Trapezium Bone Density—A Comparison of Measurements by DXA and CT
Abstract
:1. Introduction
2. Results
3. Discussion
4. Materials and Methods
4.1. Patients
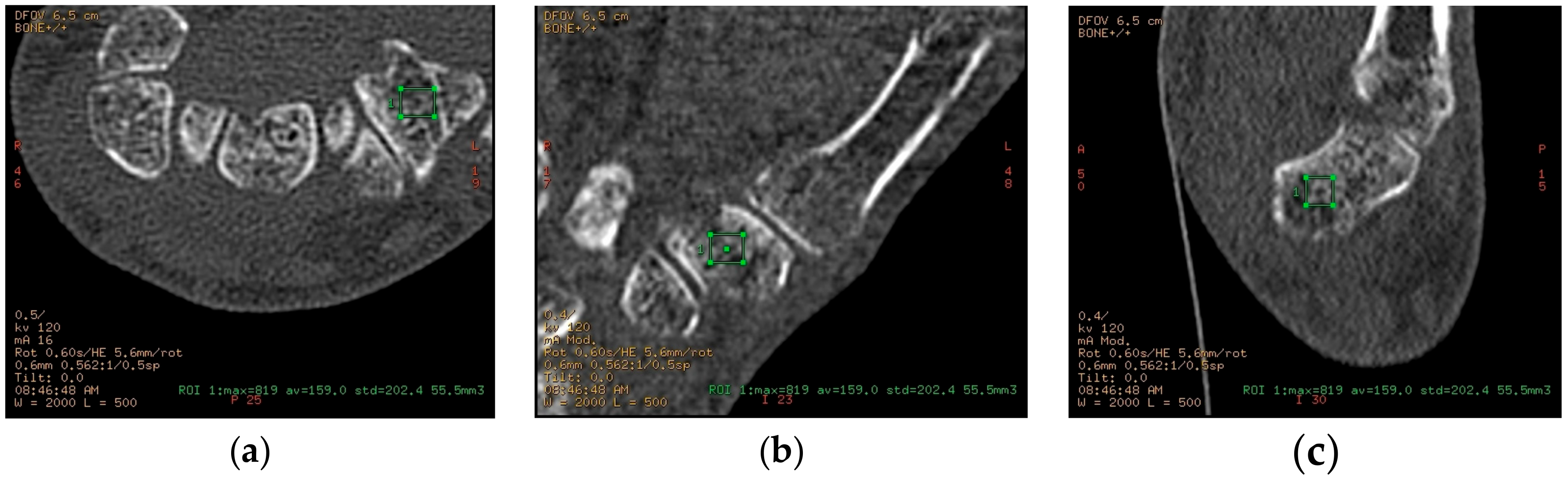
4.2. Image Acquisition and Analysis
4.3. Statistical Analysis
4.4. Reproducibility
5. Conclusions
Author Contributions
Conflicts of Interest
References
- Hansen, T.B.; Snerum, L. Elektra trapeziometacarpal prosthesis for treatment of osteoarthrosis of the basal joint of the thumb. Scand. J. Plast. Reconstr. Surg. Hand Surg. 2008, 42, 316–319. [Google Scholar] [CrossRef] [PubMed]
- Hansen, T.B.; Vainorius, D. High loosening rate of the moje acamo prosthesis for treating osteoarthritis of the trapeziometacarpal joint. J. Hand Surg. Eur. Vol. 2008, 33, 571–574. [Google Scholar] [CrossRef] [PubMed]
- Hernandez-Cortes, P.; Pajares-Lopez, M.; Robles-Molina, M.J.; Gomez-Sanchez, R.; Toledo-Romero, M.A.; De Torres-Urrea, J. Two-year outcomes of elektra prosthesis for trapeziometacarpal osteoarthritis: A longitudinal cohort study. J. Hand Surg. Eur. Vol. 2012, 37, 130–137. [Google Scholar] [CrossRef] [PubMed]
- Klahn, A.; Nygaard, M.; Gvozdenovic, R.; Boeckstyns, M.E. Elektra prosthesis for trapeziometacarpal osteoarthritis: A follow-up of 39 consecutive cases. J. Hand Surg. Eur. Vol. 2012, 37, 605–609. [Google Scholar] [CrossRef] [PubMed]
- Perez-Ubeda, M.J.; Garcia-Lopez, A.; Marco Martinez, F.; Junyent Vilanova, E.; Molina Martos, M.; Lopez-Duran Stern, L. Results of the cemented sr trapeziometacarpal prosthesis in the treatment of thumb carpometacarpal osteoarthritis. J. Hand Surg. 2003, 28, 917–925. [Google Scholar] [CrossRef]
- Wachtl, S.W.; Guggenheim, P.R.; Sennwald, G.R. Cemented and non-cemented replacements of the trapeziometacarpal joint. J. Bone Joint Surg. Br. Vol. 1998, 80, 121–125. [Google Scholar] [CrossRef]
- Hansen, T.B.; Meier, M.; Moller, M.C.; Larsen, K.; Stilling, M. Primary cup fixation with different designs of trapeziometacarpal total joint trapezium components: A radiostereometric analysis in a pig bone model. J. Bone Joint Surg. Br. Vol. 2011, 36, 285–290. [Google Scholar] [CrossRef] [PubMed]
- Brownbill, R.A.; Ilich, J.Z. Validation of the use of the hand for estimating bone mineral density in other skeletal sites by dxa in healthy and osteoarthritic women. J. Clin. Densitom. 2002, 5, 273–282. [Google Scholar] [CrossRef]
- El-Sherif, H.E.; Kamal, R.; Moawyah, O. Hand osteoarthritis and bone mineral density in postmenopausal women; clinical relevance to hand function, pain and disability. Osteoarthr. Cartil. 2008, 16, 12–17. [Google Scholar] [CrossRef] [PubMed]
- Haara, M.M.; Arokoski, J.P.; Kroger, H.; Karkkainen, A.; Manninen, P.; Knekt, P.; Impivaara, O.; Heliovaara, M. Association of radiological hand osteoarthritis with bone mineral mass: A population study. Rheumatology 2005, 44, 1549–1554. [Google Scholar] [CrossRef] [PubMed]
- Haugen, I.K.; Slatkowsky-Christensen, B.; Orstavik, R.; Kvien, T.K. Bone mineral density in patients with hand osteoarthritis compared to population controls and patients with rheumatoid arthritis. Ann. Rheum. Dis. 2007, 66, 1594–1598. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.K.; Park, S.H.; Choe, J.Y. Lower bone mineral density of forearm in postmenopausal patients with radiographic hand osteoarthritis. Rheumatol. Int. 2010, 30, 605–612. [Google Scholar] [CrossRef] [PubMed]
- Schneider, D.L.; Barrett-Connor, E.; Morton, D.J.; Weisman, M. Bone mineral density and clinical hand osteoarthritis in elderly men and women: The rancho bernardo study. J. Rheumatol. 2002, 29, 1467–1472. [Google Scholar] [PubMed]
- Zoli, A.; Lizzio, M.M.; Capuano, A.; Massafra, U.; Barini, A.; Ferraccioli, G. Osteoporosis and bone metabolism in postmenopausal women with osteoarthritis of the hand. Menopause 2006, 13, 462–466. [Google Scholar] [CrossRef] [PubMed]
- Licata, A. Bone density vs bone quality: What’s a clinician to do? Clevel. Clin. J. Med. 2009, 76, 331–336. [Google Scholar] [CrossRef] [PubMed]
- Hansen, T.B.; Mosegaard, K.B.; Sorensen, O.G.; Mortensen, J.; Stilling, M. Bone mineral density of the trapezium in osteoarthritic trapeziometacarpal joints. J. Hand Surg. Eur. Vol. 2013, 38, 875–879. [Google Scholar] [CrossRef] [PubMed]
- Schreiber, J.J.; Anderson, P.A.; Rosas, H.G.; Buchholz, A.L.; Au, A.G. Hounsfield units for assessing bone mineral density and strength: A tool for osteoporosis management. J. Bone Joint Surg. Am. Vol. 2011, 93, 1057–1063. [Google Scholar] [CrossRef] [PubMed]
- Westphal, F.M.; Bishop, N.; Honl, M.; Hille, E.; Puschel, K.; Morlock, M.M. Migration and cyclic motion of a new short-stemmed hip prosthesis—A biomechanical in vitro study. Clin. Biomech. 2006, 21, 834–840. [Google Scholar] [CrossRef] [PubMed]
- Schreiber, J.J.; McQuillan, T.J.; Halilaj, E.; Crisco, J.J.; Weiss, A.P.; Patel, T.; Kenney, D.; Ladd, A.L. Changes in local bone density in early thumb carpometacarpal joint osteoarthritis. J. Hand Surg. 2018, 43, 33–38. [Google Scholar] [CrossRef] [PubMed]
- Hansen, T.B.; Sorensen, O.G.; Kirkeby, L.; Homilius, M.; Amstrup, A.L. Computed tomography improves intra-observer reliability, but not the inter-observer reliability of the eaton-glickel classification. J. Hand Surg. Eur. Vol. 2013, 38, 187–191. [Google Scholar] [CrossRef] [PubMed]
- Eaton, R.G.; Glickel, S.Z. Trapeziometacarpal osteoarthritis. Staging as a rationale for treatment. Hand Clin. 1987, 3, 455–471. [Google Scholar] [PubMed]
- GE Healthcare Lunar. User Manual: Encore-Based Densitometer LU43616DK; Revision 16 July 2015; GE Healthcare Lunar: St. Clair, MI, USA, 2015. [Google Scholar]
- Diedenhofen, B.; Musch, J. Cocor: A comprehensive solution for the statistical comparison of correlations. PLoS ONE 2015, 10, e0121945. [Google Scholar] [CrossRef] [PubMed]

| Reference Mean And Data Mean | Data Mean | SEM | 95% CI | Reference Normal | Reference Osteopenia | Reference Osteoporosis | p Value |
|---|---|---|---|---|---|---|---|
| Total BMD | |||||||
| Males (N = 14) | 0.71 | 0.02 | 0.67–0.74 | 0.74 | 0.67 | 0.56 | 0.06 |
| Females (N = 57) | 0.61 | 0.01 | 0.58–0.63 | 0.63 | 0.57 | 0.48 | 0.05 |
| Ultra distal BMD | |||||||
| Males (N = 14) | 0.50 | 0.01 | 0.47–0.52 | 0.51 | 0.46 | 0.38 | 0.22 |
| Females (N = 57) | 0.42 | 0.01 | 0.39–0.44 | 0.43 | 0.38 | 0.32 | 0.25 |
| Radius 33% BMD | |||||||
| Males (N = 14) | 0.94 | 0.02 | 0.90–0.98 | 0.96 | 0.86 | 0.71 | 0.36 |
| Females (N = 57) | 0.81 | 0.01 | 0.78–0.83 | 0.81 | 0.72 | 0.59 | 0.74 |
| DXA And CT Mean | Mean | SEM | 95% CI |
|---|---|---|---|
| DXA circumference (g/cm2) | |||
| Male hands (n = 14) | 0.78 | 0.05 | 0.68–0.89 |
| Female hands (n = 57) | 0.65 | 0.02 | 0.62–0.69 |
| DXA inner-ellipse (g/cm2) | |||
| Male hands (n = 14) | 0.73 | 0.05 | 0.63–0.83 |
| Female hands (n = 57) | 0.61 | 0.02 | 0.57–0.64 |
| CT scans (HU) | |||
| Male hands (n = 14) | 277.43 | 17.89 | 241.75–313.11 |
| Female hands (n = 57) | 298.44 | 11.89 | 274.72–322.15 |
| DXA and CT Mean | Mean | SEM | 95% CI |
|---|---|---|---|
| DXA circumference (g/cm2) | |||
| Age group 1 (N = 24) | 0.72 | 0.03 | 0.66–0.77 |
| Age group 2 (N = 23) | 0.64 | 0.03 | 0.59–0.69 |
| Age group 3 (N = 24) | 0.69 | 0.04 | 0.62–0.77 |
| DXA inner-ellipse (g/cm2) | |||
| Age group 1 (N = 24) | 0.67 | 0.03 | 0.62–0.77 |
| Age group 2 (N = 23) | 0.59 | 0.03 | 0.53–0.64 |
| Age group 3 (N = 24) | 0.64 | 0.04 | 0.56–0.71 |
| CT scans (HU) | |||
| Age group 1 (N = 24) | 310.71 | 17.72 | 275.34–346.04 |
| Age group 2 (N = 23) | 309.72 | 20.62 | 268.59–350.85 |
| Age group 3 (N = 24) | 263.10 | 12.83 | 237.52–288.69 |
| Variable | Coef. | SEM | t | p > [t] | 95% CI |
|---|---|---|---|---|---|
| DXA circumference (g/cm2) | |||||
| Sex | −0.13 | 0.04 | −3.22 | 0.002 | −0.22–−0.05 |
| Age | −0.00 | 0.00 | −1.65 | 0.103 | −0.01–0.00 |
| DXA inner-ellipse (g/cm2) | |||||
| Sex | −0.13 | 0.04 | −3.00 | 0.004 | −2.11–−0.04 |
| Age | −0.00 | 0.00 | −1.79 | 0.078 | −0.01–0.00 |
| CT scans (HU) | |||||
| Sex | 17.24 | 25.26 | 0.68 | 0.497 | −33.16–67.65 |
| Age | −2.23 | 1.19 | −1.87 | 0.065 | −4.60–0.14 |
| Measurement | Mean (95% CI), g/cm3 | SDdif | Bias (LOA), g/cm3 | p Value | ICC |
|---|---|---|---|---|---|
| Intra-observer repeated measurements | |||||
| DXA Scan 1 (N = 71) | 0.62 (0.58–0.65) | 0.058 | −0.007 (0.114) | 0.00 | 0.93 |
| DXA Scan 2 (N = 71) | 0.64 (0.61–0.62) | 0.061 | 0.003 (0.120) | 0.00 | 0.92 |
| CT Scan (N = 71) | 294.30 (274.0–314.6) | 47.88 | 18.79 (93.84) | 0.00 | 0.84 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Breddam Mosegaard, S.; Breddam Mosegaard, K.; Bouteldja, N.; Bæk Hansen, T.; Stilling, M. Trapezium Bone Density—A Comparison of Measurements by DXA and CT. J. Funct. Biomater. 2018, 9, 9. https://doi.org/10.3390/jfb9010009
Breddam Mosegaard S, Breddam Mosegaard K, Bouteldja N, Bæk Hansen T, Stilling M. Trapezium Bone Density—A Comparison of Measurements by DXA and CT. Journal of Functional Biomaterials. 2018; 9(1):9. https://doi.org/10.3390/jfb9010009
Chicago/Turabian StyleBreddam Mosegaard, Sebastian, Kamille Breddam Mosegaard, Nadia Bouteldja, Torben Bæk Hansen, and Maiken Stilling. 2018. "Trapezium Bone Density—A Comparison of Measurements by DXA and CT" Journal of Functional Biomaterials 9, no. 1: 9. https://doi.org/10.3390/jfb9010009
APA StyleBreddam Mosegaard, S., Breddam Mosegaard, K., Bouteldja, N., Bæk Hansen, T., & Stilling, M. (2018). Trapezium Bone Density—A Comparison of Measurements by DXA and CT. Journal of Functional Biomaterials, 9(1), 9. https://doi.org/10.3390/jfb9010009







