Aloe Vera for Tissue Engineering Applications
Abstract
1. Introduction
2. Natural Polymer-Derived Biomaterials
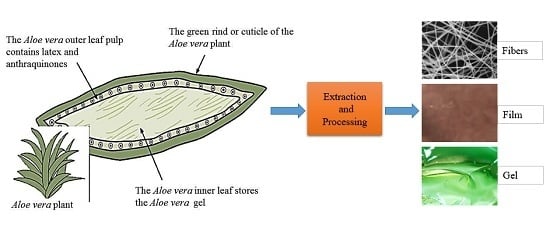
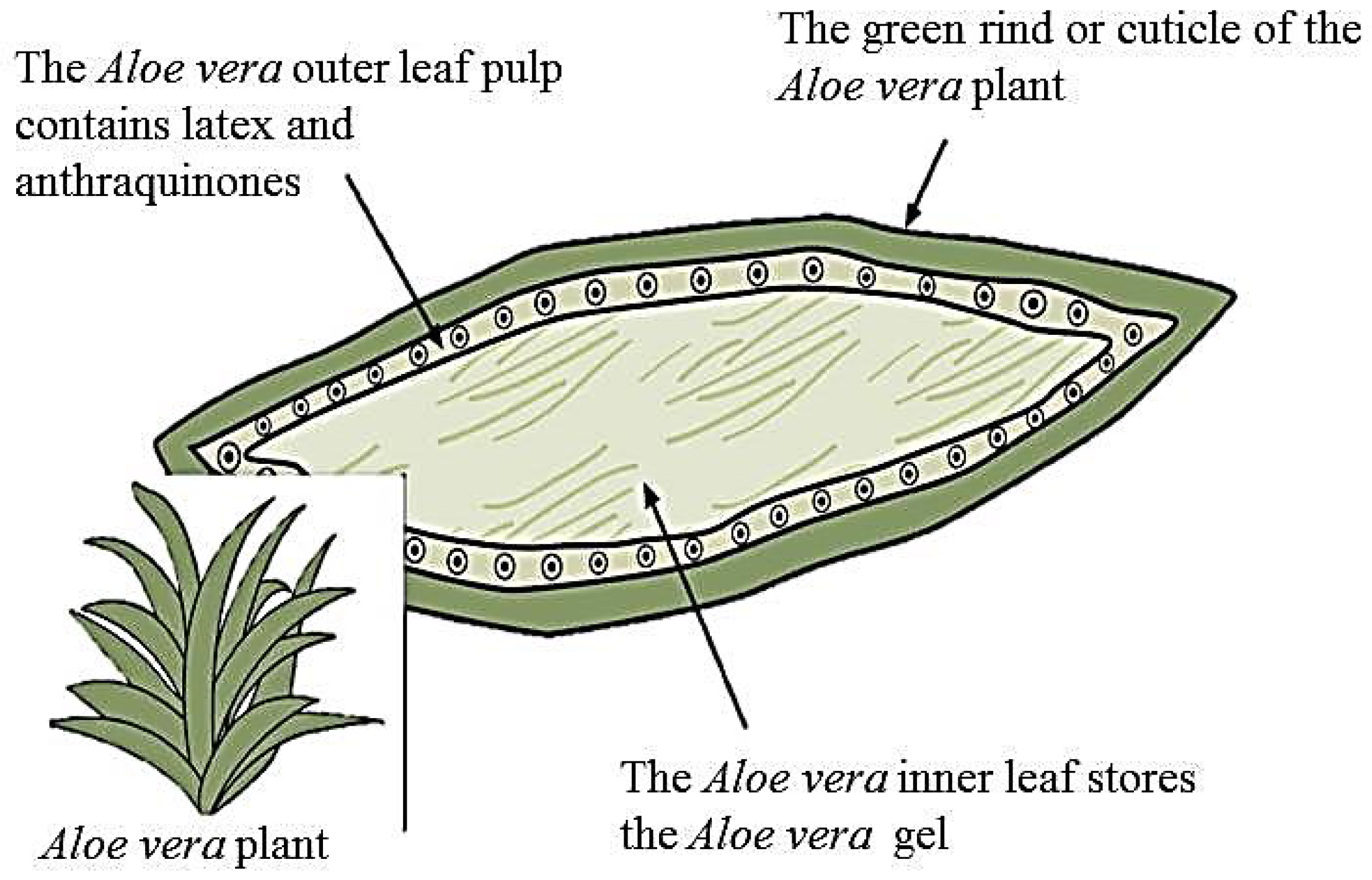
3. Aloe Vera Plant and Its Bioactive Components
4. Extraction and Processing of Aloe Vera Gel and Powder
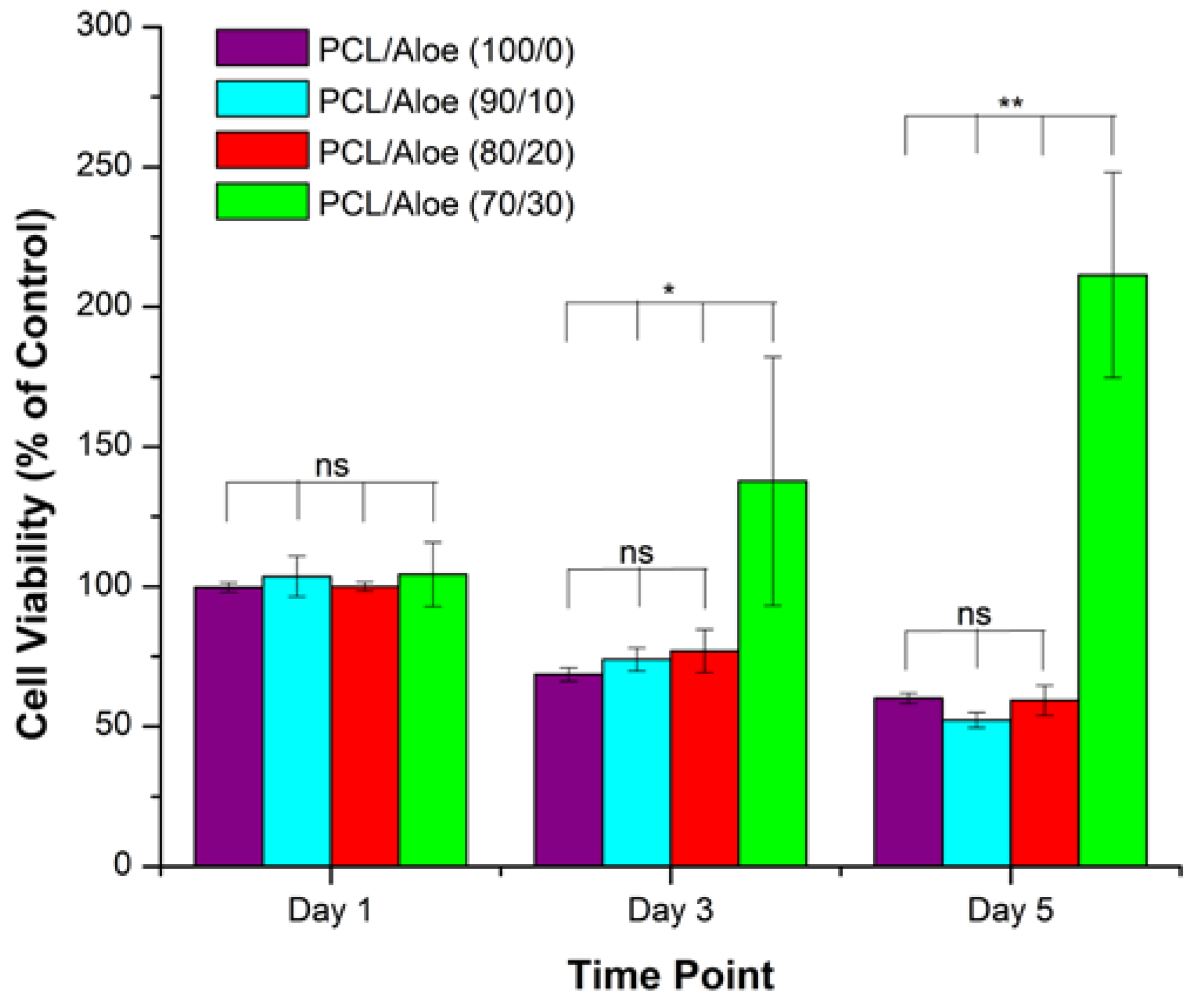
5. Tissue Engineering Prospects of Aloe Vera
6. Aloe Vera Based Tissue Engineering Scaffolds
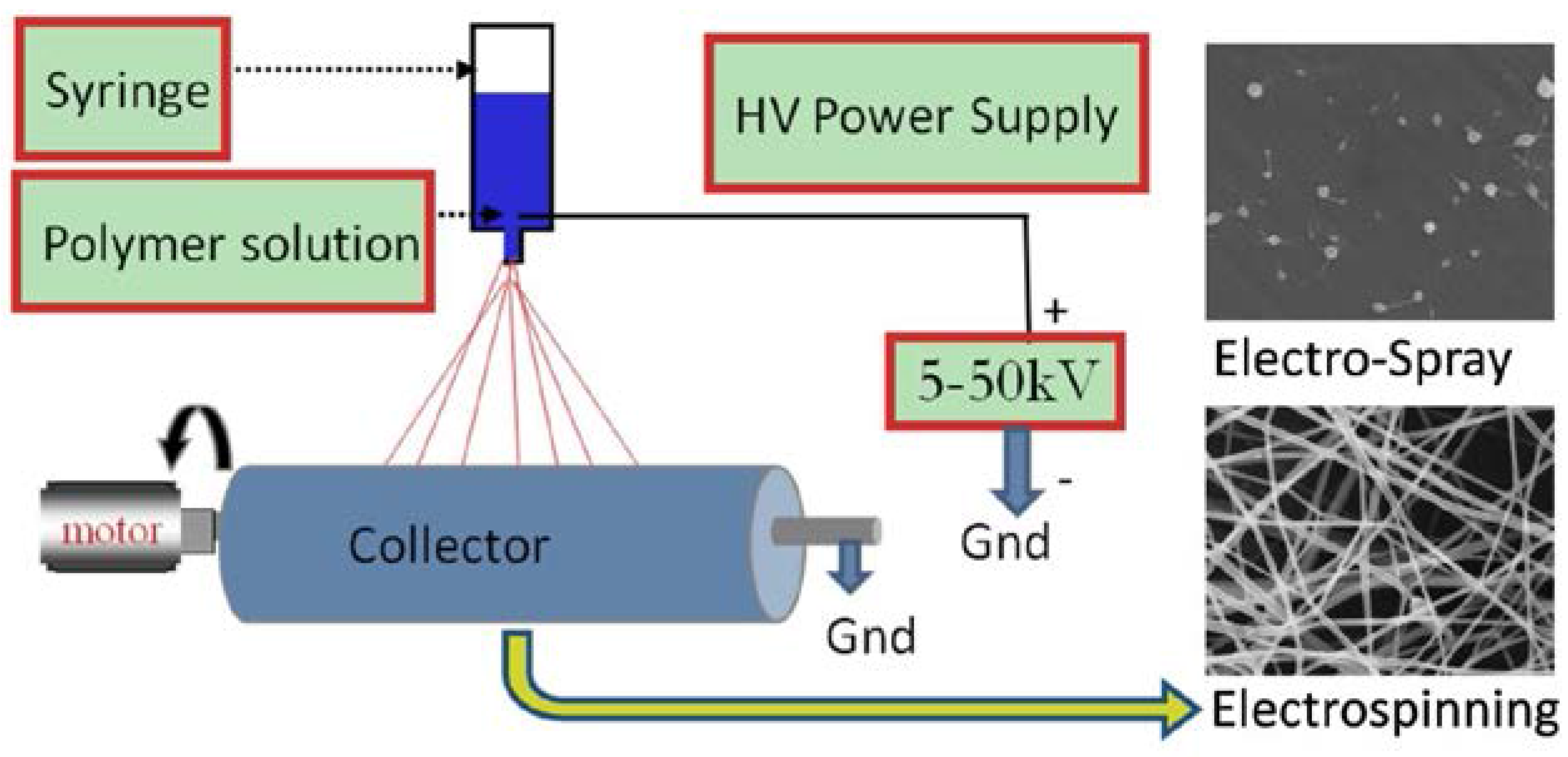
6.1. Electrospinning Technique
6.2. Freeze Drying Technique
6.3. Molecular Self-Assembly and Phase Separation Techniques
7. Aloe Vera Based Gels and Films
8. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Surjushe, A.; Vasani, R.; Saple, D.G. Aloe vera: A short review. Indian J. Dermatol. 2008, 53, 163–166. [Google Scholar] [CrossRef] [PubMed]
- Ahlawat, K.S.; Khatkar, B.S. Processing, food applications and safety of aloe vera products: A review. J. Food Sci. Technol. 2011, 48, 525–533. [Google Scholar] [CrossRef] [PubMed]
- Habeeb, F.; Shakir, E.; Bradbury, F.; Cameron, P. Screening methods used to determine the anti-microbial properties of aloe vera inner gel. Methods 2007, 42, 315–320. [Google Scholar] [CrossRef] [PubMed]
- Reynolds, T.; Dweck, A.C. Aloe vera leaf gel: A review update. J. Ethnopharmacol. 1999, 68, 3–37. [Google Scholar] [CrossRef]
- De Rodrı́guez, D.J.; Hernández-Castillo, D.; Garcı́a, R.R.; Angulo-Sánchez, J.L. Antifungal activity in vitro of aloe vera pulp and liquid fraction against plant pathogenic fungi. Ind. Crops Prod. 2005, 21, 81–87. [Google Scholar] [CrossRef]
- Athiban, P.; Borthakur, B.; Ganesan, S.; Swathika, B. Evaluation of antimicrobial efficacy of aloe vera and its effectiveness in decontaminating gutta percha cones. J. Conserv. Dent. 2012, 15, 246–248. [Google Scholar] [CrossRef] [PubMed]
- Banu, A. Efficacy of fresh aloe vera gel against multi drug resistant bacteria in infected leg ulcers. Australas. Med. J. 2012, 5, 305–309. [Google Scholar] [CrossRef] [PubMed]
- Langmead, L.; Makins, R.J.; Rampton, D.S. Anti-inflammatory effects of aloe vera gel in human colorectal mucosa in vitro. Aliment. Pharmacol. Ther. 2004, 19, 521–527. [Google Scholar] [CrossRef] [PubMed]
- Hu, Y.; Xu, J.; Hu, Q. Evaluation of Antioxidant Potential of aloe vera (Aloe barbadensis Miller) Extracts. J. Agric. Food Chem. 2003, 51, 7788–7791. [Google Scholar] [CrossRef] [PubMed]
- Davis, R.H.; Donato, J.J.; Hartman, G.M.; Haas, R.C. Anti-inflammatory and wound healing activity of a growth substance in aloe vera. J. Am. Podiatr. Med. Assoc. 1994, 84, 77–81. [Google Scholar] [CrossRef] [PubMed]
- Jithendra, P.; Rajam, A.M.; Kalaivani, T.; Mandal, A.B.; Rose, C. Preparation and characterization of aloe vera blended collagen-chitosan composite scaffold for tissue engineering applications. ACS Appl. Mater. Interfaces 2013, 5, 7291–7298. [Google Scholar] [CrossRef] [PubMed]
- Mary, S.A.; Dev, V.R.G. Electrospun herbal nanofibrous wound dressings for skin tissue engineering. J. Text. Inst. 2014, 106, 886–895. [Google Scholar] [CrossRef]
- Chithra, P.; Sajithlal, G.B.; Chandrakasan, G. Influence of aloe vera on collagen characteristics in healing dermal wounds in rats. Mol. Cell. Biochem. 1998, 181, 71–76. [Google Scholar] [CrossRef] [PubMed]
- Gupta, V.; Malhotra, S. Pharmacological attribute of aloe vera: Revalidation through experimental and clinical studies. Ayu 2012, 33, 193–196. [Google Scholar] [CrossRef] [PubMed]
- Suganya, S.; Venugopal, J.; Mary, S.A.; Ramakrishna, S.; Lakshmi, B.S.; Dev, V.R.G. Aloe vera incorporated biomimetic nanofibrous scaffold: A regenerative approach for skin tissue engineering. Iran. Polym. J. 2014, 23, 237–248. [Google Scholar] [CrossRef]
- Thompson, J.E. Topical use of aloe vera derived allantoin gel in otolaryngology. Ear Nose Throat J. 1991, 70, 56. [Google Scholar] [PubMed]
- Davis, R.H.; Stewart, G.J. Aloe vera and the inflamed synovial pouch model. J. Am. Podiatr. Med. Assoc. 1992, 82, 140–148. [Google Scholar] [CrossRef] [PubMed]
- Zhu, J.; Marchant, R.E. Design properties of hydrogel tissue-engineering scaffolds. Expert Rev. Med. Devices 2011, 8, 607–626. [Google Scholar] [CrossRef] [PubMed]
- Sell, S.A.; Wolfe, P.S.; Garg, K.; McCool, J.M.; Rodriguez, I.A.; Bowlin, G.L. The use of natural polymers in tissue engineering: A focus on electrospun extracellular matrix analogues. Polymers 2010, 2, 522–553. [Google Scholar] [CrossRef]
- Le Bao Ha, T.; Minh, T.; Nguyen, D.; Minh, D. Naturally Derived Biomaterials: Preparation and Application; Andrades, J.A., Ed.; InTech: Rijeka, Croatia, 2013. [Google Scholar]
- Shakya, A.K.; Kumar, A.; Nandakumar, K.S. Chemical cross-linking abrogates adjuvant potential of natural polymers. RSC Adv. 2014, 4, 13817–13821. [Google Scholar] [CrossRef]
- Ige, O.O.; Umoru, L.E.; Aribo, S. Natural Products: A Minefield of Biomaterials. ISRN Mater. Sci. 2012, 2012, 1–20. [Google Scholar] [CrossRef]
- Sionkowska, A. Current research on the blends of natural and synthetic polymers as new biomaterials: Review. Prog. Polym. Sci. 2011, 36, 1254–1276. [Google Scholar] [CrossRef]
- Vroman, I.; Tighzert, L. Biodegradable polymers. Materials 2009, 2, 307–344. [Google Scholar] [CrossRef]
- Ravi, S.; Chaikof, E.L. Biomaterials for vascular tissue engineering. Regen. Med. 2010, 5, 107–120. [Google Scholar] [CrossRef] [PubMed]
- Giusti, P.; Lazzeri, L.; Barbani, N.; Lelli, L.; De Petris, S.; Cascone, M.G. Blends of natural and synthetic polymers: A new route to novel biomaterials. Macromol. Symp. 1994, 78, 285–297. [Google Scholar] [CrossRef]
- Cascone, M.G.; Sim, B.; Sandra, D. Blends of synthetic and natural polymers as drug delivery systems for growth hormone. Biomaterials 1995, 16, 569–574. [Google Scholar] [CrossRef]
- Li, M.; Mondrinos, M.J.; Chen, X.; Lelkes, P.I. Electrospun blends of natural and synthetic polymers as scaffolds for tissue engineering. Conf. Proc. IEEE Eng. Med. Biol. Soc. 2005, 6, 5858–5861. [Google Scholar] [PubMed]
- Ho, M.H.; Hsieh, C.C.; Hsiao, S.W.; van Hong Thien, D. Fabrication of asymmetric chitosan GTR membranes for the treatment of periodontal disease. Carbohydr. Polym. 2010, 79, 955–963. [Google Scholar] [CrossRef]
- Haniadka, R.; Kamble, P.S.; Azmidha, A.; Mane, P.P.; Geevarughese, N.M.; Palatty, P.L.; Baliga, M.S. Aloe vera in dermatology: A brief review. Bioact. Diet. Fact. Plant Extr. Dermatol. 2009, 144, 125. [Google Scholar]
- Baruah, A.; Bordoloi, M.; Baruah, H.P. Aloe vera: A multipurpose industrial crop. Indust. Crops Prod. 2016, 94, 951–963. [Google Scholar] [CrossRef]
- Klein, A.D.; Penneys, N.S. Aloe vera. J. Am. Acad. Dermatol. 1988, 18, 714–720. [Google Scholar] [CrossRef]
- Radha, M.H.; Laxmipriya, N.P. Evaluation of biological properties and clinical effectiveness of aloe vera: A systematic review. J. Tradit. Complement. Med. 2015, 5, 21–26. [Google Scholar] [CrossRef] [PubMed]
- Hamman, J.H. Composition and applications of aloe vera leaf gel. Molecules 2008, 13, 1599–1616. [Google Scholar] [CrossRef] [PubMed]
- Chokboribal, J.; Tachaboonyakiat, W.; Sangvanich, P.; Ruangpornvisuti, V.; Jettanacheawchankit, S.; Thunyakitpisal, P. Deacetylation affects the physical properties and bioactivity of acemannan, an extracted polysaccharide from aloe vera. Carbohydr. Polym. 2015, 133, 556–566. [Google Scholar] [CrossRef] [PubMed]
- Bozzi, A.; Perrin, C.; Austin, S.; Vera, F.A. Quality and authenticity of commercial aloe vera gel powders. Food Chem. 2007, 103, 22–30. [Google Scholar] [CrossRef]
- Eamlamnam, K.; Patumraj, S.; Visedopas, N. Effects of Aloe vera and sucralfate on gastric microcirculatory changes, cytokine levels and gastric ulcer healing in rats. World J. Gastroenterol. 2006, 12, 2034–2039. [Google Scholar] [CrossRef] [PubMed]
- Werawatganon, D. Aloe vera attenuated gastric injury on indomethacin-induced gastropathy in rats. World J. Gastroenterol. 2014, 20, 18330–18337. [Google Scholar] [CrossRef] [PubMed]
- Cook, N. Flavonoids—Chemistry, metabolism, cardioprotective effects, and dietary sources. J. Nutr. Biochem. 1996, 7, 66. [Google Scholar] [CrossRef]
- Nindo, C.I.; Powers, J.R.; Tang, J. Thermal properties of Aloe vera powder and rheology of reconstituted gels. Trans. ASABE 2011, 53, 1193–1200. [Google Scholar] [CrossRef]
- Ramachandra, C.T.; Rao, P.S. Processing of Aloe vera leaf gel: A review. Am. J. Agric. Biol. Sci. 2008, 3, 502–510. [Google Scholar] [CrossRef]
- He, Q.; Changhong, L.; Kojo, E.; Tian, Z. Quality and safety assurance in the processing of Aloe vera gel juice. Food Control 2005, 16, 95–104. [Google Scholar] [CrossRef]
- Chandegara, V.K.; Varshney, A.K. Aloe vera L. processing and products: A review. Int. J. Med. Aromat. Plants 2013, 3, 492–506. [Google Scholar]
- Sehgal, I.; Winters, W.D.; Scott, M.; David, A.; Gillis, G.; Stoufflet, T.; Nair, A.; Kousoulas, K. Toxicologic assessment of a commercial decolorized whole leaf aloe vera juice, lily of the desert filtered whole leaf juice with aloesorb. J. Toxicol. 2013, 2013, 1–12. [Google Scholar] [CrossRef] [PubMed]
- David, R. Aloe vera: A Scientific Approach; Vantage Press Inc.: New York, UK, USA, 1997; pp. 290–306. [Google Scholar]
- Coats, B.C. Method of Processing Stabilized Aloe Vera gel Obtained from the Whole Aloe Vera Leaf. U.S. Patent 5,356,811, 18 October 1994. [Google Scholar]
- Eshun, K.; He, Q. Aloe vera: A valuable ingredient for the food, pharmaceutical and cosmetic industries—A review. Crit. Rev. Food Sci. Nutr. 2004, 44, 91–96. [Google Scholar] [CrossRef] [PubMed]
- Chandegara, V.K.; Varshney, A.K. Effect of centrifuge speed on gel extraction from aloe vera leaves. J. Food Process. Technol. 2014, 5, 1–6. [Google Scholar]
- Rodriguez, E.R.; Martin, J.D. Aloe vera as a functional ingredient in foods. Crit. Rev. Food Sci. Nutr. 2010, 50, 305–326. [Google Scholar] [CrossRef] [PubMed]
- Minjares-Fuentes, R.; Femenia, A.; Comas-Serra, C.; Rodriguez-Gonzalez, V.M.; Gonzalez-Laredo, R.F.; Gallegos-Infante, J.A.; Medina-Torres, L. Effect of different drying procedures on physicochemical properties and flow behavior of aloe vera (Aloe barbadensis Miller) gel. LWT Food Sci. Technol. 2016, 74, 378–386. [Google Scholar] [CrossRef]
- Medina-Torres, L.; Calderas, F.; Minjares, R.; Femenia, A.; Sánchez-Olivares, G.; Gonzalez-Laredo, F.R.; Santiago-Adame, R.; Ramirez-Nunez, D.M.; Rodríguez-Ramírez, J.; Manero, O. Structure preservation of aloe vera (barbadensis Miller) mucilage in a spray drying process. LWT Food Sci. Technol. 2016, 66, 93–100. [Google Scholar] [CrossRef]
- Cervantes, C.V. Study of spray drying of the aloe vera mucilage (Aloe vera barbadensis Miller) as a function of its rheological properties. LWT Food Sci. Technol. 2014, 55, 426–435. [Google Scholar] [CrossRef]
- Avila, H.; Rivero, J.; Herrera, F.; Fraile, G. Cytotoxicity of a low molecular weight fraction from aloe vera (Aloe barbadensis Miller) gel. Toxicon 1997, 35, 1423–1430. [Google Scholar] [CrossRef]
- Grindlay, D.; Reynolds, T. The aloe vera phenomenon: A review of the properties and modern uses of the leaf parenchyma gel. J. Ethnopharmacol. 1986, 16, 117–151. [Google Scholar] [CrossRef]
- Gentilini, R.; Bozzini, S.; Munarin, F.; Petrini, P.; Visai, L.; Tanzi, M.C. Pectins from aloe Vera: Extraction and production of gels for regenerative medicine. J. Appl. Polym. Sci. 2013, 131, 1–9. [Google Scholar]
- Salehi, M.; Farzamfar, S.; Bastami, F.; Tajerian, R. Fabrication and characterization of electrospun PLLA/collagen nanofibrous scaffold coated with chitosan to sustain release of aloe vera gel for skin tissue engineering. Biomed. Eng. Appl. Basis Commun. 2016, 28, 1650035–1650042. [Google Scholar] [CrossRef]
- Silva, S.S. An investigation of the potential application of chitosan/aloe-based membranes for regenerative medicine. Acta Biomater. 2013, 9, 6790–6797. [Google Scholar] [CrossRef] [PubMed]
- Choi, S.; Chung, M.H. A review on the relationship between aloe vera components and their biologic effects. Semin. Integr. Med. 2003, 1, 53–62. [Google Scholar] [CrossRef]
- Jaganathan, S.; Balaji, A.; Supriyanto, E.; Muhamad, I.I.; Khudzari, A.Z.M. Microwave-assisted fibrous decoration of mPE surface utilizing Aloe vera extract for tissue engineering applications. Int. J. Nanomed. 2015, 10, 5909–5923. [Google Scholar] [CrossRef] [PubMed]
- Silva, S.S.; Caridade, S.G.; Mano, J.F.; Reis, R.L. Effect of crosslinking in chitosan/aloe vera-based membranes for biomedical applications. Carbohydr. Polym. 2013, 98, 581–588. [Google Scholar] [CrossRef] [PubMed]
- Davis, R.H.; DiDonato, J.J.; Johnson, R.W.; Stewart, C.B. Aloe vera, hydrocortisone, and sterol influence on wound tensile strength and anti-inflammation. J. Am. Podiatr. Med. Assoc. 1994, 84, 614–621. [Google Scholar] [CrossRef] [PubMed]
- Chithra, P.; Sajithlal, G.B.; Chandrakasan, G. Influence of aloe vera on the glycosaminoglycans in the matrix of healing dermal wounds in rats. J. Ethnopharmacol. 1998, 59, 179–186. [Google Scholar] [CrossRef]
- Tarameshloo, M.; Norouzian, M. Aloe vera gel and thyroid hormone cream may improve wound healing in Wistar rats. Anat. Cell Biol. 2012, 45, 170–177. [Google Scholar] [CrossRef] [PubMed]
- Foster, M.; Hunter, D.; Samman, S. Evaluation of the nutritional and metabolic effects of aloe vera. In Biomolecular and Clinical Aspects, 2nd ed.; CRC Press/Taylor & Francis: Boca Raton, FL, USA, 2011; Volume 3, pp. 37–54. [Google Scholar]
- Jia, Y.; Zhao, G.; Jia, J. Preliminary evaluation: The effects of Aloe ferox Miller and Aloe arborescens Miller on wound healing. J. Ethnopharmacol. 2008, 120, 181–189. [Google Scholar] [CrossRef] [PubMed]
- Lee, M.J.; Lee, O.H.; Yoon, S.H.; Lee, S.K.; Chung, M.H. In vitro angiogenic activity of aloe vera gel on calf pulmonary artery endothelial (CPAE) cells. Arch. Pharm. Res. 1998, 21, 260–265. [Google Scholar] [CrossRef] [PubMed]
- Vijayalakshmi, D.; Dhandapani, R.; Jayaveni, S. In vitro anti-inflammatory activity of aloe vera by down regulation of MMP-9 in peripheral blood mononuclear cells. J. Ethnopharmacol. 2012, 141, 542–546. [Google Scholar] [CrossRef] [PubMed]
- Kessenbrock, K.; Plaks, V.; Werb, Z. Matrix metalloproteinases: Regulators of the tumor microenvironment. Cell 2010, 141, 52–67. [Google Scholar] [CrossRef] [PubMed]
- Loo, W.; Wang, M.; Jin, L.J.; Cheung, M.; Li, G.R. Association of matrix metalloproteinase (MMP-1, MMP-3 and MMP-9) and cyclooxygenase-2 gene polymorphisms and their proteins with chronic periodontitis. Arch. Oral Biol. 2011, 56, 1081–1090. [Google Scholar] [CrossRef] [PubMed]
- Boonyagul, S.; Banlunara, W.; Sangvanich, P.; Thunyakitpisal, P. Effect of acemannan, an extracted polysaccharide from aloe vera, on BMSCs proliferation, differentiation, extracellular matrix synthesis, mineralization, and bone formation in a tooth extraction model. Odontology 2013, 102, 310–317. [Google Scholar] [CrossRef] [PubMed]
- Jettanacheawchankit, S.; Sasithanasate, S.; Sangvanich, P.; Banlunara, W.; Thunyakitpisal, P. Acemannan Stimulates Gingival Fibroblast Proliferation; Expressions of Keratinocyte Growth Factor-1, Vascular Endothelial Growth Factor, and Type I Collagen; and Wound Healing. J. Pharmacol. Sci. 2009, 109, 525–531. [Google Scholar] [CrossRef] [PubMed]
- Jittapiromsak, N.; Sahawat, D.; Banlunara, W.; Sangvanich, P.; Thunyakitpisal, P. Acemannan, an extracted product from aloe vera, stimulates dental pulp cell proliferation, differentiation, mineralization, and dentin formation. Tissue Eng. A 2010, 16, 1997–2006. [Google Scholar] [CrossRef] [PubMed]
- Fani, M.; Kohanteb, J. Inhibitory activity of Aloe vera gel on some clinically isolated cariogenic and periodontopathic bacteria. J. Oral Sci. 2012, 54, 15–21. [Google Scholar] [CrossRef] [PubMed]
- Bashir, A.; Saeed, B.; Mujahid, T.Y.; Jehan, N. Comparative study of antimicrobial activities of aloe vera extracts and antibiotics against isolates from skin infections. Afr. J. Biotechnol. 2013, 10, 3835–3840. [Google Scholar]
- Vasita, R.; Katti, D.S. Nanofibers and their applications in tissue engineering. Int. J. Nanomed. 2006, 1, 15–30. [Google Scholar] [CrossRef]
- Ma, Z.; Kotaki, M.; Inai, R.; Ramakrishna, S. Potential of nanofiber matrix as tissue-engineering scaffolds. Tissue Eng. 2005, 11, 101–109. [Google Scholar] [CrossRef] [PubMed]
- Holzwarth, J.M.; Ma, P.X. Biomimetic nanofibrous scaffolds for bone tissue engineering. Biomaterials 2011, 32, 9622–9629. [Google Scholar] [CrossRef] [PubMed]
- Cao, H.; Liu, T.; Chew, S.Y. The application of nanofibrous scaffolds in neural tissue engineering. Adv. Drug Deliv. Rev. 2009, 61, 1055–1064. [Google Scholar] [CrossRef] [PubMed]
- James, R.; Toti, U.S.; Laurencin, C.T. Electrospun nanofibrous scaffolds for engineering soft connective tissues. Biomed. Nanotechnol. 2011, 726, 243–258. [Google Scholar]
- Dhandayuthapani, B.; Yoshida, Y.; Maekawa, T.; Kumar, D.S. Polymeric Scaffolds in Tissue Engineering Application: A Review. Int. J. Polym. Sci. 2011, 2011, 1–19. [Google Scholar] [CrossRef]
- Zhang, X.; Reagan, M.R.; Kaplan, D.L. Electrospun silk biomaterial scaffolds for regenerative medicine. Adv. Drug Deliv. Rev. 2009, 61, 988–1006. [Google Scholar] [CrossRef] [PubMed]
- Reneker, D.H.; Chun, I. Nanometre diameter fibres of polymer, produced by electrospinning. Nanotechnology 1996, 7, 216–223. [Google Scholar] [CrossRef]
- Ko, F.K. Nanofiber Technology; Kluwer Academic Publishers: Dordrecht, Netherlands, 2006; pp. 553–565. [Google Scholar]
- Li, D.; Xia, Y. Electrospinning of nanofibers: Reinventing the wheel. Adv. Mater. 2004, 16, 1151–1170. [Google Scholar] [CrossRef]
- Beachley, V.; Wen, X. Polymer nanofibrous structures: Fabrication, biofunctionalization, and cell interactions. Prog. Polym. Sci. 2010, 35, 868–892. [Google Scholar] [CrossRef] [PubMed]
- Spivak, A.F.; Dzenis, Y.A.; Reneker, D.H. A model of steady state jet in the electrospinning process. Mech. Res. Commun. 2000, 27, 37–42. [Google Scholar] [CrossRef]
- Srinivasan, G.; Reneker, D.H. Structure and morphology of small diameter electrospun aramid fibers. Polym. Int. 1995, 36, 195–201. [Google Scholar] [CrossRef]
- Reneker, D.H.; Yarin, A.L.; Fong, H. Bending instability of electrically charged liquid jets of polymer solutions in electrospinning. J. Appl. Phys. 2000, 87, 4531–4547. [Google Scholar] [CrossRef]
- Carter, P.; Bhattarai, N. Bioscaffolds: Fabrication and Performance; Elsevier: Waltham, MA, USA, 2013; Volume 7, pp. 161–188. [Google Scholar]
- Bottino, M.C.; Thomas, V.; Schmidt, G.; Vohra, Y.K.; Chu, T.-M.G.; Kowolik, M.J.; Janowski, G.M. Recent advances in the development of GTR/GBR membranes for periodontal regeneration—A materials perspective. Dent. Mater. 2012, 28, 703–721. [Google Scholar] [CrossRef] [PubMed]
- Zhao, G.; Zhang, X.; Lu, T.J.; Xu, F. Recent advances in electrospun nanofibrous scaffolds for cardiac tissue engineering. Adv. Funct. Mater. 2015, 25, 5726–5738. [Google Scholar] [CrossRef]
- Kumbar, S.G.; James, R.; Nukavarapu, S.P.; Laurencin, C.T. Electrospun nanofiber scaffolds: Engineering soft tissues. Biomed. Mater. 2008, 3, 034002–034017. [Google Scholar] [CrossRef] [PubMed]
- Alamein, M.A.; Stephens, S.; Liu, Q.; Skabo, S.; Warnke, P.H. Mass production of nanofibrous extracellular matrix with controlled 3d morphology for large-scale soft tissue regeneration. Tissue Eng. C Methods 2013, 19, 458–472. [Google Scholar] [CrossRef] [PubMed]
- Li, W.J.; Tuan, R.S. Fabrication and application of nanofibrous scaffolds in tissue engineering. Curr. Protoc. Cell Biol. 2009, 25, 1–15. [Google Scholar]
- Liang, D.; Hsiao, B.S.; Chu, B. Functional electrospun nanofibrous scaffolds for biomedical applications. Adv. Drug Deliv. Rev. 2007, 59, 1392–1412. [Google Scholar] [CrossRef] [PubMed]
- Sant, S.; Hwang, C.M.; Lee, S.-H.; Khademhosseini, A. Hybrid PGS-PCL microfibrous scaffolds with improved mechanical and biological properties. J. Tissue Eng. Regen. Med. 2011, 5, 283–291. [Google Scholar] [CrossRef] [PubMed]
- Carter, P.; Rahman, S.; Bhattarai, N. Facile fabrication of aloe vera containing PCL nanofibers for barrier membrane application. J. Biomater. Sci. Polym. Ed. 2016, 27, 692–708. [Google Scholar] [CrossRef] [PubMed]
- Sridhar, R.; Ravanan, S.; Venugopal, J.R.; Sundarrajan, S.; Pliszka, D.; Sivasubramanian, S.; Gunasekaran, P.; Prabhakaran, M.; Madhaiyan, K.; Sahayaraj, A.; et al. Curcumin- and natural extract-loaded nanofibres for potential treatment of lung and breast cancer: In vitro efficacy evaluation. J. Biomater. Sci. Polym. Ed. 2014, 25, 985–998. [Google Scholar] [CrossRef] [PubMed]
- AbdullahShukry, N.A.; Sekak, K.A.; Ahmad, M.R.; Effendi, T.J.B. Characteristics of electrospun PVA-Aloe vera nanofibres produced via electrospinning. In Proceedings of the International Colloquium in Textile Engineering, Fashion, Apparel and Design 2014; Springer: Singapore, 2014; pp. 7–11. [Google Scholar]
- Ibrahim, I.; Sekak, K.A.; Hasbullah, N. Preparation and characterization of chitosan/aloe vera composite nanofibers generated by electrostatic spinning. AIP Conf. Proc. 2015, 1674, 1–7. [Google Scholar]
- García-Orue, I.; Gainza, G.; Gutierrez, F.B.; Aguirre, J.J.; Evora, C.; Pedraz, J.L.; Hernandez, R.M.; Delgado, A.; Igartua, M. Novel nanofibrous dressings containing rhEGF and Aloe vera for wound healing applications. Int. J. Pharm. 2016. [Google Scholar] [CrossRef] [PubMed]
- Bhaarathy, V. Biologically improved nanofibrous scaffolds for cardiac tissue engineering. Mater. Sci. Eng. C 2014, 44, 268–277. [Google Scholar] [CrossRef] [PubMed]
- Wu, X.; Liu, Y.; Li, X.; Wen, P.; Zhang, Y.; Long, Y.; Wang, X.; Guo, Y.; Xing, F.; Gao, J. Preparation of aligned porous gelatin scaffolds by unidirectional freeze-drying method. Acta Biomater. 2010, 6, 1167–1177. [Google Scholar] [CrossRef] [PubMed]
- Lv, Q.; Feng, Q. Preparation of 3-D regenerated fibroin scaffolds with freeze drying method and freeze drying/foaming technique. J. Mater. Sci. Mater. Med. 2006, 17, 1349–1356. [Google Scholar] [CrossRef] [PubMed]
- Haugh, M.G.; Murphy, C.M.; O’Brien, F.J. Novel freeze-drying methods to produce a range of collagen–glycosaminoglycan scaffolds with tailored mean pore sizes. Tissue Eng. C Methods 2010, 16, 887–894. [Google Scholar] [CrossRef] [PubMed]
- Wintermantel, E. Tissue engineering scaffolds using superstructures. Biomaterials 1996, 17, 83–91. [Google Scholar] [CrossRef]
- Hutmacher, D.W. Scaffolds in tissue engineering bone and cartilage. Biomaterials 2000, 21, 2529–2543. [Google Scholar] [CrossRef]
- Hollister, S.J. Porous scaffold design for tissue engineering. Nat. Mater. 2006, 4, 518–524. [Google Scholar] [CrossRef] [PubMed]
- Lu, T.; Li, Y.; Chen, T. Techniques for fabrication and construction of three-dimensional scaffolds for tissue engineering. Int. J. Nanomed. 2013, 8, 337–350. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Huang, Z.; Kim, Y.; He, Y.; Lee, M. Guest-driven inflation of self-assembled nanofibers through hollow channel formation. J. Am. Chem. Soc. 2014, 136, 16152–16155. [Google Scholar] [CrossRef] [PubMed]
- Tekin, E.D. Molecular dynamics simulations of self-assembled peptide amphiphile based cylindrical nanofibers. RSC Adv. 2015, 5, 66582–66590. [Google Scholar] [CrossRef]
- Jun, H.-W.; Paramonov, S.E.; Hartgerink, J.D. Biomimetic self-assembled nanofibers. Soft Matter 2006, 2, 177–181. [Google Scholar] [CrossRef]
- Qin, S.Y.; Jiang, H.F.; Peng, M.Y.; Lei, Q.; Zhuo, R.X.; Zhang, X.Z. Adjustable nanofibers self-assembled from an irregular conformational peptide amphiphile. Polym. Chem. 2015, 6, 519–524. [Google Scholar] [CrossRef]
- Zhang, C.; Xue, X.; Luo, Q.; Li, Y.; Yang, K.; Zhuang, X.; Jiang, Y.; Zhang, J.; Liu, J.; Zou, G.; et al. Self-assembled peptide nanofibers designed as biological enzymes for catalyzing ester hydrolysis. ACS Nano 2014, 8, 11715–11723. [Google Scholar] [CrossRef] [PubMed]
- Cinar, G.; Ceylan, H.; Urel, M.; Erkal, T.S.; Tekin, E.D.; Tekinay, A.B.; Dana, A.; Guler, M.O. Amyloid inspired self-assembled peptide nanofibers. Biomacromolecules 2012, 13, 3377–3387. [Google Scholar] [CrossRef] [PubMed]
- Hartgerink, J.D. Self-assembly and mineralization of peptide-amphiphile nanofibers. Science 2001, 294, 1684–1688. [Google Scholar] [CrossRef] [PubMed]
- Shah, R.N.; Shah, N.A.; Del Rosario Lim, M.M.; Hsieh, C.; Nuber, G.; Stupp, S.I. Supramolecular design of self-assembling nanofibers for cartilage regeneration. Proc. Natl. Acad. Sci. USA 2010, 107, 3293–3298. [Google Scholar] [CrossRef] [PubMed]
- Cooper, A.; Zhong, C.; Kinoshita, Y.; Morrison, R.S.; Rolandi, M.; Zhang, M. Self-assembled chitin nanofiber templates for artificial neural networks. J. Mater. Chem. 2012, 22, 3105–3109. [Google Scholar] [CrossRef]
- Zhang, P.; Cheetham, A.G.; Lin, Y.-A.; Cui, H. Self-assembled tat nanofibers as effective drug carrier and transporter. ACS Nano 2013, 7, 5965–5977. [Google Scholar] [CrossRef] [PubMed]
- Goktas, M.; Cinar, G.; Orujalipoor, I.; Ide, S.; Tekinay, A.B.; Guler, M.O. Self-assembled peptide amphiphile nanofibers and PEG composite hydrogels as tunable ECM mimetic microenvironment. Biomacromolecules 2015, 16, 1247–1258. [Google Scholar] [CrossRef] [PubMed]
- Rolandi, M.; Rolandi, R. Self-assembled chitin nanofibers and applications. Adv. Colloid Interface Sci. 2014, 207, 216–222. [Google Scholar] [CrossRef] [PubMed]
- Ali, J.; Khan, A.; Kotta, S.; Ansari, S.; Sharma, R.; Kumar, A. Formulation development, optimization and evaluation of aloe vera gel for wound healing. Pharmacogn. Mag. 2013, 9, 6–10. [Google Scholar] [CrossRef] [PubMed]
- Maenthaisong, R.; Chaiyakunapruk, N.; Niruntraporn, S.; Kongkaew, C. The efficacy of aloe vera used for burn wound healing: A systematic review. Burns 2007, 33, 713–718. [Google Scholar] [CrossRef] [PubMed]
- Somboonwong, J.; Thanamittramanee, S. Therapeutic effects of aloe vera on cutaneous microcirculation and wound healing in second degree burn model in rats. J. Med. Assoc. Thail. 2000, 83, 417–425. [Google Scholar]
- Korac, R.; Khambholja, K. Potential of herbs in skin protection from ultraviolet radiation. Pharmacogn. Rev. 2011, 5, 164–173. [Google Scholar] [CrossRef] [PubMed]
- Hashemi, S.A.; Madani, S.A.; Abediankenari, S. The review on properties of aloe vera in healing of cutaneous wounds. BioMed Res. Int. 2015, 2015, 1–6. [Google Scholar] [CrossRef] [PubMed]
- Boudreau, M.D.; Beland, F.A. An evaluation of the biological and toxicological properties of Aloe Barbadensis (Miller), aloe vera. J. Environ. Sci. Health C 2006, 24, 103–154. [Google Scholar] [CrossRef] [PubMed]
- Mateescu, A.; Wang, Y.; Dostalek, J.; Jonas, U. Thin hydrogel films for optical biosensor applications. Membranes 2012, 2, 40–69. [Google Scholar] [CrossRef] [PubMed]
- Brunsen, A.; Ritz, U.; Mateescu, A.; Höfer, I.; Frank, P.; Menges, B.; Hofmann, A.; Rommens, P.M.; Knoll, W.; Jonas, U. Photocrosslinkable dextran hydrogel films as substrates for osteoblast and endothelial cell growth. J. Mater. Chem. 2012, 22, 19590–19604. [Google Scholar] [CrossRef]
- Lim, H.L.; Hwang, Y.; Kar, M.; Varghese, S. Smart hydrogels as functional biomimetic systems. Biomater. Sci. 2014, 2, 603–618. [Google Scholar] [CrossRef]
- Pourjavadi, A.; Kurdtabar, M. Collagen-based highly porous hydrogel without any porogen: Synthesis and characteristics. Eur. Polym. J. 2007, 43, 877–889. [Google Scholar] [CrossRef]
- Khademhosseini, A.; Langer, R. Microengineered hydrogels for tissue engineering. Biomaterials 2007, 28, 5087–5092. [Google Scholar] [CrossRef] [PubMed]
- Lienemann, P.S.; Lutolf, M.P.; Ehrbar, M. Biomimetic hydrogels for controlled biomolecule delivery to augment bone regeneration. Adv. Drug Deliv. Rev. 2012, 64, 1078–1089. [Google Scholar] [CrossRef] [PubMed]
- Annabi, N.; Nichol, J.W.; Zhong, X.; Ji, C.; Koshy, S.; Khademhosseini, A.; Dehghani, F. Controlling the porosity and microarchitecture of hydrogels for tissue engineering. Tissue Eng. B Rev. 2010, 16, 371–383. [Google Scholar] [CrossRef] [PubMed]
- Pereira, R.; Carvalho, A.; Vaz, D.C.; Gil, M.H. Development of novel alginate based hydrogel films for wound healing applications. Int. J. Biol. Macromol. 2013, 52, 221–230. [Google Scholar] [CrossRef] [PubMed]
- Pereira, R.; Mendes, A.; Bártolo, P. Alginate/aloe vera hydrogel films for biomedical applications. Procedia CIRP 2013, 5, 210–215. [Google Scholar] [CrossRef]
- Rahman, S.M.; Mahoney, C.; Sankar, J.; Marra, K.G.; Bhattarai, N. Synthesis and characterization of magnesium gluconate contained poly (lactic-co-glycolic acid)/chitosan microspheres. Mater. Sci. Eng. B 2016, 203, 59–66. [Google Scholar] [CrossRef]
- Khoshgozaran, S. Mechanical, physicochemical and color properties of chitosan based-films as a function of aloe vera gel incorporation. Carbohydr. Polym. 2012, 87, 2058–2062. [Google Scholar] [CrossRef]
- Pereira, R.F.; Carvalho, A.; Gil, M.H.; Mendes, A. Influence of aloe vera on water absorption and enzymatic in vitro degradation of alginate hydrogel films. Carbohydr. Polym. 2013, 98, 311–320. [Google Scholar] [CrossRef] [PubMed]
- Anjum, S.; Gupta, A.; Sharma, D.; Gautam, D.; Bhan, S.; Sharma, A.; Kapil, A.; Gupta, B. Development of novel wound care systems based on nanosilver nanohydrogels of polymethacrylic acid with aloe vera and curcumin. Mater. Sci. Eng. C 2016, 64, 157–166. [Google Scholar] [CrossRef] [PubMed]
- Tummalapalli, M.; Berthet, M.; Verrier, B.; Deopura, B.L.; Alam, M.S.; Gupta, B. Composite wound dressings of pectin and gelatin with aloe vera and curcumin as bioactive agents. Int. J. Biol. Macromol. 2016, 82, 104–113. [Google Scholar] [CrossRef] [PubMed]

| Type | Compounds |
|---|---|
| Anthraquinones/anthrones | Aloe-emodin, aloetic-acid, anthranol, aloin A and B (collectively known as barbaloin), isobarbaloin, emodin, ester of cinnamic acid |
| Carbohydrates | Pure mannan, acetylated mannan, acetylated glucomannan, glucogalactomannan, galactan, pectic substance, arabinogalactan, galactoglucoarabinomannan, galactogalacturan, xylan, cellulose |
| Enzymes | Alkaline phosphatase, amylase, carboxypeptidase, carboxylase, catalase, cyclooxidase, phosphoenolpyruvate, cyclooxygenase, superoxide dismutase, lipase, oxidase |
| Inorganic compounds | Calcium, chlorine, phosphorous, chromium, copper, magnesium, iron, manganese, potassium, sodium, zinc |
| Non-essential and essential amino acids | Alanine, arginine, aspartic acid, glutamic acid, glycine, histidine, hydroxyproline, isoleucine, leucine, lysine, methionine, proline, threonine, tyrosine, valine, phenylalanine |
| Proteins | Lectins, lectin-like substance |
| Saccharides | Mannose, glucose, l-rhamnose, aldopentose |
| Vitamins | B1, B2, B6, C, β-carotene, choline, folic acid, α-tocopherol |
| Miscellaneous | Arachidonic acid, γ-linolenic acid, potassium sorbate, steroids (campestrol, cholesterol, β-sitosterol), triglicerides, triterpenoid, gibberillin, lignins, salicylic acid, uric acid |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rahman, S.; Carter, P.; Bhattarai, N. Aloe Vera for Tissue Engineering Applications. J. Funct. Biomater. 2017, 8, 6. https://doi.org/10.3390/jfb8010006
Rahman S, Carter P, Bhattarai N. Aloe Vera for Tissue Engineering Applications. Journal of Functional Biomaterials. 2017; 8(1):6. https://doi.org/10.3390/jfb8010006
Chicago/Turabian StyleRahman, Shekh, Princeton Carter, and Narayan Bhattarai. 2017. "Aloe Vera for Tissue Engineering Applications" Journal of Functional Biomaterials 8, no. 1: 6. https://doi.org/10.3390/jfb8010006
APA StyleRahman, S., Carter, P., & Bhattarai, N. (2017). Aloe Vera for Tissue Engineering Applications. Journal of Functional Biomaterials, 8(1), 6. https://doi.org/10.3390/jfb8010006