Long-Term Antimicrobial Performance of Textiles Coated with ZnO and TiO2 Nanoparticles in a Tropical Climate
Abstract
1. Introduction
2. Materials and Methods
2.1. Preparation of the Fabric Samples
2.2. Preliminary In Vitro Tests
2.2.1. Determination of the Antibacterial Effect of Composite Materials on a Solid Nutrient Medium
2.2.2. Determination of the Antibacterial Effect of Composite Materials in a Liquid Nutrient Medium
2.3. Field Tests
2.4. Isolation of Microorganisms
2.5. Identification of Isolated Microorganisms
2.5.1. Identification of Bacteria
2.5.2. Identification of Fungi
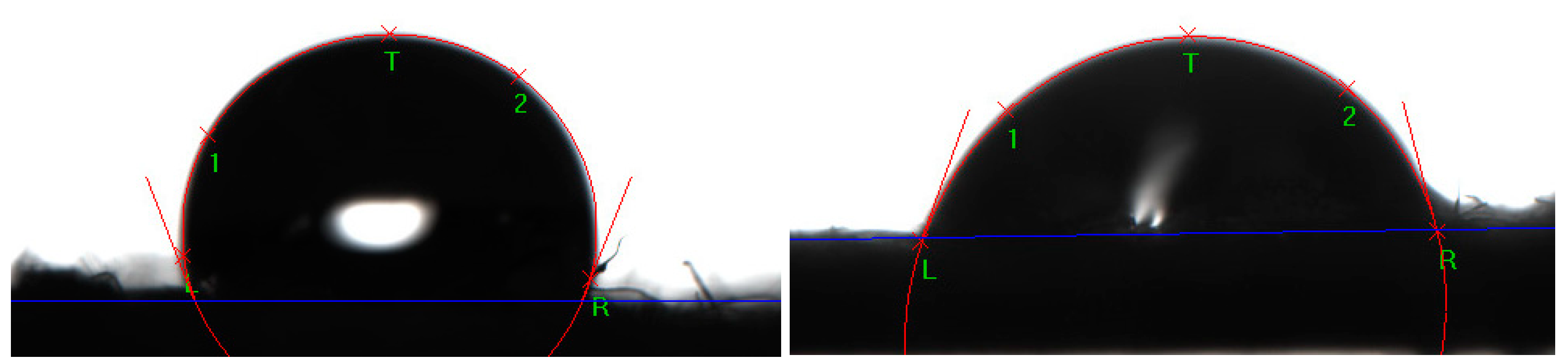
2.6. Study of the Physical and Mechanical Characteristics of Composite Materials
2.7. Statistical Analysis
3. Results and Discussion
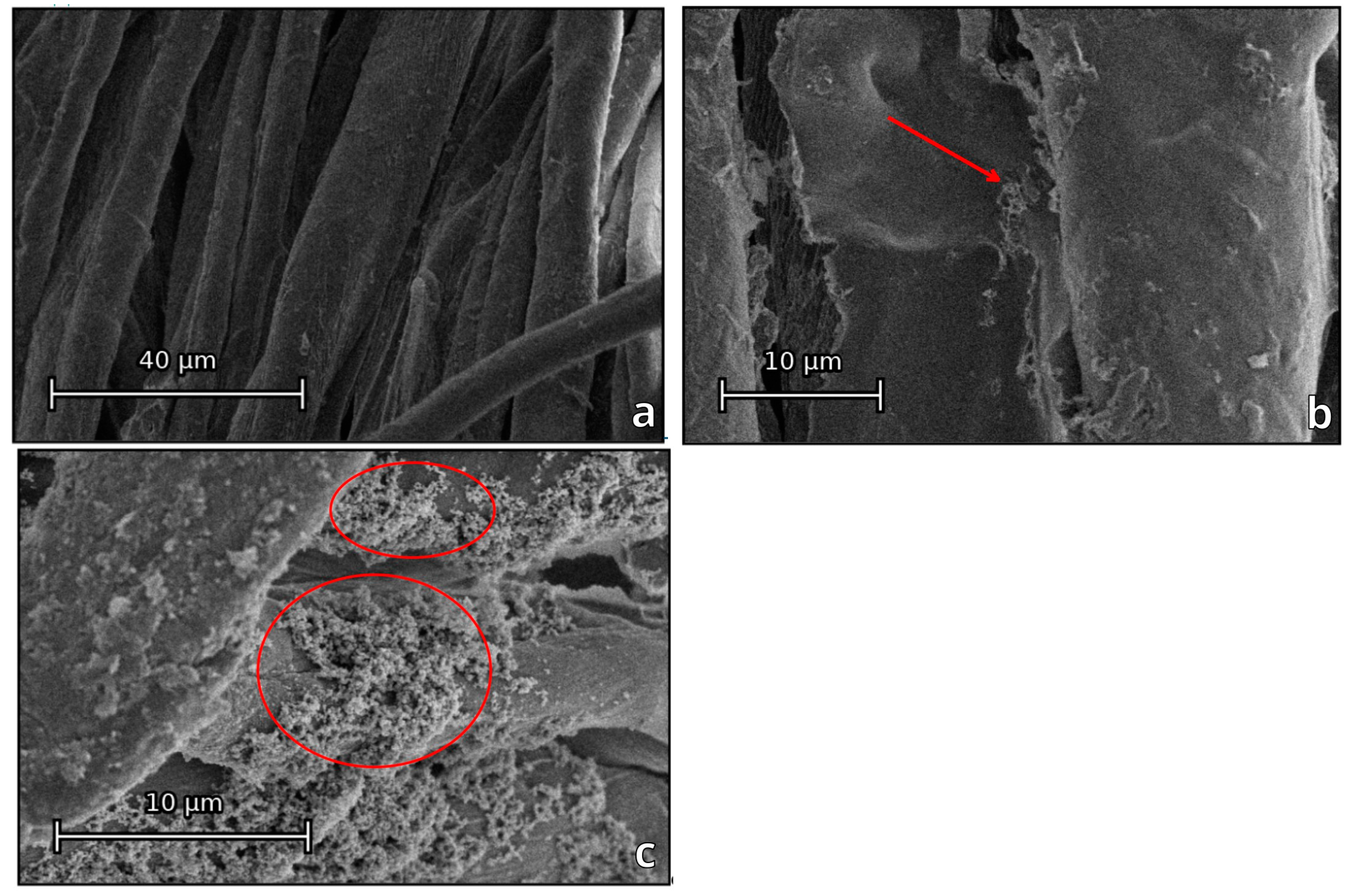
3.1. Production and Characterization of Coated Textiles
3.2. Evaluation of Antibacterial Activity in Laboratory Tests
3.3. Field Tests
3.3.1. The Number of Microorganisms on the Surface of Textiles
3.3.2. Types of Microorganisms on the Surface of Textiles
3.3.3. Identification of the Most Commonly Found Microorganisms
3.4. Tensile Strength Evaluation
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Kon, K.V.; Rai, M. (Eds.) Antibiotic Resistance: Mechanisms and New Antimicrobial Approaches; Academic Press: Cambridge, MA, USA, 2016; ISBN 978-0-12-803642-6. [Google Scholar]
- Abo-zeid, Y.; Williams, G.R. The Potential Anti-infective Applications of Metal Oxide Nanoparticles: A Systematic Review. WIREs Nanomed. Nanobiotechnol. 2020, 12, e1592. [Google Scholar] [CrossRef]
- Vallet-Regí, M.; González, B.; Izquierdo-Barba, I. Nanomaterials as Promising Alternative in the Infection Treatment. Int. J. Mol. Sci. 2019, 20, 3806. [Google Scholar] [CrossRef] [PubMed]
- Bogdan, J.; Zarzyńska, J.; Pławińska-Czarnak, J. Comparison of Infectious Agents Susceptibility to Photocatalytic Effects of Nanosized Titanium and Zinc Oxides: A Practical Approach. Nanoscale Res. Lett. 2015, 10, 309. [Google Scholar] [CrossRef]
- Lallo da Silva, B.; Abuçafy, M.P.; Berbel Manaia, E.; Oshiro Junior, J.A.; Chiari-Andréo, B.G.; Pietro, R.C.R.; Chiavacci, L.A. Relationship Between Structure and Antimicrobial Activity of Zinc Oxide Nanoparticles: An Overview. Int. J. Nanomed. 2019, 14, 9395–9410. [Google Scholar] [CrossRef]
- Lee, J.-H.; Kim, Y.-G.; Cho, M.H.; Lee, J. ZnO Nanoparticles Inhibit Pseudomonas aeruginosa Biofilm Formation and Virulence Factor Production. Microbiol. Res. 2014, 169, 888–896. [Google Scholar] [CrossRef]
- Li, M.; Zhu, L.; Lin, D. Toxicity of ZnO Nanoparticles to Escherichia coli: Mechanism and the Influence of Medium Components. Environ. Sci. Technol. 2011, 45, 1977–1983. [Google Scholar] [CrossRef]
- Khan, M.F.; Ansari, A.H.; Hameedullah, M.; Ahmad, E.; Husain, F.M.; Zia, Q.; Baig, U.; Zaheer, M.R.; Alam, M.M.; Khan, A.M.; et al. Sol-Gel Synthesis of Thorn-like ZnO Nanoparticles Endorsing Mechanical Stirring Effect and Their Antimicrobial Activities: Potential Role as Nano-Antibiotics. Sci. Rep. 2016, 6, 27689. [Google Scholar] [CrossRef] [PubMed]
- He, L.; Liu, Y.; Mustapha, A.; Lin, M. Antifungal Activity of Zinc Oxide Nanoparticles against Botrytis cinerea and Penicillium expansum. Microbiol. Res. 2011, 166, 207–215. [Google Scholar] [CrossRef]
- Lipovsky, A.; Nitzan, Y.; Gedanken, A.; Lubart, R. Antifungal Activity of ZnO Nanoparticles–the Role of ROS Mediated Cell Injury. Nanotechnology 2011, 22, 105101. [Google Scholar] [CrossRef] [PubMed]
- Ann, L.C.; Mahmud, S.; Bakhori, S.K.M.; Sirelkhatim, A.; Mohamad, D.; Hasan, H.; Seeni, A.; Rahman, R.A. Antibacterial Responses of Zinc Oxide Structures against Staphylococcus aureus, Pseudomonas aeruginosa and Streptococcus pyogenes. Ceram. Int. 2014, 40, 2993–3001. [Google Scholar] [CrossRef]
- Pasquet, J.; Chevalier, Y.; Couval, E.; Bouvier, D.; Noizet, G.; Morlière, C.; Bolzinger, M.-A. Antimicrobial Activity of Zinc Oxide Particles on Five Micro-Organisms of the Challenge Tests Related to Their Physicochemical Properties. Int. J. Pharm. 2014, 460, 92–100. [Google Scholar] [CrossRef]
- Cho, M.; Chung, H.; Choi, W.; Yoon, J. Linear Correlation between Inactivation of E. coli and OH Radical Concentration in TiO2 Photocatalytic Disinfection. Water Res. 2004, 38, 1069–1077. [Google Scholar] [CrossRef]
- Salih, F.M.; Pillay, A.E. Efficiency of Solar Water Disinfection Photocatalized by Titanium Dioxide of Varying Particle Size. J. Water Health 2007, 5, 335–340. [Google Scholar] [CrossRef]
- Kubacka, A.; Diez, M.S.; Rojo, D.; Bargiela, R.; Ciordia, S.; Zapico, I.; Albar, J.P.; Barbas, C.; Martins dos Santos, V.A.P.; Fernández-García, M.; et al. Understanding the Antimicrobial Mechanism of TiO2-Based Nanocomposite Films in a Pathogenic Bacterium. Sci. Rep. 2015, 4, 4134. [Google Scholar] [CrossRef]
- Sichel, C.; de Cara, M.; Tello, J.; Blanco, J.; Fernández-Ibáñez, P. Solar Photocatalytic Disinfection of Agricultural Pathogenic Fungi: Fusarium Species. Appl. Catal. B Environ. 2007, 74, 152–160. [Google Scholar] [CrossRef]
- Gondal, M.A.; Alzahrani, A.J.; Randhawa, M.A.; Siddiqui, M.N. Morphology and Antifungal Effect of Nano-ZnO and Nano-Pd-Doped Nano-ZnO against Aspergillus and Candida. J. Environ. Sci. Health Part A 2012, 47, 1413–1418. [Google Scholar] [CrossRef] [PubMed]
- Yu, K.-P.; Huang, Y.-T.; Yang, S.-C. The Antifungal Efficacy of Nano-Metals Supported TiO2 and Ozone on the Resistant Aspergillus niger Spore. J. Hazard. Mater. 2013, 261, 155–162. [Google Scholar] [CrossRef] [PubMed]
- Morais, D.; Guedes, R.; Lopes, M. Antimicrobial Approaches for Textiles: From Research to Market. Materials 2016, 9, 498. [Google Scholar] [CrossRef]
- FDA (Food and Drug Administration). Select Committee on GRAS Substances (SCOGS) Opinion: Zinc Salts. FDA (Food and Drug Administration): Washington, DC, USA, 2015. Available online: https://www.accessdata.fda.gov/scripts/cdrh/cfdocs/cfcfr/CFRSearch.cfm?fr=182.8991 (accessed on 8 November 2022).
- Schilling, K.; Bradford, B.; Castelli, D.; Dufour, E.; Nash, J.F.; Pape, W.; Schulte, S.; Tooley, I.; van den Bosch, J.; Schellauf, F. Human Safety Review of “Nano” Titanium Dioxide and Zinc Oxide. Photochem. Photobiol. Sci. 2010, 9, 495–509. [Google Scholar] [CrossRef] [PubMed]
- Smijs, T.; Pavel, S. Titanium Dioxide and Zinc Oxide Nanoparticles in Sunscreens: Focus on Their Safety and Effectiveness. Nanotechnol. Sci. Appl. 2011, 4, 95–112. [Google Scholar] [CrossRef] [PubMed]
- Younes, M. EFSA Panel on Food Additives and Nutrient Sources added to Food (ANS) Re-evaluation of Titanium Dioxide (E 171) as a Food Additive. EFSA J. 2016, 14, e04545. [Google Scholar] [CrossRef]
- Skocaj, M.; Filipic, M.; Petkovic, J.; Novak, S. Titanium Dioxide in Our Everyday Life; Is It Safe? Radiol. Oncol. 2011, 45, 227–247. [Google Scholar] [CrossRef] [PubMed]
- Ferdous, Z.; Nemmar, A. Health Impact of Silver Nanoparticles: A Review of the Biodistribution and Toxicity Following Various Routes of Exposure. Int. J. Mol. Sci. 2020, 21, 2375. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Zheng, J.; Yu, Y.; Su, Z.; Zhang, L.; Chen, X. Facile Synthesis of RGO–MoS2–Ag Nanocomposites with Long-Term Antimicrobial Activities. Nanotechnology 2020, 31, 125101. [Google Scholar] [CrossRef]
- Liao, C.; Li, Y.; Tjong, S. Bactericidal and Cytotoxic Properties of Silver Nanoparticles. Int. J. Mol. Sci. 2019, 20, 449. [Google Scholar] [CrossRef]
- Moritz, M.; Geszke-Moritz, M. The Newest Achievements in Synthesis, Immobilization and Practical Applications of Antibacterial Nanoparticles. Chem. Eng. J. 2013, 228, 596–613. [Google Scholar] [CrossRef]
- Abramova, A.V.; Abramov, V.O.; Gedanken, A.; Perelshtein, I.; Bayazitov, V.M. An Ultrasonic Technology for Production of Antibacterial Nanomaterials and Their Coating on Textiles. Beilstein J. Nanotechnol. 2014, 5, 532–536. [Google Scholar] [CrossRef]
- Abramova, A.V.; Abramov, V.O.; Bayazitov, V.M.; Voitov, Y.; Straumal, E.A.; Lermontov, S.A.; Cherdyntseva, T.A.; Braeutigam, P.; Weiße, M.; Günther, K. A Sol-Gel Method for Applying Nanosized Antibacterial Particles to the Surface of Textile Materials in an Ultrasonic Field. Ultrason. Sonochem. 2020, 60, 104788. [Google Scholar] [CrossRef]
- Abramov, O.V.; Gedanken, A.; Koltypin, Y.; Perkas, N.; Perelshtein, I.; Joyce, E.; Mason, T.J. Pilot Scale Sonochemical Coating of Nanoparticles onto Textiles to Produce Biocidal Fabrics. Surf. Coat. Technol. 2009, 204, 718–722. [Google Scholar] [CrossRef]
- Thaysen, A.C.; Bunker, H.J.; Butlin, K.R.; Williams, L.H. The effect of climatic exposure on textile fibres and fabrics. Ann. Appl. Biol. 1939, 26, 750–781. [Google Scholar] [CrossRef]
- Saliani, M.; Jalal, R.; Goharshadi, E.K. Effects of pH and Temperature on Antibacterial Activity of Zinc Oxide Nanofluid against E. coli O157:H7 and Staphylococcus aureus. Jundishapur J. Microbiol. 2015, 8, e17115. [Google Scholar] [CrossRef]
- de Azevedo, J.L.; Quecine, M.C. (Eds.) Diversity and Benefits of Microorganisms from the Tropics, 1st ed.; Springer International Publishing: Cham, Switzerland, 2017; ISBN 978-3-319-55804-2. [Google Scholar]
- Abramova, A.; Gedanken, A.; Popov, V.; Ooi, E.-H.; Mason, T.J.; Joyce, E.M.; Beddow, J.; Perelshtein, I.; Bayazitov, V. A Sonochemical Technology for Coating of Textiles with Antibacterial Nanoparticles and Equipment for Its Implementation. Mater. Lett. 2013, 96, 121–124. [Google Scholar] [CrossRef]
- Kamler, A.; Barinov, A. Three-Dimensional Antibacterial Material, Method for Preparation Thereof (Variants), and Unit for Implementing the Method. Patent WO-2018038627-A1, 24 August 2016. [Google Scholar]
- Datsenko, K.A.; Wanner, B.L. One-Step Inactivation of Chromosomal Genes in Escherichia coli K-12 Using PCR Products. Proc. Natl. Acad. Sci. USA 2000, 97, 6640–6645. [Google Scholar] [CrossRef]
- Baba, T.; Ara, T.; Hasegawa, M.; Takai, Y.; Okumura, Y.; Baba, M.; Datsenko, K.A.; Tomita, M.; Wanner, B.L.; Mori, H. Construction of Escherichia coli K-12 In-frame, Single-gene Knockout Mutants: The Keio Collection. Mol. Syst. Biol. 2006, 2, 2006.0008. [Google Scholar] [CrossRef]
- de Vasconcelos, A.T.R.; De Almeida, D.F.; Hungria, M.; Guimaraes, C.T.; Antônio, R.V.; Almeida, F.C.; De Almeida, L.G.; De Almeida, R.; Alves-Gomes, J.A.; Andrade, E.M.; et al. The Complete Genome Sequence of Chromobacterium violaceum Reveals Remarkable and Exploitable Bacterial Adaptability. Proc. Natl. Acad. Sci. USA 2003, 100, 11660–11665. [Google Scholar] [CrossRef]
- Veselova, M.; Lipasova, V.; Protsenko, M.A.; Buza, N.; Khmel, I.A. GacS-Dependent Regulation of Enzymic and Antifungal Activities and Synthesis of N-Acylhomoserine Lactones in Rhizospheric Strain Pseudomonas chlororaphis 449. Folia Microbiol. 2009, 54, 401–408. [Google Scholar] [CrossRef]
- Veselova, M.A.; Klein, S.; Bass, I.A.; Lipasova, V.A.; Metlitskaya, A.Z.; Ovadis, M.I.; Chernin, L.S.; Khmel, I.A. Quorum Sensing Systems of Regulation, Synthesis of Phenazine Antibiotics, and Antifungal Activity in Rhizospheric Bacterium Pseudomonas chlororaphis 449. Russ. J. Genet. 2008, 44, 1400–1408. [Google Scholar] [CrossRef]
- Weisburg, W.G.; Barns, S.M.; Pelletier, D.A.; Lane, D.J. 16S Ribosomal DNA Amplification for Phylogenetic Study. J. Bacteriol. 1991, 173, 697–703. [Google Scholar] [CrossRef]
- White, T.J.; Bruns, T.; Lee, S.J.W.T.; Taylor, J. Amplification and direct sequencing of fungal ribosomal RNA genes for phylogenetics. In PCR Protocols: A Guide to Methods and Applications; Academic Press: New York, NY, USA, 1990; Volume 18, pp. 315–322. [Google Scholar]
- Yeung, K.L.; Leung, W.K.; Yao, N.; Cao, S. Reactivity and Antimicrobial Properties of Nanostructured Titanium Dioxide. Catal. Today 2009, 143, 218–224. [Google Scholar] [CrossRef]
- Jana, T.K.; Jana, S.K.; Kumar, A.; De, K.; Maiti, R.; Mandal, A.K.; Chatterjee, T.; Chatterjee, B.K.; Chakrabarti, P.; Chatterjee, K. The Antibacterial and Anticancer Properties of Zinc Oxide Coated Iron Oxide Nanotextured Composites. Colloids Surf. B Biointerfaces 2019, 177, 512–519. [Google Scholar] [CrossRef]
- Ercan, B.; Taylor, E.; Alpaslan, E.; Webster, T.J. Diameter of Titanium Nanotubes Influences Anti-Bacterial Efficacy. Nanotechnology 2011, 22, 295102. [Google Scholar] [CrossRef] [PubMed]
- Banner, D.; Firlar, E.; Jakubonis, J.; Baggia, Y.; Finlay, J.; Shahbazian Yassar, R.; Megaridis, C.; Shokuhfar, T. Correlative Ex Situ and Liquid-Cell TEM Observation of Bacterial Cell Membrane Damage Induced by Rough Surface Topology. Int. J. Nanomed. 2020, 15, 1929–1938. [Google Scholar] [CrossRef]
- Jang, Y.; Choi, W.T.; Johnson, C.T.; García, A.J.; Singh, P.M.; Breedveld, V.; Hess, D.W.; Champion, J.A. Inhibition of Bacterial Adhesion on Nanotextured Stainless Steel 316L by Electrochemical Etching. ACS Biomater. Sci. Eng. 2018, 4, 90–97. [Google Scholar] [CrossRef]
- Khanmirzaee, S.; Montazer, M.; Pashaee, A. Dyeing of Cotton Fabric with Antibacterial Properties Using Direct Dye and CTAB. J. Nat. Fibers 2020, 17, 223–234. [Google Scholar] [CrossRef]
- McDonnell, G.; Russell, A.D. Antiseptics and Disinfectants: Activity, Action, and Resistance. Clin. Microbiol. Rev. 1999, 12, 147–179. [Google Scholar] [CrossRef]
- Al-Adham, I.S.I.; Dinning, A.J.; Eastwood, I.M.; Austin, P.; Collier, P.J. Cell Membrane Effects of Some Common Biocides. J. Ind. Microbiol. Biotechnol. 1998, 21, 6–10. [Google Scholar] [CrossRef]
- Cloete, T.E.; Jacobs, L.; Brözel, V.S. The chemical control of biofouling in industrial water systems. Biodegradation 1998, 9, 23–37. [Google Scholar] [CrossRef] [PubMed]
- Gilbert, P.; Allison, D.G.; McBain, A.J. Biofilms in Vitro and in Vivo: Do Singular Mechanisms Imply Cross-Resistance?: Biocide and antibiotic cross-tolerance. J. Appl. Microbiol. 2002, 92, 98S–110S. [Google Scholar] [CrossRef] [PubMed]
- Simões, M.; Pereira, M.O.; Machado, I.; Simões, L.C.; Vieira, M.J. Comparative Antibacterial Potential of Selected Aldehyde-Based Biocides and Surfactants against Planktonic Pseudomonas fluorescens. J. Ind. Microbiol. Biotechnol. 2006, 33, 741–749. [Google Scholar] [CrossRef] [PubMed]
- Vereshchagin, A.N.; Frolov, N.A.; Egorova, K.S.; Seitkalieva, M.M.; Ananikov, V.P. Quaternary Ammonium Compounds (QACs) and Ionic Liquids (ILs) as Biocides: From Simple Antiseptics to Tunable Antimicrobials. Int. J. Mol. Sci. 2021, 22, 6793. [Google Scholar] [CrossRef] [PubMed]
- Ali, H.; Khan, E.; Ilahi, I. Environmental Chemistry and Ecotoxicology of Hazardous Heavy Metals: Environmental Persistence, Toxicity, and Bioaccumulation. J. Chem. 2019, 2019, 6730305. [Google Scholar] [CrossRef]
- Rahman, Z.; Singh, V.P. The Relative Impact of Toxic Heavy Metals (THMs) (Arsenic (As), Cadmium (Cd), Chromium (Cr)(VI), Mercury (Hg), and Lead (Pb)) on the Total Environment: An Overview. Environ. Monit. Assess. 2019, 191, 419. [Google Scholar] [CrossRef]
- Lee, M.-Y.; Wang, W.-L.; Xu, Z.-B.; Ye, B.; Wu, Q.-Y.; Hu, H.-Y. The Application of UV/PS Oxidation for Removal of a Quaternary Ammonium Compound of Dodecyl Trimethyl Ammonium Chloride (DTAC): The Kinetics and Mechanism. Sci. Total Environ. 2019, 655, 1261–1269. [Google Scholar] [CrossRef]
- Zabielska-Matejuk, J.; Czaczyk, K. Biodegradation of New Quaternary Ammonium Compounds in Treated Wood by Mould Fungi. Wood Sci. Technol. 2006, 40, 461–475. [Google Scholar] [CrossRef]
- Kairyte, K.; Kadys, A.; Luksiene, Z. Antibacterial and Antifungal Activity of Photoactivated ZnO Nanoparticles in Suspension. J. Photochem. Photobiol. B 2013, 128, 78–84. [Google Scholar] [CrossRef]
- Najibi Ilkhechi, N.; Mozammel, M.; Yari Khosroushahi, A. Antifungal Effects of ZnO, TiO2 and ZnO-TiO2 Nanostructures on Aspergillus flavus. Pestic. Biochem. Physiol. 2021, 176, 104869. [Google Scholar] [CrossRef]
- Singh, V.K.; Mishra, A.; Jha, B. 3-Benzyl-Hexahydro-Pyrrolo[1,2-a]Pyrazine-1,4-Dione Extracted from Exiguobacterium indicum Showed Anti-Biofilm Activity against Pseudomonas aeruginosa by Attenuating Quorum Sensing. Front. Microbiol. 2019, 10, 1269. [Google Scholar] [CrossRef]
- Basumatary, B.; Das, D.; Choudhury, B.N.; Dutta, P.; Bhattacharyya, A. Isolation and Characterization of Endophytic Bacteria from Tomato Foliage and Their in Vitro Efficacy against Root-Knot Nematodes. J. Nematol. 2021, 53, 1–16. [Google Scholar] [CrossRef]
- Kiran, G.S.; Sabarathnam, B.; Thajuddin, N.; Selvin, J. Production of Glycolipid Biosurfactant from Sponge-Associated Marine Actinobacterium Brachybacterium paraconglomeratum MSA21. J. Surfactants Deterg. 2014, 17, 531–542. [Google Scholar] [CrossRef]
- Liu, Y.; Gao, J.; Bai, Z.; Wu, S.; Li, X.; Wang, N.; Du, X.; Fan, H.; Zhuang, G.; Bohu, T.; et al. Unraveling Mechanisms and Impact of Microbial Recruitment on Oilseed Rape (Brassica napus L.) and the Rhizosphere Mediated by Plant Growth-Promoting Rhizobacteria. Microorganisms 2021, 9, 161. [Google Scholar] [CrossRef] [PubMed]
- Dimopoulou, A.; Theologidis, I.; Varympopi, A.; Papafotis, D.; Mermigka, G.; Tzima, A.; Panopoulos, N.J.; Skandalis, N. Shifting Perspectives of Translational Research in Bio-Bactericides: Reviewing the Bacillus amyloliquefaciens Paradigm. Biology 2021, 10, 1202. [Google Scholar] [CrossRef]
- Ngalimat, M.S.; Yahaya, R.S.R.; Baharudin, M.M.A.-A.; Yaminudin, S.M.; Karim, M.; Ahmad, S.A.; Sabri, S. A Review on the Biotechnological Applications of the Operational Group Bacillus Amyloliquefaciens. Microorganisms 2021, 9, 614. [Google Scholar] [CrossRef]
- Li, C.; Cao, P.; Du, C.; Xu, X.; Xiang, W.; Wang, X.; Zhao, J. First Report of Leaf Spot Caused by Cladosporium tenuissimum on Panicle Hydrangea (Hydrangea paniculata) in China. Plant Dis. 2021, 105, 2240. [Google Scholar] [CrossRef]
- Nam, M.H.; Park, M.S.; Kim, H.S.; Kim, T.I.; Kim, H.G. Cladosporium cladosporioides and C. tenuissimum Cause Blossom Blight in Strawberry in Korea. Mycobiology 2015, 43, 354–359. [Google Scholar] [CrossRef]
- Rahimi, T.; Mohammadi, R. Identification of Dermatophyte and Nondermatophyte Molds Isolated from Animal Lesions Suspected to Dermatomycoses. Adv. Biomed. Res. 2020, 9, 4. [Google Scholar] [CrossRef]
- Zhang, N.; O’Donnell, K.; Sutton, D.A.; Nalim, F.A.; Summerbell, R.C.; Padhye, A.A.; Geiser, D.M. Members of the Fusarium solani Species Complex That Cause Infections in Both Humans and Plants Are Common in the Environment. J. Clin. Microbiol. 2006, 44, 2186–2190. [Google Scholar] [CrossRef] [PubMed]
- Brandt, S.C.; Ellinger, B.; van Nguyen, T.; Harder, S.; Schlüter, H.; Hahnke, R.L.; Rühl, M.; Schäfer, W.; Gand, M. Aspergillus sydowii: Genome Analysis and Characterization of Two Heterologous Expressed, Non-Redundant Xylanases. Front. Microbiol. 2020, 11, 2154. [Google Scholar] [CrossRef] [PubMed]
- Lago, M.C.; Santos, F.C.; Bueno, P.S.A.; Oliveira, M.A.S.; Barbosa-Tessmann, I.P. The Glucoamylase from Aspergillus wentii: Purification and Characterization. J. Basic Microbiol. 2021, 61, 443–458. [Google Scholar] [CrossRef] [PubMed]
- Form, I.C.; Bonus, M.; Gohlke, H.; Lin, W.; Daletos, G.; Proksch, P. Xanthone, Benzophenone and Bianthrone Derivatives from the Hypersaline Lake-Derived Fungus Aspergillus wentii. Bioorg. Med. Chem. 2019, 27, 115005. [Google Scholar] [CrossRef]
- Into, P.; Pontes, A.; Sampaio, J.P.; Limtong, S. Yeast Diversity Associated with the Phylloplane of Corn Plants Cultivated in Thailand. Microorganisms 2020, 8, 80. [Google Scholar] [CrossRef]
- Eskani, I.N.; Astuti, W.; Farida; Haerudin, A.; Setiawan, J.; Lestari, D.W.; Isnaini; Widayatno, T. Antibacterial Activities of Synthesised ZnO Nanoparticles Applied on Reactive Dyed Batik Fabrics. J. Text. Inst. 2022, 113, 430–439. [Google Scholar] [CrossRef]


| Bacteria Sample | C. violaceum CV12472 | E. coli BW25113 | P. chlororaphis 449 * | |||
|---|---|---|---|---|---|---|
| Solid Medium | Liquid Medium | Solid Medium | Liquid Medium | Solid Medium | ||
| Dilution of Bacterial Overnight Culture (Times) | Dilution of Bacterial Overnight Culture (Times) | |||||
| ×100 | ×1000 | ×1000 | ||||
| Reference sample ** | <10% a | <10% a | <10% a | <10% a | <10% a | <10% a |
| ZnO + CTAB | >50% b | <10% a | + a | >50% b | <10% a | 30–45% b |
| ZnO + TiO2 | 10–20% c | <10% a | + a | 20–50% c | <10% a | 45–50% c |
| Duration of Exposure to a Tropical Environment, Months | |||||||
|---|---|---|---|---|---|---|---|
| Type of Test Site | Type of Coating | 1 | 3 | 4 | |||
| Strains/Sample | Colonies/Sample (CFU) | Strains/Sample | Colonies/Sample (CFU) | Strains/Sample | Colonies/Sample (CFU) | ||
| Concrete open test site | Reference sample * | 15 | 9,600,000 | 15 | 11,130,000 | 15 | 12,200,000 |
| ZnO + CTAB | 8 | 95,000 (1% **) | 9 | 305,000 (2.7% **) | 9 | 650,000 (5.3% **) | |
| ZnO + TiO2 | 10 | 210,000 (2.2% **) | 9 | 246,000 (2.2% **) | 8 | 225,000 (1.8% **) | |
| Mycological shelf test site | Reference sample * | 14 | 5,950,000 | 14 | 8,140,000 | 14 | 11,650,000 |
| ZnO + CTAB | 6 | 167,000 (2.8% **) | 6 | 135,000 (1.7% **) | 7 | 91,000 (0.8% **) | |
| ZnO + TiO2 | 7 | 154,000 (2.6% **) | 7 | 153,000 (1.9% **) | 8 | 177,500 (1.5% **) | |
| Time of Exposure | Number of Species After | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| 1 Month | 3 Months | 4 Months | ||||||||
| Type of Microorganism | Bacteria | Yeast | Fungi | Bacteria | Yeast | Fungi | Bacteria | Yeast | Fungi | |
| Concrete open test site | Reference sample | 5 | 3 | 7 | 5 | 3 | 7 | 5 | 3 | 7 |
| ZnO+ CTAB | 2 | 2 | 4 | 2 | 2 | 5 | 2 | 2 | 5 | |
| ZnO + TiO2 | 2 | 3 | 6 | 2 | 3 | 5 | 2 | 3 | 5 | |
| Mycological shelf test site | Reference sample | 4 | 3 | 7 | 4 | 3 | 7 | 4 | 3 | 7 |
| ZnO + CTAB | 2 | 1 | 3 | 2 | 1 | 3 | 3 | 1 | 3 | |
| ZnO + TiO2 | 3 | 2 | 3 | 3 | 3 | 3 | 3 | 3 | 3 | |
| Species | Strain | Agreement with Genbank Database |
|---|---|---|
| Bacteria | Stenotrophomonas rhizophila | 100% |
| Exiguobacterium indicum | 99.86% | |
| Brachybacterium paraconglomeratum | 99.93% | |
| Bacillus amyloliquefaciens | 99.72% | |
| Yeast | Hannaella sinensis | 97.02% |
| Fungi | Cladosporium tenuissimum | 100% |
| Sarocladium implicatum | 100% | |
| Aspergillus sydowii | 100% | |
| Aspergillus wentii | 100% | |
| Fusarium solani-melongenae | 100% |
| Type of Exposure | Type of Coating | Duration of Exposure to the Tropical Environment | |||
|---|---|---|---|---|---|
| 0 (Before the Field Test) | 1 Month | 3 Months | 4 Months | ||
| Tensile Strength, MPa | |||||
| Concrete open test site | Reference sample, original cotton fabric without a coating | 23.1 | 17.4 | 14.1 | 13.2 |
| Fabric coated with ZnO nanoparticles + CTAB | 22.3 | 21.4 | 17.9 | 17.5 | |
| Fabric coated with ZnO + TiO2 nanoparticles | 20.9 | 18.2 | 13.6 | 9.1 | |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Veselova, V.O.; Plyuta, V.A.; Kostrov, A.N.; Vtyurina, D.N.; Abramov, V.O.; Abramova, A.V.; Voitov, Y.I.; Padiy, D.A.; Thu, V.T.H.; Hue, L.T.; et al. Long-Term Antimicrobial Performance of Textiles Coated with ZnO and TiO2 Nanoparticles in a Tropical Climate. J. Funct. Biomater. 2022, 13, 233. https://doi.org/10.3390/jfb13040233
Veselova VO, Plyuta VA, Kostrov AN, Vtyurina DN, Abramov VO, Abramova AV, Voitov YI, Padiy DA, Thu VTH, Hue LT, et al. Long-Term Antimicrobial Performance of Textiles Coated with ZnO and TiO2 Nanoparticles in a Tropical Climate. Journal of Functional Biomaterials. 2022; 13(4):233. https://doi.org/10.3390/jfb13040233
Chicago/Turabian StyleVeselova, Varvara O., Vladimir A. Plyuta, Andrei N. Kostrov, Darya N. Vtyurina, Vladimir O. Abramov, Anna V. Abramova, Yury I. Voitov, Darya A. Padiy, Vo Thi Hoai Thu, Le Thi Hue, and et al. 2022. "Long-Term Antimicrobial Performance of Textiles Coated with ZnO and TiO2 Nanoparticles in a Tropical Climate" Journal of Functional Biomaterials 13, no. 4: 233. https://doi.org/10.3390/jfb13040233
APA StyleVeselova, V. O., Plyuta, V. A., Kostrov, A. N., Vtyurina, D. N., Abramov, V. O., Abramova, A. V., Voitov, Y. I., Padiy, D. A., Thu, V. T. H., Hue, L. T., Trang, D. T. T., Baranchikov, A. E., Khmel, I. A., Nadtochenko, V. A., & Ivanov, V. K. (2022). Long-Term Antimicrobial Performance of Textiles Coated with ZnO and TiO2 Nanoparticles in a Tropical Climate. Journal of Functional Biomaterials, 13(4), 233. https://doi.org/10.3390/jfb13040233









