Patient-Specific CFD Analysis of Carotid Artery Haemodynamics: Impact of Anatomical Variations on Atherosclerotic Risk
Abstract
1. Introduction
2. Methodology
2.1. Theory
2.2. Vessel Segmentation and Geometry Construction
2.3. Meshing and Simulation
3. Results
3.1. Velocity Streamline
3.2. Pressure
3.3. Wall Shear Stress
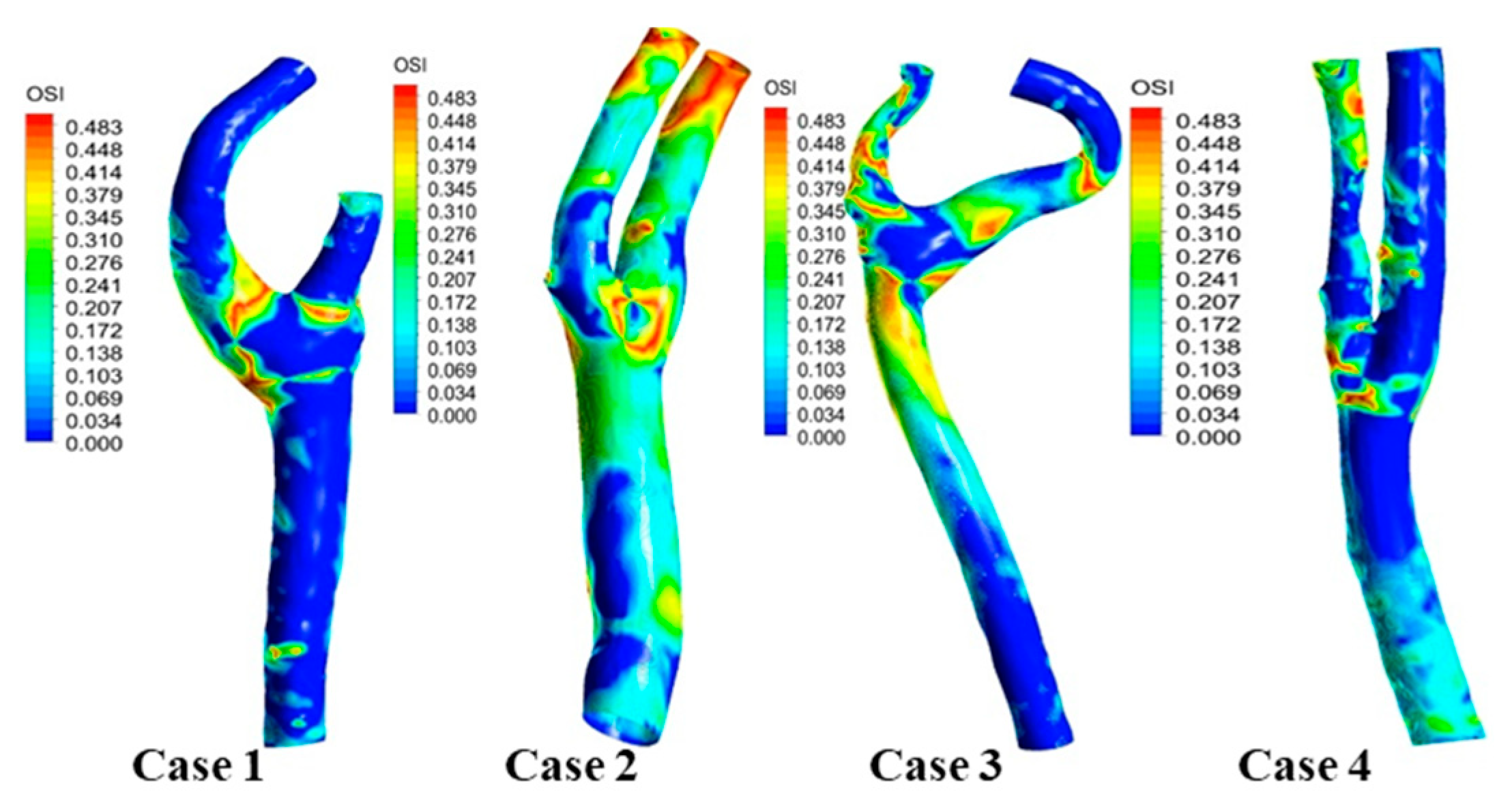
3.4. Oscillatory Shear Index
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Correction Statement
Abbreviations
| CVD | Cardiovascular diseases |
| TIA | Transient ischemic attacks |
| CFD | Computational fluid dynamics |
| CCA | Common carotid artery |
| ICA | Internal carotid artery |
| ECA | External carotid artery |
| ES | Early systole |
| PD | Peak diastole |
| ED | Early diastole |
| CY | Carreau–Yasuda |
| WSS | Wall shear stress |
| TAWSS | Time-averaged wall shear stress |
| OSI | Oscillatory shear index |
References
- Townsend, N.; Wilson, L.; Bhatnagar, P.; Wickramasinghe, K.; Rayner, M.; Nichols, M. Cardiovascular Disease in Europe: Epidemiological Update 2016. Eur. Heart J. 2016, 37, 3232–3245. [Google Scholar] [CrossRef]
- Sreeniwas Kumar, A.; Sinha, N. Cardiovascular Disease in India: A 360 Degree Overview. Med. J. Armed Forces India 2020, 76, 1–3. [Google Scholar] [CrossRef] [PubMed]
- Yang, J.; Zhang, Y.; Xue, J.; Guo, Y.; Liu, S.; Yao, Y.; Zhong, H.; Quan, A.; Yang, J. Hemodynamic Effects of Stenosis with Varying Severity in Different Segments of the Carotid Artery Using Computational Fluid Dynamics. Sci. Rep. 2025, 15, 4896. [Google Scholar] [CrossRef] [PubMed]
- Ku, D.N. Blood Flow in Arteries. Annu. Rev. Fluid Mech. 1997, 29, 399–434. [Google Scholar] [CrossRef]
- Guerciotti, B.; Vergara, C.; Azzimonti, L.; Forzenigo, L.; Buora, A.; Biondetti, P.; Domanin, M. Computational Study of the Fluid-Dynamics in Carotids before and after Endarterectomy. J. Biomech. 2016, 49, 26–38. [Google Scholar] [CrossRef]
- Ngo, M.T.; Lee, U.Y.; Ha, H.; Jin, N.; Chung, G.H.; Kwak, Y.G.; Jung, J.; Kwak, H.S. Comparison of Hemodynamic Visualization in Cerebral Arteries: Can Magnetic Resonance Imaging Replace Computational Fluid Dynamics? J. Pers. Med. 2021, 11, 253. [Google Scholar] [CrossRef]
- Ya, X.; Ma, L.; Li, H.; Ge, P.; Zheng, Z.; Mou, S.; Liu, C.; Zhang, Y.; Wang, R.; Zhang, Q.; et al. Exploring the Relationship between Hemodynamics and the Immune Microenvironment in Carotid Atherosclerosis: Insights from CFD and CyTOF Technologies. J. Cereb. Blood Flow Metab. 2024, 44, 1733–1744. [Google Scholar] [CrossRef]
- Gessaghi, V.C.; Raschi, M.A.; Tanoni, D.Y.; Perazzo, C.A.; Larreteguy, A.E. Growth Model for Cholesterol Accumulation in the Wall of a Simplified 3D Geometry of the Carotid Bifurcation. Comput. Methods Appl. Mech. Eng. 2011, 200, 2117–2125. [Google Scholar] [CrossRef]
- Bit, A.; Ghagare, D.; Rizvanov, A.A.; Chattopadhyay, H. Assessment of Influences of Stenoses in Right Carotid Artery on Left Carotid Artery Using Wall Stress Marker. BioMed Res. Int. 2017, 2017, 2935195. [Google Scholar] [CrossRef]
- Bae, T.; Ko, J.H.; Chung, J. Turbulence Intensity as an Indicator for Ischemic Stroke in the Carotid Web. World Neurosurg. 2021, 154, e443–e457. [Google Scholar] [CrossRef]
- Cao, J.; Tan, Z.; Tang, Y.; Liu, Z.; Huang, J.; Huo, G.; Yao, Z.; Zhou, Y.; Zhou, D. Hemodynamic Simulation Analysis of Tandem Carotid Artery Stenosis Based on a Patient-Specific Case. J. Biomech. 2025, 191, 112907. [Google Scholar] [CrossRef] [PubMed]
- Fu, Y.; Qiao, A.; Jin, L. The Influence of Hemodynamics on the Ulceration Plaques of Carotid Artery Stenosis. J. Mech. Med. Biol. 2015, 15, 1550008. [Google Scholar] [CrossRef]
- Massai, D.; Soloperto, G.; Gallo, D.; Xu, X.Y.; Morbiducci, U. Shear-Induced Platelet Activation and Its Relationship with Blood Flow Topology in a Numerical Model of Stenosed Carotid Bifurcation. Eur. J. Mech. B/Fluids 2012, 35, 92–101. [Google Scholar] [CrossRef]
- Urevc, J.; Zun, I.; Brumen, M.; Stok, B. Modeling the Effect of Red Blood Cells Deformability on Blood Flow Conditions in Human Carotid Artery Bifurcation. J. Biomech. Eng. 2017, 139, 011011. [Google Scholar] [CrossRef]
- Morbiducci, U.; Gallo, D.; Massai, D.; Ponzini, R.; Deriu, M.A.; Antiga, L.; Redaelli, A.; Montevecchi, F.M. On the Importance of Blood Rheology for Bulk Flow in Hemodynamic Models of the Carotid Bifurcation. J. Biomech. 2011, 44, 2427–2438. [Google Scholar] [CrossRef]
- Sia, S.F.; Zhao, X.; Yu, Y.; Zhang, Y. Multiphase Particle-in-Cell Simulation in Severe Internal Carotid Artery Stenosis. Powder Technol. 2019, 358, 62–67. [Google Scholar] [CrossRef]
- Guerciotti, B.; Vergara, C. Computational Comparison between Newtonian and Non-Newtonian Blood Rheologies in Stenotic Vessels. In Biomedical Technology: Modeling, Experiments and Simulation; Lecture Notes in Applied and Computational Mechanics; Springer: Cham, Switzerland, 2018; Volume 84, pp. 169–183. [Google Scholar]
- Lee, S.H.; Han, K.S.; Hur, N.; Cho, Y.I.; Jeong, S.K. The Effect of Patient-Specific Non-Newtonian Blood Viscosity on Arterial Hemodynamics Predictions. J. Mech. Med. Biol. 2019, 19, 1940054. [Google Scholar] [CrossRef]
- Hoi, Y.; Wasserman, B.A.; Lakatta, E.G.; Steinman, D.A. Carotid Bifurcation Hemodynamics in Older Adults: Effect of Measured versus Assumed Flow Waveform. J. Biomech. Eng. 2010, 132, 071006. [Google Scholar] [CrossRef]
- Hoi, Y.; Wasserman, B.A.; Lakatta, E.G.; Steinman, D.A. Effect of Common Carotid Artery Inlet Length on Normal Carotid Bifurcation Hemodynamics. J. Biomech. Eng. 2010, 132, 121008. [Google Scholar] [CrossRef]
- Subramaniam, T.; Rasani, M.R. Pulsatile CFD Numerical Simulation to Investigate the Effect of Various Degree and Position of Stenosis on Carotid Artery Hemodynamics. J. Adv. Res. Appl. Sci. Eng. Technol. 2022, 26, 29–40. [Google Scholar] [CrossRef]
- Nithiarasu, P.; Bevan, R.; Sazonov, I.; Van Loon, R.; Luckraz, H.; Collins, M.; Garnham, A. Influences of Domain Extensions to a Moderately Stenosed Patient-Specific Carotid Bifurcation: Investigation of Wall Quantities. Int. J. Numer. Methods Heat Fluid Flow 2011, 21, 952–979. [Google Scholar] [CrossRef]
- Morbiducci, U.; Gallo, D.; Massai, D.; Consolo, F.; Ponzini, R.; Antiga, L.; Bignardi, C.; Deriu, M.A.; Redaelli, A. Outflow Conditions for Image-Based Hemodynamic Models of the Carotid Bifurcation: Implications for Indicators of Abnormal Flow. J. Biomech. Eng. 2010, 132, 091005. [Google Scholar] [CrossRef] [PubMed]
- Rayz, V.L.; Berger, S.A.; Saloner, D. Transitional Flows in Arterial Fluid Dynamics. Comput. Methods Appl. Mech. Eng. 2007, 196, 3043–3048. [Google Scholar] [CrossRef]
- Raghuvir Pai, B.; Ayachit, A.; Abdul Khader, S.M.; Ahmed, K.A.; Rao, V.R.K.; Kamath, S.G.; Gupta, P.D. Effect of Postural Changes on Normal and Stenosed Common Carotid Artery Using FSI. Australas. Phys. Eng. Sci. Med. 2014, 37, 139–152. [Google Scholar] [CrossRef]
- Kim, C.S.; Kiris, C.; Kwak, D.; David, T. Numerical Simulation of Local Blood Flow in the Carotid and Cerebral Arteries Under Altered Gravity. J. Biomech. Eng. 2006, 128, 194–202. [Google Scholar] [CrossRef]
- Fung, Y.C. Biodynamics; Springer: New York, NY, USA, 1984. [Google Scholar] [CrossRef]
- Ferziger, J.H.; Perić, M. Computational Methods for Fluid Dynamics; Springer: Berlin/Heidelberg, Germany, 2002. [Google Scholar] [CrossRef]
- Khan, M.; Sardar, H.; Gulzar, M.M.; Alshomrani, A.S. On Multiple Solutions of Non-Newtonian Carreau Fluid Flow over an Inclined Shrinking Sheet. Results Phys. 2018, 8, 926–932. [Google Scholar] [CrossRef]
- Weddell, J.C.; Kwack, J.H.; Imoukhuede, P.I.; Masud, A. Hemodynamic Analysis in an Idealized Artery Tree: Differences in Wall Shear Stress between Newtonian and Non-Newtonian Blood Models. PLoS ONE 2015, 10, e0124575. [Google Scholar] [CrossRef]
- Gallo, D.; Steinman, D.A.; Bijari, P.B.; Morbiducci, U. Helical Flow in Carotid Bifurcation as Surrogate Marker of Exposure to Disturbed Shear. J. Biomech. 2012, 45, 2398–2404. [Google Scholar] [CrossRef]
- Li, C.H.; Gao, B.L.; Wang, J.W.; Liu, J.F.; Li, H.; Yang, S.T. Hemodynamic Factors Affecting Carotid Sinus Atherosclerotic Stenosis. World Neurosurg. 2019, 121, e262–e276. [Google Scholar] [CrossRef]
- Khader, S.M.A.; Kumar, N.; Pai, R.; Khader, S.M.A.; Kumar, N.; Pai, R. Fluid Structure Interaction Study of Stenosed Carotid Artery Considering the Effects of Blood Pressure and Altered Gravity. In Finite Element Methods and Their Applications; IntechOpen: London, UK, 2020. [Google Scholar] [CrossRef]
- Ningappa, A.H.; Patil, S.; Belur, G.S.; Barboza, A.B.V.; Kumar, N.; Ballambat, R.P.; Basri, A.A.; Khader, S.M.A.; Tamagawa, M. Influence of Altered Pressures on Flow Dynamics in Carotid Bifurcation System Using Numerical Methods. J. Adv. Res. Fluid Mech. Therm. Sci. 2022, 97, 47–61. [Google Scholar] [CrossRef]
- Perktold, K.; Rappitsch, G. Computer Simulation of Local Blood Flow and Vessel Mechanics in a Compliant Carotid Artery Bifurcation Model. Biomechanics 1995, 28, 845–856. [Google Scholar] [CrossRef]
- Gharahi, H.; Zambrano, B.A.; Zhu, D.C.; DeMarco, J.K.; Baek, S. Computational Fluid Dynamic Simulation of Human Carotid Artery Bifurcation Based on Anatomy and Volumetric Blood Flow Rate Measured with Magnetic Resonance Imaging. Int. J. Adv. Eng. Sci. Appl. Math. 2016, 8, 46–60. [Google Scholar] [CrossRef]
- Sousa, L.C.; Castro, C.F.; António, C.C.; Sousa, F.; Santos, R.; Castro, P.; Azevedo, E. Computational Simulation of Carotid Stenosis and Flow Dynamics Based on Patient Ultrasound Data—A New Tool for Risk Assessment and Surgical Planning. Adv. Med. Sci. 2016, 61, 32–39. [Google Scholar] [CrossRef]
- Harrison, G.J.; How, T.V.; Poole, R.J.; Brennan, J.A.; Naik, J.B.; Vallabhaneni, S.R.; Fisher, R.K. Closure Technique after Carotid Endarterectomy Influences Local Hemodynamics. J. Vasc. Surg. 2014, 60, 418–427. [Google Scholar] [CrossRef]
- Ku, D.N.; Giddens, D.P.; Zarins, C.K.; Glagov, S. Pulsatile Flow and Atherosclerosis in the Human Carotid Bifurcation. Positive Correlation between Plaque Location and Low Oscillating Shear Stress. Arterioscler. Off. J. Am. Heart Assoc. Inc. 1985, 5, 293–302. [Google Scholar] [CrossRef]
- He, X.; Ku, D.N. Pulsatile Flow in the Human Left Coronary Artery Bifurcation: Average Conditions. J. Biomech. Eng. 1996, 118, 74–82. [Google Scholar] [CrossRef]







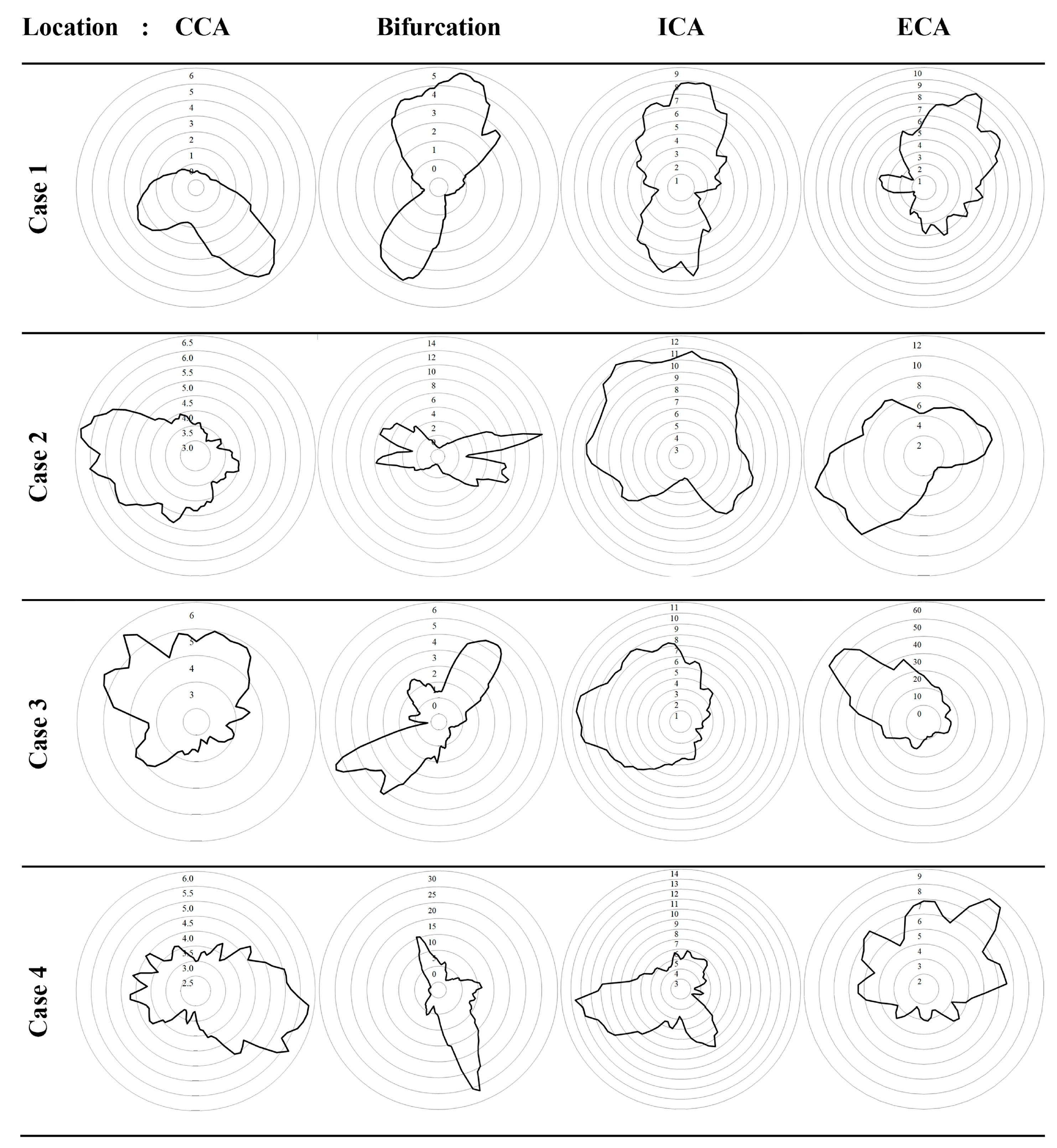


| Case | CCA Diameter (mm) | Reynolds Number (Mean Velocity) | Reynolds Number (Peak Velocity) | Womersley Number (α) |
|---|---|---|---|---|
| Case 1 | 7.33 | 579 | 1156 | 5.66 |
| Case 2 | 8.03 | 635 | 1266 | 6.21 |
| Case 3 | 5.14 | 405 | 810 | 3.97 |
| Case 4 | 6.13 | 483 | 966 | 4.74 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Ningappa, A.H.; Khader, S.M.A.; Kamat, H.; Tamagawa, M.; Kamath, G.; Pai B., R.; Koteswar, P.; Badruddin, I.A.; Zuber, M.; Mathias, K.A.; et al. Patient-Specific CFD Analysis of Carotid Artery Haemodynamics: Impact of Anatomical Variations on Atherosclerotic Risk. Computation 2026, 14, 77. https://doi.org/10.3390/computation14040077
Ningappa AH, Khader SMA, Kamat H, Tamagawa M, Kamath G, Pai B. R, Koteswar P, Badruddin IA, Zuber M, Mathias KA, et al. Patient-Specific CFD Analysis of Carotid Artery Haemodynamics: Impact of Anatomical Variations on Atherosclerotic Risk. Computation. 2026; 14(4):77. https://doi.org/10.3390/computation14040077
Chicago/Turabian StyleNingappa, Abhilash Hebbandi, S. M. Abdul Khader, Harishkumar Kamat, Masaaki Tamagawa, Ganesh Kamath, Raghuvir Pai B., Prakashini Koteswar, Irfan Anjum Badruddin, Mohammad Zuber, Kevin Amith Mathias, and et al. 2026. "Patient-Specific CFD Analysis of Carotid Artery Haemodynamics: Impact of Anatomical Variations on Atherosclerotic Risk" Computation 14, no. 4: 77. https://doi.org/10.3390/computation14040077
APA StyleNingappa, A. H., Khader, S. M. A., Kamat, H., Tamagawa, M., Kamath, G., Pai B., R., Koteswar, P., Badruddin, I. A., Zuber, M., Mathias, K. A., & Baloor, G. S. (2026). Patient-Specific CFD Analysis of Carotid Artery Haemodynamics: Impact of Anatomical Variations on Atherosclerotic Risk. Computation, 14(4), 77. https://doi.org/10.3390/computation14040077








