Temperature Sensing in the Short-Wave Infrared Spectral Region Using Core-Shell NaGdF4:Yb3+, Ho3+, Er3+@NaYF4 Nanothermometers
Abstract
:1. Introduction
2. Materials and Methods
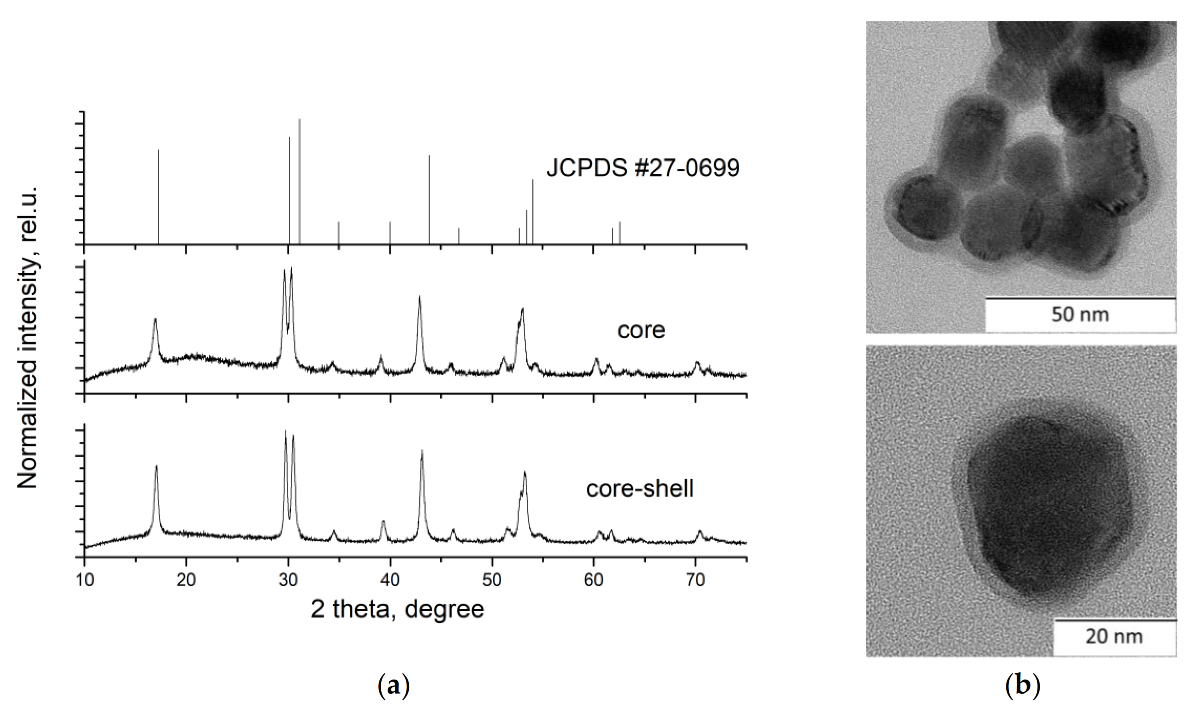
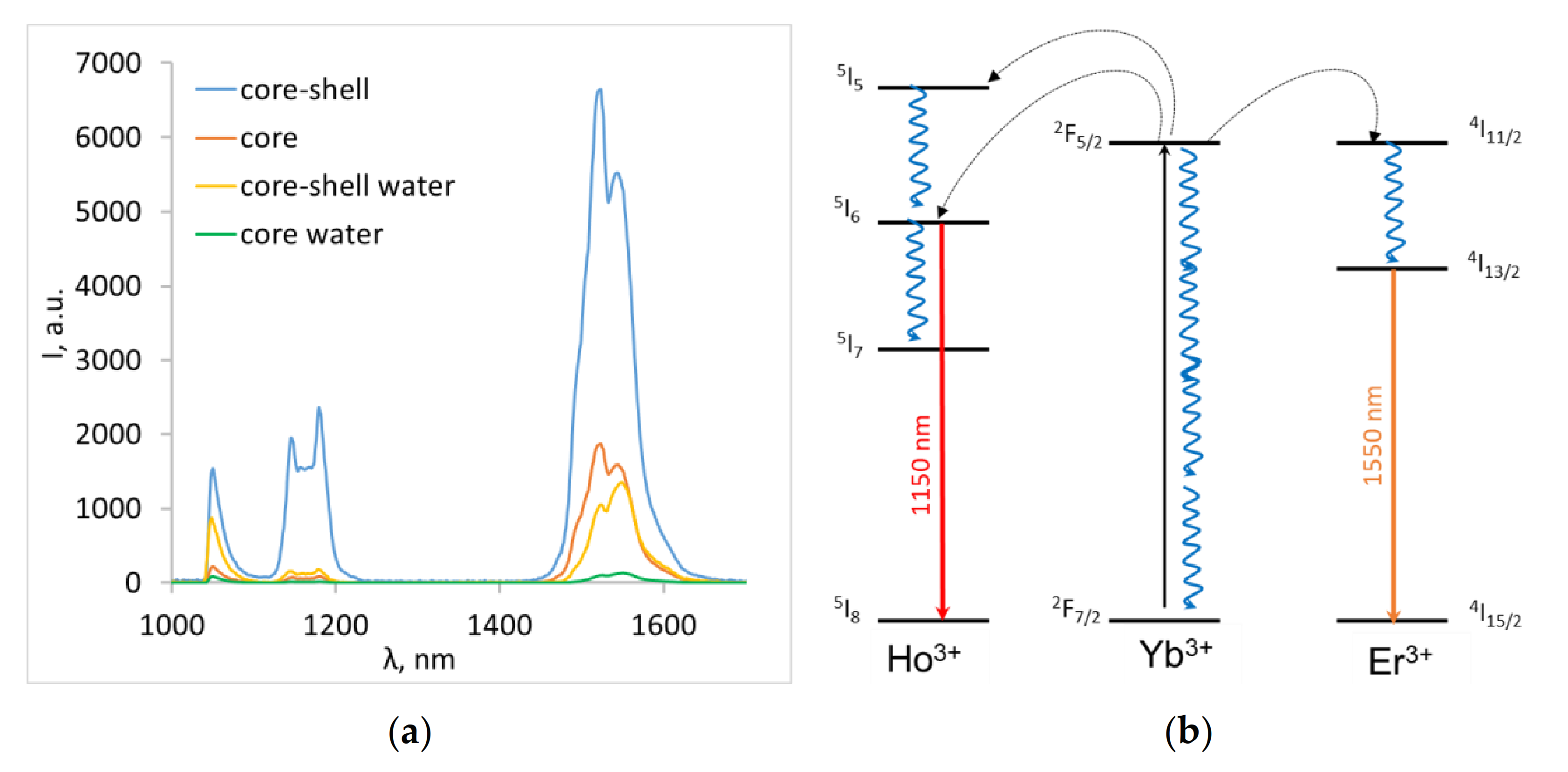
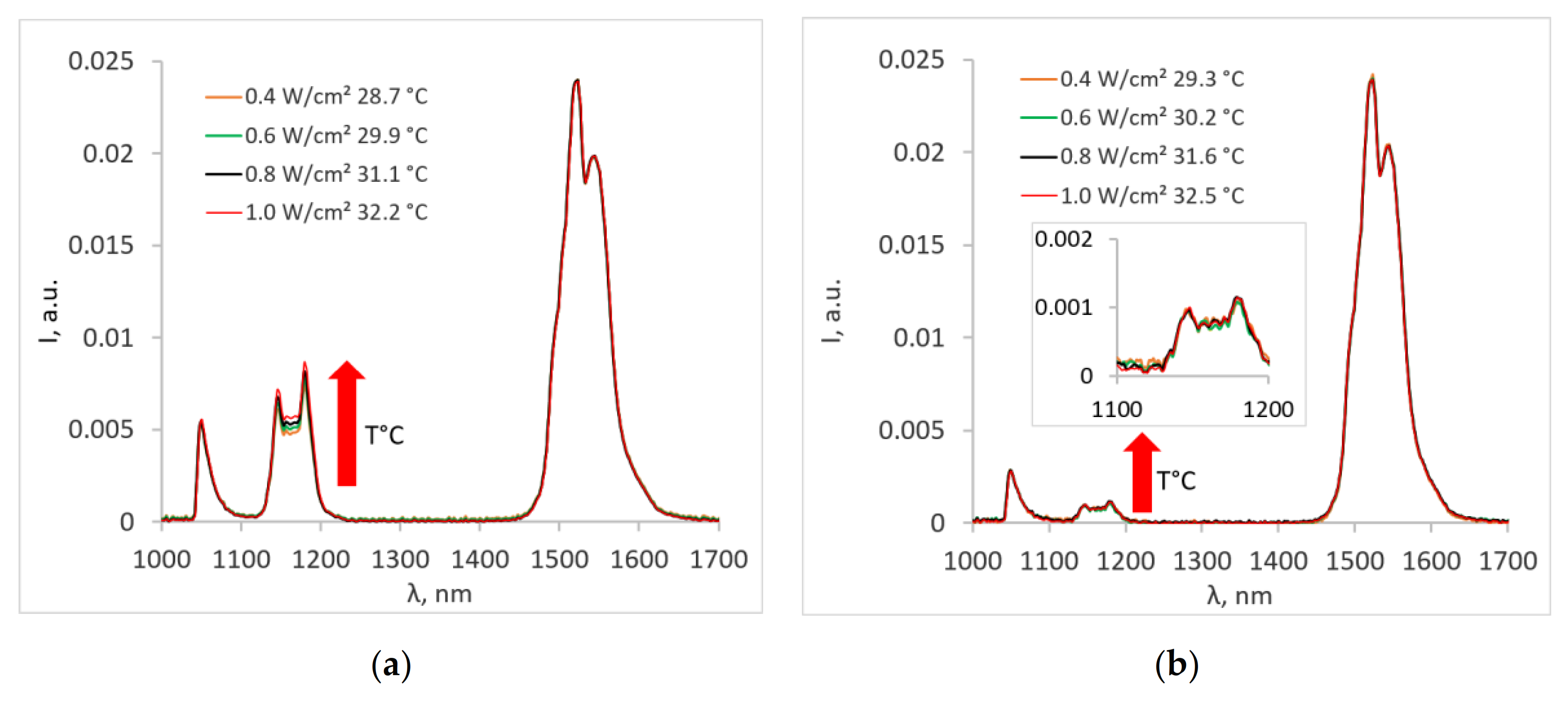
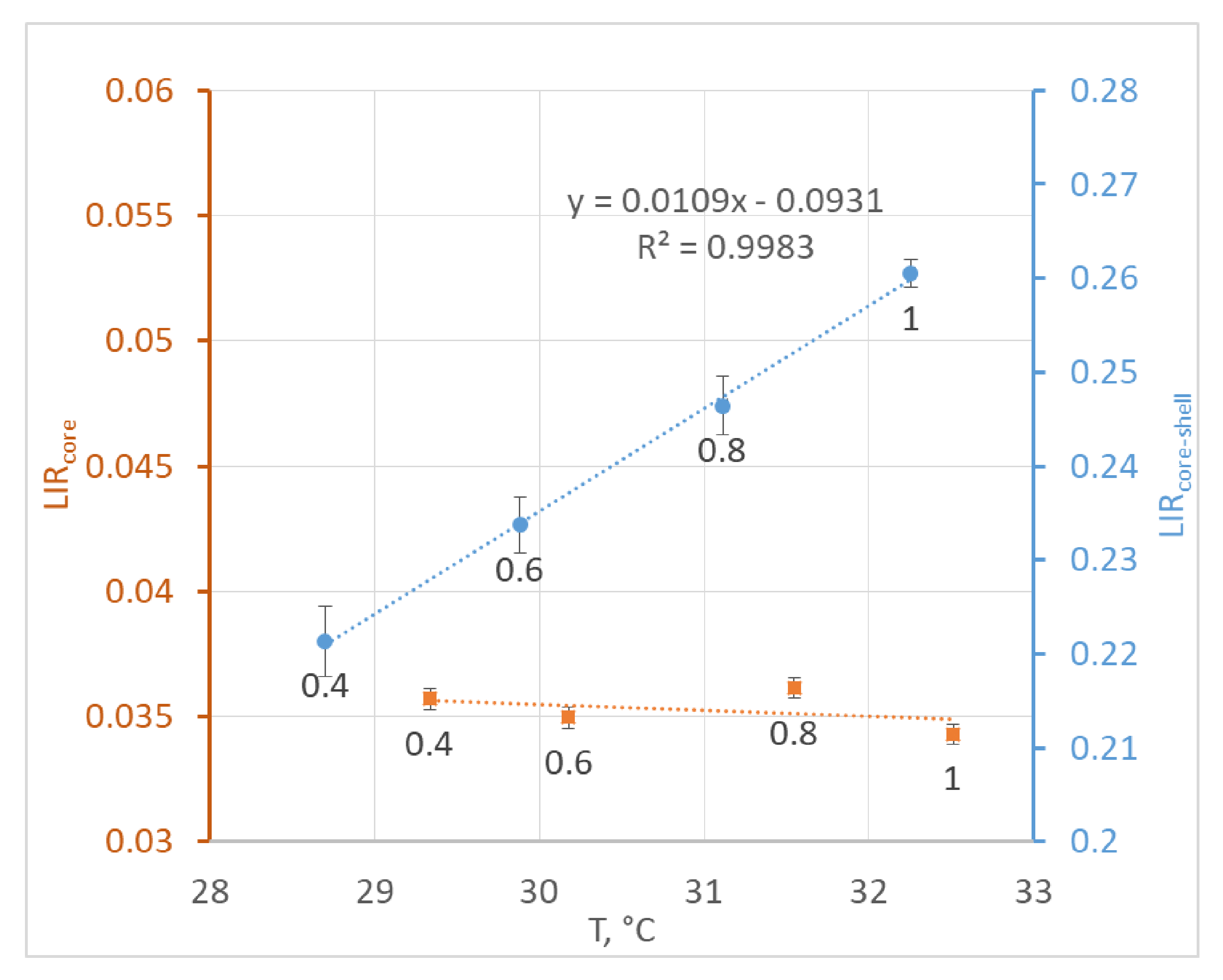
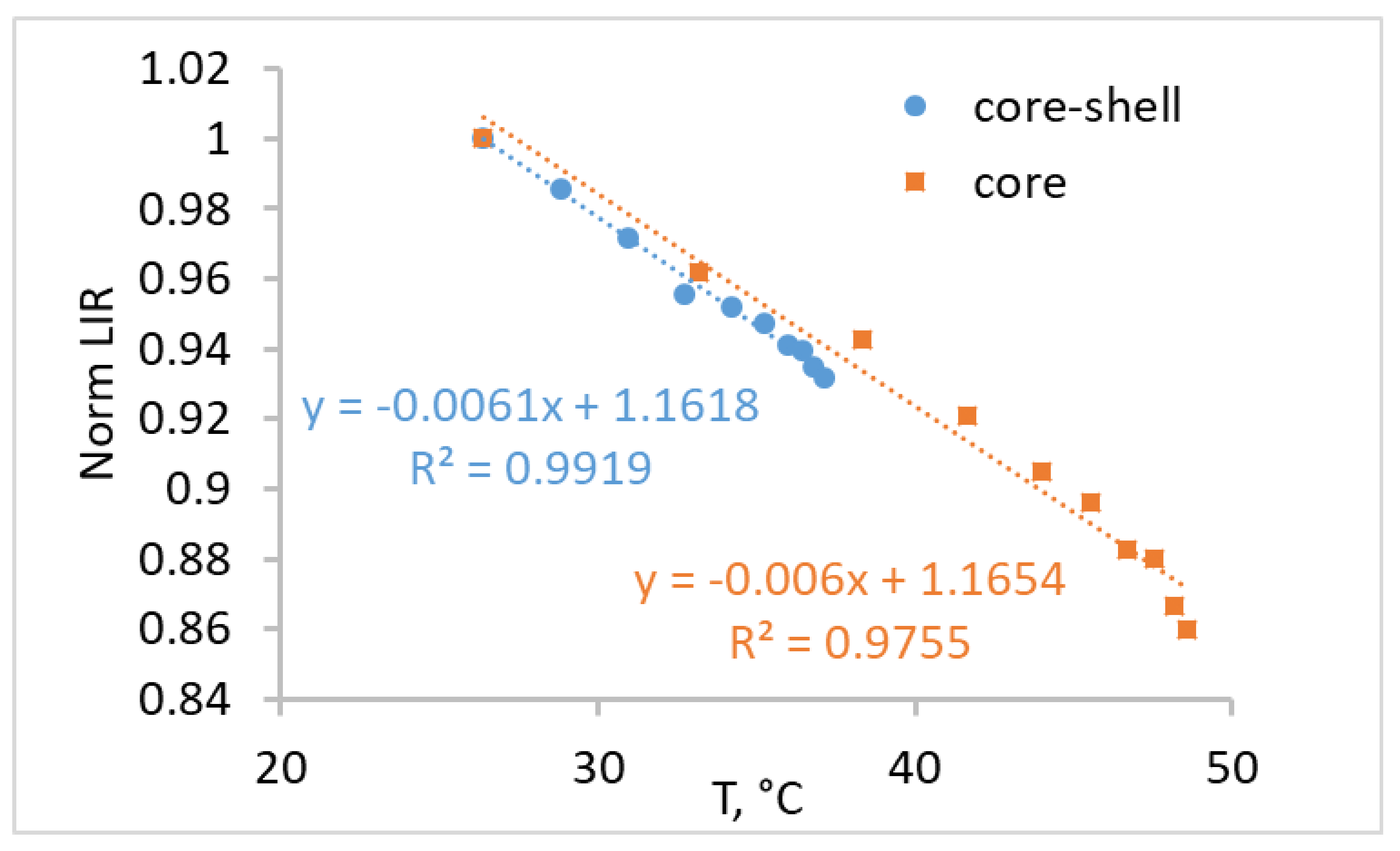
3. Results and Discussion
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Brites, C.D.S.; Martínez, E.D.; Urbano, R.R.; Rettori, C.; Carlos, L.D. Self-calibrated double luminescent thermometers through upconverting nanoparticles. Front. Chem. 2019, 7. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Brites, C.D.S.; Balabhadra, S.; Carlos, L.D. Lanthanide-Based Thermometers: At the Cutting-Edge of Luminescence Thermometry. Adv. Opt. Mater. 2019, 7, 1801239. [Google Scholar] [CrossRef] [Green Version]
- Brites, C.D.S.; Kuznetsov, S.V.; Konyushkin, V.A.; Nakladov, A.N.; Fedorov, P.P.; Carlos, L.D. Simultaneous Measurement of the Emission Quantum Yield and Local Temperature: The Illustrative Example of SrF2:Yb3+/Er3+ Single Crystals. Eur. J. Inorg. Chem. 2020. [Google Scholar] [CrossRef]
- Liang, L.; Liu, X. Nanocrystals feel the heat. Nat. Photonics 2018, 12, 124–125. [Google Scholar] [CrossRef]
- Kolesnikov, I.E.; Kalinichev, A.A.; Kurochkin, M.A.; Mamonova, D.V.; Kolesnikov, E.Y.; Kurochkin, A.V.; Lähderanta, E.; Mikhailov, M.D. Y2O3:Nd3+ nanocrystals as ratiometric luminescence thermal sensors operating in the optical windows of biological tissues. J. Lumin. 2018, 204, 506–512. [Google Scholar] [CrossRef]
- Lal, S.; Clare, S.E.; Halas, N.J. Nanoshell-enabled photothermal cancer therapy: Impending clinical impact. Acc. Chem. Res. 2008, 41, 1842–1851. [Google Scholar] [CrossRef] [PubMed]
- Lindquist, S. The heat-shock response. Ann. Rev. Biochem. 1986, 55, 1151–1191. [Google Scholar] [CrossRef]
- Begasse, M.L.; Leaver, M.; Vazquez, F.; Grill, S.W.; Hyman, A.A. Temperature dependence of cell division timing accounts for a shift in the thermal limits of c. elegans and C.briggsae. Cell Rep. 2015, 10, 647–653. [Google Scholar] [CrossRef] [Green Version]
- Jaque, D.; Martínez Maestro, L.; Del Rosal, B.; Haro-Gonzalez, P.; Benayas, A.; Plaza, J.L.; Martín Rodríguez, E.; Solé, J.G. Nanoparticles for photothermal therapies. Nanoscale 2014, 6, 9494–9530. [Google Scholar] [CrossRef]
- Ai, X.; Mu, J.; Xing, B. Recent advances of light-mediated theranostics. Theranostics 2016, 6, 2439–2457. [Google Scholar] [CrossRef]
- Hemmer, E.; Acosta-Mora, P.; Méndez-Ramos, J.; Fischer, S. Optical nanoprobes for biomedical applications: Shining a light on upconverting and near-infrared emitting nanoparticles for imaging, thermal sensing, and photodynamic therapy. J. Mater. Chem. B 2017, 5, 4365–4392. [Google Scholar] [CrossRef] [PubMed]
- Liu, H.; Fan, Y.; Wang, J.; Song, Z.; Shi, H.; Han, R.; Sha, Y.; Jiang, Y. Intracellular Temperature Sensing: An Ultra-bright Luminescent Nanothermometer with Non-sensitivity to pH and Ionic Strength. Sci. Rep. 2015, 5. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Maestro, L.M.; Rodríguez, E.M.; Rodríguez, F.S.; De La Cruz, M.C.I.; Juarranz, A.; Naccache, R.; Vetrone, F.; Jaque, D.; Capobianco, J.A.; Solé, J.G. CdSe quantum dots for two-photon fluorescence thermal imaging. Nano Lett. 2010, 10, 5109–5115. [Google Scholar] [CrossRef] [PubMed]
- De La Rosa, V.R.; Woisel, P.; Hoogenboom, R. Supramolecular control over thermoresponsive polymers. Mater. Today 2016, 19, 44–55. [Google Scholar] [CrossRef]
- Yang, G.; Liu, X.; Feng, J.; Li, S.; Li, Y. Organic Dye Thermometry. In Thermometry at the Nanoscale: Techniques and Selected Applications; Carlos, L.D., Palacio, F., Eds.; The Royal Society of Chemistry: London, UK, 2016; Volume 38, pp. 167–189. [Google Scholar]
- Paviolo, C.; Clayton, A.H.A.; Mcarthur, S.L.; Stoddart, P.R. Temperature measurement in the microscopic regime: A comparison between fluorescence lifetime- and intensity-based methods. J. Microsc. 2013, 250, 179–188. [Google Scholar] [CrossRef]
- Uchiyama, S.; Kawai, N.; De Silva, A.P.; Iwai, K. Fluorescent Polymeric and Logic Gate with Temperature and pH as Inputs. J. Am. Chem. Soc. 2004, 126, 3032–3033. [Google Scholar] [CrossRef]
- Okabe, K.; Inada, N.; Gota, C.; Harada, Y.; Funatsu, T.; Uchiyama, S. Intracellular temperature mapping with a fluorescent polymeric thermometer and fluorescence lifetime imaging microscopy. Nat. Commun. 2012, 3. [Google Scholar] [CrossRef] [Green Version]
- Zarrintaj, P.; Jouyandeh, M.; Ganjali, M.R.; Hadavand, B.S.; Mozafari, M.; Sheiko, S.S.; Vatankhah-Varnoosfaderani, M.; Gutiérrez, T.J.; Saeb, M.R. Thermo-sensitive polymers in medicine: A review. Eur. Polym. J. 2019, 117, 402–423. [Google Scholar] [CrossRef]
- Donner, J.S.; Thompson, S.A.; Kreuzer, M.P.; Baffou, G.; Quidant, R. Mapping intracellular temperature using green fluorescent protein. Nano Lett. 2012, 12, 2107–2111. [Google Scholar] [CrossRef] [Green Version]
- Zhu, X.; Feng, W.; Chang, J.; Tan, Y.W.; Li, J.; Chen, M.; Sun, Y.; Li, F. Temperature-feedback upconversion nanocomposite for accurate photothermal therapy at facile temperature. Nat. Commun. 2016, 7. [Google Scholar] [CrossRef]
- Vetrone, F.; Naccache, R.; Zamarrón, A.; De La Fuente, A.J.; Sanz-Rodríguez, F.; Maestro, L.M.; Rodriguez, E.M.; Jaque, D.; Sole, J.G.; Capobianco, J.A. Temperature sensing using fluorescent nanothermometers. ACS Nano 2010, 4, 3254–3258. [Google Scholar] [CrossRef] [PubMed]
- Weissleder, R. A clearer vision for in vivo imaging: Progress continues in the development of smaller, more penetrable probes for biological imaging. Nat. Biotechnol. 2001, 19, 316–317. [Google Scholar] [CrossRef] [PubMed]
- Jacques, S.L. Optical properties of biological tissues: A review. Phys. Med. Biol. 2013, 58, 5007–5008. [Google Scholar] [CrossRef]
- Wang, M.; Wu, C.; Sinefeld, D.; Li, B.; Xia, F.; Xu, C. Comparing the effective attenuation lengths for long wavelength in vivo imaging of the mouse brain. Biomed. Opt. Express 2018, 9, 3534–3543. [Google Scholar] [CrossRef] [PubMed]
- Mustafa, F.H.; Jaafar, M.S. Comparison of wavelength-dependent penetration depths of lasers in different types of skin in photodynamic therapy. Indian J. Phys. 2013, 87, 203–209. [Google Scholar] [CrossRef]
- Hong, G.; Diao, S.; Chang, J.; Antaris, A.L.; Chen, C.; Zhang, B.; Zhao, S.; Atochin, D.N.; Huang, P.L.; Andreasson, K.I.; et al. Through-skull fluorescence imaging of the brain in a new near-infrared window. Nat. Photonics 2014, 8, 723–730. [Google Scholar] [CrossRef]
- Zhan, Q.; He, S.; Qian, J.; Cheng, H.; Cai, F. Optimization of optical excitation of upconversion nanoparticles for rapid microscopy and deeper tissue imaging with higher quantum yield. Theranostics 2013, 3, 306–316. [Google Scholar] [CrossRef] [Green Version]
- Hemmer, E.; Benayas, A.; Légaré, F.; Vetrone, F. Exploiting the biological windows: Current perspectives on fluorescent bioprobes emitting above 1000 nm. Nanoscale Horiz. 2016, 1, 168–184. [Google Scholar] [CrossRef]
- Semonin, O.E.; Johnson, J.C.; Luther, J.M.; Midgett, A.G.; Nozik, A.J.; Beard, M.C. Absolute photoluminescence quantum yields of IR-26 Dye, PbS, and PbSe quantum dots. J. Phys. Chem. Lett. 2010, 1, 2445–2450. [Google Scholar] [CrossRef]
- Hatami, S.; Würth, C.; Kaiser, M.; Leubner, S.; Gabriel, S.; Bahrig, L.; Lesnyak, V.; Pauli, J.; Gaponik, N.; Eychmüller, A.; et al. Absolute photoluminescence quantum yields of IR26 and IR-emissive Cd1-xHgxTe and PbS quantum dots-method- and material-inherent challenges. Nanoscale 2015, 7, 133–143. [Google Scholar] [CrossRef] [Green Version]
- Thimsen, E.; Sadtler, B.; Berezin, M.Y. Shortwave-infrared (SWIR) emitters for biological imaging: A review of challenges and opportunities. Nanophotonics 2017, 6, 1043–1054. [Google Scholar] [CrossRef]
- Escudero, A.; Carrillo-Carrión, C.; Zyuzin, M.V.; Parak, W.J. Luminescent Rare-earth-based Nanoparticles: A Summarized Overview of their Synthesis, Functionalization, and Applications. Top. Curr. Chem. 2016, 374, 48. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ma, D.; Xu, X.; Hu, M.; Wang, J.; Zhang, Z.; Yang, J.; Meng, L. Rare-Earth-Based Nanoparticles with Simultaneously Enhanced Near-Infrared (NIR)-Visible (Vis) and NIR-NIR Dual-Conversion Luminescence for Multimodal Imaging. Chem. Asian J. 2016, 11, 1050–1058. [Google Scholar] [CrossRef] [PubMed]
- Zhou, H.; Sharma, M.; Berezin, O.; Zuckerman, D.; Berezin, M.Y. Nanothermometry: From Microscopy to Thermal Treatments. ChemPhysChem 2016, 17, 27–36. [Google Scholar] [CrossRef]
- Wade, S.A.; Collins, S.F.; Baxter, G.W. Fluorescence intensity ratio technique for optical fiber point temperature sensing. J. Appl. Phys. 2003, 94, 4743–4756. [Google Scholar] [CrossRef]
- Quintanilla, M.; Benayas, A.; Naccache, R.; Vetrone, F. Luminescent Nanothermometry with Lanthanide-doped Nanoparticles. In Thermometry at the Nanoscale: Techniques and Selected Applications; Carlos, L.D., Palacio, F., Eds.; The Royal Society of Chemistry: London, UK, 2016; Volume 38, pp. 124–166. [Google Scholar]
- Kamimura, M.; Matsumoto, T.; Suyari, S.; Umezawa, M.; Soga, K. Ratiometric near-infrared fluorescence nanothermometry in the OTN-NIR (NIR II/III) biological window based on rare-earth doped β-NaYF 4 nanoparticles. J. Mater. Chem. B 2017, 5, 1917–1925. [Google Scholar] [CrossRef]
- Gouveia-Neto, A.S.; Silva, J.F.; Vermelho, M.V.D.; Gomes, A.S.L.; Jacinto, C. Generation of multiwavelength light in the region of the biological windows in Tm3+-doped fiber excited at 1.064 μm. Appl. Phys. Lett. 2016, 109. [Google Scholar] [CrossRef]
- Smith, A.M.; Mancini, M.C.; Nie, S. Bioimaging: Second window for in vivo imaging. Nat. Nanotechnol. 2009, 4, 710–711. [Google Scholar] [CrossRef] [Green Version]
- Naczynski, D.J.; Tan, M.C.; Zevon, M.; Wall, B.; Kohl, J.; Kulesa, A.; Chen, S.; Roth, C.M.; Riman, R.E.; Moghe, P.V. Rare-earth-doped biological composites as in vivo shortwave infrared reporters. Nat. Commun. 2013, 4. [Google Scholar] [CrossRef]
- Sekiyama, S.; Umezawa, M.; Kuraoka, S.; Ube, T.; Kamimura, M.; Soga, K. Temperature Sensing of Deep Abdominal Region in Mice by Using Over-1000 nm Near-Infrared Luminescence of Rare-Earth-Doped NaYF4 Nanothermometer. Sci. Rep. 2018, 8, 1–12. [Google Scholar] [CrossRef]
- Wortmann, L.; Suyari, S.; Ube, T.; Kamimura, M.; Soga, K. Tuning the thermal sensitivity of β-NaYF4:Yb3+, Ho3+, Er3+ nanothermometers for optimal temperature sensing in OTN-NIR (NIR II/III) biological window. J. Lumin. 2018, 198, 236–242. [Google Scholar] [CrossRef]
- Ximendes, E.C.; Rocha, U.; Sales, T.O.; Fernández, N.; Sanz-Rodríguez, F.; Martín, I.R.; Jacinto, C.; Jaque, D. In Vivo Subcutaneous Thermal Video Recording by Supersensitive Infrared Nanothermometers. Adv. Funct. Mater. 2017, 27. [Google Scholar] [CrossRef]
- Savchuk, O.A.; Haro-González, P.; Carvajal, J.J.; Jaque, D.; Massons, J.; Aguiló, M.; Díaz, F. Er:Yb:NaY2F5O up-converting nanoparticles for sub-tissue fluorescence lifetime thermal sensing. Nanoscale 2014, 6, 9727–9733. [Google Scholar] [CrossRef] [PubMed]
- Savchuk, O.; Marti, J.J.C.; Cascales, C.; Haro-Gonzalez, P.; Sanz-Rodríguez, F.; Aguilo, M.; Diaz, F. Bifunctional Tm3+,Yb3+:GdVO4@SiO2 core-shell nanoparticles in HeLa cells: Upconversion luminescence nanothermometry in the first biological window and biolabelling in the visible. Nanomaterials 2020, 10, 993. [Google Scholar] [CrossRef] [PubMed]
- Cortelletti, P.; Skripka, A.; Facciotti, C.; Pedroni, M.; Caputo, G.; Pinna, N.; Quintanilla, M.; Benayas, A.; Vetrone, F.; Speghini, A. Tuning the sensitivity of lanthanide-activated NIR nanothermometers in the biological windows. Nanoscale 2018, 10, 2568–2576. [Google Scholar] [CrossRef] [PubMed]
- Runowski, M.; Stopikowska, N.; Szeremeta, D.; Goderski, S.; Skwierczyńska, M.; Lis, S. Upconverting Lanthanide Fluoride Core@Shell Nanorods for Luminescent Thermometry in the First and Second Biological Windows: β-NaYF4:Yb3+ − Er3+ @SiO2 Temperature Sensor. ACS Appl. Mater. Interfaces 2019, 11, 13389–13396. [Google Scholar] [CrossRef]
- Suo, H.; Guo, C.; Zheng, J.; Zhou, B.; Ma, C.; Zhao, X.; Li, T.; Guo, P.; Goldys, E.M. Sensitivity Modulation of Upconverting Thermometry through Engineering Phonon Energy of a Matrix. ACS Appl. Mater. Interfaces 2016, 8, 30312–30319. [Google Scholar] [CrossRef]
- Dong, B.; Hua, R.N.; Cao, B.S.; Li, Z.P.; He, Y.Y.; Zhang, Z.Y.; Wolfbeis, O.S. Size dependence of the upconverted luminescence of NaYF4: Er, Yb microspheres for use in ratiometric thermometry. Phys. Chem. Chem. Phys. 2014, 16, 20009–20012. [Google Scholar] [CrossRef]
- Kumar, V.; Som, S.; Dutta, S.; Das, S.; Swart, H.C. Influence of Ho3+ doping on the temperature sensing behavior of Er3+-Yb3+ doped La2CaZnO5 phosphor. RSC Adv. 2016, 6, 84914–84925. [Google Scholar] [CrossRef]
- Skripka, A.; Benayas, A.; Marin, R.; Canton, P.; Hemmer, E.; Vetrone, F. Double rare-earth nanothermometer in aqueous media: Opening the third optical transparency window to temperature sensing. Nanoscale 2017, 9, 3079–3085. [Google Scholar] [CrossRef] [Green Version]
- Prorok, K.; Pawlyta, M.; Strek, W.; Bednarkiewicz, A. Energy Migration Up-conversion of Tb3+ in Yb3+ and Nd3+ Codoped Active-Core/Active-Shell Colloidal Nanoparticles. Chem. Mater. 2016, 28, 2295–2300. [Google Scholar] [CrossRef]
- Wang, C.; Xia, H.; Feng, Z.; Zhang, Z.; Jiang, D.; Zhang, J.; He, S.; Tang, Q.; Sheng, Q.; Gu, X.; et al. Infrared spectral properties for α-NaYF4 single crystal of various Er3+ doping concentrations. Opt. Laser Technol. 2016, 82, 157–162. [Google Scholar] [CrossRef]
- Chen, G.; Shen, J.; Ohulchanskyy, T.Y.; Patel, N.J.; Kutikov, A.; Li, Z.; Song, J.; Pandey, R.K.; Agren, H.; Prasad, P.N.; et al. (α-NaYbF4:Tm3+)/CaF2 core/shell nanoparticles with efficient near-infrared to near-infrared upconversion for high-contrast deep tissue bioimaging. ACS Nano 2012, 6, 8280–8287. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chen, B.; Wang, F. Combating Concentration Quenching in Upconversion Nanoparticles. Acc. Chem. Res. 2019. [Google Scholar] [CrossRef] [PubMed]
- Shen, B.; Cheng, S.; Gu, Y.; Ni, D.; Gao, Y.; Su, Q.; Feng, W.; Li, F. Revisiting the optimized doping ratio in core/shell nanostructured upconversion particles. Nanoscale 2017, 9, 1964–1971. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; Chen, G.; Hao, S.; Yang, C. Sub-6 nm monodisperse hexagonal core/shell NaGdF4 nanocrystals with enhanced upconversion photoluminescence. Nanoscale 2017, 9, 91–98. [Google Scholar] [CrossRef] [PubMed]
- Rai, V.K.; Rai, S.B. A comparative study of FIR and FL based temperature sensing schemes: An example of Pr3+. Appl. Phys. B Lasers Opt. 2007, 87, 323–325. [Google Scholar] [CrossRef]
- Shannon, R.D. Revised effective ionic radii and systematic studies of interaction distance in halides and chalcogenides. Acta Crystallogr. A 1976, 32, 751–767. [Google Scholar] [CrossRef]
- Arppe, R.; Hyppänen, I.; Perälä, N.; Peltomaa, R.; Kaiser, M.; Würth, C.; Christ, S.; Resch-Genger, U.; Schäferling, M.; Soukka, T. Quenching of the upconversion luminescence of NaYF4:Yb3+,Er3+ and NaYF4:Yb3+,Tm3+ nanophosphors by water: The role of the sensitizer Yb3+ in non-radiative relaxation. Nanoscale 2015, 7, 11746–11757. [Google Scholar] [CrossRef] [Green Version]
- Nadort, A.; Zhao, J.; Goldys, E.M. Lanthanide upconversion luminescence at the nanoscale: Fundamentals and optical properties. Nanoscale 2016, 8, 13099–13130. [Google Scholar] [CrossRef] [Green Version]
- Miyakawa, T.; Dexter, D.L. Phonon sidebands, multiphonon relaxation of excited states, and phonon-assisted energy transfer between ions in solids. Phys. Rev. B 1970, 1, 2961–2969. [Google Scholar] [CrossRef]
- Chen, X.; Peng, D.; Ju, Q.; Wang, F. Photon upconversion in core-shell nanoparticles. Chem. Soc. Rev. 2015, 44, 1318–1330. [Google Scholar] [CrossRef] [PubMed]

© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pominova, D.; Proydakova, V.; Romanishkin, I.; Ryabova, A.; Kuznetsov, S.; Uvarov, O.; Fedorov, P.; Loschenov, V. Temperature Sensing in the Short-Wave Infrared Spectral Region Using Core-Shell NaGdF4:Yb3+, Ho3+, Er3+@NaYF4 Nanothermometers. Nanomaterials 2020, 10, 1992. https://doi.org/10.3390/nano10101992
Pominova D, Proydakova V, Romanishkin I, Ryabova A, Kuznetsov S, Uvarov O, Fedorov P, Loschenov V. Temperature Sensing in the Short-Wave Infrared Spectral Region Using Core-Shell NaGdF4:Yb3+, Ho3+, Er3+@NaYF4 Nanothermometers. Nanomaterials. 2020; 10(10):1992. https://doi.org/10.3390/nano10101992
Chicago/Turabian StylePominova, Daria, Vera Proydakova, Igor Romanishkin, Anastasia Ryabova, Sergei Kuznetsov, Oleg Uvarov, Pavel Fedorov, and Victor Loschenov. 2020. "Temperature Sensing in the Short-Wave Infrared Spectral Region Using Core-Shell NaGdF4:Yb3+, Ho3+, Er3+@NaYF4 Nanothermometers" Nanomaterials 10, no. 10: 1992. https://doi.org/10.3390/nano10101992
APA StylePominova, D., Proydakova, V., Romanishkin, I., Ryabova, A., Kuznetsov, S., Uvarov, O., Fedorov, P., & Loschenov, V. (2020). Temperature Sensing in the Short-Wave Infrared Spectral Region Using Core-Shell NaGdF4:Yb3+, Ho3+, Er3+@NaYF4 Nanothermometers. Nanomaterials, 10(10), 1992. https://doi.org/10.3390/nano10101992






