Predictive Biomarkers for the Ranking of Pulmonary Toxicity of Nanomaterials
Abstract
1. Introduction
2. Materials and Methods
2.1. Sample Nanomaterials
2.2. Animals
2.3. Intratracheal Instillation
2.4. Animals Following Intratracheal Instillation
2.5. Total RNA Extraction
2.6. Microarray Analysis
2.7. Validation of Gene Expression Data Using Quantitative Real-Time Polymerase Chain Reaction
2.8. Statistical Analysis
2.9. Histopathology and Immunohistochemistry
3. Results
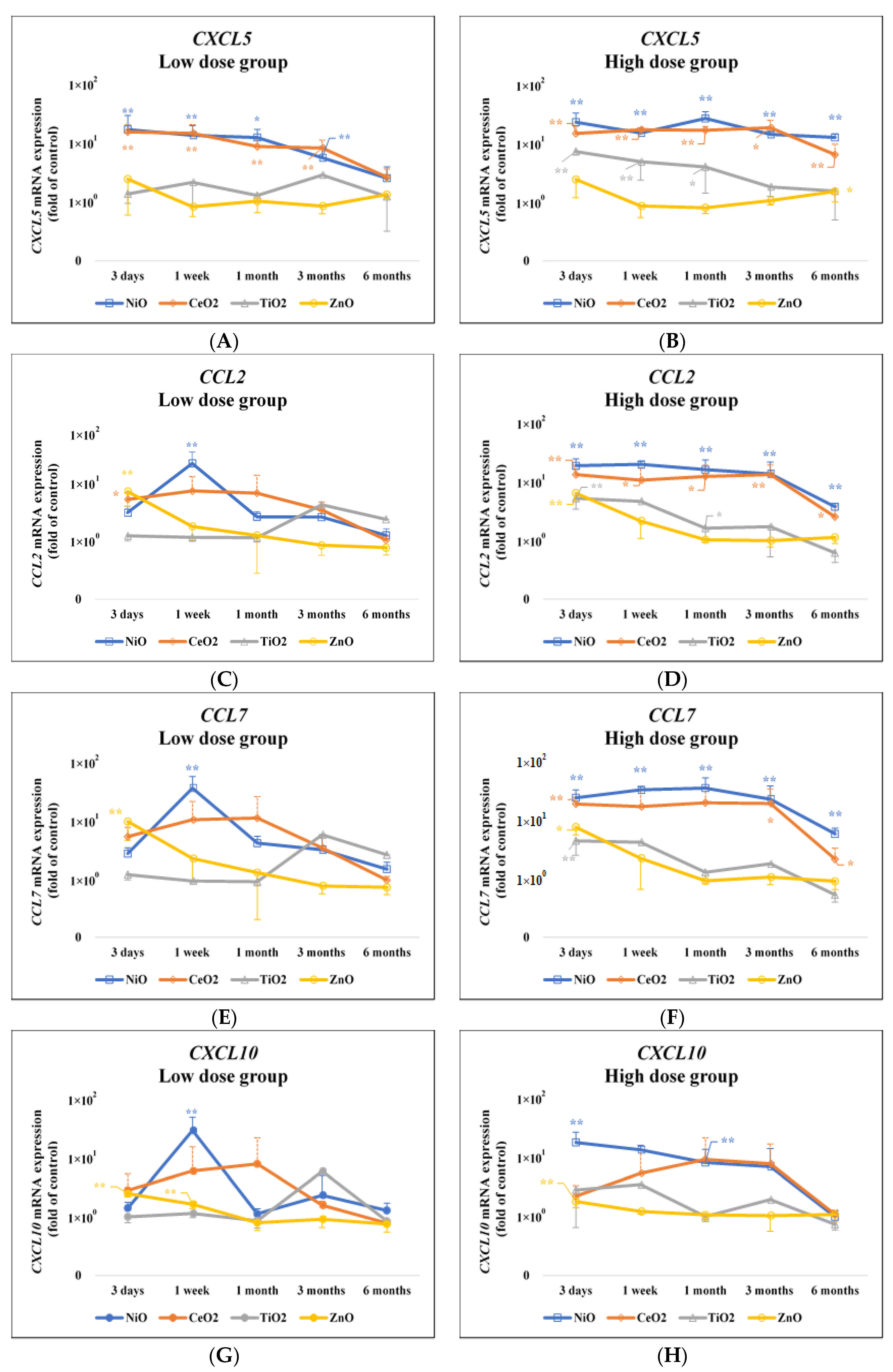
3.1. Gene Expression Analysis
3.2. Assessment of the Accuracy of Gene Expression of the Toxicity of Chemicals
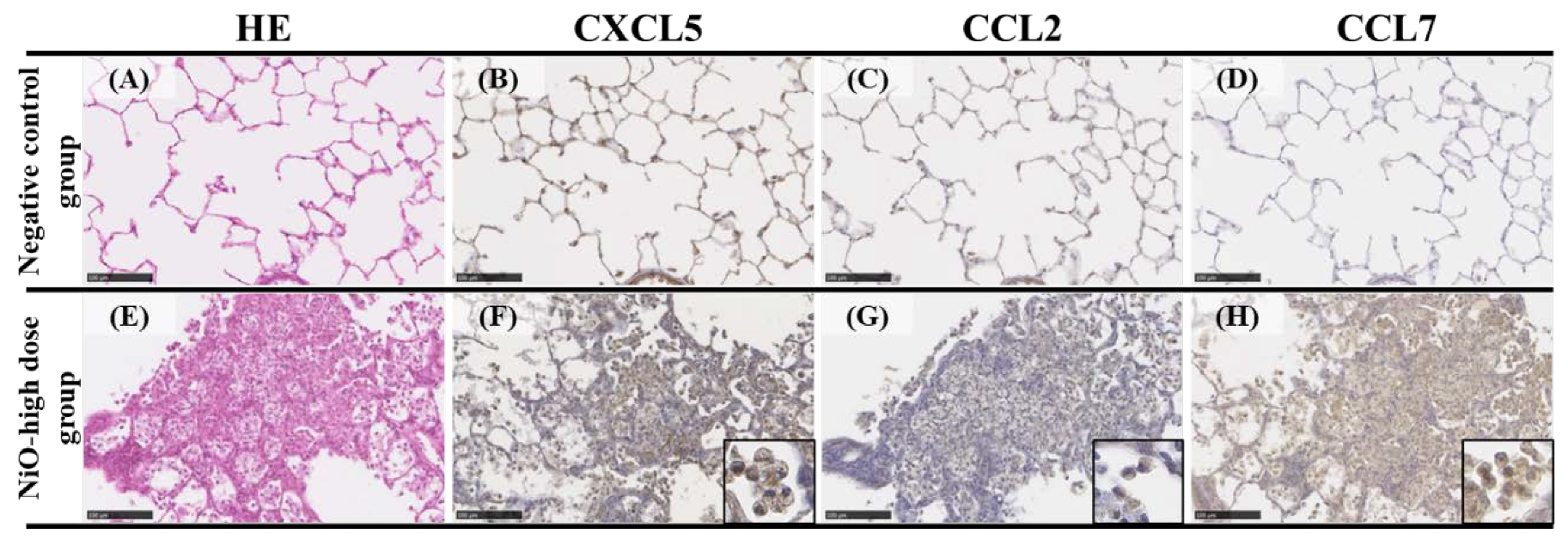
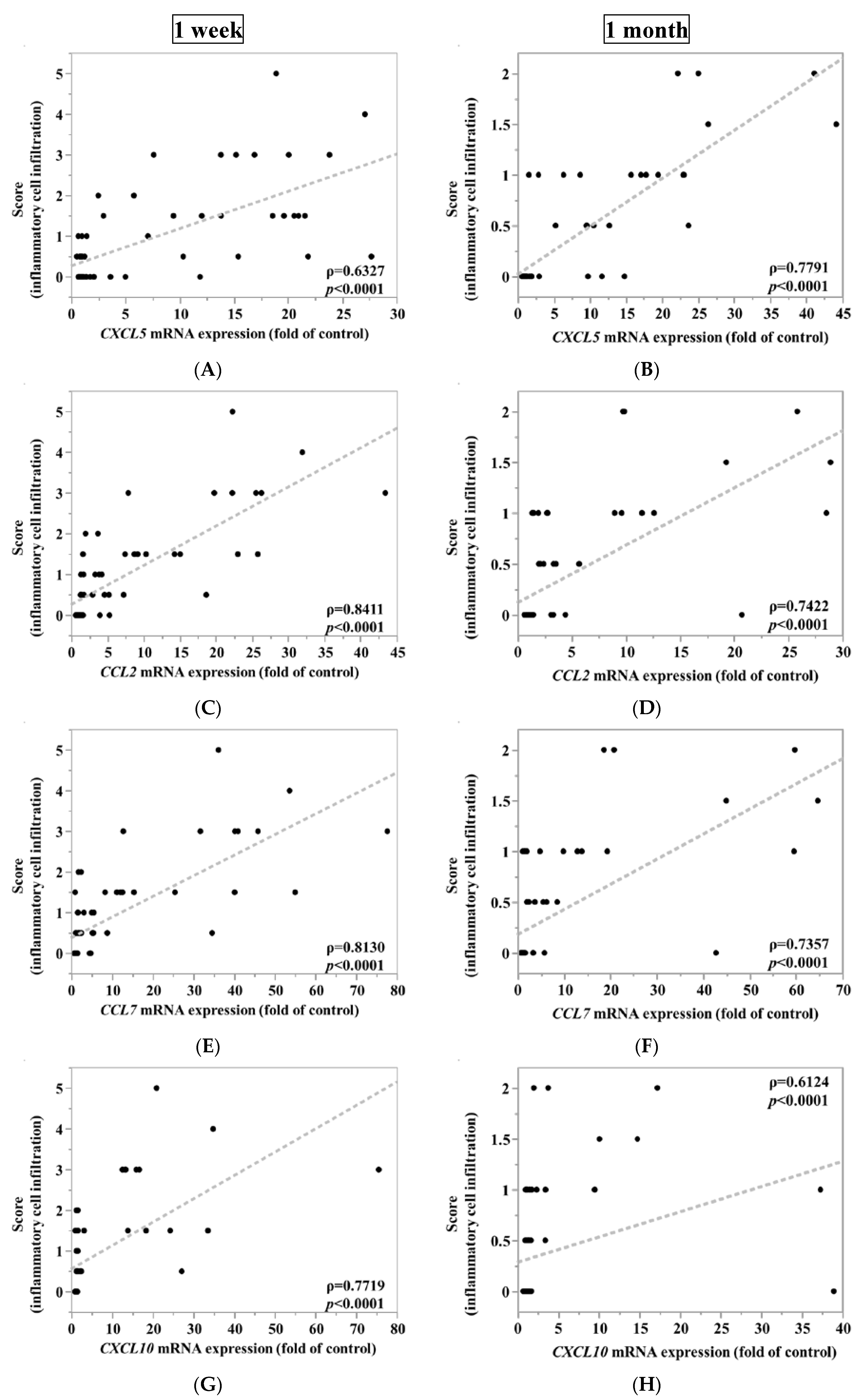
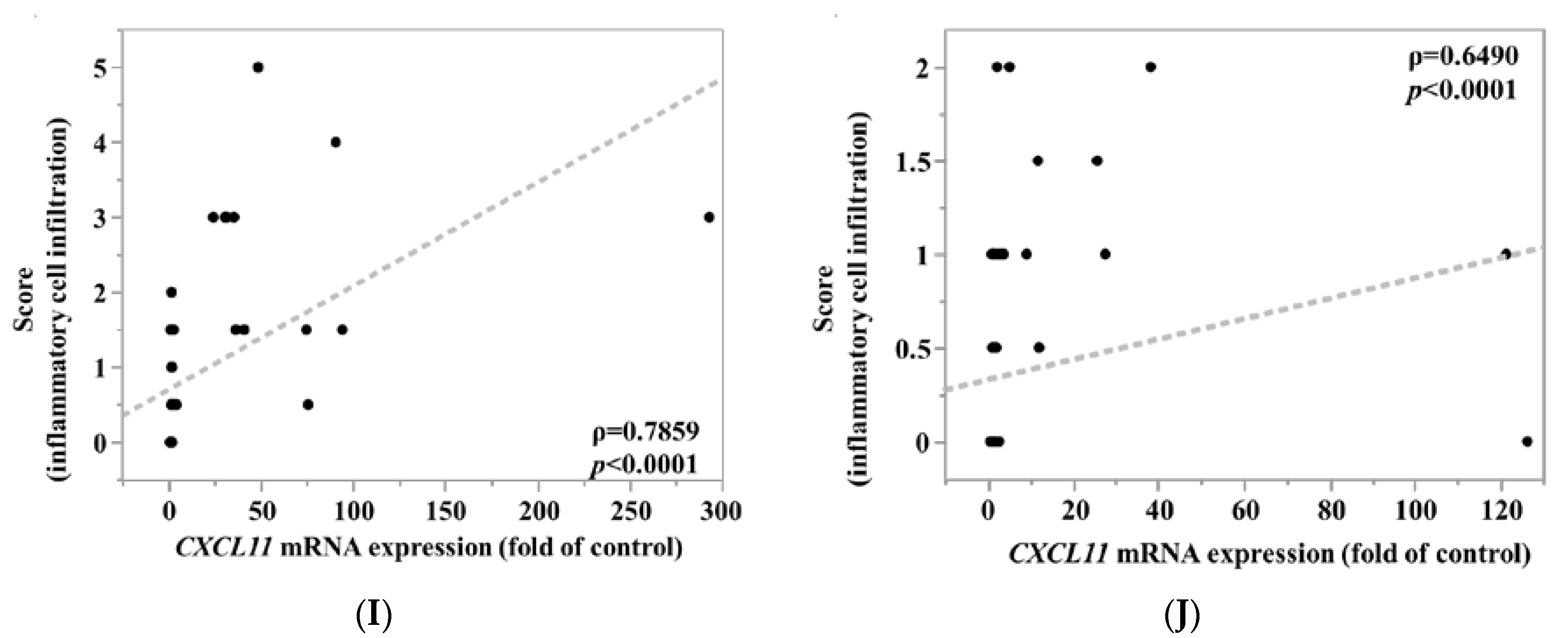
3.3. CXCL5, CCL2 and CCL7 Immunostaining
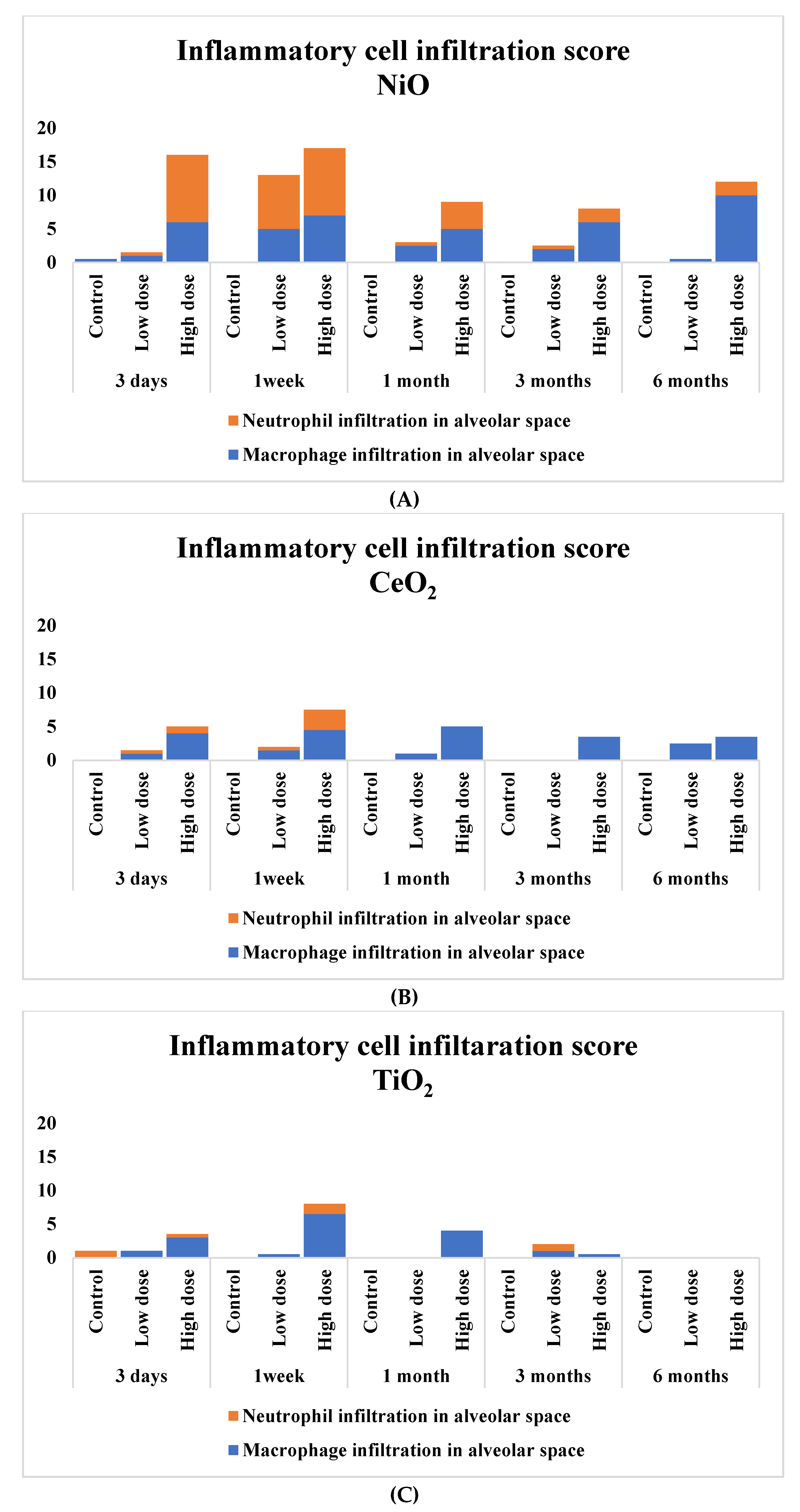
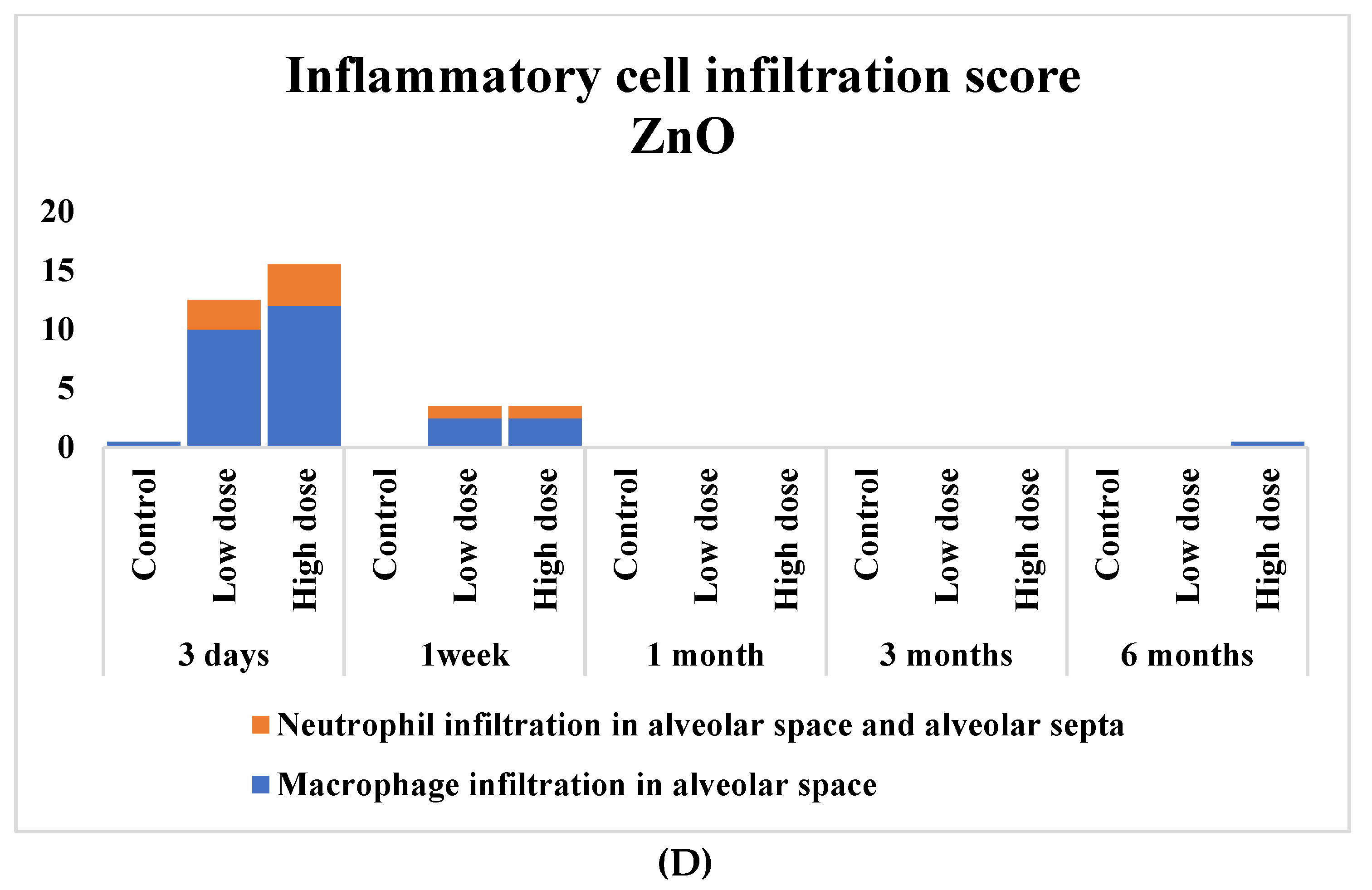
3.4. Pathological Features in the Rat Lungs
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Appendix A
| (A) Confusion matrix | |||
| Pulmonary Toxicity | |||
| High | Low | ||
| Gene expression level (fold of control) | ≧Cut off value | a (True positives) | b (False positives) |
| <Cut off value | c (False negatives) | d (True negatives) | |
| a + c | b + d | ||
| Sensitivity = = Specificity = = False positive = = False negative = = a: The number of true positives, b: The number of false positives. c: The number of false negatives, d: The number of true negatives. | |||
| (B) Example of confusion matrix. | |||
| CXCL5 at 1 week | Pulmonary Toxicity | ||
| High | Low | ||
| Gene expression level (fold of control) | ≧Cut off value | 19 | 0 |
| <Cut off value | 1 | 20 | |
| 20 | 20 | ||
| For example, in CXCL5 at 1 week after instillation. Sensitivity = = 0.95, Specificity = = 1.00, False positive = = 0.00, False negative = = 0.05. | |||
| CXCL5 | 3 Days | 1 Week | 1 Month | 3 Months | 6 Months |
|---|---|---|---|---|---|
| Low Dose Group | |||||
| NiO | 0.0037 ** | 0.0008 ** | 0.0134 * | 0.0056 ** | 0.1862 |
| CeO2 | 0.0002 ** | <0.0001 ** | 0.0003 ** | 0.0302 ** | 0.3518 |
| TiO2 | 0.4254 | 0.4474 | 0.9 | 0.2214 | 0.8551 |
| ZnO | 0.1483 | 0.6346 | 0.7478 | 0.3446 | 0.1679 |
| CCL2 | 3 Days | 1 Week | 1 Month | 3 Months | 6 Months |
| Low Dose Group | |||||
| NiO | 0.5879 | 0.0017 ** | 0.8141 | 0.8456 | 0.4627 |
| CeO2 | 0.0170 * | 0.1357 | 0.2553 | 0.5301 | 0.9883 |
| TiO2 | 0.9217 | 0.9928 | 0.737 | 0.1995 | 0.5812 |
| ZnO | 0.0011 ** | 0.926 | 0.6449 | 0.358 | 0.2674 |
| CCL7 | 3 Days | 1 Week | 1 Month | 3 Months | 6 Months |
| Low Dose Group | |||||
| NiO | 0.8425 | 0.0018 ** | 0.8701 | 0.9175 | 0.608 |
| CeO2 | 0.1495 | 0.3895 | 0.4254 | 0.8892 | 1 |
| TiO2 | 0.9424 | 0.9997 | 0.9087 | 0.2562 | 0.6004 |
| ZnO | 0.0019 ** | 0.5976 | 0.6198 | 0.3636 | 0.1821 |
| CXCL10 | 3 Days | 1 Week | 1 Month | 3 Months | 6 Months |
| Low Dose Group | |||||
| NiO | 0.9877 | 0.0053 ** | 0.9959 | 0.8678 | 0.3692 |
| CeO2 | 0.161 | 0.4941 | 0.5374 | 0.9784 | 0.3855 |
| TiO2 | 0.9956 | 0.9929 | 0.7097 | 0.212 | 0.592 |
| ZnO | <0.0001 ** | 0.0002 ** | 0.4146 | 0.3867 | 0.3168 |
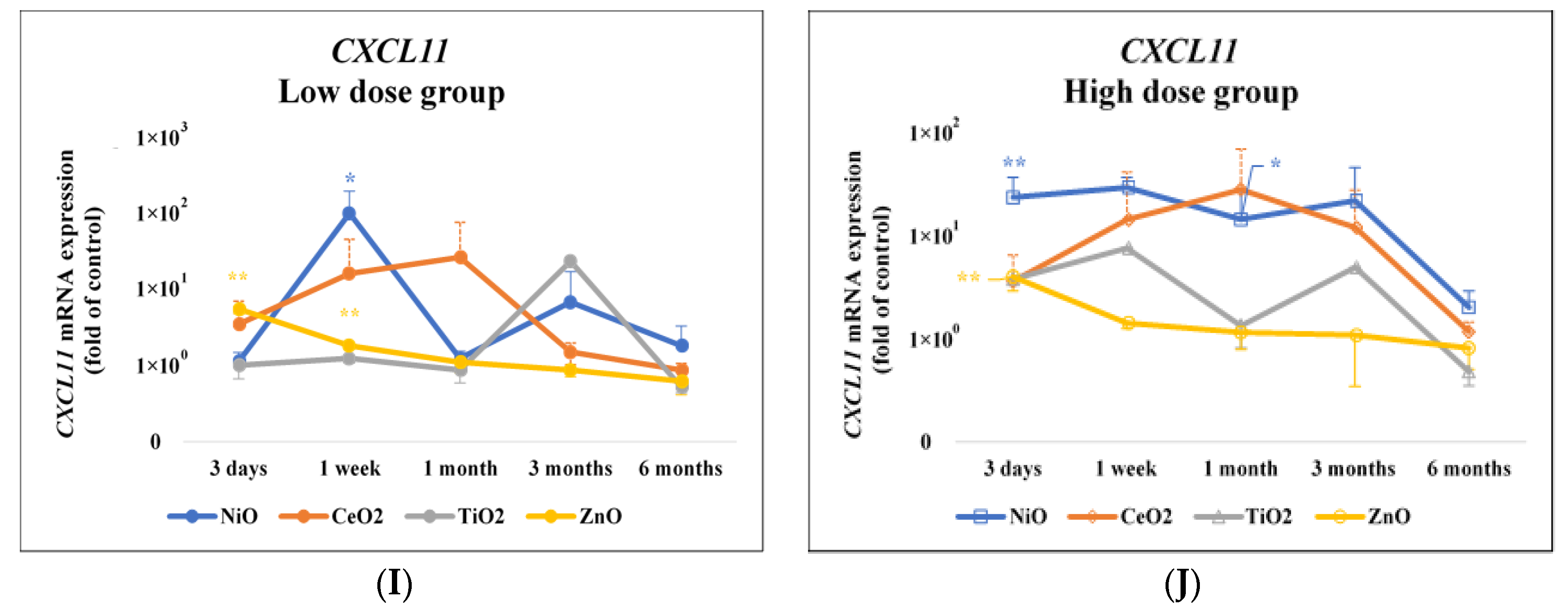
| CXCL11 | 3 Days | 1 Week | 1 Month | 3 Months | 6 Months |
| Low Dose Group | |||||
| NiO | 0.9995 | 0.0289 * | 0.9985 | 0.8235 | 0.4274 |
| CeO2 | 0.2719 | 0.5391 | 0.5132 | 0.9954 | 0.6872 |
| TiO2 | 0.9996 | 0.9982 | 0.7218 | 0.2127 | 0.2207 |
| ZnO | <0.0001 ** | 0.0038 ** | 0.7349 | 0.3887 | 0.1187 |
| CXCL5 | 3 Days | 1 Week | 1 Month | 3 Months | 6 Months |
| High dose group | |||||
| NiO | 0.0063 ** | 0.0003 ** | <0.0001 ** | <0.0001 ** | <0.0001 ** |
| CeO2 | 0.0004 ** | <0.0001 ** | <0.0001 ** | <0.0301 * | 0.0020 ** |
| TiO2 | <0.0001 ** | 0.0053 ** | 0.0133 * | 0.7013 | 0.4984 |
| ZnO | 0.1626 | 0.6737 | 0.6125 | 0.4968 | 0.0405 * |
| CCL2 | 3 Days | 1 Week | 1 Month | 3 Months | 6 Months |
| High Dose Group | |||||
| NiO | <0.0001 ** | 0.0022 ** | 0.0003 ** | 0.0025 ** | <0.0001 ** |
| CeO2 | <0.0001 ** | 0.0156 * | 0.0130 * | 0.0004 ** | 0.0177 * |
| TiO2 | <0.0001 ** | 0.1091 | 0.0267 * | 0.8836 | 0.9128 |
| ZnO | 0.0020 ** | 0.6238 | 0.9559 | 0.4076 | 0.3763 |
| CCL7 | 3 Days | 1 Week | 1 Month | 3 Months | 6 Months |
| High Dose Group | |||||
| NiO | <0.0001 ** | 0.0031 ** | 0.0004 ** | 0.0064 ** | <0.0001 ** |
| CeO2 | <0.0001 ** | 0.0889 | 0.0782 | 0.0141 * | 0.0327 * |
| TiO2 | 0.0008 ** | 0.2082 | 0.0651 | 0.9479 | 0.8681 |
| ZnO | 0.0110 * | 0.5797 | 0.9974 | 0.4146 | 0.8918 |
| CXCL10 | 3 Days | 1 Week | 1 Month | 3 Months | 6 Months |
| High Dose Group | |||||
| NiO | 0.0004 ** | 0.2108 | 0.0081 ** | 0.1018 | 0.9967 |
| CeO2 | 0.3798 | 0.5606 | 0.4095 | 0.1205 | 0.5615 |
| TiO2 | 0.0613 | 0.2908 | 0.9398 | 0.9278 | 0.3888 |
| ZnO | 0.0011 ** | 0.0655 | 0.54 | 0.4204 | 0.659 |
| CXCL11 | 3 Days | 1 Week | 1 Month | 3 Months | 6 Months |
| High Dose Group | |||||
| NiO | 0.0014 ** | 0.6045 | 0.0277 * | 0.097 | 0.2332 |
| CeO2 | 0.2058 | 0.5708 | 0.4182 | 0.1515 | 0.2836 |
| TiO2 | 0.1343 | 0.3348 | 0.7182 | 0.9359 | 0.2013 |
| ZnO | <0.0001 ** | 0.0672 | 0.4405 | 0.3996 | 0.4958 |
References
- Borm, P.J.A.; Robbins, D.; Haubold, S.; Kuhlbusch, T.; Fissan, H.; Donaldson, K.; Schins, R.; Stone, V.; Kreyling, W.; Lademann, J.; et al. The potential risks of nanomaterials: A review carried out for ECETOC. Part. Fibre Toxicol. 2006, 3, 11. [Google Scholar] [CrossRef]
- ISO/TS. ISO/TS 27687:2008(en). Nanotechnologies. Terminology and Definitions for Nano-Objects—Nanoparticle, Nanofibre and Nanoplate. Available online: https://www.iso.org/obp/ui/#iso:std:iso:ts:27687:ed-1:v2:en (accessed on 12 May 2020).
- Suzui, M.; Futakuchi, M.; Fukamachi, K.; Numano, T.; Abdelgied, M.; Takahashi, S.; Ohnishi, M.; Omori, T.; Tsuruoka, S.; Hirose, A.; et al. Multiwalled carbon nanotubes intratracheally instilled into the rat lung induce development of pleural malignant mesothelioma and lung tumors. Cancer Sci. 2016, 107, 924–935. [Google Scholar] [CrossRef] [PubMed]
- Numano, T.; Higuchi, H.; Alexander, D.B.; Alexander, W.T.; Abdelgied, M.; El-Gazzar, A.M.; Saleh, D.; Takase, H.; Hirose, A.; Naiki-Ito, A.; et al. MWCNT-7 administered to the lung by intratracheal instillation induces development of pleural mesothelioma in F344 rats. Cancer Sci. 2019, 110, 2485–2492. [Google Scholar] [CrossRef]
- Kasai, T.; Umeda, Y.; Ohnishi, M.; Mine, T.; Kondo, H.; Takeuchi, T.; Matsumoto, M.; Fukushima, S. Lung carcinogenicity of inhaled multi-walled carbon nanotube in rats. Part. Fibre Toxicol. 2016, 13, 53. [Google Scholar] [CrossRef]
- Borm, P.J.A.; Driscoll, K. Particles, inflammation and respiratory tract carcinogenesis. Toxicol. Lett. 1996, 88, 109–113. [Google Scholar]
- Shacter, E.; Weitzman, A.S. Chronic Inflammation and Cancer. Oncology 2002, 16, 217–226. [Google Scholar]
- Bellmann, B.; Muhle, H.; Creutzenberg, O.; Ernst, H.; Müller, M.; Bernstein, D.M.; Sintes, J.M.R. Calibration study on subchronic inhalation toxicity of man-made vitreous fibers in rats. Inhal. Toxicol. 2003, 15, 1147–1177. [Google Scholar] [CrossRef]
- Kim, H.; Morimoto, Y.; Ogami, A.; Nagatomo, H.; Hirohashi, M.; Oyabu, T.; Kawanami, Y.; Kuroda, E.; Higashi, T.; Tanaka, I. Differential expression of EC-SOD, Mn-SOD and CuZn-SOD in rat lung exposed to crystalline silica. J. Occup. Health 2007, 49, 242–248. [Google Scholar] [CrossRef] [PubMed]
- Nishi, K.; Morimoto, Y.; Ogami, A.; Murakami, M.; Myojo, T.; Oyabu, T.; Kadoya, C.; Yamamoto, M.; Todoroki, M.; Hirohashi, M.; et al. Expression of cytokine-induced neutrophil chemoattractant in rat lungs by intratracheal instillation of nickel oxide nanoparticles. Inhal. Toxicol. 2009, 21, 1030–1039. [Google Scholar] [CrossRef]
- Pott, F.; Ziem, U.; Reiffer, F.J.; Huth, F.; Ernst, H.; Mohr, U. Carcinogenicity studies on fibres, metal compounds, and some other dusts in rats. Exp. Pathol. 1987, 32, 129–152. [Google Scholar] [CrossRef]
- Muhle, H.; Bellmann, B.; Creutzenberg, O.; Dasenbrock, C.; Ernst, H.; Kilpper, R.; MacKenzie, J.; Morrow, P.; Mohr, U.; Takenaka, S.; et al. Pulmonary response to toner upon chronic inhalation exposure in rats. Fundam. Appl. Toxicol. 1991, 17, 280–299. [Google Scholar] [CrossRef]
- Senoh, H.; Kano, H.; Suzuki, M.; Fukushima, S.; Oshima, Y.; Kobayashi, T.; Morimoto, Y.; Izumi, H.; Ota, Y.; Takehara, H.; et al. Inter-laboratory comparison of pulmonary lesions induced by intratracheal instillation of NiO nanoparticle in rats: Histopathological examination results. J. Occup. Health 2020, 62, 1–5. [Google Scholar] [CrossRef]
- Pauluhn, J. Subchronic 13-week inhalation exposure of rats to multiwalled carbon nanotubes: Toxic effects are determined by density of agglomerate structures, not fibrillar structures. Toxicol. Sci. 2009, 113, 226–242. [Google Scholar] [CrossRef] [PubMed]
- Delorme, M.P.; Muro, Y.; Arai, T.; Banas, D.A.; Frame, S.R.; Reed, K.L.; Warheit, D.B. Ninety-day inhalation toxicity study with a vapor grown carbon nanofiber in rats. Toxicol. Sci. 2012, 128, 449–460. [Google Scholar] [CrossRef]
- Aiso, S.; Yamazaki, K.; Umeda, Y.; Asakura, M.; Kasai, T.; Takaya, M.; Toya, T.; Koda, S.; Nagano, K.; Arito, H.; et al. Pulmonary toxicity of intratracheally instilled multiwall carbon nanotubes in male fischer 344 rats. Ind. Health 2010, 48, 783–795. [Google Scholar] [CrossRef]
- Morimoto, Y.; Hirohashi, M.; Ogami, A.; Oyabu, T.; Myojo, T.; Todoroki, M.; Yamamoto, M.; Hashiba, M.; Mizuguchi, Y.; Lee, B.W.; et al. Pulmonary toxicity of well-dispersed multi-wall carbon nanotubes following inhalation and intratracheal instillation. Nanotoxicology 2012, 6, 587–599. [Google Scholar] [CrossRef]
- Morimoto, Y.; Izumi, H.; Kuroda, E. Significance of Persistent Inflammation in Respiratory Disorders Induced by Nanoparticles. J. Immunol. Res. 2014. [Google Scholar] [CrossRef] [PubMed]
- Morimoto, Y.; Izumi, H.; Yoshiura, Y.; Tomonaga, T.; Lee, B.W.; Okada, T.; Oyabu, T.; Myojo, T.; Kawai, K.; Yatera, K.; et al. Comparison of pulmonary inflammatory responses following intratracheal instillation and inhalation of nanoparticles. Nanotoxicology 2016, 10, 607–618. [Google Scholar] [CrossRef]
- Morimoto, Y.; Izumi, H.; Yoshiura, Y.; Tomonaga, T.; Oyabu, T.; Myojo, T.; Kawai, K.; Yatera, K.; Shimada, M.; Kubo, M.; et al. Pulmonary toxicity of well-dispersed cerium oxide nanoparticles following intratracheal instillation and inhalation. J. Nanoparticle Res. 2015, 17, 1–16. [Google Scholar] [CrossRef]
- Morimoto, Y.; Izumi, H.; Yoshiura, Y.; Tomonaga, T.; Oyabu, T.; Myojo, T.; Kawai, K.; Yatera, K.; Shimada, M.; Kubo, M.; et al. Evaluation of pulmonary toxicity of zinc oxide nanoparticles following inhalation and intratracheal instillation. Int. J. Mol. Sci. 2016, 17, 1241. [Google Scholar] [CrossRef]
- Keller, J.; Wohlleben, W.; Ma-Hock, L.; Strauss, V.; Gröters, S.; Küttler, K.; Wiench, K.; Herden, C.; Oberdörster, G.; van Ravenzwaay, B.; et al. Time course of lung retention and toxicity of inhaled particles: Short-term exposure to nano-Ceria. Arch. Toxicol. 2014, 88, 2033–2059. [Google Scholar] [CrossRef]
- Cho, W.-S.; Duffin, R.; Howie, S.E.M.; Scotton, C.J.; Wallace, W.A.H.; MacNee, W.; Bradley, M.; Megson, I.L.; Donaldson, K. Progressive severe lung injury by zinc oxide nanoparticles; the role of Zn2+ dissolution inside lysosomes. Part. Fibre Toxicol. 2011, 8, 27. [Google Scholar] [CrossRef]
- Oyabu, T.; Myojo, T.; Lee, B.W.; Okada, T.; Izumi, H.; Yoshiura, Y.; Tomonaga, T.; Li, Y.S.; Kawai, K.; Shimada, M.; et al. Biopersistence of NiO and TiO2 nanoparticles following intratracheal instillation and inhalation. Int. J. Mol. Sci. 2017, 18, 2757. [Google Scholar] [CrossRef]
- Dunnick, J.K.; Elwell, M.R.; Radovsky, A.E.; Benson, J.M.; Hahn, F.F.; Nikula, K.J.; Barr, E.B.; Hobbs, C.H. Comparative Carcinogenic Effects of Nickel Subsulfide, Nickel Oxide, or Nickel Sulfate Hexahydrate Chronic Exposures in the Lung. Cancer Res. 1995, 55, 5251–5256. [Google Scholar] [PubMed]
- Ma, J.Y.C.; Young, S.-H.; Mercer, R.R.; Barger, M.; Schwegler-Berry, D.; Ma, J.K.; Castranova, V. Interactive effects of cerium oxide and diesel exhaust nanoparticles on inducing pulmonary fibrosis HHS Public Access. Toxicol. Appl. Pharmacol. 2014, 278, 135–147. [Google Scholar] [CrossRef] [PubMed]
- Yoshiura, Y.; Izumi, H.; Oyabu, T.; Hashiba, M.; Kambara, T.; Mizuguchi, Y.; Lee, B.W.; Okada, T.; Tomonaga, T.; Myojo, T.; et al. Pulmonary toxicity of well-dispersed titanium dioxide nanoparticles following intratracheal instillation. J. Nanoparticle Res. 2015, 17, 241. [Google Scholar] [CrossRef] [PubMed]
- Kobayashi, N.; Naya, M.; Endoh, S.; Maru, J.; Yamamoto, K.; Nakanishi, J. Comparative pulmonary toxicity study of nano-TiO2 particles of different sizes and agglomerations in rats: Different short- and long-term post-instillation results. Toxicology 2009, 264, 110–118. [Google Scholar] [CrossRef]
- Adamcakova-Dodd, A.; Stebounova, L.V.; Kim, J.S.; Vorrink, S.U.; Ault, A.P.; O’Shaughnessy, P.T.; Grassian, V.H.; Thorne, P.S. Toxicity assessment of zinc oxide nanoparticles using sub-acute and sub-chronic murine inhalation models. Part. Fibre Toxicol. 2014, 11, 15. [Google Scholar] [CrossRef]
- TORAY. 3D-Gene®. Available online: https://www.3d-gene.com/en/#/ (accessed on 28 May 2020).
- DAVID. Functional Annotation Bioinformatics Microarray Analysis. Available online: https://david.ncifcrf.gov/ (accessed on 28 May 2020).
- Sasaki, S.; Izumi, H.; Morimoto, Y.; Sakurai, K.; Mochizuki, S. Induction of potent cell growth inhibition by schizophyllan/K-ras antisense complex in combination with gemcitabine. Bioorganic Med. Chem. 2020, 28, 115668. [Google Scholar] [CrossRef]
- Johnston, B.; Butcher, E.C. Chemokines in rapid leukocyte adhesion triggering and migration. Semin. Immunol. 2002, 14, 83–92. [Google Scholar] [CrossRef]
- Gasperini, S.; Marchi, M.; Calzetti, F.; Laudanna, C.; Vicentini, L.; Olsen, H.; Murphy, M.; Liao, F.; Farber, J.; Marco, C.A. Gene expression and production of the monokine induced by IFN-gamma (MIG), IFN-inducible T cell alpha chemoattractant (I-TAC), and IFN-gamma-inducible protein-10 (IP-10) chemokines by human neutrophils. J. Immunol. 1999, 162, 4928–4937. [Google Scholar] [PubMed]
- Hildebrandt, G.C.; Corrion, L.A.; Olkiewicz, K.M.; Lu, B.; Lowler, K.; Duffner, U.A.; Moore, B.B.; Kuziel, W.A.; Liu, C.; Cooke, K.R. Blockade of CXCR3 Receptor: Ligand Interactions Reduces Leukocyte Recruitment to the Lung and the Severity of Experimental Idiopathic Pneumonia Syndrome. J. Immunol. 2004, 173, 2050–2059. [Google Scholar] [CrossRef] [PubMed]
- Takaku, Y.; Soma, T.; Uchida, Y.; Kobayashi, T.; Nakagome, K.; Nagata, M. CXC chemokine superfamily induced by Interferon-γ in asthma: A cross-sectional observational study. Asthma Res. Pract. 2016, 2, 6. [Google Scholar] [CrossRef] [PubMed]
- Jeyaseelan, S.; Manzer, R.; Young, S.K.; Yamamoto, M.; Akira, S.; Mason, R.J.; Worthen, G.S. Induction of CXCL5 during inflammation in the rodent lung involves activation of alveolar epithelium. Am. J. Respir. Cell Mol. Biol. 2005, 32, 531–539. [Google Scholar] [CrossRef]
- Fujita, K.; Fukuda, M.; Fukui, H.; Horie, M.; Endoh, S.; Uchida, K.; Shichiri, M.; Morimoto, Y.; Ogami, A.; Iwahashi, H. Intratracheal instillation of single-wall carbon nanotubes in the rat lung induces time-dependent changes in gene expression. Nanotoxicology 2015, 9, 290–301. [Google Scholar] [CrossRef]
- Oh, J.H.; Yang, M.J.; Yang, Y.S.; Park, H.J.; Heo, S.H.; Lee, E.H.; Song, C.W.; Yoon, S. Microarray-based analysis of the lung recovery process after stainless-steel welding fume exposure in Sprague-Dawley rats. Inhal. Toxicol. 2009, 21, 347–373. [Google Scholar] [CrossRef]
- Balamayooran, G.; Batra, S.; Cai, S.; Mei, J.; Worthen, G.S.; Penn, A.L.; Jeyaseelan, S. Role of CXCL5 in leukocyte recruitment to the lungs during secondhand smoke exposure. Am. J. Respir. Cell Mol. Biol. 2012, 47, 104–111. [Google Scholar] [CrossRef]
- Fujita, K.; Morimoto, Y.; Endoh, S.; Uchida, K.; Fukui, H.; Ogami, A.; Tanaka, I.; Horie, M.; Yoshida, Y.; Iwahashi, H.; et al. Identification of potential biomarkers from gene expression profiles in rat lungs intratracheally instilled with C60 fullerenes. Toxicology 2010, 274, 34–41. [Google Scholar] [CrossRef]
- Proost, P.; Wuyts, A.; van Damme, J. Human monocyte chemotactic proteins-2 and -3: Structural and functional comparison with MCP-1. J. Leukoc. Biol. 1996, 59, 67–74. [Google Scholar] [CrossRef]
- Langley, R.J.; Mishra, N.C.; Peña-Philippides, J.C.; Rice, B.J.; Seagrave, J.C.; Singh, S.P.; Sopori, M.L. Fibrogenic and redox-related but not proinflammatory genes are upregulated in Lewis rat model of chronic silicosis. J. Toxicol. Environ. Heal. Part A 2011, 74, 1261–1279. [Google Scholar] [CrossRef]
- Fujita, K.; Fukuda, M.; Endoh, S.; Maru, J.; Kato, H.; Nakamura, A.; Shinohara, N.; Uchino, K.; Honda, K. Pulmonary and pleural inflammation after intratracheal instillation of short single-walled and multi-walled carbon nanotubes. Toxicol. Lett. 2016, 257, 23–37. [Google Scholar] [CrossRef]
- Abdelgied, M.; El-Gazzar, A.M.; Alexander, D.B.; Alexander, W.T.; Numano, T.; Iigou, M.; Naiki-Ito, A.; Takase, H.; Abdou, K.A.; Hirose, A.; et al. Potassium octatitanate fibers induce persistent lung and pleural injury and are possibly carcinogenic in male Fischer 344 rats. Cancer Sci. 2018, 109, 2164–2177. [Google Scholar] [CrossRef]
- Belperio, J.A.; Keane, M.P.; Burdick, M.D.; Lynch, J.P.; Xue, Y.Y.; Li, K.; Ross, D.J.; Strieter, R.M. Critical role for CXCR3 chemokine biology in the pathogenesis of bronchiolitis obliterans syndrome. J. Immunol. 2002, 169, 1037–1049. [Google Scholar] [CrossRef] [PubMed]
- Oyabu, T.; Morimoto, Y.; Hirohashi, M.; Horie, M.; Kambara, T.; Lee, B.W.; Hashiba, M.; Mizuguchi, Y.; Myojo, T.; Kuroda, E. Dose-dependent pulmonary response of well-dispersed titanium dioxide nanoparticles following intratracheal instillation. J. Nanoparticle Res. 2013, 15. [Google Scholar] [CrossRef]
- Morrow, P.E.; Muhle, H.; Mermelstein, R. Chronic Inhalation Study Findings as a Basis for Proposing a New Occupational Dust Exposure Limit. J. Am. Coll. Toxicol. 1991, 10, 279–290. [Google Scholar] [CrossRef]
- Bellmann, B.; Muhle, H.; Creutzenberg, O.; Mermelstein, R. Irreversible pulmonary changes induced in rat lung by dust overload. Environ. Heal. Perspect. 1992, 97, 189–191. [Google Scholar] [CrossRef] [PubMed]
- Thiefes, A.; Wolter, S.; Mushinski, J.F.; Hoffmann, E.; Dittrich-Breiholz, O.; Graue, N.; Dörrie, A.; Schneider, H.; Wirth, D.; Luckow, B.; et al. Simultaneous blockade of NFκB, JNK, and p38 MAPK by a kinase-inactive mutant of the protein kinase TAK1 sensitizes cells to apoptosis and affects a distinct spectrum of tumor necrosis target genes. J. Biol. Chem. 2005, 280, 27728–27741. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Liang, C.; Zhang, Z.K.; Pan, X.; Peng, S.; Lee, W.S.; Lu, A.; Lin, Z.; Zhang, G.; Leung, W.N.; et al. TAK1 inhibition attenuates both inflammation and fibrosis in experimental pneumoconiosis. Cell Discov. 2017, 3, 1–21. [Google Scholar] [CrossRef]

| Nanomaterials | NiO | CeO2 | TiO2 | ZnO |
|---|---|---|---|---|
| Pulmonary toxicity | High | High | Low | Low |
| Primary diameter | 19 nm | 7.8 nm | Short 12 nm Long 55 nm | 35 nm |
| Specific surface area | 57 m2/g | 101 m2/g | 111 m2/g | 31 m2/g |
| Shape | Sphere | Irregular shape | Spindle-shaped | Sphere like |
| Secondary diameter (DLS) | 39.8–47.1 nm | 2.6–9.3 nm | 20–80 nm | 17–37 nm |
| Purity | More than 99.5% | 99.9% | 99.5% | 99.9% |
| Bulk density | 6.72 g/cm3 | 7.22 g/cm3 | 4.17 g/cm3 | 5.60 g/cm3 |
| Solubility | Low (>CeO2) | Low | Low | High |
| (A) | |||
|---|---|---|---|
| mRNA Level (Fold Change of Control) | Number of Genes | ||
| Up regulation | ≧2-fold | 450 | |
| 2–4-fold | 383 | ||
| 4–8-fold | 51 | ||
| ≧8-fold | 16 | ||
| Down regulation | ≦-fold | 388 | |
| -fold | 359 | ||
| -fold | 15 | ||
| ≦-fold | 14 | ||
| (B) | |||
| Gene Symbol | Gene Description | Fold Change | |
| CXCL5 | Chemokine (C-X-C motif) ligand5 | 12.83 | |
| CCL2 | Chemokine (C-C motif) ligand2 | 17.93 | |
| CCL7 | Chemokine (C-C motif) ligand7 | 23.44 | |
| CXCL10 | Chemokine (C-X-C motif) ligand10 | 16.42 | |
| CXCL11 | Chemokine (C-X-C motif) ligand11 | 16.70 | |
| Observation Time | 3 Days | 1 Week | 1 Month | 3 Months | 6 Months |
|---|---|---|---|---|---|
| AUC | AUC | AUC | AUC | AUC | |
| (95% C.I.) | (95% C.I.) | (95% C.I.) | (95% C.I.) | (95% C.I.) | |
| (Cut-off Value) | (Cut-off Value) | (Cut-off Value) | (Cut-off Value) | (Cut-off Value) | |
| CXCL5 | 0.990 | 0.998 | 0.995 | 0.983 | 0.928 |
| (0.982–0.999) | (0.961–1.000) | (0.948–1.000) | (0.874–0.998) | (0.803–0.976) | |
| (10.008) | (10.308) | (9.513) | (3.277) | (1.934)) | |
| CCL2 | 0.693 | 0.973 | 0.980 | 0.910 | 0.848 |
| (0.501–0.835) | (0.841–0.996) | (0.904–0.996) | (0.753–0.971) | (0.674–0.937) | |
| (11.495) | (3.919) | (1.945) | (2.059) | (1.076) | |
| CCL7 | 0.680 | 0.968 | 0.993 | 0.898 | 0.893 |
| (0.487–0.826) | (0.860–0.993) | (0.933–0.999) | (0.744–0.963) | (0.720–0.964) | |
| (12.380) | (5.090) | (1.990) | (2.007) | (1.020) | |
| CXCL10 | 0.603 | 0.795 | 0.848 | 0.778 | 0.663 |
| (0.415–0.764) | (0.592–0.912) | (0.673–0.974) | (0.593–0.894) | (0.473–0.811) | |
| (8.311) | (2.088) | (1.620) | (1.466) | (1.097) | |
| CXCL11 | 0.525 | 0.890 | 0.883 | 0.813 | 0.920 |
| (0.340–0.704) | (0.744–0.958) | (0.741–0.952) | (0.612–0.922) | (0.798–0.971) | |
| (7.843) | (2.790) | (2.073) | (1.025) | (0.706) |
| Sensitivity | Specificity | False Positive | False Negative | ||||||
|---|---|---|---|---|---|---|---|---|---|
| 3 days | CXCL5 | 0.95 | (19/20) | 1.00 | (20/20) | 0.00 | (0/20) | 0.05 | (1/20) |
| CCL2 | 0.50 | (10/20) | 0.95 | (19/20) | 0.05 | (1/20) | 0.50 | (10/20) | |
| CCL7 | 0.50 | (10/20) | 0.95 | (19/20) | 0.05 | (1/20) | 0.50 | (10/20) | |
| CXCL10 | 0.30 | (6/20) | 1.00 | (20/20) | 0.00 | (0/20) | 0.70 | (14/20) | |
| CXCL11 | 0.35 | (7/20) | 0.95 | (19/20) | 0.05 | (1/20) | 0.65 | (13/20) | |
| CXCL5 + CCL2 + CCL7 | 0.90 | (18/20) | 0.05 | (1/20) | 0.95 | (19/20) | 0.10 | (2/20) | |
| 1 week | CXCL5 | 0.95 | (19/20) | 1.00 | (20/20) | 0.00 | (0/20) | 0.05 | (1/20) |
| CCL2 | 1.00 | (20/20) | 0.90 | (18/20) | 0.10 | (2/20) | 0.00 | (0/20) | |
| CCL7 | 0.95 | (19/20) | 0.90 | (18/20) | 0.10 | (2/20) | 0.05 | (1/20) | |
| CXCL10 | 0.75 | (15/20) | 0.90 | (18/20) | 0.10 | (2/20) | 0.25 | (5/20) | |
| CXCL11 | 0.75 | (15/20) | 0.95 | (19/20) | 0.05 | (1/20) | 0.25 | (5/20) | |
| CXCL5 + CCL2 + CCL7 | 1.00 | (20/20) | 0.90 | (18/20) | 0.10 | (2/20) | 0.00 | (0/20) | |
| 1 month | CXCL5 | 0.95 | (19/20) | 1.00 | (20/20) | 0.00 | (0/20) | 0.05 | (1/20) |
| CCL2 | 1.00 | (20/20) | 0.90 | (18/20) | 0.10 | (2/20) | 0.00 | (0/20) | |
| CCL7 | 1.00 | (20/20) | 0.95 | (19/20) | 0.05 | (1/20) | 0.00 | (0/20) | |
| CXCL10 | 0.65 | (13/20) | 1.00 | (20/20) | 0.00 | (0/20) | 0.35 | (7/20) | |
| CXCL11 | 0.65 | (13/20) | 0.95 | (19/20) | 0.05 | (1/20) | 0.35 | (7/20) | |
| CXCL5 + CCL2 + CCL7 | 1.00 | (20/20) | 0.90 | (18/20) | 0.10 | (2/20) | 0.00 | (0/20) | |
| 3 months | CXCL5 | 1.00 | (20/20) | 0.95 | (19/20) | 0.05 | (1/20) | 0.00 | (0/20) |
| CCL2 | 0.95 | (19/20) | 0.80 | (16/20) | 0.20 | (4/20) | 0.50 | (1/20) | |
| CCL7 | 0.95 | (19/20) | 0.80 | (16/20) | 0.20 | (4/20) | 0.50 | (1/20) | |
| CXCL10 | 0.75 | (15/20) | 0.80 | (16/20) | 0.20 | (4/20) | 0.25 | (5/20) | |
| CXCL11 | 0.90 | (18/20) | 0.75 | (15/20) | 0.25 | (5/20) | 0.10 | (2/20) | |
| CXCL5 + CCL2 + CCL7 | 1.00 | (20/20) | 0.80 | (16/20) | 0.20 | (4/20) | 0.00 | (0/20) | |
| 6 months | CXCL5 | 0.95 | (19/20) | 0.80 | (16/20) | 0.20 | (4/20) | 0.05 | (1/20) |
| CCL2 | 0.85 | (17/20) | 0.80 | (16/20) | 0.20 | (4/20) | 0.15 | (3/20) | |
| CCL7 | 0.85 | (17/20) | 0.85 | (17/20) | 0.15 | (3/20) | 0.15 | (3/20) | |
| CXCL10 | 0.55 | (11/20) | 0.80 | (16/20) | 0.20 | (4/20) | 0.45 | (9/20) | |
| CXCL11 | 1.00 | (20/20) | 0.70 | (14/20) | 0.30 | (6/20) | 0.00 | (0/20) | |
| CXCL5 + CCL2 + CCL7 | 1.00 | (20/20) | 0.30 | (6/20) | 0.70 | (14/20) | 0.00 | (0/20) | |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Nishida, C.; Izumi, H.; Tomonaga, T.; Takeshita, J.-i.; Wang, K.-Y.; Yamasaki, K.; Yatera, K.; Morimoto, Y. Predictive Biomarkers for the Ranking of Pulmonary Toxicity of Nanomaterials. Nanomaterials 2020, 10, 2032. https://doi.org/10.3390/nano10102032
Nishida C, Izumi H, Tomonaga T, Takeshita J-i, Wang K-Y, Yamasaki K, Yatera K, Morimoto Y. Predictive Biomarkers for the Ranking of Pulmonary Toxicity of Nanomaterials. Nanomaterials. 2020; 10(10):2032. https://doi.org/10.3390/nano10102032
Chicago/Turabian StyleNishida, Chinatsu, Hiroto Izumi, Taisuke Tomonaga, Jun-ichi Takeshita, Ke-Yong Wang, Kei Yamasaki, Kazuhiro Yatera, and Yasuo Morimoto. 2020. "Predictive Biomarkers for the Ranking of Pulmonary Toxicity of Nanomaterials" Nanomaterials 10, no. 10: 2032. https://doi.org/10.3390/nano10102032
APA StyleNishida, C., Izumi, H., Tomonaga, T., Takeshita, J.-i., Wang, K.-Y., Yamasaki, K., Yatera, K., & Morimoto, Y. (2020). Predictive Biomarkers for the Ranking of Pulmonary Toxicity of Nanomaterials. Nanomaterials, 10(10), 2032. https://doi.org/10.3390/nano10102032






