Transition Metal Carbide Core/Shell Nanoparticles by Ultra-Short Laser Ablation in Liquid
Abstract
1. Introduction
2. Materials and Methods
3. Results
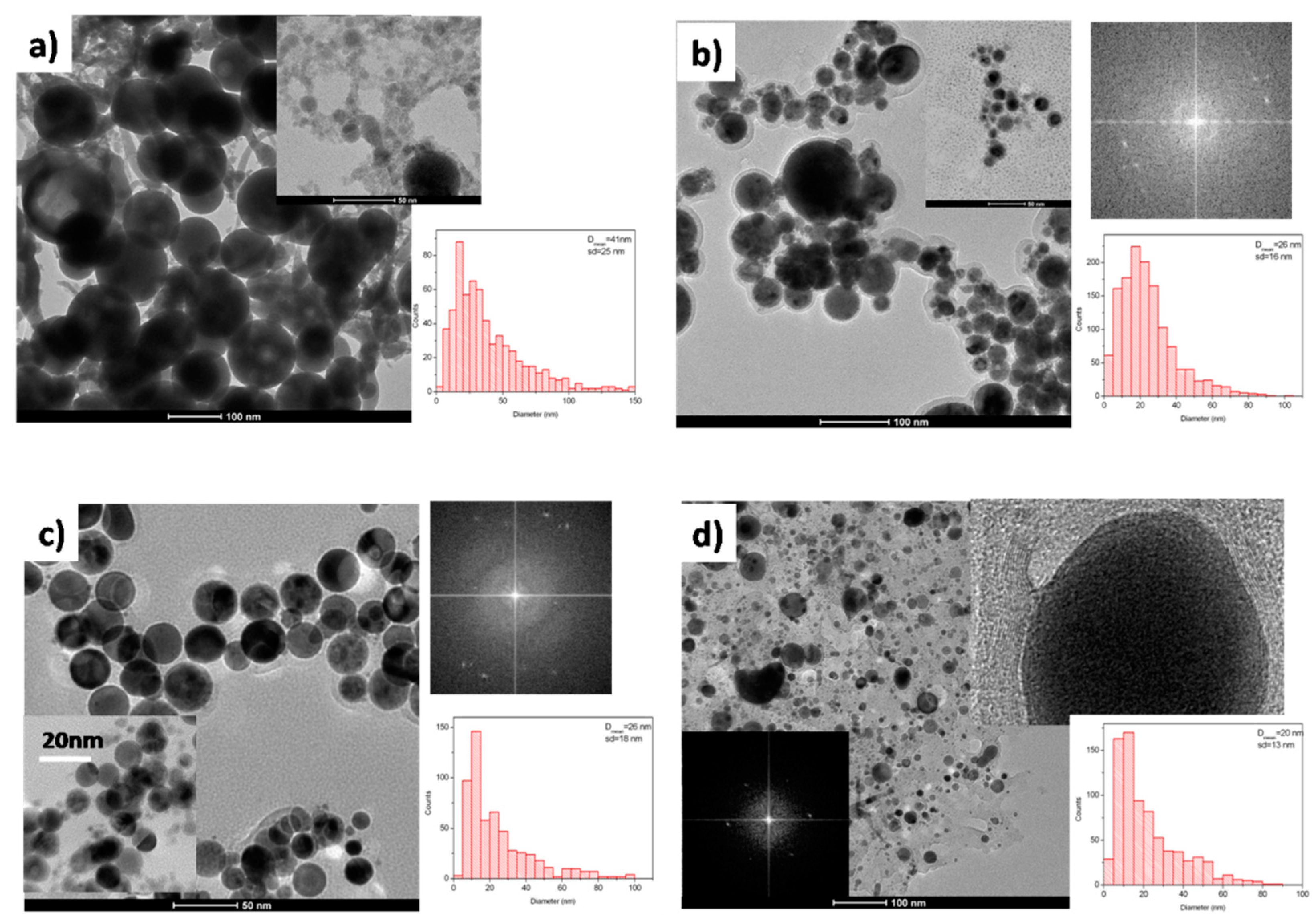
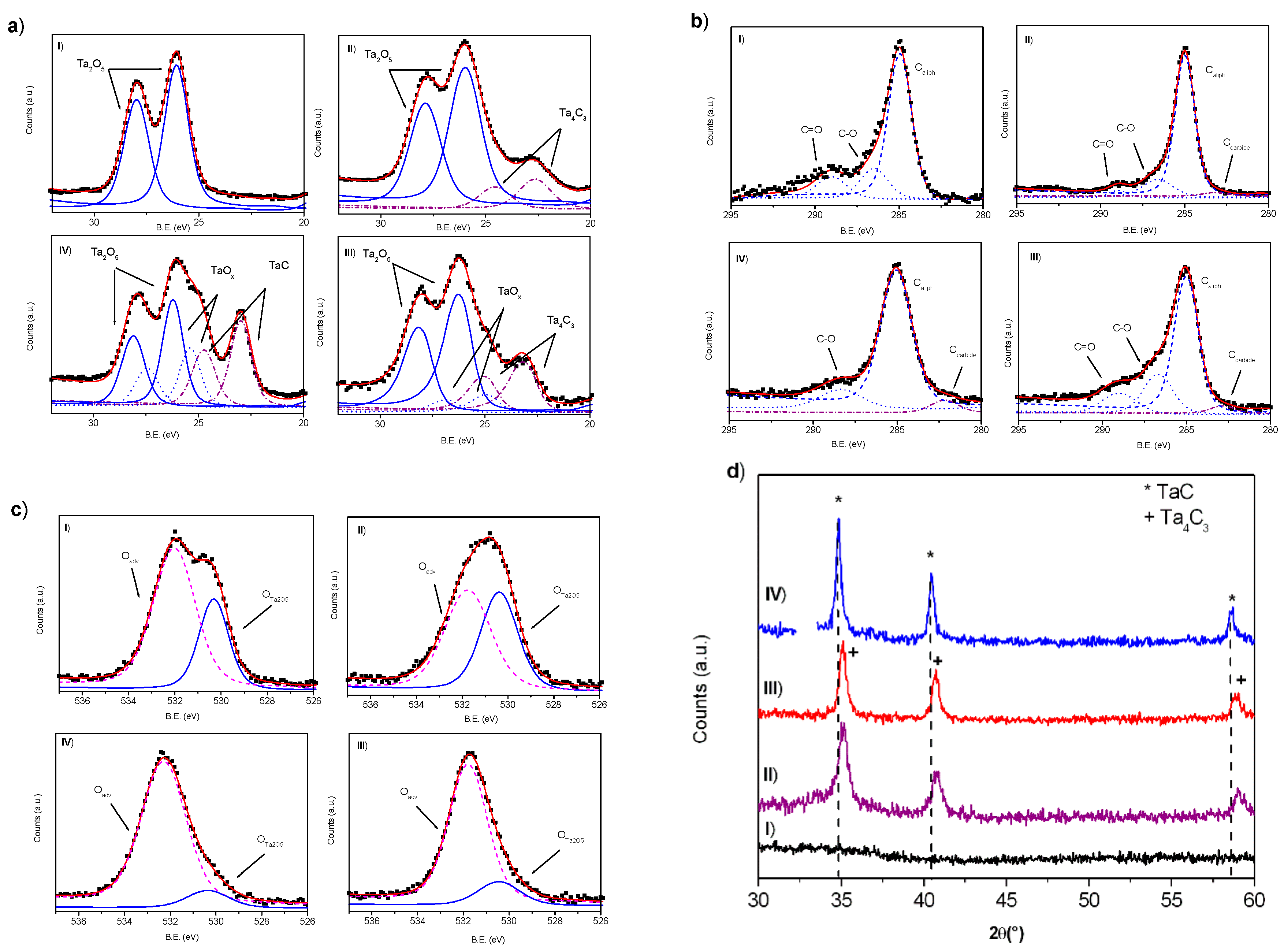
3.1. Tantalum
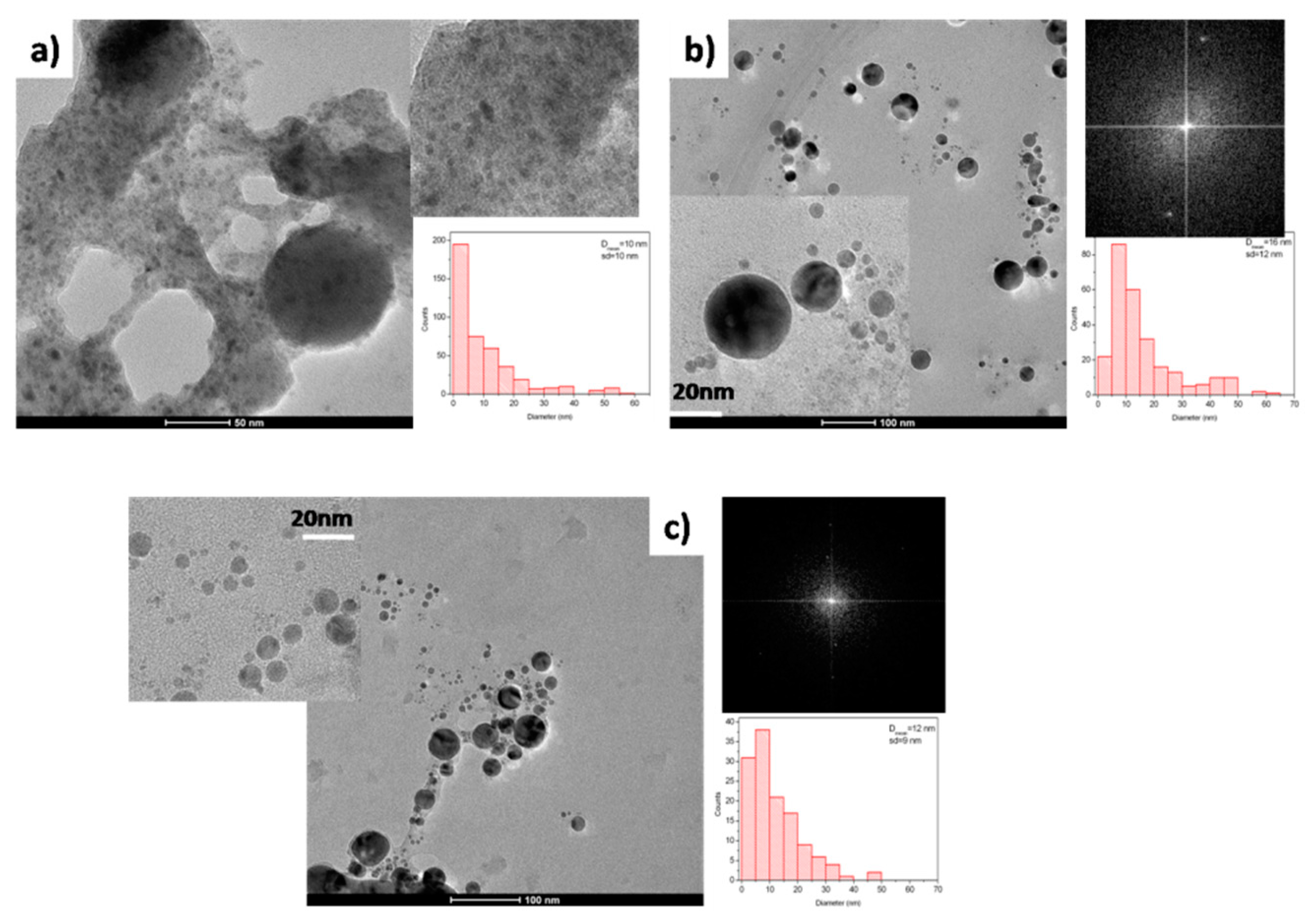
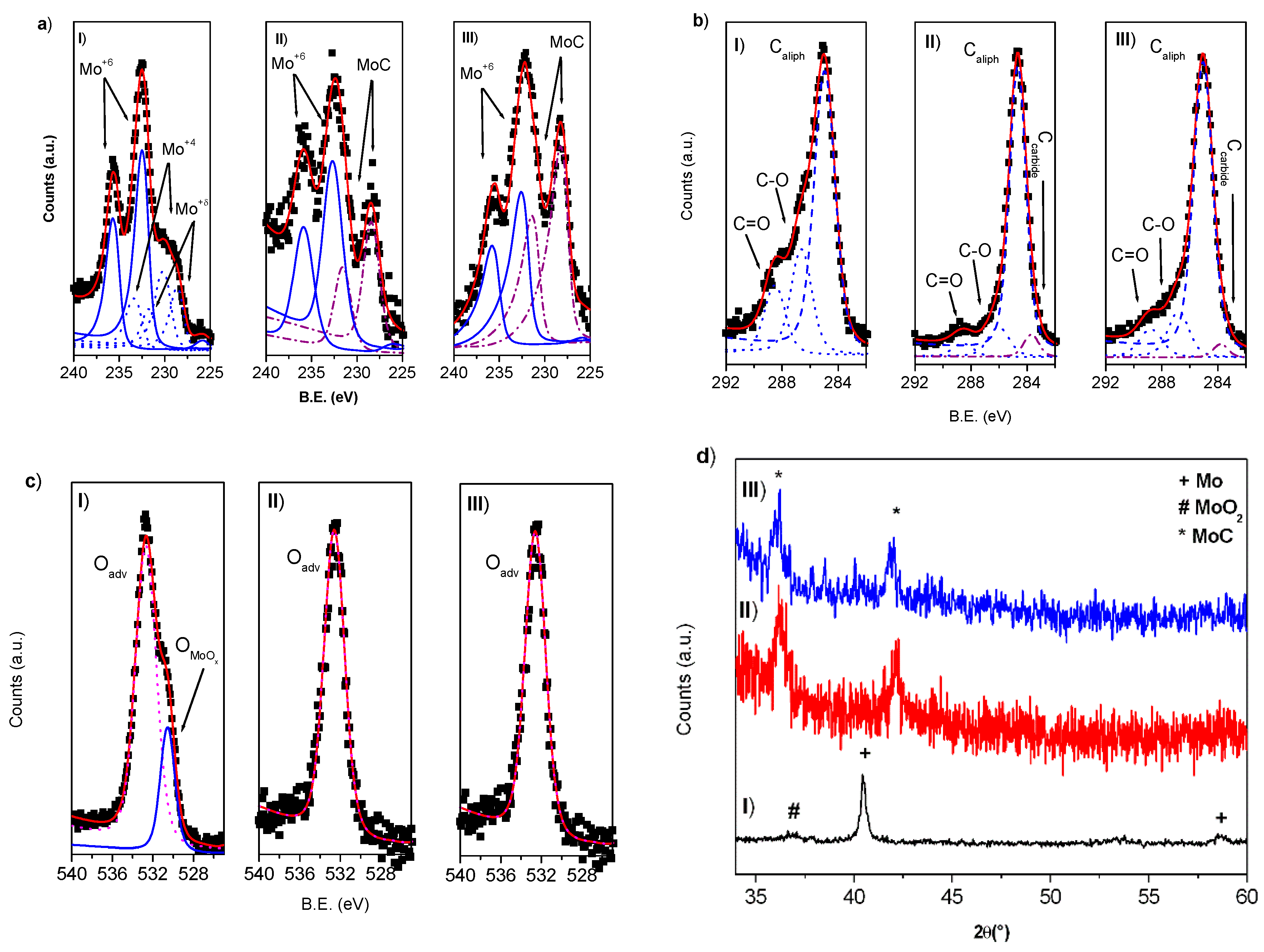
3.2. Molybdenum
4. Discussion
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Dinh, K.N.; Liang, Q.; Du, C.F.; Zhao, J.; YoongTok, A.I.; Mao, H.; Yan, Q. Nanostructures metallic transition metal carbides, nitrides, phosphides and borides for energy storage and conversion. Nano Today 2019, 25, 99–121. [Google Scholar] [CrossRef]
- Pang, J.; Sun, J.; Zheng, M.; Li, H.; Wang, Y.; Zhang, T. Transition metal carbide catalysts for biomass conversion. Appl. Catal. B 2019, 254, 510–522. [Google Scholar] [CrossRef]
- Horáček, J.; Akhmetzyanova, U.; Skuhrovcová, L.; Tišler, Z.; de Paz Carmona, H. Alumina-supported MoNx, MoCx and MoPx catalysts for the hydrotreatment of rapeseed oil. Appl. Catal. B Environ. 2020, 263, 118328. [Google Scholar] [CrossRef]
- Gouveia, J.D.; Morales-García, A.; Viñes, F.; Illas, F.; Gomes, J.R.B. MXenes as promising catalysts for water dissociation. Appl. Catal. B Environ. 2020, 260, 118191. [Google Scholar] [CrossRef]
- Song, Y.S.; Ren, J.T.; Yuan, G.; Yao, Y.; Liu, Y. Facile synthesis of Mo2C nanoparticles on N-doped carbon nanotubes with enhanced electrocatalytic activity for hydrogen evolution and oxygen reduction reactions. J. Energy Chem. 2019, 38, 68–77. [Google Scholar] [CrossRef]
- Hu, X.; Wang, Y.; Xu, M. Study of the cell responses in tantalum carbide nanoparticles-enriched polysaccharide composite hydrogel. Int. J. Biol. Macromol 2019, 135, 501–511. [Google Scholar] [CrossRef] [PubMed]
- Zhang, D.; Liu, J.; Li, P.; Tian, Z.; Liang, C. Recent advances in surfactant-free, surface-charged and defect-rich catalysts developed by laser ablation and processing in liquid. ChemNanoMat 2017, 3, 512–533. [Google Scholar] [CrossRef]
- Zhang, D.; Gokce, B.; Barcikowski, S. Laser synthesis and processing of colloids: Fundamentals and application. Chem. Rev. 2017, 117, 3990–4103. [Google Scholar] [CrossRef]
- De Bonis, A.; Santagata, A.; Galasso, A.; Laurita, A.; Teghil, R. Formation of titanium carbide (TiC) and TiC core shell nanostructures by ultra-short laser ablation of titanium carbide and metallic titanium in liquid. J. Colloid Interface Sci. 2017, 489, 76–84. [Google Scholar] [CrossRef]
- Davodi, F.; Muhlhausen, E.; Settipani, D.; Rautama, E.; Honkanen, A.; Houtari, S.; Marzun, G.; Taskinen, P.; Kallio, T. Comprehensive study to design advanced metal-carbide@graphene and metal-caarbide@iron oxide nanoparticles with tunable structure by the laser ablation in liquid. J. Colloid Interface Sci. 2019, 556, 180–192. [Google Scholar] [CrossRef]
- Amendola, V.; Meneghetti, M. What controls the composition and the structure of nanomaterials generated by laser ablation in liquid solution? Phys. Chem. Chem. Phys. 2013, 15, 3027–3046. [Google Scholar] [CrossRef] [PubMed]
- Shih, C.Y.; Streubel, R.; Heberle, J.; Letzel, A.; Shugaev, M.V.; Wu, C.; Schmidt, M.; Gokce, B.; Barcikowski, S.; Zhigilei, L.V. Two mechanisms of nanoparticle generation in picosecond laser ablation in liquids: The origin of the bimodal size distribution. Nanoscale 2018, 10, 6900–6910. [Google Scholar] [CrossRef] [PubMed]
- De Bonis, A.; Sansone, M.; D’Alessio, L.; Galasso, A.; Santagata, A.; Teghil, R. Dynamics of laser induced bubble and nanoparticles generation during ultra-short laser ablation of Pd in liquid. J. Phys. D 2013, 46, 445301. [Google Scholar] [CrossRef]
- De Bonis, A.; Galasso, A.; Santagata, A.; Teghil, R. Laser ablation of GaAs in liquid: The role of laser pulse duration. J. Phys. D 2016, 49, 035301. [Google Scholar] [CrossRef]
- Ali, N.; Bashir, S.; Kalsoom, U.; Begum, N.; Raffique, M.S.; Husinsky, W. Effect of liquid environment on the titanium surface modification by laser ablation. Appl. Surf. Sci. 2017, 405, 298–307. [Google Scholar] [CrossRef]
- MinetònArboleda, D.; Santillàn, J.M.J.; Mendoza Herrera, L.J.; Fernasndez van Raap, M.B.; Mendoza Zelis, P.; Muraca, D.; Schinca, D.C.; Scaffardi, L.B. Synthesis of Ni nanoparticles by femtosecond laser ablation in liquid: Structure and sizing. J. Phys. Chem. C 2015, 119, 13184–13193. [Google Scholar] [CrossRef]
- Mohazzab, B.F.; Jaleh, B.; Kakuee, O.; Fattah-alhosseini, A. Formation of titanium carbide on the titanium surface using laser ablation in n-heptane and investigating its corrosion resistance. Appl. Surf. Sci. 2019, 478, 623–635. [Google Scholar] [CrossRef]
- Baruah, P.K.; Sharma, A.K.; Khare, A. Role of confining liquids on the properties of Cu@Cu2O nanoparticles synthesized by pulsed laser ablation and correlative ablation study of the target surface. RSC Adv. 2019, 9, 15124–15139. [Google Scholar] [CrossRef]
- Zhang, D.; Zhang, C.; Liu, J.; Chen, Q.; Zhu, X.; Liang, C. Carbon encapsulated metal/metal carbide/metal oxide core-shell nanostructures generated by laser ablation of metals in organic solvents. ACS Appl. Nano Mater. 2019, 2, 28–39. [Google Scholar] [CrossRef]
- Marzun, G.; Bonnemann, H.; Lehmann, C.; Spliethoff, B.; Weidenthaler, C.; Barcikowski, S. Role of dissolved and molecular oxygen on Cu and PtCu alloy particle structure during laser ablation synthesis in liquids. ChemPhysChem 2017, 18, 1175–1184. [Google Scholar] [CrossRef]
- Franzel, L.; Phumisithikul, K.; Bertino, M.F.; Carpenter, E.E. Synthesis of multiphasic inhomogeneous Mo/MoCnanoparticles by pulsed laser ablation. J. Nanopart. Res. 2013, 15, 2032. [Google Scholar] [CrossRef]
- Zhang, H.; Liang, C.; Liu, J.; Tin, Z.; Shao, G. The formation of onion-like carbon-encapsulated cobalt carbide core/shell nanoparticles by the laser ablation of metallic cobalt in acetone. Carbon 2013, 55, 108–115. [Google Scholar] [CrossRef]
- Zhang, H.; Liu, J.; Tian, Z.; Ye, Y.; Cai, Y.; Liang, C. A general strategy towards transition metal carbide/carbon core/shell nanospheres and their application for supercapacitor electrode. Carbon 2016, 100, 590–599. [Google Scholar] [CrossRef]
- Castle, J.E.; Salvi, A.M. Chemical state information from the near-peak region of the X-ray photoelectron background. J. Electron Spectrosc. 2001, 114, 1103–1113. [Google Scholar] [CrossRef]
- Zhang, D.; Ma, Z.; Spasova, M.; Yelsukova, A.E.; Lu, S.; Farle, M.; Wiedwald, U.; Gokce, B. Formation mechanism of laser-syntesized iron-manganese alloy nanoparticles, manganese oxide nanosheets and nanofibers. Part. Part. Syst. Charact. 2017, 34, 1600225. [Google Scholar] [CrossRef]
- Gritsenko, V.A.; Perevalov, T.V.; Voronkovskij, V.A.; Gismatulin, A.A.; Kruchinin, V.N.; Aliev, V.S.; Pustovarov, V.A.; Prosvirin, I.P. Charge transport and the nature of traps in oxygen deficient tantalum oxide. ACS Appl. Mater. Interfaces 2018, 10, 3769–3775. [Google Scholar] [CrossRef]
- West, R.C. Handbook of Chemistry and Physics, 69th ed.; CRC Press: Boca Raton, FL, USA, 1988–1989. [Google Scholar]
- Li, Q.; Liang, C.; Tian, Z.; Zhang, J.; Zhang, H.; Cai, W. Core–shell TaxO@Ta2O5 structured nanoparticles: Laser ablation synthesis in liquid, structure and photocatalytic property. CrystEngComm 2012, 14, 3236. [Google Scholar] [CrossRef]
- Manukumar, K.N.; Kishore, B.; Manjunath, K.; Nagaraju, G. Mesoporous Ta2O5 nanoparticles as an anode material for lithium ion battery and an efficient photocatalyst for hydrogen evolution. Int. J. Hydrog. Energy 2018, 43, 18125–18135. [Google Scholar] [CrossRef]
- Alhajri, N.S.; Yoshida, H.; Anjum, D.H.; Garcia-Esparza, A.T.; Kubota, J.; Domen, K.; Takanabe, K. Synthesis of tantalum carbide and nitride nanoparticles using a reactive mesoporous template for electrochemical hydrogen evolution. J. Mater. Chem. A 2013, 1, 12606–12616. [Google Scholar] [CrossRef]
- Brar, L.K.; Gupta, A.; Pandey, O.P. Influence of carbon content of nano-TaC powders on the electrocatalytic and photocatalytic properties. Catal. Today 2019, 325, 98–108. [Google Scholar] [CrossRef]
- Teghil, R.; De Bonis, A.; Galasso, A.; Villani, P.; Santagata, A. Femtosecond pulsed laser ablation deposition of tantalum carbide. Appl. Surf. Sci. 2007, 254, 1220–1223. [Google Scholar] [CrossRef]
- Sheng Moo, J.G.; Awaludin, Z.; Okajima, T.; Ohsaka, T. An XPS depth-profile study on electrochemically deposited TaOx. J. Solid State Electrochem. 2013, 17, 3115–3123. [Google Scholar]
- Li, P.G.; Lei, M.; Sun, Z.B.; Cao, L.Z.; Guo, Y.F.; Guo, X.; Tang, W.H. C3N4 as precursor for the synthesis of NbC, TaC and WC nanoparticles. J. Alloys Compd. 2007, 430, 237–240. [Google Scholar] [CrossRef]
- Li, P.; Qui, Y.; Liu, S.; Li, H.; Zhao, S.; Diao, J.; Guo, X. Heterogeneous Mo2C/Fe5C2 nanoparticles embedded in nitrogen-doped carbon s efficient electrocatalysts for the oxygen reduction reaction. Eur. J. Inorg. Chem. 2019, 27, 3235–3241. [Google Scholar] [CrossRef]
- Madrigal-Camacho, M.; Vilichis-Nestor, A.R.; Camacho-Lopez, M.; Camacho-Lopez, M.A. Synthesis of MoC@graphite NPs by short and ultrashort pulses laser ablation in toluene under N2 atmosphere. Diam. Relat. Mater. 2018, 82, 63–69. [Google Scholar] [CrossRef]
- Hamdan, M.A.; Loridant, S.; Jahjah, M.; Pinel, C.; Perret, N. TiO2-supported molybdenum carbide: An active catalyst for the aqueous phase hydrogenation for succinic acid. Appl. Catal. A 2019, 571, 71–81. [Google Scholar] [CrossRef]
- Wu, C.; Zhigilei, L.V. Microscopic mechanism of laser spallation and ablation of metal targets from large-scale molecular dynamics simulations. Appl. Phys. A 2014, 114, 11–32. [Google Scholar] [CrossRef]
- Cristoforetti, G.; Pitzalis, E.; Spiniello, R.; Ishak, R.; Muniz Miranda, M. Production of palladium nanoparticles by pulsed laser ablation in water and their characterization. J. Phys. Chem. C 2010, 115, 5073–5083. [Google Scholar] [CrossRef]
- De Bonis, A.; Sansone, M.; Galasso, A.; Santagata, A.; Teghil, R. The role of the solvent in the ultra-short laser ablation of palladium target in liquid. Appl. Phys. A Mater. Sci. Process. 2014, 117, 211–216. [Google Scholar] [CrossRef]
- Haxhiaj, I.; Tigges, S.; Firla, D.; Zhang, X.; Hagemann, U.; Kondo, T.; Nakamura, J.; Marzun, G.; Barcikowski, S. Platinum nanoparticles supported on reduced graphene oxide prepared in situ by a continuous one-step laser process. Appl. Surf. Sci. 2019, 469, 811–820. [Google Scholar] [CrossRef]
- Gentile, A.; Giacco, D.; De Bonis, A.; Teghil, R.; Marrani, A.G.; Brutti, S. Synergistic electro-catalysis of Pd/PdO nanoparticles and Cr (II)-doped NiCo2O4 nanofibers in aprotic Li-O2 batteries. J. Electrochem. Soc. 2018, 165, A3605–A3612. [Google Scholar] [CrossRef]
- Niessen, A.K.; De Boer, F.R. The enthalpy of formation of solid borides, carbides, nitrides, silicides and phosphides of transition and noble metals. J. Less Common Met. 1981, 82, 75–80. [Google Scholar] [CrossRef]
- Yang, S.; Cai, W.; Zhang, H.; Zeng, H.; Lei, Y. A General Strategy for Fabricating Unique Carbide Nanostructures with Excitation Wavelength-Dependent Light Emissions. J. Phys. Chem. C 2011, 115, 7279–7284. [Google Scholar] [CrossRef]
- De Bonis, A.; Lovaglio, T.; Galasso, A.; Santagata, A.; Teghil, R. Iron and iron oxide nanoparticles obtained by ultra-short laser ablation in liquid. Appl. Surf. Sci. 2015, 353, 433–438. [Google Scholar] [CrossRef]
- Rotzoll, G. High-temperature pyrolysis of ethanol. J. Anal. Appl. Pyrolysis 1985, 9, 43–52. [Google Scholar] [CrossRef]
- Nikov, R.G.; Nedyalkov, N.N.; Atanasov, P.A.; Karashanova, D.B. Laser-assisted fabrication and size distribution modification of colloidal gold nanostructures by nanosecond laser ablation in different liquids. Appl. Phys. A 2017, 123, 490. [Google Scholar] [CrossRef]
- Amendola, V.; Rizzi, G.A.; Polizzi, S.; Meneghetti, M. Synthesis of Gold Nanoparticles by Laser Ablation in Toluene: Quenching and Recovery of the Surface Plasmon Absorption. J. Phys. Chem. B 2005, 109, 23125–23128. [Google Scholar] [CrossRef]
- Cristoforetti, G.; Pitzalis, E.; Spiniello, R.; Ishack, R.; Giammanco, F.; Muniz-Miranda, M.; Caporali, S. Physico-chemical properties of Pd nanoparticles produced by Pulsed Laser Ablation in different organic solvents. Appl. Surf. Sci. 2012, 258, 3289–3297. [Google Scholar] [CrossRef]
- Boyer, P.; Meunier, M. Modeling solvent influence on growth mechanism of nanoparticles (Au, Co,) synthesized by surfactant free laser process. J. Phys. Chem. C 2012, 116, 8014–8019. [Google Scholar] [CrossRef]
- Mohapatra, D.; Badrayyana, S.; Parida, S. Facile wick-and-oil flame synthesis of high-quality hydrophilic onionlike carbon nanoparticles. Mater. Chem. Phys. 2016, 174, 112–119. [Google Scholar] [CrossRef]
- Joly-Pottuz, L.; Vacher, B.; Ohmae, N.; Martin, J.; Epicier, T. Anti-wear and friction reducing mechanisms of carbon nano-onions as lubricant additives. Tribol. Lett. 2008, 30, 69–80. [Google Scholar] [CrossRef]
- Keller, N.N.; Maksimova, N.I.; Roddatis, V.V.; Schur, M.; Mestl, G.; Butenko, Y.V.; Kuznetsov, V.L.; Schlogl, R. The catalytic use of onion-like carbon materials for styrene synthesis by oxidative dehydrogenation of ethylbenzene. Angew. Chem. Int. Ed. 2002, 41, 1885–1888. [Google Scholar] [CrossRef]
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
De Bonis, A.; Curcio, M.; Santagata, A.; Galasso, A.; Teghil, R. Transition Metal Carbide Core/Shell Nanoparticles by Ultra-Short Laser Ablation in Liquid. Nanomaterials 2020, 10, 145. https://doi.org/10.3390/nano10010145
De Bonis A, Curcio M, Santagata A, Galasso A, Teghil R. Transition Metal Carbide Core/Shell Nanoparticles by Ultra-Short Laser Ablation in Liquid. Nanomaterials. 2020; 10(1):145. https://doi.org/10.3390/nano10010145
Chicago/Turabian StyleDe Bonis, Angela, Mariangela Curcio, Antonio Santagata, Agostino Galasso, and Roberto Teghil. 2020. "Transition Metal Carbide Core/Shell Nanoparticles by Ultra-Short Laser Ablation in Liquid" Nanomaterials 10, no. 1: 145. https://doi.org/10.3390/nano10010145
APA StyleDe Bonis, A., Curcio, M., Santagata, A., Galasso, A., & Teghil, R. (2020). Transition Metal Carbide Core/Shell Nanoparticles by Ultra-Short Laser Ablation in Liquid. Nanomaterials, 10(1), 145. https://doi.org/10.3390/nano10010145








