Complications Related to Metal-on-Metal Articulation in Trapeziometacarpal Joint Total Joint Arthroplasty
Abstract
:1. Introduction
2. Results
Case Presentation
Case 1

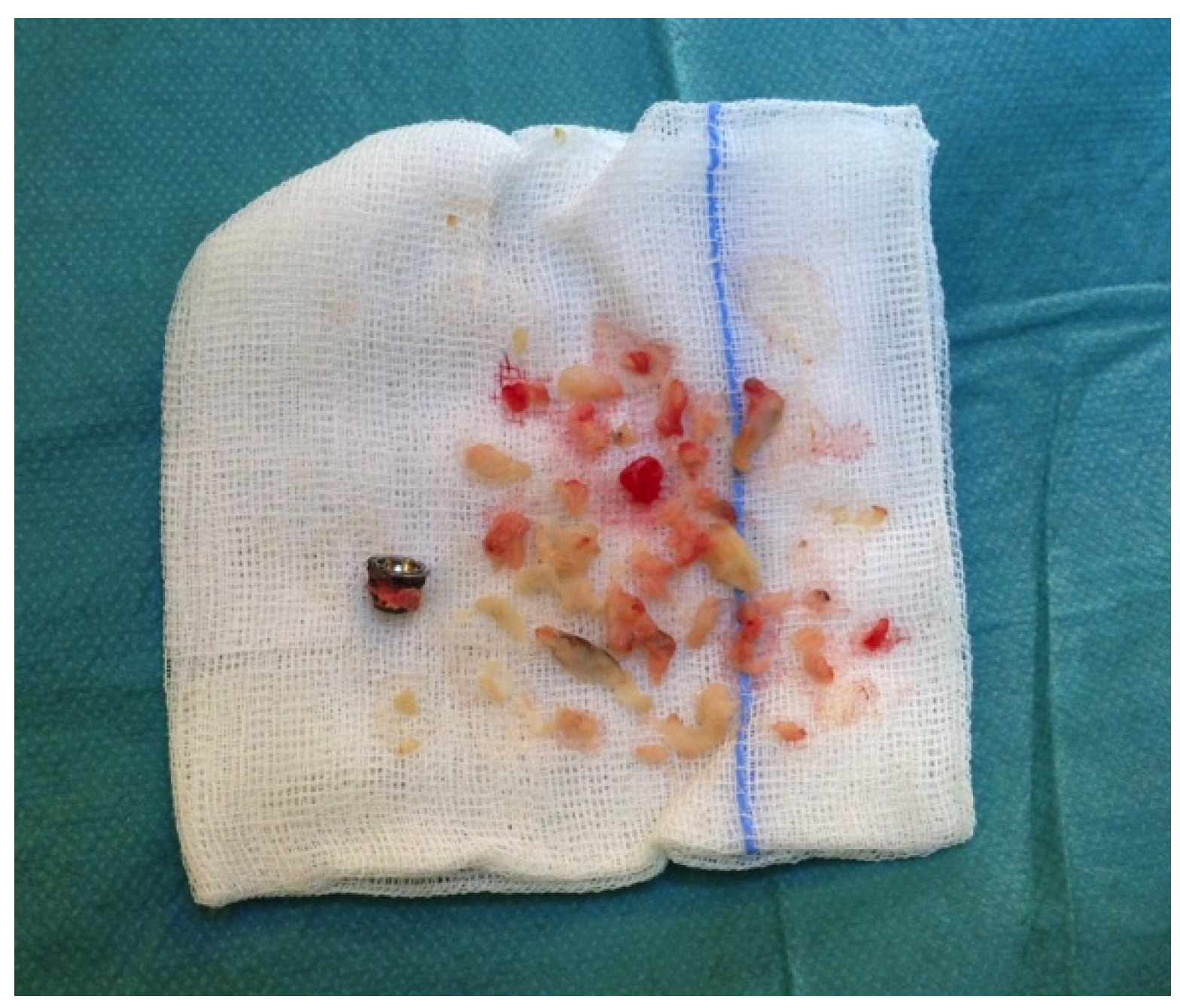
Case 2
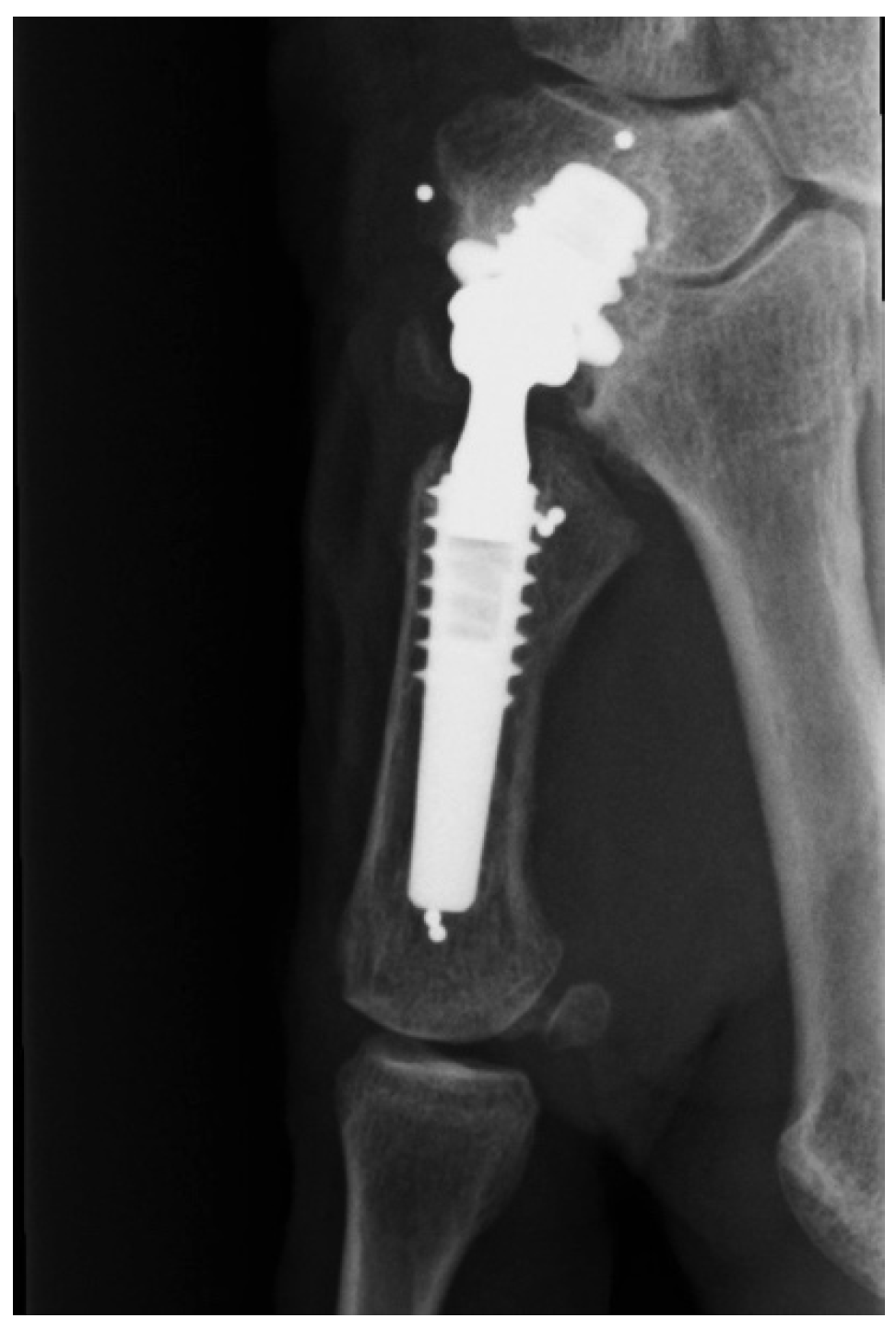
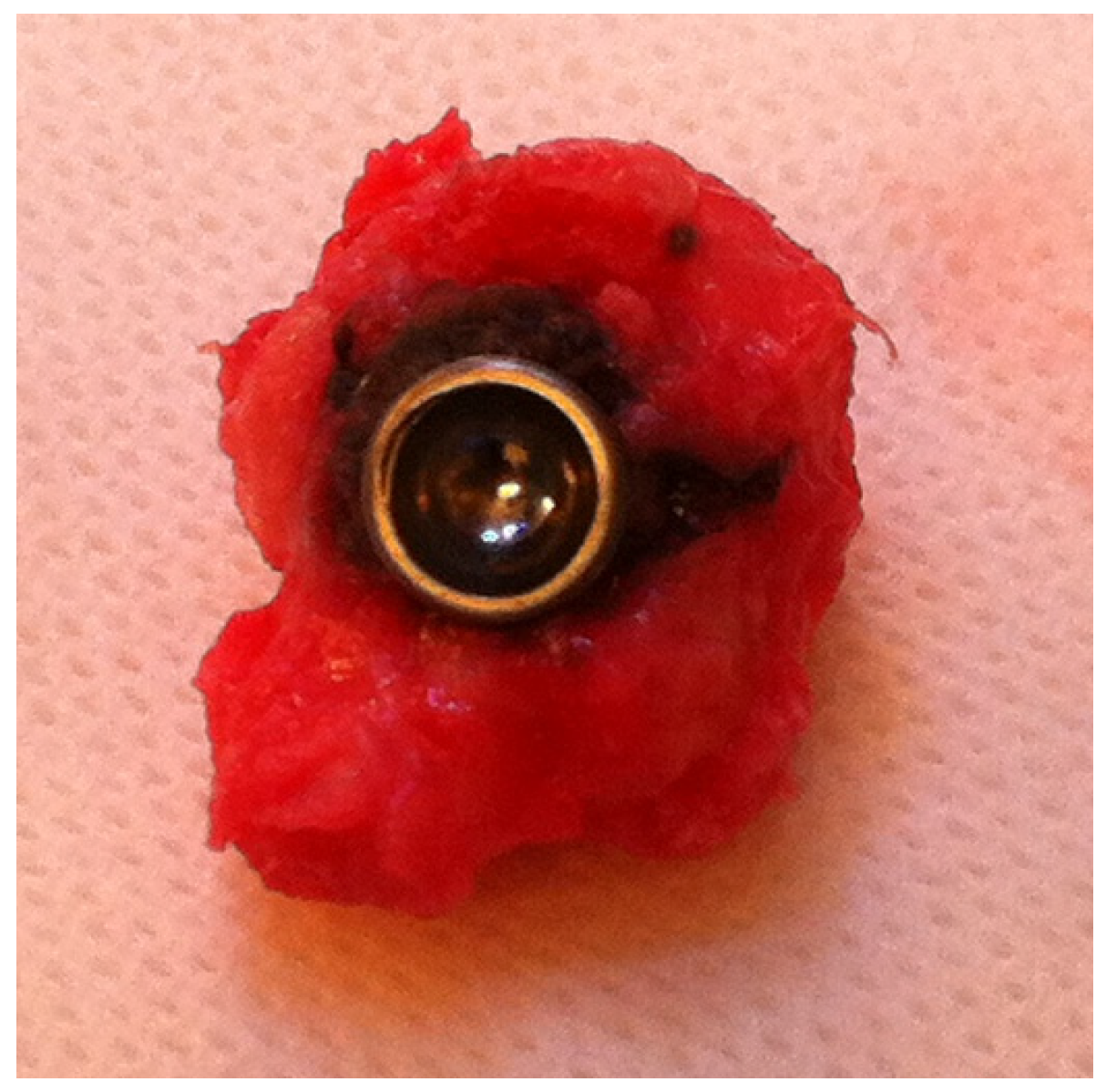
3. Discussion
4. Conclusions
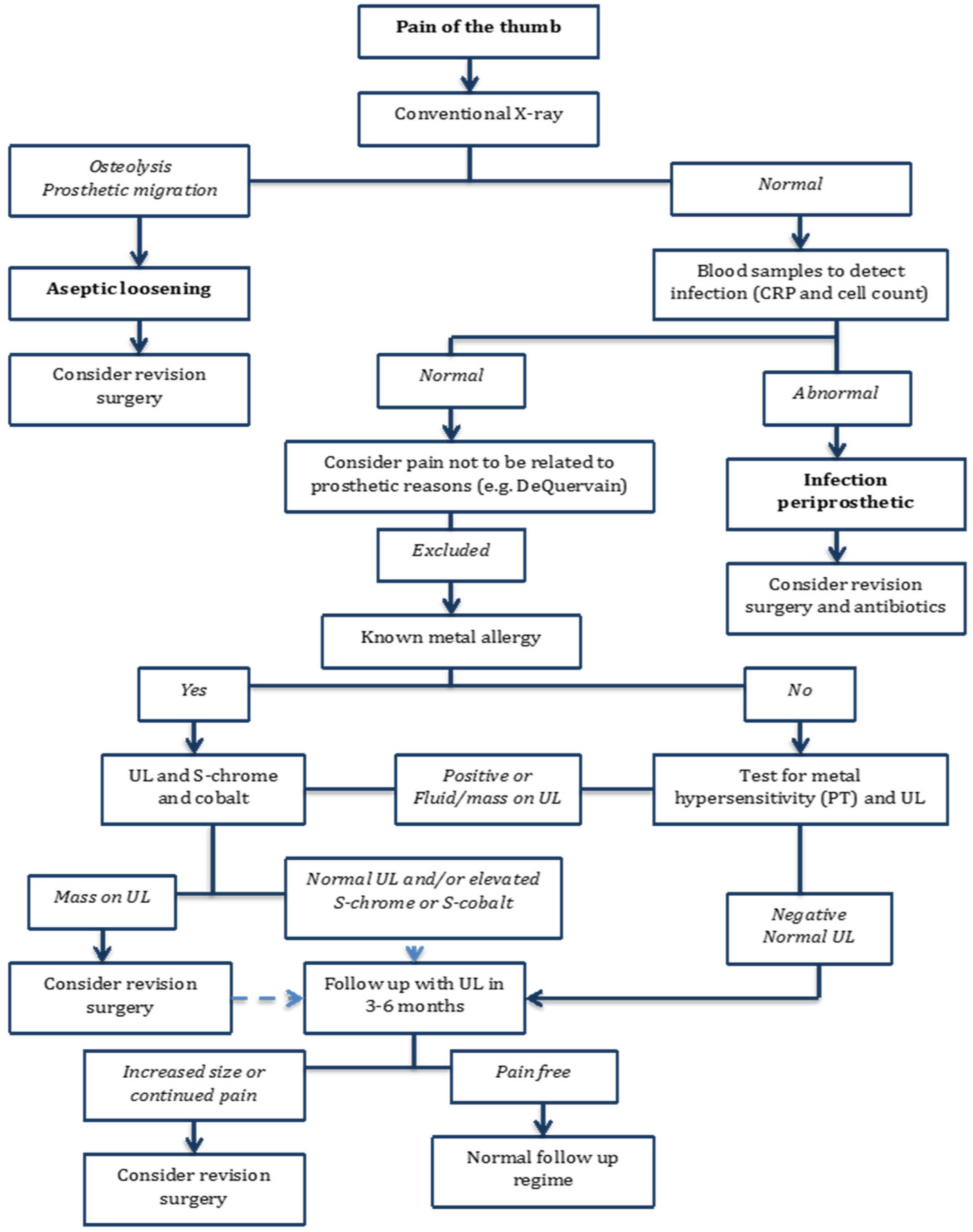
Author Contributions
Conflicts of Interest
References
- Wajon, A.; Carr, E.; Edmunds, I.; Ada, L. Surgery for thumb (trapeziometacarpal joint) osteoarthritis. Cochrane Database Syst. Rev. 2009, 4. [Google Scholar] [CrossRef]
- Kaszap, B.; Daecke, W.; Jung, M. Outcome comparison of primary trapeziectomy versus secondary trapeziectomy following failed total trapeziometacarpal joint replacement. J. Hand Surg. Am. 2013, 38, 863–871. [Google Scholar] [CrossRef] [PubMed]
- Hentz, V.R. Surgical treatment of trapeziometacarpal joint arthritis—A historical perspective. Clin. Orthop. Relat. Res. 2014, 472, 1184–1189. [Google Scholar] [CrossRef] [PubMed]
- Regnard, P.J. Electra trapeziometacarpal prosthesis: Results of the first 100 cases. J. Hand Surg. Eur. 2006, 31, 621–628. [Google Scholar] [CrossRef]
- Ulrich-Vinther, M.; Puggaard, H.; Lange, B. Prospective 1-year follow-up study comparing joint prosthesis with tendon interposition arhtroplasty in treatment of trapeziometacarpal osteoarthritis. J. Hand Surg. Am. 2008, 33, 1369–1377. [Google Scholar] [CrossRef] [PubMed]
- Hasegawa, M.; Yoshida, K.; Wakabayashi, H.; Sudo, A. Cobalt and chromium ion release after large-diameter metal-on-metal total hip arthroplasty. J. Arthroplast. 2012, 27, 990–996. [Google Scholar] [CrossRef]
- Matthies, A.K.; Racasan, R.; Bills, P.; Blunt, L.; Cro, S.; Panagiotidou, A.; Blunn, G.; Skinner, J.; Hart, A.J. Material loss at the taper junction of retrieved large head metal-on-metal total hip replacement. J. Orthop. Res. 2013, 31, 1677–1685. [Google Scholar] [PubMed]
- Campbell, J.R.; Estey, M.P. Metal release from hip prostheses: Cobalt and chromium toxicity and the role of the clinical laboratory. Clin. Chem. Lab. Med. 2013, 51, 213–220. [Google Scholar] [CrossRef] [PubMed]
- Lombardi, A.V.; Barrack, R.L.; Berend, K.R.; Cuckler, J.M.; Jacobs, J.J.; Mont, M.A.; Schmalzried, T.P. The hip society—Algorithmic approach to diagnosis and management of metal-on-metal arhtroplasty. J. Bone. Joint Surg. Br. 2012, 94, 14–18. [Google Scholar] [CrossRef] [PubMed]
- Zeng, Y.; Feng, W. Metal allergy in patients with total hip replacement: A review. J. Int. Med. Res. 2013, 41, 247–252. [Google Scholar] [CrossRef] [PubMed]
- Bayley, N.; Khan, H.; Grosso, P.; Hupel, T.; Stevens, D.; Snider, M.; Schemitsch, E.; Kuzyk, P. What are the predictors and prevalence of pseudotumor and elevated metal ions after large-diameter metal-on-metal THA? Clin. Orthop. Relat. Res. 2015, 473, 477–484. [Google Scholar] [CrossRef] [PubMed]
- Willert, H.G.; Buchhorn, G.H.; Fayyazi, A.; Flury, R.; Windler, M.; Köster, G.; Lohmann, C.H. Metal-on-metal bearings and hypersensitivity in patients with artificial hip joints. A clinical and histomorphological study. J. Bone Joint Surg. Am. 2005, 87, 28–36. [Google Scholar] [CrossRef] [PubMed]
- Almousa, S.A.; Greidanus, N.V.; Masri, B.A.; Duncan, C.P.; Garbuz, D.S. The natural history of inflammatory pseudotumours in asymptomatic patients after metal-on-metal hip arthroplasty. Clin. Orthop. Relat. Res. 2013, 471, 3814–3821. [Google Scholar] [CrossRef] [PubMed]
- Amini, M.H.; Mayes, W.H.; Tzeng, A.; Tzeng, T.H.; Saleh, K.J.; Mihalko, W.M. Evaluation and management of metal hypersensitivity in total joint arthroplasty: A systematic review. J. Long Term Eff. Med. Implants 2014, 24, 25–36. [Google Scholar] [CrossRef] [PubMed]
- BS ISO 5832-12:2007 Implants for Surgery—Metallic Materials—Part 12: Wrought Cobalt-Chromium-Molybedenium Alloy; ISO: Geneva, Switzerland, 2007.
- Kwon, Y.M.; Thomas, P.; Summer, B.; Pandit, H.; Taylor, A.; Beard, D.; Murray, D.W.; Gill, H.S. Lymphocyte proliferation responses in patients with pseudotumors following metal-on-metal hip resurfacing arthroplasty. J. Orthop. Res. 2010, 28, 444–450. [Google Scholar] [PubMed]
- Stürup, J.; Dahl, L.B.; Jensen, K.E.; Larsen, A.B.; Gebuhr, P. Few adverse reactions to metal on metal articulation in total hip arthroplasty in a review study on 358 consecutive cases with 1 to 5 years follow-up. Open Orthop. J. 2012, 6, 366–370. [Google Scholar] [CrossRef] [PubMed]
- Matthies, A.K.; Skinner, J.A.; Osmani, H.; Henckel, J.; Hart, A.J. Pseudotumors are common in well-positioned low-wearing metal-on-metal hips. Clin. Orthop. Relat. Res. 2012, 470, 1895–1906. [Google Scholar] [CrossRef] [PubMed]
- Bisschop, R.; Boomsma, M.F.; Van Raay, J.J.; Tiebosch, A.T.; Maas, M.; Gerritsma, C.L. High prevalence of pseudotumors in patients with a Birmingham hip resurfacing prosthesis: A prospective cohort study of one hundred and twenty-nine patients. J. Bone Joint Surg. Am. 2013, 95, 1554–1560. [Google Scholar] [CrossRef] [PubMed]
- Chang, E.Y.; McAnally, J.L.; Van Horne, J.R.; Van Horne, J.G.; Wolfson, T.; Gamst, A.; Chung, C.B. Relationship of plasma metal ions and clinical and imaging findings in patients with ASR XL metal-on-metal total hip replacement. J. Bone Joint Surg. Am. 2013, 95, 2015–2020. [Google Scholar] [CrossRef] [PubMed]
- Bisseling, P.; José, M.H. Metal ion levels and functional results following resurfacing hip arthroplasty versus conventional small-diameter metal-on-metal total hip arthroplasty; a 3 to 5 year follow-up of a randomized controlled trial. J. Arthroplast. 2015, 30, 61–67. [Google Scholar] [CrossRef]
- Lohmann, C.H.; Meyer, H.; Nuechtern, J.V.; Singh, G.; Junk-Jantsch, S.; Schmotzer, H.; Morlock, M.M.; Pflüger, G. Periprosthetic tissue metal content but not serum metal content predicts the type of tissue response in failed small-diameter metal-on-metal total hip arthroplasties. J. Bone Joint Surg. Am. 2013, 95, 1561–1568. [Google Scholar] [CrossRef] [PubMed]
- Hallab, N.J.; Anderson, S.; Caicedo, M.; Skipor, A.; Campbell, P.; Jacobs, J.J. Immune responses correlate with serum-metal in metal-on-metal hip arthroplasty. J. Arthroplast. 2004, 19, 88–93. [Google Scholar] [CrossRef]
- Hasegawa, M.; Miyamoto, N.; Miyazaki, S.; Wakabayashi, H.; Sudo, A. Longitudinal magnetic resonance imaging of pseudotumors following metal-on-metal total hip arthroplasty. J. Arthroplast. 2014, 29, 2236–2238. [Google Scholar] [CrossRef]
- Smith, E.; Mehta, A.J.; Statham, B.N. Metal sensitivity to Elektra prostheses—Two cases from a metal on metal implant for hand joint replacement. Contact Dermatit. 2009, 60. [Google Scholar] [CrossRef]
- Lohmann, C.H.; Nuechtern, J.V.; Willert, H.G.; Junk-Jantsch, S.; Ruether, W.; Pflueger, G. Hypersensitivity reations in total hip arthroplasty. Orthopedics 2007, 30, 760–761. [Google Scholar] [PubMed]
- Hart, A.J.; Sabah, S.; Henckel, J.; Lewis, A.; Cobb, J.; Sampson, B.; Mitchell, A.; Skinner, J.A. The painful metal-on-metal hip resurfacing. J. Bone Joint Surg. Br. 2009, 91, 738–744. [Google Scholar] [CrossRef] [PubMed]
- Mahendra, G.; Pandit, H.; Kliskey, K.; Murray, D.; Gill, H.S.; Athanasou, N. Necrotic and inflammatory changes in metal-on-metal resurfacing hip arthroplasties. Acta Orthop. 2009, 80, 653–659. [Google Scholar] [CrossRef] [PubMed]
- Hart, A.J.; Satchithananda, K.; Liddle, A.D.; Sabah, S.A.; McRobbie, D.; Henckel, J.; Cobb, J.P.; Skinner, J.A.; Mitchell, A.W. Pseudotumors in association with well-functioning metal-on-metal hip prostheses: A case-control study using three-dimensional computed tomography and magnetic resonance imaging. J. Bone Joint Surg. Am. 2012, 94, 317–325. [Google Scholar] [PubMed]
- Liddle, A.D.; Satchithananda, K.; Henckel, J.; Sabah, S.A.; Vipulendran, K.V.; Lewis, A.; Skinner, J.A.; Mitchell, A.W.; Hart, A.J. Revision of metal-on-metal hip arthroplasty in a tertiary center—A prospective study of 39 hips with between 1 and 4 years follow up. Acta Orthop. 2013, 84, 237–245. [Google Scholar] [CrossRef] [PubMed]
- Hansen, T.B.; Dremstrup, L.; Stilling, M. Patients with metal-on-metal articulation in trapeziometacarpal total joint arthroplasty may have elevated serum chrome and cobalt. J. Hand Surg. Eur. 2013, 38, 860–865. [Google Scholar] [CrossRef]
- Brown, G.C.; Lckshin, M.D.; Salvati, E.A.; Bullough, P.G. Sensitivity to metal as a possible cause of sterile loosening after cobalt-chromium total hip replacement arhtroplasty. J. Bone Joint Surg. Am. 1977, 59, 164–168. [Google Scholar] [PubMed]
- Schalock, P.C.; Menné, T.; Johansen, J.D.; Taylor, J.S.; Maibach, H.I.; Lidén, C.; Bruze, M.; Thyssen, J.P. Hypersensitivity reactions to metallic implants—Diagnostic algorithm and suggested patch test series for clinical use. Contact Dermat. 2011, 66, 4–19. [Google Scholar] [CrossRef]
- Cousen, P.J.; Gawkrodger, D.J. Metal allergy and second-generation metal-on-metal arthroplasties. Contact Dermat. 2011, 66, 55–62. [Google Scholar] [CrossRef]
- Garbuz, D.S.; Hargreaves, B.A.; Duncan, C.P.; Masri, B.A.; Wilson, D.R.; Forster, B.B. Diagnostic accuracy of MRI versus ultrasound for detecting pseudtotumors in asymptomatic metal-on-metal THA. Clin. Orthop Relat. Res. 2014, 472, 417–423. [Google Scholar] [CrossRef] [PubMed]
- Medical Device Alert, MDA/2012/036; Medicines and Healthcare Products Regulatory Agency: London, UK, 2012.
© 2015 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Frølich, C.; Hansen, T.B. Complications Related to Metal-on-Metal Articulation in Trapeziometacarpal Joint Total Joint Arthroplasty. J. Funct. Biomater. 2015, 6, 318-327. https://doi.org/10.3390/jfb6020318
Frølich C, Hansen TB. Complications Related to Metal-on-Metal Articulation in Trapeziometacarpal Joint Total Joint Arthroplasty. Journal of Functional Biomaterials. 2015; 6(2):318-327. https://doi.org/10.3390/jfb6020318
Chicago/Turabian StyleFrølich, Christina, and Torben Bæk Hansen. 2015. "Complications Related to Metal-on-Metal Articulation in Trapeziometacarpal Joint Total Joint Arthroplasty" Journal of Functional Biomaterials 6, no. 2: 318-327. https://doi.org/10.3390/jfb6020318
APA StyleFrølich, C., & Hansen, T. B. (2015). Complications Related to Metal-on-Metal Articulation in Trapeziometacarpal Joint Total Joint Arthroplasty. Journal of Functional Biomaterials, 6(2), 318-327. https://doi.org/10.3390/jfb6020318




