A Critical Review of Commercial Collagen-Based Scaffolds in Bone Regeneration: Functional Properties and Clinical Evidence from Infuse® Bone Graft
Abstract
1. Introduction
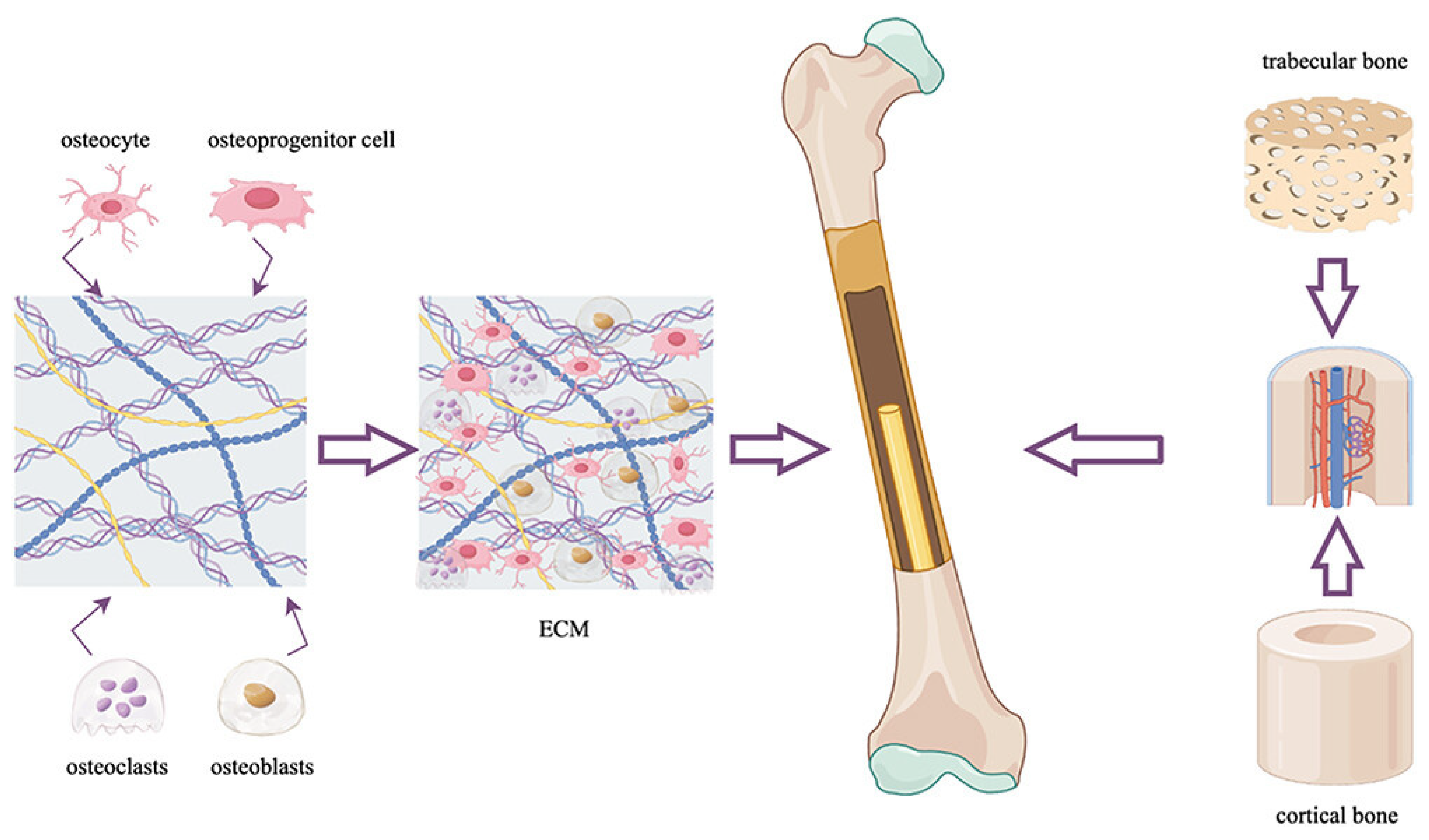
2. Composition Considerations and Commercial Advances
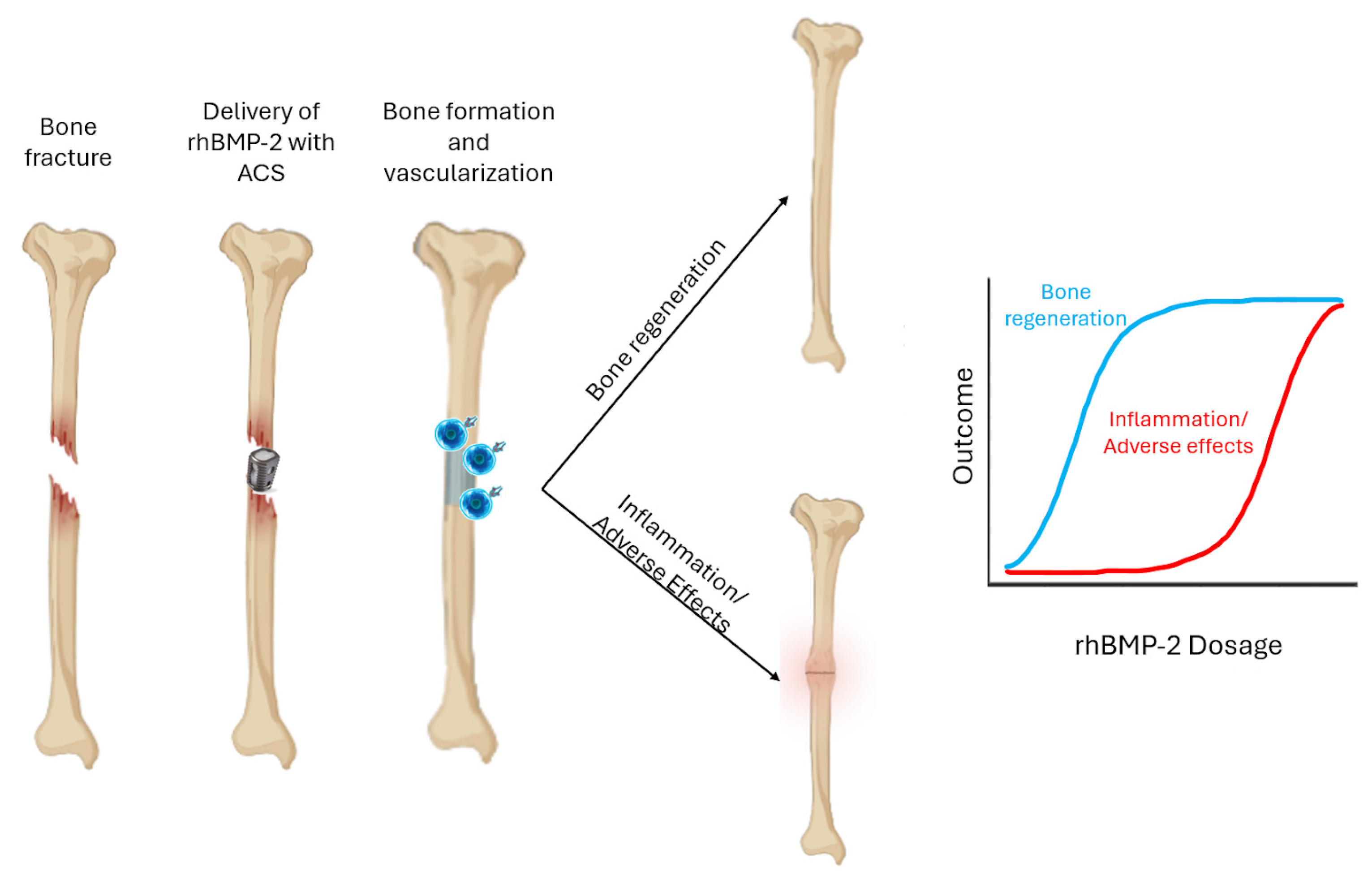
3. Tissue-Level Response to rhBMP-2-Based Bone Grafts
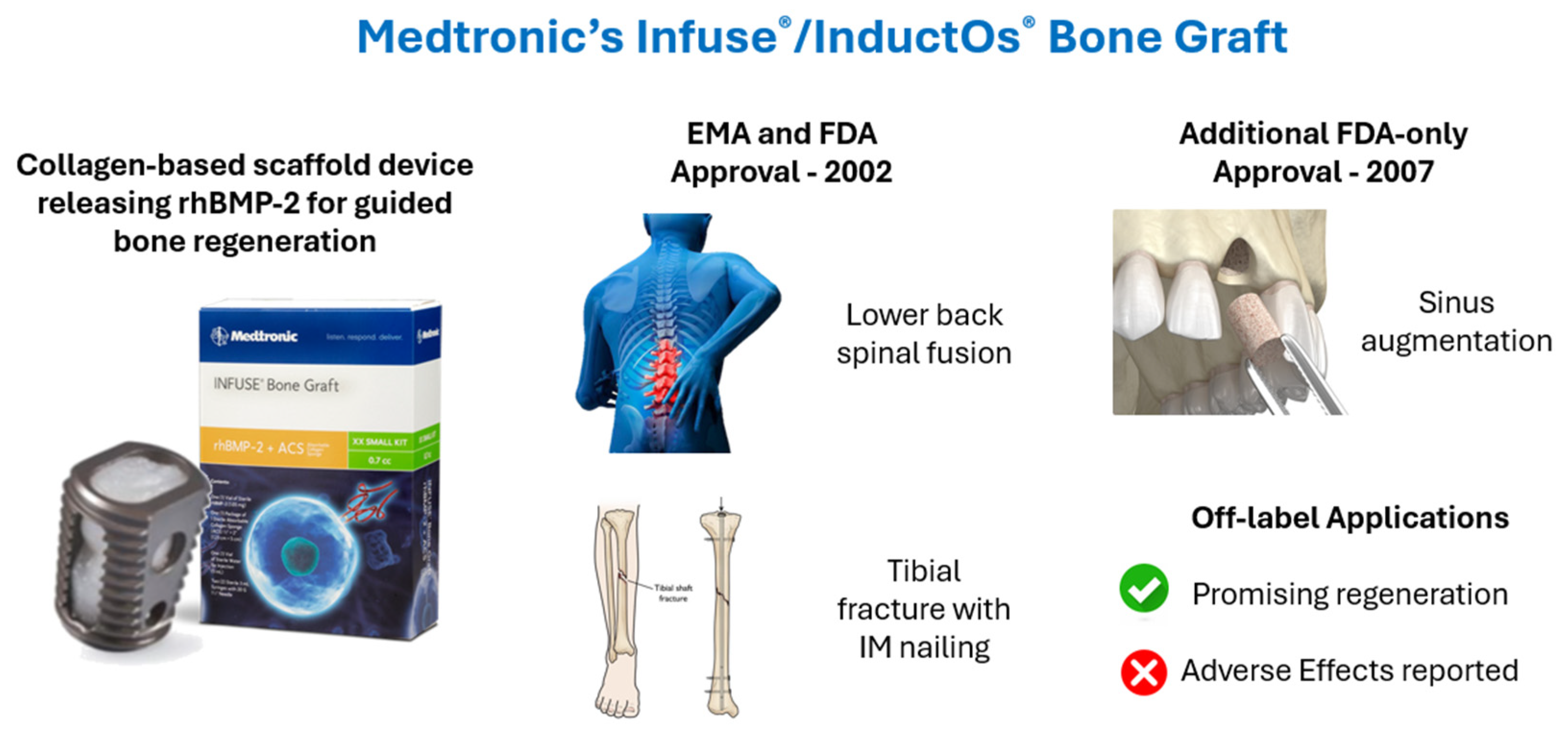
4. Approved Clinical Applications
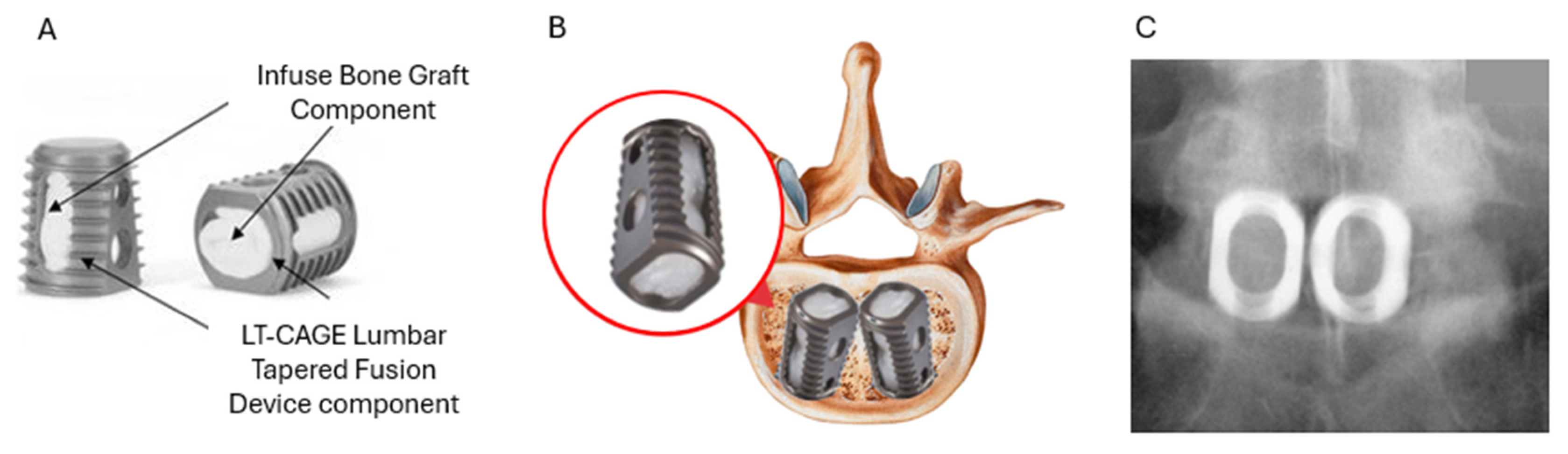
4.1. Spinal Fusion
4.2. Open Tibial Fracture
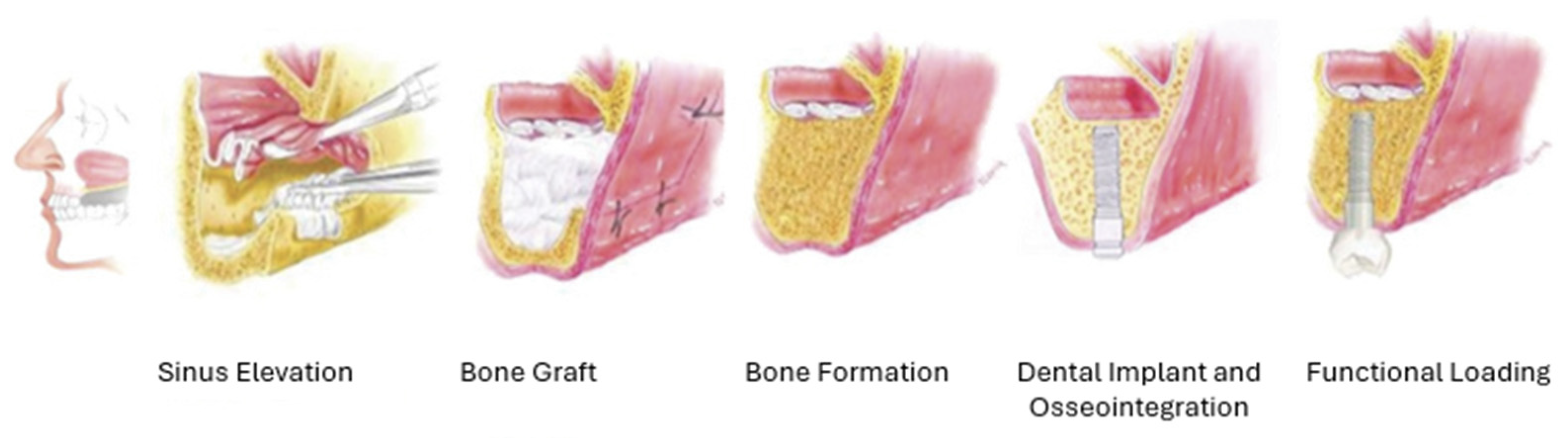
4.3. Maxillary Augmentation
5. Cost
6. Future Perspectives
6.1. Spinal Fusion
6.2. Open Tibial Fracture
6.3. Maxillary Augmentation
7. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| rhBMP-2 | Recombinant human BMP-2 |
| ECM | Extracellular Matrix |
| ACS | Absorbable Collagen Sponge |
| TLIF | Transforaminal Lumbar Interbody Fusion |
| BESTT | BMP-2 Evaluation in Surgery for Tibial Trauma |
| METRC | Major Extremity Trauma Research Center |
| ODI | Oswestry Disability Index |
| GTR | Guided Tissue Regeneration |
| GBR | Guided Bone Regeneration |
| VEGF | Vascular endothelial growth factor |
| HO | Heterotopic Ossification |
| ICBG | Iliac Crest Bone Graft |
| PLF | Posterolateral Fusion |
| PLIF | Posterior Lumbar Interbody Fusion |
| ALIF | Anterior Lumbar Interbody Fusion |
| CT | Computed Tomography |
| MIS | Minimally Invasive Surgery |
References
- Berthiaume, T.J.; Yarmush, M.L. Tissue Engineering and Regenerative Medicine: History, Progress, and Challenges. Annu. Rev. Chem. Biomol. Eng. 2011, 2, 403–430. [Google Scholar] [CrossRef]
- Eltom, A.; Zhong, G.; Muhammad, A. Scaffold Techniques and Designs in Tissue Engineering Functions and Purposes: A Review. Adv. Mater. Sci. Eng. 2019, 2019, 3429527. [Google Scholar] [CrossRef]
- Schmalz, G.; Widbiller, M.; Galler, K. Material Tissue Interaction—From Toxicity to Tissue Regeneration. Oper. Dent. 2016, 41, 117–131. [Google Scholar] [CrossRef]
- Gautschi, O.P.; Schatlo, B.; Schaller, K.; Tessitore, E. Clinically Relevant Complications Related to Pedicle Screw Placement in Thoracolumbar Surgery and Their Management: A Literature Review of 35,630 Pedicle Screws. Neurosurg. Focus 2011, 31, E8. [Google Scholar] [CrossRef] [PubMed]
- Ramos, D.M.; Dhandapani, R.; Subramanian, A.; Sethuraman, S.; Kumbar, S.G. Clinical Complications of Biodegradable Screws for Ligament Injuries. Mater. Sci. Eng. C 2020, 109, 110423. [Google Scholar] [CrossRef] [PubMed]
- Schepers, T.; Van Lieshout, E.M.M.; De Vries, M.R.; Van Der Elst, M. Complications of Syndesmotic Screw Removal. Foot Ankle Int. 2011, 32, 1040–1044. [Google Scholar] [CrossRef] [PubMed]
- Francis, A.P.; Augustus, A.R.; Chandramohan, S.; Bhat, S.A.; Priya, V.V.; Rajagopalan, R. A Review on Biomaterials-Based Scaffold: An Emerging Tool for Bone Tissue Engineering. Mater. Today Commun. 2023, 34, 105124. [Google Scholar] [CrossRef]
- Kavitha Sri, A.; Arthi, C.; Neya, N.R.; Hikku, G.S. Nano-Hydroxyapatite/Collagen Composite as Scaffold Material for Bone Regeneration. Biomed. Mater. 2023, 18, 032002. [Google Scholar] [CrossRef]
- Geevarghese, R.; Sajjadi, S.S.; Hudecki, A.; Sajjadi, S.; Jalal, N.R.; Madrakian, T.; Ahmadi, M.; Włodarczyk-Biegun, M.K.; Ghavami, S.; Likus, W.; et al. Biodegradable and Non-Biodegradable Biomaterials and Their Effect on Cell Differentiation. Int. J. Mol. Sci. 2022, 23, 16185. [Google Scholar] [CrossRef]
- Dias, J.R.; Ribeiro, N.; Baptista-Silva, S.; Costa-Pinto, A.R.; Alves, N.; Oliveira, A.L. In Situ Enabling Approaches for Tissue Regeneration: Current Challenges and New Developments. Front. Bioeng. Biotechnol. 2020, 8, 85. [Google Scholar] [CrossRef]
- Katrilaka, C.; Karipidou, N.; Petrou, N.; Manglaris, C.; Katrilakas, G.; Tzavellas, A.N.; Pitou, M.; Tsiridis, E.E.; Choli-Papadopoulou, T.; Aggeli, A. Freeze-Drying Process for the Fabrication of Collagen-Based Sponges as Medical Devices in Biomedical Engineering. Materials 2023, 16, 4425. [Google Scholar] [CrossRef]
- Amini, A.R.; Laurencin, C.T.; Nukavarapu, S.P. Bone Tissue Engineering: Recent Advances and Challenges. Crit. Rev. Biomed. Eng. 2012, 40, 363–408. [Google Scholar] [CrossRef]
- O’Keefe, R.J.; Mao, J. Bone Tissue Engineering and Regeneration: From Discovery to the Clinic—An Overview. Tissue Eng. Part B Rev. 2011, 17, 389–392. [Google Scholar] [CrossRef]
- Battafarano, G.; Rossi, M.; De Martino, V.; Marampon, F.; Borro, L.; Secinaro, A.; Del Fattore, A. Strategies for Bone Regeneration: From Graft to Tissue Engineering. Int. J. Mol. Sci. 2021, 22, 1128. [Google Scholar] [CrossRef]
- Zhang, J.; Zhang, W.; Yue, W.; Qin, W.; Zhao, Y.; Xu, G. Research Progress of Bone Grafting: A Comprehensive Review. Int. J. Nanomed. 2025, 20, 4729–4757. [Google Scholar] [CrossRef]
- Dimitriou, R.; Jones, E.; McGonagle, D.; Giannoudis, P.V. Bone Regeneration: Current Concepts and Future Directions. BMC Med. 2011, 9, 66. [Google Scholar] [CrossRef] [PubMed]
- Viola, A.; Appiah, J.; Donnally, C.J.; Kim, Y.H.; Shenoy, K. Bone Graft Options in Spinal Fusion: A Review of Current Options and the Use of Mesenchymal Cellular Bone Matrices. World Neurosurg. 2022, 158, 182–188. [Google Scholar] [CrossRef] [PubMed]
- Eleswarapu, A.; Rowan, F.A.; Le, H.; Wick, J.B.; Roberto, R.F.; Javidan, Y.; Klineberg, E.O. Efficacy, Cost, and Complications of Demineralized Bone Matrix in Instrumented Lumbar Fusion: Comparison With rhBMP-2. Glob. Spine J. 2021, 11, 1223–1229. [Google Scholar] [CrossRef] [PubMed]
- Cohen, J.D.; Kanim, L.E.; Trontis, A.J.; Bae, H.W. Allografts and Spinal Fusion. Int. J. Spine Surg. 2021, 15, 68–93. [Google Scholar] [CrossRef]
- Santoro, A.; Voto, A.; Fortino, L.; Guida, R.; Laudisio, C.; Cillo, M.; D’Ursi, A.M. Bone Defect Treatment in Regenerative Medicine: Exploring Natural and Synthetic Bone Substitutes. Int. J. Mol. Sci. 2025, 26, 3085. [Google Scholar] [CrossRef]
- Kim, K.D.; Batchelder, C.A.; Koleva, P.; Ghaffari-Rafi, A.; Karnati, T.; Goodrich, D.; Castillo, J.; Lee, C. In Vivo Performance of a Novel Hyper-Crosslinked Carbohydrate Polymer Bone Graft Substitute for Spinal Fusion. Bioengineering 2025, 12, 243. [Google Scholar] [CrossRef]
- De Pace, R.; Molinari, S.; Mazzoni, E.; Perale, G. Bone Regeneration: A Review of Current Treatment Strategies. J. Clin. Med. 2025, 14, 1838. [Google Scholar] [CrossRef] [PubMed]
- Kowalski, M.; Hasson, M.; Hammond, K.; Labib, S.; Patel, J.M. Biomaterials in Connective Tissue Regeneration and Rehabilitation. In Regenerative Rehabilitation; Greising, S.M., Call, J.A., Eds.; Physiology in Health and Disease; Springer International Publishing: Cham, Switzerland, 2022; pp. 291–330. ISBN 978-3-030-95883-1. [Google Scholar]
- Jeon, E.Y.; Joo, K.I.; Cha, H.J. Body Temperature-Activated Protein-Based Injectable Adhesive Hydrogel Incorporated with Decellularized Adipose Extracellular Matrix for Tissue-Specific Regenerative Stem Cell Therapy. Acta Biomater. 2020, 114, 244–255. [Google Scholar] [CrossRef] [PubMed]
- Burkus, J.K.; Heim, S.E.; Gornet, M.F.; Zdeblick, T.A. Is INFUSE Bone Graft Superior to Autograft Bone? An Integrated Analysis of Clinical Trials Using the LT-CAGE Lumbar Tapered Fusion Device. J. Spinal Disord. Tech. 2003, 16, 113–122. [Google Scholar] [CrossRef] [PubMed]
- Lykissas, M.; Gkiatas, I. Use of Recombinant Human Bone Morphogenetic Protein-2 in Spine Surgery. World J. Orthop. 2017, 8, 531. [Google Scholar] [CrossRef]
- Lisa, K. A Randomized Controlled Trial Comparing rhBMP-2/Absorbable Collagen Sponge Versus Autograft for the Treatment of Tibia Fractures With Critical Size Defects. J. Orthop. Trauma 2019, 33, 384–391. [Google Scholar] [CrossRef]
- Davies, R.P.W.; Aggeli, A. Self-assembly of Amphiphilic Β-sheet Peptide Tapes Based on Aliphatic Side Chains. J. Pept. Sci. 2011, 17, 107–114. [Google Scholar] [CrossRef]
- Koopmans, R.J.; Aggeli, A. Nanobiotechnology—Quo Vadis? Curr. Opin. Microbiol. 2010, 13, 327–334. [Google Scholar] [CrossRef]
- Maude, S.; Miles, D.E.; Felton, S.H.; Ingram, J.; Carrick, L.M.; Wilcox, R.K.; Ingham, E.; Aggeli, A. De Novo Designed Positively Charged Tape-Forming Peptides: Self-Assembly and Gelation in Physiological Solutions and Their Evaluation as 3D Matrices for Cell Growth. Soft Matter 2011, 7, 8085. [Google Scholar] [CrossRef]
- Theocharis, A.D.; Skandalis, S.S.; Gialeli, C.; Karamanos, N.K. Extracellular Matrix Structure. Adv. Drug Deliv. Rev. 2016, 97, 4–27. [Google Scholar] [CrossRef]
- Causa, F.; Netti, P.A.; Ambrosio, L. A Multi-Functional Scaffold for Tissue Regeneration: The Need to Engineer a Tissue Analogue. Biomaterials 2007, 28, 5093–5099. [Google Scholar] [CrossRef]
- Tassieri, M.; Waigh, T.A.; Trinick, J.; Aggeli, A.; Evans, R.M.L. Analysis of the Linear Viscoelasticity of Polyelectrolytes by Magnetic Microrheometry—Pulsed Creep Experiments and the One Particle Response. J. Rheol. 2010, 54, 117–131. [Google Scholar] [CrossRef]
- Kontakis, M.G.; Moulin, M.; Andersson, B.; Norein, N.; Samanta, A.; Stelzl, C.; Engberg, A.; Diez-Escudero, A.; Kreuger, J.; Hailer, N.P. Trabecular-Bone Mimicking Osteoconductive Collagen Scaffolds: An Optimized 3D Printing Approach Using Freeform Reversible Embedding of Suspended Hydrogels. 3D Print Med. 2025, 11, 11. [Google Scholar] [CrossRef]
- Polo-Corrales, L.; Latorre-Esteves, M.; Ramirez-Vick, J.E. Scaffold Design for Bone Regeneration. J. Nanosci. Nanotech. 2014, 14, 15–56. [Google Scholar] [CrossRef] [PubMed]
- Kisiel, M.; Klar, A.S.; Ventura, M.; Buijs, J.; Mafina, M.-K.; Cool, S.M.; Hilborn, J. Complexation and Sequestration of BMP-2 from an ECM Mimetic Hyaluronan Gel for Improved Bone Formation. PLoS ONE 2013, 8, e78551. [Google Scholar] [CrossRef] [PubMed]
- Malham, G.M.; Louie, P.K.; Brazenor, G.A.; Mobbs, R.J.; Walsh, W.R.; Sethi, R.K. Recombinant Human Bone Morphogenetic Protein-2 in Spine Surgery: Recommendations for Use and Alternative Bone Substitutes—A Narrative Review. J. Spine Surg. 2022, 8, 477–490. [Google Scholar] [CrossRef]
- Fan, L.; Ren, Y.; Emmert, S.; Vučković, I.; Stojanovic, S.; Najman, S.; Schnettler, R.; Barbeck, M.; Schenke-Layland, K.; Xiong, X. The Use of Collagen-Based Materials in Bone Tissue Engineering. Int. J. Mol. Sci. 2023, 24, 3744. [Google Scholar] [CrossRef]
- Gillman, C.E.; Jayasuriya, A.C. FDA-Approved Bone Grafts and Bone Graft Substitute Devices in Bone Regeneration. Mater. Sci. Eng. C 2021, 130, 112466. [Google Scholar] [CrossRef]
- Lyu, S.; Siegel, R.A. (Eds.) Drug-Device Combinations for Chronic Diseases; Wiley: Hoboken, NJ, USA, 2016; ISBN 978-1-118-12000-2. [Google Scholar]
- McKay, W.F.; Peckham, S.M.; Badura, J.M. A Comprehensive Clinical Review of Recombinant Human Bone Morphogenetic Protein-2 (INFUSE® Bone Graft). Int. Orthop. SICO 2007, 31, 729–734. [Google Scholar] [CrossRef]
- Ramly, E.P.; Alfonso, A.R.; Kantar, R.S.; Wang, M.M.; Siso, J.R.D.; Ibrahim, A.; Coelho, P.G.; Flores, R.L. Safety and Efficacy of Recombinant Human Bone Morphogenetic Protein-2 (rhBMP-2) in Craniofacial Surgery. Plast. Reconstr. Surg.—Glob. Open 2019, 7, e2347. [Google Scholar] [CrossRef]
- Jung, N.; Park, J.; Park, S.-H.; Oh, S.; Kim, S.; Cho, S.-W.; Kim, J.-E.; Moon, H.S.; Park, Y.-B. Improving Bone Formation by Guided Bone Regeneration Using a Collagen Membrane with rhBMP-2: A Novel Concept. J. Funct. Biomater. 2023, 14, 170. [Google Scholar] [CrossRef] [PubMed]
- Von Benecke, J.P.; Tarsitano, E.; Zimmermann, L.-M.A.; Shakesheff, K.M.; Walsh, W.R.; Bae, H.W. A Narrative Review on Recombinant Human Bone Morphogenetic Protein 2: Where Are We Now? Cureus 2024. [Google Scholar] [CrossRef] [PubMed]
- Wang, D.; Qi, G.; Zhang, M.; Carlson, B.; Gernon, M.; Burton, D.; Sun, X.S.; Wang, J. Peptide Hydrogel for Sustained Release of Recombinant Human Bone Morphogenetic Protein-2 In Vitro. J. Funct. Biomater. 2024, 15, 369. [Google Scholar] [CrossRef]
- Inductos. European Medicines Agency. Available online: https://www.ema.europa.eu/en/documents/overview/inductos-epar-summary-public_en.pdf (accessed on 4 July 2025).
- Medtronic. Available online: https://www.medtronic.com/ (accessed on 4 July 2025).
- What Is a Dental Bone Graft? Available online: https://westlittlerockdentist.com/what-is-a-dental-bone-graft/ (accessed on 4 July 2025).
- Kwon, J.-W.; Cho, J.H.; Lee, J.B.; Kim, J.H. Evaluation of the Fusion Rate and Safety of Escherichia Coli-Derived rhBMP-2 in Transforaminal Lumbar Interbody Fusion for Patients with Degenerative Lumbar Disease: A Prospective, Multicenter, Single-Arm Trial. J. Clin. Med. 2024, 13, 1733. [Google Scholar] [CrossRef]
- Goulding, S.; Sullivan, A.; O’Keeffe, G.; Collins, L. The Potential of Bone Morphogenetic Protein 2 as a Neurotrophic Factor for Parkinson’s Disease. Neural Regen. Res. 2020, 15, 1432. [Google Scholar] [CrossRef]
- Wang, Y.; Zhao-Ying, C.; Jia-Qi, C.; Hui-Min, C. BMP2 Promotes the Differentiation of Neural Stem Cells into Dopaminergic Neurons in Vitro via miR-145-Mediated Upregulation of Nurr1 Expression. Am. J. Transl. Res. 2016, 8, 3689–3699. [Google Scholar]
- Epstein, N. Pros, Cons, and Costs of INFUSE in Spinal Surgery. Surg. Neurol. Int. 2011, 2, 10. [Google Scholar] [CrossRef]
- Medtronic Expands Use of Controversial Infuse Product. Available online: https://www.mddionline.com/product-development/medtronic-expands-use-of-controversial-infuse-product (accessed on 4 July 2025).
- Infuse Bone Graft. Available online: https://www.drugwatch.com/infuse/ (accessed on 4 July 2025).
- Burkus, J.K.; Gornet, M.F.; Dickman, C.A.; Zdeblick, T.A. Anterior Lumbar Interbody Fusion Using rhBMP-2 With Tapered Interbody Cages. J. Spinal Disord. Tech. 2002, 15, 337–349. [Google Scholar] [CrossRef]
- Kleeman, T.J.; Michael Ahn, U.; Talbot-Kleeman, A. Laparoscopic Anterior Lumbar Interbody Fusion With rhBMP-2: A Prospective Study of Clinical and Radiographic Outcomes. Spine 2001, 26, 2751–2756. [Google Scholar] [CrossRef]
- James, A.W.; LaChaud, G.; Shen, J.; Asatrian, G.; Nguyen, V.; Zhang, X.; Ting, K.; Soo, C. A Review of the Clinical Side Effects of Bone Morphogenetic Protein-2. Tissue Eng. Part B: Rev. 2016, 22, 284–297. [Google Scholar] [CrossRef]
- Carragee, E.J.; Hurwitz, E.L.; Weiner, B.K. A Critical Review of Recombinant Human Bone Morphogenetic Protein-2 Trials in Spinal Surgery: Emerging Safety Concerns and Lessons Learned. Spine J. 2011, 11, 471–491. [Google Scholar] [CrossRef]
- Halloran, D.; Durbano, H.W.; Nohe, A. Bone Morphogenetic Protein-2 in Development and Bone Homeostasis. J. Dev. Biol. 2020, 8, 19. [Google Scholar] [CrossRef]
- Sethi, A.; Craig, J.; Bartol, S.; Chen, W.; Jacobson, M.; Coe, C.; Vaidya, R. Radiographic and CT Evaluation of Recombinant Human Bone Morphogenetic Protein-2–Assisted Spinal Interbody Fusion. Am. J. Roentgenol. 2011, 197, W128–W133. [Google Scholar] [CrossRef]
- Owens, K.; Glassman, S.D.; Howard, J.M.; Djurasovic, M.; Witten, J.L.; Carreon, L.Y. Perioperative Complications with rhBMP-2 in Transforaminal Lumbar Interbody Fusion. Eur. Spine J. 2011, 20, 612–617. [Google Scholar] [CrossRef] [PubMed]
- Girasole, G.; Muro, G.; Mintz, A.; Chertoff, J. Transforaminal Lumbar Interbody Fusion Rates in Patients Using a Novel Titanium Implant and Demineralized Cancellous Allograft Bone Sponge. Int. J. Spine Surg. 2013, 7, e95–e100. [Google Scholar] [CrossRef] [PubMed]
- Tafazal, S.I.; Sell, P.J. Incidental Durotomy in Lumbar Spine Surgery: Incidence and Management. Eur. Spine J. 2005, 14, 287–290. [Google Scholar] [CrossRef] [PubMed]
- Wong, A.P.; Smith, Z.A.; Nixon, A.T.; Lawton, C.D.; Dahdaleh, N.S.; Wong, R.H.; Auffinger, B.; Lam, S.; Song, J.K.; Liu, J.C.; et al. Intraoperative and Perioperative Complications in Minimally Invasive Transforaminal Lumbar Interbody Fusion: A Review of 513 Patients. J. Neurosurg. Spine 2015, 22, 487–495. [Google Scholar] [CrossRef]
- Ghobrial, G.M.; Theofanis, T.; Darden, B.V.; Arnold, P.; Fehlings, M.G.; Harrop, J.S. Unintended Durotomy in Lumbar Degenerative Spinal Surgery: A 10-Year Systematic Review of the Literature. Neurosurg. Focus 2015, 39, E8. [Google Scholar] [CrossRef]
- Carragee, E.J.; Chu, G.; Rohatgi, R.; Hurwitz, E.L.; Weiner, B.K.; Yoon, S.T.; Comer, G.; Kopjar, B. Cancer Risk After Use of Recombinant Bone Morphogenetic Protein-2 for Spinal Arthrodesis. J. Bone Jt. Surg. 2013, 95, 1537–1545. [Google Scholar] [CrossRef]
- Simmonds, M.C.; Brown, J.V.E.; Heirs, M.K.; Higgins, J.P.T.; Mannion, R.J.; Rodgers, M.A.; Stewart, L.A. Safety and Effectiveness of Recombinant Human Bone Morphogenetic Protein-2 for Spinal Fusion: A Meta-Analysis of Individual-Participant Data. Ann. Intern. Med. 2013, 158, 877–889. [Google Scholar] [CrossRef]
- Fu, R.; Selph, S.; McDonagh, M.; Peterson, K.; Tiwari, A.; Chou, R.; Helfand, M. Effectiveness and Harms of Recombinant Human Bone Morphogenetic Protein-2 in Spine Fusion: A Systematic Review and Meta-Analysis. Ann. Intern. Med. 2013, 158, 890–902. [Google Scholar] [CrossRef] [PubMed]
- Cooper, G.S.; Kou, T.D. Risk of Cancer After Lumbar Fusion Surgery With Recombinant Human Bone Morphogenic Protein-2 (Rh-BMP-2). Spine 2013, 38, 1862–1868. [Google Scholar] [CrossRef] [PubMed]
- Dettori, J.R.; Chapman, J.R.; DeVine, J.G.; McGuire, R.A.; Junge, M.R.; Norvell, D.C. Longer Follow-up Continues to Reveal No Increased Risk of Cancer with the Use of Recombinant Human Bone Morphogenetic Protein in Spine Fusion. Spine J. 2019, 19, 1640–1647. [Google Scholar] [CrossRef] [PubMed]
- Freeman, I.; Cohen, S. The Influence of the Sequential Delivery of Angiogenic Factors from Affinity-Binding Alginate Scaffolds on Vascularization. Biomaterials 2009, 30, 2122–2131. [Google Scholar] [CrossRef]
- Lee, K.; Silva, E.A.; Mooney, D.J. Growth Factor Delivery-Based Tissue Engineering: General Approaches and a Review of Recent Developments. J. R. Soc. Interface 2011, 8, 153–170. [Google Scholar] [CrossRef]
- BMP-2 Evaluation in Surgery for Tibial Trauma (BESTT) Study Group. Recombinant Human Bone Morphogenetic Protein-2 for Treatment of Open Tibial Fractures: A Prospective, Controlled, Randomized Study of Four Hundred and Fifty Patients. J. Bone Jt. Surg. 2002, 84, 2123–2134. [Google Scholar] [CrossRef]
- Elniel, A.R.; Giannoudis, P.V. Open Fractures of the Lower Extremity: Current Management and Clinical Outcomes. EFORT Open Rev. 2018, 3, 316–325. [Google Scholar] [CrossRef]
- Troeltzsch, M.; Troeltzsch, M.; Kauffmann, P.; Gruber, R.; Brockmeyer, P.; Moser, N.; Rau, A.; Schliephake, H. Clinical Efficacy of Grafting Materials in Alveolar Ridge Augmentation: A Systematic Review. J. Cranio-Maxillofac. Surg. 2016, 44, 1618–1629. [Google Scholar] [CrossRef]
- Aro, H.T.; Govender, S.; Patel, A.D.; Hernigou, P.; Perera De Gregorio, A.; Popescu, G.I.; Golden, J.D.; Christensen, J.; Valentin, A. Recombinant Human Bone Morphogenetic Protein-2: A Randomized Trial in Open Tibial Fractures Treated with Reamed Nail Fixation. J. Bone Jt. Surg. 2011, 93, 801–808. [Google Scholar] [CrossRef]
- O’Toole, R.V.; Gary, J.L.; Reider, L.; Bosse, M.J.; Gordon, W.T.; Hutson, J.; Quinnan, S.M.; Castillo, R.C.; Scharfstein, D.O.; MacKenzie, E.J.; et al. A Prospective Randomized Trial to Assess Fixation Strategies for Severe Open Tibia Fractures: Modern Ring External Fixators Versus Internal Fixation (FIXIT Study). J. Orthop. Trauma 2017, 31, S10–S17. [Google Scholar] [CrossRef]
- Intramedullary Nailing (Basic Technique). Available online: https://surgeryreference.aofoundation.org/orthopedic-trauma/adult-trauma/basic-technique/basic-principles-of-im-nailing#insertion-of-guide-wires (accessed on 4 July 2025).
- Infuse Bone Graft rhBMP2 Instructions for Preparation and Surgical Application for Open Tibial Fracture 2005. Available online: https://gahpl.com/pdf/biologics/InfuseBMP.pdf (accessed on 4 July 2025).
- Wei, S.; Cai, X.; Huang, J.; Xu, F.; Liu, X.; Wang, Q. Recombinant Human BMP-2 for the Treatment of Open Tibial Fractures. Orthopedics 2012, 35, e847–e854. [Google Scholar] [CrossRef]
- Jones, A.L.; Bucholz, R.W.; Bosse, M.J.; Mirza, S.K.; Lyon, T.R.; Webb, L.X.; Pollak, A.N.; Golden, J.D.; Valentin-Opran, A. Recombinant Human BMP-2 and Allograft Compared with Autogenous Bone Graft for Reconstruction of Diaphyseal Tibial Fractures with Cortical Defects: A Randomized, Controlled Trial. J. Bone Jt. Surg. 2006, 88, 1431–1441. [Google Scholar] [CrossRef] [PubMed]
- Alt, V.; Borgman, B.; Eicher, A.; Heiss, C.; Kanakaris, N.K.; Giannoudis, P.V.; Song, F. Effects of Recombinant Human Bone Morphogenetic Protein-2 (rhBMP-2) in Grade III Open Tibia Fractures Treated with Unreamed Nails—A Clinical and Health-Economic Analysis. Injury 2015, 46, 2267–2272. [Google Scholar] [CrossRef] [PubMed]
- Triplett, R.G.; Nevins, M.; Marx, R.E.; Spagnoli, D.B.; Oates, T.W.; Moy, P.K.; Boyne, P.J. Pivotal, Randomized, Parallel Evaluation of Recombinant Human Bone Morphogenetic Protein-2/Absorbable Collagen Sponge and Autogenous Bone Graft for Maxillary Sinus Floor Augmentation. J. Oral Maxillofac. Surg. 2009, 67, 1947–1960. [Google Scholar] [CrossRef] [PubMed]
- Tiwana, P.S.; Kushner, G.M.; Haug, R.H. Maxillary Sinus Augmentation. Dent. Clin. N. Am. 2006, 50, 409–424. [Google Scholar] [CrossRef]
- Starch-Jensen, T.; Jensen, J.D. Maxillary Sinus Floor Augmentation: A Review of Selected Treatment Modalities. J. Oral Maxillofac. Res. 2017, 8, e3. [Google Scholar] [CrossRef]
- Alraei, K.; Shrqawi, J.; Alarusi, K. Application of Recombinant Human BMP-2 with Bone Marrow Aspirate Concentrate and Platelet-Rich Fibrin in Titanium Mesh for Vertical Maxillary Defect Reconstruction Prior to Implant Placement. Case Rep. Dent. 2021, 2021, 6691022. [Google Scholar] [CrossRef]
- Woo, E.J. Adverse Events Reported After the Use of Recombinant Human Bone Morphogenetic Protein 2. J. Oral Maxillofac. Surg. 2012, 70, 765–767. [Google Scholar] [CrossRef]
- Arias-Irimia, O.; Dorado, C.B.; Moreno, G.G.; Brinkmann, J.C.; Martínez-González, J.M. Pre-operative Measurement of the Volume of Bone Graft in Sinus Lifts Using C Ompu D Ent. Clin. Oral Implant. Res. 2012, 23, 1070–1074. [Google Scholar] [CrossRef]
- Bathla, S.; Fry, R.; Majumdar, K. Maxillary Sinus Augmentation. J. Indian Soc. Periodontol 2018, 22, 468. [Google Scholar] [CrossRef]
- Egol, K.A.; Nauth, A.; Lee, M.; Pape, H.-C.; Watson, J.T.; Borrelli, J. Bone Grafting: Sourcing, Timing, Strategies, and Alternatives. J. Orthop. Trauma 2015, 29, S10–S14. [Google Scholar] [CrossRef] [PubMed]
- Ackerman, S.J.; Mafilios, M.S.; Polly, D.W. Economic Evaluation of Bone Morphogenetic Protein Versus Autogenous Iliac Crest Bone Graft in Single-Level Anterior Lumbar Fusion: An Evidence-Based Modeling Approach. Spine 2002, 27, S94–S99. [Google Scholar] [CrossRef] [PubMed]
- Polly, D.W.; Ackerman, S.J.; Shaffrey, C.I.; Ogilvie, J.W.; Wang, J.C.; Stralka, S.W.; Mafilios, M.S.; Heim, S.E.; Sandhu, H.S. A Cost Analysis of Bone Morphogenetic Protein Versus Autogenous Iliac Crest Bone Graft in Single-Level Anterior Lumbar Fusion. Orthopedics 2003, 26, 1027–1037. [Google Scholar] [CrossRef]
- On, S.-W.; Park, S.-Y.; Yi, S.-M.; Park, I.-Y.; Byun, S.-H.; Yang, B.-E. Current Status of Recombinant Human Bone Morphogenetic Protein-2 (rhBMP-2) in Maxillofacial Surgery: Should It Be Continued? Bioengineering 2023, 10, 1005. [Google Scholar] [CrossRef]
- Carreon, L.Y.; Glassman, S.D.; Djurasovic, M.; Campbell, M.J.; Puno, R.M.; Johnson, J.R.; Dimar, J.R. RhBMP-2 Versus Iliac Crest Bone Graft for Lumbar Spine Fusion in Patients Over 60 Years of Age: A Cost-Utility Study. Spine 2009, 34, 238–243. [Google Scholar] [CrossRef]
- Kelly, M.P.; Vaughn, O.L.A.; Anderson, P.A. Systematic Review and Meta-Analysis of Recombinant Human Bone Morphogenetic Protein-2 in Localized Alveolar Ridge and Maxillary Sinus Augmentation. J. Oral Maxillofac. Surg. 2016, 74, 928–939. [Google Scholar] [CrossRef]
- Mehta, S.; Blagg, R.; Willcockson, J.; Gociman, B.; Yamashiro, D.; Siddiqi, F. Cost-Effectiveness Analysis of Demineralized Bone Matrix and rhBMP-2 versus Autologous Iliac Crest Bone Grafting in Alveolar Cleft Patients. Plast. Reconstr. Surg. 2018, 142, 737–743. [Google Scholar] [CrossRef]
- Dickinson, B.P.; Ashley, R.K.; Wasson, K.L.; O’Hara, C.; Gabbay, J.; Heller, J.B.; Bradley, J.P. Reduced Morbidity and Improved Healing with Bone Morphogenic Protein-2 in Older Patients with Alveolar Cleft Defects. Plast. Reconstr. Surg. 2008, 121, 209–217. [Google Scholar] [CrossRef]
- Lee, H.Y.; An, S.B.; Hwang, S.Y.; Hwang, G.Y.; Lee, H.-L.; Park, H.J.; Shin, J.; Kim, K.N.; Wee, S.W.; Yoon, S.L.; et al. Synergistic Enhancement of Spinal Fusion in Preclinical Models Using Low-Dose rhBMP-2 and Stromal Vascular Fraction in an Injectable Hydrogel Composite. Mater. Today Bio 2025, 30, 101379. [Google Scholar] [CrossRef]
- Lee, H.-Y.; Kang, J.-I.; Lee, H.-L.; Hwang, G.-Y.; Kim, K.-N.; Ha, Y. Concentration-Dependent Efficacy of Recombinant Human Bone Morphogenetic Protein-2 Using a HA/β-TCP Hydrogel Carrier in a Mini-Pig Vertebral Oblique Lateral Interbody Fusion Model. Int. J. Mol. Sci. 2023, 24, 892. [Google Scholar] [CrossRef]
- Lyu, H.-Z.; Lee, J.H. The Efficacy of rhBMP-2 Loaded Hydrogel Composite on Bone Formation around Dental Implants in Mandible Bone Defects of Minipigs. Biomater. Res. 2020, 24, 5. [Google Scholar] [CrossRef]
- Barnes, B.; Boden, S.; Louis-Ugbo, J.; Tomak, P.; Park, J.-S.; Park, M.-S.; Minamide, A. Lower Dose of rhBMP-2 Achieves Spine Fusion When Combined With an Osteoconductive Bulking Agent in Non-Human Primates. Spine 2005, 30, 1127–1133. [Google Scholar] [CrossRef] [PubMed]
- Allen, R.T.; Lee, Y.-P.; Stimson, E.; Garfin, S. Bone Morphogenetic Protein-2 (BMP-2) in the Treatment of Pyogenic Vertebral Osteomyelitis. Spine 2007, 32, 2996–3006. [Google Scholar] [CrossRef] [PubMed]
- Elnaggar, A.G.; Habib, H.-E.A.-A. Idiopathic Pyogenic Thoracic and Lumbar Spondylodiscitis: Outcome of Long Segment Fixation. Egypt J. Neurosurg. 2024, 39, 53. [Google Scholar] [CrossRef]
- Yaw Tee, L.Y.; Hunter, S.; Baker, J.F. BMP Use in the Surgical Treatment of Pyogenic Spondylodiscitis: Is It Safe? J. Clin. Neurosci. 2022, 95, 94–98. [Google Scholar] [CrossRef]
- Lin, L.; Huang, X.; Li, Z.; Zhang, G.; Yu, H.; Wan, Y.; Zhou, C.; Zhou, L. Freeze-Drying Platforms Design for Batch Fabrication of Haversian System Mimicking Scaffolds with Enhanced Osteogenesis. Front. Bioeng. Biotechnol. 2022, 10, 1013528. [Google Scholar] [CrossRef]
- Qin, Q.; Lee, S.; Patel, N.; Walden, K.; Gomez-Salazar, M.; Levi, B.; James, A.W. Neurovascular Coupling in Bone Regeneration. Exp. Mol. Med. 2022, 54, 1844–1849. [Google Scholar] [CrossRef]
- Odgren, P.R.; Gartland, A.; Mason-Savas, A.; Marks, S.C. Bone Structure. In Encyclopedia of Endocrine Diseases; Elsevier: Amsterdam, The Netherlands, 2004; pp. 392–400. ISBN 978-0-12-475570-3. [Google Scholar]
- Dahl, A.C.E.; Thompson, M.S. Mechanobiology of Bone. In Comprehensive Biotechnology; Elsevier: Amsterdam, The Netherlands, 2011; pp. 217–236. ISBN 978-0-08-088504-9. [Google Scholar]
- Patterson-Kane, J.C.; Firth, E.C. Tendon, Ligament, Bone, and Cartilage: Anatomy, Physiology, and Adaptations to Exercise and Training. In The Athletic Horse; Elsevier: Amsterdam, The Netherlands, 2014; pp. 202–242. ISBN 978-0-7216-0075-8. [Google Scholar]
- Klein, M.J. Biology of Normal Bone. In Pathobiology of Human Disease; Elsevier: Amsterdam, The Netherlands, 2014; pp. 753–768. ISBN 978-0-12-386457-4. [Google Scholar]
- Li, L.; Wang, P.; Liang, H.; Jin, J.; Zhang, Y.; Shi, J.; Zhang, Y.; He, S.; Mao, H.; Xue, B.; et al. Design of a Haversian System-like Gradient Porous Scaffold Based on Triply Periodic Minimal Surfaces for Promoting Bone Regeneration. J. Adv. Res. 2023, 54, 89–104. [Google Scholar] [CrossRef]
- Bakalova, L.P.; Andreasen, C.M.; Thomsen, J.S.; Brüel, A.; Hauge, E.-M.; Kiil, B.J.; Delaisse, J.-M.; Andersen, T.L.; Kersh, M.E. Intracortical Bone Mechanics Are Related to Pore Morphology and Remodeling in Human Bone. J. Bone Miner. Res. 2018, 33, 2177–2185. [Google Scholar] [CrossRef]
- Hassan, M.G.; Horenberg, A.L.; Coler-Reilly, A.; Grayson, W.L.; Scheller, E.L. Role of the Peripheral Nervous System in Skeletal Development and Regeneration: Controversies and Clinical Implications. Curr. Osteoporos. Rep. 2023, 21, 503–518. [Google Scholar] [CrossRef]
- Ji-Ye, H.; Xin-Feng, Z.; Lei-Sheng, J. Autonomic Control of Bone Formation. In Handbook of Clinical Neurology; Elsevier: Amsterdam, The Netherlands, 2013; Volume 117, pp. 161–171. ISBN 978-0-444-53491-0. [Google Scholar]
- Ducy, P. Regulation of Bone Remodeling by Central and Peripheral Nervous Signals. In Principles of Bone Biology; Elsevier: Amsterdam, The Netherlands, 2020; pp. 809–823. ISBN 978-0-12-814841-9. [Google Scholar]
- Tajima, T.; Menuki, K.; Okuma, K.F.; Tsukamoto, M.; Fukuda, H.; Okada, Y.; Kosugi, K.; Yamanaka, Y.; Uchida, S.; Sakai, A. Cortical Bone Loss Due to Skeletal Unloading in Aldehyde Dehydrogenase 2 Gene Knockout Mice Is Associated with Decreased PTH Receptor Expression in Osteocytes. Bone 2018, 110, 254–266. [Google Scholar] [CrossRef]
- McMurtrey, R.J. Patterned and Functionalized Nanofiber Scaffolds in Three-Dimensional Hydrogel Constructs Enhance Neurite Outgrowth and Directional Control. J. Neural Eng. 2014, 11, 066009. [Google Scholar] [CrossRef] [PubMed]
- Anitha, M.; Shahnavaz, N.; Qayed, E.; Joseph, I.; Gossrau, G.; Mwangi, S.; Sitaraman, S.V.; Greene, J.G.; Srinivasan, S. BMP2 Promotes Differentiation of Nitrergic and Catecholaminergic Enteric Neurons through a Smad1-Dependent Pathway. Am. J. Physiol.-Gastrointest. Liver Physiol. 2010, 298, G375–G383. [Google Scholar] [CrossRef] [PubMed]
- Hegarty, S.V.; Sullivan, A.M.; O’Keeffe, G.W. BMP2 and GDF5 Induce Neuronal Differentiation through a Smad Dependant Pathway in a Model of Human Midbrain Dopaminergic Neurons. Mol. Cell. Neurosci. 2013, 56, 263–271. [Google Scholar] [CrossRef] [PubMed]
- Ashraf, N.S.; Sutton, J.R.; Yang, Y.; Ranxhi, B.; Libohova, K.; Shaw, E.D.; Barget, A.J.; Todi, S.V.; Paulson, H.L.; Costa, M.D.C. Druggable Genome Screen Identifies New Regulators of the Abundance and Toxicity of ATXN3, the Spinocerebellar Ataxia Type 3 Disease Protein. Neurobiol. Dis. 2020, 137, 104697. [Google Scholar] [CrossRef]
- Zhong, J.; Zou, H. BMP Signaling in Axon Regeneration. Curr. Opin. Neurobiol. 2014, 27, 127–134. [Google Scholar] [CrossRef]
- Benavente, F.; Pinto, C.; Parada, M.; Henríquez, J.P.; Osses, N. Bone Morphogenetic Protein 2 Inhibits Neurite Outgrowth of Motor Neuron-like NSC-34 Cells and Up-regulates Its Type II Receptor. J. Neurochem. 2012, 122, 594–604. [Google Scholar] [CrossRef]
- Niu, H.; Ma, Y.; Wu, G.; Duan, B.; Wang, Y.; Yuan, Y.; Liu, C. Multicellularity-Interweaved Bone Regeneration of BMP-2-Loaded Scaffold with Orchestrated Kinetics of Resorption and Osteogenesis. Biomaterials 2019, 216, 119216. [Google Scholar] [CrossRef]
- Arzaghi, H.; Adel, B.; Jafari, H.; Askarian-Amiri, S.; Shiralizadeh Dezfuli, A.; Akbarzadeh, A.; Pazoki-Toroudi, H. Nanomaterial Integration into the Scaffolding Materials for Nerve Tissue Engineering: A Review. Rev. Neurosci. 2020, 31, 843–872. [Google Scholar] [CrossRef]
- Shi, S.; Ou, X.; Cheng, D. How Advancing Is Peripheral Nerve Regeneration Using Nanofiber Scaffolds? A Comprehensive Review of the Literature. Int. J. Nanomed. 2023, 18, 6763–6779. [Google Scholar] [CrossRef]
- Qian, J.; Lin, Z.; Liu, Y.; Wang, Z.; Lin, Y.; Gong, C.; Ruan, R.; Zhang, J.; Yang, H. Functionalization Strategies of Electrospun Nanofibrous Scaffolds for Nerve Tissue Engineering. Smart Mater. Med. 2021, 2, 260–279. [Google Scholar] [CrossRef]
- Xu, J.; Fahmy-Garcia, S.; Wesdorp, M.A.; Kops, N.; Forte, L.; De Luca, C.; Misciagna, M.M.; Dolcini, L.; Filardo, G.; Labberté, M.; et al. Effectiveness of BMP-2 and PDGF-BB Adsorption onto a Collagen/Collagen-Magnesium-Hydroxyapatite Scaffold in Weight-Bearing and Non-Weight-Bearing Osteochondral Defect Bone Repair: In Vitro, Ex Vivo and In Vivo Evaluation. J. Funct. Biomater. 2023, 14, 111. [Google Scholar] [CrossRef]
- Wang, J.; Xiao, L.; Wang, W.; Zhang, D.; Ma, Y.; Zhang, Y.; Wang, X. The Auxiliary Role of Heparin in Bone Regeneration and Its Application in Bone Substitute Materials. Front. Bioeng. Biotechnol. 2022, 10, 837172. [Google Scholar] [CrossRef]
- Kitahara, T.; Tateiwa, D.; Hirai, H.; Ikuta, M.; Furuichi, T.; Bun, M.; Ukon, Y.; Kanie, Y.; Furuya, M.; Fujimori, T.; et al. rhBMP-2-Loaded Hydroxyapatite/Beta-Tricalcium Phosphate Microsphere/Hydrogel Composite Promotes Bone Regeneration in a Novel Rat Femoral Nonunion Model. Front. Bioeng. Biotechnol. 2024, 12, 1461260. [Google Scholar] [CrossRef] [PubMed]
- Ball, J.R.; Shelby, T.; Hernandez, F.; Mayfield, C.K.; Lieberman, J.R. Delivery of Growth Factors to Enhance Bone Repair. Bioengineering 2023, 10, 1252. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.; Lee, S.H.; Choi, B.S.; Kim, S. Comparison of Four Bone Substitute Types in Sinus Augmentation with Perforated Schneiderian Membrane: An Experimental Study. J. Periodontol. 2025, JPER.24-0663. [Google Scholar] [CrossRef]
- Balaji, D.P.; Dinç Kalayci, P.; Ramkumar, S.S. (Eds.) Advances in Sports Science and Technology, 1st ed.; CRC Press, Taylor & Francis Group: Boca Raton, FL, USA, 2025; ISBN 978-1-003-61628-3. [Google Scholar]
- Stacchi, C.; Coyac, B.R.; Helms, J.A. Biomechanical Basis for Bone Healing and Osseointegration of Implants in Sinus Grafts. Clin. Implant. Dent. Rel. Res. 2025, 27, e13424. [Google Scholar] [CrossRef]
- Duarte, N.D.; Frigério, P.B.; Chica, G.E.A.; Okamoto, R.; Buchaim, R.L.; Buchaim, D.V.; Messora, M.R.; Issa, J.P.M. Biomaterials for Guided Tissue Regeneration and Guided Bone Regeneration: A Review. Dent. J. 2025, 13, 179. [Google Scholar] [CrossRef]
- Fabozzi, R.; Bianchetti, F.; Baldi, D.; Sanchez, C.Y.; Bagnasco, F.; De Angelis, N. The Effectiveness and Complication Rate of Resorbable Biopolymers in Oral Surgery: A Systematic Review. Dent. J. 2025, 13, 264. [Google Scholar] [CrossRef]
| Advantages vs. Autograft | Disadvantages (Side Effects) |
|---|---|
| No tissue harvesting surgery required | High rates of ectopic bone growth (typically between 33% to 70%) |
| ~8% improved fusion rate | 29% rate of radiculitis |
| Lower reoperation rate | 19% rate of dural tear |
| Reduced blood loss | 5% rate of hematoma/seroma |
| Reduced patient-reported pain | Reported cases of Vertebrae osteolysis, Device subsidence and dislodgement, Soft tissue swelling and inflammation, Cervical swelling-related death, Connection to cancer, Retrograde ejaculation, Wound dehiscence, Increased infection, Increased neurologic deficits, Arachnoiditis |
| Better patient-reported overall health score | |
| Shorter surgery time | |
| Shorter hospital stay | |
| Faster return to work |
| Adverse Event (Harmonized Definition) | Rate/Range (Study Context) | Follow-Up Window | Key References | Typical Absolute Fusion Gain vs. Control (~24 Months) |
|---|---|---|---|---|
| Heterotopic/ectopic bone (radiographic, CT-detected) | 13–75% radiographic; symptomatic far lower | 6–12 months CT | [26,57,58,60] | n/a |
| Heterotopic/ectopic bone (symptomatic, requiring treatment/revision) | 0–2% | 6–24 months | [26,57,61] | n/a |
| Radiculitis (new/worsened radicular pain) | 7–14% (posterior fusions) | Early postop—3 months | [57,61] | n/a |
| Osteolysis/osteoclastic resorption (adjacent to cage, CT) | 5–20% (series-dependent) | 3–12 months CT | [57,60,62] | n/a |
| Seroma/hematoma (wound collections) | 1–3% | 30–90 days | [18,57,61] | n/a |
| Wound infection/dehiscence | 2–4% | 30–90 days | [18,57,61] | n/a |
| Urinary retention | 2–8% | In—hospital–30 days | [57,61] | n/a |
| Ileus | 1–3% | In—hospital–30 days | [57,61] | n/a |
| Respiratory complications (pneumonia, aspiration) | 0.5–1% | In—hospital–30 days | [57,61] | n/a |
| Incidental durotomy (approach-related, not BMP-specific) | 3–10% (higher in revisions; 1.8–6.2% MIS-TLIF) | Intra-op | [58,63,64,65] | n/a |
| Retrograde ejaculation (ALIF, male) | 2–8% overall; 7.2% rhBMP-2 vs. 0.6% control (L5–S1 RCT) | 3–24 months | [37,57,58] | ALIF: +6–16% vs. ICBG ([25,37]) |
| Fusion efficacy (PLF/PLIF/TLIF) | – | – | [18,25,37] | PLF/PLIF/TLIF: +5–10% vs. ICBG |
| Clinical Application | Spinal (Lumbar) Fusion | Open Tibial Fracture | Maxillary Augmentation |
|---|---|---|---|
| FDA Approval Year: | 2002 | 2004 | 2007 |
| EMA Approval Year: | 2002 | 2002 | - |
| Major Advantage: | High vertebral fusion rate without harvest surgery | Higher bone formation rate compared to no grafting | High bone formation rate without harvest surgery |
| Major Disadvantage: | Many concerning side effects | Lack of data, possible regeneration inferiority compared to autograft | Some side effects |
| Cost Per Package: | USD 2500–6000 | ||
| Total Cost Relative to Autograft Alternative: | Likely comparable, but definitive conclusion is difficult. Cost is related to clinical success. | ||
| Approach | Key Mechanism | BMP-2 Role | Evidence and Findings |
|---|---|---|---|
| Aligned nanofiber and hydrogel scaffolds | Laminin-coated electrospun fibers guide axon growth within a hydrogel matrix | Co-delivered with BMP 2 to support osteogenesis and neural guidance | Guides neurite outgrowth significantly; enhanced neurite tracking in laminin-functionalized hydrogel |
| BMP 2-loaded nanofibers (e.g., PCL/HA) | Fibrous scaffold mimicking ECM with sustained BMP 2 release | Encourages bone formation; may support neurogenic niche | PLGA/HA fibrous scaffolds with BMP 2 mimic ECM, though neural data limited |
| Magnetic nanoparticles (MNPs) | MNPs guide neurite extension via external magnetic fields | Can be co-functionalized with BMP 2 to hybridize osteo/neuroinduction | Promising concept in SCI repair; shown neurite extension under guidance |
| BMP 2-mRNA exosome hydrogels | Exosomes deliver BMP 2 mRNA, enabling sustained release from host cells | Supports osteogenesis; hypothesized to exert neuroimmunomodulation | Engineered BMP 2 exosomes in hydrogel accelerated bone repair |
| Nanofiber–hydrogel composite for SCI | Composite scaffolds bridge injury sites and support axon regrowth | Could be adapted to BMP 2 delivery for combined therapy | Improved neural repair after spinal cord contusion in rats |
| Exosome-functionalized MOF scaffolds | Exosome-coated magnesium scaffolds drive osteogenesis/angiogenesis | Platform could be repurposed for targeted BMP 2 neurodelivery | Enhanced osteogenic and angiogenic markers observed |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Karipidou, N.; Gorley, J.P.M.; Katrilaka, C.; Manglaris, C.; Tzavellas, A.N.; Pitou, M.; Cheva, A.; Michailidis, N.; Tsiridis, E.E.; Choli-Papadopoulou, T.; et al. A Critical Review of Commercial Collagen-Based Scaffolds in Bone Regeneration: Functional Properties and Clinical Evidence from Infuse® Bone Graft. J. Funct. Biomater. 2025, 16, 313. https://doi.org/10.3390/jfb16090313
Karipidou N, Gorley JPM, Katrilaka C, Manglaris C, Tzavellas AN, Pitou M, Cheva A, Michailidis N, Tsiridis EE, Choli-Papadopoulou T, et al. A Critical Review of Commercial Collagen-Based Scaffolds in Bone Regeneration: Functional Properties and Clinical Evidence from Infuse® Bone Graft. Journal of Functional Biomaterials. 2025; 16(9):313. https://doi.org/10.3390/jfb16090313
Chicago/Turabian StyleKaripidou, Niki, John Paul Muller Gorley, Chrysoula Katrilaka, Chris Manglaris, Anastasios Nektarios Tzavellas, Maria Pitou, Angeliki Cheva, Nikolaos Michailidis, Eleftherios E. Tsiridis, Theodora Choli-Papadopoulou, and et al. 2025. "A Critical Review of Commercial Collagen-Based Scaffolds in Bone Regeneration: Functional Properties and Clinical Evidence from Infuse® Bone Graft" Journal of Functional Biomaterials 16, no. 9: 313. https://doi.org/10.3390/jfb16090313
APA StyleKaripidou, N., Gorley, J. P. M., Katrilaka, C., Manglaris, C., Tzavellas, A. N., Pitou, M., Cheva, A., Michailidis, N., Tsiridis, E. E., Choli-Papadopoulou, T., & Aggeli, A. (2025). A Critical Review of Commercial Collagen-Based Scaffolds in Bone Regeneration: Functional Properties and Clinical Evidence from Infuse® Bone Graft. Journal of Functional Biomaterials, 16(9), 313. https://doi.org/10.3390/jfb16090313








