Bioactive Glasses: From Parent 45S5 Composition to Scaffold-Assisted Tissue-Healing Therapies
Abstract
:1. Introduction
2. BGs: A Historical Overview
3. Interaction of BGs with the Physiological Environment: General Features
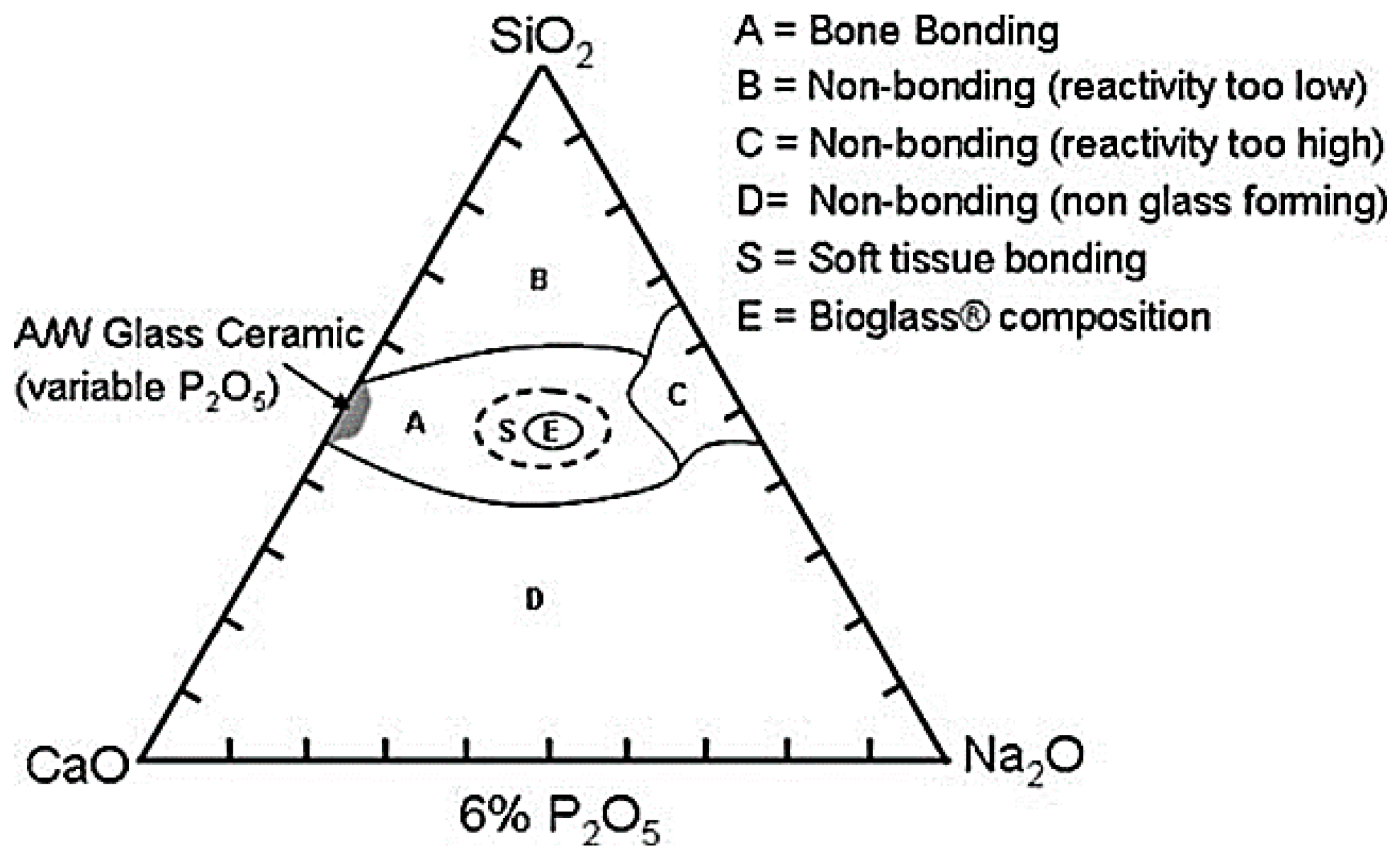
3.1. Classes of Bioactivity
- Osteo-integration, which is the ability to establish a chemical bond with the physiological tissue without the formation of a fibrous layer around the implant [38];
- Osteo-induction, which stimulates and activates pluripotent stem cells leading to their differentiation to an osteoblastic phenotype [39]. During this process, which is mediated by a signalling cascade, some extracellular and intracellular receptors are involved, the most important of which belong to the TGF-beta superfamily [38];
- Osteogenesis, that involves the synthesis of new bone by osteoblasts that are present within the graft (if cell-seeded biomaterial constructs are used) or have colonized it after implantation [39].
- Class A bioactive materials: These materials lead to both osteo-conductive and osteo-productive effects because of the rapid reaction mechanism that takes place on the surface of the material and which leads to the dissolution of critical concentration of soluble silica and Ca ions. Class A BGs are able to promote the colonization of their surface by osteogenic stem cells directly inside the surgery-derived bone defect that results in the rapid formation of osteoid bridges between particles, followed by the mineralization of the matrix and the formation of mature bone structures. This phenomenon determines both an extracellular and an intracellular response at the interface because of the interaction with the ions released from the surface [10].
- Class B bioactive materials: unlike the previous class, such implants show only osteo-conductive properties determined exclusively by extracellular factors [40]; a typical example is hydroxyapatite.
3.2. Mechanism of Bioactivity: Creation of the Material–Host Tissue Bond
- Formation of the hydroxycarbonateapatite (HCA) layer, which takes place during the first 5 steps of reactions, culminating in the crystallization of the amorphous calcium phosphate film [2];
- Dissolution of ionic products from BG surface and osteogenesis, which leads to the mineralization of the extracellular matrix (ECM) [23].
3.2.1. Formation of the HCA Layer
- Ionic exchange between Ca2+ and Na+ ions in the material and H+ and H3O+ ions coming from the surrounding environment. Silanol bonds (Si–OH) are established on the surface of the material. An increase of the solution pH is observed due to the release of alkaline ions and a silica-rich layer forms on the surface of the glass. (PO4)3− ions are released too, if they are present in the starting composition.The chemical reaction is reported below:Si–O–Na+ + H+ + OH− → Si–OH+ + Na+ (aq) + OH−
- 2.
- High local value of pH determines the breaking of O–Si–O bonds operated by OH− groups, which cause the breaking of the silica network. Soluble silica is released in the form of Si(OH)4 and the silanol groups are exposed on the surface of the material, directly in contact with the solution. The equation describing the mechanism is reported below:Si–O–Si + H2O → Si–OH + OH–Si
- 3.
- Silanol groups condensation and re-polymerization of an amorphous silica-rich layer poor in Na+ and Ca2+ ions. The thickness of this layer varies between 1 and 2 µm.It is possible to observe also an increase in the proportion of bridging oxygen during leaching.
- 4.
- Migration to the surface of Ca ions and phosphate groups through the silica rich layer both from the material and from the solution. On the silica-rich layer a second layer forms, which is composed of amorphous calcium phosphate (ACP). The formation of this second layer was confirmed by surface-sensitive shallow-angle X-ray diffraction (XRD) analysis [23].
- 5.
- Hydroxyl and carbonated groups are incorporated from the solution while the process of dissolution of the glass continues starting from the surface. The amorphous layer crystallizes becoming HCA. The resulting surface is very similar to the nano-crystalline mineral phase of the physiological bone tissue both from a compositional and a structural point of view and this allows the direct anchoring of the implant to the living tissue.
3.2.2. Ionic Dissolution Products and Osteogenesis
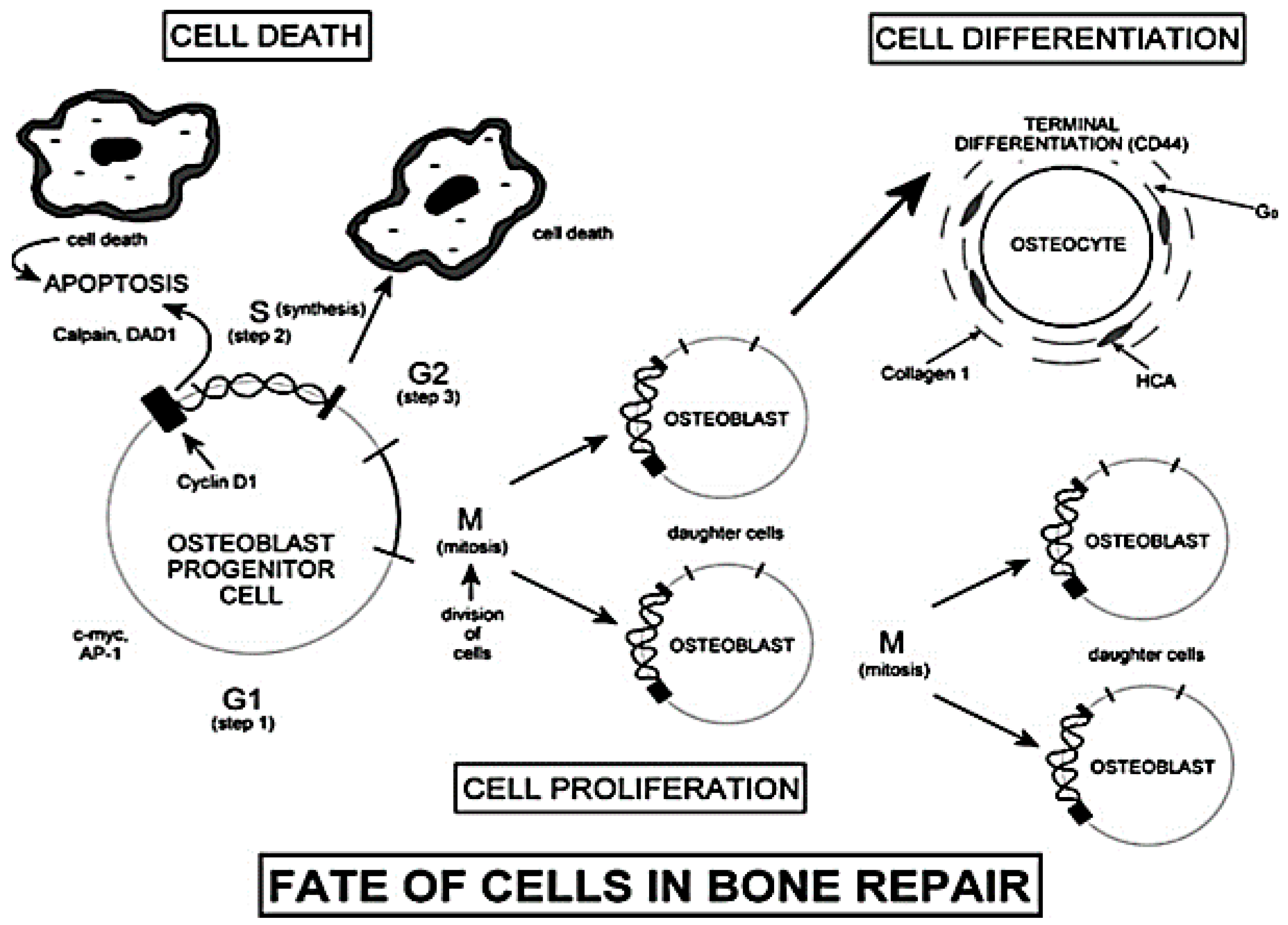
3.2.3. Influence and Genetic Control of the Ionic Dissolution Products on the Osteoblasts Cell Cycle
3.3. Effect of Doping Elements and Glass Properties
3.4. Influence of the Atomic Structure on Dissolution Rate and HCA Nucleation
4. Use and Application Fields of BGs in Clinical Practice
4.1. Bone Repair and Orthopedic Surgery
- Two surgical procedures are required, the first for harvesting—which may be painful and stressful for the patient—and the second for implantation;
- Haematoma formation, blood loss and infections;
- Arterial and ureteral injuries;
- Cosmetic defects;
- Limited availability (bone graft substitute);
4.2. Chondrogenesis and Soft Tissue Repair
5. Manufacturing Processes for the Production of BGs
5.1. Melt-Derived BGs
- Direct forming via casting into molds, quenching into water, or drawing into continuous fiber;
- Thermally treating the glass above Tg in order to allow the sintering of particles into a porous architecture, drawing of fibers from a pre-form, or sealing particles to obtain coatings on a surface [82].
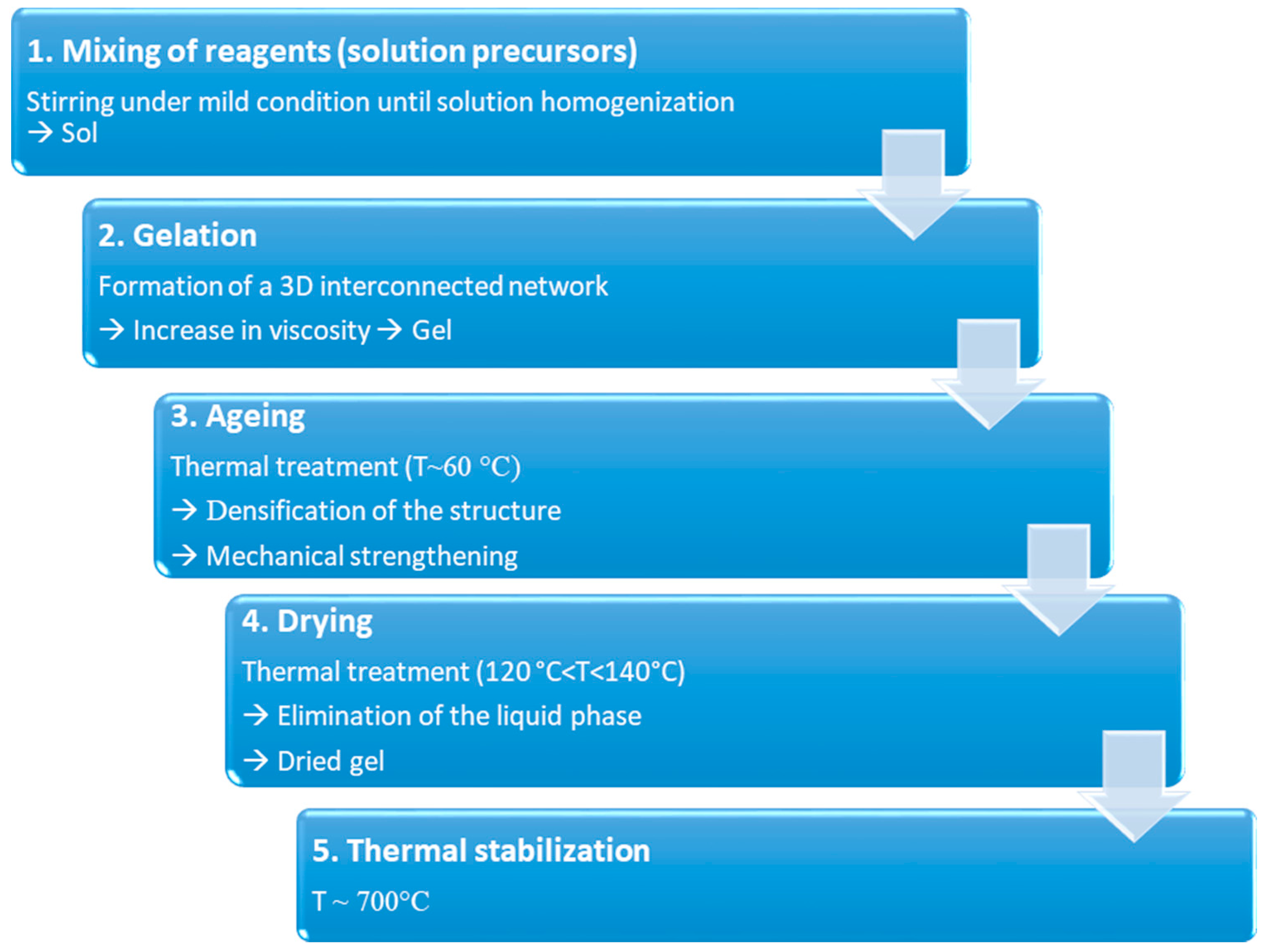
5.2. Sol–gel Derived BGs
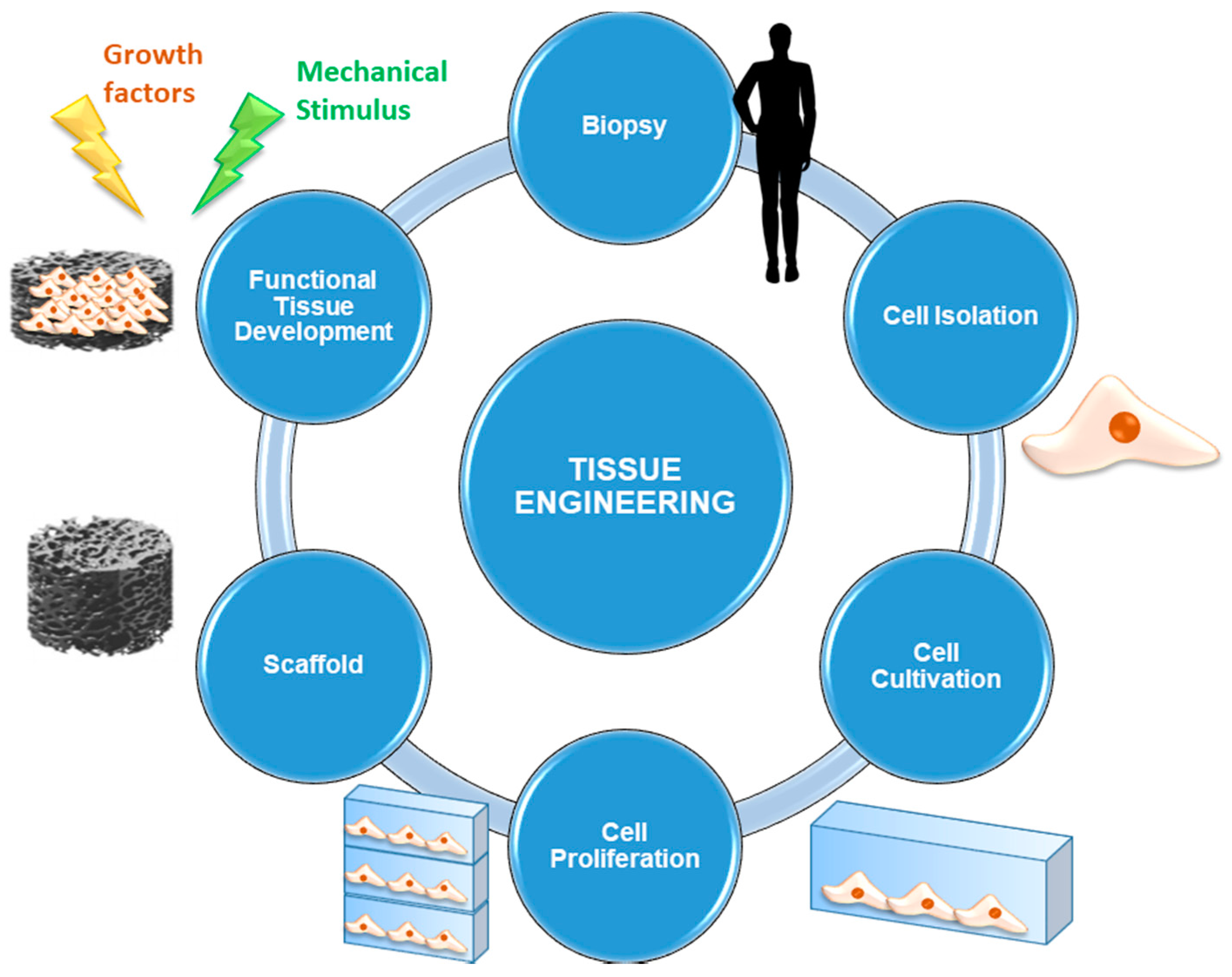
6. BGs in Tissue Engineering
- Cells are the fundamental element without which the synthesis of new tissue cannot occur. Usually, autologous cells are used: they are directly taken from the site of concern by biopsy in order to avoid the risk of rejection deriving from immune response. Alternatively, it is also possible to use stem cells, that is, undifferentiated cells that are able to evolve to multiple cell lines under the supply of appropriate stimulation protocols. Multipotent stem cells are currently used: they are taken mainly from bone marrow or other tissues, such as the adipose one, easily available in human body.
- Scaffolds represent 3D (porous) structures that are able to provide physical support to cells by stimulating cell adhesion, migration, proliferation, and differentiation processes. Currently, several types of scaffolds are available, according to the material they are made of. They may be either natural (generally derived from ECM extracted by the patient or by donors, or made by biopolymers) or synthetic (consisting of materials designed ad hoc to mimic the characteristics of the physiological tissue).
- Signals can be biological, chemical or physical-mechanical. Opportune stimulating procedures are able to influence cell pathways during the processes of proliferation and differentiation by favoring the evolution towards specific phenotypes. These signals are of considerable importance since they are able to ensure cell survival and, therefore, it is necessary that all the cells seeded on the scaffold are affected by them in the same way and with the same efficiency [98].
6.1. BG Scaffolds for Bone TE
- Biocompatibility and bioactivity The scaffold must not release toxic products within the physiological environment and it must be able to promote anchoring of osteogenic cells that trigger the formation of new bone tissue. After the implantation, the scaffold must produce a negligible immune response in order to prevent the activation of inflammatory patterns which might compromise the healing process [103]. Furthermore, since scaffolds are usually not intended as permanents implants, the constituent materials should exhibit suitable bioactivity and dissolution kinetics comparable to tissue healing rates, in order to allow cells to produce the new extracellular matrix by themselves and permit tissue to regenerate as the scaffold degrades [43];
- Capability to create a bond with living bone without the formation of a scar layer at the interface [23];
- Porous and interconnected structure in order to facilitate nutrients exchange, cell migration, and formation of a vascular network to allow tissue oxygenation [104]. An ideal bone scaffold should have an interconnected porous structure, that is, it should be highly permeable with porosity >80–90 vol % and pore diameters in the range of 10–500 μm for cell seeding, tissue ingrowth and vascularization as well as nutrients delivery and waste removal [100]. However, the minimum porosity value admitted is 50 vol %, sufficient to satisfy the necessary requirement for tissue ingrowth [43]. A bimodal pore size distribution has to be preferred in order to mimic the morphologic characteristics of cancellous bone: pores below 50 μm (preferably ≈ 2–10 μm) were found to facilitate the interaction between cells and materials and osteo-integration, while, on the other hand, pores of 100–500 μm enhance new bone formation, bone ingrowth, and capillaries formation (direct osteogenesis) [100];
- Adaptability in shape and size (mouldability) to completely fill bone defects [23];
- Suitable degradation rate in order to match the time required for the tissue regeneration and osteoclastic remodeling [23];
- Maintenance of mechanical properties during degradation and remodeling and load-sharing with host tissue. Ideally, a scaffold should exhibit mechanical properties consistent with the anatomical site of concern. This often represents a clinical challenge considering orthopedics applications. As regards bone, it is necessary to consider the variation of the tissue healing rates depending on the aging process. It is always recommended to consider this aspect together with porosity requirements, since a balance is needed for ensuring stability and integrity of the structure [23,43,100,105];
- Relatively easy fabrication, production process scalability, and low fabrication costs for large-scale production [23];
- Sterilization and suitability according to regulations for the usage of biomedical devices [23].
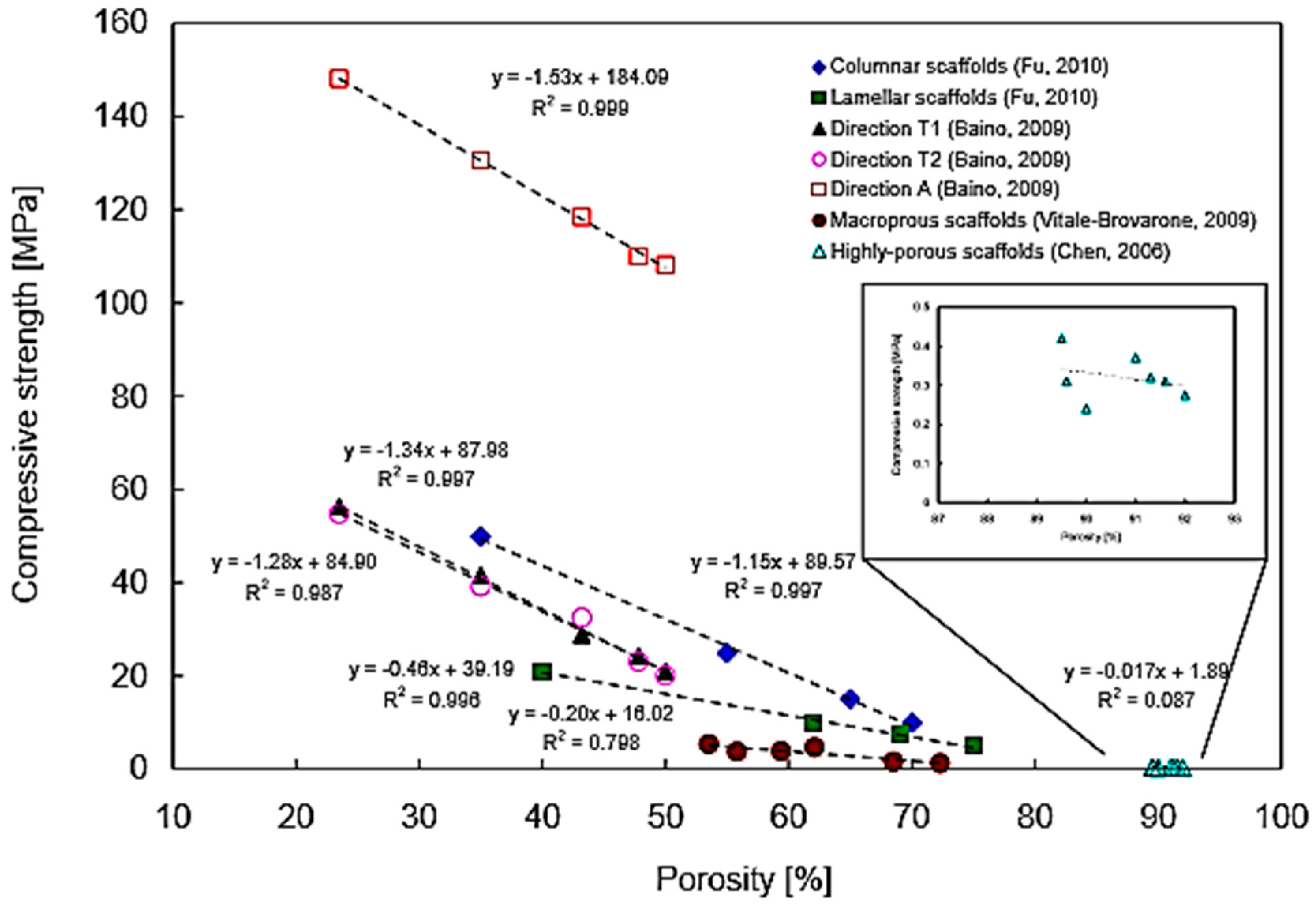
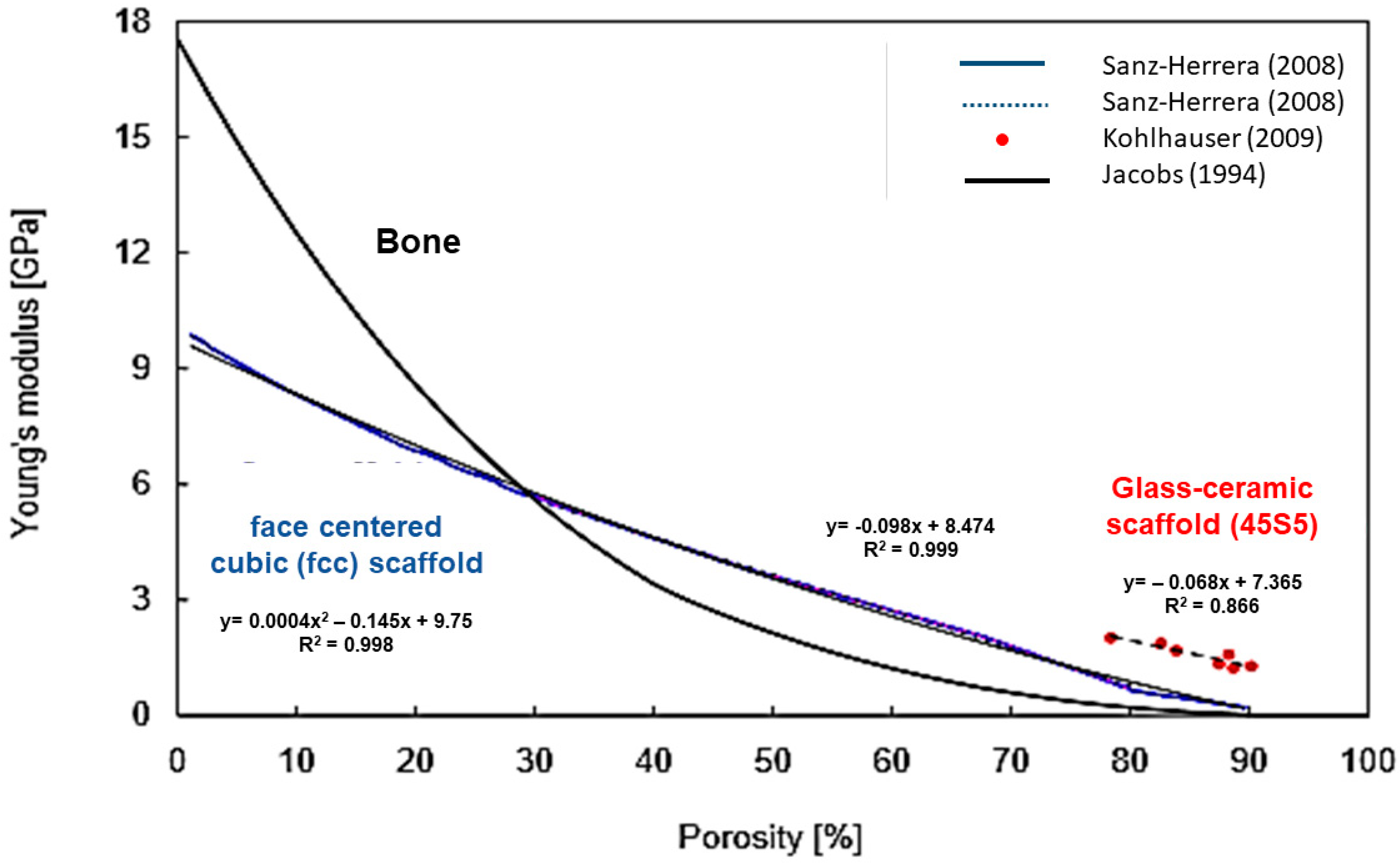
6.2. Mechanical Behavior of Silicate BG Scaffolds
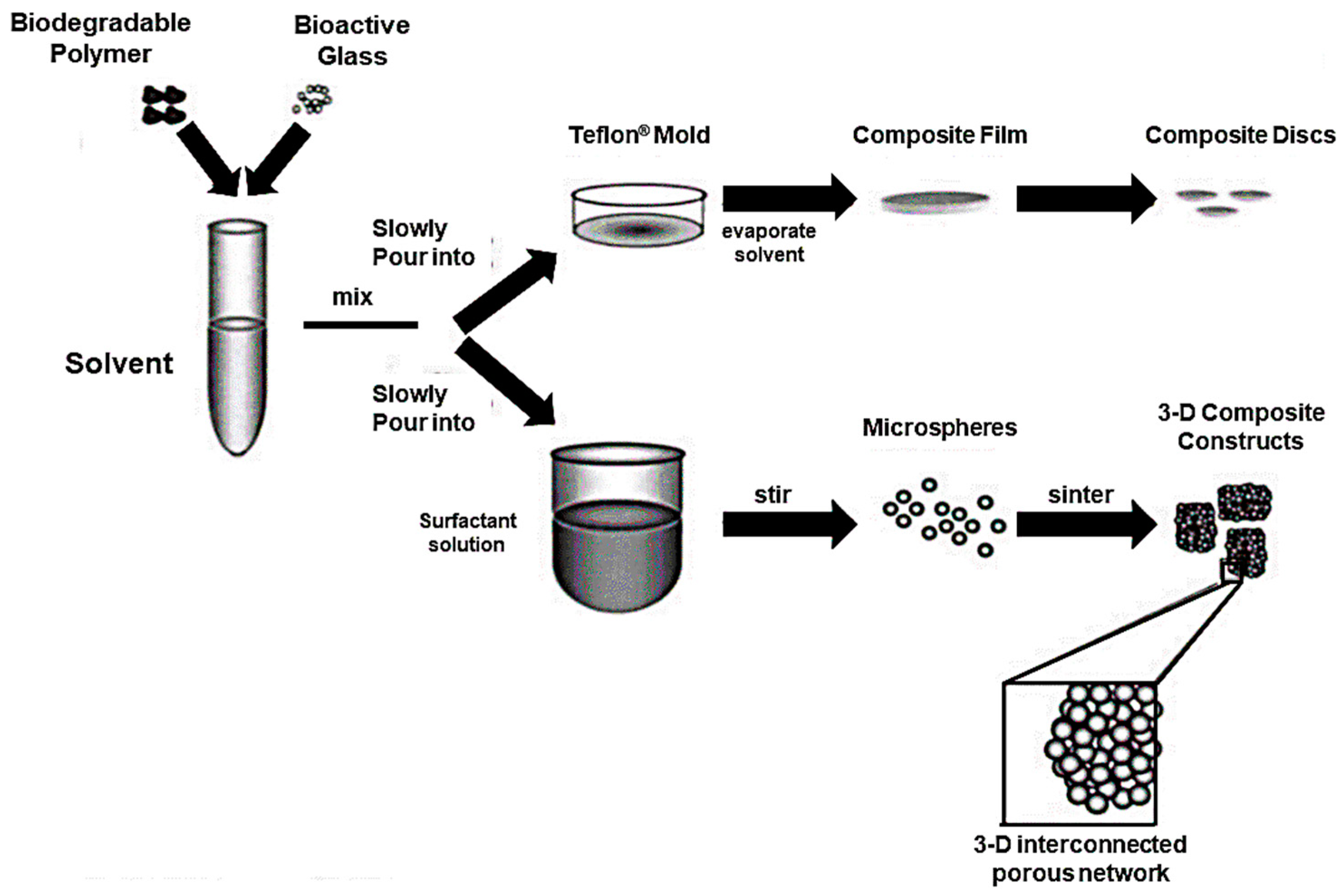
6.3. Scaffolds for Bone TE: Design and Manufacturing
- Particles sintering around a template. During the sintering process the glass particles are heated above Tg, determining their fusion in the contact points. During this procedure the amorphous structure of the glass is preserved by maintaining the temperature below the onset of crystallization. As a result, the definition of sintering window is given as the temperature range limited by Tg (lower limit) and Tx (upper limit). The sintering procedure turns out to be more effective when the particle size is small enough to avoid the formation of large defects by allowing the creation of a highly sintered and close-packed structure. On the other hand, smaller particles are responsible for higher values of surface area, which result in the enhancement of the crystallization process.
- 2.
- Foaming process: Foaming technique aims at the production of 3D highly interconnected porous structures and involves the introduction of gas bubbles into a slurry or sol.
- 3.
- Solid Free Form Techniques: SFF techniques allow the realization of a design-controlled scaffold manufacturing process. By simply varying processing parameters, it is possible to obtain a precise control on the final 3D structure.
- Production of a computer-generated model of the wanted structure by the use of a CAD software;
- Segmentation of the model into cross-sections;
- Implementation of the data;
- Production of the physical model [122].
7. Towards the Future: The Potential of Borate and Phosphate BGs in TE Approaches
Conflicts of Interest
References
- Hench, L.L.; Thompson, I. Twenty-first century challenges for biomaterials. J. R. Soc. Interface 2010, 7, S379–S391. [Google Scholar] [CrossRef] [PubMed]
- Hench, L.L. The story of Bioglass®. J. Mater. Sci. Mater. Med. 2006, 17, 967–978. [Google Scholar] [CrossRef] [PubMed]
- Hench, L.L. Biomaterials: A forecast for the future. Biomaterials 1998, 19, 1419–1423. [Google Scholar] [CrossRef]
- Hoppe, A.; Güldal, N.S.; Boccaccini, A.R. A review of the biological response to ionic dissolution products from bioactive glasses and glass-ceramics. Biomaterials 2011, 32, 2757–2774. [Google Scholar] [CrossRef] [PubMed]
- Andersson, Ö.H.; Karlsson, K.H.; Kangasniemi, K. Calcium phosphate formation at the surface of bioactive glass in vivo. J. Non-Cryst. Solids 1990, 119, 290–296. [Google Scholar] [CrossRef]
- Andersson, Ö.H.; Kangasniemi, I. Calcium phosphate formation at the surface of bioactive glass in vitro. J. Biomed. Mater. Res. A 1991, 25, 1019–1030. [Google Scholar] [CrossRef] [PubMed]
- Brink, M.; Turunen, T.; Happonen, R.P.; Yli-Urpo, A. Compositional dependence of bioactivity of glasses in the system Na2O–K2O–MgO–CaO–B2O3–P2O5–SiO2. J. Biomed. Mater. Res. 1997, 37, 114–121. [Google Scholar] [CrossRef]
- Oudadesse, H.; Mami, M.; Doebez-Sridi, R.; Pellen-Mussi, P.; Jeanne, S.; Zrineh, A.; Cathelineau, G. Study of the bioactivity of various mineral compositions of bioactive glasses. Bioceram. Dev. Appl. 2011, 1. [Google Scholar] [CrossRef]
- Vallet-Regí, M.; Ragel, C.V.; Salinas, A.J. Glasses with medical applications. Eur. J. Inorg. Chem. 2003, 2003, 1029–1042. [Google Scholar] [CrossRef]
- Hench, L.L. Chronology of bioactive glass development and clinical applications. New J. Glass Ceram. 2013, 3, 67–73. [Google Scholar] [CrossRef]
- Baino, F.; Novajra, G.; Vitale-Brovarone, C. Bioceramics and scaffolds: A winning combination for tissue engineering. Front. Bioeng. Biotechnol. 2015, 3, 202. [Google Scholar] [CrossRef] [PubMed]
- Baino, F.; Vitale-Brovarone, C. Three-dimensional glass-derived scaffolds for bone tissue engineering: Current trends and forecasts for the future. J. Biomed. Mater. Res. A 2011, 97, 514–535. [Google Scholar] [CrossRef] [PubMed]
- Fu, Q.; Saiz, E.; Tomsia, A.P. Direct ink writing of highly porous and strong glass scaffolds for load-bearing bone defects repair and regeneration. Acta Biomater. 2011, 7, 3547–3554. [Google Scholar] [CrossRef] [PubMed]
- Hench, L.L.; Jones, J.R. Bioactive glasses: Frontiers and challenges. Front. Bioeng. Biotechnol. 2015, 3, 194. [Google Scholar] [CrossRef] [PubMed]
- Miguez-Pacheco, V.; Hench, L.L.; Boccaccini, A.R. Bioactive glasses beyond bone and teeth: Emerging applications in contact with soft tissues. Acta Biomater. 2015, 13, 1–15. [Google Scholar] [CrossRef] [PubMed]
- Baino, F.; Novajra, G.; Miguez-Pacheco, V.; Boccaccini, A.R.; Vitale-Brovarone, C. Bioactive glasses: Special applications outside the skeletal system. J. Non-Cryst. Solids 2016, 432, 15–30. [Google Scholar] [CrossRef]
- Gorustovich, A.A.; Roether, J.A.; Boccaccini, A.R. Effect of bioactive glasses on angiogenesis: A review of in vitro and in vivo evidences. Tissue Eng. Part B Rev. 2009, 16, 199–207. [Google Scholar] [CrossRef] [PubMed]
- Hoppe, A.; Mouriño, V.; Boccaccini, A.R. Therapeutic inorganic ions in bioactive glasses to enhance bone formation and beyond. Biomater. Sci. 2013, 1, 254–256. [Google Scholar] [CrossRef]
- Suominen, E.; Aho, A.J.; Vedel, E.; Kangasniemi, I.; Uusipaikka, E.; Yli-Urpo, A. Subchondral bone and cartilage repair with bioactive glasses, hydroxyapatite, and hydroxyapatite-glass composite. J. Biomed. Mater. Res. 1996, 32, 543–551. [Google Scholar] [CrossRef]
- Lovett, M.; Lee, K.; Edwards, A.; Kaplan, D.L. Vascularization strategies for tissue engineering. Tissue Eng. Part B Rev. 2009, 15, 353–370. [Google Scholar] [CrossRef] [PubMed]
- Hench, L.L.; West, J.K. The sol–gel process. Chem. Rev. 1990, 90, 33–72. [Google Scholar] [CrossRef]
- Hench, L.L. Bioceramics: From concept to clinic. J. Am. Ceram. Soc. 1991, 74, 1487–1510. [Google Scholar] [CrossRef]
- Jones, J.R. Review of bioactive glass: From Hench to hybrids. Acta Biomater. 2013, 9, 4457–4486. [Google Scholar] [CrossRef] [PubMed]
- Detsch, R.; Alles, S.; Hum, J.; Westenberger, P.; Sieker, F.; Heusinger, D.; Kasper, C.; Boccaccini, A.R. Osteogenic differentiation of umbilical cord and adipose derived stem cells onto highly porous 45S5 Bioglass®-based scaffolds. J. Biomed. Mater. Res. A 2015, 103, 1029–1037. [Google Scholar] [CrossRef] [PubMed]
- Tsigkou, O.; Jones, J.R.; Polak, J.M.; Stevens, M.M. Differentiation of fetal osteoblasts and formation of mineralized bone nodules by 45S5 Bioglass® conditioned medium in the absence of osteogenic supplements. Biomaterials 2009, 30, 3542–3550. [Google Scholar] [CrossRef] [PubMed]
- Xynos, I.D.; Edgar, A.J.; Buttery, L.D.K.; Hench, L.L.; Polak, J.M. Gene-expression profiling of human osteoblasts following treatment with the ionic products of Bioglass® 45S5 dissolution. J. Biomed. Mater. Res. 2001, 55, 151–157. [Google Scholar] [CrossRef]
- Chen, Q.Z.; Thompson, I.D.; Boccaccini, A.R. 45S5 Bioglass®-derived glass-ceramic scaffolds for bone tissue engineering. Biomaterials 2006, 27, 2414–2425. [Google Scholar] [CrossRef] [PubMed]
- Chen, Q.Z.; Thouas, G.A. Fabrication and characterization of sol–gel derived 45S5 Bioglass®-ceramic scaffolds. Acta Biomater. 2011, 7, 3616–3626. [Google Scholar] [CrossRef] [PubMed]
- Qian, J.; Kang, Y.; Zhang, W. Fabrication and characterization of biomorphic 45S5 Bioglass scaffold from sugarcane. Mater. Sci. Eng. C 2009, 29, 1361–1364. [Google Scholar] [CrossRef]
- Boccardi, E.; Philippart, A.; Juhasz-Bortuzzo, J.A.; Novajra, G.; Vitale-Brovarone, C.; Boccaccini, A.R. Characterisation of Bioglass based foams developed via replication of natural marine sponges. Adv. Appl. Ceram. 2015, 114, S56–S62. [Google Scholar] [CrossRef]
- Liu, X.; Rahaman, M.N.; Hilmas, G.E.; Bal, B.S. Mechanical properties of bioactive glass (13-93) scaffolds fabricated by robotic deposition for structural bone repair. Acta Biomater. 2013, 9, 7025–7034. [Google Scholar] [CrossRef] [PubMed]
- Eqtesadi, S.; Motealleh, A.; Miranda, P.; Pajares, A.; Lemos, A.; Ferreira, J.M.F. Robocasting of 45S5 bioactive glass scaffolds for bone tissue engineering. J. Eur. Ceram. Soc. 2014, 34, 107–118. [Google Scholar] [CrossRef]
- Motealleh, A.; Eqtesadi, S.; Civantos, A.; Pajares, A.; Miranda, P. Robocast 45S5 Bioglass scaffolds: In vitro behavior. J. Mater. Sci. 2017, 52, 9179–9191. [Google Scholar] [CrossRef]
- Meincke, T.; Miguez-Pacheco, V.; Hoffmann, D.; Boccaccini, A.R.; Taylor, R.N.K. Engineering the surface functionality of 45S5 bioactive glass-based scaffolds by the heterogeneous nucleation and growth of silver particles. J. Mater. Sci. 2017, 52, 9082–9090. [Google Scholar] [CrossRef]
- Ghoreishian, S.M.; Jamalpoor, M. Clinical, radiographic and histologic comparison of ridge augumentation with bioactive glass alone and in combination with autogenous bone graft. Dent. Res. J. 2006, 2, 1–9. [Google Scholar]
- Laurencin, C.; Khan, Y.; El-Amin, S.F. Bone graft substitutes. Expert Rev. Med. Devices 2006, 3, 49–57. [Google Scholar] [CrossRef] [PubMed]
- Strnad, Z.; Sestak, J. Bio-compatible ceramics as mimetic material for bone tissue substitution. In Proceedings of the Second International Conference on Intelligent Processing and Manufacturing of Materials (PMM ’99), Honolulu, HI, USA, 10–15 July 1999; Volume 1, pp. 431–436. [Google Scholar]
- Giannoudis, P.V.; Dinopoulos, H.; Tsiridis, E. Bone substitutes: An update. Injury 2005, 36, S20–S27. [Google Scholar] [CrossRef] [PubMed]
- Moore, W.R.; Graves, S.E.; Bain, G.I. Synthetic bone graft substitutes. ANZ J. Surg. 2001, 71, 354–361. [Google Scholar] [CrossRef] [PubMed]
- Oonishi, H.; Hench, L.L.; Wilson, J.; Sugihara, F.; Tsuji, E.; Matsuura, M.; Kin, S.; Yamamoto, T.; Mizokawa, S. Quantitative comparison of bone growth behaviour in granules of Bioglass, A-W glass-ceramic, and hydroxyapatite. J. Biomed. Mater. Res. 2000, 51, 37–46. [Google Scholar] [CrossRef]
- Cao, W.; Hench, L.L. Bioactive materials. Ceram. Int. 1996, 22, 493–507. [Google Scholar] [CrossRef]
- Greenspan, D.C. Bioactive glass: Mechanism of bone bonding. Tandläkartidn. Ǻrk 1999, 91, 1–32. [Google Scholar]
- Rahaman, M.N.; Day, D.E.; Bal, B.S.; Fu, Q.; Jung, S.B.; Bonewald, L.F.; Tomsia, A.P. Bioactive glass in tissue engineering. Acta Biomater. 2011, 7, 2355–2373. [Google Scholar] [CrossRef] [PubMed]
- Abou Neel, E.A.; Pickup, D.M.; Valappil, S.P.; Newport, R.J.; Knowles, J.C. Bioactive functional materials: A perspective on phosphate-based glasses. J. Mater. Chem. 2009, 19, 690–701. [Google Scholar] [CrossRef]
- Hench, L.L. Genetic design of bioactive glass. J. Eur. Ceram. Soc. 2009, 29, 1257–1265. [Google Scholar] [CrossRef]
- Xynos, I.D.; Edgar, A.J.; Buttery, L.D.K.; Hench, L.L.; Polak, J.M. Ionic products of bioactive glass dissolution increase proliferation of human osteoblasts and induce insulin-like growth factors II mRNA expression and protein synthesis. Biochem. Biophys. Res. Commun. 2000, 276, 461–465. [Google Scholar] [CrossRef] [PubMed]
- Rabiee, S.M.; Nazparvar, N.; Azizian, M.; Vashaee, D.; Tayebi, L. Effect of ion substitution on properties of bioactive glasses: A review. Ceram. Int. 2015, 41, 7241–7251. [Google Scholar] [CrossRef]
- Yamaguchi, M. Role of zinc in bone formation and bone resorption. J. Trace Elem. Exp. Med. 1998, 11, 119–135. [Google Scholar] [CrossRef]
- Yamaguchi, M.; Oishi, H.; Suketa, Y. Stimulatory effect of zinc in bone formation in tissue culture. Biochem. Pharmacol. 1987, 36, 4007–4012. [Google Scholar] [CrossRef]
- Fung, E.B.; Kwiatkowski, J.L.; Huang, J.N.; Gildengorin, G.; king, J.C.; Vichinsky, E.P. Zinc supplementation improves bone density in patients with thalassemia: A double-blind, randomized, placebo-controlled trial 1–3. Am. J. Clin. Nutr. 2013, 98, 960–971. [Google Scholar] [CrossRef] [PubMed]
- Aina, V.; Malavasi, G.; Fiorio Pla, A.; Munaron, L.; Monterra, C. Zinc-containing bioactive glasses: Surface reactivity and behaviour towards endothelial cells. Acta Biomater. 2009, 5, 1211–1222. [Google Scholar] [CrossRef] [PubMed]
- Sharabi, S.; Hesaraki, S.; Moemeni, S.; Khorami, M. Structural discrepancies and in vitro nanoapatite formation ability of sol–gel derived glasses doped with different bone stimulator ions. Ceram. Int. 2011, 37, 2737–2746. [Google Scholar] [CrossRef]
- Oudadesse, H.; Dietrich, E.; Gal, Y.L.; Pellen, P.; Bureau, B.; Mostafa, A.A.; Cathelineau, G. Apatite forming ability and cytocompatibility of pure Zn-doped bioactive glasses. Biomed. Mater. 2011, 6, 035006. [Google Scholar] [CrossRef] [PubMed]
- Lusvardi, G.; Zaffe, D.; Menabue, L.; Bertoldi, C.; Malavasi, G.; Consolo, U. In vitro and in vivo behaviour of zinc-doped phosphosilicate glasses. Acta Biomater. 2009, 5, 419–428. [Google Scholar] [CrossRef] [PubMed]
- O’Donnell, M.D.; Hill, R.G. Influence of strontium and the importance of glass chemistry and structure when designing bioactive glasses for bone regeneration. Acta Biomater. 2010, 6, 2382–2385. [Google Scholar] [CrossRef] [PubMed]
- Capuccini, C.; Torricelli, P.; Sima, F.; Boanini, E.; Ristoscu, C.; Bracci, B.; Socol, G.; Fini, M.; Mihailescu, I.N.; Bigi, A. Strontium-substituted hydroxyapatite coatings synthetized by pulsed-laser deposition: In vitro osteoblast and osteoclast response. Acta Biomater. 2008, 4, 1885–1893. [Google Scholar] [CrossRef] [PubMed]
- Bonnelye, E.; Chabadel, A.; Saltel, F.; Jurdic, P. Dual effect of strontium renelate: Stimulation of osteoblast differentiation and inhibition of osteoclast formation and response in vitro. Bone 2008, 42, 129–138. [Google Scholar] [CrossRef] [PubMed]
- Du, J.; Xiang, Y. Effect of strontium substitution on the structure, ionic diffusion and dynamic properties of 45S5 bioactive glasses. J. Non-Cryst. Solids 2012, 358, 1059–1071. [Google Scholar] [CrossRef]
- Du, J.; Xiang, Y. Investigating the structure-diffusion-bioactivity relationship of strontium containing bioactive glasses using molecular dynamics based computer stimulation. J. Non-Cryst. Solids 2016, 432, 35–40. [Google Scholar] [CrossRef]
- Isaac, J.; Nohra, J.; Lao, J.; Jallot, E.; Nedelec, J.M.; Berdal, A.; Sautier, J.M. Effect of strontium-doped bioactive glass on the differentiation of cultured osteogenic cells. Eur. Cells Mater. 2011, 21, 130–143. [Google Scholar] [CrossRef]
- Chopra, I. The increasing use of silver-based products as antimicrobial agents: A useful development or a cause for concern? J. Antimicrob. Chemother. 2007, 59, 587–590. [Google Scholar] [CrossRef] [PubMed]
- Zhu, H.; Hu, C.; Zhang, F.; Feng, X.; Li, J.; Liu, T.; Chen, J.; Zhang, J. Preparation and antibacterial property of silver-containing mesoporous 58S bioactive glass. Mater. Sci. Eng. C 2014, 42, 22–30. [Google Scholar] [CrossRef] [PubMed]
- Balamurugan, A.; Balossier, G.; Laurent-Maquin, D.; Pina, S.; Rebelo, A.H.; Faure, J.; Ferreira, J.M. An in vitro biological and anti-bacterial study on a sol–gel derived silver-incorporated Bioglass system. Dent. Mater. 2008, 24, 1343–1351. [Google Scholar] [CrossRef] [PubMed]
- El-Kady, A.M.; Ali, A.F.; Rizk, R.A.; Ahmed, M.M. Synthesis, characterizatiom and microbiological response of silver doped bioactive glass nanoparticles. Ceram. Int. 2012, 38, 177–188. [Google Scholar] [CrossRef]
- Vernè, E.; Di Nunzio, S.; Bosetti, M.; Appendino, P.; Brovarone, C.V.; Maina, G.; Cannas, M. Surface characterization of silver-doped bioactive glass. Biomaterials 2005, 26, 5111–5119. [Google Scholar] [CrossRef] [PubMed]
- Bellantone, M.; Williams, H.D.; Hench, L.L. Broad-spectrum bactericidal activity of Ag2O-doped bioactive glass. Antimicrob. Agents Chemother. 2002, 46, 1940–1945. [Google Scholar] [CrossRef] [PubMed]
- Bellantone, M.; Coleman, N.J.; Hench, L.L. Bacteriostatic action of a novel four-component bioactive glass. J. Biomed. Mater. Res. 2000, 51, 484–490. [Google Scholar] [CrossRef]
- El-Rashidy, A.A.; Waly, G.; Gad, A.; Hashem, A.A.; Balasubramanian, P.; Kaya, S.; Boccaccini, A.R.; Sami, I. Preparation and in vitro characterization of silver-doped bioactive glass nanoparticles fabricated using a sol–gel process and modified Stöber method. J. Non-Cryst. Solids 2018, 483, 26–36. [Google Scholar] [CrossRef]
- Miola, M.; Vernè, E.; Vitale-Brovarone, C.; Baino, F. Antibacterial Bioglass-derived scaffolds: Innovative synthesis approach and characterization. Int. J. Appl. Glass Sci. 2016, 7, 238–247. [Google Scholar] [CrossRef]
- Zhao, S.; Li, L.; Wang, H.; Zhang, Y.; Cheng, X.; Zhou, N.; Rahaman, M.N.; Liu, Z.; Huang, W.; Zhang, C. Wound dressing composed of copper-doped borate bioactive glass microfibers stimulate angiogenesis and heal full-thickness skin defects in a rodent model. Biomaterials 2015, 53, 379–391. [Google Scholar] [CrossRef] [PubMed]
- Kargozar, S.; Lotfibakhshaiesh, N.; Ai, J.; Mozafari, M.; Brouki Milan, P.; Hamzehlou, S.; Barati, M.; Baino, F.; Hill, R.G.; Joghataei, M.T. Strontium- and cobalt-substituted bioactive glasses seeded with human umbilical cord perivascular cells to promote bone regeneration via enhanced osteogenic and angiogenic activities. Acta Biomater. 2017, 58, 502–514. [Google Scholar] [CrossRef] [PubMed]
- Hill, R.G.; Brauer, D.S. Predicting the bioactivity of glasses using the network connectivity or split network models. J. Non-Cryst. Solids 2011, 357, 3884–3887. [Google Scholar] [CrossRef]
- Martin, R.A.; Yue, S.; Hanna, J.V.; Lee, P.D.; Newport, R.J.; Smith, M.E.; Jones, J.R. Characterizing the hierarchical structures of bioactive sol–gel silicate glass and hybrid scaffolds for bone regeneration. Philos. Trans. R. Soc. A Math. Phys. Eng. Sci. 2012, 370, 1422–1443. [Google Scholar] [CrossRef] [PubMed]
- Edén, M. The split network analysis for exploring composition-structure correlations in multi-component glasses: I. Rationalizing bioactivity-composition trends of bioglasses. J. Non-Cryst. Solids 2011, 357, 1595–1602. [Google Scholar] [CrossRef]
- Calori, G.M.; Mazza, E.; Colombo, M.; Ripamonti, C. The use of bone-graft substitutes in large bone defects: Any specific needs? Injury 2011, 42, S56–S63. [Google Scholar] [CrossRef] [PubMed]
- Lewandrowski, K.U.; Gresser, J.D.; Wise, D.L.; Trantolo, D.J. Bioresorbable bone graft substitutes of different osteoconductivities: A histologic evaluation of osteointegration of poly(propylene glycol-co-fumaric acid)-based cement implants in rats. Biomaterials 2000, 21, 757–764. [Google Scholar] [CrossRef]
- Mauffrey, C.; Seligson, D.; Lichte, P.; Pape, H.C.; Al-Rayyan, M. Bone graft substitutes for articular support and metaphyseal comminution: What are the options? Injury 2011, 42, S35–S39. [Google Scholar] [CrossRef] [PubMed]
- Baino, F.; Vernè, E. Glass-based coatings on biomedical implants: A state-of-the-art review. Biomed. Glasses 2017, 3. [Google Scholar] [CrossRef]
- Kargozar, S.; Hamzehlou, S.; Baino, F. Potential of bioactive glasses for cardiac and pulmonary tissue engineering. Materials 2017, 10, 1429. [Google Scholar] [CrossRef] [PubMed]
- Baino, F. How can Bioactive glasses be useful in ocular surgery? J. Biomed. Mater. Res. A 2015, 103, 1259–1275. [Google Scholar] [CrossRef] [PubMed]
- Ylänen, H. Bioactive Glasses: Materials, Properties and Applications, 2nd ed.; Woodhead Publishing: Sawston, UK, 2017; ISBN 9780081009376. [Google Scholar]
- Boccaccini, A.R.; Brauer, D.S.; Hupa, L. Bioactive Glasses: Fundamentals, Technology and Applications; Royal Society of Chemistry: London, UK, 2016; ISBN 9781782629764. [Google Scholar]
- Sepulveda, P.; Jones, J.R.; Hench, L.L. Characterization of melt-derived 45S5 and sol–gel-derived 58S bioactive glasses. J. Biomed. Mater. Res. 2001, 58, 734–740. [Google Scholar] [CrossRef] [PubMed]
- Sepulveda, P.; Jones, J.R.; Hench, L.L. In vitro dissolution of melt derived 45S5 and sol–gel derived 58S bioactive glasses. Biomed. Mater. Res. 2002, 61, 301–311. [Google Scholar] [CrossRef] [PubMed]
- Owens, G.J.; Singh, R.K.; Foroutan, F.; Alqaysi, M.; Han, C.-M.; Mahapatra, C.; Kim, H.-W.; Knowles, J.C. Sol–gel based materials for biomedical applications. Prog. Mater. Sci. 2016, 77, 1–79. [Google Scholar] [CrossRef]
- Jones, J.R.; Ehrenfried, L.M.; Hench, L.L. Optimising bioactive glass scaffolds for bone tissue engineering. Biomaterials 2006, 27, 964–973. [Google Scholar] [CrossRef] [PubMed]
- Pereira, M.M.; Clark, A.E.; Hench, L.L. Calcium phosphate formation on sol–gel-derived bioactive glass in vitro. J. Biomed. Mater. Res. A 1994, 28, 693–698. [Google Scholar] [CrossRef] [PubMed]
- Li, R.; Clark, A.E.; Hench, L.L. An investigation of bioactive glass powders by sol–gel processing. J. Appl. Biomater. 1991, 2, 231–239. [Google Scholar] [CrossRef] [PubMed]
- Jones, J.R.; Lee, P.D.; Hench, L.L. Hierarchical porous materials for tissue engineering. Philos. Trans. A Math. Phys. Eng. Sci. 2006, 364, 263–281. [Google Scholar] [CrossRef] [PubMed]
- Kresge, C.T.; Leonowicz, M.E.; Roth, W.J.; Vartuli, J.C.; Beck, J.S. Ordered mesoporous molecular sieves synthesized by a liquid-crystal template mechanism. Nature 1992, 359, 710–712. [Google Scholar] [CrossRef]
- Arcos, D.; Vallet-Regi, M. Bioceramics for drug delivery. Acta Mater. 2013, 61, 890–911. [Google Scholar] [CrossRef]
- Pereira, M.M.; Clark, A.E.; Hench, L.L. Effect of Texture on the Rate of Hydroxyapatite Formation on Gel-Silica Surface. J. Am. Ceram. Soc. 1995, 78, 2463–2468. [Google Scholar] [CrossRef]
- Gupta, R.; Kumar, A. Bioactive materials for biomedical applications using sol–gel technology. Biomed. Mater. 2008, 3, 034005. [Google Scholar] [CrossRef] [PubMed]
- Avnir, D. Organic chemistry within ceramic matrixes: Doped sol–gel materials. Acc. Chem. Res. 1995, 28, 328–334. [Google Scholar] [CrossRef]
- Arcos, D.; Vallet-Regí, M. Sol–gel silica-based biomaterials and bone tissue regeneration. Acta Biomater. 2010, 6, 2874–2888. [Google Scholar] [CrossRef] [PubMed]
- Saravanapavan, P.; Jones, J.R.; Pryce, R.S.; Hench, L.L. Bioactivity of gel-glass powders in the CaO–SiO2 system: A comparison with ternary (CaO–P2O5–SiO2) and quaternary glasses (SiO2–CaO–P2O5–Na2O). J. Biomed. Mater. Res. A 2003, 66, 110–119. [Google Scholar] [CrossRef] [PubMed]
- Wu, C.; Chang, J. Multifunctional mesoporous bioactive glasses for effective delivery of therapeutic ions and drug/growth factors. J. Control. Release 2014, 193, 282–295. [Google Scholar] [CrossRef] [PubMed]
- Lanza, R.; Langer, R.; Vacanti, J. Principles of Tissue Engineering, 3rd ed.; Academic Press: Cambridge, MA, USA, 2011; ISBN 9780080548845. [Google Scholar]
- Karp, J.M.; Dalton, P.D.; Shoichet, M.S. Scaffolds for tissue engineering. MRS Bull. 2003, 28, 301–306. [Google Scholar] [CrossRef]
- Gerhardt, L.-C.; Boccaccini, A.R. Bioactive glass and glass-ceramic scaffolds for bone tissue engineering. Materials 2010, 3, 3867–3910. [Google Scholar] [CrossRef] [PubMed]
- Sola, A.; Bellucci, D.; Cannillo, V.; Cattini, A. Bioactive glass coatings: A review. Surf. Eng. 2011, 27, 560–572. [Google Scholar] [CrossRef]
- Karageorgiou, V.; Kaplan, D. Porosity of 3D biomaterial scaffolds and osteogenesis. Biomaterials 2005, 26, 5474–5491. [Google Scholar] [CrossRef] [PubMed]
- O’Brien, F.J. Biomaterials and scaffolds for tissue engineering. Mater. Today 2011, 14, 88–95. [Google Scholar] [CrossRef]
- Dee, K.C.; Puleo, D.; Bizios, R. Engineering of materials for biomedical applications. Mater. Today 2000, 3, 7–10. [Google Scholar] [CrossRef]
- Hollister, S.J. Scaffolds design and manufacturing: From concept to clinic. Adv. Mater. 2009, 21, 3330–3342. [Google Scholar] [CrossRef] [PubMed]
- Chen, Q.; Baino, F.; Spriano, S.; Pugno, N.M.; Vitale-Brovarone, C. Modelling of the strength-porosity relationship in glass-ceramic foam scaffolds for bone repair. J. Eur. Ceram. Soc. 2014, 34, 2663–2673. [Google Scholar] [CrossRef]
- Liverani, L.; Hum, J.; Boccaccini, A.R. Proteins as coating materials on bioactive glass-based composite foams. In Proceedings of the Syntactic and Composite Foams V, Siracusa, Italy, 26–31 May 2017. [Google Scholar]
- Ma, J.; Chen, C.Z.; Wang, D.G.; Meng, X.G.; Shi, J.Z. Influence of the sintering temperature on the structural feature and bioactivity of sol–gel derived SiO2–CaO–P2O5 Bioglass. Ceram. Int. 2010, 36, 1911–1916. [Google Scholar] [CrossRef]
- Liu, X.; Rahaman, M.N.; Fu, Q. Bone regeneration in strong porous bioactive glass (13-93) scaffolds with an oriented microstructure implanted in rat calvarial defects. Acta Biomater. 2013, 9, 4889–4898. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.; Rahaman, M.N.; Fu, Q.; Tomsia, A.P. Porous and strong bioactive glass (13-93) scaffolds prepared by unidirectional freezing of camphene-based suspensions. Acta Biomater. 2012, 8, 415–423. [Google Scholar] [CrossRef] [PubMed]
- Fu, Q.; Rahaman, M.N.; Sonny Bal, B.; Brown, R.F.; Day, D.E. Mechanical and in vitro performance of 13-93 bioactive glass scaffolds prepared by a polymer foam replication technique. Acta Biomater. 2008, 4, 1854–1864. [Google Scholar] [CrossRef] [PubMed]
- Rezwan, K.; Chen, Q.Z.; Blaker, J.J.; Boccaccini, A.R. Biodegradable and bioactive porous polymer/inorganic composite scaffolds for bone tissue engineering. Biomaterials 2006, 27, 3413–3431. [Google Scholar] [CrossRef] [PubMed]
- Lu, H.H.; El-Amin, S.F.; Scott, K.D.; Laurencin, C.T. Three-dimensional, bioactive, biodegradable, polymer-bioactive glass composite scaffolds with improved mechanical properties support collagen synthesis and mineralization of human osteoblast-like cells in vitro. J. Biomed. Mater. Res. 2003, 64A, 465–474. [Google Scholar] [CrossRef] [PubMed]
- Fu, Q.; Jia, W.; Lau, G.Y.; Tomsia, A.P. Strength, toughness, and reliability of a porous glass/biopolymer composite scaffold. J. Biomed. Mater. Res. B Appl. Biomater. 2018, 106, 1209–1217. [Google Scholar] [CrossRef] [PubMed]
- Gao, C.; Feng, P.; Peng, S.; Shuai, C. Carbon nanotube, graphene and boron nitride nanotube reinforced bioactive ceramics for bone repair. Acta Biomater. 2017, 61, 1–20. [Google Scholar] [CrossRef] [PubMed]
- Erasmus, E.P.; Johnson, O.T.; Sigalas, I.; Massera, J. Effects of sintering temperature on crystallization and fabrication of porous bioactive glass scaffolds for bone regeneration. Sci. Rep. 2017, 7, 6046. [Google Scholar] [CrossRef] [PubMed]
- Baino, F.; Caddeo, S.; Novajra, G.; Vitale-Brovarone, C. Using porous bioceramic scaffolds to model healthy and osteoporotic bone. J. Eur. Ceram. Soc. 2016, 36, 2175–2182. [Google Scholar] [CrossRef]
- Sarkar, S.K.; Lee, B.T. Hard tissue regeneration using bone substitutes: An update on innovations in materials. Korean J. Intern. Med. 2015, 30, 279–293. [Google Scholar] [CrossRef] [PubMed]
- Mallick, K.K. Freeze casting of porous bioactive glass and bioceramics. J. Am. Ceram. Soc. 2009, 92, 85–94. [Google Scholar] [CrossRef]
- Sepulveda, P.; Jones, J.R.; Hench, L.L. Bioactive sol–gel foams for tissue repair. J. Biomed. Mater. Res. 2002, 59, 340–348. [Google Scholar] [CrossRef] [PubMed]
- Wu, Z.Y.; Hill, R.G.; Yue, S.; Nightingale, D.; Lee, P.D.; Jones, J.R. Melt-derived bioactive glass scaffolds produced by a gel-cast foaming technique. Acta Biomater. 2011, 7, 1807–1816. [Google Scholar] [CrossRef] [PubMed]
- Sachlos, E.; Czernuszka, J.T. Making tissue engineering scaffolds work. Review: The application of solid freeform fabrication technology to the production of tissue engineering scaffolds. Eur. Cells Mater. 2003, 5, 29–40. [Google Scholar] [CrossRef]
- Lu, H.; Zhang, T.; Wang, X.P.; Fang, Q.F. Electrospun submicron bioactive glass fibers for bone tissue scaffold. J. Mater. Sci. Mater. Med. 2009, 20, 793–798. [Google Scholar] [CrossRef] [PubMed]
- Bose, S.; Roy, M.; Bandyopadhyay, A. Recent advances in bone tissue engineering scaffolds. Trends Biotechnol. 2012, 30, 546–554. [Google Scholar] [CrossRef] [PubMed]
- Fu, Q.; Rahaman, M.N.; Fu, H.; Liu, X. Silicate, borosilicate, and borate bioactive glass scaffolds with controllable degradation rate for bone tissue engineering applications. I. Preparation and in vitro degradation. J. Biomed. Mater. Res. A 2010, 95, 164–171. [Google Scholar] [CrossRef] [PubMed]
- Hakki, S.S.; Bozkurt, B.S.; Hakki, E.E. Boron regulates mineralized tissue-associated proteins in osteoblasts (MC3T3-E1). J. Trace Elem. Med. Biol. 2010, 24, 243–250. [Google Scholar] [CrossRef] [PubMed]
- Naghii, M.R.; Torkaman, G.; Mofid, M. Effects of boron and calcium supplementation on mechanical properties of bone in rats. Biofactors 2006, 28, 195–201. [Google Scholar] [CrossRef] [PubMed]
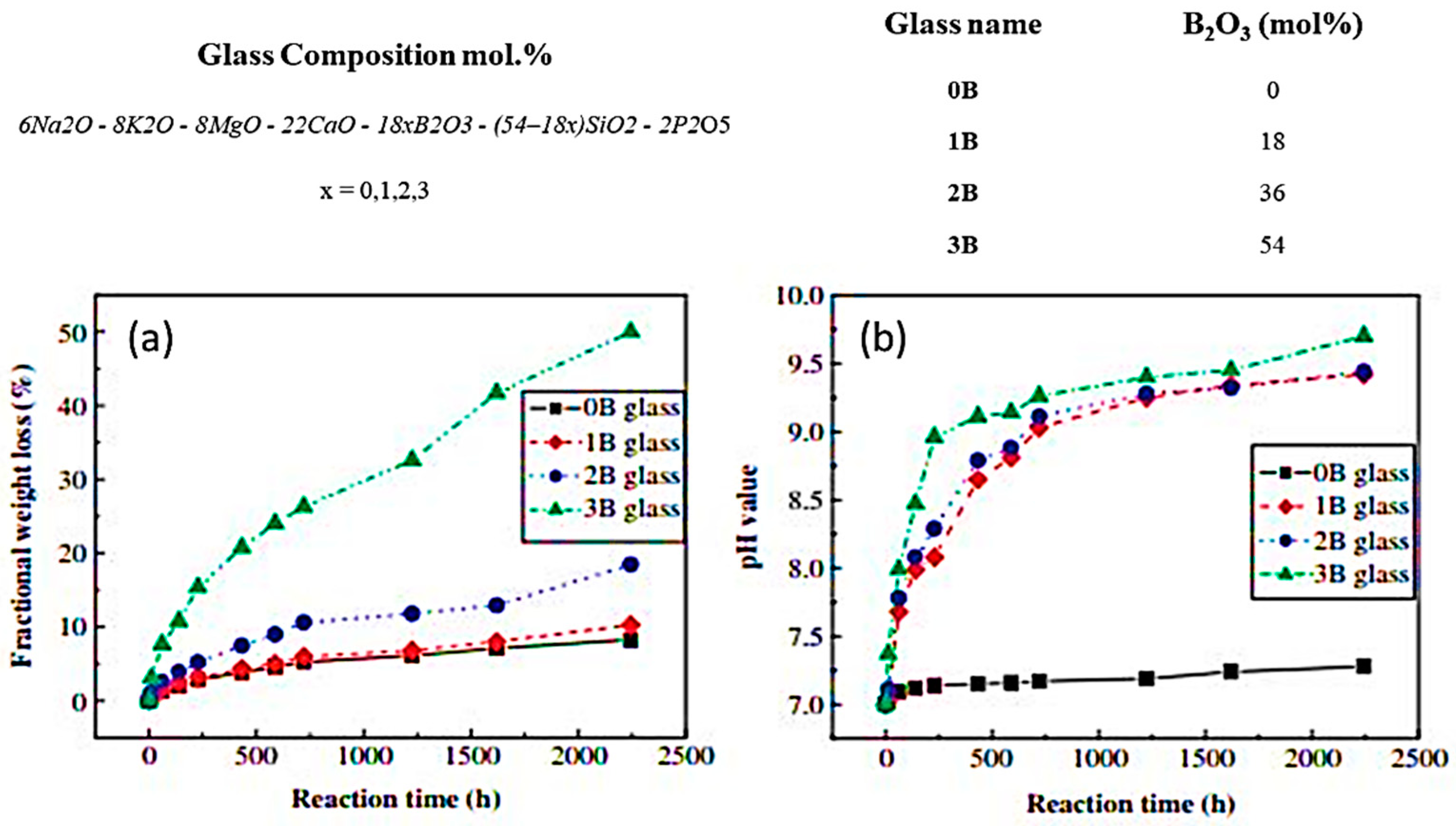
- Yao, A.; Wang, D.; Huang, W.; Day, D.E. In vitro bioactive characteristics of borate-based glasses with controllable degradation behaviour. J. Am. Ceram. Soc. 2007, 90, 303–306. [Google Scholar] [CrossRef]
- Wright, A.C.; Dalba, G.; Rocca, F.; Vedishcheva, N.M. Borate versus silicate glasses: Why are they so different? Phys. Chem. Glasses Eur. J. Glass Sci. Technol. B 2010, 51, 233–265. [Google Scholar]
- Huang, W.; Day, D.E.; Kittiratanapiboon, K.; Rahaman, M.N. Kinetics and mechanism of the conversion of silicate (45S5), borate and borosilicate glasses to hydroxyapatite in diluite phosphate solutions. J. Mater. Sci. Mater. Med. 2006, 17, 583–596. [Google Scholar] [CrossRef] [PubMed]
- Bi, L.; Rahaman, M.N.; Day, D.E.; Brown, Z.; Samujh, C.; Liu, X.; Mohammadkhah, A.; Dusevich, V.; Eick, J.D.; Bonewald, L.F. Effect of bioactive borate glass microstructure on bone regeneration, angiogenesis and hydroxyapatite conversion in a rat calvarial defect model. Acta Biomater. 2013, 9, 8015–8026. [Google Scholar] [CrossRef] [PubMed]
- Gu, Y.; Wang, G.; Zhang, X.; Zhang, Y.; Zhang, C.; Liu, X.; Rahaman, M.N.; Huang, W.; Pan, H. Biodegradable borosilicate bioactive glass scaffolds with a trabecular microstructurefor bone repair. Mater. Sci. Eng. C 2014, 36, 294–300. [Google Scholar] [CrossRef] [PubMed]
- Wu, C.; Miron, R.; Sculean, A.; Kaskel, S.; Doert, T.; Schulze, R.; Zhang, Y. Proliferation, differentiation and gene expression of osteoblasts in boron-containing associated with dexamethasone deliver from mesoporous bioactive glass scaffolds. Biomaterials 2011, 32, 7068–7078. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.; Jia, W.; Gu, Y.; Xiao, W.; Liu, X.; Wang, D.; Zhang, C.; Huang, W.; Rahaman, M.N.; Day, D.E.; et al. Teicoplanin-loaded borate bioactive glass implants for treating chronic bone infection in a rabbit tibia osteomyelitis model. Biomaterials 2010, 31, 5865–5874. [Google Scholar] [CrossRef] [PubMed]
- Gu, Y.; Huang, W.; Rahaman, M.N.; Day, D.E. Bone regeneration in rat calvarian defects implanted with fibrous scaffolds composed of a mixture of silicate and borate bioactive glasses. Acta Biomater. 2013, 9, 9126–9136. [Google Scholar] [CrossRef] [PubMed]
- Xie, Z.; Liu, X.; Jia, W.; Zhang, C.; Huang, W.; Wang, J. Treatment of osteomyelitis and repair of bone defect by degradable bioactive borate glass releasing vancomycin. J. Control. Release 2009, 139, 118–126. [Google Scholar] [CrossRef] [PubMed]
- Jia, W.T.; Zhang, X.; Luo, S.H.; Liu, X.; Huang, W.H.; Rahaman, M.N.; Day, D.E.; Zhang, C.Q.; Xie, Z.P.; Wang, J.Q. Novel borate glass/chitosan composite as a delivery vehicle for teicoplanin in the treatment of chronic osteomyelitis. Acta Biomater. 2010, 6, 812–819. [Google Scholar] [CrossRef] [PubMed]
- Chen, S.; Yang, Q.; Brow, R.K.; Liu, K.; Brow, K.A.; Ma, Y.; Shi, H. In vitro stimulation of vascular endothelial growth factor by borate-based glass fibers under dynamic flow conditions. Mater. Sci. Eng. C 2017, 73, 447–455. [Google Scholar] [CrossRef] [PubMed]
- Zhou, J.; Wang, H.; Zhao, S.; Zhou, N.; Li, L.; Huang, W.; Wang, D.; Zhang, C. In vivo and in vitro studies of borate-basedglass micro-fibers for dermal repairing. Mater. Sci. Eng. C 2016, 60, 437–445. [Google Scholar] [CrossRef] [PubMed]
- Wray, P. Cotton candy’ that heals. Am. Ceram. Soc. Bull. 2011, 90, 24–31. [Google Scholar]
- Jung, D.; Day, T.; Stoecker, W.; Taylor, P. Treatment of non-healing diabetic venous stasis ulcers with bioactive glass nanofibers. Wound Repair Regen. 2011, 19, A30. [Google Scholar]
- Knowles, J.C. Phosphate based glasses for biomedical applications. J. Mater. Chem. 2003, 13, 2395–2401. [Google Scholar] [CrossRef]




| Composition (wt %) | 45S5 Bioglass (NovaBone) | S53P4 (AbminDent 1) | A–W Glass-Ceramic (Cerabone) |
|---|---|---|---|
| Na2O | 24.5 | 23 | 0 |
| CaO | 24.5 | 20 | 44.7 |
| CaF2 | 0 | 0 | 0.5 |
| MgO | 0 | 0 | 4.6 |
| P2O5 | 6 | 4 | 16.2 |
| SiO2 | 45 | 53 | 34 |
| Phases | Glass | Glass | Apatite Beta-Wollastonite Glass |
| Class of Bioactivity | A | B | B |
| Class A Bioactivity (wt %) | Class B Bioactivity (wt %) | |
|---|---|---|
| SiO2 | 42–50 | 52–58 |
| Na2O | 14–28 | 3–20 |
| CaO | 12–26 | 8–20 |
| P2O5 | 3–9 | 3–12 |
| Al2O3 | 0–1 | 0–3 |
| MgO | 0–3 | 0–12 |
| K2O | 0–6 | 0–12 |
| CaF2 | 0–12 | 0–18 |
| Stage | Reaction Event |
|---|---|
| 11 | Crystallization of matrix |
| 10 | Cellular attachment |
| 9 | Differentiation of steam cells |
| 8 | Attachment of steam cells |
| 7 | Action of macrophages |
| 6 | Adsorption of biological moieties |
| 5 | Nucleation and crystallization of calcium phosphate to HCA |
| 4 | Precipitation of amorphous calcium phosphate |
| 3–2 | Dissolution and re-polymerization of surface silica |
| 1 | Ion exchange |
| 0 | Initial glass surface |
| Material Property | Trabecular Bone | Cortical Bone | 45S5 Bioglass |
|---|---|---|---|
| Compressive strength (MPa) | 0.1–16 | 130–200 | 500 |
| Tensile strength (MPa) | n.a. | 50–151 | 42 |
| Compressive elastic modulus (GPa) | 0.12–1.1 | 11.5–17 | n.a. |
| Young’s modulus (GPa) | 0.05–0.5 | 7–30 | 35 |
| Fracture toughness (MPa·m1/2) | n.a. | 2–12 | 0.7–1.1 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Fiume, E.; Barberi, J.; Verné, E.; Baino, F. Bioactive Glasses: From Parent 45S5 Composition to Scaffold-Assisted Tissue-Healing Therapies. J. Funct. Biomater. 2018, 9, 24. https://doi.org/10.3390/jfb9010024
Fiume E, Barberi J, Verné E, Baino F. Bioactive Glasses: From Parent 45S5 Composition to Scaffold-Assisted Tissue-Healing Therapies. Journal of Functional Biomaterials. 2018; 9(1):24. https://doi.org/10.3390/jfb9010024
Chicago/Turabian StyleFiume, Elisa, Jacopo Barberi, Enrica Verné, and Francesco Baino. 2018. "Bioactive Glasses: From Parent 45S5 Composition to Scaffold-Assisted Tissue-Healing Therapies" Journal of Functional Biomaterials 9, no. 1: 24. https://doi.org/10.3390/jfb9010024
APA StyleFiume, E., Barberi, J., Verné, E., & Baino, F. (2018). Bioactive Glasses: From Parent 45S5 Composition to Scaffold-Assisted Tissue-Healing Therapies. Journal of Functional Biomaterials, 9(1), 24. https://doi.org/10.3390/jfb9010024







