6-Aza-2-Thio-Thymine Stabilized Gold Nanoclusters as Photoluminescent Probe for Protein Detection
Abstract
1. Introduction
2. Materials and Methods
2.1. Chemical and Reagents
2.2. Instruments
2.3. Preparation of ATT-AuNCs
2.4. Sample Analysis
3. Results and Discussion
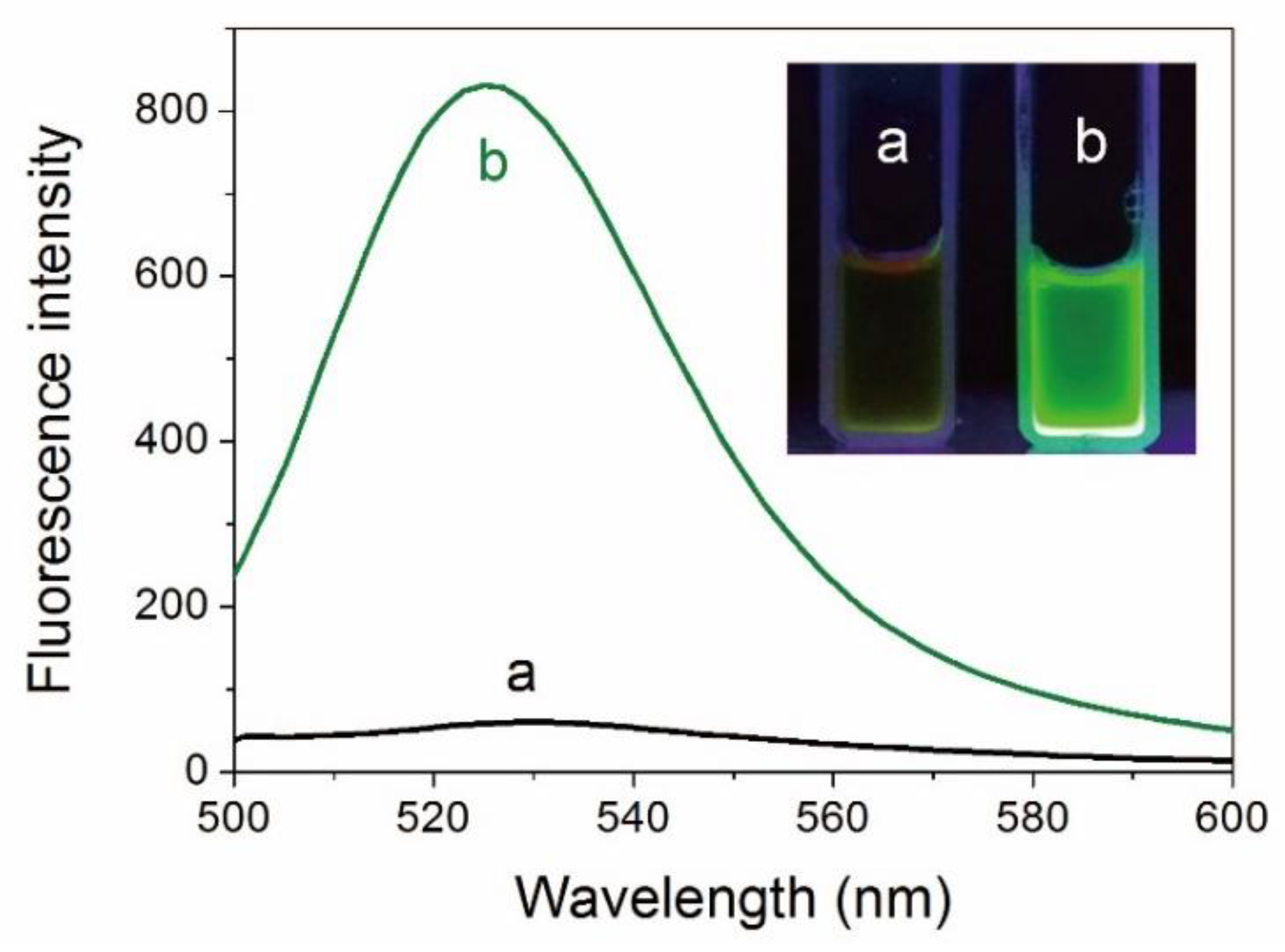
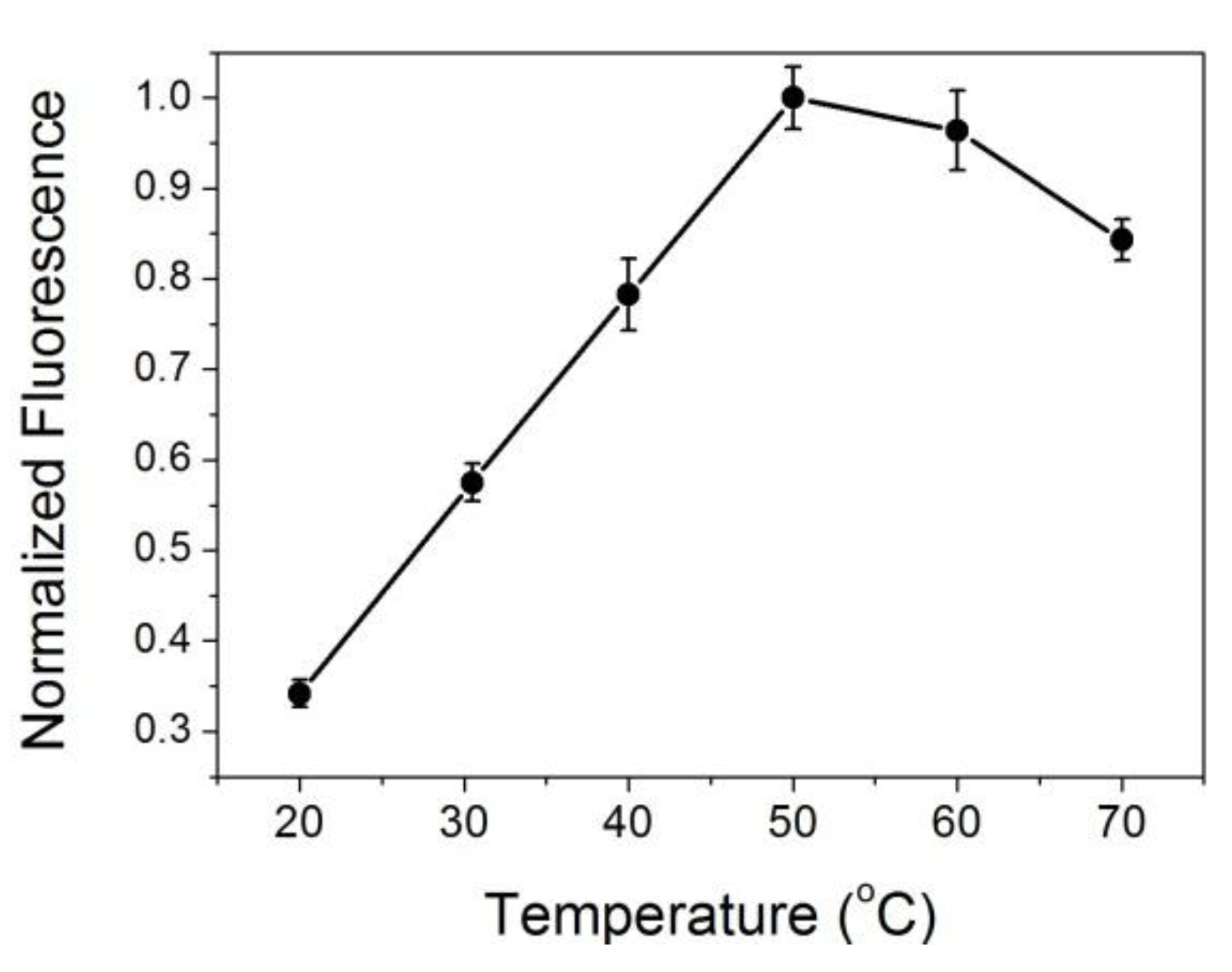
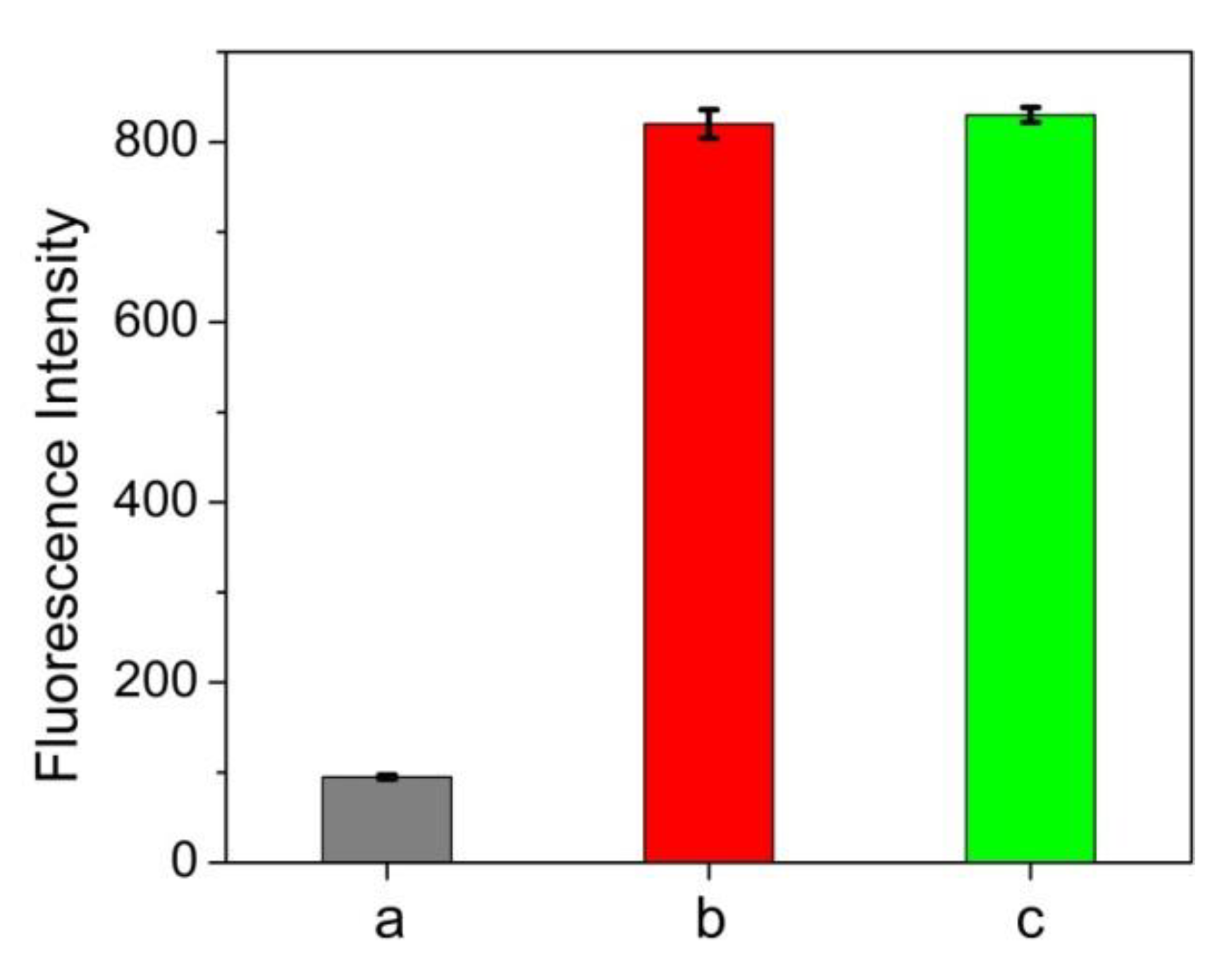
3.1. Interaction between Protein and ATT-AuNCs
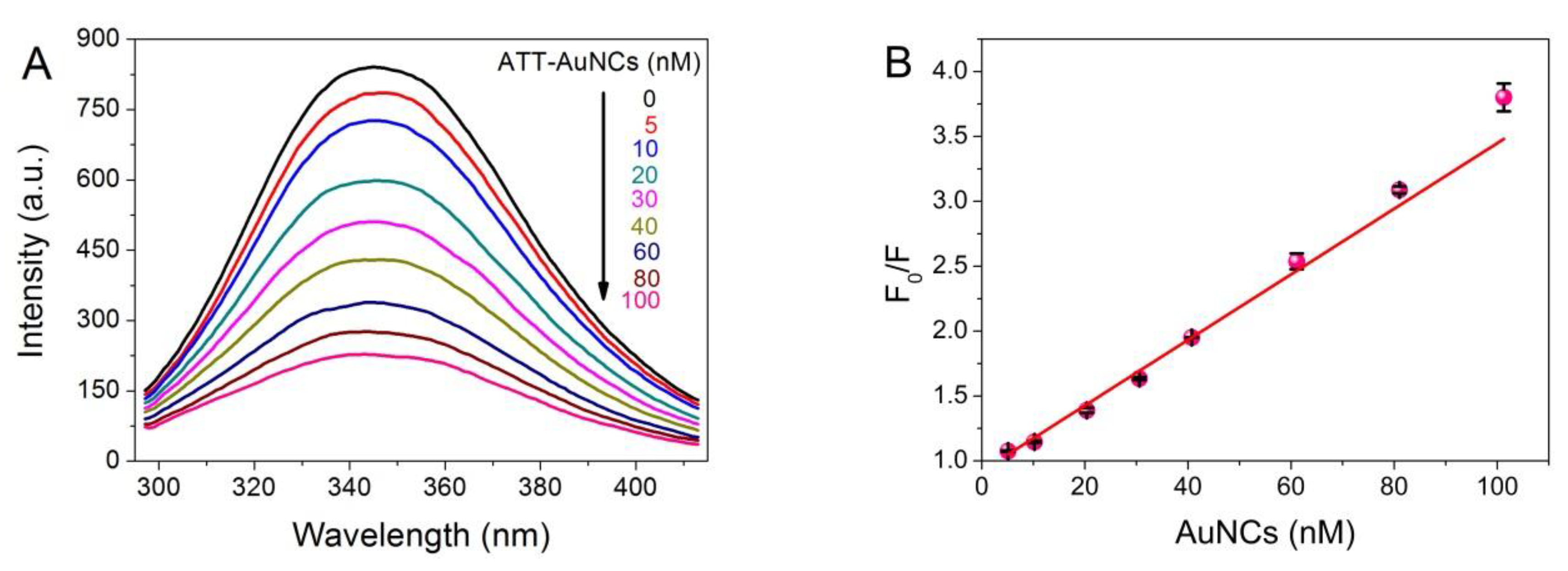
3.2. Analytical Performance
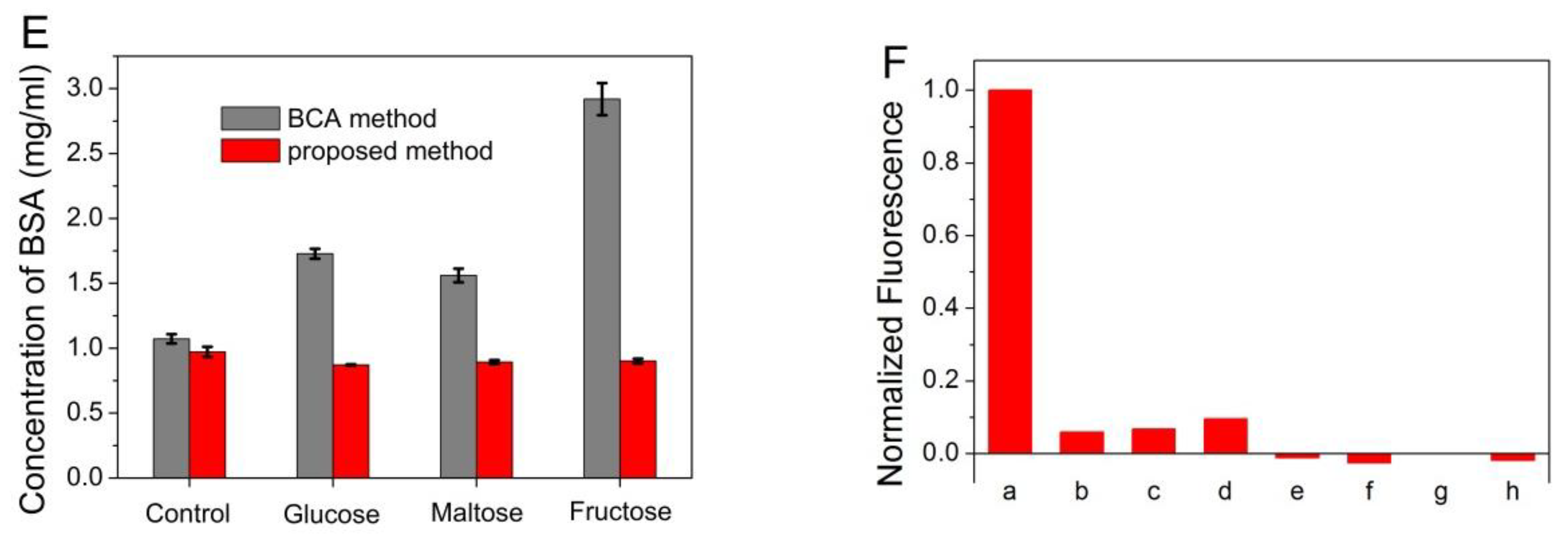
3.3. Determination of Total Protein in Human Plasma and Serum
3.4. Determination of Proteins in Milk Samples
3.5. Determination of Cellular Total Protein Concentration
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Smith, P.K.; Krohn, R.I.; Hermanson, G.T.; Mallia, A.K.; Gartner, F.H.; Provenzano, M.D.; Fujimoto, E.K.; Goeke, N.M.; Olson, B.J.; Klenk, D.C. Measurement of Protein Using Bicinchoninic Acid. Anal. Biochem. 1985, 150, 76–85. [Google Scholar] [CrossRef]
- Lowry, O.H.; Nira, J.; Rosebrough, A.; Lewis, F.; Rose, J.R. Protein Measurement with the Folin Phenol Reagent. J. Biol. Chem. 1951, 193, 265–275. [Google Scholar] [PubMed]
- Lorenzen, A.; Kennedy, S.W. A Fluorescence-Based Protein Assay for Use with a Microplate Reader. Anal. Biochem. 1993, 214, 346–348. [Google Scholar] [CrossRef] [PubMed]
- Han, X.X.; Zhao, B.; Ozaki, Y. Surface-Enhanced Raman Scattering for Protein Detection. Anal. Bioanal. Chem. 2009, 394, 1719–1727. [Google Scholar] [CrossRef] [PubMed]
- Xie, J.P.; Zheng, Y.G.; Ying, J.Y. Protein-Directed Synthesis of Highly Fluorescent Gold Nanoclusters. J. Am. Chem. Soc. 2009, 131, 888–889. [Google Scholar] [CrossRef] [PubMed]
- Jin, R.C. Quantum Sized, Thiolate-Protected Gold Nanoclusters. Nanoscale 2010, 2, 343–362. [Google Scholar] [CrossRef] [PubMed]
- Qian, H.F.; Zhu, M.Z.; Wu, Z.K.; Jin, R.C. Quantum Sized Gold Nanoclusters with Atomic Precision. Acc. Chem. Res. 2012, 45, 1470–1479. [Google Scholar] [CrossRef]
- Deng, H.H.; Shi, X.Q.; Wang, F.F.; Peng, H.P.; Liu, A.L.; Xia, X.H.; Chen, W. Fabrication of Water-Soluble, Green-Emitting Gold Nanoclusters with a 65% Photoluminescence Quantum Yield Via Host-Guest Recognition. Chem. Mater. 2017, 29, 1362–1369. [Google Scholar] [CrossRef]
- Peng, H.P.; Huang, Z.N.; Sheng, Y.L.; Zhang, X.P.; Deng, H.H.; Chen, W.; Liu, J.W. Pre-Oxidation of Gold Nanoclusters Results in a 66% Anodic Electrochemiluminescence Yield and Drives Mechanistic Insights. Angew. Chem. Int. Ed. 2019, 58, 11691–11694. [Google Scholar] [CrossRef] [PubMed]
- Shang, L.; Dong, S.J.; Nienhaus, G.U. Ultra-Small Fluorescent Metal Nanoclusters: Synthesis and Biological Applications. Nano Today 2011, 6, 401–418. [Google Scholar] [CrossRef]
- Lu, Y.Z.; Chen, W. Sub-Nanometre Sized Metal Clusters: From Synthetic Challenges to the Unique Property Discoveries. Chem. Soc. Rev. 2012, 41, 3594–3623. [Google Scholar] [CrossRef] [PubMed]
- Cui, M.; Zhao, Y.; Song, Q. Synthesis, Optical Properties and Applications of Ultra-Small Luminescent Gold Nanoclusters. TrAC-Trend Anal. Chem. 2014, 57, 73–82. [Google Scholar] [CrossRef]
- Shang, L.; Doerlich, R.M.; Brandholt, S.; Schneider, R.; Trouillet, V.; Bruns, M.; Gerthsen, D.; Nienhaus, G.U. Facile Preparation of Water-Soluble Fluorescent Gold Nanoclusters for Cellular Imaging Applications. Nanoscale 2011, 3, 2009–2014. [Google Scholar] [CrossRef] [PubMed]
- Tian, D.H.; Qian, Z.S.; Xia, Y.S.; Zhu, C.Q. Gold Nanocluster-Based Fluorescent Probes for near-Infrared and Turn-on Sensing of Glutathione in Living Cells. Langmuir 2012, 28, 3945–3951. [Google Scholar] [CrossRef]
- Deng, H.H.; Wu, G.W.; Zou, Z.Q.; Peng, H.P.; Liu, A.L.; Lin, X.H.; Xia, X.H.; Chen, W. pH-Sensitive Gold Nanoclusters: Preparation and Analytical Applications for Urea, Urease, and Urease Inhibitor Detection. Chem. Comm. 2015, 51, 7847–7850. [Google Scholar] [CrossRef]
- Deng, H.H.; Zhang, L.N.; He, S.B.; Liu, A.L.; Li, G.W.; Lin, X.H.; Xia, X.H.; Chen, W. Methionine-Directed Fabrication of Gold Nanoclusters with Yellow Fluorescent Emission for Cu2+ Sensing. Biosens. Bioelectron. 2015, 65, 397–403. [Google Scholar] [CrossRef]
- Martin-Barreiro, A.; Marcos, S.; Fuente, J.M.; Grazu, V.; Galban, J. Gold Nanocluster Fluorescence as an Indicator for Optical Enzymatic Nanobiosensors: Choline and Acetylcholine Determination. Sens. Actuat. B-Chem. 2018, 277, 261–270. [Google Scholar] [CrossRef]
- Huang, K.Y.; He, H.X.; He, S.B.; Zhang, X.P.; Peng, H.P.; Lin, Z.; Deng, H.H.; Xia, X.H.; Chen, W. Gold Nanocluster-Based Fluorescence Turn-Off Probe for Sensing of Doxorubicin by Photoinduced Electron Transfer. Sens. Actuat. B-Chem. 2019, 296, 126656. [Google Scholar] [CrossRef]
- Nel, A.E.; Madler, L.; Velegol, D.; Xia, T.; Hoek, E.M.V.; Somasundaran, P.; Klaessig, F.; Castranova, V.; Thompson, M. Understanding Biophysicochemical Interactions at the Nano-Bio Interface. Nat. Mater. 2009, 8, 543–557. [Google Scholar] [CrossRef]
- Wang, J.; Jensen, U.B.; Jensen, G.V.; Shipovskov, S.; Balakrishnan, V.S.; Otzen, D.; Pedersen, J.S.; Besenbacher, F.; Sutherland, D.S. Soft Interactions at Nanoparticles Alter Protein Function and Conformation in a Size Dependent Manner. Nano Lett. 2011, 11, 4985–4991. [Google Scholar] [CrossRef]
- Mahmoudi, M.; Lynch, I.; Ejtehadi, M.R.; Monopoli, M.P.; Bombelli, F.B.; Laurent, S. Protein-Nanoparticle Interactions: Opportunities and Challenges. Chem. Rev. 2011, 111, 5610–5637. [Google Scholar] [CrossRef] [PubMed]
- Huang, Z.; Wang, H.; Yang, W. Gold Nanoparticle-Based Facile Detection of Human Serum Albumin and Its Application as an Inhibit Logic Gate. ACS Appl. Mater. Inter. 2015, 7, 8990–8998. [Google Scholar] [CrossRef] [PubMed]
- Lacerda, S.H.; Paoli, D.; Park, J.J.; Meuse, C.; Pristinski, D.; Becker, M.L.; Karim, A.; Douglas, J.F. Interaction of Gold Nanoparticles with Common Human Blood Proteins. ACS Nano 2010, 4, 365–379. [Google Scholar] [CrossRef] [PubMed]
- Shang, L.; Brandholt, S.; Stockmar, F.; Trouillet, V.; Bruns, M.; Nienhaus, G.U. Effect of Protein Adsorption on the Fluorescence of Ultrasmall Gold Nanoclusters. Small 2012, 8, 661–665. [Google Scholar] [CrossRef] [PubMed]
- Xu, S.; Lu, X.; Yao, C.; Huang, F.; Jiang, H.; Hua, W.; Na, N.; Liu, H.; Ouyang, J. A Visual Sensor Array for Pattern Recognition Analysis of Proteins Using Novel Blue-Emitting Fluorescent Gold Nanoclusters. Anal. Chem. 2014, 86, 11634–11639. [Google Scholar] [CrossRef] [PubMed]
- Deng, H.H.; Shi, X.Q.; Peng, H.P.; Zhuang, Q.Q.; Yang, Y.; Liu, A.L.; Xia, X.H.; Chen, W. Gold Nanoparticle-Based Photoluminescent Nanoswitch Controlled by Host-Guest Recognition and Enzymatic Hydrolysis for Arginase Activity Assay. ACS Appl. Mater. Interfaces 2018, 10, 5358–5364. [Google Scholar] [CrossRef]
- Deng, H.H.; Wang, F.F.; Shi, X.Q.; Peng, H.P.; Liu, A.L.; Xia, X.H.; Chen, W. Water-Soluble Gold Nanoclusters Prepared by Protein-Ligand Interaction as Fluorescent Probe for Real-Time Assay of Pyrophospatase Activity. Biosens. Bioelectron. 2016, 83, 1–8. [Google Scholar] [CrossRef]
- Kang, J.; Liu, Y.; Xie, M.; Li, S.; Jiang, M.; Wang, Y. Interactions of Human Serum Albumin with Chlorogenic Acid and Ferulic Acid. BBA-Gen. Subj. 2004, 1674, 205–214. [Google Scholar] [CrossRef]
- Snyder, S.; Pendergraph, B. Detection and Evaluation of Chronic Kidney Disease. Interventions 2005, 100, 24–25. [Google Scholar]
- Tibblin, G.; Bergentz, S.E.; Bjure, J.; Wilhelmsen, L. Hematocrit, Plasma Protein, Plasma Volume, and Viscosity in Early Hypertensive Disease. Am. Heart J. 1966, 72, 165–176. [Google Scholar] [CrossRef]
- Ingelfinger, J.R. Melamine and the Global Implications of Food Contamination. New Eng. J. Med. 2008, 359, 2745–2748. [Google Scholar] [CrossRef] [PubMed]
- Kobayashi, T.; Okada, A.; Fujii, Y.; Niimi, K.; Hamamoto, S.; Yasui, T.; Tozawa, K.; Kohri, K. The Mechanism of Renal Stone Formation and Renal Failure Induced by Administration of Melamine and Cyanuric Acid. Urol. Res. 2010, 38, 117–125. [Google Scholar] [CrossRef] [PubMed]
- Finete, V.D.L.M.; Gouvêa, M.M.; de Carvalho Marques, F.F.; Netto, A.D.P. Is It Possible to Screen for Milk or Whey Protein Adulteration with Melamine, Urea and Ammonium Sulphate, Combining Kjeldahl and Classical Spectrophotometric Methods? Food Chem. 2013, 141, 3649–3655. [Google Scholar] [CrossRef]
- Chiodoni, C.; Colombo, M.P.; Sangaletti, S. Matricellular Proteins: From Homeostasis to Inflammation, Cancer, and Metastasis. Cancer Metastasis Rev. 2010, 29, 295–307. [Google Scholar] [CrossRef] [PubMed]
- De Mejia, E.G.; Dia, V.P. The Role of Nutraceutical Proteins and Peptides in Apoptosis, Angiogenesis, and Metastasis of Cancer Cells. Cancer Metastasis Rev. 2010, 29, 511–528. [Google Scholar] [CrossRef] [PubMed]

| T (°C) | K (108 L mol−1) | ΔH (kJ mol−1) | ΔG (kJ) | ΔS (J mol−1 K−1) | R | SD |
|---|---|---|---|---|---|---|
| 21 | 5.70 | 17.583 | −49.275 | 227.380 | 0.9996 | 0.97 |
| 33 | 7.38 | −51.948 | 0.9978 | 2.41 | ||
| 38 | 8.51 | −53.167 | 0.9997 | 0.70 |
| Sample | Proposed Method (mg/mL, n = 3) | BCA Method (mg/mL, n = 3) | F-Test 1 | t-Test 1 |
|---|---|---|---|---|
| 1 | 62.60 ± 1.35 | 62.84 ± 1.27 | 1.13 | 0.22 |
| 2 | 61.73 ± 0.28 | 62.20 ± 1.17 | 17.46 | 0.68 |
| 3 | 58.08 ± 1.15 | 58.55 ± 1.26 | 1.21 | 0.48 |
| 4 | 58.95 ± 0.73 | 58.05 ± 0.78 | 1.14 | 1.46 |
| 5 | 65.18 ± 2.62 | 65.95 ± 1.42 | 3.40 | 0.45 |
| 6 | 63.26 ± 1.88 | 64.80 ± 1.66 | 1.28 | 1.07 |
| Sample | Proposed Method (mg/mL, n = 3) | BCA Method (mg/mL, n = 3) | F-Test 1 | t-Test 1 |
|---|---|---|---|---|
| 1 | 59.15 ± 0.82 | 61.25 ± 2.78 | 11.49 | 1.26 |
| 2 | 65.28 ± 1.63 | 64.86 ± 1.96 | 1.45 | 0.29 |
| 3 | 64.81 ± 1.45 | 65.02 ± 0.52 | 7.78 | 0.24 |
| 4 | 70.56 ± 0.48 | 71.74 ± 1.93 | 16.17 | 1.03 |
| Sample | Proposed Method (mg/mL, n = 3) | BCA Method (mg/mL, n = 3) | F-Test 1 | t-Test 1 |
|---|---|---|---|---|
| 1 | 31.17 ± 0.18 | 32.29 ± 0.43 | 5.71 | 4.16 |
| 2 | 32.44 ± 0.19 | 32.01 ± 0.52 | 7.49 | 1.34 |
| 3 | 30.01 ± 0.16 | 30.05 ± 0.50 | 9.77 | 0.14 |
| Sample | Proposed Method (mg/mL, n = 3) | BCA Method (mg/mL, n = 3) | F-Test 1 | t-Test 1 |
|---|---|---|---|---|
| 1 | 5.52 ± 0.034 | 5.51 ± 0.026 | 1.71 | 0.40 |
| 2 | 6.77 ± 0.046 | 6.84 ± 0.072 | 2.45 | 1.42 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Deng, H.-H.; Shi, X.-Q.; Balasubramanian, P.; Huang, K.-Y.; Xu, Y.-Y.; Huang, Z.-N.; Peng, H.-P.; Chen, W. 6-Aza-2-Thio-Thymine Stabilized Gold Nanoclusters as Photoluminescent Probe for Protein Detection. Nanomaterials 2020, 10, 281. https://doi.org/10.3390/nano10020281
Deng H-H, Shi X-Q, Balasubramanian P, Huang K-Y, Xu Y-Y, Huang Z-N, Peng H-P, Chen W. 6-Aza-2-Thio-Thymine Stabilized Gold Nanoclusters as Photoluminescent Probe for Protein Detection. Nanomaterials. 2020; 10(2):281. https://doi.org/10.3390/nano10020281
Chicago/Turabian StyleDeng, Hao-Hua, Xiao-Qiong Shi, Paramasivam Balasubramanian, Kai-Yuan Huang, Ying-Ying Xu, Zhong-Nan Huang, Hua-Ping Peng, and Wei Chen. 2020. "6-Aza-2-Thio-Thymine Stabilized Gold Nanoclusters as Photoluminescent Probe for Protein Detection" Nanomaterials 10, no. 2: 281. https://doi.org/10.3390/nano10020281
APA StyleDeng, H.-H., Shi, X.-Q., Balasubramanian, P., Huang, K.-Y., Xu, Y.-Y., Huang, Z.-N., Peng, H.-P., & Chen, W. (2020). 6-Aza-2-Thio-Thymine Stabilized Gold Nanoclusters as Photoluminescent Probe for Protein Detection. Nanomaterials, 10(2), 281. https://doi.org/10.3390/nano10020281





