Effects of Buffer Gases on Graphene Flakes Synthesis in Thermal Plasma Process at Atmospheric Pressure
Abstract
1. Introduction
2. Experimental Methods
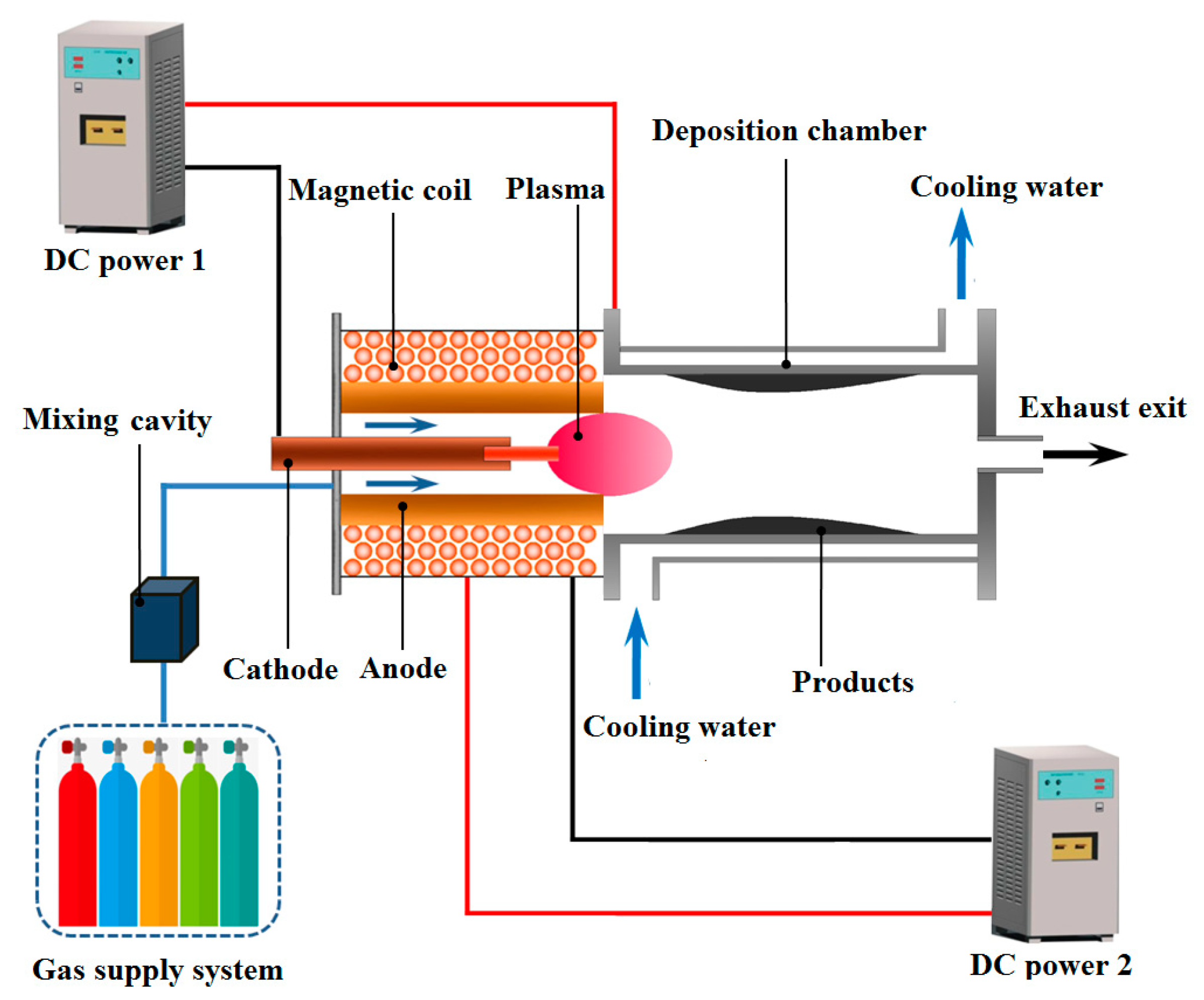
2.1. Experimental Apparatus
2.2. Experimental Parameters
2.3. Characterization
3. Results
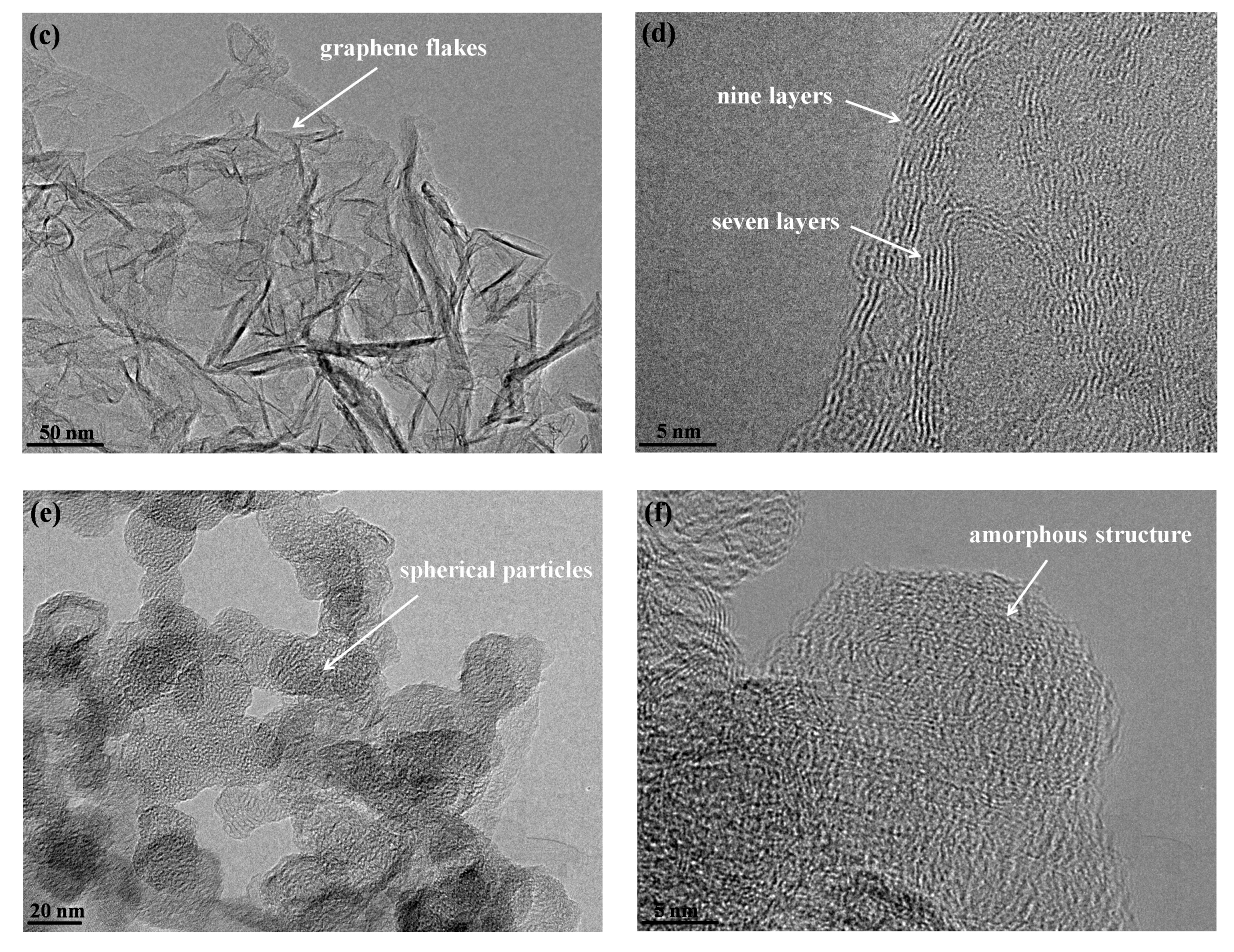
3.1. TEM Images
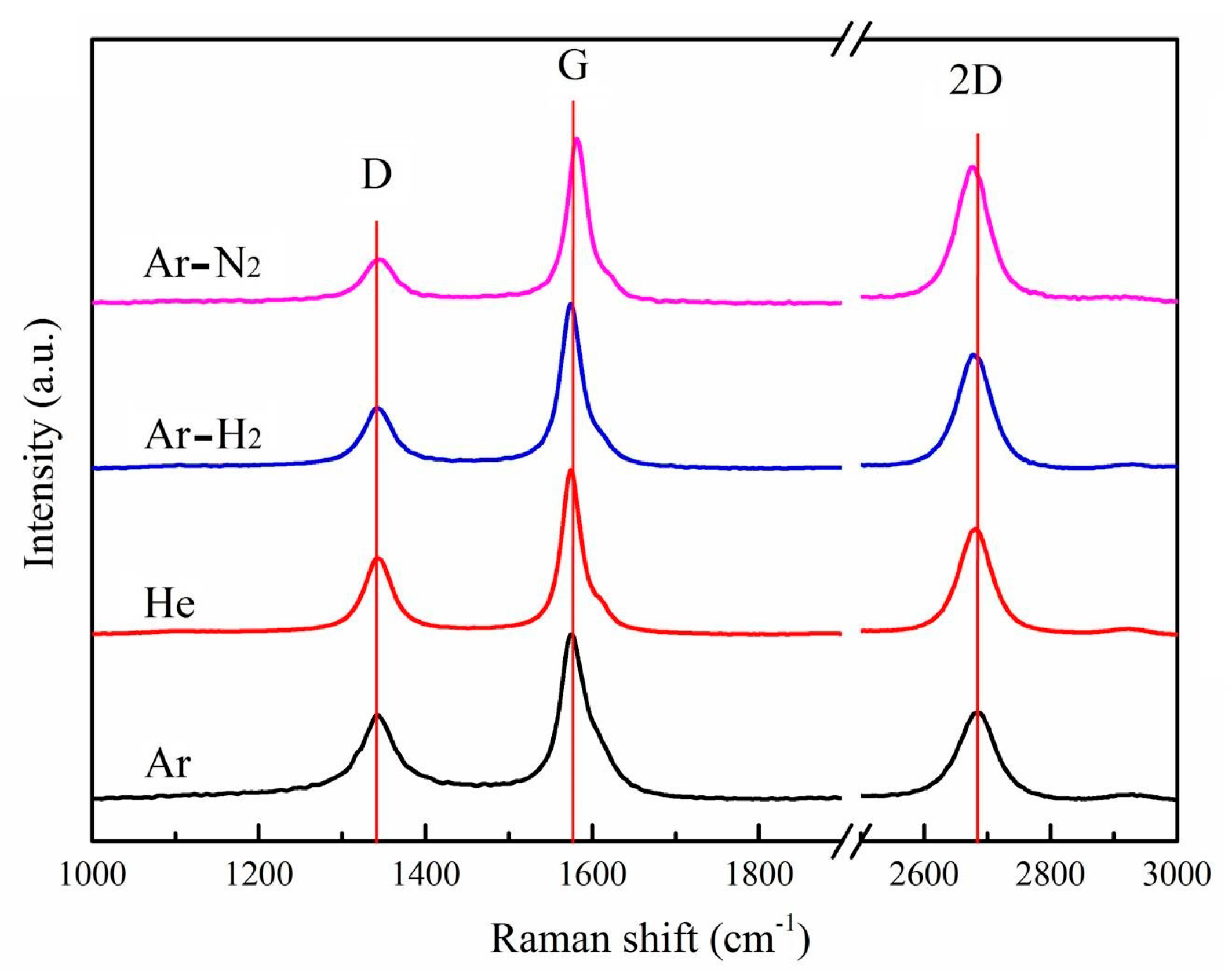
3.2. Raman Spectroscopy
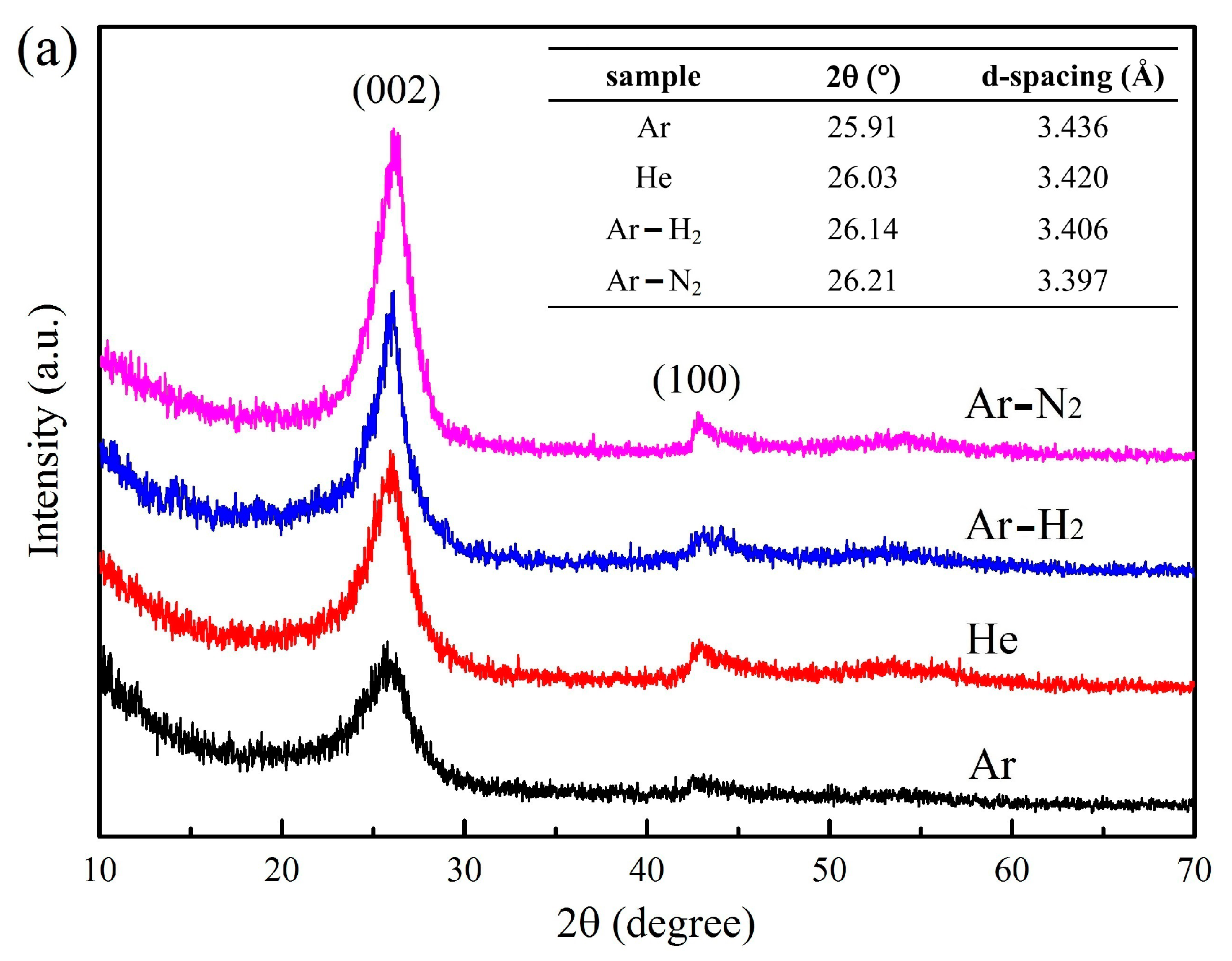
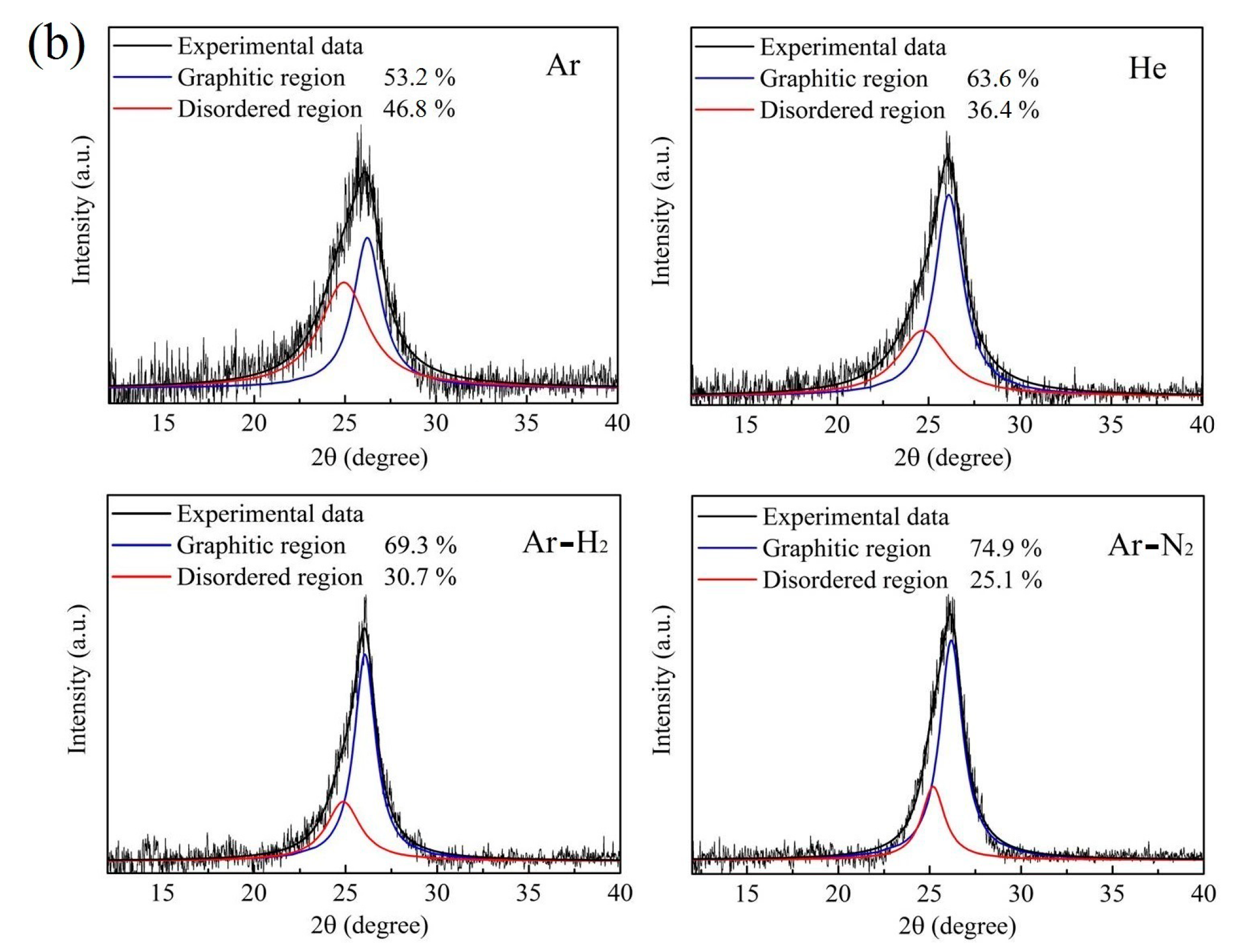
3.3. XRD Patterns
3.4. XPS Spectra
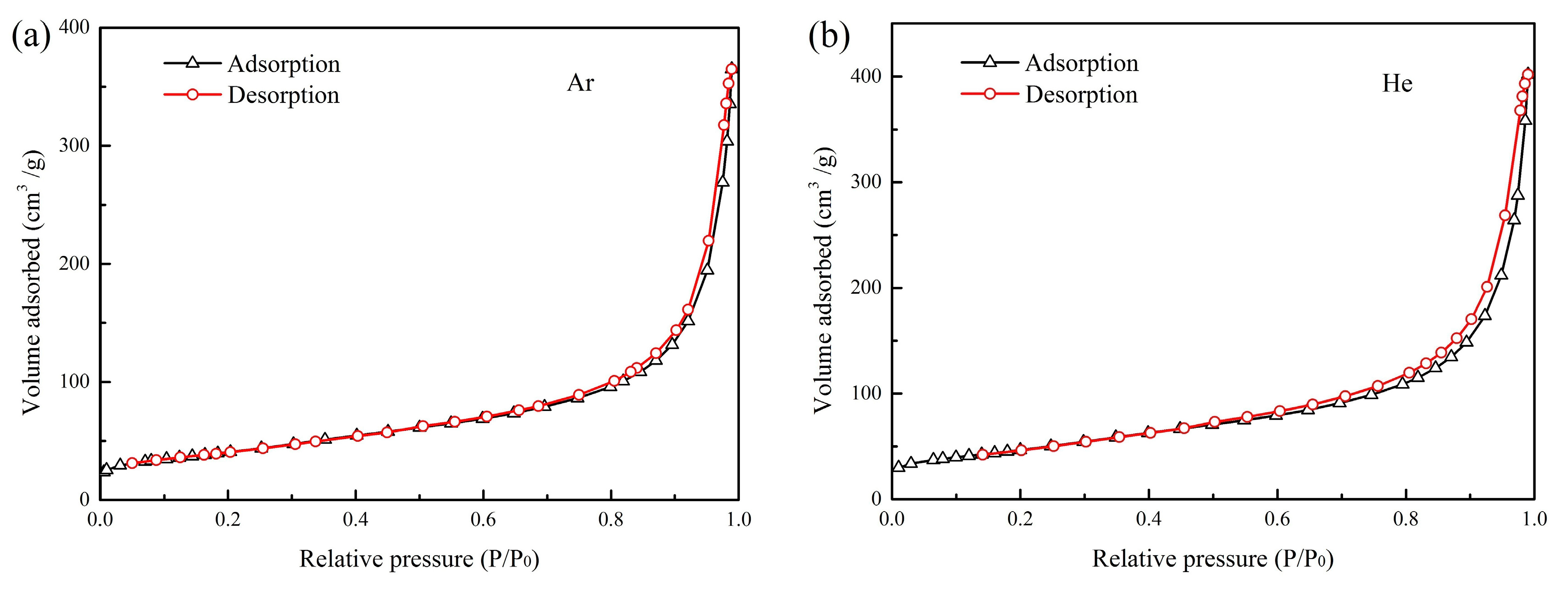
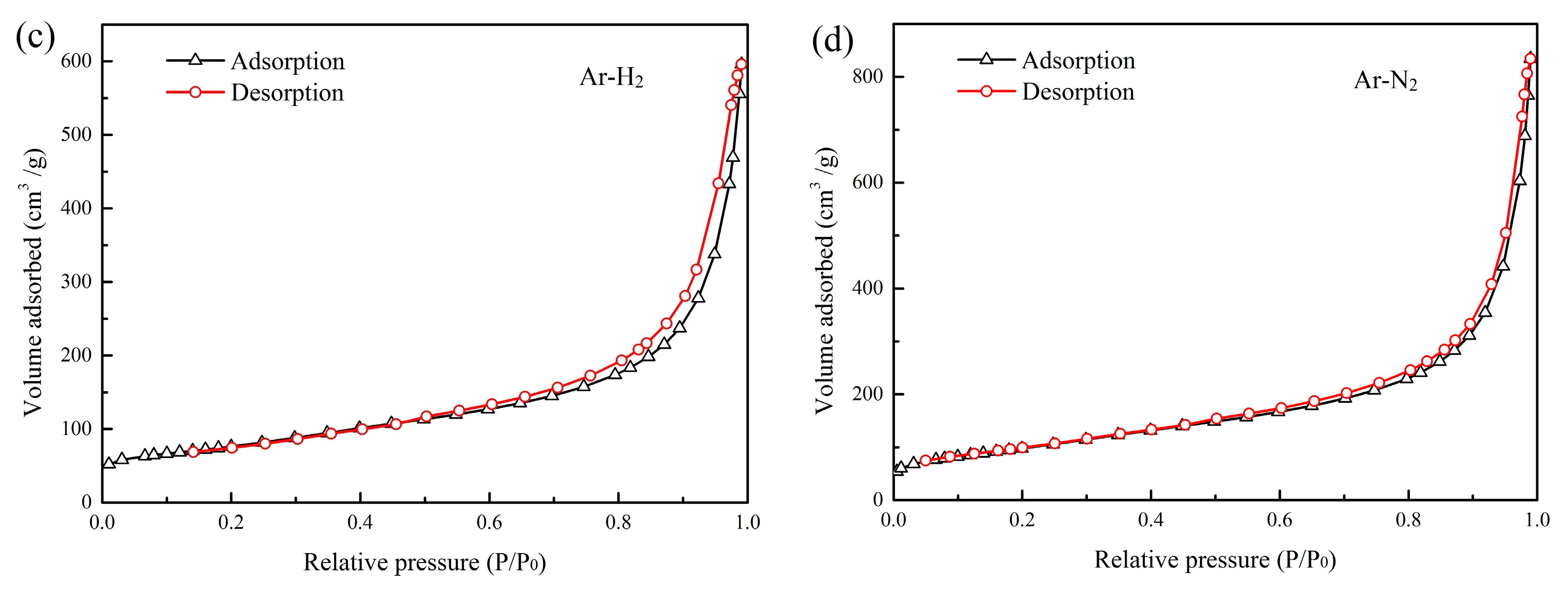
3.5. BET Surface Area
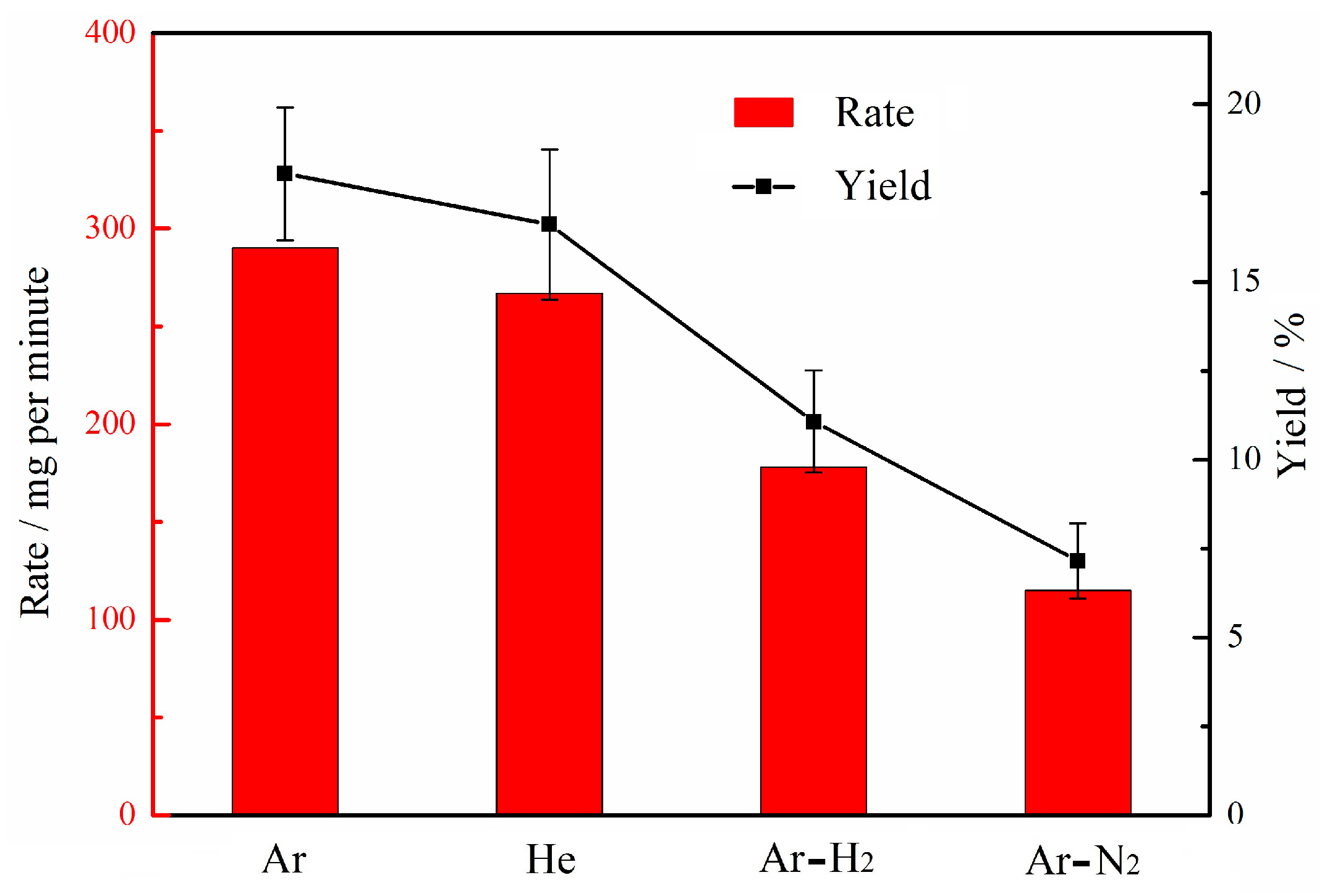
3.6. Synthesis Rate/Yield
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Novoselov, K.S.; Geim, A.K.; Morozov, S.V.; Jiang, D.; Zhang, Y.; Dubonos, S.V.; Grigorieva, I.V.; Firsov, A.A. Electric Field Effect in Atomically Thin Carbon Films. Science 2004, 306, 666–669. [Google Scholar] [CrossRef]
- Page, A.; Ding, F.; Irle, S.; Morokuma, K. Insights into Carbon Nanotube and Graphene Formation Mechanisms from Molecular Simulations: A Review. Rep. Prog. Phys. 2015, 78, 036501. [Google Scholar] [CrossRef]
- Zhu, Y.; Ji, H.; Cheng, H.-M.; Ruoff, R.S. Mass Production and Industrial Applications of Graphene Materials. Natl. Sci. Rev. 2017, 5, 90–101. [Google Scholar] [CrossRef]
- Chun, L.; Gaoquan, S. Three-Dimensional Graphene Architectures. Nanoscale 2012, 4, 5549–5563. [Google Scholar]
- Reina, A.; Jia, X.; Ho, J.; Nezich, D.; Son, H.; Bulovic, V.; Dresselhaus, M.S.; Kong, J. Large Area, Few-Layer Graphene Films on Arbitrary Substrates by Chemical Vapor Deposition. Nano Lett. 2008, 9, 30–35. [Google Scholar] [CrossRef]
- Malesevic, A.; Vitchev, R.; Schouteden, K.; Volodin, A.; Zhang, L.; Van Tendeloo, G.; Vanhulsel, A.; Van Haesendonck, C. Synthesis of Few-Layer Graphene Via Microwave Plasma-Enhanced Chemical Vapour Deposition. Nanotechnology 2008, 19, 305604. [Google Scholar] [CrossRef]
- Jernigan, G.G.; VanMil, B.L.; Tedesco, J.L.; Tischler, J.G.; Glaser, E.R.; Davidson, A., III; Campbell, P.M.; Gaskill, D.K. Comparison of Epitaxial Graphene on Si-Face and C-Face 4h Sic Formed by Ultrahigh Vacuum and Rf Furnace Production. Nano Lett. 2009, 9, 2605–2609. [Google Scholar] [CrossRef]
- Emtsev, K.V.; Bostwick, A.; Horn, K.; Jobst, J.; Kellogg, G.L.; Ley, L.; McChesney, J.L.; Ohta, T.; Reshanov, S.A.; Röhrl, J. Towards Wafer-Size Graphene Layers by Atmospheric Pressure Graphitization of Silicon Carbide. Nat. Mater. 2009, 8, 203–207. [Google Scholar] [CrossRef] [PubMed]
- Zhu, Y.; Murali, S.; Cai, W.; Li, X.; Suk, J.W.; Potts, J.R.; Ruoff, R.S. Graphene and Graphene Oxide: Synthesis, Properties, and Applications. Adv. Mater. 2010, 22, 3906–3924. [Google Scholar] [CrossRef] [PubMed]
- Subrahmanyam, K.; Panchakarla, L.; Govindaraj, A.; Rao, C. Simple Method of Preparing Graphene Flakes by an Arc-Discharge Method. J. Phys. Chem. C 2009, 113, 4257–4259. [Google Scholar] [CrossRef]
- Kim, K.S.; Hong, S.H.; Lee, K.-S.; Ju, W.T. Continuous Synthesis of Nanostructured Sheetlike Carbons by Thermal Plasma Decomposition of Methane. IEEE Trans. Plasma Sci. 2007, 35, 434–443. [Google Scholar] [CrossRef]
- Kim, J.; Heo, S.B.; Gu, G.H.; Suh, J.S. Fabrication of Graphene Flakes Composed of Multi-Layer Graphene Sheets Using a Thermal Plasma Jet System. Nanotechnology 2010, 21, 095601. [Google Scholar] [CrossRef]
- Shavelkina, M.; Amirov, R.; Bilera, I. Formation of Carbon Nanostructures by the Plasma Jets: Synthesis, Characterization, Application. Mater. Today Proc. 2018, 5, 25956–25961. [Google Scholar] [CrossRef]
- Dato, A. Graphene Synthesized in Atmospheric Plasmas—A Review. J. Mater. Res. 2019, 34, 214–230. [Google Scholar] [CrossRef]
- Dato, A.; Frenklach, M. Substrate-Free Microwave Synthesis of Graphene: Experimental Conditions and Hydrocarbon Precursors. NewJ. Phys. 2010, 12, 125013. [Google Scholar] [CrossRef]
- Dato, A.; Lee, Z.; Jeon, K.-J.; Erni, R.; Radmilovic, V.; Richardson, T.J.; Frenklach, M. Clean and Highly Ordered Graphene Synthesized in the Gas Phase. Chem. Commun. 2009, 40, 6095–6097. [Google Scholar] [CrossRef] [PubMed]
- Dato, A.; Radmilovic, V.; Lee, Z.; Phillips, J.; Frenklach, M. Substrate-Free Gas-Phase Synthesis of Graphene Sheets. Nano Lett. 2008, 8, 2012–2016. [Google Scholar] [CrossRef]
- Tatarova, E.; Henriques, J.; Luhrs, C.; Dias, A.; Phillips, J.; Abrashev, M.; Ferreira, C. Microwave Plasma Based Single Step Method for Free Standing Graphene Synthesis at Atmospheric Conditions. Appl. Phys. Lett. 2013, 103, 134101. [Google Scholar] [CrossRef]
- Tatarova, E.; Dias, A.; Henriques, J.; do Rego, A.B.; Ferraria, A.; Abrashev, M.; Luhrs, C.C.; Phillips, J.; Dias, F.; Ferreira, C. Microwave Plasmas Applied for the Synthesis of Free Standing Graphene Sheets. J. Phys. D Appl. Phys. 2014, 47, 385501. [Google Scholar] [CrossRef]
- Bundaleska, N.; Tsyganov, D.; Dias, A.; Felizardo, E.; Henriques, J.; Dias, F.; Abrashev, M.; Kissovski, J.; Tatarova, E. Microwave Plasma Enabled Synthesis of Free Standing Carbon Nanostructures at Atmospheric Pressure Conditions. Phys. Chem. Chem. Phys. 2018, 20, 13810–13824. [Google Scholar] [CrossRef]
- Melero, C.; Rincón, R.; Muñoz, J.; Zhang, G.; Sun, S.; Perez, A.; Royuela, O.; González-Gago, C.; Calzada, M. Scalable Graphene Production from Ethanol Decomposition by Microwave Argon Plasma Torch. Plasma Phys.Control.Fusion 2017, 60, 014009. [Google Scholar] [CrossRef]
- Rincón, R.; Melero, C.; Jiménez, M.; Calzada, M. Synthesis of Multi-Layer Graphene and Multi-Wall Carbon Nanotubes from Direct Decomposition of Ethanol by Microwave Plasma without Using Metal Catalysts. Plasma Sources Sci. Technol. 2015, 24, 032005. [Google Scholar] [CrossRef]
- Pristavita, R.; Mendoza-Gonzalez, N.-Y.; Meunier, J.-L.; Berk, D. Carbon Blacks Produced by Thermal Plasma: The Influence of the Reactor Geometry on the Product Morphology. Plasma Chem.Plasma Process. 2010, 30, 267–279. [Google Scholar] [CrossRef]
- Pristavita, R.; Mendoza-Gonzalez, N.-Y.; Meunier, J.-L.; Berk, D. Carbon Nanoparticle Production by Inductively Coupled Thermal Plasmas: Controlling the Thermal History of Particle Nucleation. Plasma Chem.Plasma Process. 2011, 31, 851–866. [Google Scholar] [CrossRef]
- Mohanta, A.; Lanfant, B.; Asfaha, M.; Leparoux, M. Methane Dissociation Process in Inductively Coupled Ar/H2/Ch4 Plasma for Graphene Nano-Flakes Production. Appl. Phys. Lett. 2017, 110, 093109. [Google Scholar] [CrossRef]
- Pristavita, R.; Meunier, J.-L.; Berk, D. Carbon Nano-Flakes Produced by an Inductively Coupled Thermal Plasma System for Catalyst Applications. Plasma Chem. Plasma Process. 2011, 31, 393–403. [Google Scholar] [CrossRef]
- Meunier, J.-L.; Mendoza-Gonzalez, N.-Y.; Pristavita, R.; Binny, D.; Berk, D. Two-Dimensional Geometry Control of Graphene Nanoflakes Produced by Thermal Plasma for Catalyst Applications. Plasma Chem. Plasma Process. 2014, 34, 505–521. [Google Scholar] [CrossRef]
- Mohanta, A.; Lanfant, B.; Leparoux, M. Induction Plasma Synthesis of Graphene Nano-Flakes with in Situ Investigation of Ar–H2–CH4 Plasma by Optical Emission Spectroscopy. Plasma Chem. Plasma Process. 2019, 39, 1161–1179. [Google Scholar] [CrossRef]
- Zhang, H.; Cao, T.; Cheng, Y. Preparation of Few-Layer Graphene Nanosheets by Radio-Frequency Induction Thermal Plasma. Carbon 2015, 86, 38–45. [Google Scholar] [CrossRef]
- Lee, M.W.; Kim, H.-Y.; Yoon, H.; Kim, J.; Suh, J.S. Fabrication of Dispersible Graphene Flakes Using Thermal Plasma Jet and Their Thin Films for Solar Cells. Carbon 2016, 106, 48–55. [Google Scholar] [CrossRef]
- Wang, C.; Sun, L.; Dai, X.; Li, D.; Chen, X.; Xia, W.; Xia, W. Continuous Synthesis of Graphene Nano-Flakes by Magnetically Rotating Arc at Atmospheric Pressure. Carbon 2019, 148, 394–402. [Google Scholar] [CrossRef]
- Chen, X.; Wang, C.; Song, M.; Ma, J.; Ye, T.; Xia, W. The Morphological Transformation of Carbon Materials from Nanospheres to Graphene Nanoflakes by Thermal Plasmas. Carbon 2019, 155, 521–530. [Google Scholar] [CrossRef]
- Wang, C.; Sun, L.; Chen, X.; Song, M.; Xia, W. Products on Electrodes in an Argon-Methane Magnetically Rotating Arc at Atmospheric Pressure. Fuller. Nanotub. Carbon Nanostruct. 2019, 27, 498–505. [Google Scholar] [CrossRef]
- Shen, B.; Ding, J.; Yan, X.; Feng, W.; Li, J.; Xue, Q. Influence of Different Buffer Gases on Synthesis of Few-Layered Graphene by Arc Discharge Method. Appl. Surf. Sci. 2012, 258, 4523–4531. [Google Scholar] [CrossRef]
- Li, N.; Wang, Z.; Zhao, K.; Shi, Z.; Gu, Z.; Xu, S. Large Scale Synthesis of N-Doped Multi-Layered Graphene Sheets by Simple Arc-Discharge Method. Carbon 2010, 48, 255–259. [Google Scholar] [CrossRef]
- Li, B.; Nan, Y.; Cao, H.; Yan, P.; Zhao, S.; Zhao, X.; Song, X. Arc Plasma-Assisted Hydrogenation of Few-Layer Graphene in Methane-Hydrogen Atmospheres. Diam. Relat. Mater. 2017, 76, 44–49. [Google Scholar] [CrossRef]
- Li, B.; Song, X.; Zhang, P. Raman-Assessed Structural Evolution of as-Deposited Few-Layer Graphene by He/H2 Arc Discharge During Rapid-Cooling Thinning Treatment. Carbon 2014, 66, 426–435. [Google Scholar] [CrossRef]
- Qin, B.; Zhang, T.; Chen, H.; Ma, Y. The Growth Mechanism of Few-Layer Graphene in the Arc Discharge Process. Carbon 2016, 102, 494–498. [Google Scholar] [CrossRef]
- Zhang, D.; Ye, K.; Yao, Y.; Liang, F.; Qu, T.; Ma, W.; Yang, B.; Dai, Y.; Watanabe, T. Controllable Synthesis of Carbon Nanomaterials by Direct Current Arc Discharge from the Inner Wall of the Chamber. Carbon 2019, 142, 278–284. [Google Scholar] [CrossRef]
- Sedelnikova, O.; Fedoseeva, Y.V.; Romanenko, A.; Gusel‘nikov, A.; Vilkov, O.Y.; Maksimovskiy, E.; Bychanok, D.; Kuzhir, P.; Bulusheva, L.; Okotrub, A. Effect of Boron and Nitrogen Additives on Structure and Transport Properties of Arc-Produced Carbon. Carbon 2019, 143, 660–668. [Google Scholar] [CrossRef]
- Dacheng, W.; Yunqi, L.; Yu, W.; Hongliang, Z.; Liping, H.; Gui, Y. Synthesis of N-Doped Graphene by Chemical Vapor Deposition and Its Electrical Properties. Nano Lett. 2009, 9, 1752. [Google Scholar]
- Ivan, V.; Murari, R.; Pasquale, F.; Sheng, D.; Panos, D.; Gyula, E.; Sergei, S. Role of Hydrogen in Chemical Vapor Deposition Growth of Large Single-Crystal Graphene. ACS Nano 2011, 5, 6069–6076. [Google Scholar]
- Lin, L.; Deng, B.; Sun, J.; Peng, H.; Liu, Z. Bridging the Gap between Reality and Ideal in Chemical Vapor Deposition Growth of Graphene. Chem. Rev. 2018, 118, 9281–9343. [Google Scholar] [CrossRef] [PubMed]
- Wang, C.; Li, W.W.; Zhang, X.N.; Liao, M.R.; Zha, J.; Xia, W.D. Observation of Thermal Cathodic Hot Spots in a Magnetically Rotating Arc Plasma Generator. IEEE Trans. Plasma Sci. 2015, 43, 3716–3720. [Google Scholar] [CrossRef]
- Wang, C.; Li, J.; Zhang, Z.; Ye, L.; Xia, W.; Xia, W. An Experimental Investigation of Cathode Spot Motion in a Magnetically Rotating Arc Plasma Generator at Atmospheric Pressure. Plasma Chem. Plasma Process. 2019, 39, 259–276. [Google Scholar] [CrossRef]
- Wang, C.; Sun, Q.; Sun, L.; Lu, Z.; Xia, W.; Xia, W. Spot and Diffuse Mode of Cathode Attachments in a Magnetically Rotating Arc Plasma Generator at Atmospheric Pressure. J. Appl. Phys. 2019, 125, 033301. [Google Scholar] [CrossRef]
- Vander Wal, R.L.; Tomasek, A.J.; Street, K.; Hull, D.R.; Thompson, W.K. Carbon Nanostructure Examined by Lattice Fringe Analysis of High-Resolution Transmission Electron Microscopy Images. Appl. Spectrosc. 2004, 58, 230–237. [Google Scholar] [CrossRef]
- Song, X.; Liu, Y.; Zhu, J. Synthesis of Polyhedral Graphite in a Forced Flow Arc Discharge. Mater. Lett. 2007, 61, 4781–4783. [Google Scholar] [CrossRef]
- Singh, M.; Sengupta, A.; Zeller, K.; Skoptsov, G.; Vander Wal, R.L. Effect of Hydrogen Concentration on Graphene Synthesis Using Microwave-Driven Plasma-Mediated Methane Cracking. Carbon 2019, 143, 802–813. [Google Scholar] [CrossRef]
- Zielinski, T.; Kijenski, J. Plasma Carbon Black—The New Active Additive for Plastics. Compos. Part A Appl. Sci.Manuf. 2005, 36, 467–471. [Google Scholar] [CrossRef]
- Gonzalez-Aguilar, J.; Moreno, M.; Fulcheri, L. Carbon Nanostructures Production by Gas-Phase Plasma Processes at Atmospheric Pressure. J. Phys. D Appl. Phys. 2007, 40, 2361–2374. [Google Scholar] [CrossRef]
- Moreno-Couranjou, M.; Monthioux, M.; Gonzalez-Aguilar, J.; Fulcheri, L. A Non-Thermal Plasma Process for the Gas Phase Synthesis of Carbon Nanoparticles. Carbon 2009, 47, 2310–2321. [Google Scholar] [CrossRef]
- Wang, C.; Lu, Z.; Li, D.; Xia, W.; Xia, W. Effect of the Magnetic Field on the Magnetically Stabilized Gliding Arc Discharge and Its Application in the Preparation of Carbon Black Nanoparticles. Plasma Chem. Plasma Process. 2018, 38, 1223–1238. [Google Scholar] [CrossRef]
- Fabry, F.; Flamant, G.; Fulcheri, L. Carbon Black Processing by Thermal Plasma. Analysis of the Particle Formation Mechanism. Chem. Eng. Sci. 2001, 56, 2123–2132. [Google Scholar] [CrossRef]
- Wang, F.; Hong, R. Continuous Preparation of Structure-Controlled Carbon Nanoparticle Via Arc Plasma and the Reinforcement of Polymeric Composites. Chem. Eng. J. 2017, 328, 1098–1111. [Google Scholar] [CrossRef]
- Nemanich, R.J.; Solin, S. First-and Second-Order Raman Scattering from Finite-Size Crystals of Graphite. Phys. Rev. B 1979, 20, 392–401. [Google Scholar] [CrossRef]
- Livneh, T.; Haslett, T.L.; Moskovits, M. Distinguishing Disorder-Induced Bands from Allowed Raman Bands in Graphite. Phys. Rev. B 2002, 66, 195110. [Google Scholar] [CrossRef]
- Ni, Z.; Wang, Y.; Yu, T.; Shen, Z. Raman Spectroscopy and Imaging of Graphene. Nano Res. 2008, 1, 273–291. [Google Scholar] [CrossRef]
- Sun, D.; Hong, R.; Liu, J.; Wang, F.; Wang, Y. Preparation of Carbon Nanomaterials Using Two-Group Arc Discharge Plasma. Chem. Eng. J. 2016, 303, 217–230. [Google Scholar] [CrossRef]
- Ferrari, A.C.; Basko, D.M. Raman Spectroscopy as a Versatile Tool for Studying the Properties of Graphene. Nat. Nanotech. 2013, 8, 235–246. [Google Scholar] [CrossRef]
- Ferrari, A.; Rodil, S.; Robertson, J. Interpretation of Infrared and Raman Spectra of Amorphous Carbon Nitrides. Phys. Rev. B 2003, 67, 155306. [Google Scholar] [CrossRef]
- Jiang, Y.; Chowdhury, S.; Balasubramanian, R. New Insights into the Role of Nitrogen-Bonding Configurations in Enhancing the Photocatalytic Activity of Nitrogen-Doped Graphene Aerogels. J. Colloid Interface Sci. 2019, 534, 574–585. [Google Scholar] [CrossRef] [PubMed]
- Shi, Z.; Kutana, A.; Yakobson, B.I. How Much N-Doping Can Graphene Sustain? J. Phys. Chem. Lett. 2015, 6, 106–112. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.; Hong, R.; Feng, W.; Badami, D. Synthesis of Structure Controlled Carbon Nanomaterials by Ac Arc Plasma Process. Powder Technol. 2014, 256, 158–165. [Google Scholar] [CrossRef]
- Cao, B.; Liu, H.; Xu, B.; Lei, Y.; Chen, X.; Song, H. Mesoporous Soft Carbon as an Anode Material for Sodium Ion Batteries with Superior Rate and Cycling Performance. J. Mater. Chem. A 2016, 4, 6472–6478. [Google Scholar] [CrossRef]
- Ji, C.; Yao, B.; Li, C.; Shi, G. An Improved Hummers Method for Eco-Friendly Synthesis of Graphene Oxide. Carbon 2013, 64, 225–229. [Google Scholar]
- Yuan, H.; Yan, F.; Li, C.; Zhu, C.; Zhang, X.; Chen, Y. Nickel Nanoparticle Encapsulated in Few-Layer Nitrogen-Doped Graphene Supported by Nitrogen-Doped Graphite Sheets as a High-Performance Electromagnetic Wave Absorbing Material. Acs Appl. Mater. Interfaces 2017, 10, 1399–1407. [Google Scholar] [CrossRef]
- Zhou, Y.; Wang, N.; Muhammad, J.; Wang, D.; Duan, Y.; Zhang, X.; Dong, X.; Zhang, Z. Graphene Nanoflakes with Optimized Nitrogen Doping Fabricated by Arc Discharge Method as Highly Efficient Absorbers toward Microwave Absorption. Carbon 2019, 148, 204–213. [Google Scholar] [CrossRef]
- Zafar, Z.; Zhen, H.N.; Xing, W.; Zhi, X.S.; Hai, Y.N.; Jing, B.; Li, T.S. Evolution of Raman Spectra in Nitrogen Doped Graphene. Carbon 2013, 61, 57–62. [Google Scholar] [CrossRef]
- Wang, H.; Maiyalagan, T.; Wang, X. Review on Recent Progress in Nitrogen-Doped Graphene: Synthesis, Characterization, and Its Potential Applications. Acs Catal. 2012, 2, 781–794. [Google Scholar] [CrossRef]
- Li, O.L.; Chiba, S.; Wada, Y.; Panomsuwan, G.; Ishizaki, T. Synthesis of Graphitic-N and Amino-N in Nitrogen-Doped Carbon Via a Solution Plasma Process and Exploration of Their Synergic Effect for Advanced Oxygen Reduction Reaction. J. Mater. Chem. A 2017, 5, 2073–2082. [Google Scholar] [CrossRef]
- Ma, L.-P.; Wu, Z.-S.; Li, J.; Wu, E.-D.; Ren, W.-C.; Cheng, H.-M. Hydrogen Adsorption Behavior of Graphene above Critical Temperature. Int. J. Hydrogen Energy 2009, 34, 2329–2332. [Google Scholar] [CrossRef]
- Zhou, H.; Watanabe, J.; Miyake, M.; Ogino, A.; Nagatsu, M.; Zhan, R. Optical and Mass Spectroscopy Measurements of Ar/Ch4/H2 Microwave Plasma for Nano-Crystalline Diamond Film Deposition. Diam. Relat. Mater. 2007, 16, 675–678. [Google Scholar] [CrossRef]
- Gordillo-Vázquez, F.J.; Albella, J. Distinct Nonequilibrium Plasma Chemistry of C 2 Affecting the Synthesis of Nanodiamond Thin Films from C 2 H 2 (1%)/H 2/Ar-Rich Plasmas. J. Appl. Phys. 2003, 94, 6085–6090. [Google Scholar] [CrossRef]
- Wu, A.; Li, X.; Yan, J.; Yang, J.; Du, C.; Zhu, F.; Qian, J. Co-Generation of Hydrogen and Carbon Aerosol from Coalbed Methane Surrogate Using Rotating Gliding Arc Plasma. Appl. Energy 2017, 195, 67–79. [Google Scholar] [CrossRef]
- Zhang, H.; Du, C.; Wu, A.; Bo, Z.; Yan, J.; Li, X. Rotating Gliding Arc Assisted Methane Decomposition in Nitrogen for Hydrogen Production. Int. J. Hydrogen Energy 2014, 39, 12620–12635. [Google Scholar] [CrossRef]
- Whitesides, R.; Frenklach, M. Detailed Kinetic Monte Carlo Simulations of Graphene-Edge Growth. J. Phys. Chem. A 2009, 114, 689–703. [Google Scholar] [CrossRef]
- Wang, X.; Lin, X.; Mesleh, M.; Jarrold, M.; Dravid, V.; Ketterson, J.; Chang, R. The Effect of Hydrogen on the Formation of Carbon Nanotubes and Fullerenes. J. Mater. Res. 1995, 10, 1977–1983. [Google Scholar] [CrossRef]
- Xie, W.G.; Jian, C.; Chen, J.; Ming, W.W.; Deng, S.Z.; Xu, N.S. Study on Effect of Hydrogen Treatment on Amorphous Carbon Film Using Scanning Probe Microscopy. Ultramicroscopy 2009, 109, 451–456. [Google Scholar] [CrossRef]
- Wang, X.; Lin, X.; Dravid, V.; Ketterson, J.; Chang, R.P. Carbon Nanotubes Synthesized in a Hydrogen Arc Discharge. Appl. Phys. Lett. 1995, 66, 2430–2432. [Google Scholar] [CrossRef]
- Sun, L.; Wang, C.; Zhou, Y.; Zhang, X.; Cai, B.; Qiu, J. Flowing Nitrogen Assisted-Arc Discharge Synthesis of Nitrogen-Doped Single-Walled Carbon Nanohorns. Appl. Surf. Sci. 2013, 277, 88–93. [Google Scholar] [CrossRef]
- Mandumpal, J.; Gemming, S.; Seifert, G. Curvature Effects of Nitrogen on Graphitic Sheets: Structures and Energetics. Chem. Phys. Lett. 2007, 447, 115–120. [Google Scholar] [CrossRef]
- Broitman, E.; Hellgren, N.; Järrendahl, K.; Johansson, M.; Olafsson, S.; Radnóczi, G.; Sundgren, J.-E.; Hultman, L. Electrical and Optical Properties of CNx (0≤ x ≤ 0.25) Films Deposited by Reactive Magnetron Sputtering. J. Appl. Phys. 2001, 89, 1184–1190. [Google Scholar] [CrossRef]



| Test | Input Power I/U/P | Feedstock Gas | Buffer Gas |
|---|---|---|---|
| Ar | 85 A/61 V/~5.2 kW | C3H8 1 slm | Ar 35 slm |
| He | 92 A/103 V/~9.5 kW | C3H8 1 slm | He 35 slm |
| Ar-H2 | 90 A/84 V/~7.6 kW | C3H8 1 slm | Ar (32 slm), H2 (3 slm) |
| Ar-N2 | 95 A/78 V/~7.4 kW | C3H8 1 slm | Ar (32 slm), N2 (3 slm) |
| Sample | Position (cm−1) | FWHM (cm−1) | ID/IG Value | I2D/IG Value |
|---|---|---|---|---|
| Ar | 1575.58 | 42.37 | 0.54 | 0.53 |
| He | 1573.28 | 31.40 | 0.46 | 0.64 |
| Ar-H2 | 1571.17 | 30.81 | 0.37 | 0.69 |
| Ar-N2 | 1581.56 | 29.36 | 0.25 | 0.82 |
| Sample | C | O | N |
|---|---|---|---|
| Ar | 98.9% | 1.1% | - |
| He | 98.7% | 1.3% | - |
| Ar-H2 | 98.9% | 1.1% | - |
| Ar-N2 | 95.8% | 2.3% | 1.9% |
| N2 Flow Rate | N-Doped Content | Pyridinic N | Pyrrolic N | Graphitic N | Oxidized N |
|---|---|---|---|---|---|
| 1 slm | 0.7% | 0.16% | 0.31% | 0.12% | 0.11% |
| 3 slm | 1.9% | 0.43% | 0.85% | 0.36% | 0.26% |
| 5 slm | 2.8% | 0.76% | 1.31% | 0.41% | 0.32% |
| Sample | BET Surface Area (m2/g) | Pore Volume (cm3/g) | Average Pore Size (nm) |
|---|---|---|---|
| Ar | 138.26 | 0.57 | 19.18 |
| He | 172.63 | 0.68 | 16.26 |
| Ar-H2 | 281.94 | 0.94 | 14.89 |
| Ar-N2 | 353.77 | 1.31 | 13.21 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wang, C.; Song, M.; Chen, X.; Li, D.; Xia, W.; Xia, W. Effects of Buffer Gases on Graphene Flakes Synthesis in Thermal Plasma Process at Atmospheric Pressure. Nanomaterials 2020, 10, 309. https://doi.org/10.3390/nano10020309
Wang C, Song M, Chen X, Li D, Xia W, Xia W. Effects of Buffer Gases on Graphene Flakes Synthesis in Thermal Plasma Process at Atmospheric Pressure. Nanomaterials. 2020; 10(2):309. https://doi.org/10.3390/nano10020309
Chicago/Turabian StyleWang, Cheng, Ming Song, Xianhui Chen, Dongning Li, Weiluo Xia, and Weidong Xia. 2020. "Effects of Buffer Gases on Graphene Flakes Synthesis in Thermal Plasma Process at Atmospheric Pressure" Nanomaterials 10, no. 2: 309. https://doi.org/10.3390/nano10020309
APA StyleWang, C., Song, M., Chen, X., Li, D., Xia, W., & Xia, W. (2020). Effects of Buffer Gases on Graphene Flakes Synthesis in Thermal Plasma Process at Atmospheric Pressure. Nanomaterials, 10(2), 309. https://doi.org/10.3390/nano10020309




