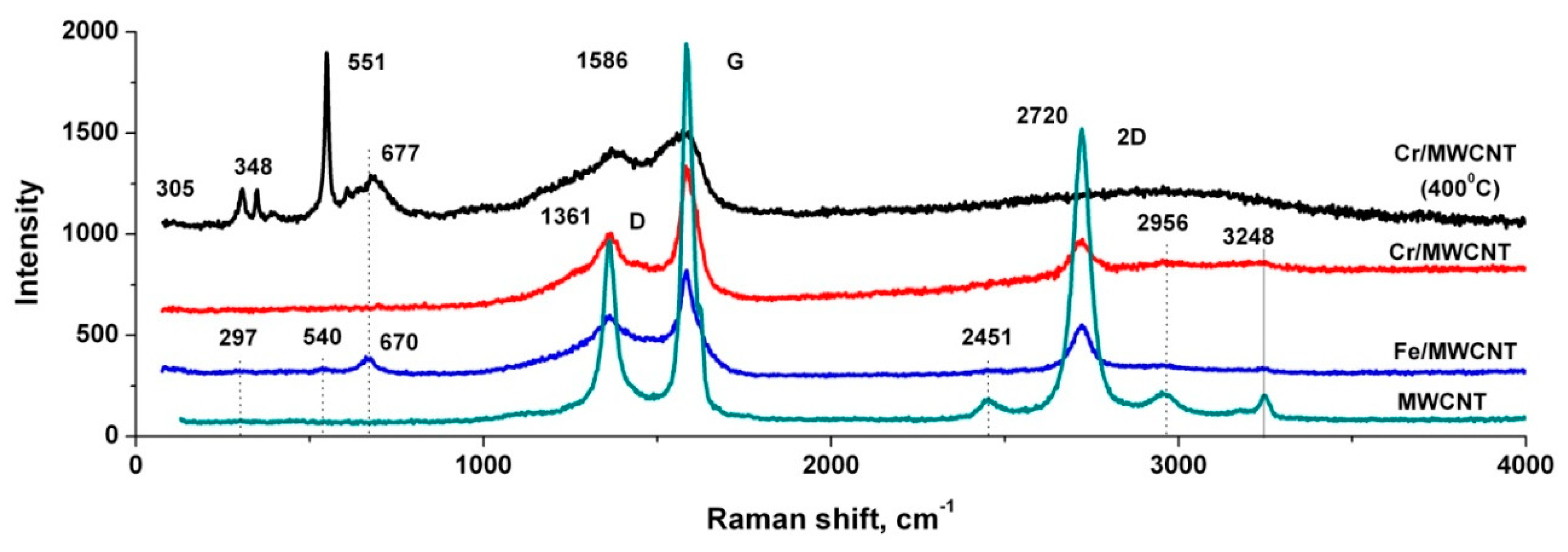
3.4. Raman Spectra of the Nanocomposites and Initial MWCNTs
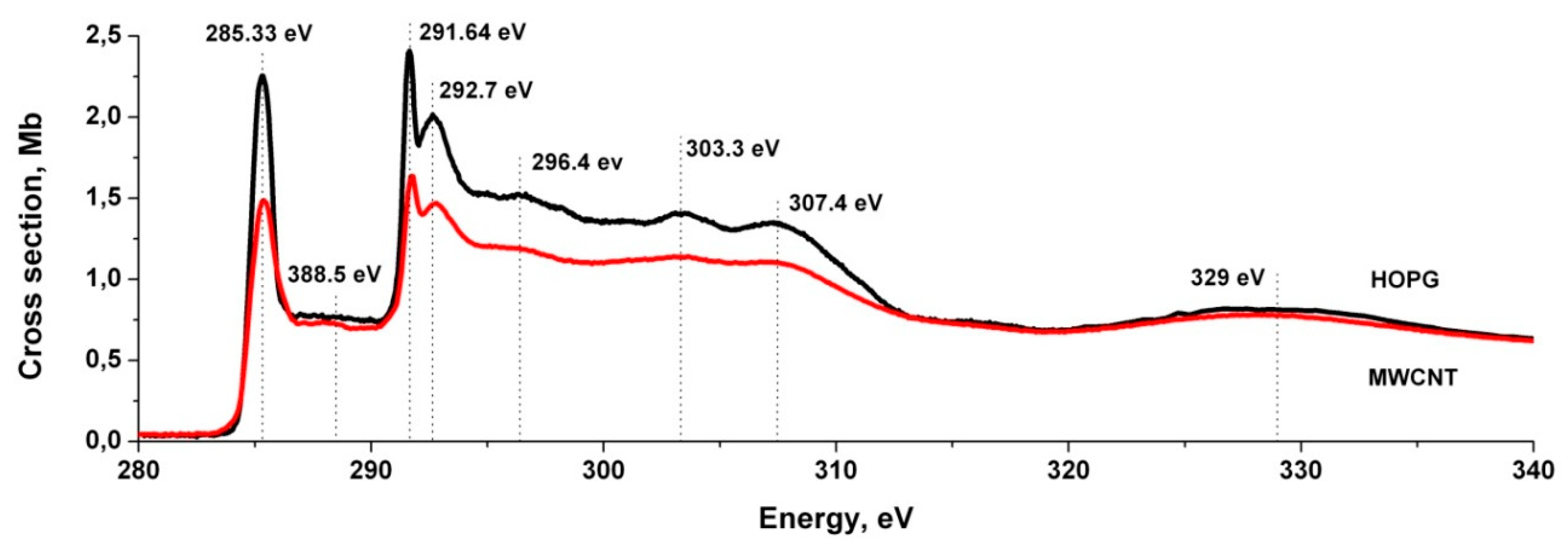
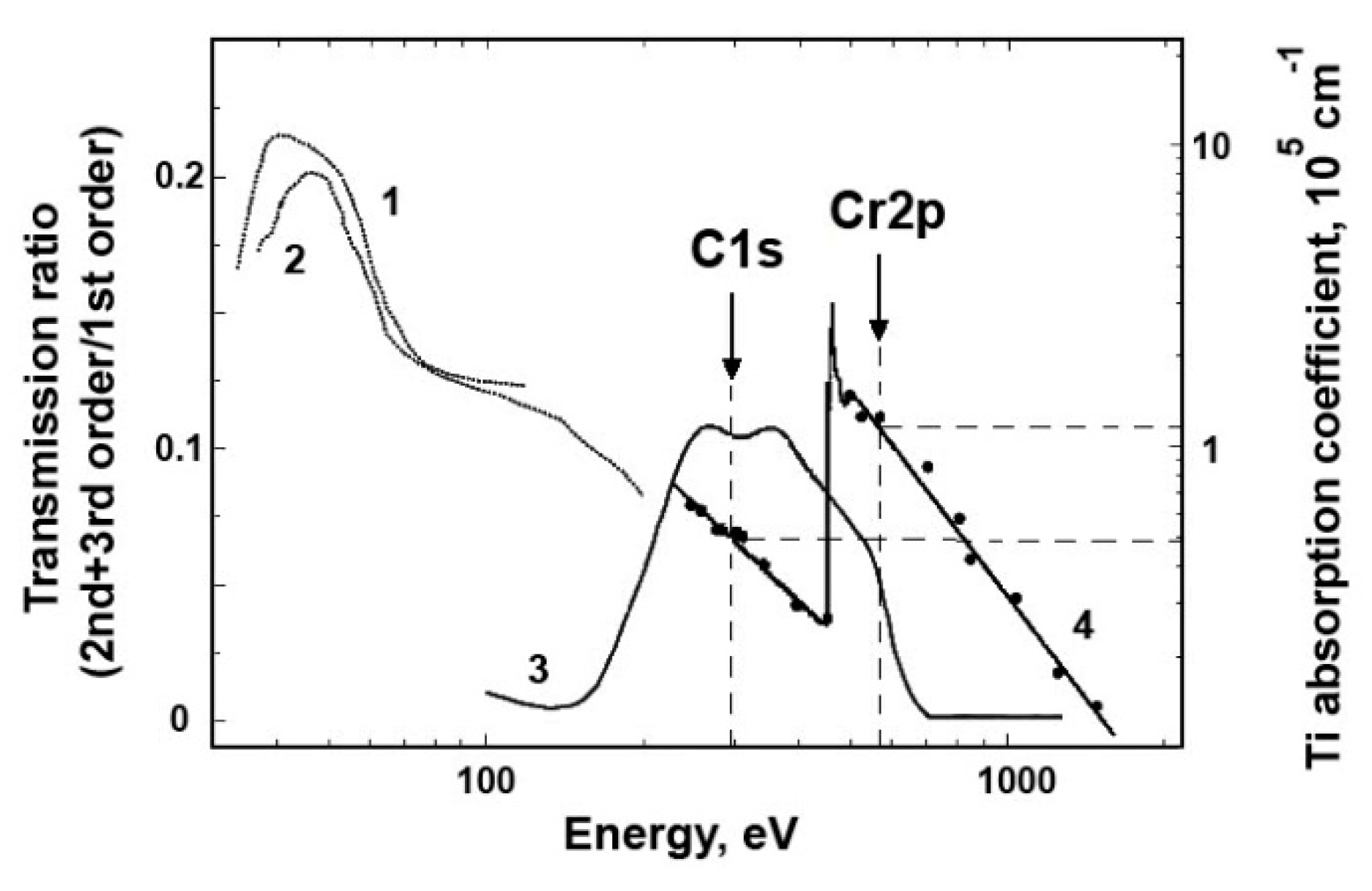
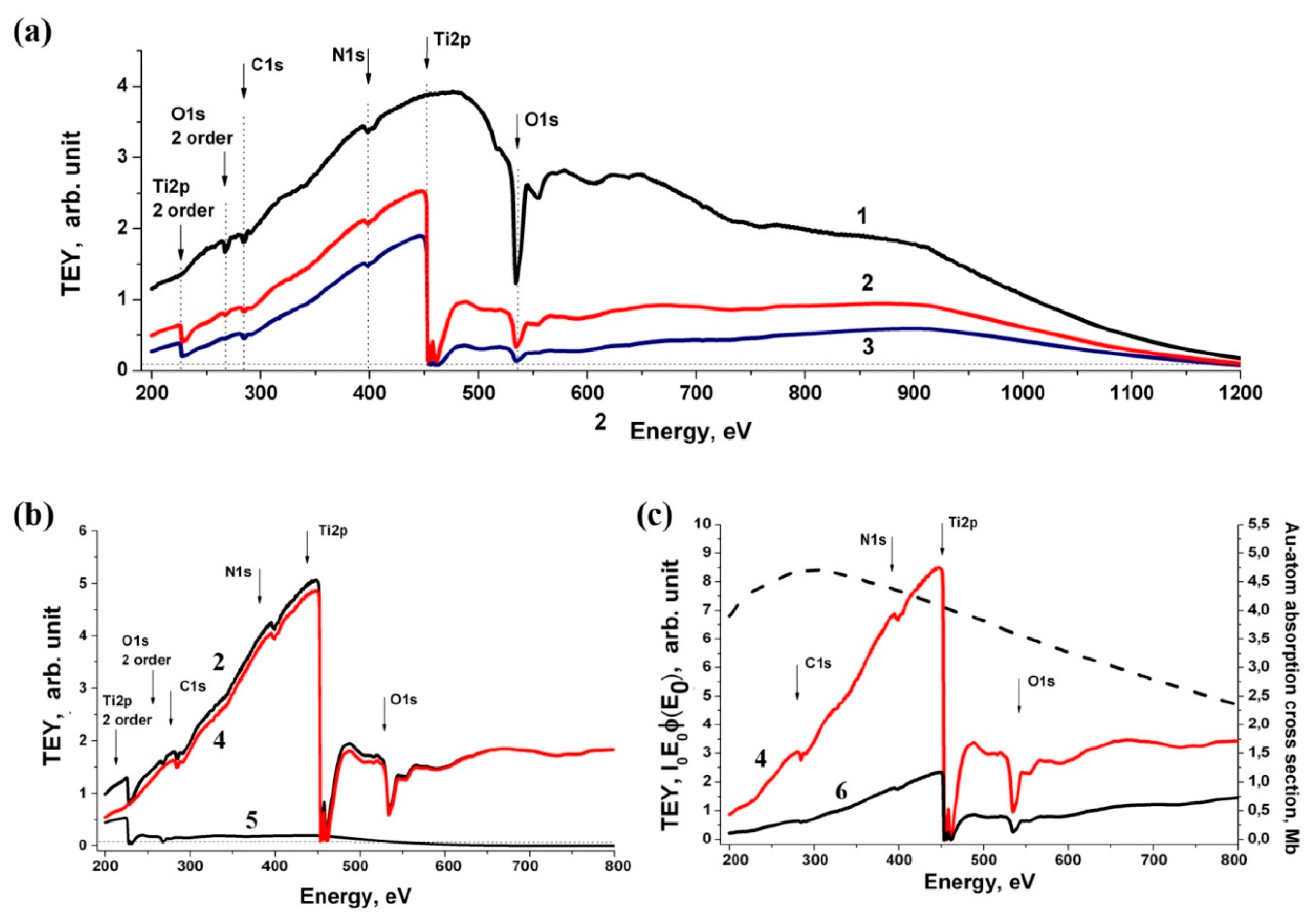
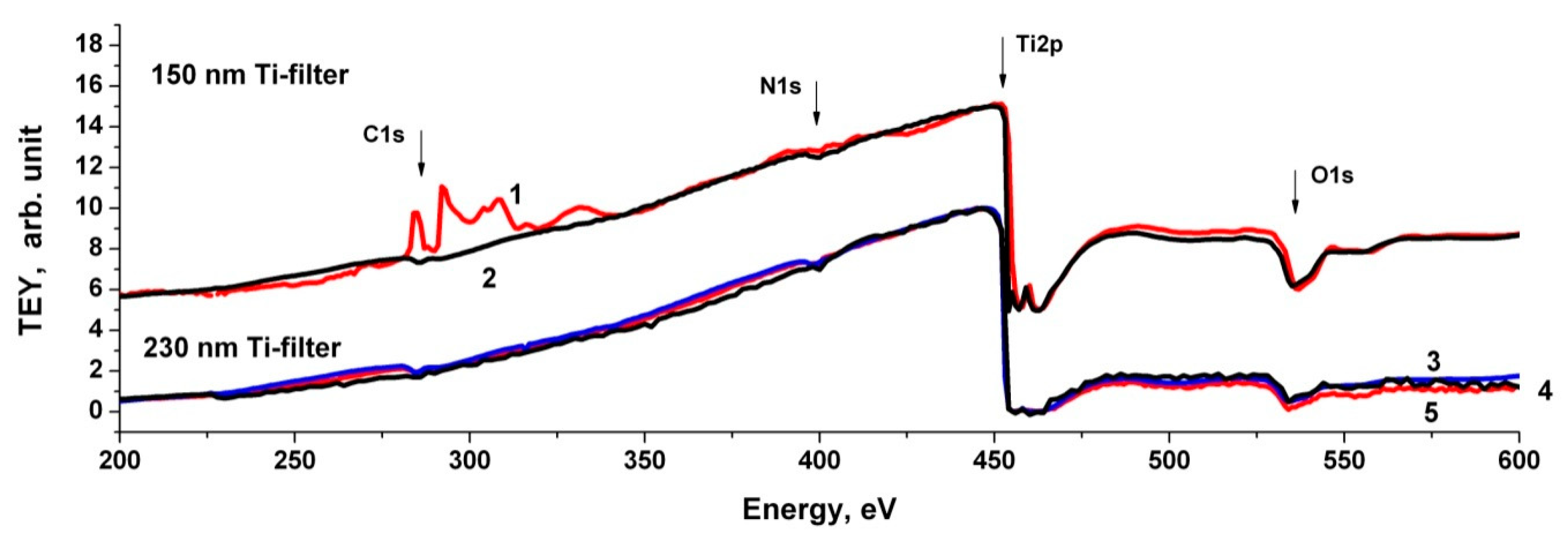
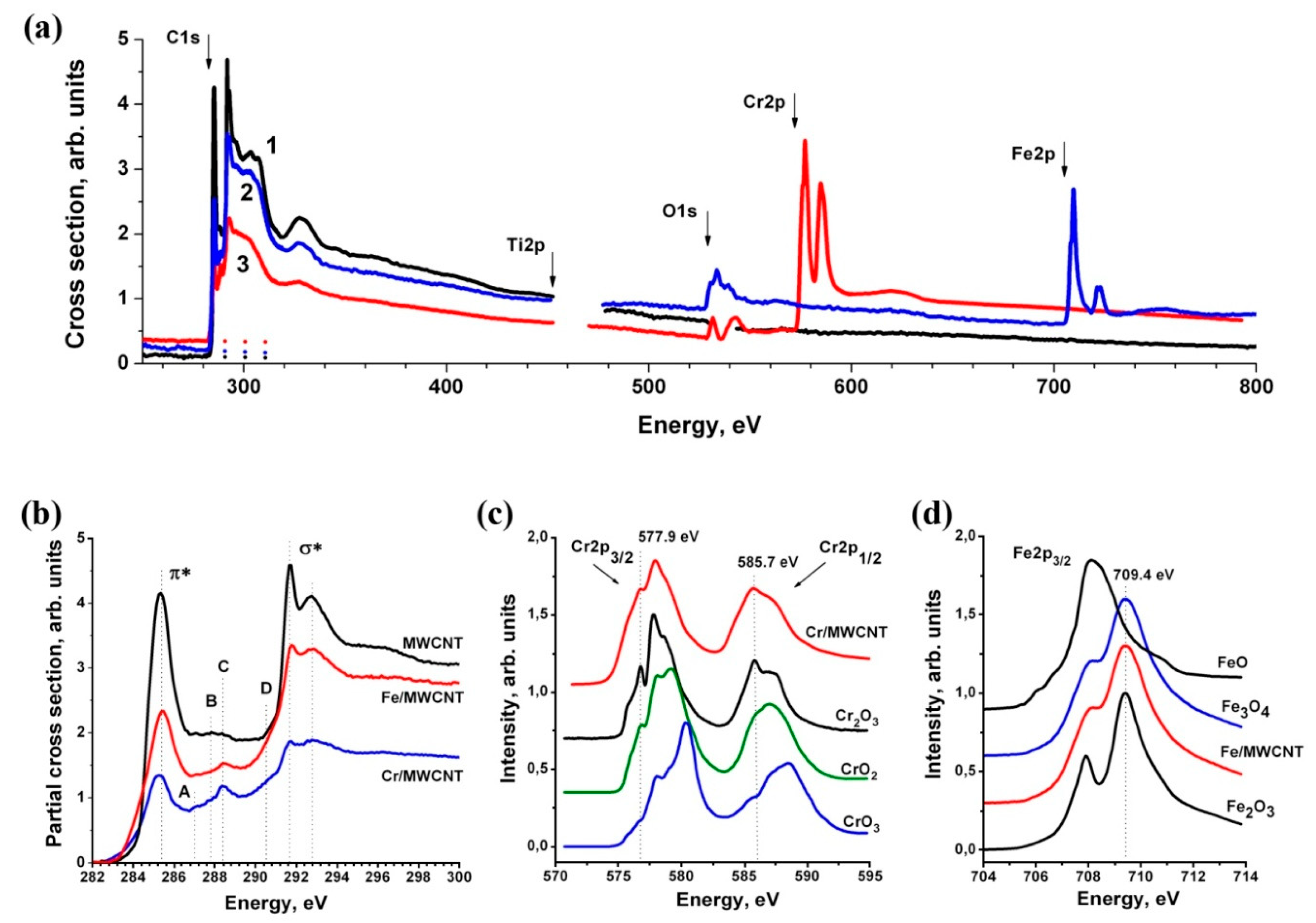
3.5. Near-Edge X-ray Absorption Fine Structure (NEXAFS) Spectroscopy Research
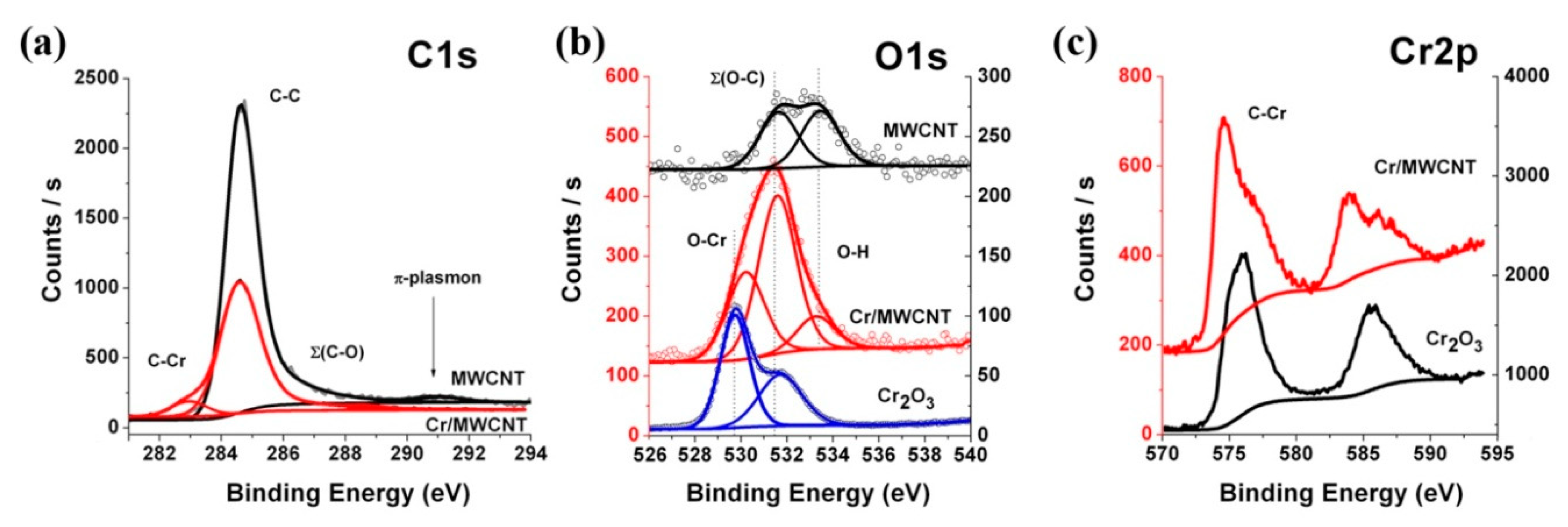
3.6. XPS Spectroscopy Research
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Rao, C.N.R.; Cheetham, A.K.J. Science and technology of nanomaterials: Current status and future prospects. J. Mater. Chem. 2001, 11, 2887–2894. [Google Scholar] [CrossRef]
- Daoush, W.M.; Lim, B.K.; Mo, C.B.; Nam, D.H.; Hong, S.H. Electrical and mechanical properties of carbon nanotube reinforced coppernanocomposites fabricated by electroless deposition process. Mater. Sci. Eng. A 2009, 513–514, 247–253. [Google Scholar] [CrossRef]
- Aborkin, A.; Khorkov, K.; Prusov, E.; Kremlev, K.; Perezhogin, I.; Alymov, M. Effect of Increasing the Strength of Aluminum Matrix Nanocomposites Reinforced with Microadditions of Multiwalled Carbon Nanotubes Coated with TiC Nanoparticles. Nanomaterials 2019, 9, 1596. [Google Scholar] [CrossRef] [PubMed]
- Eder, D. Carbon Nanotube-Inorganic Hybrids. Chem. Rev. 2010, 110, 1348–1385. [Google Scholar] [CrossRef] [PubMed]
- Ebbesen, T. Wetting, filling and decorating carbon nanotubes. J. Phys. Chem. Solids 1996, 57, 951–955. [Google Scholar] [CrossRef]
- Rakov, E.G. Chemistry of Carbon Nanotubes. In Nanomaterials Handbook; Golotsi, Y., Ed.; CRC Press: Boca Raton, FL, USA, 2006; Volume 56, pp. 105–175. [Google Scholar]
- Mu, S.C.; Tang, H.L.; Qian, S.H. Hydrogen storage in carbon nanotubes modified by microwaveplasma etching and Pd decoration. Carbon 2006, 44, 762–767. [Google Scholar] [CrossRef]
- Gao, C.; Li, W.; Maekawa, T. Magnetic Carbon Nanotubes: Synthesis by Electrostatic Self-Assembly Approach and Application in Biomanipulations. J. Phys. Chem. B 2006, 110, 7213–7220. [Google Scholar] [CrossRef] [PubMed]
- Zhao, X.; Jiang, P.; Chu, W.; Mu, S.; Liu, D.; Song, L.; Liu, L.; Luo, S.; Zhang, Z.; Xiang, Y.; et al. The growth of carbon nanostructures in the channels of aligned carbon nanotube. Carbon 2006, 44, 1298–1352. [Google Scholar] [CrossRef]
- Tersoff, J. A barrier falls. Nature 2003, 424, 622–623. [Google Scholar] [CrossRef]
- Kadomtseva, A.V.; Vorotyntsev, A.V.; Vorotyntsev, V.M.; Petukhov, A.N.; Ob’edkov, A.M.; Kremlev, K.V.; Kaverin, B.S. Effect of the Catalytic System Based on Multi-Walled Carbon Nanotubes Modified with Copper Nanoparticles on the Kinetics of Catalytic Reduction of Germanium Tetrachloride by Hydrogen. Russ. J. Appl. Chem. 2015, 88, 595–602. [Google Scholar] [CrossRef]
- Liu, Q.; Ren, W.; Chen, Z.-G.; Liu, B.; Yu, B.; Li, F.; Cong, H.; Cheng, H.-M. Direct synthesis of carbon nanotubes decorated with size-controllable Fe nanoparticles encapsulated by graphitic layers. Carbon 2008, 46, 1417–1423. [Google Scholar] [CrossRef]
- Ansari, S.; Khorshidi, A.; Shariati, S. Chemoselective reduction of nitro and nitrilecompounds using an Fe3O4-MWCNTs@PEI-Ag nanocomposite as a reusable catalyst. RSC Adv. 2020, 10, 3554–3565. [Google Scholar] [CrossRef]
- Islam, M.R.; Ferdous, M.; Sujan, M.I.; Mao, X.; Zeng, H.; Azam, M.S. Recyclable Ag-decorated highly carbonaceous magnetic nanocomposites for the removal of organic pollutants. J. Colloid Interface Sci. 2020, 562, 52–62. [Google Scholar] [CrossRef]
- Merryfield, R.; McDaniel, M.; Parks, G. An XPS Study of the Phillips Cr/Silica Polymerization Catalyst. J. Catal. 1982, 77, 348–359. [Google Scholar] [CrossRef]
- Sanfilippo, D.; Miracca, I. Dehydrogenation of paraffins: Synergies between catalystdesign and reactor engineering. Catal. Today 2006, 111, 133–139. [Google Scholar] [CrossRef]
- Kim, D.S.; Wachs, I.E. Surface Chemistry of Supported Chromium Oxide Catalysts. J. Catal. 1993, 142, 166–171. [Google Scholar] [CrossRef]
- An, G.; Zhang, Y.; Liu, Z.; Miao, Z.; Han, B.; Miao, S.; Li, J. Preparation of porous chromium oxide nanotubes using carbon nanotubes as templates and their application as an ethanol sensor. Nanotechnology 2008, 19, 035504. [Google Scholar] [CrossRef]
- Kremlev, K.V.; Ob’edkov, A.M.; Ketkov, S.Y.; Kaverin, B.S.; Semenov, N.M.; Gusev, S.A.; Andreev, P.V. Deposition of Nanocrystalline Nonstoichiometric Chromium Oxide Coatings on the Surface of Multiwalled Carbon Nanotubes by Chromium Acetylacetonate Vapor Pyrolysis. Tech. Phys. Lett. 2017, 43, 71–78. [Google Scholar] [CrossRef]
- Sivkov, V.N.; Ob’edkov, A.M.; Petrova, O.V.; Nekipelov, S.V.; Kremlev, K.V.; Kaverin, B.S.; Semenov, N.M.; Gusev, S.A. X-Ray and Synchrotron Investigations of Heterogeneous Systems Based on Multiwalled Carbon Nanotubes. Phys. Solid State 2015, 57, 197–204. [Google Scholar] [CrossRef]
- Sivkov, V.N.; Petrova, O.V.; Nekipelov, S.V.; Obiedkov, A.M.; Kaverin, B.S.; Kirillov, A.I.; Semenov, N.M.; Domrachev, G.A.; Egorov, V.A.; Gusev, S.A.; et al. NEXAFS Study of the Composite Materials MWCNTs-Pyrolytic Metals by Synchrotron Radiation. Fuller. Nanotub. Carbon. Nanostruct. 2014, 23, 1536–4046. [Google Scholar] [CrossRef]
- Sivkov, V.N.; Lomov, A.A.; Vasil’ev, A.L.; Nekipelov, S.V.; Petrova, O.V. X-Ray and Synchrotron Studies of Porous Silicon. Semiconductors 2013, 47, 1051–1057. [Google Scholar] [CrossRef]
- Petrenko, I.; Summers, A.P.; Simon, P.; Zoltowska-Aksamitowska, S.; Motylenko, M.; Schimpf, C.; Rafaja, D.; Roth, F.; Kummer, K.; Brendler, E.; et al. Extreme biomimetics: Preservation of molecular detail in centimeter-scale samples of biological meshes laid down by sponges. Sci. Adv. 2019, 5, eaax2805. [Google Scholar] [CrossRef] [PubMed]
- Ob’edkov, A.M.; Kaverin, B.S.; Egorov, V.A.; Semenov, N.M.; Ketkov, S.Y.; Domrachev, G.A.; Kremlev, K.V.; Gusev, S.A.; Perevezentsev, V.N.; Moskvichev, A.N.; et al. Macroscopic cylinders on the basis of radial-oriented multi-wall car-bon nanotubes. Lett. Mater. 2012, 2, 152–156. [Google Scholar] [CrossRef]
- Gorovikov, S.A.; Molodtsov, S.L.; Follath, R. Optical design of the high-energy resolution beamline at a dipole magnet of BESSY II. Nucl. Instrum. Methods Phys. Res. A 1998, 411, 506–512. [Google Scholar] [CrossRef]
- Fedoseenko, S.I.; Vyalikh, D.V.; Iossifov, I.F.; Follath, R.; Gorovikov, S.A.; Püttner, R.; Schmidt, J.S.; Molodtsov, S.L.; Adamchuk, V.K.; Gudat, W.; et al. Commissioning results and performance of the high-resolution Russian-German Beamline at BESSY II. Nucl. Instrum. Methods Phys. Res. A 2003, 505, 718–728. [Google Scholar] [CrossRef]
- Batson, P.E. Carbon 1s near-edge-absorption fine structure in graphite. Phys. Rev. B 1993, 48, 2608–2610. [Google Scholar] [CrossRef]
- Henke, B.L.; Gullikson, E.M.; Devis, J.L. X-ray interactions: Photoabsorption, scattering, transmission and reflection at E = 50–30000 eV, Z = 1–92. At. Data Nucl. Data Tables 1993, 54, 181. [Google Scholar] [CrossRef]
- Kummer, K.; Sivkov, V.N.; Vyalikh, D.V.; Maslyuk, V.V.; Bluher, A.; Nekipelov, S.V.; Bredow, T.; Mertig, I.; Mertig, M.; Molodtsov, S.L. Oscillator strength of the peptide bond π* resonances at all relevant x-ray absorption edges. Phys. Rev. B 2009, 80, 155433. [Google Scholar] [CrossRef]
- Zhu, W.Z.; Miser, D.E.; Chan, W.G.; Hajaligol, M.R. Characterization of multiwalled carbon nanotubes prepared by carbon arc cathode deposit. Mater. Chem. Phys. 2003, 82, 638–647. [Google Scholar] [CrossRef]
- Belin, T.; Epron, F. Characterization methods of carbon nanotubes: A review. Mater. Sci. Eng. B 2005, 119, 105–118. [Google Scholar] [CrossRef]
- Zhao, H.; Andro, Y. Raman spectra and X-ray diffraction patterns of carbon nanotubes by hydrogen Arc discharge. Jpn. J. Appl. Phys. 1998, 37, 4846–4849. [Google Scholar] [CrossRef]
- Huang, W.; Wang, Y.; Luo, G.; Wei, F. 99.9% purity multi-walled carbonnanotubes by vacuum high-temperature annealing. Carbon 2003, 41, 2585–2590. [Google Scholar] [CrossRef]
- Banerjee, S.; Hemraj-Benny, T.; Balasubramanian, M.; Fischer, D.A.; Misewich, J.A.; Wong, S.S. Surface Chemistry and Structure of Purified, Ozonized, Multiwalled Carbon Nanotubes Probed by NEXAFS and Vibrational Spectroscopies. ChemPhysChem 2004, 5, 1416–1422. [Google Scholar] [CrossRef] [PubMed]
- Banerjee, S.; Hemraj-Benny, T.; Sambasivan, S.; Fischer, D.A.; James, A.; Misewich, J.A.; Wong, S.S. Near-Edge X-ray Absorption Fine Structure Investigations of Order in Carbon Nanotube-Based Systems. J. Phys. Chem. B 2005, 109, 8489–8495. [Google Scholar] [CrossRef]
- Brzhezinskaya, M.M.; Muradyan, V.E.; Vinogradov, N.A.; Preobrajenski, A.B.; Gudat, W.; Vinogradov, A.S. Electronic structure of fluorinated multiwalled carbon nanotubes studied using x-ray absorption and photoelectron spectroscopy. Phys. Rev. B 2009, 79, 155439. [Google Scholar] [CrossRef]
- Tran, N.H.; Wilson, M.A.; Milev, A.S.; Bartlett, J.R.; Lamb, R.N.; Martin, D.; Kannangara, G.S.K. Photoemission and absorption spectroscopy of carbon nanotube interfacial interaction. Adv. Colloid Interface Sci. 2009, 145, 23–41. [Google Scholar] [CrossRef]
- De Faria, D.L.A.; Venancio Silva, S.; De Oliveira, M.T. Raman Microspectroscopy of Some Iron Oxides and Oxyhydroxides. J. Raman Spectrosc. 1997, 28, 873–878. [Google Scholar] [CrossRef]
- Balberg, I.; Pinch, H.L. The optical absorption of iron oxides. J. Magn. Magn. Mater. 1978, 7, 12–15. [Google Scholar] [CrossRef]
- McClure, D.S. Comparison of the Crystal Fields and Optical Spectra of Cr2O3 and Ruby. J. Chem. Phys. 1963, 38, 2289–2294. [Google Scholar] [CrossRef]
- Li, Q.; Gou, Y.; Wang, T.-G.; Gu, T.; Yu, Q.; Wang, L. Study on Local Residual Stress in a Nanocrystalline Cr2O3 Coating by Micro-Raman Spectroscopy. Coatings 2019, 9, 500. [Google Scholar] [CrossRef]
- Birnie, J.; Craggs, C.; Gardiner, D.J.; Graves, P.R. Ex situ and in situ determination of stress distributions in chromium oxide films by Raman microscopy. Corros. Sci. 1992, 33, 1–12. [Google Scholar] [CrossRef]
- Calvarin, G.; Huntz, A.M.; Le Goff, A.H.; Joiret, S.; Bernard, M.C. Oxide scale stress determination by Raman spectroscopy application to the NiCr/Cr2O3 system and influence of yttrium. Scr. Mater. 1998, 38, 1649–1658. [Google Scholar] [CrossRef]
- Gudat, W.; Kunz, C. Close Similarity between Photoelectric Yield and Photoabsorption Spectra in the Soft-X-Ray Range. Phys. Rev. Lett. 1972, 29, 169–172. [Google Scholar] [CrossRef]
- Henke, B.L.; Smith, J.A.; Attwood, D.T. 0.1–10 keV X-ray induced electron emissions from solids—Models and secondary electron measurements. J. Appl. Phys. 1977, 48, 1852–1866. [Google Scholar] [CrossRef]
- Henke, B.L.; Knauer, J.P.; Premarate, K. The characterization of x-ray photocathodes in the 0.1–10 keV photon energy region. J. Appl. Phys. 1981, 52, 1509–1520. [Google Scholar] [CrossRef]
- Nekipelov, S.V.; Sivkov, V.N. Metallic titanium X-ray absorption cross section spectral dependences in the energy range 240–1500 eV. Phys. Solid State 1994, 36, 1512. [Google Scholar]
- Sonntag, B.; Haensel, R.; Kunz, C. Optical absorption measurements of the transition metals Ti, V, Cr, Mn, Fe, Co, Ni in the region of 3p electron transitions. Solid State Commun. 1969, 7, 597–599. [Google Scholar] [CrossRef]
- Denley, D.; Williams, R.S.; Perfetti, P.; Shirley, D.A.; Stöhr, J. X-ray absorption fine structure above the Ti L-edge. Phys. Rev. B 1979, 19, 1762–1768. [Google Scholar] [CrossRef]
- Regan, T.J.; Ohldag, H.; Stamm, C.; Nolting, F.; Luning, J.; Stöhr, J.; White, R.L. Chemical effects at metal/oxide interfaces studied by x-ray-absorption spectroscopy. Phys. Rev. B 2001, 64, 214422. [Google Scholar] [CrossRef]
- Gago, R.; Vinnichenko, M.; Hübner, R.; Redondo-Cubero, A. Bonding structure and morphology of chromium oxide films grown by pulsed-DC reactive magnetron sputter deposition. J. Alloys Compd. 2016, 672, 529–535. [Google Scholar] [CrossRef]
- Dedkov, Y.S.; Vinogradov, A.S.; Fonin, M.; König, C.; Vyalikh, D.V.; Preobrajenski, A.B.; Krasnikov, S.A.; Kleimenov, Y.; Nesterov, M.A.; Rüdiger, U.; et al. Correlations in the electronic structure of half-metallic ferromagnetic CrO2 films: An x-ray absorption and resonant photoemission spectroscopy study. Phys. Rev. B. 2005, 72, 060401. [Google Scholar] [CrossRef]
- Wurth, W.; Stöhr, J.; Feulner, P.; Pan, X.; Bauchspiess, K.R.; Baba, Y.; Hudel, E.; Rocker, G.; Menzel, D. Bonding, structure, and magnetism of physisorbed and chemisorbed O2 on Pt(111). Phys. Rev. Lett. 1990, 65, 2426–2429. [Google Scholar] [CrossRef]
- Jeong, H.-K.; Noh, H.-J.; Kim, J.-Y.; Jin, M.H.; Park, C.Y.; Lee, Y.H. X-ray absorption spectroscopy of graphite oxide. Eur. Lett. 2008, 82, 67004. [Google Scholar] [CrossRef]
- Madix, R.J.; Solomon, J.L.; Stöhr, J. The orientation of the carbonate anion on Ag (110). Surf. Sci. 1988, 197, L253–L259. [Google Scholar] [CrossRef]
- Chen, J.C. NEXAFS investigations of transition metal oxides, nitrides, carbides, sulfides and other interstitial compounds. Surf. Sci. Rep. 1997, 30, 1–152. [Google Scholar] [CrossRef]
- Jin, Y.; Xu, H.; Datye, A.K. Electron Energy Loss Spectroscopy (EELS) of Iron Fischer–Tropsch Catalysts. Microsc. Microanal. 2006, 12, 124–134. [Google Scholar] [CrossRef]
- Vinogradov, N.A.; Zakharov, A.A.; Kocevski, V.; Rusz, J.; Simonov, K.A.; Eriksson, O.; Mikkelsen, A.; Lundgren, E.; Vinogradov, A.S.; Mårtensson, N.; et al. Formation and Structure of Graphene Waves on Fe(110). Phys. Rev. Lett. 2012, 109, 026101. [Google Scholar] [CrossRef]
- McGuire, G.E. Carbon. In Auger Electron Spectroscopy Reference Manual; Plenum Press: New York, NY, USA, 1979; pp. 41–43. [Google Scholar]
- Seah, M.P.; Dench, W.A. Quantitative electron spectroscopy of surfaces: A standard data base for electron inelastic mean free paths in solids. Surf. Interface Anal. 1979, 1, 2–11. [Google Scholar] [CrossRef]
- Skryleva, E.A.; Parkhomenko, Y.N.; Karnaukh, I.M.; Zhukova, E.A.; Karaeva, A.R.; Mordkovich, V.Z. XPS characterization of MWCNT and C60-based composites. Fuller. Nanotub. Carbon. Nanostruct. 2016, 24, 535–540. [Google Scholar] [CrossRef]
- Singer, G.; Siedlaczek, P.; Sinn, G.; Rennhofer, H.; Mičušík, M.; Omastová, M.; Unterlass, M.M.; Wendrinsky, J.; Milotti, V.; Fedi, F.; et al. Acid Free Oxidation and Simple Dispersion Method of MWCNT for High-Performance CFRP. Nanomaterials 2018, 8, 912. [Google Scholar] [CrossRef]
- Ago, H.; Kugler, T.; Cacialli, F.; Salaneck, W.R.; Shaffer, M.S.P.; Windle, A.H.; Friend, R.H. Work Functions and Surface Functional Groups of Multiwall Carbon Nanotubes. J. Phys. Chem. B 1999, 103, 8116–8121. [Google Scholar] [CrossRef]
- Okpalugo, T.I.T.; Papakonstantinou, P.; Murphy, H.; McLaughlin, J.; Brown, N.M.D. High resolution XPS characterization of chemical functionalised MWCNTs and SWCNTs. Carbon 2005, 43, 153–161. [Google Scholar] [CrossRef]
- Minati, L.; Speranza, G.; Bernagozzi, I.; Torrengo, S.; Toniutti, L.; Rossi, B.; Ferrari, M.; Chiasera, A. Investigation on the Electronic and Optical Properties of Short Oxidized Multiwalled Carbon Nanotubes. J. Phys. Chem. C 2010, 114, 11068–11073. [Google Scholar] [CrossRef]
- Jang, M.-H.; Hwang, Y.S. Effects of functionalized multi-walled carbon nanotubes on toxicity and bioaccumulation of lead in Daphnia magna. PLoS ONE 2018, 13, e0194935. [Google Scholar] [CrossRef]
- National Institute of Standards and Technology. NIST Electron Inelastic-Mean-Free-Path Database: Version 1.2; National Institute of Standards and Technology: Gaithersburg, MD, USA, 2010. Available online: https://www.nist.gov/srd/nist-standard-reference-database-71 (accessed on 31 January 2020). [CrossRef]
- Teghil, R.; Santagata, A.; De Bonis, A.; Galasso, A.; Villani, P. Chromium carbide thin films deposited by ultra-short pulse laser deposition. Appl. Surf. Sci. 2009, 255, 7729–7733. [Google Scholar] [CrossRef]
- Zhao, D.; Jiang, X.; Wang, Y.; Duan, W.; Wang, L. Microstructure evolution, wear and corrosion resistance of Cr-C nanocomposite coatings in seawater. Appl. Surf. Sci. 2018, 457, 914–924. [Google Scholar] [CrossRef]
- Kawabata, A.; Yoshinaka, M.; Hirota, K.; Yamaguchi, O. Hot isostatic pressing and characterization of sol-gel-derived chromium (III) oxide. J. Am. Ceram. Soc. 1995, 78, 2271–2273. [Google Scholar] [CrossRef]
- Balachandran, U.; Siegel, R.W.; Liao, Y.X.; Askew, T.R. Synthesis, sintering, and magnetic properties of nanophase Cr2O3. Nanostruct. Mater. 1995, 5, 505–512. [Google Scholar] [CrossRef]
- Pei, Z.Z.; Zhang, X. Controlled synthesis of large-sized Cr2O3 via hydrothermal reduction. Mater. Lett. 2013, 93, 377–379. [Google Scholar] [CrossRef]
- Ma, H.; Xu, Y.; Rong, Z.; Cheng, X.; Gao, S.; Zhang, X.; Zhao, H.; Huo, L. Highly toluene sensing performance based on monodispersed Cr2O3 porous microspheres. Sens. Actuators B Chem. 2012, 174, 325–331. [Google Scholar] [CrossRef]
- Chen, M.L.; Oh, W.C.; Zhang, F.J. Photonic activity for MB solution of metal oxide/CNT catalysts derived from different organometallic compounds. Fuller. Nanotub. Carbon. Nanostruct. 2012, 20, 127–137. [Google Scholar] [CrossRef]
- Jia, B.; Gao, L.; Sun, J. Self-assembly of magnetite beads along multiwalled carbon nanotubes via a simple hydrothermal process. Carbon 2007, 45, 1476–1481. [Google Scholar] [CrossRef]
- Sun, Z.; Yuan, H.; Liu, Z.; Han, B.; Zhang, X. A Highly efficient chemical sensor material for H2S: α-Fe2O3 nanotubes fabricated using carbon nanotube templates. Adv. Mater. 2005, 17, 2993–2997. [Google Scholar] [CrossRef]
- Hildreth, O.; Cola, B.; Graham, S.; Wong, C.P. Conformally coating vertically aligned carbon nanotube arrays using thermal decomposition of iron pentacarbonyl. J. Vac. Sci. Technol. B 2012, 30, 03D101. [Google Scholar] [CrossRef]

© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sivkov, D.; Petrova, O.; Mingaleva, A.; Ob’edkov, A.; Kaverin, B.; Gusev, S.; Vilkov, I.; Isaenko, S.; Bogachuk, D.; Skandakov, R.; et al. The Structure and Chemical Composition of the Cr and Fe Pyrolytic Coatings on the MWCNTs’ Surface According to NEXAFS and XPS Spectroscopy. Nanomaterials 2020, 10, 374. https://doi.org/10.3390/nano10020374
Sivkov D, Petrova O, Mingaleva A, Ob’edkov A, Kaverin B, Gusev S, Vilkov I, Isaenko S, Bogachuk D, Skandakov R, et al. The Structure and Chemical Composition of the Cr and Fe Pyrolytic Coatings on the MWCNTs’ Surface According to NEXAFS and XPS Spectroscopy. Nanomaterials. 2020; 10(2):374. https://doi.org/10.3390/nano10020374
Chicago/Turabian StyleSivkov, Danil, Olga Petrova, Alena Mingaleva, Anatoly Ob’edkov, Boris Kaverin, Sergey Gusev, Ilya Vilkov, Sergey Isaenko, Dmitriy Bogachuk, Roman Skandakov, and et al. 2020. "The Structure and Chemical Composition of the Cr and Fe Pyrolytic Coatings on the MWCNTs’ Surface According to NEXAFS and XPS Spectroscopy" Nanomaterials 10, no. 2: 374. https://doi.org/10.3390/nano10020374
APA StyleSivkov, D., Petrova, O., Mingaleva, A., Ob’edkov, A., Kaverin, B., Gusev, S., Vilkov, I., Isaenko, S., Bogachuk, D., Skandakov, R., Sivkov, V., & Nekipelov, S. (2020). The Structure and Chemical Composition of the Cr and Fe Pyrolytic Coatings on the MWCNTs’ Surface According to NEXAFS and XPS Spectroscopy. Nanomaterials, 10(2), 374. https://doi.org/10.3390/nano10020374






