Potential Hazard of Lanthanides and Lanthanide-Based Nanoparticles to Aquatic Ecosystems: Data Gaps, Challenges and Future Research Needs Derived from Bibliometric Analysis
Abstract
1. Introduction
2. Existing Ecotoxicological Knowledge on Lanthanides: Identifying the Data Gaps
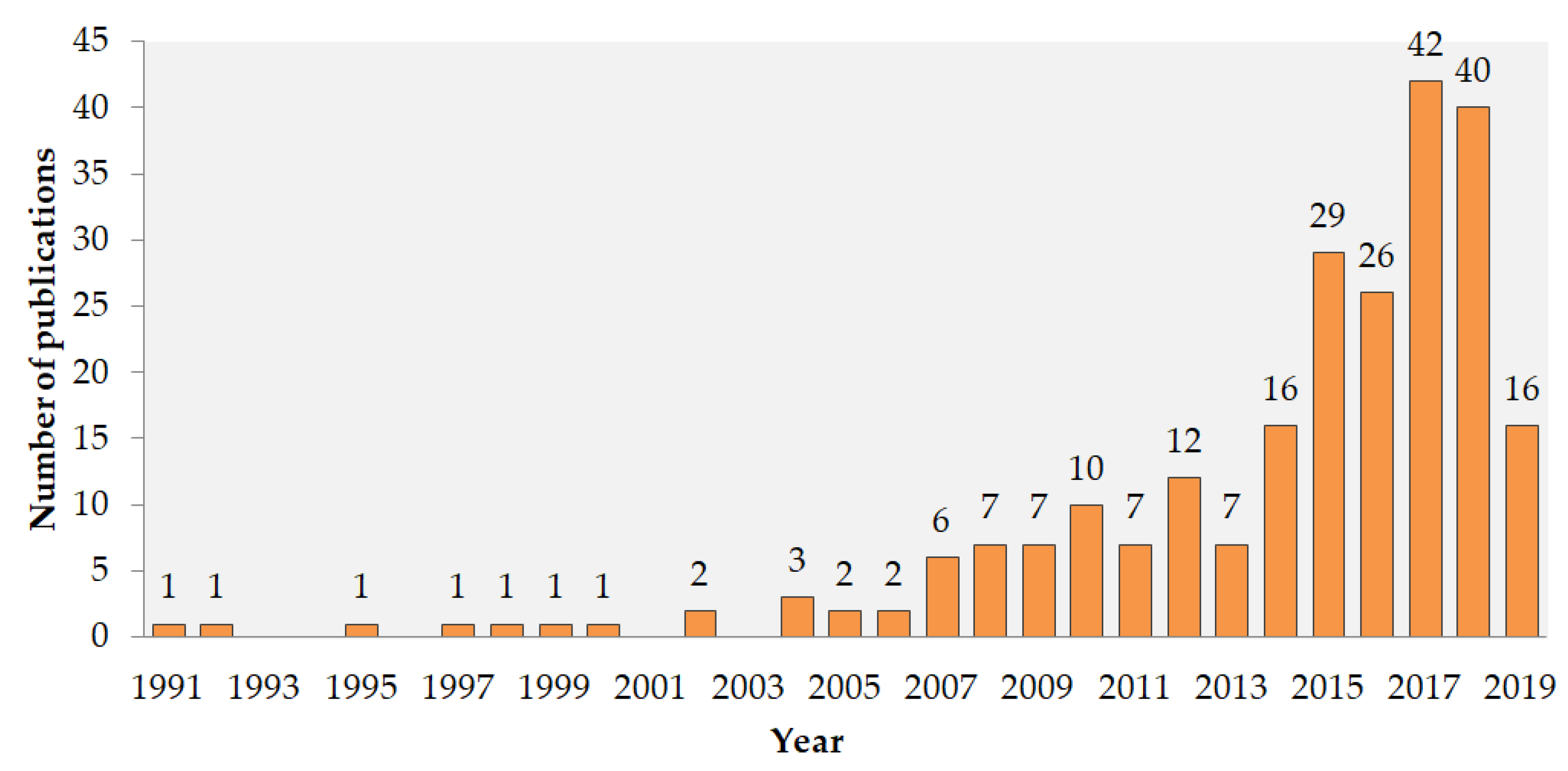
2.1. Choice of the Key-Words for the Search in WoS (Web of Science)
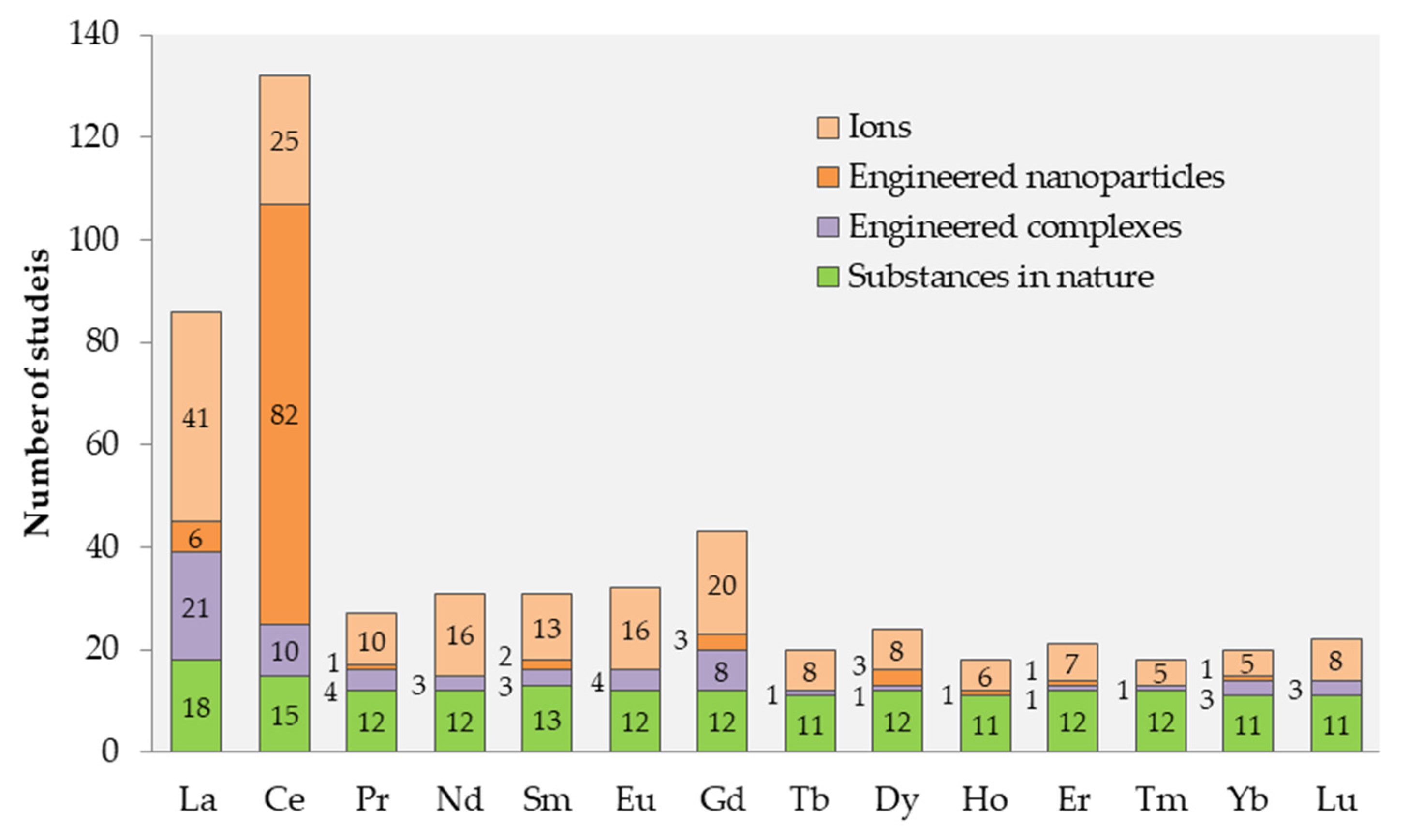
2.2. Information on Different Ln Compounds: WoS
2.3. Analysis of the Existing Information Describing the Environmental Hazard of Ln
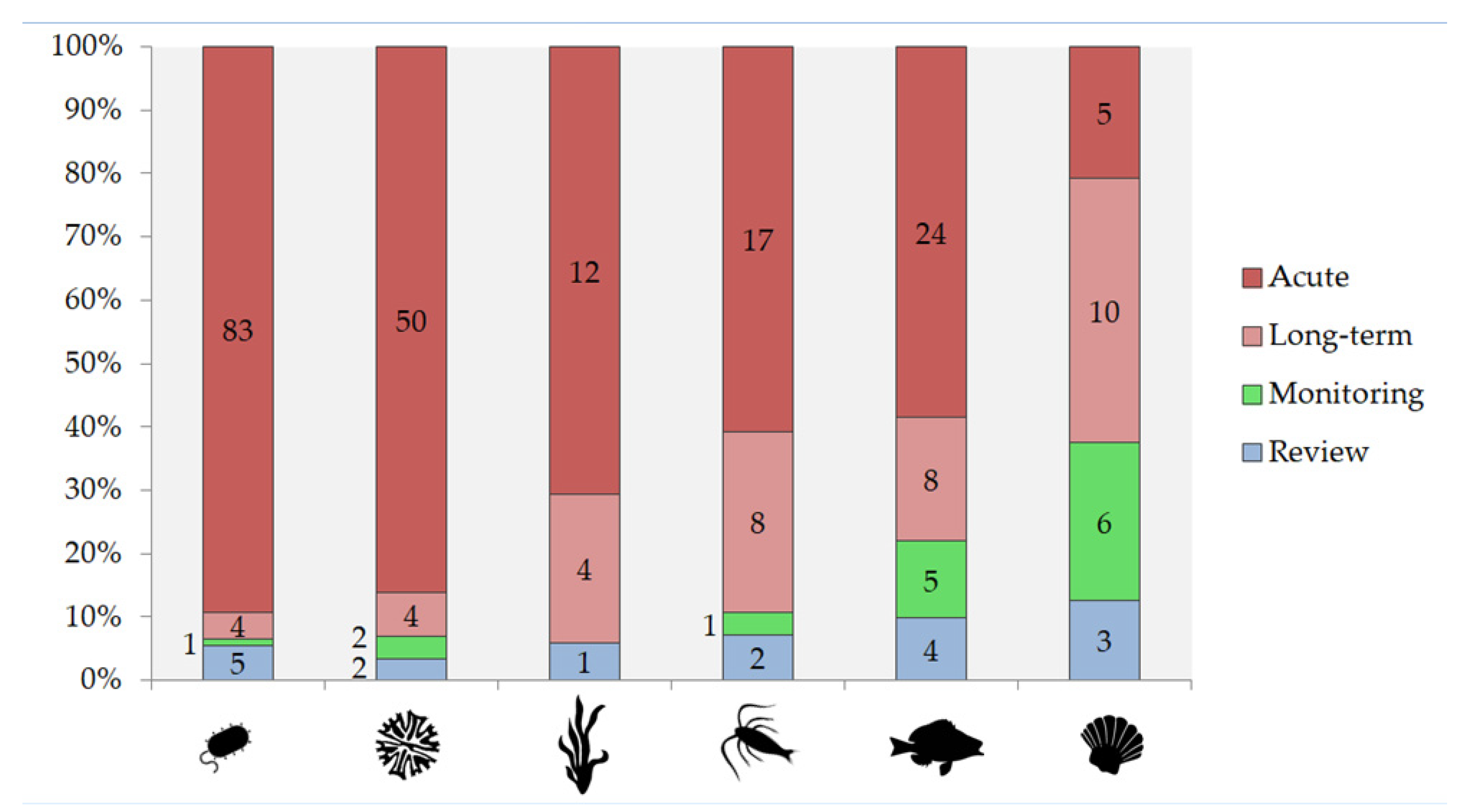
2.3.1. Acute and Long-Term Exposure
2.3.2. Information on Toxicity of Ln for Benthic Organisms
2.3.3. Bioaccumulation Studies
3. Environmental Safety Assessment of Ln Compounds
3.1. Potential Hazard of Ln to Aquatic Ecosystems: State of the Art
3.1.1. Toxicity to Aquatic Biota
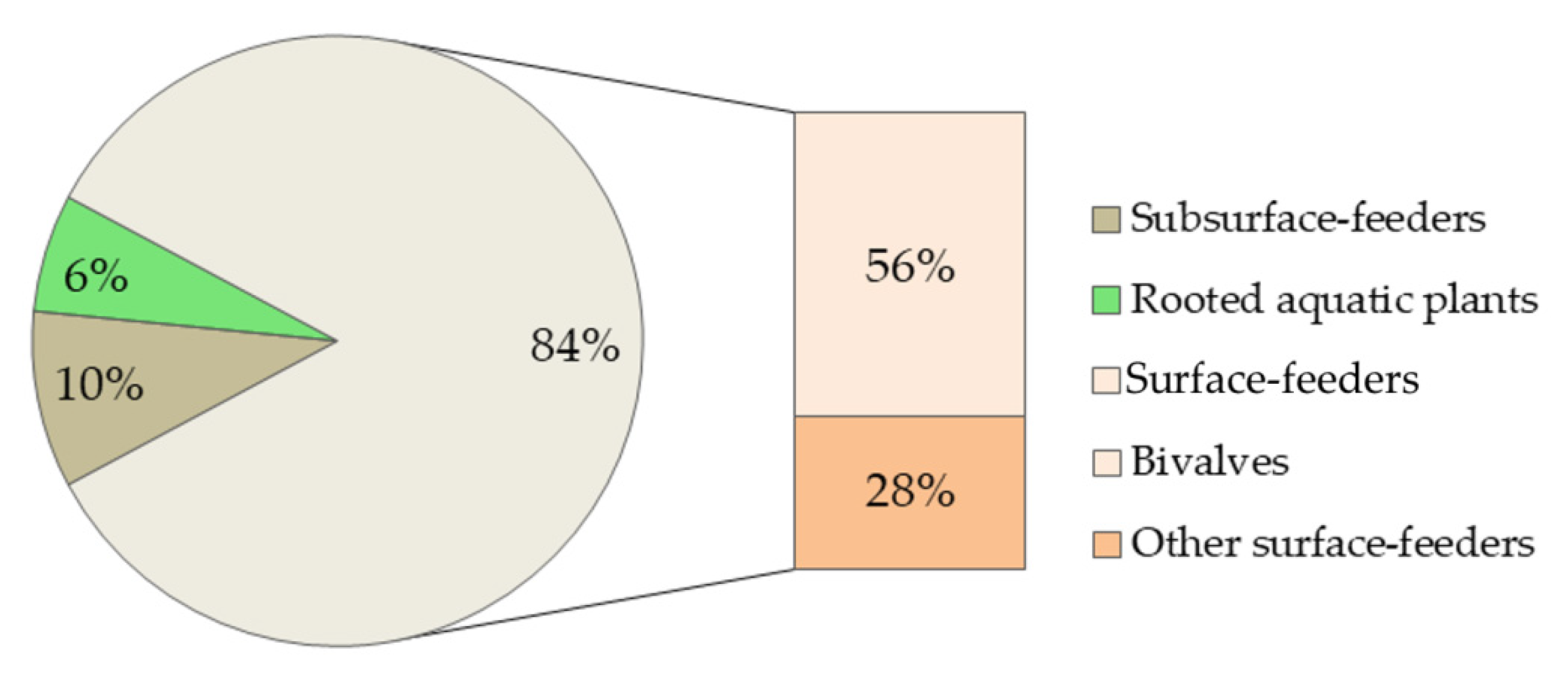
3.1.2. Bioaccumulation of Ln
3.2. Environmental Exposure Levels of Ln
4. Uncertainties in Evaluation of Potential Hazard of Ln Compounds to the Aquatic Organisms
4.1. Behaviour of Ln in the Test Environment
4.2. Ecological Realism of the Laboratory Toxicity Test Results
4.3. Ln as a Uniform Group of Elements
5. Summary
- The most ecotoxicologically studied Ln are Ce and Ln. Practically no information was found for Nd, Tb, Tm, and Yb. More attention in scientific research could be drawn to Ln with lower atomic mass, as they are more abundant and tend to bioaccumulate more than heavy Ln.
- There is a considerable lack of long-term ecotoxicity data from environmentally relevant exposure conditions (Ln concentrations and test media), although these data are the most relevant for an evaluation of the potential hazard of anthropogenic Ln to aquatic ecosystems.
- As Ln tend accumulate in the sediments, more attention should be paid to the adverse effects of Ln to bottom-dwelling species, especially to sediment-digesting ones that are underrepresented in the current literature.
- Although the current environmental concentrations of Ln are still too low to cause adverse effects, remarkable bioaccumulation of Ln in some aquatic plant species is a warning sign. Therefore, additional information on the Ln bioaccumulation potential at all food chain levels is needed.
- There is an urgent need for additional knowledge on the behaviour of Ln in the aquatic environment.
Author Contributions
Funding
Conflicts of Interest
References
- Campbell, G.A. Rare earth metals: A strategic concern. Miner. Econ. 2014, 27, 21–31. [Google Scholar] [CrossRef]
- Balaram, V. Rare earth elements: A review of applications, occurrence, exploration, analysis, recycling, and environmental impact. Geosci. Front. 2019, 10, 1285–1303. [Google Scholar] [CrossRef]
- Lian, H.; Hou, Z.; Shang, M.; Geng, D.; Zhang, Y.; Lin, J. Rare earth ions doped phosphors for improving efficiencies of solar cells. Energy 2013, 57, 270–283. [Google Scholar] [CrossRef]
- Asian Metal. Available online: http://metalpedia.asianmetal.com/metal/rare_earth/application.shtml (accessed on 18 June 2019).
- European Commission. Communication (COM(2012) 572 Final) from the Commission to the European Parliament, the Council and the European Economic and Social Committee: Second Regulatory Review on Nanomaterials; European Commission: Brussels, Belgium, 2013. [Google Scholar]
- Grand View Research. Available online: https://www.grandviewresearch.com/industry-analysis/cerium-oxide-nanoparticles-market (accessed on 12 June 2019).
- Engström, M.; Klasson, A.; Pedersen, H.; Vahlberg, C.; Käll, P.-O.; Uvdal, K. High proton relaxivity for gadolinium oxide nanoparticles. Magn. Reson. Mater. Phys. Biol. Med. 2006, 19, 180–186. [Google Scholar] [CrossRef] [PubMed]
- Balusamy, B.; Kandhasamy, Y.G.; Senthamizhan, A.; Chandrasekaran, G.; Subramanian, M.S.; Kumaravel, T.S. Characterization and bacterial toxicity of lanthanum oxide bulk and nanoparticles. J. Rare Earths 2012, 30, 1298–1302. [Google Scholar] [CrossRef]
- AL-Shawafi, W.M.; Salah, N.; Alshahrie, A.; Ahmed, Y.M.; Moselhy, S.S.; Hammad, A.H.; Hussain, M.A.; Memic, A. Size controlled ultrafine CeO2 nanoparticles produced by the microwave assisted route and their antimicrobial activity. J. Mater. Sci. Mater. Med. 2017, 28. [Google Scholar] [CrossRef]
- Farias, I.A.P.; Dos Santos, C.C.L.; Sampaio, F.C. Antimicrobial activity of cerium oxide nanoparticles on opportunistic microorganisms: A systematic review. BioMed Res. Int. 2018, 1–14. [Google Scholar] [CrossRef]
- Krishnamoorthy, K.; Veerapandian, M.; Zhang, L.H.; Yun, K.; Kim, S.J. Surface chemistry of cerium oxide nanocubes: Toxicity against pathogenic bacteria and their mechanistic study. J. Ind. Eng. Chem. 2014, 20, 3513–3517. [Google Scholar] [CrossRef]
- Aashima, S.K.P.; Singh, S.; Mehta, S.K. Biocompatible gadolinium oxide nanoparticles as efficient agent against pathogenic bacteria. J. Colloid Interface Sci. 2018, 529, 496–504. [Google Scholar] [CrossRef]
- Aramesh-Boroujeni, Z.; Khorasani-Motlagh, M.; Noroozifar, M. Multispectroscopic DNA-binding studies of a terbium(III) complex containing 2,2’-bipyridine ligand. J. Biomol. Struct. Dyn. 2016, 34, 414–426. [Google Scholar] [CrossRef]
- Muthukumaran, M.; Venkateswara Raju, C.; Sumathi, C.; Ravi, G.; Solairaj, D.; Rameshthangam, P.; Wilson, J.; Rajendran, S.; Alwarappan, S. Cerium doped nickel-oxide nanostructures for riboflavin biosensing and antibacterial applications. New J. Chem. 2016, 40, 2741–2748. [Google Scholar] [CrossRef]
- Tyler, G. Rare earth elements in soil and plant systems—A review. Plant Soil 2004, 267, 191–206. [Google Scholar] [CrossRef]
- Volokh, A.A.; Gorbunov, A.V.; Gundorina, S.F.; Revich, B.A.; Frontasyeva, M.V.; Pal, C.S. Phosphorus fertilizer production as a source of rare-earth elements pollution of the environment. Sci. Total Environ. 1990, 95, 141–148. [Google Scholar] [CrossRef]
- Keller, A.A.; McFerran, S.; Lazareva, A.; Suh, S. Global life cycle releases of engineered nanomaterials. J. Nanopart. Res. 2013, 15, 1692. [Google Scholar] [CrossRef]
- Garner, K.L.; Suh, S.; Keller, A.A. Assessing the risk of engineered nanomaterials in the environment: Development and application of the nanoFate model. Environ. Sci. Technol. 2017, 51, 5541–5551. [Google Scholar] [CrossRef]
- Peters, R.J.B.; van Bemmel, G.; Milani, N.B.L.; den Hertog, G.C.T.; Undas, A.K.; van der Lee, M.; Bouwmeester, H. Detection of nanoparticles in Dutch surface waters. Sci. Total Environ. 2018, 621, 210–218. [Google Scholar] [CrossRef]
- Telgmann, L.; Sperling, M.; Karst, U. Determination of gadolinium-based MRI contrast agents in biological and environmental samples: A review. Anal. Chim. Acta 2013, 764, 1–16. [Google Scholar] [CrossRef]
- Hatje, V.; Bruland, K.W.; Flegal, A.R. Increases in anthropogenic gadolinium anomalies and rare earth element concentrations in San Francisco Bay over a 20 year record. Environ. Sci. Technol. 2016, 50, 4159–4168. [Google Scholar] [CrossRef]
- Birka, M.; Roscher, J.; Holtkamp, M. Investigating the stability of gadolinium based contrast agents towards UV radiation. Water Res. 2016, 91, 244–250. [Google Scholar] [CrossRef]
- Sauve, S.; Desrosiers, M. A review of what is an emerging contaminant. Chem. Cent. J. 2014, 8. [Google Scholar] [CrossRef]
- Bondarenko, O.; Juganson, K.; Ivask, A.; Kasemets, K.; Mortimer, M.; Kahru, A. Toxicity of Ag, CuO and ZnO nanoparticles to selected environmentally relevant test organisms and mammalian cells in vitro: A critical review. Arch. Toxicol. 2013, 87, 1181–1200. [Google Scholar] [CrossRef] [PubMed]
- Heinlaan, M.; Ivask, A.; Blinova, I.; Dubourguier, H.-C.; Kahru, A. Toxicity of nanosized and bulk ZnO, CuO and TiO2 to bacteria Vibrio fischeri and crustaceans Daphnia magna and Thamnocephalus platyurus. Chemosphere 2008, 71, 1308–1316. [Google Scholar] [CrossRef] [PubMed]
- Aruoja, V.; Pokhrel, S.; Sihtmäe, M.; Mortimer, M.; Lutz Mädlerb, L.; Kahru, A. Toxicity of 12 metal-based nanoparticles to algae, bacteria and protozoa. Environ. Sci. Nano 2015, 2, 630–644. [Google Scholar] [CrossRef]
- Anaya, N.M.; Solomon, F.; Oyanedel-Craver, V. Effects of dysprosium oxide nanoparticles on Escherichia coli. Environ. Sci. Nano 2016, 3, 67–73. [Google Scholar] [CrossRef]
- Röhder, L.A.; Brandt, T.; Sigg, L.; Behra, R. Influence of agglomeration of cerium oxide nanoparticles and speciation of cerium(III) on short term effects to the green algae Chlamydomonas reinhardtii. Aquat. Toxicol. 2014, 152, 121–130. [Google Scholar] [CrossRef] [PubMed]
- Kosak née Röhder, L.A.; Brandt, T.; Sigg, L.; Behra, R. Uptake and effects of cerium (III) and cerium oxide nanoparticles to Chlamydomonas reinhardtii. Aquat. Toxicol. 2018, 197, 41–46. [Google Scholar] [CrossRef] [PubMed]
- Gonzalez, V.; Vignati, D.A.L.; Pons, M.N.; Montarges-Pelletier, E.; Bojic, C.; Giamberini, L. Lanthanide ecotoxicity: First attempt to measure environmental risk for aquatic organisms. Environ. Pollut. 2015, 199, 139–147. [Google Scholar] [CrossRef]
- Cobelo-García, A.; Filella, M.; Croot, P.; Frazzoli, C.; Du Laing, G.; Ospina-Alvarez, N.; Rauch, S.; Salaun, P.; Schäfer, J.; Zimmermann, S. COST action TD1407: Network on technology-critical elements (NOTICE)—from environmental processes to human health threats. Environ. Sci. Pollut. Res. 2015, 22, 15188–15194. [Google Scholar] [CrossRef]
- Tchounwou, P.B.; Yedjou, C.G.; Patlolla, A.K.; Sutton, D.J. Heavy metal toxicity and the environment. Exp. Suppl. 2012, 101, 133–164. [Google Scholar] [CrossRef]
- Pang, X.; Li, D.; Peng, A. Application of rare-earth elements in the agriculture of China and its environmental behavior in soil. J. Soils Sediments 2001, 1, 124–129. [Google Scholar] [CrossRef]
- Hanana, H.; Turcotte, P.; André, C.; Gagnon, C.; Gagné, F. Comparative study of the effects of gadolinium chloride and gadolinium – based magnetic resonance imaging contrast agent on freshwater mussel, Dreissena plymorpha. Chemosphere 2017, 181, 197–207. [Google Scholar] [CrossRef] [PubMed]
- Rim, K.-T. Effects of rare earth elements on the environment and human health: A literature review. Toxicol. Environ. Health Sci. 2016, 8, 189–200. [Google Scholar] [CrossRef]
- Pagano, G.; Aliberti, F.; Guida, M.; Oral, R.; Siciliano, A.; Trifuoggi, M.; Tommasi, F. Rare earth elements in human and animal health: State of art and research priorities. Environ. Res. 2015, 142, 215–220. [Google Scholar] [CrossRef] [PubMed]
- Wang, B.; Yan, L.; Huo, W.; Lu, Q.; Cheng, Z.; Zhang, J.; Li, Z. Rare earth elements and hypertension risk among housewives: A pilot study in Shanxi Province, China. Environ. Pollut. 2017, 220, 837–842. [Google Scholar] [CrossRef]
- Poniedziałek, B.; Rzymski, P.; Pięt, M.; Niedzielski, P.; Mleczek, M.; Wilczak, M.; Rzymski, P. Rare-earth elements in human colostrum milk. Environ. Sci. Pollut. Res. 2017, 24, 26148–26154. [Google Scholar] [CrossRef]
- Marzec-Wróblewska, U.; Kamiński, P.; Łakota, P.; Ludwikowski, G.; Szymański, M.; Wasilow, K.; Stuczyński, T.; Buciński, A.; Jerzak, L. Determination of rare earth elements in human sperm and association with semen quality. Arch. Environ. Contam. Toxicol. 2015, 69, 191–201. [Google Scholar] [CrossRef]
- Herrmann, H.; Nolde, J.; Berger, S.; Heise, S. Aquatic ecotoxicity of lanthanum—A review and an attempt to derive water and sediment quality criteria. Ecotoxicol. Environ. Saf. 2016, 124, 213–238. [Google Scholar] [CrossRef]
- Dahle, J.T.; Arai, Y. Environmental geochemistry of cerium: Applications and toxicology of cerium oxide nanoparticles. Int. J. Environ. Res. Public Health 2015, 12, 1253–1278. [Google Scholar] [CrossRef]
- Blaise, C.; Gagné, F.; Harwood, M.; Quinn, B.; Hanana, H. Ecotoxicity responses of the freshwater cnidarian Hydra attenuata to 11 rare earth elements. Ecotoxicol. Environ. Saf. 2018, 163, 486–491. [Google Scholar] [CrossRef]
- Gonzalez, V.; Vignati, D.A.L.; Leyval, C.; Giamberini, L. Environmental fate and ecotoxicity of lanthanides: Are they a uniform group beyond chemistry? Environ. Int. 2014, 71, 148–157. [Google Scholar] [CrossRef]
- Sneller, F.E.C.; Kalf, D.F.; Weltje, L.; Van Wezel, A.P. Maximum Permissible Concentrations and Negligible Concentrations for Rare Earth Elements (REEs). RIVM report 601501 011 2000, 1–66. [Google Scholar]
- European Commission. Regulation (EC) No 1272/2008 of the Europaen Parliament and of the Council of 16 December 2008 on classification, labelling and packaging of substances and mixtures, amending and repealing Directives 67/548/EEC and 1999/45/EC, and amending Regulation (EC). 2008. Available online: http://ec.europa.eu/growth/sectors/chemicals/classification-labelling/ (accessed on 16 June 2019).
- ECHA (European Chemical Agency). Guidance on Information Requirements and Chemical Safety Sssessment. Chapter R.7b: Endpoint Specific Guidance; European Chemical Agency: Helsinki, Finland, 2017. [CrossRef]
- Gwenzi, W.; Mangori, L.; Danha, C.; Chaukura, N.; Dunjana, N.; Sanganyado, E. Sources, behaviour, and environmental and human health risks of high-technology rare earth elements as emerging contaminants. Sci. Total Environ. 2018, 636, 299–313. [Google Scholar] [CrossRef]
- Bishop, W.M.; McNabb, T.; Cormican, I.; Willis, B.E.; Hyde, S. Operational evaluation of Phoslock phosphorus locking technology in Laguna Niguel Lake, California. Water Air Soil Pollut. 2014, 225. [Google Scholar] [CrossRef]
- Zhang, J.; Zhang, T.; Lu, Q.; Cai, S.; Chu, W.; Qiu, H.; Xu, T.; Li, F.; Xu, Q. Oxidative effects, nutrients and metabolic changes in aquatic macrophyte, Elodea nuttallii, following exposure to lanthanum. Ecotoxicol. Environ. Saf. 2015, 115, 159–165. [Google Scholar] [CrossRef]
- Waajen, G.; van Oosterhout, F.; Lürling, M. Bio-accumulation of lanthanum from lanthanum modified bentonite treatments in lake restoration. Environ. Pollut. 2017, 230, 911–918. [Google Scholar] [CrossRef] [PubMed]
- Guo, X.; Feng, C. Biological toxicity response of Asian Clam (Corbicula fluminea) to pollutants in surface water and sediment. Sci. Total Environ. 2018, 631–632, 56–70. [Google Scholar] [CrossRef] [PubMed]
- Copetti, D.; Finsterle, K.; Marziali, L.; Stefani, F.; Tartari, G.; Douglas, G.; Reitzel, K.; Spears, B.M.; Winfield, I.J.; Crosa, G.; et al. Eutrophication management in surface waters using lanthanum modified bentonite: A review. Water Res. 2016, 97, 162–174. [Google Scholar] [CrossRef]
- Bonnail, E.; Pérez-López, R.; Sarmiento, A.M.; Nieto, J.M.; DelValls, T.Á. A novel approach for acid mine drainage pollution biomonitoring using rare earth elements bioaccumulated in the freshwater clam Corbicula fluminea. J. Hazards Mater. 2017, 338, 466–471. [Google Scholar] [CrossRef]
- Hanana, H.; Turcotte, P.; Dubé, M.; Gagnon, C.; Gagné, F. Response of the freshwater mussel, Dreissena polymorpha to sub-lethal concentrations of samarium and yttrium after chronic exposure. Ecotoxicol. Environ. Saf. 2018, 165, 662–670. [Google Scholar] [CrossRef]
- Henriques, B.; Coppola, F.; Monteiro, R.; Pinto, J.; Viana, T.; Pretti, C.; Soares, A.; Freitas, R.; Pereira, E. Toxicological assessment of anthropogenic gadolinium in seawater: Biochemical effects in mussels Mytilus galloprovincialis. Sci. Total Environ. 2019, 664, 626–634. [Google Scholar] [CrossRef]
- Pinto, J.; Costa, M.; Leite, C.; Borges, C.; Coppola, F.; Henriques, B.; Monteiro, R.; Russo, T.; Di Cosmo, A.; Soares, A.M.V.M.; et al. Ecotoxicological effects of lanthanum in Mytilus galloprovincialis: Biochemical and histopathological impacts. Aquat. Toxicol. 2019, 181–192. [Google Scholar] [CrossRef] [PubMed]
- Van Oosterhout, F.; Goitom, E.; Roessink, I.; Lürling, M. Lanthanum from a modified clay used in eutrophication control is bioavailable to the marbled crayfish (Procambarus fallax f. virginalis). PLoS ONE 2014, 9. [Google Scholar] [CrossRef] [PubMed]
- Zhao, X.; Yu, M.; Xu, D.; Liu, A.; Hou, X.; Hao, F.; Long, Y.; Zhou, Q.; Jiang, G. Distribution, bioaccumulation, trophic transfer, and influences of CeO2 nanoparticles in a constructed aquatic food web. Environ. Sci. Technol. 2017, 51, 5205–5214. [Google Scholar] [CrossRef] [PubMed]
- Amyot, M.; Clayden, M.G.; Macmillan, G.A.; Perron, T.; Arscott-Gauvin, A. Fate and trophic transfer of rare earth elements in temperate lake food webs. Environ. Sci. Technol. 2017, 51, 6009–6017. [Google Scholar] [CrossRef]
- Bosco-Santos, A.; Luiz-Silva, W.; Silva-Filho, E.V.; de Souza, M.D.C.; Dantas, E.L.; Navarro, M.S. Fractionation of rare earth and other trace elements in crabs, Ucides cordatus, from a subtropical mangrove affected by fertilizer industry. J. Environ. Sci. 2017, 54, 69–76. [Google Scholar] [CrossRef]
- Chassard-Bouchaud, C.; Escaig, F.; Boumati, P.; Galle, P. Microanalysis and image processing of stable and radioactive elements in ecotoxicology. Current developments using SIMS microscope and electron microprobe. Biol. Cell 1992, 74, 59–74. [Google Scholar] [CrossRef]
- Fuad, M.M.; Shazili, N.A.M.; Faridah, M. Trace metals and rare earth elements in Rock Oyster Saccostrea cucullata along the east coast of Peninsular Malaysia. Aquat. Ecosyst. Health Manag. 2013, 16, 78–87. [Google Scholar] [CrossRef]
- MacMillan, G.A.; Chételat, J.; Heath, J.P.; Mickpegak, R.; Amyot, M. Rare earth elements in freshwater, marine, and terrestrial ecosystems in the eastern Canadian Arctic. Environ. Sci. Processes Impacts 2017, 19, 1336–1345. [Google Scholar] [CrossRef]
- Merschel, G.; Bau, M. Rare earth elements in the aragonitic shell of freshwater mussel Corbicula fluminea and the bioavailability of anthropogenic lanthanum, samarium and gadolinium in river water. Sci. Total Environ. 2015, 533, 91–101. [Google Scholar] [CrossRef]
- Pratas, J.; Favas, P.J.C.; Varun, M.; D’Souza, R.; Paul, M.S. Distribution of rare earth elements, thorium and uranium in streams and aquatic mosses of Central Portugal. Environ. Earth Sci. 2017, 76. [Google Scholar] [CrossRef]
- Rodriguez-Hernandez, Á.; Zumbado, M.; Henriquez-Hernandez, A.L.; Boada, L.D.; Luzardo, O.P. Dietary intake of essential, toxic, and potentially toxic elements from Mussels (Mytilus spp.) in the Spanish population: A nutritional assessment. Nutrients 2019, 11, 864. [Google Scholar] [CrossRef] [PubMed]
- Squadrone, S.; Brizio, P.; Stella, C.; Prearo, M.; Pastorino, P.; Serracca, L.; Ercolini, C.; Abete, M.C. Presence of trace metals in aquaculture marine ecosystems of the northwestern Mediterranean Sea (Italy). Environ. Pollut. 2016, 215, 77–83. [Google Scholar] [CrossRef] [PubMed]
- Zhao, L.; Liu, L. Assessing the impact of lanthanum on the bivalve Corbicula fluminea in the Rhine River. Sci. Total Environ. J. 2018, 641, 830–839. [Google Scholar] [CrossRef] [PubMed]
- Pagano, G.; Guida, M.; Siciliano, A.; Oral, R.; Koçba, F.; Palumbo, A.; Castellano, I.; Migliaccio, O.; Thomas, P.J.; Trifuoggi, M. Comparative toxicities of selected rare earth elements: Sea urchin embryogenesis and fertilization damage with redox and cytogenetic effects. Environ. Res. 2016, 147, 453–460. [Google Scholar] [CrossRef] [PubMed]
- Peng, C.; Chen, Y.; Pu, Z.; Zhao, Q.; Tong, X.; Chen, Y.; Jiang, L. CeO2 nanoparticles alter the outcome of species interactions. Nanotoxicology 2017, 11, 625–636. [Google Scholar] [CrossRef] [PubMed]
- Kurvet, I.; Juganson, K.; Vija, H.; Sihtmäe, M.; Blinova, I.; Syvertsen-Wiig, G.; Kahru, A. Toxicity of nine (doped) rare earth metal oxides and respective individual metals to aquatic microorganisms Vibrio fischeri and Tetrahymena thermophila. Materials 2017, 10, 754. [Google Scholar] [CrossRef]
- Bour, A.; Mouchet, F.; Verneuil, L.; Evariste, L.; Silvestre, J.; Pinelli, E.; Gauthier, L. Toxicity of CeO2 nanoparticles at different trophic levels - Effects on diatoms, chironomids and amphibians. Chemosphere 2015, 120, 230–236. [Google Scholar] [CrossRef]
- Dogra, Y.; Arkill, K.P.; Elgy, C.; Stolpe, B.; Lead, J.; Valsami-Jones, E.; Tyler, C.R.; Galloway, T.S. Cerium oxide nanoparticles induce oxidative stress in the sediment-dwelling amphipod Corophium volutator. Nanotoxicology 2016, 10, 480–487. [Google Scholar] [CrossRef] [PubMed]
- Gaiser, B.K.; Fernandes, T.F.; Jepson, M.A.; Lead, J.R.; Tyler, C.R.; Baalousha, M.; Biswas, A.; Britton, G.J.; Cole, P.A.; Johnston, B.D.; et al. Interspecies comparisons on the uptake and toxicity of silver and cerium dioxide nanoparticles. Environ. Toxicol. Chem. 2012, 31, 144–154. [Google Scholar] [CrossRef]
- Van Hoecke, K.; Quik, J.T.K.; Mankiewicz-Boczek, J.; De Schamphelaere, K.A.C.; Elsaesser, A.; Van der Meeren, P.; Barnes, C.; Mckerr, G.; Howard, C.V.; Van de Meent, D.; et al. Fate and effects of CeO2 nanoparticles in aquatic ecotoxicity tests. Environ. Sci. Technol. 2009, 43, 4537–4546. [Google Scholar] [CrossRef]
- Tang, J.; Zhu, N.; Zhu, Y.; Zamir, S.M.; Wu, Y. Sustainable pollutant removal by periphytic biofilm via microbial composition shifts induced by uneven distribution of CeO2 nanoparticles. Bioresour. Technol. 2018, 248, 75–81. [Google Scholar] [CrossRef] [PubMed]
- Joonas, E.; Aruoja, V.; Olli, K.; Syvertsen-wiig, G.; Vija, H.; Kahru, A. Potency of (doped) rare earth oxide particles and their constituent metals to inhibit algal growth and induce direct toxic effects. Sci. Total Environ. J. 2017, 594, 478–486. [Google Scholar] [CrossRef] [PubMed]
- Blinova, I.; Vija, H.; Lukjanova, A.; Muna, M.; Syvertsen-wiig, G.; Kahru, A. Assessment of the hazard of nine (doped) lanthanides-based ceramic oxides to four aquatic species. Sci. Total Environ. 2018, 612, 1171–1176. [Google Scholar] [CrossRef] [PubMed]
- Gaiser, B.K.; Biswas, A.; Rosenkranz, P.; Jepson, M.A.; Lead, J.R.; Stone, V.; Tyler, C.R.; Fernandes, T.F. Effects of silver and cerium dioxide micro- and nano-sized particles on Daphnia magna. J. Environ. Monit. 2011, 13, 1227–1235. [Google Scholar] [CrossRef]
- Juganson, K.; Ivask, A.; Blinova, I.; Mortimer, M.; Kahru, A. NanoE-Tox: New and in-depth database concerning ecotoxicity of nanomaterials. Beilstein J. Nanotechnol. 2015, 6, 1788–1804. [Google Scholar] [CrossRef] [PubMed]
- Blaise, C.; Gagné, F.; Férard, J.F.; Eullaffroy, P. Ecotoxicity of selected nano-materials to aquatic organisms. Environ. Toxicol. 2008, 23, 591–598. [Google Scholar] [CrossRef] [PubMed]
- Manier, N.; Bado-nilles, A.; Delalain, P.; Aguerre-chariol, O.; Pandard, P. Ecotoxicity of non-aged and aged CeO2 nanomaterials towards freshwater microalgae. Environ. Pollut. 2013, 180, 63–70. [Google Scholar] [CrossRef] [PubMed]
- He, X.; Pan, Y.; Zhang, J.; Li, Y.; Ma, Y.; Zhang, P.; Ding, Y.; Zhang, J.; Wu, Z.; Zhao, Y.; et al. Quantifying the total ionic release from nanoparticles after particle-cell contact. Environ. Pollut. 2015, 196, 194–200. [Google Scholar] [CrossRef]
- Thill, A.; Zeyons, O.; Spalla, O.; Chauvat, F.; Rose, J.; Auffan, M. Cytotoxicity of CeO2 nanoparticles physico-chemical insight of the cytotoxicity mechanism. Environ. Sci. Technol. 2006, 40, 6151–6156. [Google Scholar] [CrossRef]
- Xie, C.; Zhang, J.; Ma, Y.; Ding, Y.; Zhang, P.; Zheng, L.; Chai, Z.; Zhao, Y.; Zhang, Z.; He, X. Bacillus subtilis causes dissolution of ceria nanoparticles at the nano-bio interface. Environ. Sci. Nano 2019, 6, 216–223. [Google Scholar] [CrossRef]
- Angel, B.M.; Vallotton, P.; Apte, S.C. On the mechanism of nanoparticulate CeO2 toxicity to freshwater algae. Aquat. Toxicol. 2015, 168, 90–97. [Google Scholar] [CrossRef] [PubMed]
- Garaud, M.; Trapp, J.; Devin, S.; Cossu-Leguille, C.; Pain-Devin, S.; Felten, V.; Giamberini, L. Multibiomarker assessment of cerium dioxide nanoparticle (nCeO) sublethal effects on two freshwater invertebrates, Dreissena polymorpha and Gammarus roeseli. Aquat. Toxicol. 2015, 158, 63–74. [Google Scholar] [CrossRef] [PubMed]
- Taylor, N.S.; Merrifield, R.; Williams, T.D.; Chipman, J.K.; Lead, J.R.; Viant, M.R. Molecular toxicity of cerium oxide nanoparticles to the freshwater alga Chlamydomonas reinhardtii is associated with supra-environmental exposure concentrations. Nanotoxicology 2015, 10, 32–41. [Google Scholar] [CrossRef] [PubMed]
- David, E.M.D.S.; Royam, M.M.; Sekar, S.K.R.; Manivannan, B.; Soman, S.J.; Mukherjee, A.; Natarajan, C. Toxicity, uptake, and accumulation of nano and bulk cerium oxide particles in Artemia salina. Environ. Sci. Pollut. Res. 2017, 24, 24187–24200. [Google Scholar] [CrossRef]
- Rodea-Palomares, I.; Gonzalo, S.; Santiago-Morales, J.; Leganés, F.; García-Calvo, E.; Rosal, R.; Fernández-Piñas, F. An insight into the mechanisms of nanoceria toxicity in aquatic photosynthetic organisms. Aquat. Toxicol. 2012, 122–123, 133–143. [Google Scholar] [CrossRef]
- Bellio, P.; Luzi, C.; Mancini, A.; Cracchiolo, S.; Passacantando, M.; Di Pietro, L.; Perilli, M.; Amicosante, G.; Santucci, S.; Celenza, G. Cerium oxide nanoparticles as potential antibiotic adjuvant. Effects of CeO2 nanoparticles on bacterial outer membrane permeability. Biochim. Biophys. Acta Biomembr. 2018, 1860, 2428–2435. [Google Scholar] [CrossRef]
- Rodea-Palomares, I.; Boltes, K.; Fernandez-Pinas, F.; Leganes, F.; Garcia-Calvo, E.; Santiago, J.; Rosal, R. Physicochemical Characterization and Ecotoxicological Assessment of CeO2 Nanoparticles Using Two Aquatic Microorganisms. Toxicol. Sci. 2011, 119, 135–145. [Google Scholar] [CrossRef]
- Forest, V.; Leclerc, L.; Hochepied, J.F.; Trouvé, A.; Sarry, G.; Pourchez, J. Impact of cerium oxide nanoparticles shape on their in vitro cellular toxicity. Toxicol. In Vitro. 2017, 38, 136–141. [Google Scholar] [CrossRef]
- Unnithan, A.R.; Sasikala, A.R.K.; Sathishkumar, Y.; Lee, Y.S.; Park, C.H.; Kim, C.S. Nanoceria doped electrospun antibacterial composite mats for potential biomedical applications. Ceramics Int. 2014, 40, 12003–12012. [Google Scholar] [CrossRef]
- Blinova, I.; Lukjanova, A.; Muna, M.; Vija, H.; Kahru, A. Evaluation of the potential hazard of lanthanides to freshwater microcrustaceans. Sci. Total Environ. 2018, 642, 1100–1107. [Google Scholar] [CrossRef]
- Barry, M.J.; Meehan, B.J. The acute and chronic toxicity of lanthanum to Daphnia carinata. Chemosphere 2000, 41, 1669–1674. [Google Scholar] [CrossRef]
- Stauber, J.L. Toxicity Testing of Modified Clay Leachates Using Freshwater Organisms. Report No: ET/IR267R 2000, 1–27. [Google Scholar]
- Filipi, R.; Nesmerak, K.; Rucki, M.; Roth, Z.; Hanzlikova, I.; Tichy, M. Acute toxicity of rare earth elements and their compounds. Chem. Listy 2007, 101, 793–798. [Google Scholar]
- Ji, L.; Liu, J.; Song, W.; Li, S.; Miao, D. Effects of dietary europium complex and europium(III) on cultured pearl colour in the pearl oyster Pinctada martensii. Aquac. Res. 2013, 44, 1300–1306. [Google Scholar] [CrossRef]
- Romero-Freire, A.; Minguez, L.; Pelletier, M.; Cayer, A.; Caillet, C.; Devin, S.; Gross, E.M.; Guérold, F.; Pain-devin, S.; Vignati, D.A.L.; et al. Assessment of baseline ecotoxicity of sediments from a prospective mining area enriched in light rare earth elements. Sci. Total Environ. 2018, 612, 831–839. [Google Scholar] [CrossRef]
- Paquet, N.; Indiketi, N.; Dalencourt, C.; Larivière, D.; Roberge, S.; Gruyer, N.; Triffault-Bouchet, G.; Fortin, C. Toxicity of tailing leachates from a niobium mine toward three aquatic organisms. Ecotoxicol. Environ. Saf. 2019, 176, 355–363. [Google Scholar] [CrossRef]
- Correia, A.T.; Rebelo, D.; Marques, J.; Nunes, B. Effects of the chronic exposure to cerium dioxide nanoparticles in Oncorhynchus mykiss: Assessment of oxidative stress, neurotoxicity and histological alterations. Environ. Toxicol. Pharmacol. 2019, 68, 27–36. [Google Scholar] [CrossRef]
- Xu, Q.; Jiang, Y.; Chu, W.; Su, C.; Hu, D.; Lu, Q.; Zhang, T. Response of Spirodela polyrhiza to cerium: Subcellular distribution, growth and biochemical changes. Ecotoxicol. Environ. Saf. 2017, 139, 56–64. [Google Scholar] [CrossRef]
- Xu, T.; Su, C.; Hu, D.; Li, F.; Lu, Q.; Zhang, T.; Xu, Q. Molecular distribution and toxicity assessment of praseodymium by Spirodela polyrrhiza. J. Hazards Mater. 2016, 312, 132–140. [Google Scholar] [CrossRef]
- Zicari, M.A.; D’Aquino, L.; Paradiso, A.; Mastrolitti, S.; Tommasi, F. Effect of cerium on growth and antioxidant metabolism of Lemna minor L. Ecotoxicol. Environ. Saf. 2018, 163, 536–543. [Google Scholar] [CrossRef]
- Shen, F.; Wang, L.; Zhou, Q.; Huang, X. Effects of lanthanum on Microcystis aeruginosa: Attention to the changes in composition and content of cellular microcystins. Aquat. Toxicol. 2018, 196, 9–16. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Li, J.; Lü, Y.; Jin, H.; Deng, S.; Zeng, Y. Effects of cerium on growth and physiological characteristics of Anabaena flosaquae. J. Rare Earths 2012, 30, 1287–1292. [Google Scholar] [CrossRef]
- Edahbi, M.; Plante, B.; Benzaazoua, M. Environmental challenges and identification of the knowledge gaps associated with REE mine wastes management. J. Clean. Prod. 2019, 212, 1232–1241. [Google Scholar] [CrossRef]
- Bau, M.; Dulski, P. Anthropogenic origin of positive gadolinium anomalies in river waters. Earth Planet. Sci. Lett. 1996, 143, 245–255. [Google Scholar] [CrossRef]
- Bau, M.; Knappe, A.; Dulski, P. Anthropogenic gadolinium as a micropollutant in river waters in Pennsylvania and in Lake Erie, northeastern United States. Chem. Erde Geochem. 2006, 66, 143–152. [Google Scholar] [CrossRef]
- Goldstein, S.J.; Jacobsen, S.B. Rare earth elements in river waters. Earth Planet. Sci. Lett. 1988, 89, 35–47. [Google Scholar] [CrossRef]
- Han, G.; Liu, C.Q. Dissolved rare earth elements in river waters draining karst terrains in Guizhou Province, China. Aquat. Geochem. 2007, 13, 95–107. [Google Scholar] [CrossRef]
- Ingri, J.; Widerlund, A.; Land, M.; Gustafsson, Ö.; Andersson, P.; Öhlander, B. Temporal variations in the fractionation of the rare earth elements in a Boreal river; the role of colloidal particles. Chem. Geol. 2000, 166, 23–45. [Google Scholar] [CrossRef]
- Kharitonova, N.A.; Vakh, E.A. Rare earth elements in surface waters of Priamurye. Features of accumulation and fractionation. Tomsk State Univ. J. 2015, 232–244. [Google Scholar] [CrossRef]
- Neal, C. Rare earth element concentrations in dissolved and acid available participate forms for eastern UK rivers. Hydrol. Earth Syst. Sci. 2007, 11, 313–327. [Google Scholar] [CrossRef]
- Spears, B.M.; Lürling, M.; Yasseri, S.; Castro-Castellon, A.T.; Gibbs, M.; Meis, S.; McDonald, C.; McIntosh, J.; Sleep, D.; Van Oosterhout, F. Lake responses following lanthanum-modified bentonite clay (Phoslock®) application: An analysis of water column lanthanum data from 16 case study lakes. Water Res. 2013, 47, 5930–5942. [Google Scholar] [CrossRef] [PubMed]
- Weltje, L.; Heidenreich, H.; Zhu, W.; Wolterbeek, H.T.; Korhammer, S.; de Goeij, J.J.; Markert, B. Lanthanide concentrations in freshwater plants and molluscs, related to those in surface water, pore water and sediment: A case study in the Netherlands. Sci. Total Environ. 2002, 286, 191–214. [Google Scholar] [CrossRef]
- Rzymski, P.; Klimaszyk, P.; Niedzielski, P.; Marszelewski, W.; Borowiak, D.; Nowiński, K.; Baikenzheyeva, A.; Kurmanbayev, R.; Aladin, N. Pollution with trace elements and rare-earth metals in the lower course of Syr Darya River and Small Aral Sea, Kazakhstan. Chemosphere 2019, 234, 81–88. [Google Scholar] [CrossRef] [PubMed]
- Leybourne, M.I.; Johannesson, K.H. Rare earth elements (REE) and yttrium in stream waters, stream sediments, and Fe–Mn oxyhydroxides: Fractionation, speciation, and controls over REE + Y patterns in the surface environment. Geochim. Cosmochim. Acta 2008, 72, 5962–5983. [Google Scholar] [CrossRef]
- Neal, C.; Whitehead, P.G.; Jeffery, H.; Neal, M. The water quality of the River Carnon, west Cornwall, November 1992 to March 1994: The impacts of Wheal Jane discharges. Sci. Total Environ. 2005, 338, 23–39. [Google Scholar] [CrossRef]
- Chudaeva, V.A.; Chudaev, O.V. Accumulation and fractionation of rare earth elements in surface waters of the Russian Far East under the conditions of natural and anthropogenic anomalies. Geochem. Int. 2011, 49, 523–549. [Google Scholar] [CrossRef]
- Migaszewski, Z.M.; Gałuszka, A.; Dołęgowska, S. Rare earth and trace element signatures for assessing an impact of rock mining and processing on the environment: Wiśniówka case study, south-central Poland. Environ. Sci. Pollut. Res. 2016, 23, 24943–24959. [Google Scholar] [CrossRef]
- Noller, B.N. Non-radiological contaminants from uranium mining and milling at Ranger, Jabiru, Northern Territory, Australia. Environ. Monit. Assess. 1991, 19, 383–400. [Google Scholar] [CrossRef]
- Stauber, J.L.; Binet, M.T. Canning River Phoslock field trial —ecotoxicity testing final report, Centre for advanced analytical chemistry, CSIRO Energy technology, Bangor, NSW Australia: 2000. Available online: http://www.phoslock.com.au/site/content/scientificreport/Eco-Toxicity_Report_by_CSIRO_Australia_Stauber__Binet_2000_Canning_river_phoslock_field_trial_-_ecotoxicity_testing_final_report.pdf (accessed on 17 June 2019).
- Stewart, B.W.; Capo, R.C.; Hedin, B.C.; Hedin, R.S. Rare earth element resources in coal mine drainage and treatment precipitates in the Appalachian Basin, USA. Int. J. Coal Geol. 2017, 169, 28–39. [Google Scholar] [CrossRef]
- Van Koetsem, F.; Geremew, T.T.; Wallaert, E.; Verbeken, K.; Van der Meeren, P.; Du Laing, G. Fate of engineered nanomaterials in surface water: Factors affecting interactions of Ag and CeO2 nanoparticles with (re)suspended sediments. Ecol. Eng. 2015, 80, 140–150. [Google Scholar] [CrossRef]
- Booth, A.; Størseth, T.; Altin, D.; Fornara, A.; Ahniyaz, A.; Jungnickel, H.; Laux, P.; Luch, A.; Sørensen, L. Freshwater dispersion stability of PAA-stabilised cerium oxide nanoparticles and toxicity towards Pseudokirchneriella subcapitata. Sci. Total Environ. 2015, 505, 596–605. [Google Scholar] [CrossRef] [PubMed]
- Alam, B.; Philippe, A.; Rosenfeldt, R.R.; Seitz, F.; Dey, S.; Bundschuh, M.; Schaumann, G.E.; Brenner, S.A. Synthesis, characterization, and ecotoxicity of CeO2 nanoparticles with differing properties. J Nanopart. Res. 2016, 18, 303. [Google Scholar] [CrossRef]
- Goodhead, R.M.; Johnston, B.D.; Cole, P.A.; Baalousha, M.; Hodgson, D.; Iguchi, T.; Lead, J.R.; Tyler, C.R. Does natural organic matter increase the bioavailability of cerium dioxide nanoparticles to fish? Environ. Chem. 2015, 12, 673. [Google Scholar] [CrossRef]
- Zhang, Y.; Blewett, T.A.; Val, A.L.; Goss, G.G. UV-induced toxicity of cerium oxide nanoparticles (CeO2 NPs) and the protective properties of natural organic matter (NOM) from the Rio Negro Amazon River. Environ. Sci. Nano 2018, 5, 476–486. [Google Scholar] [CrossRef]
- Pulido-Reyes, G.; Rodea-Palomares, I.; Das, S.; Sakthivel, T.S.; Leganes, F.; Rosal, R.; Seal, S.; Fernández-Pinãs, F. Untangling the biological effects of cerium oxide nanoparticles: The role of surface valence states. Sci. Rep. 2015, 5, 15613. [Google Scholar] [CrossRef]
- El-Akl, P.; Smith, S.; Wilkinson, K.J. Linking the chemical speciation of cerium to its bioavailability in water for a freshwater alga. Environ. Toxicol. Chem. 2015, 34, 1711–1719. [Google Scholar] [CrossRef]
- Weltje, L.; Verhoof, L.R.C.W.; Verweij, W.; Hamers, T. Lutetium speciation and toxicity in a microbial bioassay: Testing the free-ion model for lanthanides. Environ. Sci. Technol. 2004, 38, 6597–6604. [Google Scholar] [CrossRef]
- Lürling, M.; Tolman, Y. Effects of lanthanum and lanthanum-modified clay on growth, survival and reproduction of Daphnia magna. Water Res. 2010, 44, 309–319. [Google Scholar] [CrossRef]
- Tang, H.; Wang, X.; Shuai, W.; Liu, Y. Immobilization of rare earth elements of the mine tailings using phosphates and lime. Procedia Environ. Sci. 2016, 31, 255–263. [Google Scholar] [CrossRef][Green Version]
- Tang, J.; Johannesson, K.H. Speciation of rare earth elements in natural terrestrial waters: Assessing the role of dissolved organic matter from the modeling approach. Geochim. Cosmochim. Acta 2003, 67, 2321–2339. [Google Scholar] [CrossRef]
- Johannesson, K.H.; Tang, J.; Daniels, J.M.; Bounds, W.J.; Burdige, D.J. Rare earth element concentrations and speciation in organic-rich blackwaters of the Great Dismal Swamp, Virginia, USA. Chem. Geol. 2004, 209, 271–294. [Google Scholar] [CrossRef]
- Rowell, J.A.; Fillion, M.A.; Smith, S.; Wilkinson, K.J. Determination of the speciation and bioavailability of samarium to Chlamydomonas reinhardtii in the presence of natural organic matter. Environ. Toxicol. Chem. 2018, 37, 1623–1631. [Google Scholar] [CrossRef] [PubMed]
- Tang, J.; Johannesson, K.H. Ligand extraction of rare earth elements from aquifer sediments: Implications for rare earth element complexation with organic matter in natural waters. Geochim. Cosmochim. Acta 2010, 74, 6690–6705. [Google Scholar] [CrossRef]
- Figueiredo, C.; Grilo, T.F.; Lopes, C.; Brito, P.; Diniz, M.; Caetano, M.; Rosa, R.; Raimundo, J. Accumulation, elimination and neuro-oxidative damage under lanthanum exposure in glass eels (Anguilla anguilla). Chemosphere 2018, 206, 414–423. [Google Scholar] [CrossRef] [PubMed]
- Yang, G.; Tan, Q.G.; Zhu, L.; Wilkinson, K.J. The role of complexation and competition in the biouptake of europium by a unicellular alga. Environ. Toxicol. Chem. 2014, 33, 2609–2615. [Google Scholar] [CrossRef] [PubMed]
- Ma, Y.; Wang, J.; Peng, C.; Ding, Y.; He, X.; Zhang, P.; Li, N.; Lan, T.; Wang, D.; Zhang, Z.; et al. Toxicity of cerium and thorium on Daphnia magna. Ecotoxicol. Environ. Saf. 2016, 134, 226–232. [Google Scholar] [CrossRef] [PubMed]
- Cotton, S. Lanthanide and Actinide Chemistry; John Wiley & Sons: Chippenham, UK, 2006. [Google Scholar]
- Eriksson, J.; Gilek, M.; Rudén, C. Regulating Chemical Risks: European and Global Challenge; Springer: Dordrecht, Netherlands, 2010. [Google Scholar]
- Merrington, G.; An, Y.; Grist, E.P.M.; Jeong, S.; Rattikansukha, C.; Roe, S.; Schneider, U.; Sthiannopkao, S.; Suter, G.W.; Dam, R.V.; et al. Water quality guidelines for chemicals: Learning lessons to deliver meaningful environmental metrics. Environ. Sci. Pollut. Res. 2014, 21, 6–16. [Google Scholar] [CrossRef]
- Vighi, M.; Villa, S. Ecotoxicology: The Challenges for the 21st Century. Toxics 2013, 1, 18–35. [Google Scholar] [CrossRef]
- Briffa, S.M.; Lynch, I.; Hapiuk, D.; Valsami-Jones, E. Physical and chemical transformations of zirconium doped ceria nanoparticles in the presence of phosphate: Increasing realism in environmental fate and behaviour experiments. Environ. Pollut. 2019, 252, 974–981. [Google Scholar] [CrossRef]
- Gaiser, B.K.; Fernandes, T.F.; Jepson, M.; Lead, J.R.; Tyler, C.R.; Stone, V. Assessing exposure, uptake and toxicity of silver and cerium dioxide nanoparticles from contaminated environments. Environ. Health 2009, 8, 1–4. [Google Scholar] [CrossRef]
- Johnston, B.D.; Scown, T.M.; Moger, J.; Cumberland, S.A.; Baalousha, M.; Linge, K.; Van Aerle, R.; Jarvis, K.; Lead, J.R.; Tyler, C.R. Bioavailability of nanoscale metal oxides TiO2, CeO2, and ZnO to fish. Environ. Sci. Technol. 2010, 44, 1144–1151. [Google Scholar] [CrossRef] [PubMed]
- Hirano, S.; Suzuki, K.T. Exposure, metabolism, and toxicity of rare earths and related compounds. Environ. Health Perspect. 1996, 104, 85–95. [Google Scholar] [CrossRef] [PubMed]
- Tai, P.; Zhao, Q.; Su, D.; Li, P.; Stagnitti, F. Biological toxicity of lanthanide elements on algae. Chemosphere 2010, 80, 1031–1035. [Google Scholar] [CrossRef] [PubMed]
- Haferburg, G.; Merten, D.; Büchel, G.; Kothe, E. Biosorption of metal and salt tolerant microbial isolates from a former uranium mining area. Their impact on changes in rare earth element patterns in acid mine drainage. J. Basic Microbiol. 2007, 47, 474–484. [Google Scholar] [CrossRef]
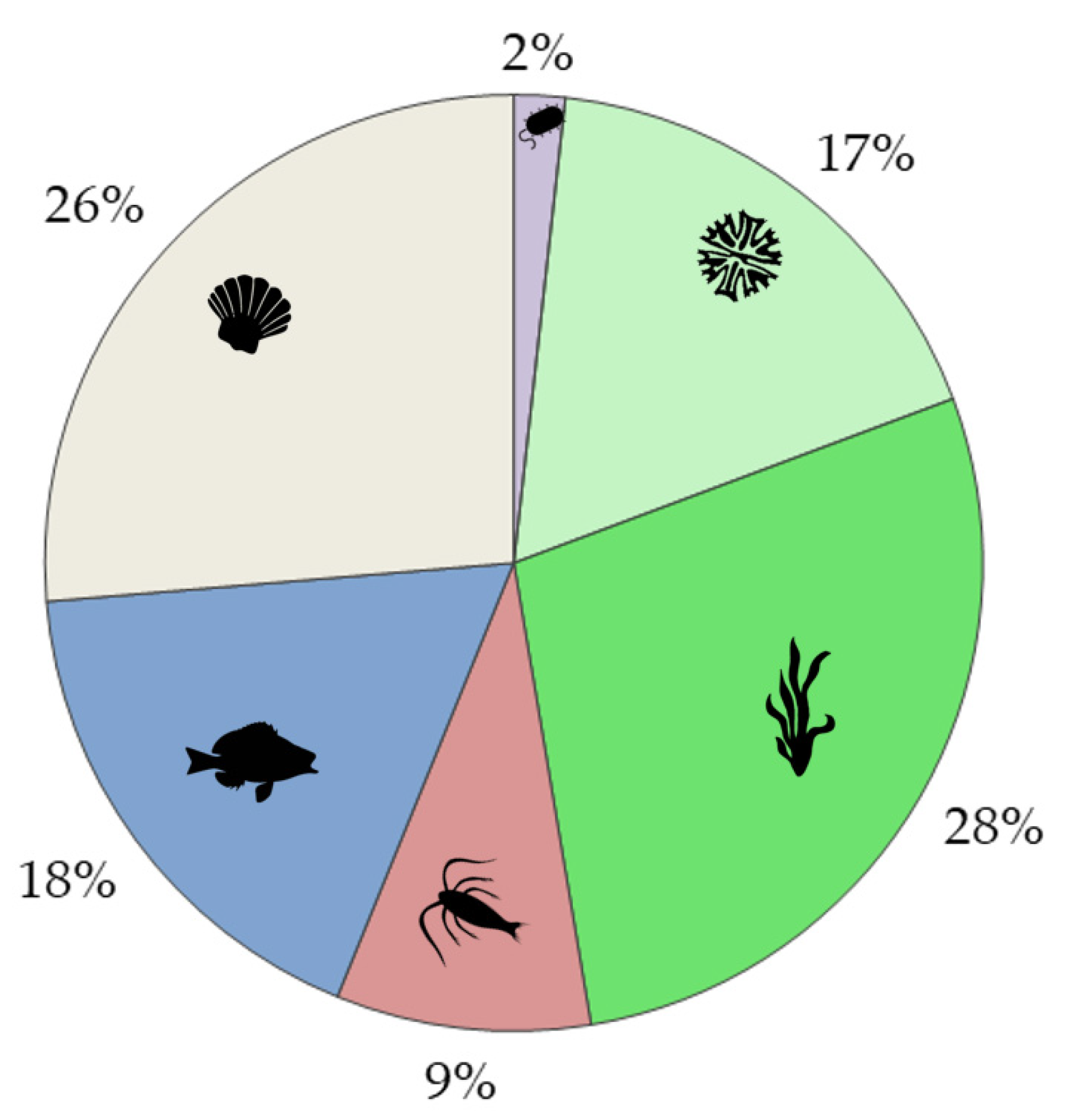
| Ecological Group | Ln, REE 8 | La | Ce | Pr | Nd | Sm | Eu | Gd | Tb | Dy | Ho | Er | Tm | Yb | Lu | Total * |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Microorganims 1 | 19 | 11 | 48 | 3 | 1 | 3 | 3 | 5 | 1 | 3 | 1 | 1 | 0 | 1 | 3 | 93 |
| Phytoplankton 2 | 21 | 13 | 28 | 1 | 0 | 4 | 2 | 5 | 1 | 0 | 1 | 1 | 1 | 0 | 1 | 58 |
| Macrophytes 3 | 13 | 6 | 5 | 1 | 1 | 0 | 0 | 0 | 0 | 0 | 0 | 1 | 0 | 1 | 0 | 17 |
| Zooplankton 4 | 13 | 7 | 14 | 0 | 0 | 1 | 0 | 2 | 0 | 1 | 1 | 1 | 0 | 0 | 1 | 28 |
| Nekton 5 | 17 | 13 | 22 | 1 | 0 | 1 | 2 | 3 | 0 | 0 | 1 | 1 | 0 | 0 | 1 | 41 |
| Benthos 6 | 17 | 8 | 7 | 0 | 0 | 2 | 1 | 3 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 24 |
| Bioaccumulation 7 | 37 | 12 | 20 | 1 | 0 | 2 | 3 | 2 | 0 | 0 | 0 | 1 | 1 | 1 | 0 | 51 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Blinova, I.; Muna, M.; Heinlaan, M.; Lukjanova, A.; Kahru, A. Potential Hazard of Lanthanides and Lanthanide-Based Nanoparticles to Aquatic Ecosystems: Data Gaps, Challenges and Future Research Needs Derived from Bibliometric Analysis. Nanomaterials 2020, 10, 328. https://doi.org/10.3390/nano10020328
Blinova I, Muna M, Heinlaan M, Lukjanova A, Kahru A. Potential Hazard of Lanthanides and Lanthanide-Based Nanoparticles to Aquatic Ecosystems: Data Gaps, Challenges and Future Research Needs Derived from Bibliometric Analysis. Nanomaterials. 2020; 10(2):328. https://doi.org/10.3390/nano10020328
Chicago/Turabian StyleBlinova, Irina, Marge Muna, Margit Heinlaan, Aljona Lukjanova, and Anne Kahru. 2020. "Potential Hazard of Lanthanides and Lanthanide-Based Nanoparticles to Aquatic Ecosystems: Data Gaps, Challenges and Future Research Needs Derived from Bibliometric Analysis" Nanomaterials 10, no. 2: 328. https://doi.org/10.3390/nano10020328
APA StyleBlinova, I., Muna, M., Heinlaan, M., Lukjanova, A., & Kahru, A. (2020). Potential Hazard of Lanthanides and Lanthanide-Based Nanoparticles to Aquatic Ecosystems: Data Gaps, Challenges and Future Research Needs Derived from Bibliometric Analysis. Nanomaterials, 10(2), 328. https://doi.org/10.3390/nano10020328





