Sublethal Effects of Crude Oil and Chemical Dispersants on Multiple Life History Stages of the Eastern Oyster, Crassostrea virginica
Abstract
1. Introduction
2. Materials and Methods
2.1. Preparation of WAF and CEWAF
2.2. Veliger Swimming
2.3. Pediveliger Settlement
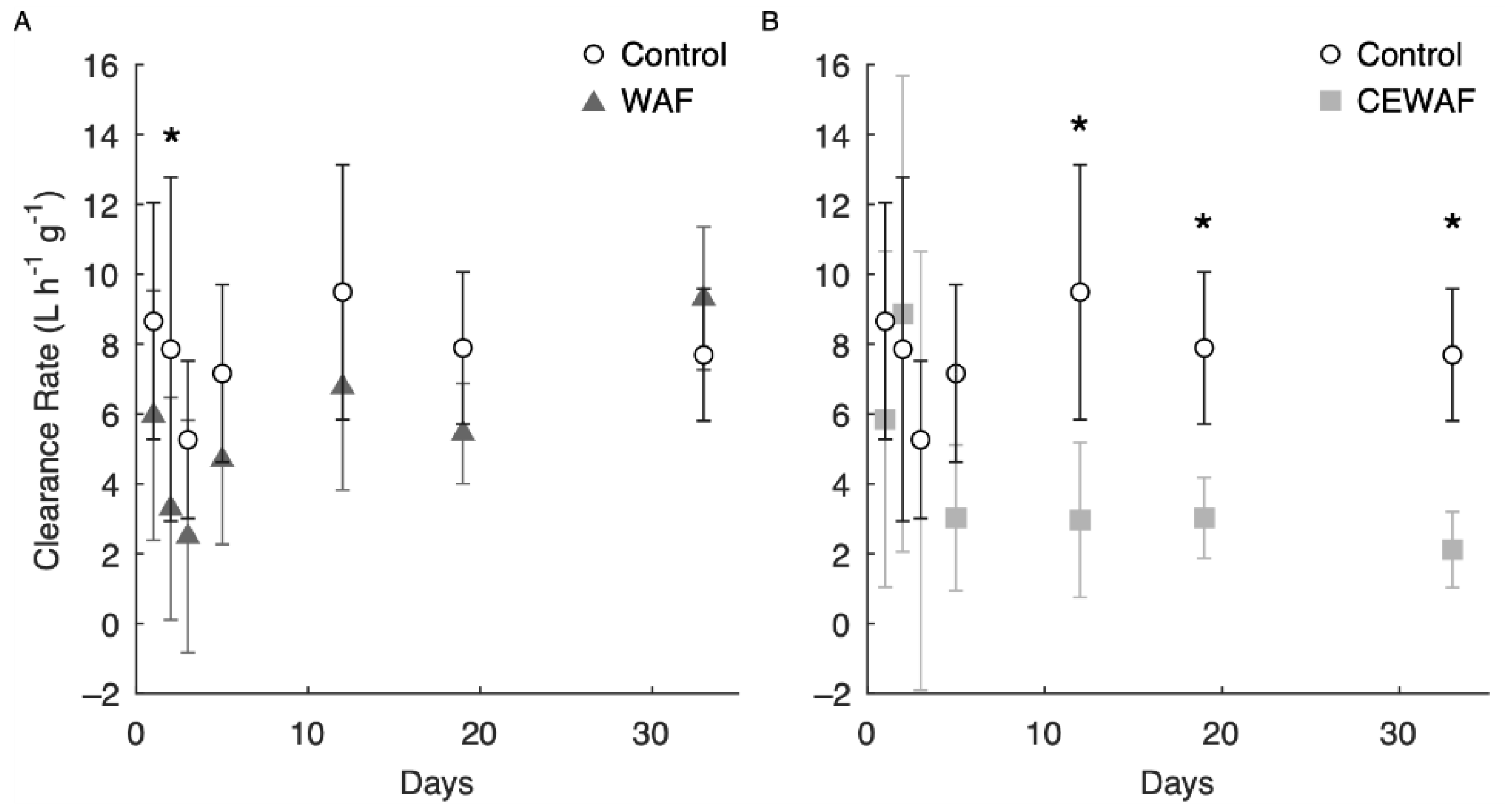
2.4. Adult Clearance Rate
2.5. Statistical Analyses
3. Results
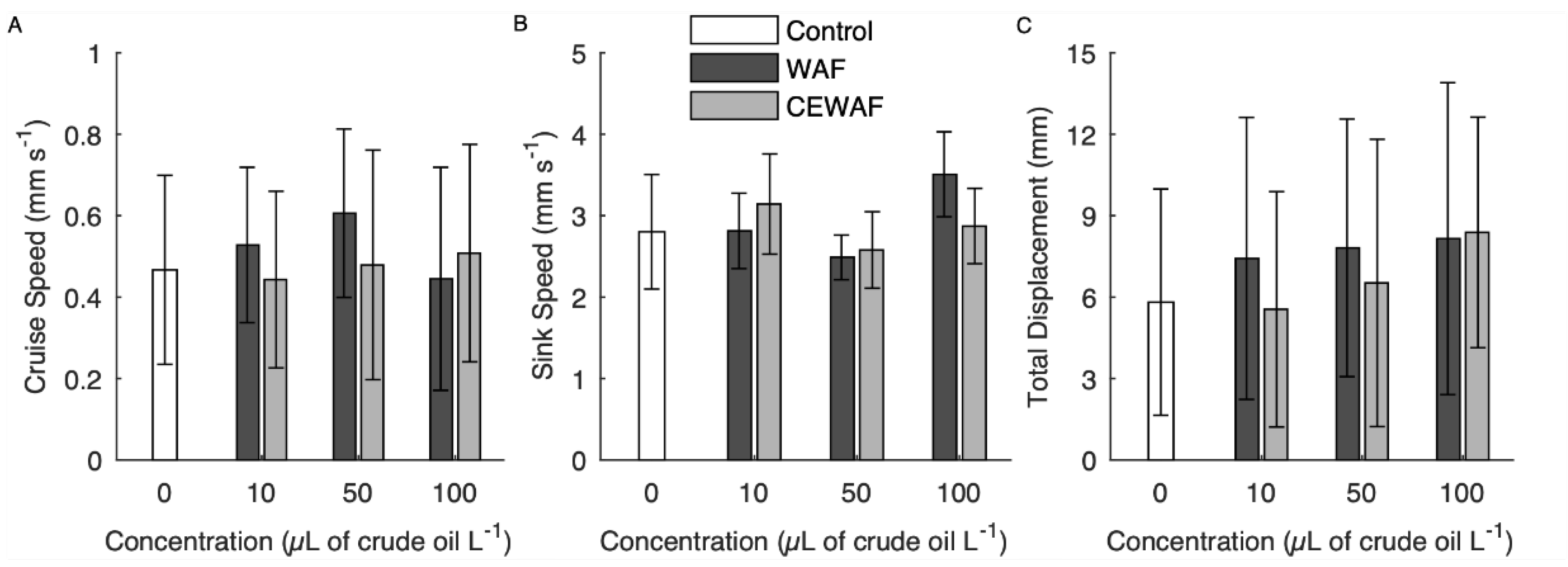
3.1. Veliger Swimming
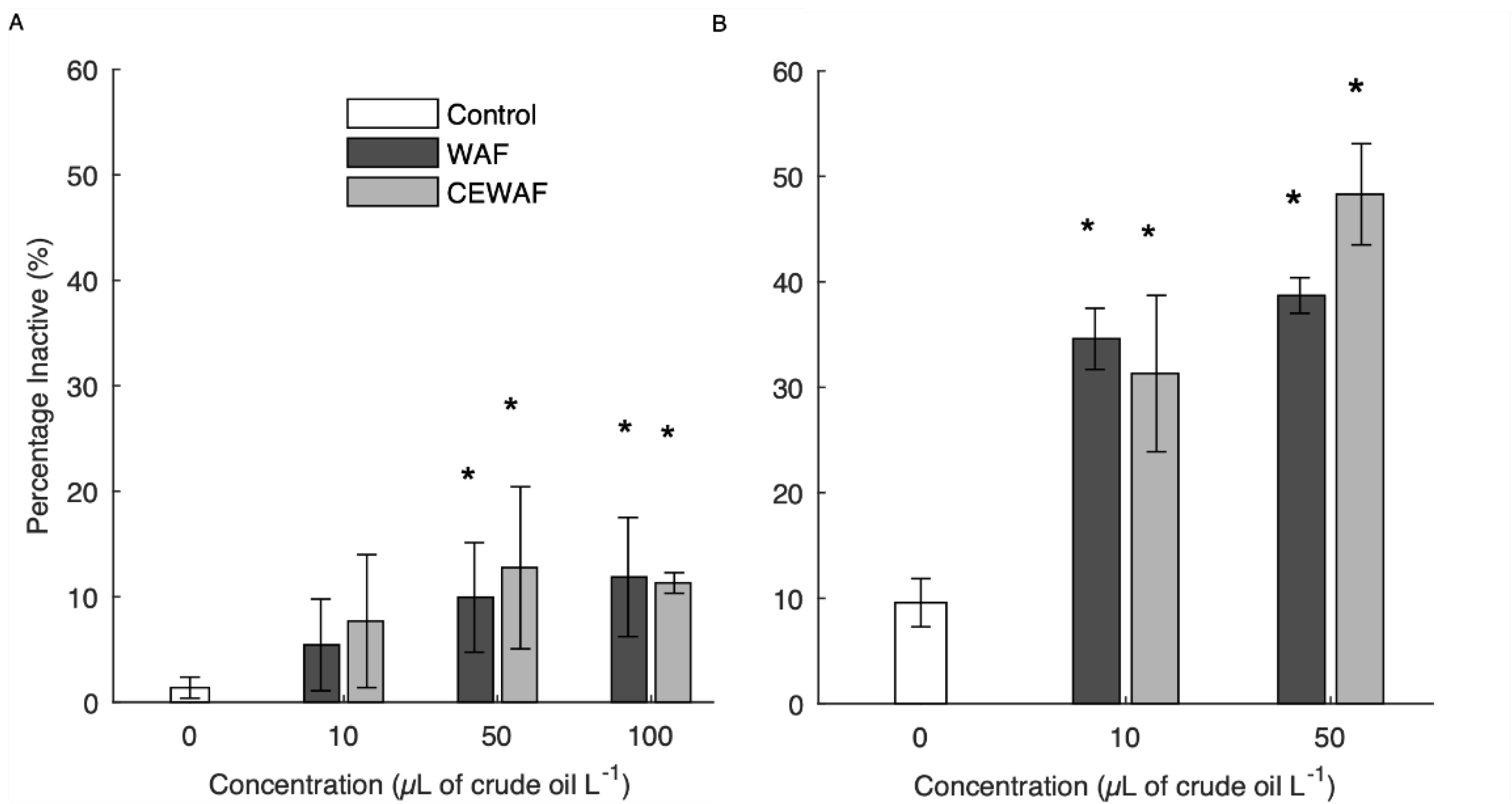
3.2. WAF/CEWAF Induced Inactivity
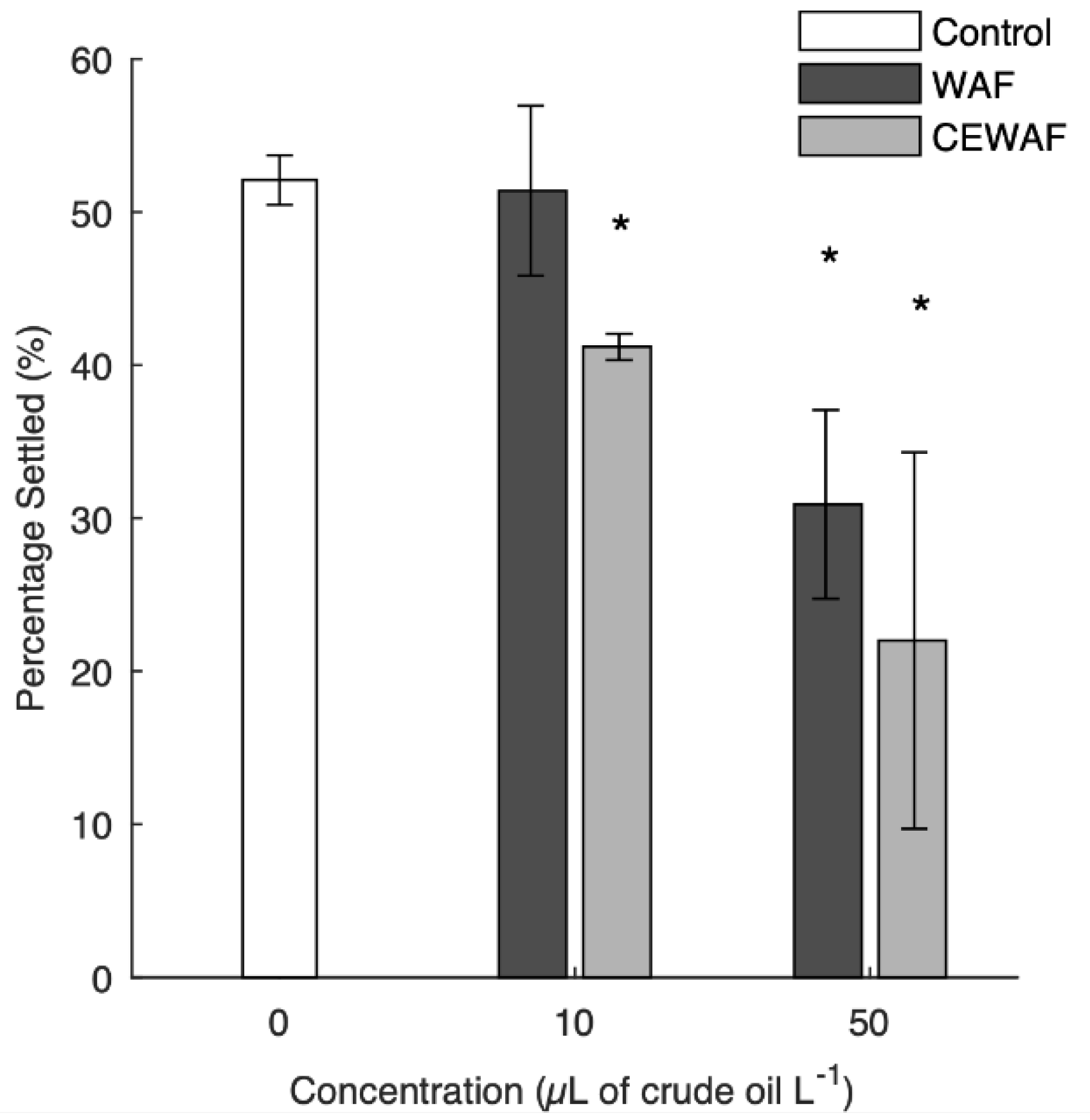
3.3. Pediveliger Settlement
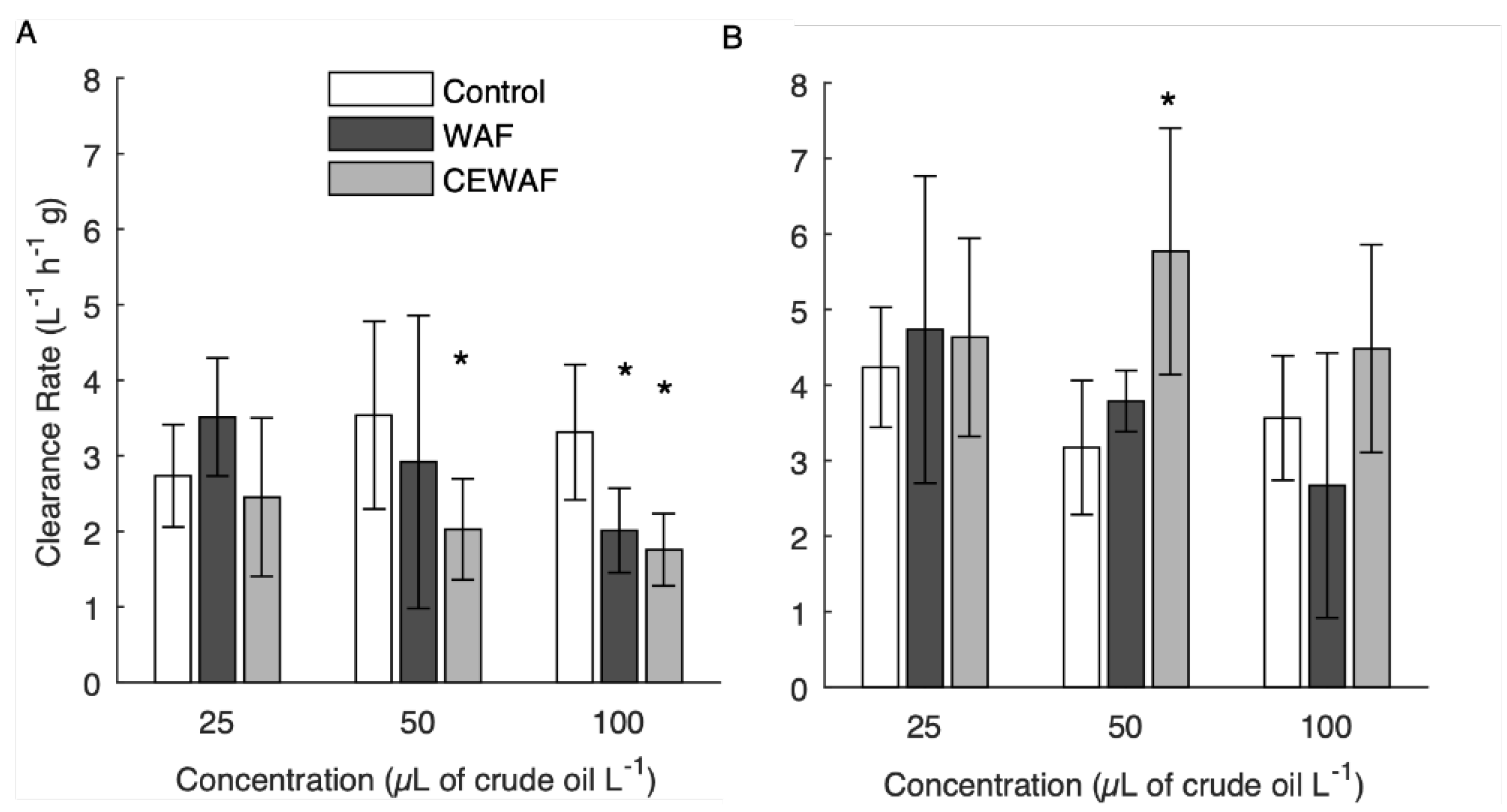
3.4. Short-Term Adult Clearance Rates
3.5. Long-Term Adult Clearance Rates
4. Discussion
4.1. Veliger and Pediveliger Inactivity
4.2. Veliger Swimming
4.3. Pediveliger Settlement
4.4. Adult Clearance Rates
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Graham, B.; Reilly, W.K.; Beinecke, F.; Boesch, D.F.; Garcia, T.D.; Murray, C.A.; Ulmer, F. Deep Water: The Gulf Oil Disaster and the Future of Offshore Drilling; Report to the President; National Commission on the BP Deepwater Horizon Oil Spill and Offshore Drilling: Washington, DC, USA, 2011; pp. 1–398. Available online: https://digital.library.unt.edu/ark:/67531/metadc123527 (accessed on 10 October 2020).
- Nixon, Z.; Zengel, S.; Michel, J. NOAA Technical Report: Categorization of Shoreline Oiling from the Deepwater Horizon Oil Spill; Research Planning Inc.: Columbia, SC, USA, 2015. Available online: https://www.fws.gov/doiddata/dwh-ar-documents/901/DWH-AR0270684.pdf (accessed on 10 October 2020).
- Zukunft, P.F. Summary Report for SubSea and SubSurface Oil and Dispersant Detection: Sampling and Monitoring; DIANE Publishing: Darby, PA, USA, 2010. Available online: https://www.restorethegulf.gov/sites/default/files/documents/pdf/osat_report_final_17dec.pdf (accessed on 10 October 2020).
- Li, Z.; Lee, K.; King, T.; Boufadel, M.C.; Venosa, A.D. Assessment of chemical dispersant effectiveness in a wave tank under regular non-breaking and breaking wave conditions. Mar. Pollut. Bull. 2008, 56, 903–912. [Google Scholar] [CrossRef] [PubMed]
- Kujawinski, E.B.; Soule, M.C.K.; Valentine, D.L.; Boysen, A.K.; Longnecker, K.; Redmond, M.C. Fate of Dispersants Associated with the Deepwater Horizon Oil Spill. Environ. Sci. Technol. 2011, 45, 1298–1306. [Google Scholar] [CrossRef] [PubMed]
- Reddy, C.M.; Arey, J.S.; Seewald, J.S.; Sylva, S.P.; Lemkau, K.L.; Nelson, R.K.; Carmichael, C.A.; McIntyre, C.P.; Fenwick, J.; Ventura, G.T.; et al. Composition and fate of gas and oil released to the water column during the Deepwater Horizon oil spill. Proc. Natl. Acad. Sci. USA 2012, 109, 20229–20234. [Google Scholar] [CrossRef] [PubMed]
- Harry, H. Oysters of the northwestern Gulf of Mexico. Tex. Conchol. 1985, 23, 14–19. [Google Scholar]
- Grabowski, J.H.; Brumbaugh, R.D.; Conrad, R.F.; Keeler, A.G.; Opaluch, J.J.; Peterson, C.H.; Piehler, M.F.; Powers, S.P.; Smyth, A.R. Economic Valuation of Ecosystem Services Provided by Oyster Reefs. Bioscience 2012, 62, 900–909. [Google Scholar] [CrossRef]
- Peterson, C.; Grabowski, J.; Powers, S. Estimated enhancement of fish production resulting from restoring oyster reef habitat: Quantitative valuation. Mar. Ecol. Prog. Ser. 2003, 264, 249–264. [Google Scholar] [CrossRef]
- Tews, J.; Brose, U.; Grimm, V.; Tielbörger, K.; Wichmann, M.C.; Schwager, M.; Jeltsch, F. Animal species diversity driven by habitat heterogeneity/diversity: The importance of keystone structures. J. Biogeogr. 2003, 31, 79–92. [Google Scholar] [CrossRef]
- Seed, R. Predator-prey relationships between the mud crab Panopeus herbstii, the blue crab, Callinectes sapidus and the Atlantic ribbed mussel Geukensia (=Modiolus) demissa. Estuar. Coast. Mar. Sci. 1980, 11, 445–458. [Google Scholar] [CrossRef]
- Coen, L.D.; Luckenbach, M.W.; Breitburg, D.L. The role of oyster reefs as essential fish habitat: A review of current knowledge and some new perspectives. Am. Fish. Soc. Symp. 1999, 22, 438–454. [Google Scholar]
- Butler, R.W.; Kirbyson, J.W. Oyster Predation by the Black Oystercatcher in British Columbia. Condor 1979, 81, 433. [Google Scholar] [CrossRef]
- Berringan, M.; Candies, T.; Cirino, J.; Dugas, R.; Dyer, C.; Gray, J.; Herrington, T.; Keithly, W.; Leard, R.; Nelson, J.; et al. The Oyster Fishery of the Gulf of Mexico, United States: A Regional Management Plan; Gulf States Marine Fisheries Commission: Ocean Springs, MS, USA, 1991. [Google Scholar]
- Vignier, J.; Donaghy, L.; Soudant, P.; Chu, F.L.E.; Morris, J.M.; Carney, M.W.; Lay, C.; Krasnec, M.; Robert, R.; Volety, A.K. Impacts of Deepwater Horizon oil and associated dispersant on early development of the Eastern oyster Crassostrea virginica. Mar. Pollut. Bull. 2015, 100, 426–437. [Google Scholar] [CrossRef]
- Vignier, J.; Soudant, P.; Chu, F.; Morris, J.; Carney, M.; Lay, C.; Krasnec, M.; Robert, R.; Volety, A.K. Lethal and sub-lethal effects of Deepwater Horizon slick oil and dispersant on oyster (Crassostrea virginica) larvae. Mar. Environ. Res. 2016, 120, 20–31. [Google Scholar] [CrossRef] [PubMed]
- Supan, J. Evaluation of a leased oyster bottom in Mississippi Sound. Gulf Caribb. Res. 1983, 7, 261–266. [Google Scholar] [CrossRef][Green Version]
- Andrews, J. Mollusca: Pelecypods and Lesser Classes. In Reproduction of Marine Invertebrates; Giese, A., Pearse, J., Eds.; Academic Press: New York, NY, USA, 1979; pp. 293–341. [Google Scholar]
- Newell, R.I.E.; Koch, E.W. Modeling seagrass density and distribution in response to changes in turbidity stemming from bivalve filtration and seagrass sediment stabilization. Estuaries 2004, 27, 793–806. [Google Scholar] [CrossRef]
- Zu Ermgassen, P.S.E.; Spalding, M.D.; Grizzle, R.E.; Brumbaugh, R.D. Quantifying the Loss of a Marine Ecosystem Service: Filtration by the Eastern Oyster in US Estuaries. Estuaries Coasts 2013, 36, 36–43. [Google Scholar] [CrossRef]
- Saiz, E.; Movilla, J.; Yebra, L.; Barata, C.; Calbet, A. Lethal and sublethal effects of naphthalene and 1,2-dimethylnaphthalene on naupliar and adult stages of the marine cyclopoid copepod Oithona davisae. Environ. Pollut. 2009, 157, 1219–1226. [Google Scholar] [CrossRef] [PubMed]
- Almeda, R.; Wambaugh, Z.; Wang, Z.; Hyatt, C.; Liu, Z.; Buskey, E.J. Interactions between Zooplankton and Crude Oil: Toxic Effects and Bioaccumulation of Polycyclic Aromatic Hydrocarbons. PLoS ONE 2013, 8, e67212. [Google Scholar] [CrossRef] [PubMed]
- Couillard, C.M.; Lee, K.; Légaré, B.; King, T.L. Effect of dispersant on the composition of the water-accommodated fraction of crude oil and its toxicity to larval marine fish. Environ. Toxicol. Chem. 2005, 24, 1496–1504. [Google Scholar] [CrossRef]
- Pie, H.V.; Mitchelmore, C.L. Acute toxicity of current and alternative oil spill chemical dispersants to early life stage blue crabs (Callinectes sapidus). Chemosphere 2015, 128, 14–20. [Google Scholar] [CrossRef] [PubMed]
- Rico-Martínez, R.; Snell, T.W.; Shearer, T.L. Synergistic toxicity of Macondo crude oil and dispersant Corexit 9500A® to the Brachionus plicatilis species complex (Rotifera). Environ. Pollut. 2013, 173, 5–10. [Google Scholar] [CrossRef]
- Kim, M.-C.; Cho, S.-M.; Jeong, W.-G. Short-term physiological response of the Pacific oyster, Crassostrea gigas, on exposure to varying levels of polycyclic aromatic hydrocarbon. Aquac. Res. 2007, 38, 1612–1618. [Google Scholar] [CrossRef]
- Sigler, M.; Leibovitz, L. Acute toxicity of oil and bilge cleaners to larval American oysters (Crassostrea virginica). Bull. Environ. Contam. Toxicol. 1982, 29, 137–145. [Google Scholar] [CrossRef] [PubMed]
- Laramore, S.; Krebs, W.; Garr, A. Effects of Macondo Canyon 252 Oil (Naturally and Chemically Dispersed) on Larval Crassostrea virginica (Gmelin, 1791). J. Shellfish. Res. 2014, 33, 709–718. [Google Scholar] [CrossRef]
- Stefansson, E.S.; Langdon, C.J.; Pargee, S.M.; Blunt, S.M.; Gage, S.J.; Stubblefield, W.A. Acute effects of non-weathered and weathered crude oil and dispersant associated with the Deepwater Horizon incident on the development of marine bivalve and echinoderm larvae. Environ. Toxicol. Chem. 2016, 35, 2016–2028. [Google Scholar] [CrossRef]
- Langdon, C.; Stefansson, E.S.; Pargee, S.M.; Blunt, S.M.; Gage, S.J.; Stubblefield, W.A. Chronic effects of non-weathered and weathered crude oil and dispersant associated with the Deepwater Horizon incident on development of larvae of the eastern oyster, Crassostrea virginica. Environ. Toxicol. Chem. 2016, 35, 2029–2040. [Google Scholar] [CrossRef]
- Finch, B.E.; Stefansson, E.S.; Langdon, C.J.; Pargee, S.M.; Blunt, S.M.; Gage, S.J.; Stubblefield, W.A. Photo-enhanced toxicity of two weathered Macondo crude oils to early life stages of the eastern oyster (Crassostrea virginica). Mar. Pollut. Bull. 2016, 113, 316–323. [Google Scholar] [CrossRef] [PubMed]
- Bacosa, H.P.; Erdner, D.L.; Liu, Z. Differentiating the roles of photooxidation and biodegradation in the weathering of Light Louisiana Sweet crude oil in surface water from the Deepwater Horizon site. Mar. Pollut. Bull. 2015, 95, 265–272. [Google Scholar] [CrossRef]
- Aurand, D.; Coelho, G. Cooperative Aquatic Toxicity Testing of Dispersed Oil and the Chemical Response to Oil Spills: Ecological Effects Research Forum (CROSERF); Technical Report 07-03; Ecosystem Management & Associates, Inc.: Lusby, MD, USA, 2005. [Google Scholar]
- Barron, M.G.; Ka’Aihue, L. Critical evaluation of CROSERF test methods for oil dispersant toxicity testing under subarctic conditions. Mar. Pollut. Bull. 2003, 46, 1191–1199. [Google Scholar] [CrossRef]
- Clark, J.R.; Bragin, G.E.; Febbo, E.J.; Letinski, D.J. Toxicity of physically and chemically dispersed oils under continuous and environmentally realistic exposure conditions: Applicability to dispersant use decisions in spill response planning. In Proceedings of the International Oil Spill Conference (IOSC 2001, No. 2)., Tampa, FL, USA, March 2001; pp. 1249–1255. [Google Scholar]
- Singer, M.; Aurand, D.; Bragin, G.; Clark, J.; Coelho, G.; Sowby, M.; Tjeerdema, R. Standardization of the Preparation and Quantitation of Water-accommodated Fractions of Petroleum for Toxicity Testing. Mar. Pollut. Bull. 2000, 40, 1007–1016. [Google Scholar] [CrossRef]
- Singer, M.M.; Aurand, D.V.; Coelho, G.M.; Bragin, G.E.; Clark, J.R.; Sowby, M.; Tjeerdema, R.S. Making, measuring, and using water-accommodated fractions of petroleum for toxicity testing. In Proceedings of the International Oil Spill Conference (IOSC 2001, No. 2), Tampa, FL, USA, March 2001; pp. 1269–1274. [Google Scholar]
- Law, R.; Dawes, V.; Woodhead, R.; Matthiessen, P. Polycyclic aromatic hydrocarbons (PAH) in seawater around England and Wales. Mar. Pollut. Bull. 1997, 34, 306–322. [Google Scholar] [CrossRef]
- Dupuy, C.; Le Gall, S.; Hartmann, H.; Bréret, M. Retention of ciliates and flagellates by the oyster Crassostrea gigas in French Atlantic coastal ponds: Protists as a trophic link between bacterioplankton and benthic suspension-feeders. Mar. Ecol. Prog. Ser. 1999, 177, 165–175. [Google Scholar] [CrossRef]
- Mo, C.; Neilson, B. Standardization of oyster soft tissue dry weight measurements. Water Res. 1994, 28, 243–246. [Google Scholar] [CrossRef]
- Riisgård, H.U. Efficiency of particle retention and filtration rate in 6 species of Efficiency of particle retention and filtration rate in 6 species of Northeast American bivalves. Mar. Ecol. Prog. Ser. 1988, 45, 217–223. [Google Scholar] [CrossRef]
- George-Ares, A.; Clark, J. Aquatic toxicity of two Corexit® dispersants. Chemosphere 2000, 40, 897–906. [Google Scholar] [CrossRef]
- Goodbody-Gringley, G.; Wetzel, D.L.; Gillon, D.; Pulster, E.; Miller, A.; Ritchie, K.B. Toxicity of Deepwater Horizon Source Oil and the Chemical Dispersant, Corexit® 9500, to Coral Larvae. PLoS ONE 2013, 8, e45574. [Google Scholar] [CrossRef]
- Salehi, M.; Rodriguez, R.; Boettcher, A.; Powers, S.P.; Geitner, N.; Ladner, D.A.; Rikard, S.; Whelton, A.J. Impact of dispersant on early life stages of the water flea Daphnia magna and the eastern oyster Crassostrea virginica. J. Appl. Toxicol. 2017, 37, 1464–1470. [Google Scholar] [CrossRef]
- Jeong, W.-G.; Cho, S.-M. The effects of polycyclic aromatic hydrocarbon exposure on the fertilization and larval development of the Pacific oyster, Crassostrea gigas. J. Shellfish Res. 2005, 24, 209–213. [Google Scholar]
- Beiras, R.; His, E. Effects of dissolved mercury on embryo-genesis, survival, growth and metamorphosis of Crassostrea gigas oyster larvae. Mar. Ecol. Prog. Ser. 1994, 113, 95–103. [Google Scholar] [CrossRef]
- Almeda, R.; Connelly, T.L.; Buskey, E.J. How much crude oil can zooplankton ingest? Estimating the quantity of dispersed crude oil defecated by planktonic copepods. Environ. Pollut. 2016, 208, 645–654. [Google Scholar] [CrossRef]
- Jiang, Z.; Huang, Y.; Chen, Q.; Zeng, J.; Xu, X. Acute toxicity of crude oil water accommodated fraction on marine copepods: The relative importance of acclimatization temperature and body size. Mar. Environ. Res. 2012, 81, 12–17. [Google Scholar] [CrossRef]
- Almeda, R.; Connelly, T.L.; Buskey, E.J. Novel insight into the role of heterotrophic dinoflagellates in the fate of crude oil in the sea. Sci. Rep. 2014, 4, 7560. [Google Scholar] [CrossRef] [PubMed]
- Donahue, W.; Wang, R.T.; Welch, M.; Nicol, J. Effects of water-soluble components of petroleum oils and aromatic hydrocarbons on barnacle larvae. Environ. Pollut. 1977, 13, 187–202. [Google Scholar] [CrossRef]
- Cohen, J.H.; McCormick, L.R.; Burkhardt, S.M. Effects of Dispersant and Oil on Survival and Swimming Activity in a Marine Copepod. Bull. Environ. Contam. Toxicol. 2014, 92, 381–387. [Google Scholar] [CrossRef] [PubMed]
- Hansen, B.H.; Altin, D.; Olsen, A.J.; Nordtug, T. Acute toxicity of naturally and chemically dispersed oil on the filter-feeding copepod Calanus finmarchicus. Ecotoxicol. Environ. Saf. 2012, 86, 38–46. [Google Scholar] [CrossRef] [PubMed]
- Hansen, B.H.; Altin, D.; Nordtug, T.; Øverjordet, I.B.; Olsen, A.J.; Krause, D.; Størdal, I.; Størseth, T.R. Exposure to crude oil micro-droplets causes reduced food uptake in copepods associated with alteration in their metabolic profiles. Aquat. Toxicol. 2017, 184, 94–102. [Google Scholar] [CrossRef] [PubMed]
- Titelman, J.; Kiørboe, T. Motility of copepod nauplii and implications for food encounter. Mar. Ecol. Prog. Ser. 2003, 247, 123–135. [Google Scholar] [CrossRef]
- Dekshenieks, M.M.; Hofmann, E.E.; Klinck, J.M.; Powell, E.N. Modeling the vertical distribution of oyster larvae in response to environmental conditions. Mar. Ecol. Prog. Ser. 1996, 136, 97–110. [Google Scholar] [CrossRef]
- Wisely, B.; Blick, R. Mortality of marine invertebrate larvae in mercury, copper, and zinc solutions. Mar. Freshw. Res. 1967, 18, 63–72. [Google Scholar] [CrossRef]
- Finelli, C.M.; Wethey, D.S. Behavior of oyster (Crassostrea virginica) larvae in flume boundary layer flows. Mar. Biol. 2003, 143, 703–711. [Google Scholar] [CrossRef]
- Baker, S.; Mann, R. Description of metamorphic phases in the oyster Crassostrea virginica and effects of hypoxia on metamorphosis. Mar. Ecol. Prog. Ser. 1994, 104, 91–99. [Google Scholar] [CrossRef]
- Smith, C.M.; Hackney, C.T. The Effects of Hydrocarbons on the Setting of the American Oyster, Crassostrea virginica, in Intertidal Habitats in Southeastern North Carolina. Estuaries 1989, 12, 42–48. [Google Scholar] [CrossRef]
- Axiak, V.; George, J.J. Effects of exposure to petroleum hydrocarbons on the gill functions and ciliary activities of a marine bivalve. Mar. Biol. 1987, 94, 241–249. [Google Scholar] [CrossRef]
- Donkin, P.; Widdows, J.; Evans, S.V.; Worrall, C.M.; Carr, M. Quantitative structure-activity relationships for the effect of hydrophobic organic chemicals on rate of feeding by mussels (Mytilus edulis). Aquat. Toxicol. 1989, 14, 277–293. [Google Scholar] [CrossRef]
- Toro, B.; Navarro, J.; Palma-Fleming, H. Use of clearance rate in Choromytilus chorus (Bivalvia: Mytilidae) as a non-destructive biomarker of aquatic pollution. Rev. Chil. Hist. Nat. 2003, 76, 267–274. [Google Scholar] [CrossRef]
- Redmond, K.J.; Berry, M.; Pampanin, D.M.; Andersen, O.K. Valve gape behaviour of mussels (Mytilus edulis) exposed to dispersed crude oil as an environmental monitoring endpoint. Mar. Pollut. Bull. 2017, 117, 330–339. [Google Scholar] [CrossRef]
- Yamada, M.; Takada, H.; Toyoda, K.; Yoshida, A.; Shibata, A.; Nomura, H.; Wada, M.; Nishimura, M.; Okamoto, K.; Ohwada, K. Study on the fate of petroleum-derived polycyclic aromatic hydrocarbons (PAHs) and the effect of chemical dispersant using an enclosed ecosystem, mesocosm. Mar. Pollut. Bull. 2003, 47, 105–113. [Google Scholar] [CrossRef]
- Wolfe, M.; Schwartz, G.; Singaram, S.; E Mielbrecht, E.; Tjeerdema, R.S.; Sowby, M.L. Influence of dispersants on the bioavailability and trophic transfer of petroleum hydrocarbons to larval topsmelt (Atherinops affinis). Aquat. Toxicol. 2001, 52, 49–60. [Google Scholar] [CrossRef]
- Wu, D.; Wang, Z.; Hollebone, B.; McIntosh, S.; King, T.; Hodson, P.V. Comparative toxicity of four chemically dispersed and undispersed crude oils to rainbow trout embryos. Environ. Toxicol. Chem. 2012, 31, 754–765. [Google Scholar] [CrossRef]
- Soniat, T.M.; King, S.M.; Tarr, M.A.; Thorne, M.A. Chemical and Physiological Measures on Oysters (Crassostrea virginica) from Oil-Exposed Sites in Louisiana. J. Shellfish. Res. 2011, 30, 713–717. [Google Scholar] [CrossRef]
| [Crude Oil] | [PAH] µg L−1 | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| µL L−1 | mg L−1 | Nap | Ace | Acy | Flu | Phe | An | Flua | Pyr | BaA | Chr | BbF |
| 10 | 8.5 | 8.45 | 0.85 | 0.14 | 2.82 | 6.08 | 0.08 | 0.15 | 0.31 | 0.14 | 1.94 | 0.2 |
| 25 | 21.1 | 21.12 | 2.14 | 0.35 | 7.06 | 15.21 | 0.2 | 0.38 | 0.77 | 0.35 | 4.85 | 0.49 |
| 50 | 42.3 | 42.23 | 4.27 | 0.7 | 14.11 | 30.42 | 0.4 | 0.76 | 1.54 | 0.7 | 9.7 | 0.98 |
| 100 | 84.5 | 84.46 | 8.54 | 1.4 | 28.23 | 60.83 | 0.8 | 1.53 | 3.08 | 1.4 | 19.39 | 1.96 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Garcia, S.M.; Du Clos, K.T.; Hawkins, O.H.; Gemmell, B.J. Sublethal Effects of Crude Oil and Chemical Dispersants on Multiple Life History Stages of the Eastern Oyster, Crassostrea virginica. J. Mar. Sci. Eng. 2020, 8, 808. https://doi.org/10.3390/jmse8100808
Garcia SM, Du Clos KT, Hawkins OH, Gemmell BJ. Sublethal Effects of Crude Oil and Chemical Dispersants on Multiple Life History Stages of the Eastern Oyster, Crassostrea virginica. Journal of Marine Science and Engineering. 2020; 8(10):808. https://doi.org/10.3390/jmse8100808
Chicago/Turabian StyleGarcia, Sara M., Kevin T. Du Clos, Olivia H. Hawkins, and Brad J. Gemmell. 2020. "Sublethal Effects of Crude Oil and Chemical Dispersants on Multiple Life History Stages of the Eastern Oyster, Crassostrea virginica" Journal of Marine Science and Engineering 8, no. 10: 808. https://doi.org/10.3390/jmse8100808
APA StyleGarcia, S. M., Du Clos, K. T., Hawkins, O. H., & Gemmell, B. J. (2020). Sublethal Effects of Crude Oil and Chemical Dispersants on Multiple Life History Stages of the Eastern Oyster, Crassostrea virginica. Journal of Marine Science and Engineering, 8(10), 808. https://doi.org/10.3390/jmse8100808






