Quantification of Microplastics in North-Western Mediterranean Harbors: Seasonality and Biofilm-Related Metallic Contaminants
Abstract
1. Introduction
2. Materials and Methods
2.1. Sampling
2.2. Sample Treatment
3. Results and Discussion
3.1. Microplastics Areal Concentrations in the North-Western Mediterranean Harbors
3.2. Spatial Variability of Microplastics Areal Concentrations in the North-Western Mediterranean Harbors
3.3. Influence of Seasonality on Microplastics Areal Concentrations
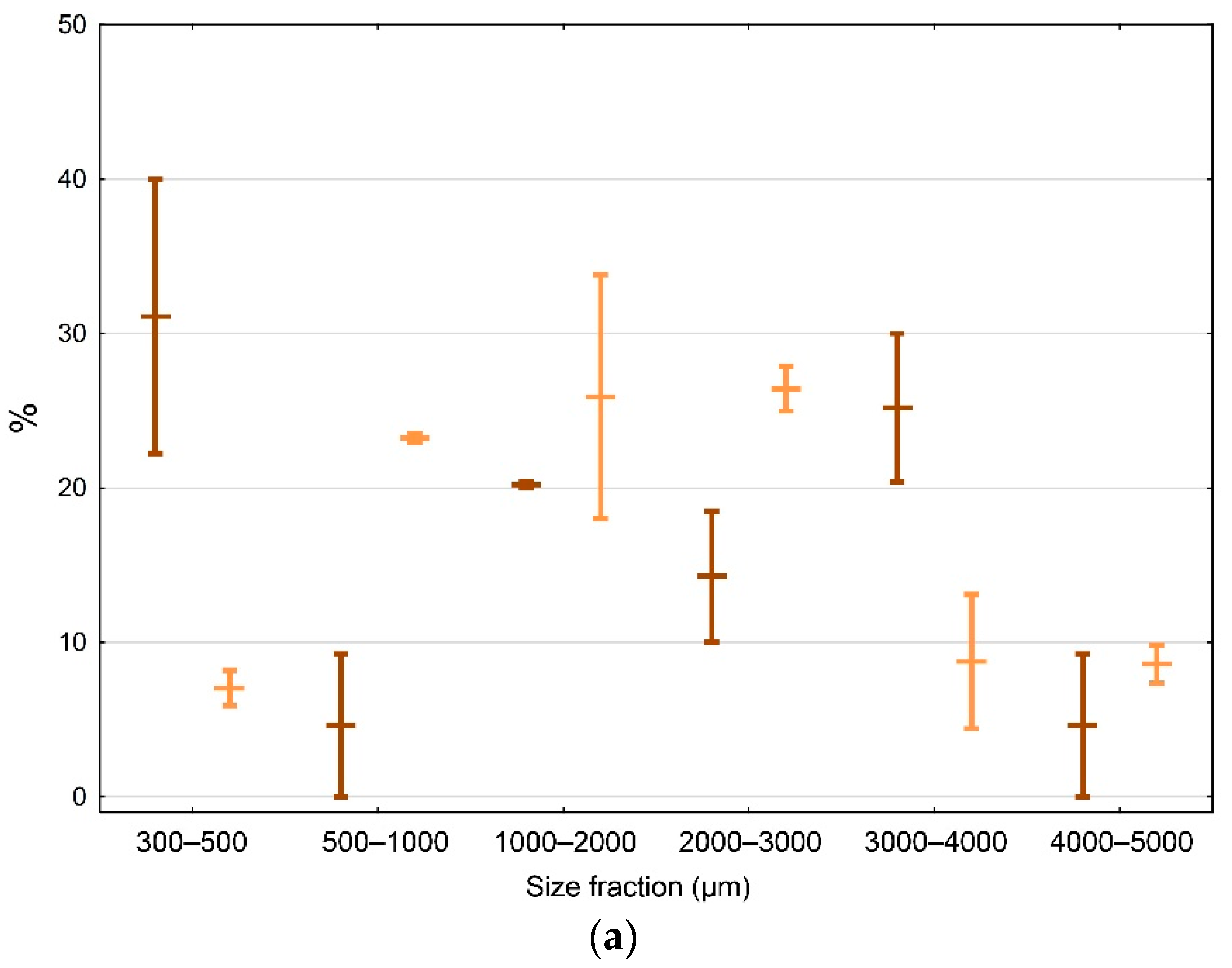
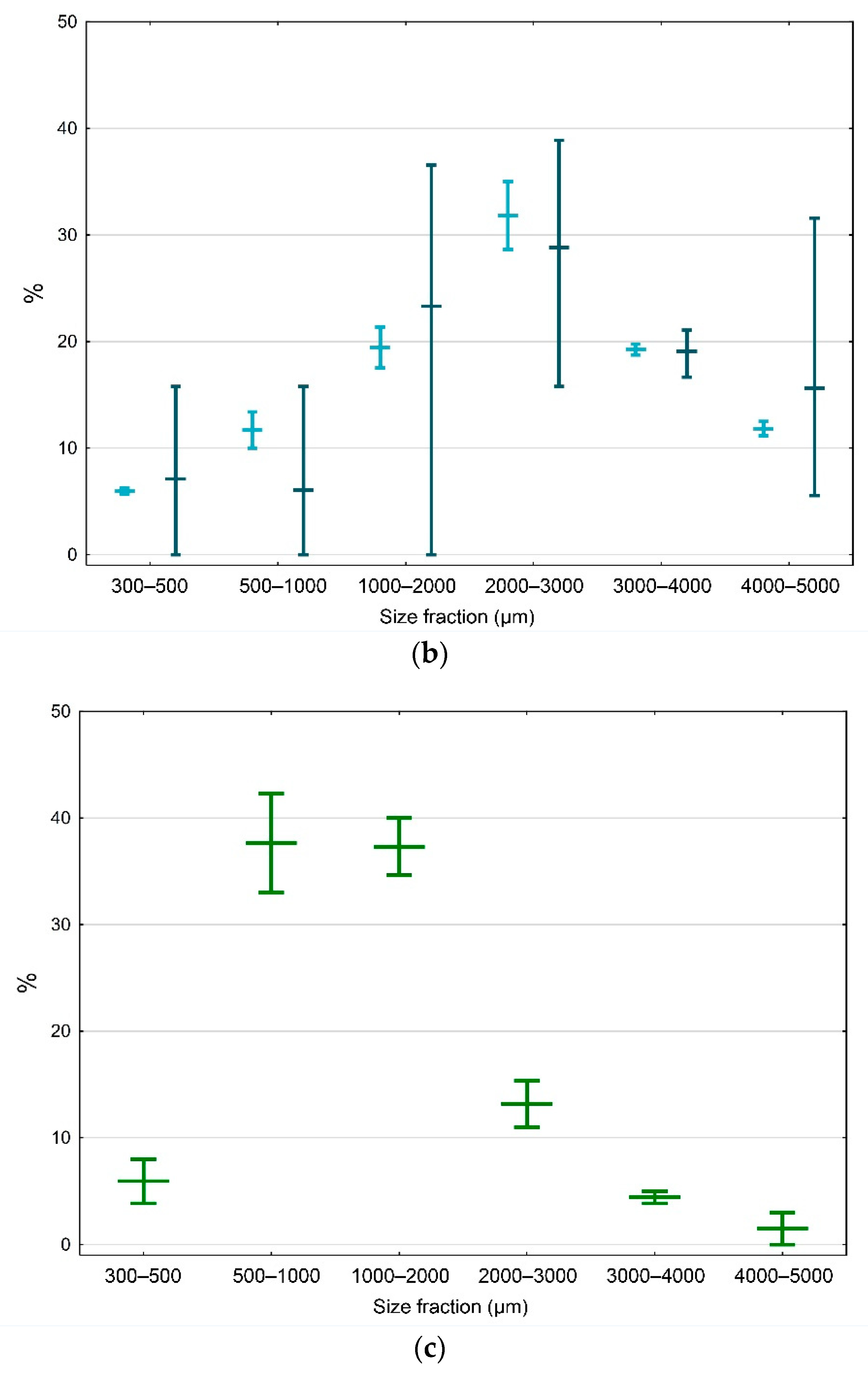
3.4. Size Distribution of Microplastics in the North-Western Mediterranean Harbors
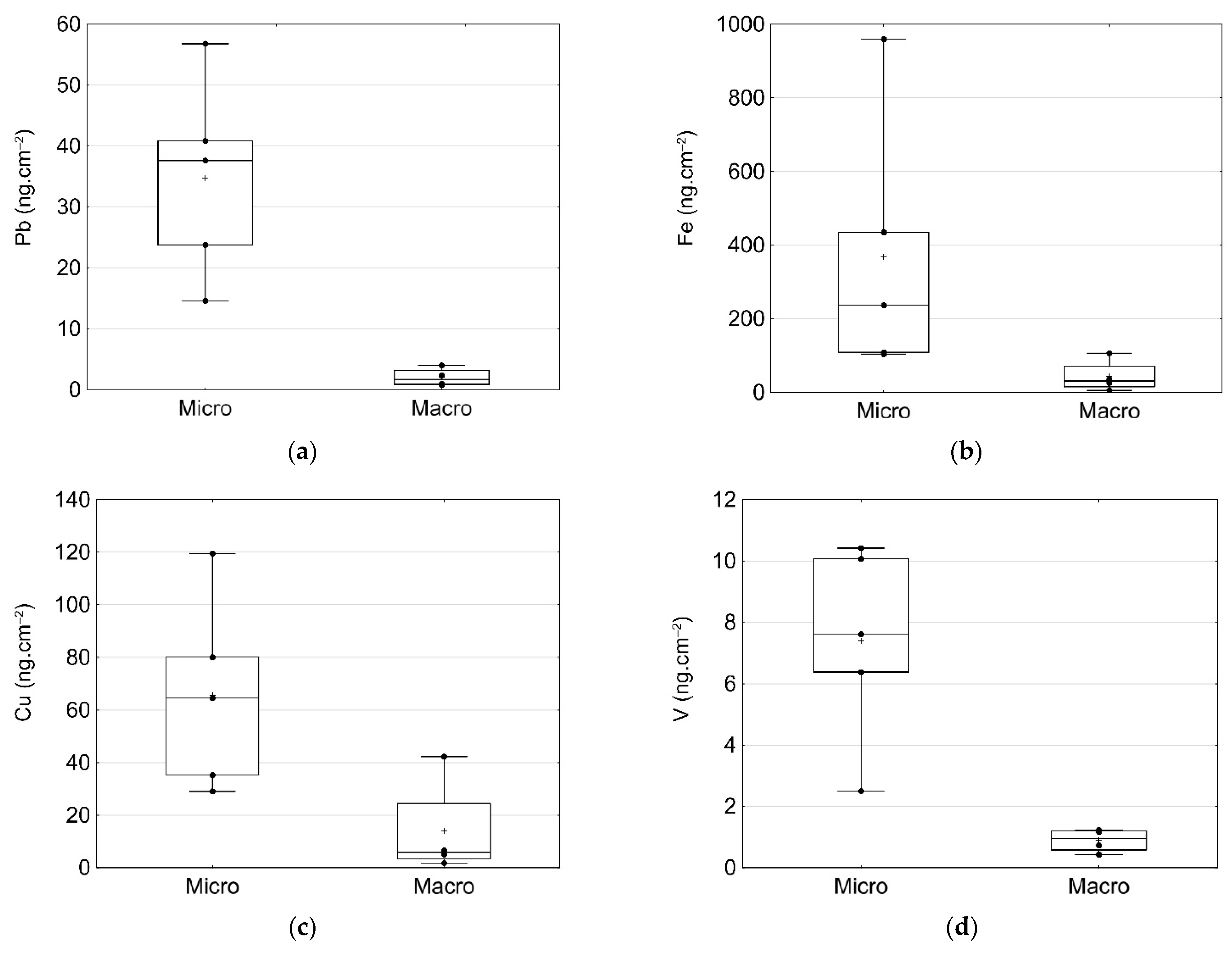
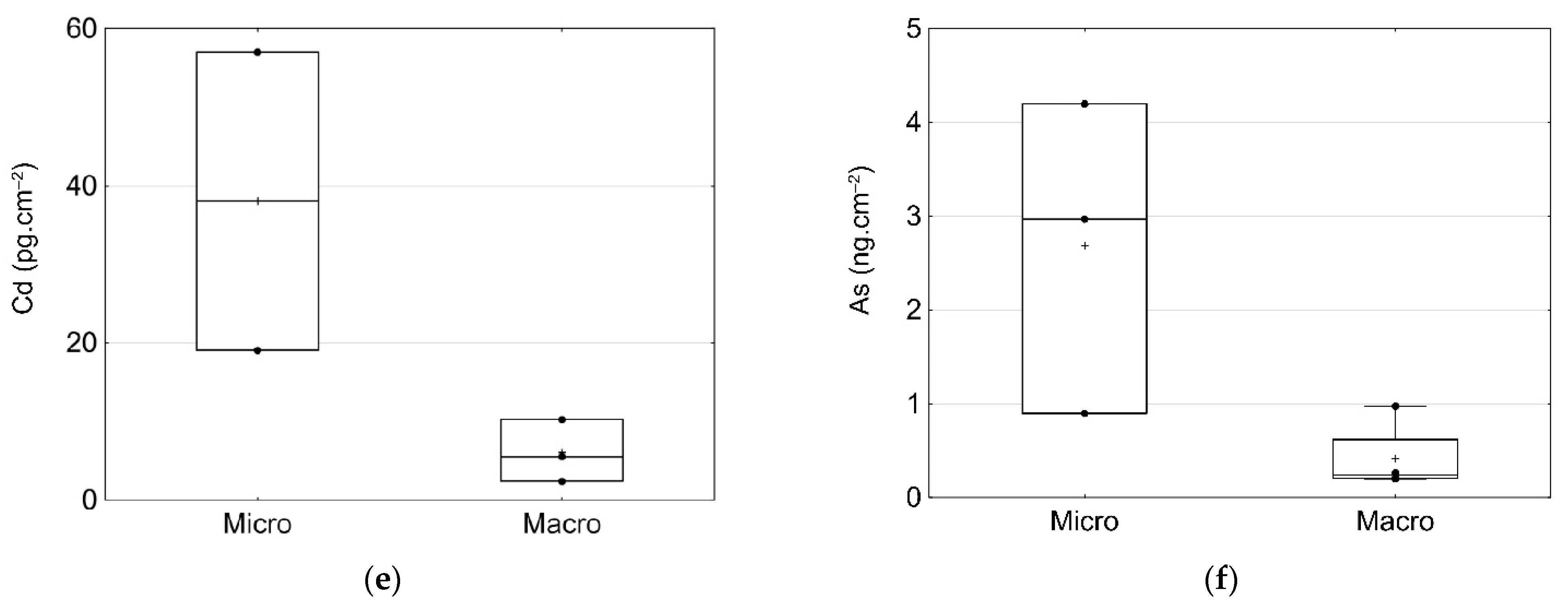
3.5. Inorganic Contaminants
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Goldstein, M.C.; Rosenberg, M.; Cheng, L. Increased Oceanic Microplastic Debris Enhances Oviposition in an Endemic Pelagic Insect. Biol. Lett. 2012, 8, 817–820. [Google Scholar] [CrossRef]
- Cole, M.; Lindeque, P.; Halsband, C.; Galloway, T.S. Microplastics as Contaminants in the Marine Environment: A Review. Mar. Pollut. Bull. 2011, 62, 2588–2597. [Google Scholar] [CrossRef] [PubMed]
- Coll, M.; Piroddi, C.; Steenbeek, J.; Kaschner, K.; Ben Rais Lasram, F.; Aguzzi, J.; Ballesteros, E.; Bianchi, C.N.; Corbera, J.; Dailianis, T.; et al. The Biodiversity of the Mediterranean Sea: Estimates, Patterns, and Threats. PLoS ONE 2010, 5, e11842. [Google Scholar] [CrossRef]
- Lotze, H.K.; Coll, M.; Dunne, J.A. Historical Changes in Marine Resources, Food-Web Structure and Ecosystem Functioning in the Adriatic Sea, Mediterranean. Ecosystems 2011, 14, 198–222. [Google Scholar] [CrossRef]
- De Madron, X.D.; Guieu, C.; Sempéré, R.; Conan, P.; Cossa, D.; D’Ortenzio, F.; Estournel, C.; Gazeau, F.; Rabouille, C.; Stemmann, L.; et al. Marine Ecosystems’ Responses to Climatic and Anthropogenic Forcings in the Mediterranean. Prog. Oceanogr. 2011, 91, 97–166. [Google Scholar] [CrossRef]
- Cózar, A.; Sanz-Martín, M.; Martí, E.; González-Gordillo, J.I.; Ubeda, B.; Gálvez, J.Á.; Irigoien, X.; Duarte, C.M. Plastic Accumulation in the Mediterranean Sea. PLoS ONE 2015, 10, e0121762. [Google Scholar] [CrossRef]
- Van Sebille, E.; Wilcox, C.; Lebreton, L.; Maximenko, N.; Hardesty, B.D.; van Franeker, J.A.; Eriksen, M.; Siegel, D.; Galgani, F.; Law, K.L. A Global Inventory of Small Floating Plastic Debris. Environ. Res. Lett. 2015, 10, 124006. [Google Scholar] [CrossRef]
- Moore, C.J.; Moore, S.L.; Leecaster, M.K.; Weisberg, S.B. A Comparison of Plastic and Plankton in the North Pacific Central Gyre. Mar. Pollut. Bull. 2001, 42, 1297–1300. [Google Scholar] [CrossRef]
- Jambeck, J.R.; Geyer, R.; Wilcox, C.; Siegler, T.R.; Perryman, M.; Andrady, A.; Narayan, R.; Law, K.L. Plastic Waste Inputs from Land into the Ocean. Science 2015, 347, 768–771. [Google Scholar] [CrossRef]
- Pedrotti, M.L.; Petit, S.; Elineau, A.; Bruzaud, S.; Crebassa, J.-C.; Dumontet, B.; Martí, E.; Gorsky, G.; Cózar, A. Changes in the Floating Plastic Pollution of the Mediterranean Sea in Relation to the Distance to Land. PLoS ONE 2016, 11, e0161581. [Google Scholar] [CrossRef]
- Ross, J.B.; Parker, R.; Strickland, M. A Survey of Shoreline Litter in Halifax Harbour 1989. Mar. Pollut. Bull. 1991, 22, 245–248. [Google Scholar] [CrossRef]
- Glaser, J.A. The Importance of Biofilms to the Fate and Effects of Microplastics. In Bacterial Biofilms; Dincer, S., Sümengen Özdenefe, M., Arkut, A., Eds.; IntechOpen: London, UK, 2020. [Google Scholar] [CrossRef]
- Flemming, H.-C.; Wingender, J. The Biofilm Matrix. Nat. Rev. Microbiol. 2010, 8, 623–633. [Google Scholar] [CrossRef]
- Duong, T.T.; Morin, S.; Coste, M.; Herlory, O.; Feurtet-Mazel, A.; Boudou, A. Experimental Toxicity and Bioaccumulation of Cadmium in Freshwater Periphytic Diatoms in Relation with Biofilm Maturity. Sci. Total Environ. 2010, 408, 552–562. [Google Scholar] [CrossRef]
- Richard, H.; Carpenter, E.J.; Komada, T.; Palmer, P.T.; Rochman, C.M. Biofilm Facilitates Metal Accumulation onto Microplastics in Estuarine Waters. Sci. Total Environ. 2019, 683, 600–608. [Google Scholar] [CrossRef]
- Rochman, C.M.; Hentschel, B.T.; Teh, S.J. Long-Term Sorption of Metals Is Similar among Plastic Types: Implications for Plastic Debris in Aquatic Environments. PLoS ONE 2014, 9, e85433. [Google Scholar] [CrossRef]
- Roane, T.M.; Pepper, I.L.; Gentry, T.J. Microorganisms and Metal Pollutants. In Environmental Microbiology; Elsevier: Amsterdam, The Netherlands, 2015; pp. 415–439. [Google Scholar] [CrossRef]
- Mazoyer, C.; Vanneste, H.; Dufresne, C.; Ourmières, Y.; Magaldi, M.G.; Molcard, A. Impact of Wind-Driven Circulation on Contaminant Dispersion in a Semi-Enclosed Bay. Estuar. Coast. Shelf Sci. 2020, 233, 106529. [Google Scholar] [CrossRef]
- Cutroneo, L.; Carbone, C.; Consani, S.; Vagge, G.; Canepa, G.; Capello, M. Environmental Complexity of a Port: Evidence from Circulation of the Water Masses, and Composition and Contamination of Bottom Sediments. Mar. Pollut. Bull. 2017, 119, 184–194. [Google Scholar] [CrossRef] [PubMed]
- Cutroneo, L.; Reboa, A.; Besio, G.; Borgogno, F.; Canesi, L.; Canuto, S.; Dara, M.; Enrile, F.; Forioso, I.; Greco, G.; et al. Correction to: Microplastics in Seawater: Sampling Strategies, Laboratory Methodologies, and Identification Techniques Applied to Port Environment. Environ. Sci. Pollut. Res. 2020, 27, 8938–8952. [Google Scholar] [CrossRef] [PubMed]
- Covernton, G.A.; Pearce, C.M.; Gurney-Smith, H.J.; Chastain, S.G.; Ross, P.S.; Dower, J.F.; Dudas, S.E. Size and Shape Matter: A Preliminary Analysis of Microplastic Sampling Technique in Seawater Studies with Implications for Ecological Risk Assessment. Sci. Total Environ. 2019, 667, 124–132. [Google Scholar] [CrossRef]
- Gorsky, G.; Ohman, M.D.; Picheral, M.; Gasparini, S.; Stemmann, L.; Romagnan, J.-B.; Cawood, A.; Pesant, S.; Garcia-Comas, C.; Prejger, F. Digital Zooplankton Image Analysis Using the ZooScan Integrated System. J. Plankton Res. 2010, 32, 285–303. [Google Scholar] [CrossRef]
- De Haan, W.P.; Sanchez-Vidal, A.; Canals, M. Floating Microplastics and Aggregate Formation in the Western Mediterranean Sea. Mar. Pollut. Bull. 2019, 140, 523–535. [Google Scholar] [CrossRef]
- Schmidt, N.; Thibault, D.; Galgani, F.; Paluselli, A.; Sempéré, R. Occurrence of Microplastics in Surface Waters of the Gulf of Lion (NW Mediterranean Sea). Prog. Oceanogr. 2018, 163, 214–220. [Google Scholar] [CrossRef]
- Moore, C.J.; Moore, S.L.; Weisberg, S.B.; Lattin, G.L.; Zellers, A.F. A Comparison of Neustonic Plastic and Zooplankton Abundance in Southern California’s Coastal Waters. Mar. Pollut. Bull. 2002, 44, 1035–1038. [Google Scholar] [CrossRef]
- Antonella, A.; Léa, D.; Alex, A.; Fabrizio, A.; Asunción, B.; Ilaria, C.; Lara, C.; Roberto, C.; Gaëlle, D.; Delphine, G.; et al. Floating Marine Macro Litter: Density Reference Values and Monitoring Protocol Settings from Coast to Offshore. Results from the MEDSEALITTER Project. Mar. Pollut. Bull. 2020, 160, 111647. [Google Scholar] [CrossRef]
- Claessens, M.; Meester, S.D.; Landuyt, L.V.; Clerck, K.D.; Janssen, C.R. Occurrence and Distribution of Microplastics in Marine Sediments along the Belgian Coast. Mar. Pollut. Bull. 2011, 62, 2199–2204. [Google Scholar] [CrossRef]
- Desforges, J.-P.W.; Galbraith, M.; Dangerfield, N.; Ross, P.S. Widespread Distribution of Microplastics in Subsurface Seawater in the NE Pacific Ocean. Mar. Pollut. Bull. 2014, 79, 94–99. [Google Scholar] [CrossRef]
- Capello, M.; Cutroneo, L.; Castellano, M.; Orsi, M.; Pieracci, A.; Maria Bertolotto, R.; Povero, P.; Tucci, S. Physical and Sedimentological Characterisation of Dredged Sediments. Chem. Ecol. 2010, 26 (Suppl. 1), 359–369. [Google Scholar] [CrossRef]
- Boucher, J.; Friot, D. Primary Microplastics in the Oceans: A Global Evaluation of Sources; IUCN International Union for Conservation of Nature: Gland, Switzerland, 2017. [Google Scholar] [CrossRef]
- Zhang, H. Transport of Microplastics in Coastal Seas. Estuar. Coast. Shelf Sci. 2017, 199, 74–86. [Google Scholar] [CrossRef]
- Auta, H.S.; Emenike, C.U.; Fauziah, S.H. Distribution and Importance of Microplastics in the Marine Environment: A Review of the Sources, Fate, Effects, and Potential Solutions. Environ. Int. 2017, 102, 165–176. [Google Scholar] [CrossRef]
- Lebreton, L.C.M.; van der Zwet, J.; Damsteeg, J.-W.; Slat, B.; Andrady, A.; Reisser, J. River Plastic Emissions to the World’s Oceans. Nat. Commun. 2017, 8, 15611. [Google Scholar] [CrossRef]
- Castro-Jiménez, J.; González-Fernández, D.; Fornier, M.; Schmidt, N.; Sempéré, R. Macro-Litter in Surface Waters from the Rhone River: Plastic Pollution and Loading to the NW Mediterranean Sea. Mar. Pollut. Bull. 2019, 146, 60–66. [Google Scholar] [CrossRef]
- Galgani, F.; Hanke, G.; Werner, S.; De Vrees, L. Marine Litter within the European Marine Strategy Framework Directive. ICES J. Mar. Sci. 2013, 70, 1055–1064. [Google Scholar] [CrossRef]
- Isobe, A.; Uchida, K.; Tokai, T.; Iwasaki, S. East Asian Seas: A Hot Spot of Pelagic Microplastics. Mar. Pollut. Bull. 2015, 101, 618–623. [Google Scholar] [CrossRef] [PubMed]
- Defontaine, S.; Sous, D.; Tesan, J.; Monperrus, M.; Lenoble, V.; Lanceleur, L. Microplastics in a Salt-Wedge Estuary: Vertical Structure and Tidal Dynamics. Mar. Pollut. Bull. 2020, 160, 111688. [Google Scholar] [CrossRef]
- Cozar, A.; Echevarria, F.; Gonzalez-Gordillo, J.I.; Irigoien, X.; Ubeda, B.; Hernandez-Leon, S.; Palma, A.T.; Navarro, S.; Garcia-de-Lomas, J.; Ruiz, A.; et al. Plastic Debris in the Open Ocean. Proc. Natl. Acad. Sci. USA 2014, 111, 10239–10244. [Google Scholar] [CrossRef] [PubMed]
- Weinstein, J.E.; Crocker, B.K.; Gray, A.D. From Macroplastic to Microplastic: Degradation of High-Density Polyethylene, Polypropylene, and Polystyrene in a Salt Marsh Habitat: Degradation of Plastic in a Salt Marsh Habitat. Environ. Toxicol. Chem. 2016, 35, 1632–1640. [Google Scholar] [CrossRef]
- Andrady, A.L. Microplastics in the Marine Environment. Mar. Pollut. Bull. 2011, 62, 1596–1605. [Google Scholar] [CrossRef] [PubMed]
- Lindeque, P.K.; Cole, M.; Coppock, R.L.; Lewis, C.N.; Miller, R.Z.; Watts, A.J.R.; Wilson-McNeal, A.; Wright, S.L.; Galloway, T.S. Are We Underestimating Microplastic Abundance in the Marine Environment? A Comparison of Microplastic Capture with Nets of Different Mesh-Size. Environ. Pollut. 2020, 265, 114721. [Google Scholar] [CrossRef]
- Wright, R.J.; Erni-Cassola, G.; Zadjelovic, V.; Latva, M.; Christie-Oleza, J.A. Marine Plastic Debris: A New Surface for Microbial Colonization. Environ. Sci. Technol. 2020, 54, 11657–11672. [Google Scholar] [CrossRef]
- Kooi, M.; van Nes, E.H.; Scheffer, M.; Koelmans, A.A. Ups and Downs in the Ocean: Effects of Biofouling on Vertical Transport of Microplastics. Environ. Sci. Technol. 2017, 51, 7963–7971. [Google Scholar] [CrossRef]
- Lehtiniemi, M.; Hartikainen, S.; Näkki, P.; Engström-Öst, J.; Koistinen, A.; Setälä, O. Size Matters More than Shape: Ingestion of Primary and Secondary Microplastics by Small Predators. Food Webs 2018, 17, e00097. [Google Scholar] [CrossRef]
- Pellini, G.; Gomiero, A.; Fortibuoni, T.; Ferrà, C.; Grati, F.; Tassetti, A.N.; Polidori, P.; Fabi, G.; Scarcella, G. Characterization of Microplastic Litter in the Gastrointestinal Tract of Solea Solea from the Adriatic Sea. Environ. Pollut. 2018, 234, 943–952. [Google Scholar] [CrossRef]
- Leslie, H.A.; Brandsma, S.H.; van Velzen, M.J.M.; Vethaak, A.D. Microplastics En Route: Field Measurements in the Dutch River Delta and Amsterdam Canals, Wastewater Treatment Plants, North Sea Sediments and Biota. Environ. Int. 2017, 101, 133–142. [Google Scholar] [CrossRef]
- Simpson, S.; Batley, G.E. Sediment Quality Assessment: A Practical Guide, 2nd ed.; Simpson, S., Batley, G.E., Eds.; CSIRO Publishing: Clayton, Australia, 2015. [Google Scholar]
- Laumond, F.; Copin-Montegut, G.; Courau, P.; Nicolas, E. Cadmium, Copper and Lead in the Western Mediterranean Sea. Mar. Chem. 1984, 15, 251–261. [Google Scholar] [CrossRef]
- Orani, A.M.; Barats, A.; Zitte, W.; Morrow, C.; Thomas, O.P. Comparative Study on the Bioaccumulation and Biotransformation of Arsenic by Some Northeastern Atlantic and Northwestern Mediterranean Sponges. Chemosphere 2018, 201, 826–839. [Google Scholar] [CrossRef]
- Sherrell, R.M.; Boyle, E.A. Zinc, Chromium, Vanadium and Iron in the Mediterranean Sea. Deep Sea Research Part A. Oceanogr. Res. Pap. 1988, 35, 1319–1334. [Google Scholar] [CrossRef]
- Friese, K.; Mages, M.; Wendt-Potthoff, K.; Neu, T.R. Determination of Heavy Metals in Biofilms from the River Elbe by Total-Reflection X-Ray Fluorescence Spectrometry. Spectrochim. Acta Part B At. Spectrosc. 1997, 52, 1019–1025. [Google Scholar] [CrossRef]
- Tien, C.-J.; Chen, C.S. Patterns of Metal Accumulation by Natural River Biofilms during Their Growth and Seasonal Succession. Arch. Environ. Contam. Toxicol. 2013, 64, 605–616. [Google Scholar] [CrossRef] [PubMed]
- Prunier, J.; Maurice, L.; Perez, E.; Gigault, J.; Pierson Wickmann, A.-C.; Davranche, M.; Halle, A. ter. Trace Metals in Polyethylene Debris from the North Atlantic Subtropical Gyre. Environ. Pollut. 2019, 245, 371–379. [Google Scholar] [CrossRef] [PubMed]
- Tessier, E.; Garnier, C.; Mullot, J.-U.; Lenoble, V.; Arnaud, M.; Raynaud, M.; Mounier, S. Study of the Spatial and Historical Distribution of Sediment Inorganic Contamination in the Toulon Bay (France). Mar. Pollut. Bull. 2011, 62, 2075–2086. [Google Scholar] [CrossRef]
- Holmes, L.A.; Turner, A.; Thompson, R.C. Adsorption of Trace Metals to Plastic Resin Pellets in the Marine Environment. Environ. Pollut. 2012, 160, 42–48. [Google Scholar] [CrossRef]
- Ashton, K.; Holmes, L.; Turner, A. Association of Metals with Plastic Production Pellets in the Marine Environment. Mar. Pollut. Bull. 2010, 60, 2050–2055. [Google Scholar] [CrossRef] [PubMed]
- Coclet, C.; Garnier, C.; Durrieu, G.; Omanović, D.; D’Onofrio, S.; Le Poupon, C.; Mullot, J.-U.; Briand, J.-F.; Misson, B. Changes in Bacterioplankton Communities Resulting From Direct and Indirect Interactions With Trace Metal Gradients in an Urbanized Marine Coastal Area. Front. Microbiol. 2019, 10, 257. [Google Scholar] [CrossRef]
- Mato, Y.; Isobe, T.; Takada, H.; Kanehiro, H.; Ohtake, C.; Kaminuma, T. Plastic Resin Pellets as a Transport Medium for Toxic Chemicals in the Marine Environment. Environ. Sci. Technol. 2001, 35, 318–324. [Google Scholar] [CrossRef]
- Artham, T.; Sudhakar, M.; Venkatesan, R.; Madhavan Nair, C.; Murty, K.V.G.K.; Doble, M. Biofouling and Stability of Synthetic Polymers in Sea Water. Int. Biodeterior. Biodegrad. 2009, 63, 884–890. [Google Scholar] [CrossRef]
- Kalčíková, G.; Skalar, T.; Marolt, G.; Jemec Kokalj, A. An Environmental Concentration of Aged Microplastics with Adsorbed Silver Significantly Affects Aquatic Organisms. Water Res. 2020, 175, 115644. [Google Scholar] [CrossRef]
- Koelmans, A.A.; Besseling, E.; Foekema, E.M. Leaching of Plastic Additives to Marine Organisms. Environ. Pollut. 2014, 187, 49–54. [Google Scholar] [CrossRef]
- Brennecke, D.; Duarte, B.; Paiva, F.; Caçador, I.; Canning-Clode, J. Microplastics as Vector for Heavy Metal Contamination from the Marine Environment. Estuar. Coast. Shelf Sci. 2016, 178, 189–195. [Google Scholar] [CrossRef]
- Layglon, N.; Misson, B.; Durieu, G.; Coclet, C.; D’Onofrio, S.; Dang, D.H.; François, D.; Mullot, J.-U.; Mounier, S.; Lenoble, V.; et al. Long-Term Monitoring Emphasizes Impacts of the Dredging on Dissolved Cu and Pb Contamination along with Ultraplankton Distribution and Structure in Toulon Bay (NW Mediterranean Sea, France). Mar. Pollut. Bull. 2020, 156, 111196. [Google Scholar] [CrossRef] [PubMed]
- Cutroneo, L.; Castellano, M.; Pieracci, A.; Povero, P.; Tucci, S.; Capello, M. The Use of a Combined Monitoring System for Following a Turbid Plume Generated by Dredging Activities in a Port. J. Soils Sediments 2012, 12, 797–809. [Google Scholar] [CrossRef]
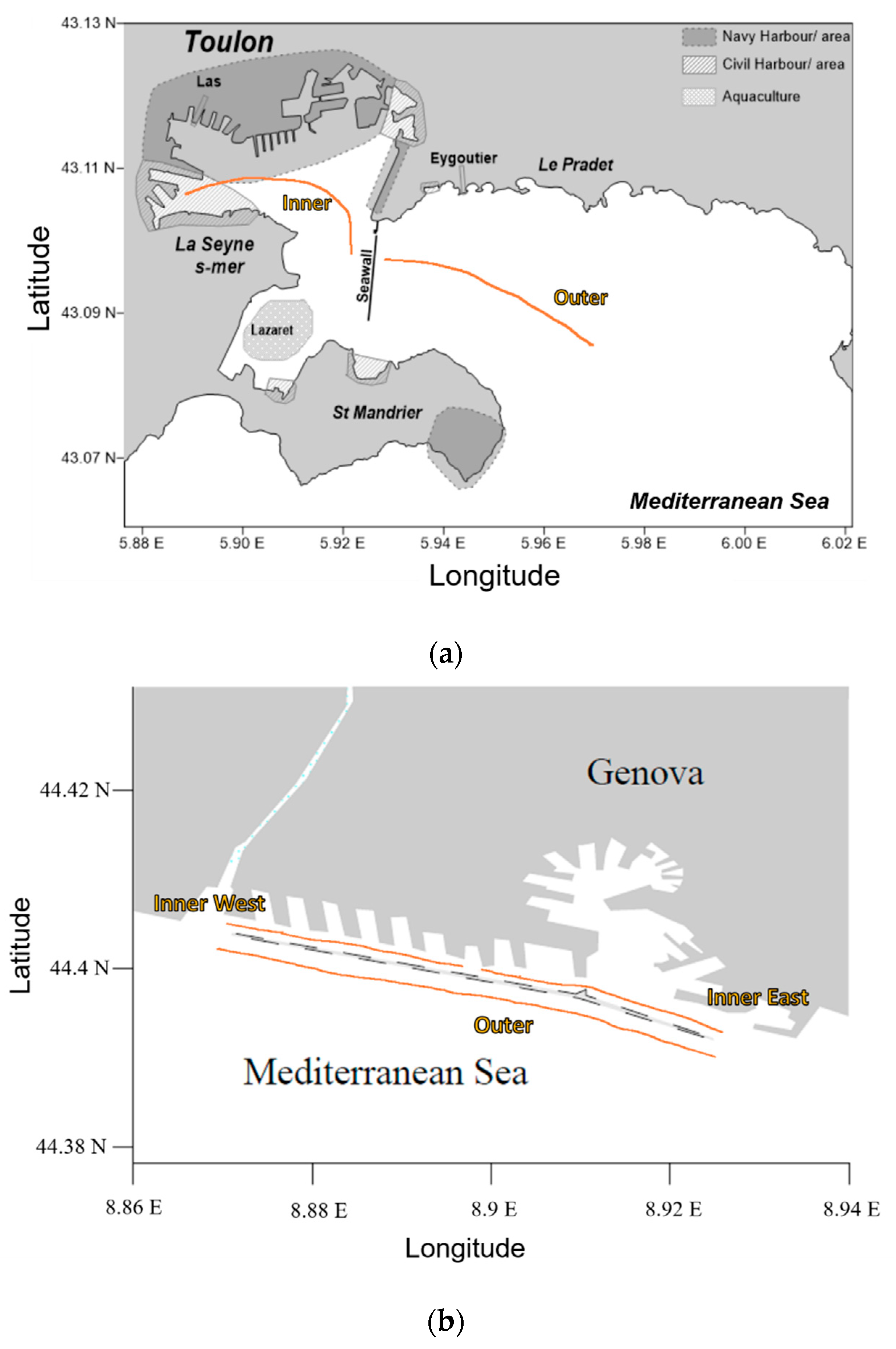
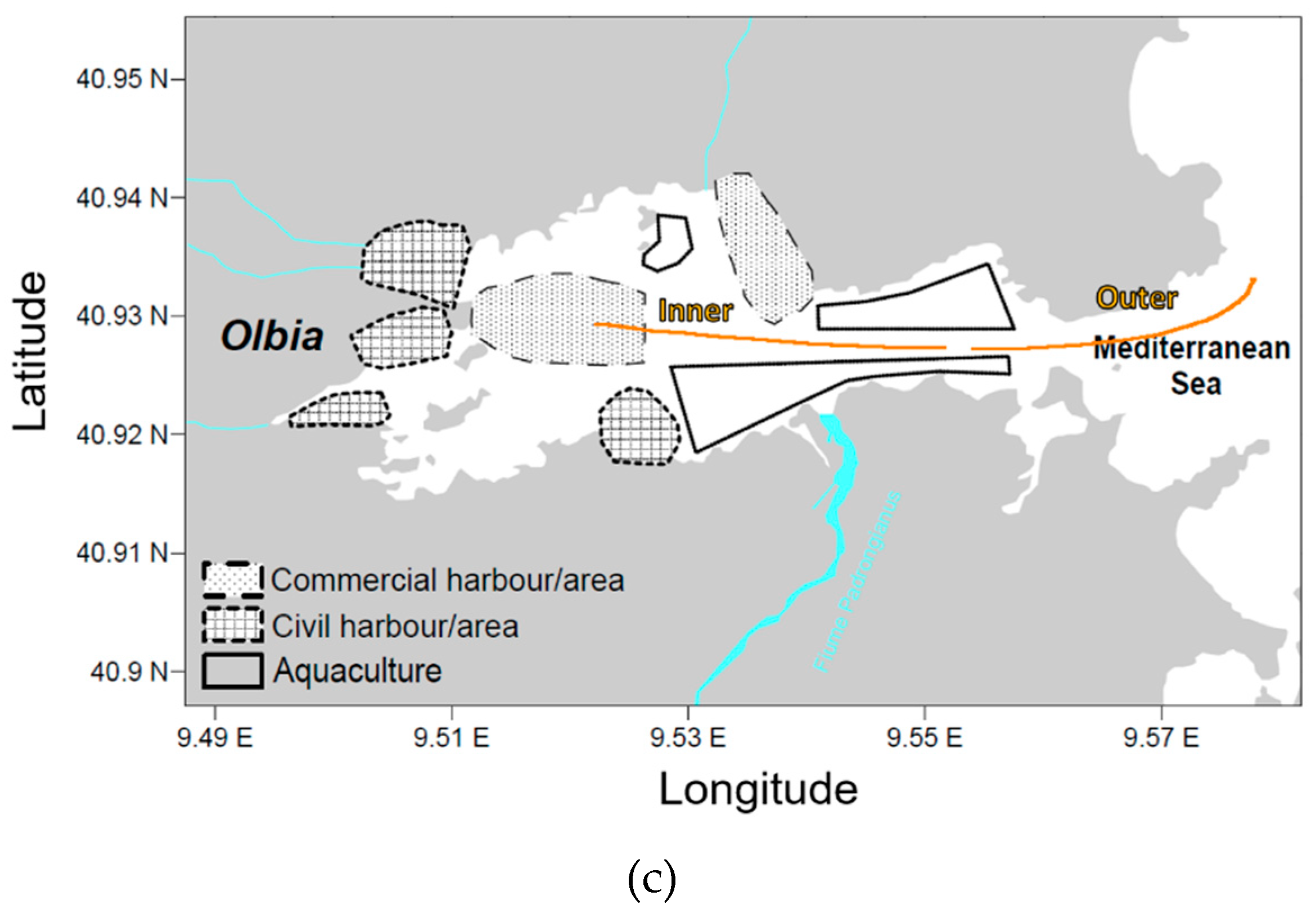
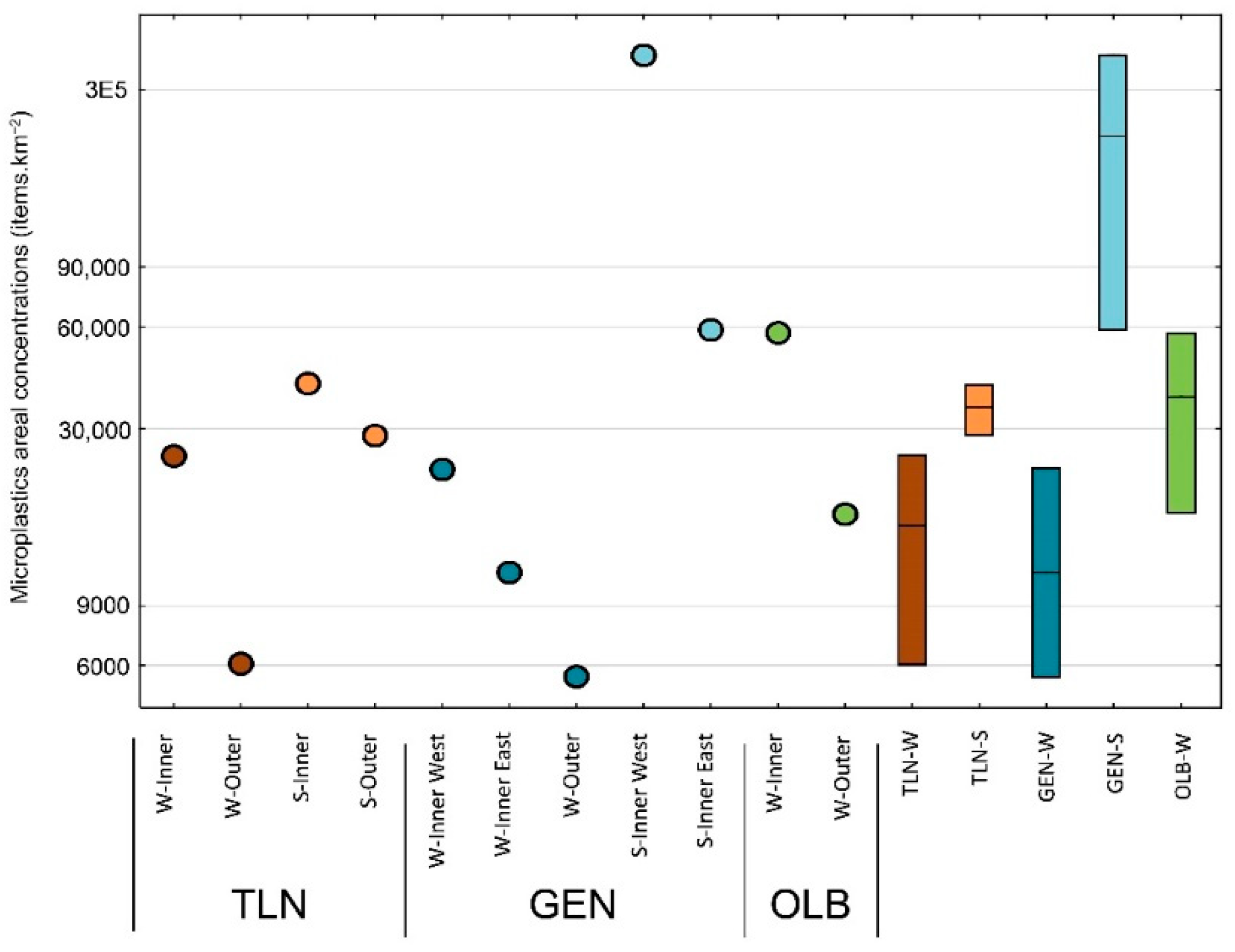
| Site | Season | Zone | Volume | Surface | Items | Concentrations | [g·km−2] |
|---|---|---|---|---|---|---|---|
| [m3] | [m2] | [Items·km−2] | |||||
| TLN | W | Inner | 217 | 2166 | 54 | 24,931 | 36.00 |
| Outer | 164 | 1644 | 10 | 6083 | 0.06 | ||
| TLN | S | Inner | 167 | 1666 | 68 | 40,816 | 33.20 |
| Outer | 213 | 2128 | 61 | 28,665 | 8.18 | ||
| GEN | W | Inner-West | 180 | 1800 | 41 | 22,778 | 15.00 |
| Inner-East | 168 | 1680 | 19 | 11,310 | 9.30 | ||
| Outer | 323 | 3228 | 18 | 5576 | 1.90 | ||
| GEN | S | Inner-West | 136 | 1158 | 440 | 379,965 | 551.00 |
| Outer | 116 | 1362 | 80 | 58,737 | 130.00 | ||
| OLB | W | Inner | 174 | 1736 | 100 | 57,604 | 6.96 |
| Outer | 155 | 1547 | 26 | 16,807 | 0.36 |
| Sample | Site | Pb | Fe | Cu | V | Cd | As |
|---|---|---|---|---|---|---|---|
| mg·kg−1 | mg·kg−1 | mg·kg−1 | mg·kg−1 | µg·kg−1 | µg·kg−1 | ||
| TLN-W-Inner | TLN | 1.8 | 22 | 2.54 | 1.53 | 3 | 862 |
| TLN-W-Outer | <DL | 181.8 | 46 | 1.982 | 2821 | 400.11 | |
| TLN-S-Inner | 0.66 | 8 | 15.1 | 1.11 | 24 | 444.35 | |
| TLN-S-Outer | 2.08 | 71.3 | 2.57 | 1.087 | 9.98 | 504 | |
| GEN-W-Inner West | GEN | 11.72 | 89.76 | 6 | 2.152 | <DL | 185 |
| GEN-W-Inner East | 3.78 | 37.6 | 12.74 | 0.4 | <DL | <DL | |
| GEN-W-Outer | 7.97 | 187 | 23.3 | 1.25 | <DL | <DL | |
| GEN-S-Inner West | 1.342 | 10 | 3.2 | 0.7 | 5.2 | 273 | |
| GEN-S-Inner East | 1.89 | 5.2 | 3.25 | 0.51 | 0.96 | 211 | |
| OLB-W-Inner | OLB | 3.79 | 80.4 | 0.728 | 0.309 | 1.7 | 258 |
| OLB-W-Outer | 12.98 | 25,393 | 29.6 | 21.5 | 176 | 969 | |
| Analysis of variance | H = 4.06 | H = 2.00 | H = 0.06 | H = 0.65 | H = 2.00 | H = 4.31 | |
| p = 0.131 | p = 0.367 | p = 0.968 | p = 0.720 | p = 0.367 | p = 0.115 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tesán Onrubia, J.A.; Djaoudi, K.; Borgogno, F.; Canuto, S.; Angeletti, B.; Besio, G.; Capello, M.; Cutroneo, L.; Stocchino, A.; Mounier, S.; et al. Quantification of Microplastics in North-Western Mediterranean Harbors: Seasonality and Biofilm-Related Metallic Contaminants. J. Mar. Sci. Eng. 2021, 9, 337. https://doi.org/10.3390/jmse9030337
Tesán Onrubia JA, Djaoudi K, Borgogno F, Canuto S, Angeletti B, Besio G, Capello M, Cutroneo L, Stocchino A, Mounier S, et al. Quantification of Microplastics in North-Western Mediterranean Harbors: Seasonality and Biofilm-Related Metallic Contaminants. Journal of Marine Science and Engineering. 2021; 9(3):337. https://doi.org/10.3390/jmse9030337
Chicago/Turabian StyleTesán Onrubia, Javier A., Kahina Djaoudi, Franco Borgogno, Susanna Canuto, Bernard Angeletti, Giovanni Besio, Marco Capello, Laura Cutroneo, Alessandro Stocchino, Stéphane Mounier, and et al. 2021. "Quantification of Microplastics in North-Western Mediterranean Harbors: Seasonality and Biofilm-Related Metallic Contaminants" Journal of Marine Science and Engineering 9, no. 3: 337. https://doi.org/10.3390/jmse9030337
APA StyleTesán Onrubia, J. A., Djaoudi, K., Borgogno, F., Canuto, S., Angeletti, B., Besio, G., Capello, M., Cutroneo, L., Stocchino, A., Mounier, S., & Lenoble, V. (2021). Quantification of Microplastics in North-Western Mediterranean Harbors: Seasonality and Biofilm-Related Metallic Contaminants. Journal of Marine Science and Engineering, 9(3), 337. https://doi.org/10.3390/jmse9030337










