Surface Activation of Calcium Zirconate-Calcium Stabilized Zirconia Eutectic Ceramics with Bioactive Wollastonite-Tricalcium Phosphate Coatings
Abstract
:1. Introduction
2. Materials and Methods
3. Results
3.1. CZO-CSZ Eutectic
3.2. Alloying of CZO-CSZ Eutectic with W-TCP
3.3. Cladding of CZO-CSZ Eutectic with W-TCP
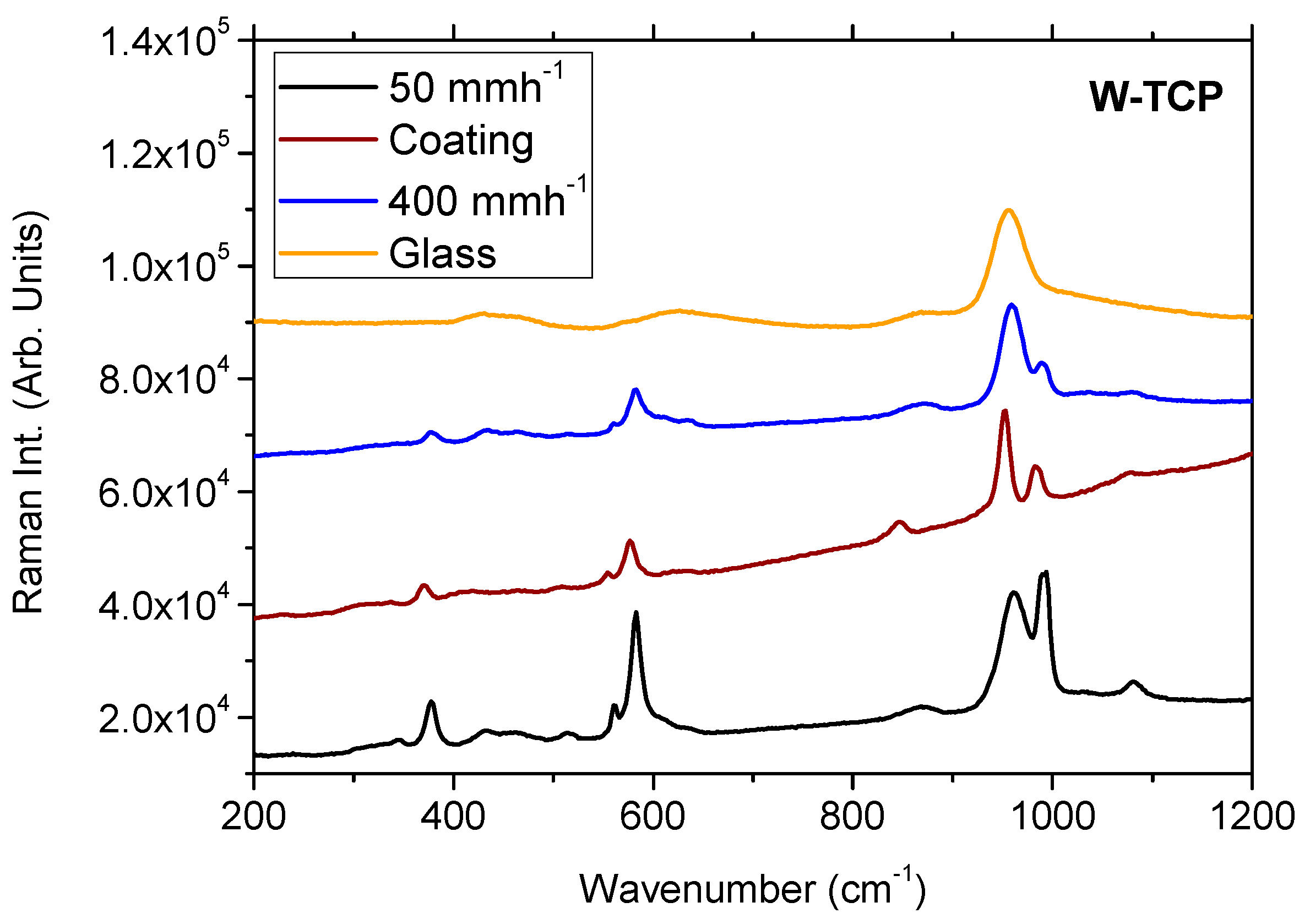
3.4. Micro-Raman Characterization
4. Conclusions
- -
- The microstructure and mechanical properties of CZO-CSZ DSEC have been studied. Because of their excellent hardness, resistance, toughness, and microstructure stability, they exhibit good characteristics for use as biomaterials.
- -
- Laser alloying and cladding can modify in an effective way CZO-CSZ bioinert eutectics with desired biological properties, making them useful as load-bearing implants for clinical applications.
- -
- The as-formed hybrid material is robust, with excellent adhesion between the coating and the ceramic substrate.
- -
- Raman studies allowed for distinguishing the structure of the CZO-CSZ matrix from the W-TCP coating. Furthermore, the Raman spectra of the coating presented features of a glass-ceramic material.
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Manicone, P.F.; Iommetti, P.R.; Raffaelli, L. An overview of zirconia ceramics: Basic properties and clinical applications. J. Dent. 2007, 35, 819–826. [Google Scholar] [CrossRef] [PubMed]
- Piconi, C.; Maccauro, G. Zirconia as a ceramic biomaterial. Biomaterials 1999, 20, 1–25. [Google Scholar] [CrossRef] [PubMed]
- Carter, C.B.; Norton, M.G. Chapter: Ceramics in Biology and Medicine. In Ceramics Materials: Science and Engineering; Springer: Berlin/Heidelberg, Germany, 2007; pp. 635–651. [Google Scholar] [CrossRef]
- Fan, G.; Zhang, J.; Shen, Z.; Dong, D.; Su, H. Integrated processing of Al2O3/ZrO2 eutectic implants with bioactive Ca-P coatings by laser cladding. J. Mater. Res. Technol. 2022, 18, 2842–2852. [Google Scholar] [CrossRef]
- Wang, S.H.; Sola, D.; Peña, J.I. Laser-Induced Surface Modification on Wollastonite-Tricalcium Phosphate and Magnesium Oxide-Magnesium Stabilized Zirconia Eutectics for Bone Restoring Applications. Appl. Sci. 2022, 12, 12188. [Google Scholar] [CrossRef]
- Kulkarni, A.A.; Kohanek, J.; Tyler, K.I.; Hanson, E.; Kim, D.U.; Thornton, K.; Braun, P.V. Template-Directed Solidification of Eutectic Optical Materials. Adv. Op. Mater. 2018, 6, 1800071. [Google Scholar] [CrossRef]
- Merino, R.I.; Pardo, J.A.; Peña, J.I.; de la Fuente, G.F.; Larrea, A.; Orera, V.M. Luminescence properties of ZrO2-CaO eutectic crystals with ordered lamellar microstructure activated with Er3+ ions. Phys. Rev. B 1997, 56, 109076. [Google Scholar] [CrossRef]
- Merino, R.I.; Peña, J.I.; Orera, V.M.; de la Fuente, G.F. Conductivity anisotropy in directionally solidified CaZrO3-CaSZ and MgO-MgSZ eutectics. Solid State Ion. 1997, 100, 313–318. [Google Scholar] [CrossRef]
- Peña, J.I.; Merino, R.I.; de la Fuente, G.F.; Orera, V.M. Aligned ZrO2(c)-CaZrO3 eutectics grown by the laser floating zone method: Electrical and optical properties. Adv. Mater. 1996, 8, 909–912. [Google Scholar] [CrossRef]
- Zhu, L.; Xue, P.; Lan, Q.; Meng, G.; Ren, Y.; Yang, Z.; Xu, P.; Liu, Z. Recent research and development status of laser cladding: A review. Opt. Laser Technol. 2021, 138, 106915. [Google Scholar] [CrossRef]
- Chakraborty, R.; Raza, M.S.; Datta, S.; Saha, P. Synthesis and characterization of nickel free titanium–hydroxyapatite composite coating over Nitinol surface through in-situ laser cladding and alloying. Surf. Coat. Technol. 2019, 358, 539–550. [Google Scholar] [CrossRef]
- De Aza, A.H.; Velásquez, P.; Alemany, M.I.; Pena, P.; De Aza, P.N. In Situ bone-like apatite formation from a bioeutectic® ceramic in SBF dynamic flow. J. Am. Ceram. Soc. 2007, 90, 1200–1207. [Google Scholar] [CrossRef]
- Rodrigo-Vázquez, C.S.; Bartolomé, J.F.; Rodríguez, M.A.; De Aza, A.H. Microstructural development and mechanical performance of CaSiO3-Ca3(PO3)2 bioceramics following the addition of CaSiO3-Ca3 (PO4)2-MgCa(SiO3)2 eutectic glass. Ceram. Int. 2021, 47, 5502–5509. [Google Scholar] [CrossRef]
- De Aza, P.N.; De Aza, A.H.; De Aza, S. Crystalline bioceramic materials. Bol. Soc. Esp. Ceram. Vidr. 2005, 44, 135–145. [Google Scholar] [CrossRef]
- Rey-García, F.; Ibáñez, R.; Angurel, L.A.; Costa, F.M.; de la Fuente, G.F. Laser Floating Zone Growth: Overview, Singular Materials, Broad Applications, and Future Perspectives. Crystals. 2021, 11, 38. [Google Scholar] [CrossRef]
- Magallanes-Perdomo, M.; Pena, P.; De Aza, P.N.; Carrodeguas, R.G.; Rodríguez, M.A.; Turrillas, X.; De Aza, S.; De Aza, A.H. Devitrification studies of wollastonite–tricalcium phosphate eutectic glass. Acta Biomater. 2009, 5, 3057–3066. [Google Scholar] [CrossRef] [PubMed]
- Kokubo, T.; Takadama, H. How useful is SBF in predicting in vivo bone bioactivity? Biomaterials 2006, 27, 2907–2915. [Google Scholar] [CrossRef]
- de Francisco, I.; Merino, R.I.; Orera, V.M.; Larrea, A.; Peña, J.I. Growth of Al2O3/ZrO2(Y2O3) eutectic rods by the laser floating zone technique: Effect of the rotation. J. Eur. Ceram. Soc. 2005, 25, 1341–1350. [Google Scholar] [CrossRef]
- Llorca, F.J.; Orera, V.M. Directionally solidified eutectic ceramic oxides. Prog. Mater. Sci. 2006, 51, 711–809. [Google Scholar] [CrossRef]
- Silva, A.P.; Booth, F.; Garrido, L.; Aglietti, E.; Pena, P.; Baudín, C. Young’s modulus and hardness of multiphase CaZrO3-MgO ceramics by micro and nanoindentation. J. Eur. Ceram. Soc. 2018, 38, 2194–2201. [Google Scholar] [CrossRef]
- Lang, T.J.F.; You, J.-G.; Zhang, X.F.; Luo, X.D.; Zheng, S.Y. Effect of MgO on thermal shock resistance of CaZrO3 ceramic. Ceram. Int. 2018, 44, 22176–22180. [Google Scholar] [CrossRef]
- Davidge, R.; Tappin, G. The Effects of Temperature and Environment on the Strength of Two Polycrystalline Aluminas. Proc. Brit. Ceram. 1970, 15, 47. [Google Scholar]
- Yang, H.; Ohishi, Y.; Kurosaki, K.; Muta, H.; Yamanaka, S. Thermomechanical properties of calcium series perovskite-type oxides. J. Alloys Compd. 2010, 504, 201–204. [Google Scholar] [CrossRef]
- Maurya, R.; Gupta, A.; Omar, S.; Balani, K. Effect of sintering on mechanical properties of ceria reinforced yttria stabilized zirconia. Ceram. Int. 2016, 42, 11393–11403. [Google Scholar] [CrossRef]
- Hulse, C.O.; Batt, J.A. Effect of Eutectic Microstructures on the Mechanical Properties of Ceramic Oxides. Final Report, 1 February 1969–31 December 1973; Technical Report, AD-781995, UARL-N-910803-10; United Aircraft Research Laboratories: Hartford, CT, USA, May 1974. [Google Scholar]
- Hayashi, H.; Saitou, T.; Maruyama, N.; Inaba, H.; Kawamura, K.; Mori, M. Thermal Expansion Coefficient of Yttria Stabilized Zirconia for Various Yttria Contents. Solid State Ion. 2005, 176, 613–619. [Google Scholar] [CrossRef]
- Carneiro, P.M.C.; Maceiras, A.; Nunes-Pereira, J.; Silva, P.D.; Silva, A.P.; Baudín, C. Property characterization and numerical modelling of the thermal conductivity of CaZrO3-MgO ceramic composites. J. Eur. Ceram. Soc. 2021, 41, 7241–7252. [Google Scholar] [CrossRef]
- Booth, F.; Garrido, L.; Aglietti, E.; Silva, A.; Pena, P.; Baudín, C. CaZrO3–MgO structural ceramics obtained by reaction sintering of dolomite-zirconia mixtures. J. Eur. Ceram. Soc. 2016, 36, 2611–2626. [Google Scholar] [CrossRef]
- Zhang, J.; Wang, Z.G.; Song, X.; Xie, M.; Zhang, Y.; Mu, R.; An, S. Tailoring the thermal conductivity of Sc2O3–Yb2O3 Co-stabilized ZrO2 thermal barrier materials and mechanism insight. Ceram. Int. 2023, 49, 13601–13609. [Google Scholar] [CrossRef]
- Kaur, G.; Pandey, O.P.; Singh, K.; Homa, D.; Scott, B.; Pickrell, G. A review of bioactive glasses: Their structure, properties, fabrication and apatite formation. J. Biomed. Mater. Res. 2014, 102, 254–274. [Google Scholar] [CrossRef]
- Canillas, M.; de Aza, A.H.; Rodríguez, M.A. Calcium phosphates in biomedical engineering. In Reference Module in Materials Science and Materials Engineering; Elsevier: Amsterdam, The Netherlands, 2021; pp. 595–600. [Google Scholar] [CrossRef]
- Song, X.; Ding, Y.; Zhang, J.; Jiang, C.; Liu, Z.; Lin, C.; Zheng, W.; Zeng, Y. Thermophysical and mechanical properties of cubic, tetragonal and monoclinic ZrO2. J. Mater. Res. Technol. 2023, 23, 648–655. [Google Scholar] [CrossRef]
- Htun, Z.L.; Ahmad, N.; Thant, A.A.; Ahmad-Fauzi, M.N. Characterization of CaO-ZrO2 Reinforced HAp Biocomposite for Strength and Toughness Improvement. Procedia Chem. 2016, 19, 510–516. [Google Scholar] [CrossRef]
- Vassala, M.F.; Nunes-Pereira, J.; Miguel, S.P.; Correia, I.J.; Silva, A.P. Microstructural, mechanical and biological properties of hydroxyapatite—CaZrO3 biocomposites. Ceram. Int. 2019, 45, 8195–8203. [Google Scholar] [CrossRef]
- Pollet, M.; Daturi, M.; Marinel, S. Vibrational spectroscopy study of the lattice defects in CaZrO3 ceramics. J. Eur. Ceram. Soc. 2004, 24, 1805–1809. [Google Scholar] [CrossRef]
- Sola, D.; Balda, R.; Peña, J.I.; Fernandez, J. Site-selective laser spectroscopy of Nd3+ ions in 0.8CaSiO3-0.2Ca3(PO4)2 biocompatible eutectic glass-ceramics. Opt. Express 2012, 20, 10701–10711. [Google Scholar] [CrossRef] [PubMed]
- Sola, D.; Balda, R.; Al-Saleh, M.; Peña, J.I.; Fernandez, J. Time-resolved fluorescence line-narrowing of Eu3+ in biocompatible eutectic glass-ceramics. Opt. Express 2013, 21, 6561–6571. [Google Scholar] [CrossRef]






| O | Ca | Zr | O | Ca | Zr | |
|---|---|---|---|---|---|---|
| Theoretical (at%) | Experimental (at%) | |||||
| Eutectic | 61.54 | 15.38 | 23.08 | 59.94 | 15.71 | 24.35 |
| CSZ | 63.64 | 9.09 | 27.27 | 61.68 | 9.87 | 28.45 |
| CaZrO3 | 60 | 20 | 20 | 58.39 | 20.93 | 20.68 |
| Growth Conditions | HV (GPa) | Toughness (Indentation) (MPa m1/2) | Flexural Strength (MPa) | Phase Interspacing λ (µm) |
|---|---|---|---|---|
| 300 mm/h | 9.13 ± 0.2 | 2.85 ± 0.24 | 735 ± 24.46 | 1.21 |
| at. % | O | Ca | Zr | P | Si |
|---|---|---|---|---|---|
| Coating average composition | 59.6 (60.60) | 23.06 (21.21) | 6.29 (6.06) | 10.46 (12.12) | |
| Phase 1 | 59.11 | 24.12 | 6.84 | 9.17 | |
| Phase 2 globular | 61.79 | 23.76 | 8.35 | 6.5 | |
| Interface | 61.67 | 9.9 | 26.59 | 1.84 | |
| CZO-CSZ DSEC | 58.45 | 15.62 | 25.93 |
| Sample | Frequency (cm−1) |
|---|---|
| CZO-CSZ DSEC | 145, 190, 212, 227, 234, 262.5, 286.5, 308, 358, 392, 436, 448, 488, 545.5, 607, 660, 818 |
| W-TCP Eut. Coating | 136, 370, 506, 554, 577, 845, 953, 983, 1076 |
| W-TCP Eut. Glass-ceramics | 344, 377, 432, 516, 560, 581, 870, 958, 993, 1079 |
| W-TCP Eut. Glass | 425, 458, 623, 870, 955 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sola, D.; Chueca, E.; Wang, S.; Peña, J.I. Surface Activation of Calcium Zirconate-Calcium Stabilized Zirconia Eutectic Ceramics with Bioactive Wollastonite-Tricalcium Phosphate Coatings. J. Funct. Biomater. 2023, 14, 510. https://doi.org/10.3390/jfb14100510
Sola D, Chueca E, Wang S, Peña JI. Surface Activation of Calcium Zirconate-Calcium Stabilized Zirconia Eutectic Ceramics with Bioactive Wollastonite-Tricalcium Phosphate Coatings. Journal of Functional Biomaterials. 2023; 14(10):510. https://doi.org/10.3390/jfb14100510
Chicago/Turabian StyleSola, Daniel, Eloy Chueca, Shunheng Wang, and José Ignacio Peña. 2023. "Surface Activation of Calcium Zirconate-Calcium Stabilized Zirconia Eutectic Ceramics with Bioactive Wollastonite-Tricalcium Phosphate Coatings" Journal of Functional Biomaterials 14, no. 10: 510. https://doi.org/10.3390/jfb14100510
APA StyleSola, D., Chueca, E., Wang, S., & Peña, J. I. (2023). Surface Activation of Calcium Zirconate-Calcium Stabilized Zirconia Eutectic Ceramics with Bioactive Wollastonite-Tricalcium Phosphate Coatings. Journal of Functional Biomaterials, 14(10), 510. https://doi.org/10.3390/jfb14100510








