Nanomedicine Approaches for Corneal Diseases
Abstract
:1. Introduction
2. Pathology of Corneal Diseases
2.1. Corneal Infection
2.2. Corneal Scarring/Fibrosis
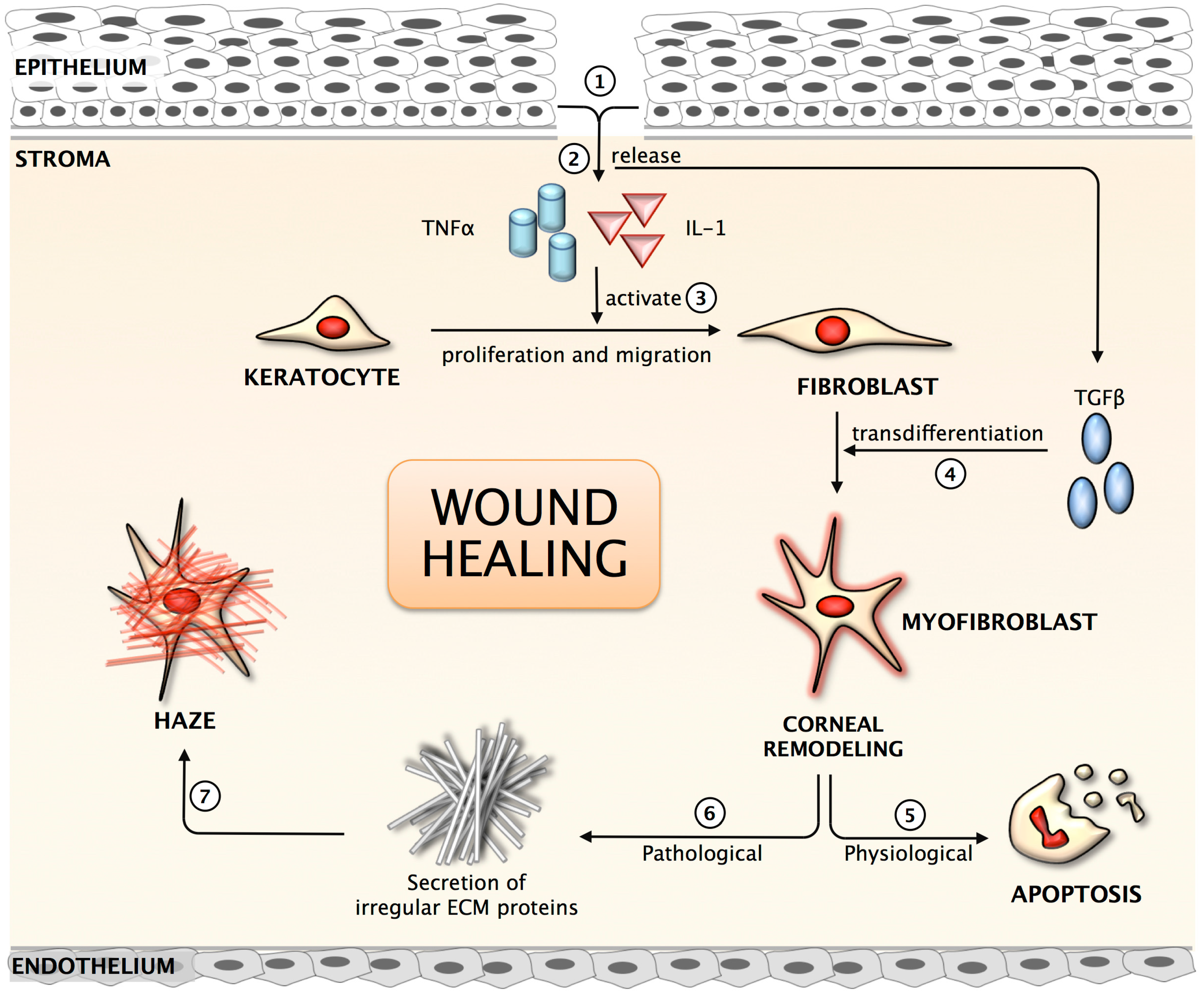
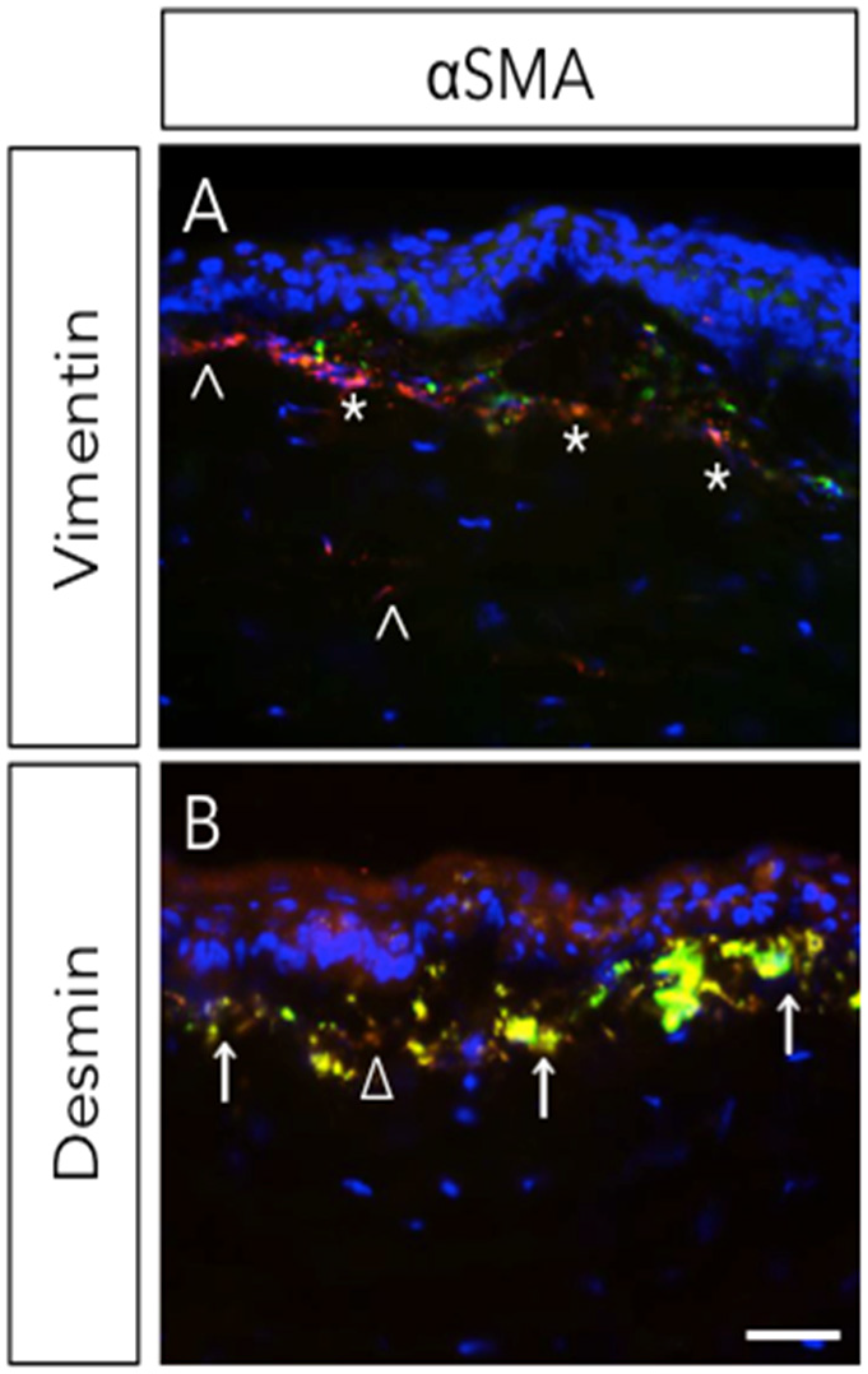
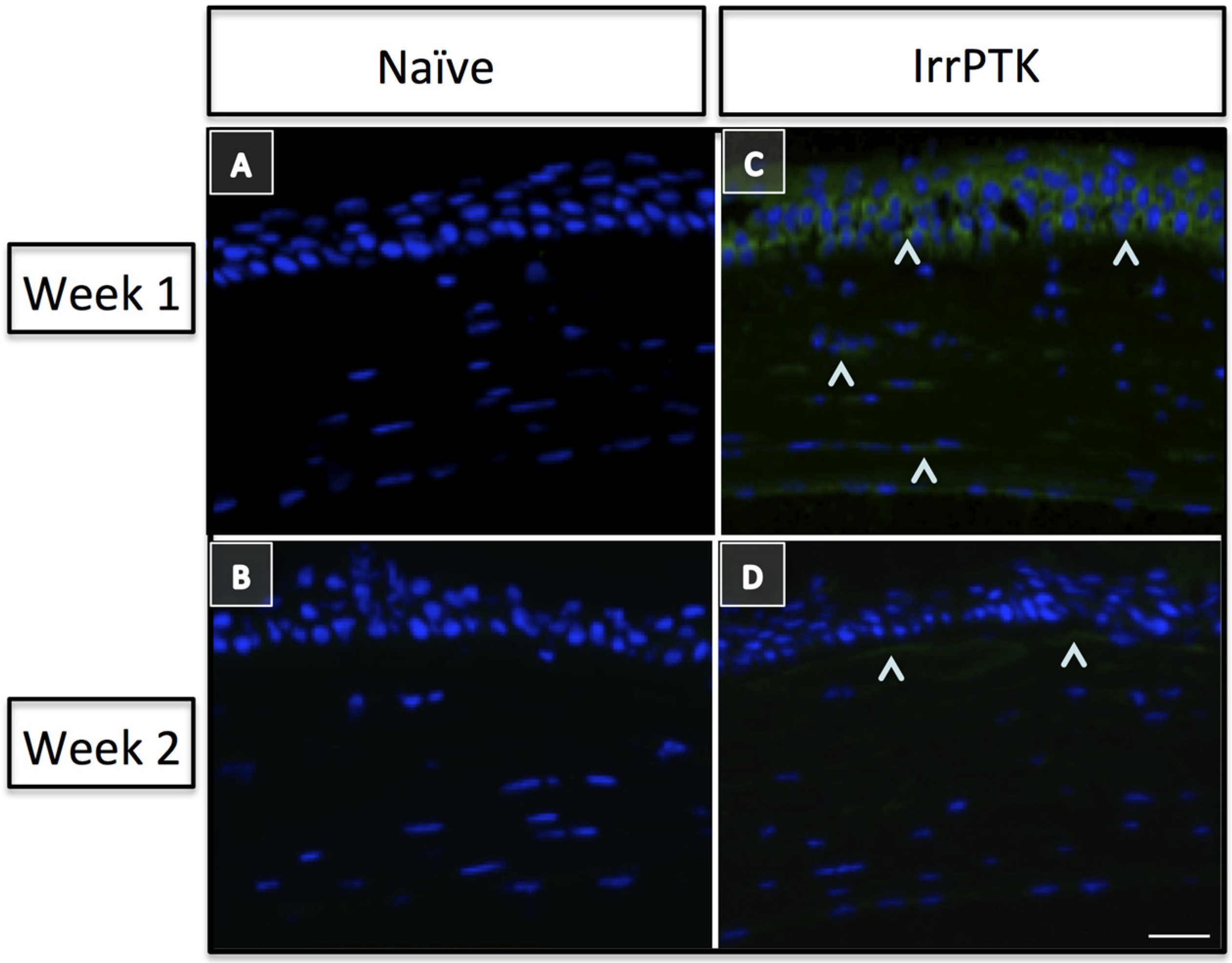
2.3. Corneal Neovascularization
3. Nanomedicine for Corneal Diseases
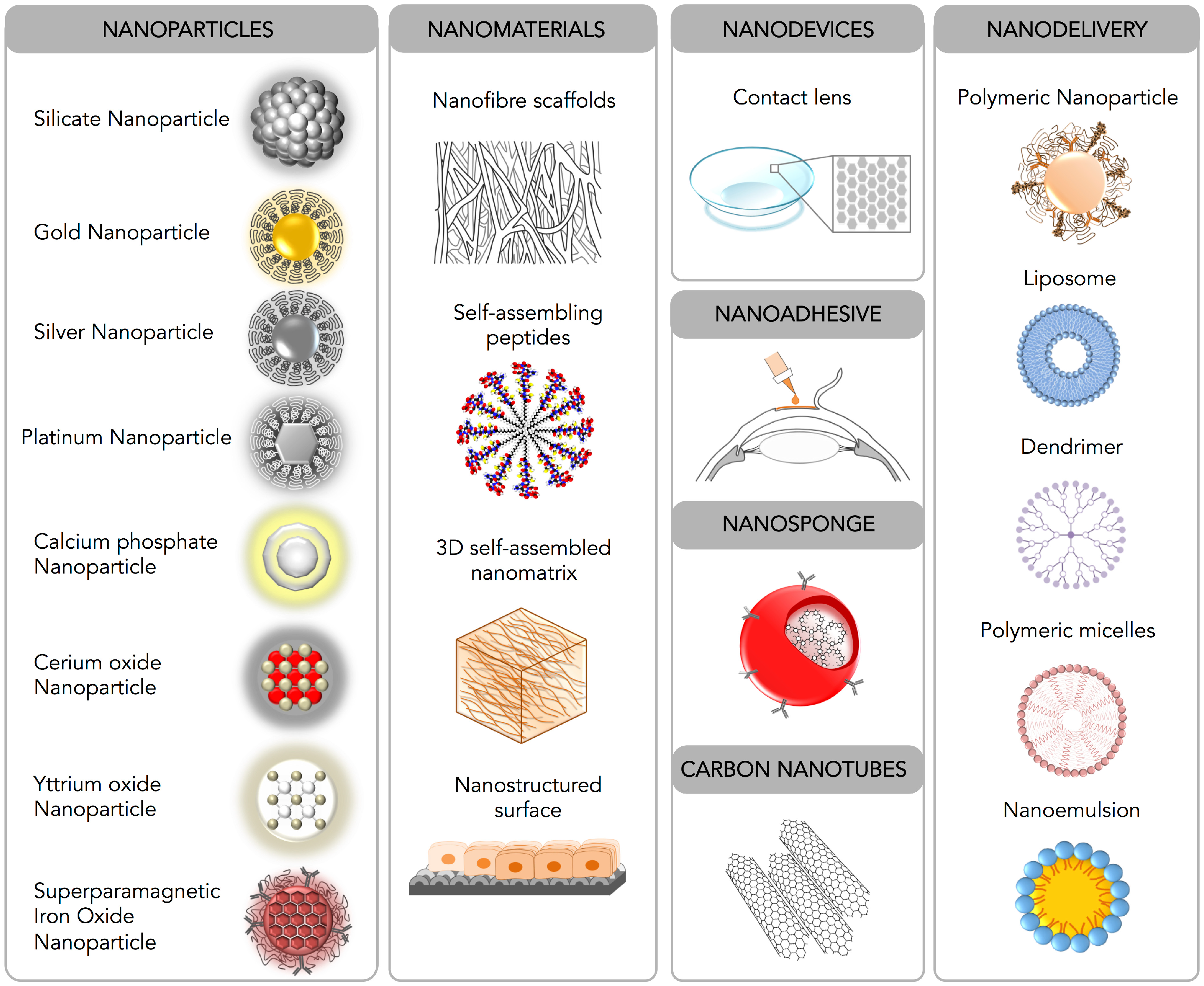
3.1. Nanoparticles
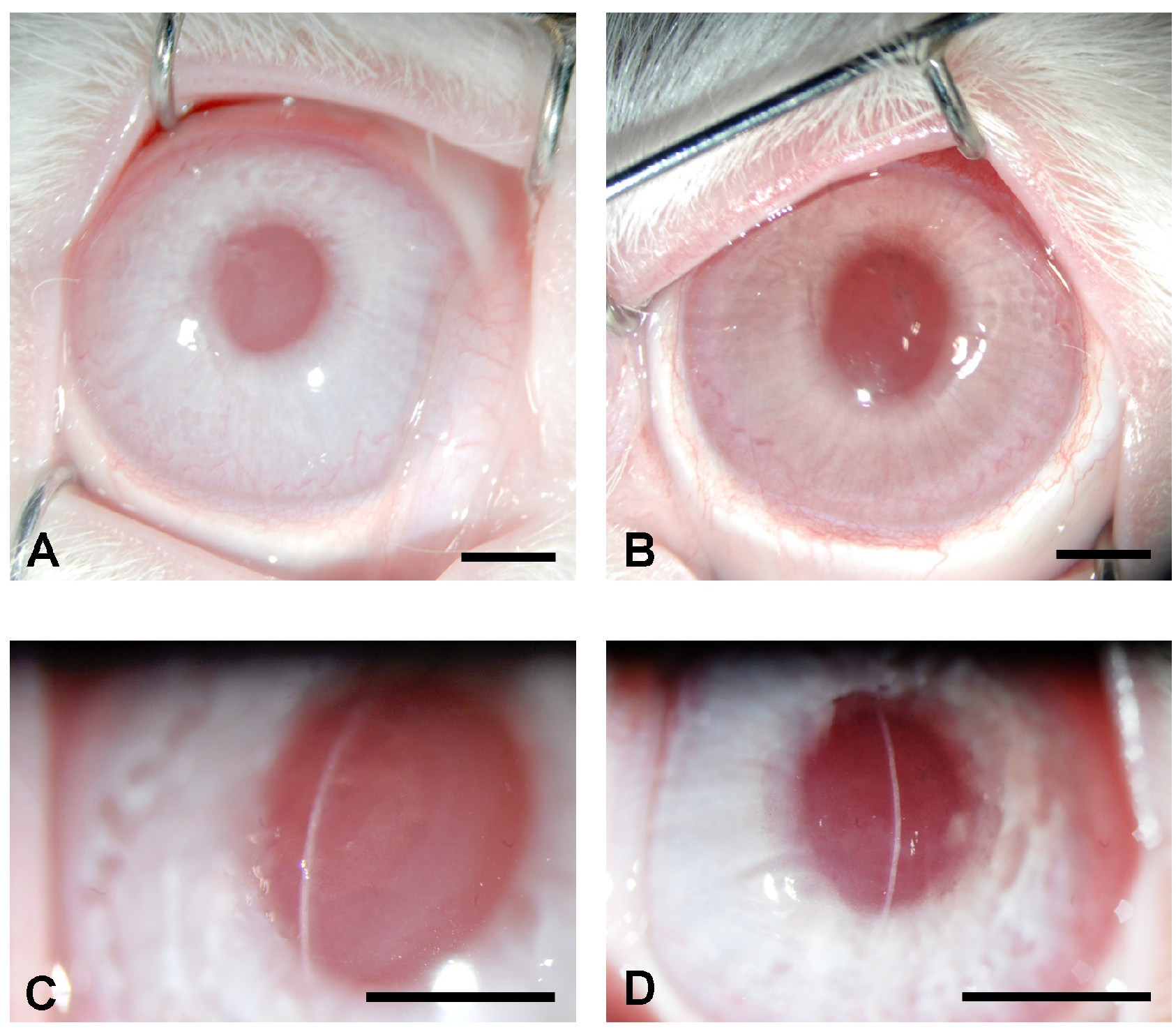
3.2. Nanofiber Scaffolds
3.3. Nanodevices
3.4. Nanoadhesives, Nanosponges and Carbon Nanotubes
3.5. Nanodelivery
| Antimicrobials | Liposome properties | Experiments | Results | Ref. | |
|---|---|---|---|---|---|
| Lipid Composition | Particle size (nm) | ||||
| Itraconazole | PC:Chol:SA (7:2:1) | 276.5 | Rabbit model of microbial keratitis (Strains: Aspergilus flavus URM 6029) | Liposomal formulations decreased fungal burden compared to free drug | [134] |
| Fluconazole | N/A | N/A | Rabbit model of microbial keratitis (Strains: C. albicans) | Complete healing occurred in 86% animals given liposomal formulations compared to 50% in the free drug group. Decreased instillation frequency, duration of recovery and healing compared to free drug | [135] |
| Tobramycin | Multivesicular liposomes | 103–105 | Rabbit model of microbial keratitis (Strains: P. aeruginosa) | Liposomal formulations combined with fibrin sealants require 5-fold less tobramycin compared to eye drops | [136] |
| Tobramycin | Hexadecylphosphate (1:2) (Solid-lipid NPs) | 80 | In vivo pharmaco kinetics in rabbits | SLN increases the bioavailability of tobramycin compared to commercial eye drops | [137] |
| Gentamycin | Phosphatidic acid, PC, a-tocopherol (1:19:0.22) | 100–1000 | In vivo pharmacokinetics by subconjunctival injections in pigmented rabbits | Gentamycin availability increased in the cornea | [138] |
| Ciprofloxacin | PC:Cho:DODAB | 530 ± 25 | In vivo pharmacokinetics in rabbits | Higher AUC and 3-fold enhanced bioavailability for the liposomal formulations compared to eye drop instillations | [139] |
| DPPC:Cho:DODAB | 619 ± 71 | ||||
| DMPC | 580 ± 197 | ||||
| Ciprofloxacin | PC:Chol (5:3) | 1630 | Ex vivo corneal permeability in rabbits | 3-fold increase in transcorneal permeation was observed compared to free drug. Addition of carbopol increased the transcorneal efficiency by about 5 times compared to eye drop | [140] |
| PC:Chol :SA (5:3:1) | 1850 | ||||
| PC:Chol :SA (5:3:1) coated with carbopol gel | – | ||||
| Ciprofloxacin | Lecithin:Cho (7:2) | 338 | In vitro antimicrobial assays | Two different liposomal formulations (MLV and REV) of cioprofloxacin were coated onto contact lenses and the MLVs coated lenses showed better zone of inhibition compared to the REVs-coated lenses | [141] |
| Norfloxacin | DMPC | 1090 | Ex vivo corneal permeability in porcine | Corneal retention of norfloxacin increased for DSPC liposomes | [142] |
| DPPC | 1410 | ||||
| DSPC | 2230 | ||||
4. Future Directions
5. Conclusions
Acknowledgements
Author Contributions
Conflicts of Interest
References
- Klintworth, G.K. The cornea—Structure and macromolecules in health and disease. A review. Am. J. Pathol. 1977, 89, 718–808. [Google Scholar] [PubMed]
- Böhnke, M.; Masters, B.R. Confocal microscopy of the cornea. Prog. Retin. Eye Res. 1999, 18, 553–628. [Google Scholar] [CrossRef] [PubMed]
- Jester, J.V.; Moller-Pedersen, T.; Huang, J.; Sax, C.M.; Kays, W.T.; Cavangh, H.D.; Petroll, W.M.; Piatigorsky, J. The cellular basis of corneal transparency: Evidence for ‘corneal crystallins’. J. Cell Sci. 1999, 112, 613–622. [Google Scholar] [PubMed]
- Kinoshita, S.; Adachi, W.S.; Nishida, K.Y.N.; Quantock, A.J.; Okubo, K. Characteristics of the human ocular surface epithelium. Prog. Retin. Eye Res. 2001, 20, 639–673. [Google Scholar] [CrossRef] [PubMed]
- Tuft, S.J.; Coster, D.J. The corneal endothelium. Eye (Lond) 1990, 4, 389–424. [Google Scholar] [CrossRef]
- Hanlon, S.D.; Behzad, A.R.; Sakai, L.Y.; Burns, A.R. Corneal Stroma Microfibrils. Exp. Eye Res. 2015, 132, 198–207. [Google Scholar] [CrossRef] [PubMed]
- Yamanaka, O.; Liu, C.Y.; Kao, W.W. Fibrosis in the anterior segments of the eye. Endocr. Metab. Immune Disord. Drug Targets 2010, 10, 331–335. [Google Scholar] [CrossRef] [PubMed]
- Hassell, J.R.; Birk, D.E. The molecular basis of corneal transparency. Exp. Eye Res. 2010, 91, 326–335. [Google Scholar] [CrossRef] [PubMed]
- McCluskey, P.; Powell, R.J. The eye in systemic inflammatory diseases. Lancet 2004, 364, 2125–2133. [Google Scholar] [CrossRef] [PubMed]
- Shoham, A.; Hadziahmetovic, M.; Dunaief, J.L.; Mydlarski, M.B.; Schipper, H.M. Oxidative stress in diseases of the human cornea. Free Rad. Biol. Med. 2008, 45, 1047–1055. [Google Scholar] [CrossRef] [PubMed]
- Wilson, S.E.; Chaurasia, S.S.; Medeiros, F.W. Apoptosis in the initiation, modulation and termination of the corneal wound healing response. Exp. Eye Res. 2007, 85, 305–311. [Google Scholar] [CrossRef] [PubMed]
- Cintron, C.; Schneider, H.; Kublin, C.L. Corneal scar formation. Exp. Eye Res. 1973, 17, 215–259. [Google Scholar] [CrossRef]
- Tandon, A.; Tovey, J.C.; Sharma, A.; Gupta, R.; Mohan, R.R. Role of transforming growth factor Beta in corneal function, biology and pathology. Curr. Mol. Med. 2010, 10, 565–578. [Google Scholar] [PubMed]
- Chaurasia, S.S.; Kaur, H.; de Medeiros, F.W.; Smith, S.D.; Wilson, S.E. Dynamics of the expression of intermediate filaments vimentin and desmin during myofibroblast differentiation after corneal injury. Exp. Eye Res. 2009, 89, 590–596. [Google Scholar] [CrossRef] [PubMed]
- Nien, C.J.; Flynn, K.J.; Chang, M.; Brown, D.; Jester, J.V. Reducing peak corneal haze after photorefractive keratectomy in rabbits: Prednisolone acetate 1.00% versus cyclosporine A 0.05%. J. Cataract Refract. Surg. 2011, 37, 937–944. [Google Scholar] [CrossRef] [PubMed]
- Srinivasan, M.; Mascarenhas, J.; Rajaraman, R.; Ravindran, M.; Lalitha, P.; Glidden, D.V.; Ray, K.J.; Hong, K.C.; Oldenburg, C.E.; Lee, S.M.; et al. Corticosteroids for bacterial keratitis: The steroids for corneal ulcers trial (SCUT). Arch. Ophthalmol. 2012, 130, 143–150. [Google Scholar] [CrossRef] [PubMed]
- Camellin, M. Laser epithelial keratomileusis with mitomycin C: indications and limits. J. Refract. Surg. 2004, 20, S693–S698. [Google Scholar] [PubMed]
- Safianik, B.; Ben-Zion, I.; Garzozi, H.J. Serious corneoscleral complications after pterygium excision with mitomycin C. Br. J. Ophthalmol. 2002, 86, 357–358. [Google Scholar] [CrossRef] [PubMed]
- Wagner, V.; Dullaart, A.; Bock, A.K.; Zweck, A. The emerging nanomedicine landscape. Nature Biotechnol. 2006, 24, 1211–1218. [Google Scholar] [CrossRef]
- Wickline, S.A.; Lanza, G.M. Nanotechnology for molecular imaging and targeted therapy. Circulation. 2003, 107, 1092–1095. [Google Scholar] [CrossRef] [PubMed]
- Kim, B.Y.S.; Rutka, J.T.; Chan, W.C. Nanomedicine. N. Engl. J. Med. 2010, 363, 2434–2443. [Google Scholar] [CrossRef] [PubMed]
- Diebolda, Y.; Calongea, M. Applications of nanoparticles in ophthalmology. Prog. Retin. Eye Res. 2010, 29, 596–609. [Google Scholar] [CrossRef] [PubMed]
- Raju, H.B.; Goldberg, J.L. Nanotechnology for ocular therapeutics and tissue repair. Expert Rev. Ophthalmol. 2008, 3, 431–436. [Google Scholar] [CrossRef]
- Zarbin, M.A.; Arlow, T.; Ritch, R. Regenerative nanomedicine in vision restoration. Mayo Clin. Proc. 2013, 88, 1480–1490. [Google Scholar] [CrossRef] [PubMed]
- Sharma, A.; Tandon, A.; Tovey, J.C.; Gupta, R.; Robertson, J.D.; Fortune, J.A.; Klibanov, A.M.; Cowden, J.W.; Rieger, F.G.; Mohan, R.R. Polyethylenimine-conjugated gold nanoparticles: Gene transfer potential and low toxicity in the cornea. Nanomedicine. 2011, 7, 505–513. [Google Scholar] [CrossRef] [PubMed]
- Sharma, A.; Rodier, J.T.; Tandon, A.; Klibanov, A.M.; Mohan, R.R. Attenuation of corneal myofibroblast development through nanoparticle-mediated soluble transforming growth factor-β type II receptor (sTGFβRII) gene transfer. Mol. Vis. 2012, 18, 2598–2607. [Google Scholar] [PubMed]
- Chowdhury, S.; Guha, R.; Trivedi, R.; Kompella, U.B.; Konar, A.; Hazra, S. Pirfenidone nanoparticles improve corneal wound healing and prevent scarring following alkali burn. PLoS One 2013, 8, e70528. [Google Scholar] [CrossRef] [PubMed]
- Tandon, A.; Sharma, A.; Rodier, J.T.; Klibanov, A.M.; Rieger, F.G.; Mohan, R.R. BMP7 gene transfer via gold nanoparticles into stroma inhibits corneal fibrosis in vivo. PLoS One 2013, 8, e66434. [Google Scholar] [CrossRef] [PubMed]
- Evans, D.J.; McNamara, N.A.; Fleiszig, S.M. Life at front: Dissecting bacterial-host interactions at the ocular surface. Ocul. Surf. 2007, 5, 213–227. [Google Scholar] [CrossRef] [PubMed]
- Gipson, I.K. Distribution of mucins at the ocular surface. Exp. Eye Res. 2004, 78, 379–388. [Google Scholar] [CrossRef] [PubMed]
- Flanagan, J.L.; Willcox, M.D.P. Role of lactoferrin in the tear film. Biochimie 2009, 91, 35–43. [Google Scholar] [CrossRef] [PubMed]
- Hanes, R.J.; Tighe, P.J.; Dua, H.S. Antimicrobial defensin peptides of the human ocular surface. Br. J. Ophthalmol. 1999, 83, 737–741. [Google Scholar] [CrossRef] [PubMed]
- Huang, L.C.; Jean, D.; Proske, R.J.; Reins, R.Y.; McDermott, A.M. Ocular surface expression and in vitro activity of antimicrobial peptides. Curr. Eye Res. 2007, 32, 595–609. [Google Scholar] [CrossRef] [PubMed]
- Jeng, B.H.; McLeod, S.D. Microbial keratitis. Br. J. Ophthalmol. 2003, 87, 805–806. [Google Scholar] [CrossRef] [PubMed]
- Green, M.; Apel, A.; Stapleton, F. Risk factors and causative organisms in microbial keratitis. Cornea. 2008, 27, 22–27. [Google Scholar] [CrossRef] [PubMed]
- Bourcier, T.; Thomas, F.; Borderie, V.; Chaumeil, C.; Laroche, L. Bacterial keratitis: predisposing factors, clinical, and microbiological review of 300 cases. Br. J. Ophthalmol. 2003, 87, 834–848. [Google Scholar] [CrossRef] [PubMed]
- Sirikul, T.; Prabriputaloong, T.; Smathivat, A.; Chuck, R.S.; Vongthongsri, A. Predisposing factors and etiologic diagnosis of ulcerative keratitis. Cornea 2008, 27, 283–287. [Google Scholar] [CrossRef] [PubMed]
- Bourlais, C.L.; Acar, L.; Zia, H.; Sado, P.A.; Needham, T.; Leverge, R. Ophthalmic drug delivery systems-recent advances. Prog. Retin. Eye Res. 1998, 17, 33–58. [Google Scholar] [CrossRef] [PubMed]
- Thompson, A.M. Ocular toxicity of fluoroquinolones. Clin. Exp. Ophthalmol. 2007, 35, 566–577. [Google Scholar] [CrossRef]
- Kuwabara, T.; Perkins, D.G.; Cogan, D.G. Sliding of the epithelium in experimental corneal wounds. Invest. Ophthalmol. Vis. Sci. 1976, 15, 4–14. [Google Scholar]
- Wilson, S.E.; Liu, J.J.; Mohan, R.R. Stromal-Epithelial interactions in the cornea. Prog. Retin. Eye Res. 1999, 18, 293–309. [Google Scholar] [CrossRef] [PubMed]
- Salomão, M.Q.; Chaurasia, S.S.; Sinha-Roy, A.; Ambrósio, R., Jr.; Esposito, A.; Sepulveda, R.; Agrawal, V.; Wilson, S.E. Corneal wound healing after ultraviolet-A/riboflavin collagen cross-linking: A rabbit study. J. Refract. Surg. 2011, 27, 401–407. [Google Scholar] [CrossRef] [PubMed]
- Kuo, I.C. Corneal wound healing. Curr. Opin. Ophthalmol. 2004, 15, 311–315. [Google Scholar] [CrossRef] [PubMed]
- Jester, J.V.; Barry, P.A.; Lind, G.J.; Petroll, W.M.; Garana, R.; Cavanagh, H.D. Corneal keratocytes: in situ and in vitro organization of cytoskeletal contractile proteins. Invest. Ophthalmol. Vis. Sci. 1994, 35, 730–743. [Google Scholar] [PubMed]
- Fini, M.E. Keratocyte and fibroblast phenotypes in the repairing cornea. Prog. Retin. Eye Res. 1999, 18, 529–551. [Google Scholar] [CrossRef] [PubMed]
- Zieske, J.D. Extracellular matrix and wound healing. Curr. Opin. Ophthalmol. 2001, 12, 237–241. [Google Scholar] [CrossRef] [PubMed]
- Chaurasia, S.S.; Perera, P.R.; Poh, R.; Lim, R.R.; Wong, T.T.; Mehta, J.S. Hevin plays a pivotal role in corneal wound healing. PLoS One. 2013, 8, e81544. [Google Scholar] [CrossRef] [PubMed]
- Tombran-Tink, J.; Chader, G.; Johnson, L. PEDF: A pigment epithelium-derived factor with potent neuronal differentiative activity. Exp. Eye Res. 1991, 53, 411–414. [Google Scholar] [CrossRef] [PubMed]
- Matsui, T.; Nishino, Y.; Maeda, S.; Yamagishi, S. PEDF-derived peptide inhibits corneal angiogenesis by suppressing VEGF expression. Microvasc. Res. 2012, 84, 105–108. [Google Scholar] [CrossRef] [PubMed]
- Ambati, B.K.; Nozaki, M.; Singh, N.; Takeda, A.; Jani, P.D.; Suthar, T.; Albuquerque, R.J.; Richter, E.; Sakurai, E.; Newcomb, M.T.; et al. Corneal avascularity is due to soluble VEGF receptor-1. Nature 2006, 443, 993–997. [Google Scholar] [CrossRef] [PubMed]
- Cursiefen, C.; Masli, S.; Ng, T.F.; Dana, M.R.; Bornstein, P.; Lawler, J.; Streilein, J.W. Roles of thrombospondin-1 and -2 in regulating corneal and iris angiogenesis. Invest. Ophthalmol. Vis. Sci. 2004, 45, 1117–1124. [Google Scholar] [CrossRef] [PubMed]
- Peek, R.; van Gelderen, B.E.; Bruinenberg, M.; Kijlstra, A. Molecular cloning of a new angiopoietinlike factor from the human cornea. Invest. Ophthalmol. Vis. Sci. 1998, 39, 1782–1788. [Google Scholar] [PubMed]
- Makino, Y.; Cao, R.; Svensson, K.; Bertilsson, G.; Asman, M.; Tanaka, H.; Cao, Y.; Berkenstam, A.; Poellinger, L. Inhibitory PAS domain protein is a negative regulator of hypoxia-inducible gene expression. Nature 2001, 414, 550–554. [Google Scholar] [CrossRef] [PubMed]
- Streilein, J.W. New thoughts on the immunology of corneal transplantation. Eye 2003, 17, 943–948. [Google Scholar] [CrossRef] [PubMed]
- Cursiefen, C. Immune privilege and angiogenic privilege of the cornea. Chem. Immunol. Allergy 2007, 92, 50–57. [Google Scholar] [PubMed]
- Azar, D.T. Corneal angiogenic privilege: Angiogenic and antiangiogenic factors in corneal avascularity, vasculogenesis, and wound healing (an American Ophthalmological Society thesis). Trans. Am. Ophthalmol. Soc. 2006, 104, 264–302. [Google Scholar] [PubMed]
- Chan, C.K.; Pham, L.N.; Zhou, J.; Spee, C.; Ryan, S.J.; Hinton, D.R. Differential expression of pro- and antiangiogenic factors in mouse strain- dependent hypoxia-induced retinal neovascularization. Lab. Invest. 2005, 85, 721–733. [Google Scholar] [CrossRef] [PubMed]
- Ogawa, S.; Yoshida, S.; Ono, M.; Onoue, H.; Ito, Y.; Ishibashi, T.; Inomata, H.; Kuwano, M. Induction of macrophage inflammatory protein-1alpha and vascular endothelial growth factor during inflammatory neovascularization in the mouse cornea. Angiogenesis 1999, 3, 327–334. [Google Scholar] [CrossRef] [PubMed]
- Li, Z.R.; Li, Y.P.; Lin, M.L.; Su, W.R.; Zhang, W.X.; Zhang, Y.; Yao, L.; Liang, D. Activated macrophages induce neovascularization through upregulation of MMP-9 and VEGF in rat corneas. Cornea 2012, 31, 1028–1035. [Google Scholar] [CrossRef] [PubMed]
- Chang, J.H.; Gabison, E.E.; Kato, T.; Azar, D.T. Corneal neovascularization. Curr. Opin. Ophthalmol. 2001, 12, 242–249. [Google Scholar] [CrossRef] [PubMed]
- Mirabelli, P.; Peebo, B.B.; Xeroudaki, M.; Koulikovska, M.; Lagali, N. Early effects of dexamethasone and anti-VEGF therapy in an inflammatory corneal neovascularization model. Exp. Eye Res. 2014, 125, 118–127. [Google Scholar] [CrossRef] [PubMed]
- Shakiba, Y.; Mansouri, K.; Arshadi, D.; Rezaei, N. Corneal neovascularization: molecular events and therapeutic options. Recent Pat. Inflamm. Allergy Drug Discov. 2009, 3, 221–231. [Google Scholar] [CrossRef] [PubMed]
- Cai, X.; Conley, S.; Naash, M. Nanoparticle applications in ocular gene therapy. Vision Res. 2008, 48, 319–324. [Google Scholar] [CrossRef] [PubMed]
- Pissuwan, D.; Niidome, T.; Cortie, M.B. The forthcoming applications of gold nanoparticles in drug and gene delivery systems. J. Control. Release. 2011, 149, 65–71. [Google Scholar] [CrossRef] [PubMed]
- Ghosh, P.S.; Kim, C.K.; Han, G.; Forbes, N.S.; Rotello, V.M. Efficient gene delivery vectors by tuning the surface charge density of amino acid-functionalized gold nanoparticles. ACS Nano. 2008, 2, 2213–2218. [Google Scholar] [CrossRef] [PubMed]
- Zhou, X.; Zhang, X.; Yu, X.; Zha, X.; Fu, Q.; Liu, B.; Wang, X.; Chen, Y.; Shan, Y.; Jin, Y.; et al. The effect of conjugation to gold nanoparticles on the ability of low molecular weight chitosan to transfer DNA vaccine. Biomaterials 2008, 29, 111–117. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.; Takahashi, M.; Shimizu, T.; Shirasawa, T.; Kajita, M.; Kanayama, A.; Miyamoto, Y. Effects of a potent antioxidant, platinum nanoparticle, on the lifespan of Caenorhabditis elegans. Mech. Ageing Dev. 2008, 129, 322–331. [Google Scholar] [CrossRef] [PubMed]
- Nagarwal, R.C.; Singh, P.N.; Kant, S.; Maiti, P.; Pandit, J.K. Chitosan nanoparticles of 5-fluorouracil for ophthalmic delivery: Characterization, in vitro and in vivo study. Chem. Pharm. Bull. 2011, 59, 272–278. [Google Scholar] [CrossRef] [PubMed]
- Jain, G.K.; Pathan, S.A.; Akhter, S.; Jayabalan, N.; Talegaonkar, S.; Khar, R.K.; Ahmad, F.J. Microscopic and spectroscopic evaluation of novel PLGA-chitosan Nanoplexes as an ocular delivery system. Colloids Surf. B Biointerfaces 2011, 82, 397–403. [Google Scholar] [CrossRef] [PubMed]
- De la Fuente, M.; Seijo, B.; Alonso, M.J. Bioadhesive hyaluronan-chitosan nanoparticles can transport genes across the ocular mucosa and transfect ocular tissue. Gene Ther. 2008, 15, 668–676. [Google Scholar] [CrossRef] [PubMed]
- Contreras-Ruiz, L.; de la Fuente, M.; Párraga, J.E.; López-García, A.; Fernández, I.; Seijo, B.; Sánchez, A.; Calonge, M.; Diebold, Y. Intracellular trafficking of hyaluronic acid-chitosan oligomer-based nanoparticles in cultured human ocular surface cells. Mol. Vis. 2011, 17, 279–290. [Google Scholar] [PubMed]
- Ludwing, A. The use of mucoadhesive polymers in ocular drug delivery. Adv. Drug Deliv. Rev. 2005, 57, 1595–1693. [Google Scholar] [CrossRef] [PubMed]
- Hu, J.; Kovtun, A.; Tomaszewski, A.; Singer, B.B.; Seitz, B.; Epple, M.; Steuhl, K.P.; Ergün, S.; Fuchsluger, T.A. A new tool for the transfection of corneal endothelial cells: calcium phosphate nanoparticles. Acta. Biomater. 2012, 8, 1156–1163. [Google Scholar] [CrossRef] [PubMed]
- Ellis-Behnke, R.G.; Liang, Y.X.; You, S.W.; Tay, D.K.; Zhang, S.; So, K.F.; Schneider, G.E. Nano neuro knitting: Peptide nanofiber scaffold for brain repair and axon regeneration with functional return of vision. Proc. Natl. Acad. Sci. USA 2006, 103, 5054–5059. [Google Scholar] [CrossRef] [PubMed]
- Ellenberg, D.; Shi, J.; Jain, S.; Chang, J.H.; Ripps, H.; Brady, S.; Melhem, E.R.; Lakkis, F.; Adamis, A.; Chen, D.F.; et al. Impediments to eye transplantation: ocular viability following optic-nerve transection or enucleation. Br. J. Ophthalmol. 2009, 93, 1134–1140. [Google Scholar] [CrossRef] [PubMed]
- Yang, J.M.; Yamato, M.; Nishida, K.; Ohki, T.; Kanzaki, M.; Sekine, H.; Shimizu, T.; Okano, T. Cell delivery in regenerative medicine: the cell sheet engineering approach. J. Control. Release 2006, 116, 193–203. [Google Scholar] [CrossRef] [PubMed]
- Ma, X.Y.; Bao, H.J.; Cui, L.; Zou, J. The graft of autologous adipose-derived stem cells in the corneal stromal after mechanic damage. PLoS One 2013, 8, e76103. [Google Scholar] [CrossRef] [PubMed]
- Duan, X.; Sheardown, H. Dendrimer crosslinked collagen as a corneal tissue engineering scaffold: Mechanical properties and corneal epithelial cell interactions. Biomaterials 2006, 27, 4608–4617. [Google Scholar] [CrossRef] [PubMed]
- Peer, D.; Karp, J.M.; Hong, S.; Farokhzad, O.C.; Margalit, R.; Langer, R. Nanocarriers as an emerging platform for cancer therapy. Nat. Nanotechnol. 2007, 2, 751–760. [Google Scholar] [CrossRef] [PubMed]
- McCarthy, T.D.; Karellas, P.; Henderson, S.A.; Giannis, M.; O’Keefe, D.F.; Heery, G.; Paull, J.R.; Matthews, B.R.; Holan, G. Dendrimers as drugs: Discovery and preclinical and clinical development of dendrimer-based microbicides for HIV and STI prevention. Mol. Pharm. 2005, 2, 312–318. [Google Scholar] [CrossRef] [PubMed]
- Davis, M.E.; Zuckerman, J.E.; Choi, C.H.J.; Seligson, D.; Tolcher, A.; Alabi, C.A.; Yen, Y.; Heidel, J.D.; Ribas, A. Evidence of RNAi in humans from systemically administered siRNA via targeted nanoparticles. Nature 2010, 464, 1067–1070. [Google Scholar] [CrossRef] [PubMed]
- Resch-Genger, U.; Grabolle, M.; Cavaliere-Jaricot, S.; Nitschke, R.; Nann, T. Quantum dots versus organic dyes as fluorescent labels. Nat. Methods 2008, 5, 763–775. [Google Scholar] [CrossRef] [PubMed]
- Sperling, R.A.; Gil, P.R.; Zhang, F.; Zanella, M.; Parak, W.J. Biological applications of gold nanoparticles. Chem. Soc. Rev. 2008, 37, 1896–1908. [Google Scholar] [CrossRef] [PubMed]
- Liu, Z.A.; Li, X.L.; Tabakman, S.M.; Jiang, K.L.; Fan, S.S.; Dai, H.J. Multiplexed multicolor Raman imaging of live cells with isotopically modified single walled carbon nanotubes. J. Am. Chem. Soc. 2008, 130, 13540–13541. [Google Scholar] [CrossRef] [PubMed]
- Xu, J.; Li, X.; Sun, F. Preparation and evaluation of a contact lens vehicle for puerarin delivery. J. Biomater. Sci. Polym. Ed. 2010, 21, 271–288. [Google Scholar] [CrossRef] [PubMed]
- Peng, C.C.; Chauhan, A.J. Extended cyclosporine delivery by silicone-hydrogel contact lenses. J. Control. Release 2011, 154, 267–274. [Google Scholar] [CrossRef]
- Garhwal, R.; Shady, S.F.; Ellis, E.J.; Ellis, J.Y.; Leahy, C.C.; McCarthy, S.P.; Crawford, K.S.; Gaines, P. Sustained ocular delivery of ciprofloxacin using nanospheres and conventional contact lens materials. Invest. Ophthalmol. Vis. Sci. 2012, 53, 1341–1352. [Google Scholar] [CrossRef] [PubMed]
- Hiratani, H.; Fujiwara, A.; Tamiya, Y.; Mizutani, Y.; Alvarez-Lorenzo, C. Ocular release of timolol from molecularly imprinted soft contact lenses. Biomaterials 2005, 26, 1293–1298. [Google Scholar] [CrossRef] [PubMed]
- Alvarez-Lorenzo, C.; Yañez, F.; Barreiro-Iglesias, R.; Concheiro, A. Imprinted soft contact lenses as norfloxacin delivery systems. J. Control. Release 2006, 113, 236–244. [Google Scholar] [CrossRef] [PubMed]
- Tieppo, A.; Pate, K.M.; Byrne, M.E. In vitro controlled release of an anti-inflammatory from daily disposable therapeutic contact lenses under physiological ocular tear flow. Eur. J. Pharm. Biopharm. 2012, 81, 170–177. [Google Scholar] [CrossRef] [PubMed]
- Von der Mark, K.; Park, J.; Bauer, S.; Schmuki, P. Nanoscale engineering of biomimetic surfaces: Cues from the extracellular matrix. Cell Tissue Res. 2010, 339, 131–153. [Google Scholar] [CrossRef] [PubMed]
- Ahmed, R.Z.; Patil, G.; Zaheer, Z. Nanosponges—A completely new nano-horizon: Pharmaceutical applications and recent advances. Drug Dev. Ind. Pharm. 2013, 39, 1263–1272. [Google Scholar] [CrossRef] [PubMed]
- Swaminathan, S.; Vavia, P.R.; Trotta, F.; Cavalli, R. Nanosponges encapsulating dexamethasone for ocular delivery: Formulation design, physicochemical characterization, safety and corneal permeability assessment. J. Biomed. Nanotechnol. 2013, 9, 998–1007. [Google Scholar] [CrossRef] [PubMed]
- Saito, R.; Dresselhaus, G.; Dresselhaus, M.S. Physical Properties of Carbon Nanotubes; Imperial College Press: London, UK, 1998. [Google Scholar]
- Ajayan, P.M.; Zhou, O.Z. Applications of carbon nanotubes. In Topics in Applied Physics, Carbon Nanotubes; Dresselhaus, M., Dresselhaus, G., Avouris, P., Eds.; Springer-Verlag GmbH: Heidelberg, Germany, 2001; Volume 80, pp. 391–425. [Google Scholar]
- Wong, B.S.; Yoong, S.L.; Jagusiak, A.; Panczyk, T.; Ho, H.K.; Ang, W.H.; Pastorin, G. Carbon nanotubes for delivery of small molecule drugs. Adv. Drug Deliv. Rev. 2013, 65, 1964–2015. [Google Scholar] [CrossRef] [PubMed]
- Souza, J.G.; Dias, K.; Pereira, T.A.; Bernardi, D.S.; Lopez, R.F. Topical delivery of ocular therapeutics: Carrier systems and physical methods. J. Pharm. Pharmacol. 2014, 66, 507–530. [Google Scholar] [CrossRef] [PubMed]
- Gulsen, D.; Chauhan, A. Dispersion of microemulsion drops in HEMA hydrogel: A potential ophthalmic drug delivery vehicle. Int. J. Pharm. 2005, 292, 95–117. [Google Scholar] [CrossRef] [PubMed]
- Müller, R.H.; Jacobs, C.; Kayser, O. Nanosuspensions as particulate drug formulations in therapy. Rationale for development and what we can expect for the future. Adv. Drug Deliv. Rev. 2001, 47, 3–19. [Google Scholar] [CrossRef] [PubMed]
- Kayser, O.; Lemke, A.; Hernández-Trejo, N. The impact of nanobiotechnology on the development of new drug delivery systems. Curr. Pharm. Biotechnol. 2005, 6, 3–5. [Google Scholar] [PubMed]
- Sahoo, S.K.; Dilnawaz, F.; Krishnakumar, S. Nanotechnology in ocular drug delivery. Drug. Discov. Today 2008, 13, 144–151. [Google Scholar] [CrossRef] [PubMed]
- El-Sousi, S.; Nácher, A.; Mura, C.; Catalán-Latorre, A.; Merino, V.; Merino-Sanjuán, M.; Díez-Sales, O. Hydroxypropylmethylcellulose films for the ophthalmic delivery of diclofenac sodium. J. Pharm. Pharmacol. 2013, 65, 193–200. [Google Scholar] [CrossRef] [PubMed]
- Ticho, U.; Blementhal, M.; Zonis, S.; Gal, A.; Blank, I.; Mazor, Z.W. Piloplex, a new long-acting pilocarpine polymer salt. A: Long-term study. Br. J. Ophthalmol. 1979, 63, 45–47. [Google Scholar] [CrossRef] [PubMed]
- Kompella, U.B.; Sundaram, S.; Raghava, S.; Escobar, E.R. Luteinizing hormone-releasing hormone agonist and transferri functionalizations enhance nanoparticle delivery in a novel bovine ex vivo eye model. Mol. Vis. 2006, 12, 1185–1198. [Google Scholar] [PubMed]
- Jwala, J.; Boddu, S.H.; Shah, S.; Sirimulla, S.; Pal, D.; Mitra, A.K. Ocular sustained release nanoparticles containing stereoisomeric dipeptide prodrugs of acyclovir. J. Ocul. Pharmacol. Ther. 2011, 27, 163–172. [Google Scholar] [CrossRef] [PubMed]
- Giannavola, C.; Bucolo, C.; Maltese, A.; Paolino, D.; Vandelli, M.A.; Puglisi, G.; Lee, V.H.L.; Fresta, M. Influence of preparation conditions on acyclovir-loaded poly-D,L-lactic acid nanospheres and effect of PEG coating on ocular drug bioavailability. Pharm. Res. 2003, 20, 584–590. [Google Scholar] [CrossRef] [PubMed]
- Qaddoumi, M.G.; Ueda, H.; Yang, J.; Davda, J.; Labhasetwar, V.; Lee, V.H.L. The characteristics and mechanisms of uptake of PLGA nanoparticles in rabbit conjunctival epithelial cell layers. Pharm. Res. 2004, 21, 641–648. [Google Scholar] [CrossRef] [PubMed]
- Marchal-Heussler, L.; Sirbat, D.; Hoffman, M.; Maincent, P. Poly(epsilon-caprolactone) nanocapsules in carteolol ophthalmic delivery. Pharm. Res. 1993, 10, 386–390. [Google Scholar] [CrossRef] [PubMed]
- Qazi, Y.; Stagg, B.; Singh, N.; Singh, S.; Zhang, X.; Luo, L.; Simonis, J.; Kompella, U.B.; Ambati, B.K. Nanoparticle-mediated delivery of shRNA.VEGF-A plasmids regresses corneal neovascularization. Invest. Ophthalmol. Vis. Sci. 2012, 53, 2837–2844. [Google Scholar] [CrossRef] [PubMed]
- Araújo, L.M.; Thomazine, J.A.; Lopez, R.F. Development of microemulsions to topically deliver 5-aminolevulinic acid in photo-dynamic therapy. Eur. J. Pharm. Biopharm. 2010, 75, 48–55. [Google Scholar] [CrossRef] [PubMed]
- Smolin, G.; Okumoto, M.; Feiler, S.; Condon, D. Iodoxuridine-liposome therapy for herpes simplex keratitis. Am. J. Ophthalmol. 1981, 91, 220–226. [Google Scholar] [CrossRef] [PubMed]
- White, C.J.; McBride, M.K.; Pate, K.M.; Tieppo, A.; Byrne, M.E. Extended release of high molecular weight hydroxypropyl methylcellulose from molecularly imprinted, extended wear silicone hydrogel contact lenses. Biomaterials 2011, 32, 5698–5705. [Google Scholar] [CrossRef] [PubMed]
- Benita, S.; Plenecassagne, J.D.; Cavé, G.; Drouin, D.; Le Hao Dong, P.; Sincholle, D. Pilocarpine hydrochloride liposomes: Characterization in vitro and preliminary evaluation in vivo in rabbit eye. J. Microencapsul. 1984, 1, 203–216. [Google Scholar] [CrossRef] [PubMed]
- Singh, K.; Mezei, M. Liposomal ophthalmic drug delivery system. I. Triamcinalone acetonide. Int. J. Pharm. 1983, 16, 339–344. [Google Scholar] [CrossRef]
- Singh, K.; Mezei, M. Liposomal ophthalmic drug delivery system. II. Dihydrostreptomycin sulphate. Int. J. Pharm. 1984, 19, 263–269. [Google Scholar] [CrossRef]
- Navath, R.S.; Kurtoglu, Y.E.; Wang, B.; Kannan, S.; Romero, R.; Kannan, R.M. Dendrimer-drug conjugates for tailored intracellular drug release based on glutathione levels. Bioconjug. Chem. 2008, 19, 2446–2455. [Google Scholar] [CrossRef] [PubMed]
- Antoni, P.; Hed, Y.; Nordberg, A.; Nyström, D.; von Holst, H.; Hult, A.; Malkoch, M. Bifunctional dendrimers: From robust synthesis and accelerated one-pot postfunctionalization strategy to potential applications. Angew. Chem. Int. Ed. Engl. 2009, 48, 2126–2130. [Google Scholar] [CrossRef] [PubMed]
- Khandare, J.; Kolhe, P.; Pillai, O.; Kannan, S.; Lieh-Lai, M.; Kannan, R.M. Synthesis, cellular transport, and activity of polyamidoamine dendrimer-methylprednisolone conjugates. Bioconjug. Chem. 2005, 16, 330–337. [Google Scholar] [CrossRef] [PubMed]
- Mintzer, M.A.; Grinstaff, M.W. Biomedical applications of dendrimers: A tutorial. Chem. Soc. Rev. 2011, 40, 173–190. [Google Scholar] [CrossRef] [PubMed]
- Liu, M.; Fréchet, J.M. Designing dendrimers for drug delivery. Pharm. Sci. Technolo. Today 1999, 2, 393–401. [Google Scholar] [CrossRef] [PubMed]
- Tomalia, D.A.; Baker, H.; Dewald, J.; Hall, M.; Kallos, G.; Martin, S.; Roeck, J.; Ryder, J.; Smith, P. A new class of polymers: starburst dendritic macromolecules. Polym. J. 1985, 17, 117–132. [Google Scholar] [CrossRef]
- Kolhe, P.; Misra, E.; Kannan, R.M.; Kannan, S.; Lieh-Lai, M. Drug complexation, in vitro release and cellular entry of dendrimers and hyperbranched polymers. Int. J. Pharm. 2003, 259, 143–160. [Google Scholar] [CrossRef] [PubMed]
- Souza, J.G.; Dias, K.; Silva, S.A.; de Rezende, L.C.; Rocha, E.M.; Emery, F.S.; Lopez, R.F. Transcorneal iontophoresis of dendrimers: PAMAM corneal penetration and dexamethasone delivery. J. Control. Release 2015, 200, 115–124. [Google Scholar] [CrossRef] [PubMed]
- Calabretta, M.K.; Kumar, A.; McDermott, A.M.; Cai, C. Antibacterial activities of poly(amidoamine) dendrimers terminated with amino and poly(ethylene glycol) groups. Biomacromolecules 2007, 8, 1807–1811. [Google Scholar] [CrossRef] [PubMed]
- Calvo, P.; Alonso, M.J.; Vila-Jato, J.L.; Robinson, J.R. Improved ocular bioavailability of indomethacin by novel ocular drug carriers. J. Pharm. Pharmacol. 1996, 48, 1147–1152. [Google Scholar] [CrossRef] [PubMed]
- De Campos, A.M.; Sánchez, A.; Gref, R.; Calvo, P.; Alonso, M.J. The effect of a PEG versus a chitosan coating on the interaction of drug colloidal carriers with the ocular mucosa. Eur. J. Pharm. Sci. 2003, 20, 73–81. [Google Scholar] [CrossRef] [PubMed]
- Järvinen, K.; Järvinen, T.; Urtti, A. Ocular absorption following topical delivery. Adv. Drug Deliv. Rev. 1995, 16, 3–19. [Google Scholar] [CrossRef]
- Di Tommaso, C.; Bourges, J.L.; Valamanesh, F.; Trubitsyn, G.; Torriglia, A.; Jeanny, J.C.; Behar-Cohen, F.; Gurny, R.; Möller, M. Novel micelle carriers for cyclosporin A topical ocular delivery: in vivo corneal penetration, ocular distribution and efficacy studies. Eur. J. Pharm. Biopharm. 2012, 81, 257–264. [Google Scholar] [CrossRef] [PubMed]
- Tadros, T.; Izquierdo, P.; Esquena, J.; Solans, C. Formation and stability of nanoemulsions. Adv. Colloid Interface Sci. 2004, 108–109, 303–318. [Google Scholar]
- Date, A.A.; Desai, N.; Dixit, R.; Nagarsenker, M. Self-nanoemulsifying drug delivery systems: formulation insights, applications and advances. Nanomedicine (Lond) 2010, 5, 1595–1616. [Google Scholar] [CrossRef]
- Solans, C.; Esquena, J.; Forgiarini, A.M.; Uson, N.; Morales, D.; Izquierdo, P.; Azemar, N. Nano-emulsions: Formation, properties, and applications. In Adsorption and Aggregation of Surfactants in Solution; Mittal, K.L., Shah, D.O., Eds.; Marcel Dekker, Inc.: New York, NY, USA, 2002; Volume 109, pp. 524–554. [Google Scholar]
- Anton, N.; Benoit, J.P.; Saulnier, P. Design and production of nanoparticles formulated from nano-emulsion templates—A review. J. Control. Release 2008, 128, 185–199. [Google Scholar] [CrossRef] [PubMed]
- Calvo, P.; Vila-Jato, J.L.; Alonso, M.J. Comparative in vitro evaluation of several colloidal systems, nanoparticles, nanocapsules, and nanoemulsions, as ocular drug carriers. J. Pharm. Sci. 1996, 85, 530–536. [Google Scholar] [CrossRef] [PubMed]
- Erdem, E.; Kandemir, H.; Arıkan-Akdağlı, S.; Esen, E.; Açıkalın, A.; Yağmur, M.; Ilkit, M. Aspergillus terreus infection in a sutureless self-sealing incision made during cataract surgery. Mycopathologia 2015, 179, 129–134. [Google Scholar] [CrossRef] [PubMed]
- Habib, F.S.; Fouad, E.A.; Abdel-Rhaman, M.S.; Fathalla, D. Liposomes as an ocular delivery system of fluconazole: In vitro studies. Acta. Ophthalmol. 2010, 88, 901–904. [Google Scholar] [CrossRef] [PubMed]
- Frucht-Perry, J.; Assil, K.K.; Ziegler, E.; Douglas, H.; Brown, S.I.; Schanzlin, D.J.; Weinreb, R.N. Fibrin-enmeshed tobramycin liposomes: single application topical therapy of Pseudomonas keratitis. Cornea 1992, 11, 393–397. [Google Scholar] [CrossRef] [PubMed]
- Cavalli, R.; Gasco, M.R.; Chetoni, P.; Burgalassi, S.; Saettone, M.F. Solid lipid nanoparticles (SLN) as ocular delivery system for tobramycin. Int. J. Pharm. 2002, 238, 241–245. [Google Scholar] [CrossRef] [PubMed]
- Barza, M.; Baum, J.; Szoka, F., Jr. Pharmacokinetics of subconjunctival liposome-encapsulated gentamicin in normal rabbit eyes. Invest. Ophthalmol. Vis. Sci. 1984, 25, 486–490. [Google Scholar] [PubMed]
- Taha, E.I.; El-Anazi, M.H.; El-Bagory, I.M.; Bayomi, M.A. Design of liposomal colloidal systems for ocular delivery of ciprofloxacin. Saudi. Pharm. J. 2014, 22, 231–239. [Google Scholar] [CrossRef] [PubMed]
- Hosny, K.M. Ciprofloxacin as ocular liposomal hydrogel. AAPS PharmSciTech. 2010, 11, 241–246. [Google Scholar] [CrossRef] [PubMed]
- Jain, R.L.; Shastri, J.P. Study of ocular drug delivery system using drug-loaded liposomes. Int. J. Pharm. Investig. 2011, 1, 35–41. [Google Scholar] [CrossRef] [PubMed]
- Lin, H.H.; Ko, S.M.; Hsu, L.R.; Tsai, Y.H. The preparation of norfloxacin-loaded liposomes and their in vitro evaluation in pig’s eye. J. Pharm. Pharmacol. 1996, 48, 801–805. [Google Scholar] [CrossRef] [PubMed]
- Sumer, B.; Gao, J. Theranostic nanomedicine for cancer. Nanomedicine 2008, 3, 137–140. [Google Scholar] [CrossRef] [PubMed]
- Zarbin, M.A.; Montemagno, C.; Leary, J.F.; Ritch, R. Nanomedicine in ophthalmology: The new frontier. Am. J. Ophthalmol. 2010, 150, 144–162. [Google Scholar] [CrossRef] [PubMed]
© 2015 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chaurasia, S.S.; Lim, R.R.; Lakshminarayanan, R.; Mohan, R.R. Nanomedicine Approaches for Corneal Diseases. J. Funct. Biomater. 2015, 6, 277-298. https://doi.org/10.3390/jfb6020277
Chaurasia SS, Lim RR, Lakshminarayanan R, Mohan RR. Nanomedicine Approaches for Corneal Diseases. Journal of Functional Biomaterials. 2015; 6(2):277-298. https://doi.org/10.3390/jfb6020277
Chicago/Turabian StyleChaurasia, Shyam S., Rayne R. Lim, Rajamani Lakshminarayanan, and Rajiv R. Mohan. 2015. "Nanomedicine Approaches for Corneal Diseases" Journal of Functional Biomaterials 6, no. 2: 277-298. https://doi.org/10.3390/jfb6020277
APA StyleChaurasia, S. S., Lim, R. R., Lakshminarayanan, R., & Mohan, R. R. (2015). Nanomedicine Approaches for Corneal Diseases. Journal of Functional Biomaterials, 6(2), 277-298. https://doi.org/10.3390/jfb6020277







