The Active Reef Restoration Toolbox is a Vehicle for Coral Resilience and Adaptation in a Changing World
Abstract
:1. Introduction
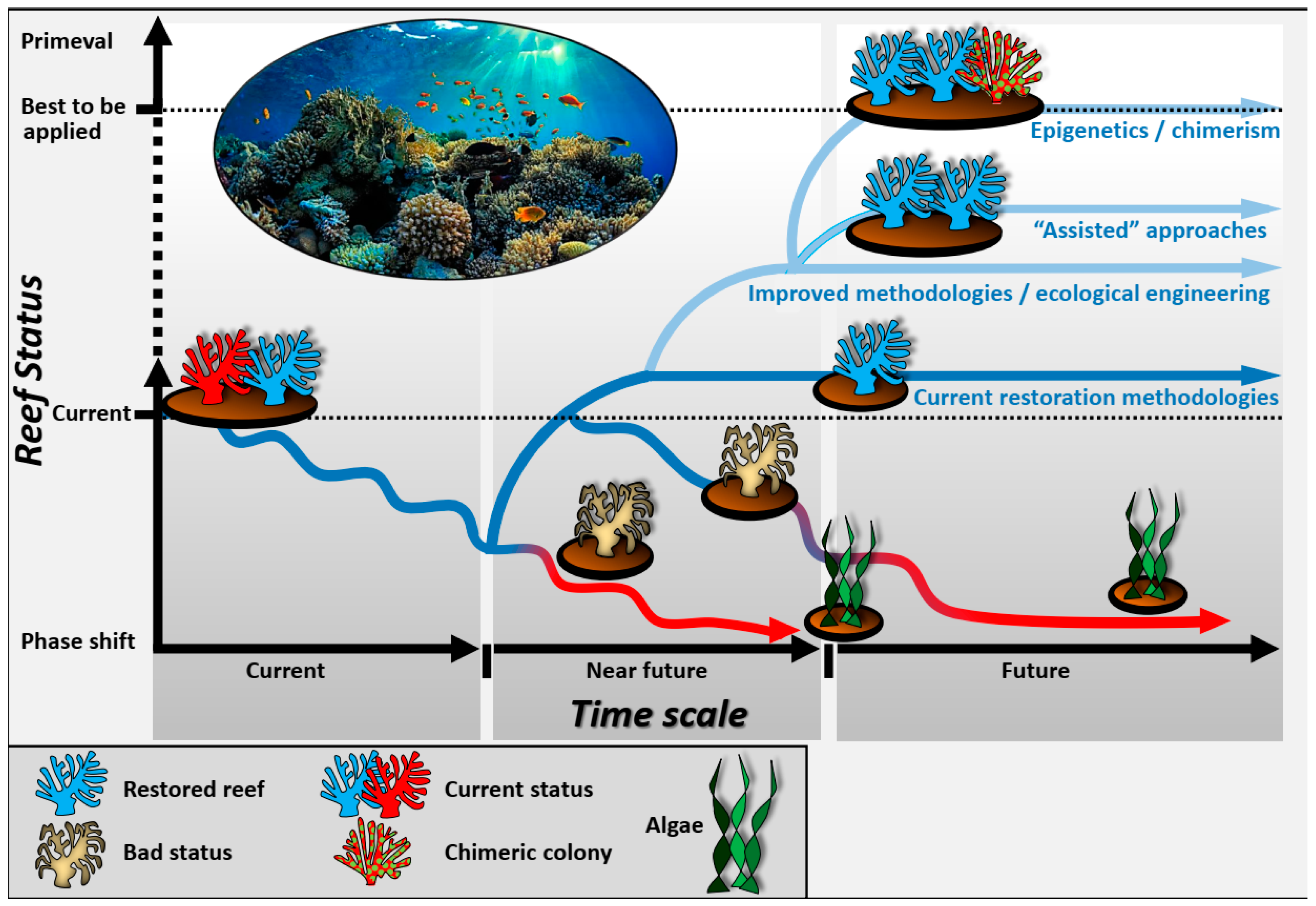
2. Defining the Toolbox
2.1. Improved Gardening Methodologies
2.2. Ecological Engineering
2.3. Assisted Migration/Colonization
2.4. Assisted Genetics/Evolution
2.5. Assisted Microbiome
2.6. Epigenetics
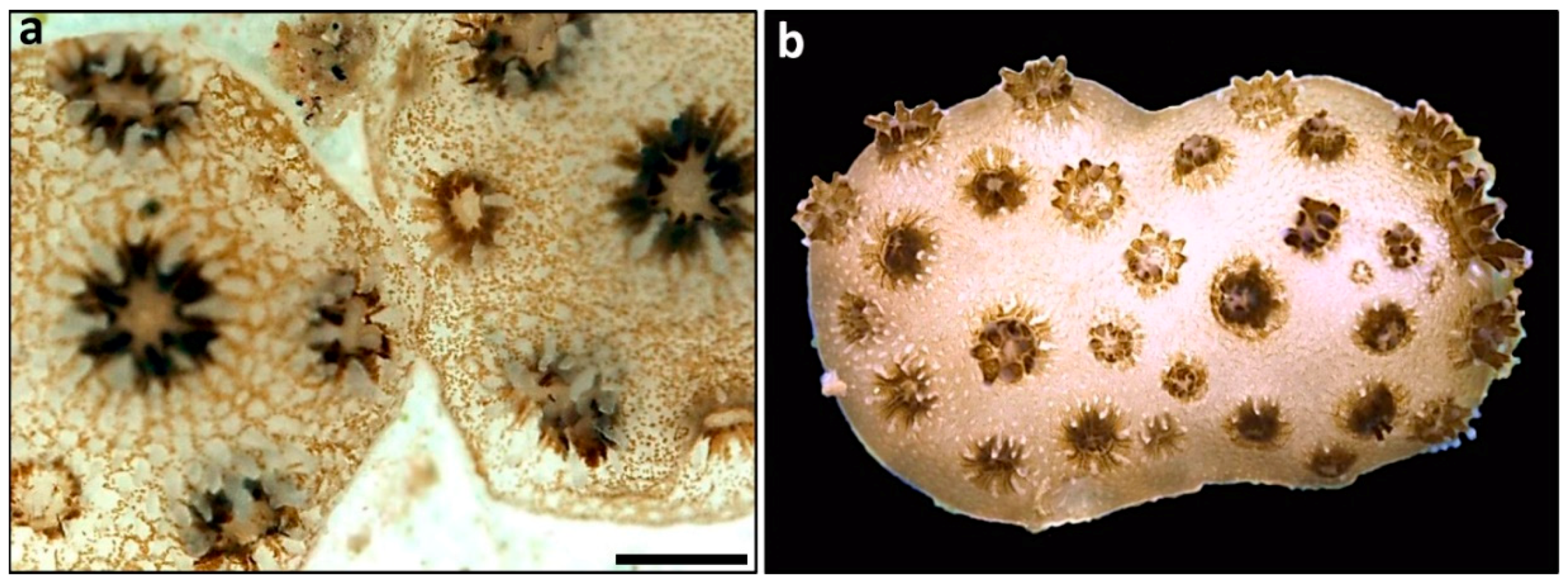
2.7. Coral Chimerism
3. Discussion
Funding
Acknowledgments
Conflicts of Interest
References
- Rinkevich, B. Climate change and active reef restoration—Ways of constructing the “reefs of tomorrow”. J. Mar. Sci. Eng. 2015, 3, 111–127. [Google Scholar] [CrossRef]
- Sully, S.; Burkepile, D.E.; Donovan, M.K.; Hodgson, G.; Van Woesik, R. A global analysis of coral bleaching over the past two decades. Nat. Commun. 2019, 10, 1264. [Google Scholar] [CrossRef] [PubMed]
- Jones, K.R.; Klein, C.J.; Halpern, B.S.; Venter, O.; Grantham, H.; Kuempel, C.D.; Shumway, N.; Friedlander, A.M.; Possingham, H.P.; Watson, J.E. The location and protection status of Earth’s diminishing marine wilderness. Curr. Biol. 2018, 28, 2506–2512. [Google Scholar] [CrossRef] [PubMed]
- Bruno, J.F.; Selig, E.R. Regional decline of coral cover in the Indo-Pacific: Timing, extent, and subregional comparisons. PLoS ONE 2007, 2, e711. [Google Scholar] [CrossRef] [PubMed]
- De’ath, G.; Fabricius, K.E.; Sweatman, H.; Puotinen, M. The 27-year decline of coral cover on the great barrier reef and its causes. Proc. Natl. Acad. Sci. USA 2012, 109, 17995–17999. [Google Scholar] [CrossRef] [PubMed]
- Trenberth, K.E.; Cheng, L.; Jacobs, P.; Zhang, Y.; Fasullo, J. Hurricane Harvey links to ocean heat content and climate change adaptation. Earths Future 2018, 6, 730–744. [Google Scholar] [CrossRef]
- Hughes, T.P.; Kerry, J.T.; Baird, A.H.; Connolly, S.R.; Chase, T.J.; Dietzel, A.; Hill, T.; Hoey, A.S.; Hoogenboom, M.O.; Jacobson, M.; et al. Global warming impairs stock—Recruitment dynamics of corals. Nature 2019, 568, 387–390. [Google Scholar] [CrossRef] [PubMed]
- IPCC. Climate Change 2014: Synthesis Report. Contribution of Working Groups I, II and III to the Fifth Assessment Report of the IPCC. In Proccedings of the Intergovernmental Panel on Climate Change; IPCC: Geneva, Switzerland, 2014. [Google Scholar]
- Fong, C.R.; Bittick, S.J.; Fong, P. Simultaneous synergist, antagonistic and additive interactions between multiple local stressors all degrade algal turf communities on coral reefs. J. Ecol. 2018, 106, 1390–1400. [Google Scholar] [CrossRef]
- Fine, M.; Cinar, M.; Voolstra, C.R.; Safa, A.; Rinkevich, B.; Laffoley, D.; Hilmi, N.; Allemand, D. Coral reefs of the Red sea-challenges and potential solutions. Reg. Stud. Mar. Sci. 2019, 25, 100498. [Google Scholar] [CrossRef]
- Rinkevich, B. Management of coral reefs: We have gone wrong when neglecting active reef restoration. Mar. Pollut. Bull. 2008, 56, 1821–1824. [Google Scholar] [CrossRef]
- Miller, K.I.; Russ, G.R. Studies of no-take marine reserves: Methods for differentiating reserve and habitat effects. Ocean Coast. Manag. 2014, 96, 51–60. [Google Scholar] [CrossRef]
- Rinkevich, B. Restoration strategies for coral reefs damaged by recreational activities: The use of sexual and asexual recruits. Restor. Ecol. 1995, 3, 241–251. [Google Scholar] [CrossRef]
- Rinkevich, B. Steps towards the evaluation of coral reef restoration by using small branch fragments. Mar. Biol. 2000, 136, 807–812. [Google Scholar] [CrossRef]
- Rinkevich, B. Conservation of coral reefs through active restoration measures: Recent approaches and last decade progress. Environ. Sci. Technol. 2005, 39, 4333–4342. [Google Scholar] [CrossRef] [PubMed]
- Rinkevich, B. The coral gardening concept and the use of underwater nurseries; lesson learned from silvics and silviculture. In Coral Reef Restoration Handbook; Precht, W.F., Ed.; CRC Press: Boca Raton, FL, USA, 2006; pp. 291–301. [Google Scholar]
- Rinkevich, B. Rebuilding coral reefs: Does active reef restoration lead to sustainable reefs? Curr. Opin. Environ. Sustain. 2014, 7, 28–36. [Google Scholar] [CrossRef]
- Epstein, N.; Bak, R.P.M.; Rinkevich, B. Strategies for gardening denuded coral reef areas: The applicability of using different types of coral material for reef restoration. Restor. Ecol. 2001, 9, 432–442. [Google Scholar] [CrossRef]
- Epstein, N.; Bak, R.P.M.; Rinkevich, B. Applying forest restoration principles to coral reef rehabilitation. Aquat. Conserv. 2003, 13, 387–395. [Google Scholar] [CrossRef]
- Rinkevich, B. The quandary for active and passive reef restoration in a changing world. In Active Coral Reef Restoration: Techniques for a Changing Planet; Vaughan, D., Ed.; J. Ross Publishing: Fort Lauderdale, FL, USA, in press.
- Lirman, D.; Schopmeyer, S. Ecological solutions to reef degradation: Optimizing coral reef restoration in the Caribbean and Western Atlantic. Peer J. 2016, 4, e2597. [Google Scholar] [CrossRef]
- Bradshaw, A.D. Underlying principles of restoration. Can. J. Fish. Aquat. Sci. 1996, 53 (Suppl. 1), 3–9. [Google Scholar] [CrossRef]
- Holl, K.D.; Aide, T.M. When and where to actively restore ecosystems? For. Ecol. Manag. 2011, 261, 1558–1563. [Google Scholar] [CrossRef]
- Clewell, A.F.; Aronson, J. Motivations for the restoration of ecosystems. Conserv. Biol. 2006, 20, 420–428. [Google Scholar] [CrossRef] [PubMed]
- Shafir, S.; Van Rijn, J.; Rinkevich, B. Steps in the construction of underwater coral nursery, an essential component in reef restoration acts. J. Mar. Biol. 2006, 149, 679–687. [Google Scholar] [CrossRef]
- Shafir, S.; van Rijn, J.; Rinkevich, B. A mid-water coral nursery. In Proceedings of the 10th International Coral Reef Symposium, Okinawa, Japan, 28 June–2 July 2004; pp. 1674–1679. [Google Scholar]
- Shafir, S.; Rinkevich, B. The underwater silviculture approach for reef restoration: An emergent aquaculture theme. In Aquaculture Research Trends; Schwartz, S.H., Ed.; Nova Science Publications: New York, NY, USA, 2008; pp. 279–295. [Google Scholar]
- Shafir, S.; Rinkevich, B. Integrated long term mid-water coral nurseries: A management instrument evolving into a floating ecosystem. Univ. Maurit. Res. J. 2010, 16, 365–379. [Google Scholar]
- Shafir, S.; Abady, S.; Rinkevich, B. Improved sustainable maintenance for mid-water coral nursery by the application of an anti-fouling agent. J. Exp. Mar. Biol. Ecol. 2009, 368, 124–128. [Google Scholar] [CrossRef]
- Baria, M.V.B.; Guest, J.R.; Edwards, A.J.; Aliño, P.M.; Heyward, A.J.; Gomez, E.D. Caging enhances post-settlement survival of juveniles of the scleractinian coral Acropora tenuis. J. Exp. Mar. Biol. Ecol. 2010, 394, 149–153. [Google Scholar] [CrossRef]
- Liñán-Cabello, M.A.; Flores-Ramírez, L.A.; Laurel-Sandoval, M.A.; Mendoza, E.G.; Santiago, O.S.; Delgadillo-Nuño, M.A. Acclimation in Pocillopora spp. during a coral restoration program in Carrizales Bay, Colima, Mexico. Mar. Freshw. Behav. Physiol. 2011, 44, 61–72. [Google Scholar] [CrossRef]
- Linden, B.; Rinkevich, B. Creating stocks of young colonies from brooding-coral larvae, amenable to active reef restoration. J. Exp. Mar. Biol. Ecol. 2011, 398, 40–46. [Google Scholar] [CrossRef]
- Linden, B.; Vermeij, M.J.A.; Rinkevich, B. The coral settlement box: A simple device to produce coral stock from brooded coral larvae entirely in situ. Ecol. Eng. 2019, 132, 115–119. [Google Scholar] [CrossRef]
- Nakamura, R.; Ando, W.; Yamamoto, H.; Kitano, M.; Sato, A.; Nakamura, M.; Kayanne, H.; Omori, M. Corals mass-cultured from eggs and transplanted as juveniles to their native, remote coral reef. Mar. Ecol. Prog. Ser. 2011, 436, 161–168. [Google Scholar] [CrossRef]
- Nedimyer, K.; Gaines, K.; Roach, S. Coral Tree Nursery©: An innovative approach to growing corals in an ocean-based field nursery. Aquac. Aquar. Conserv. Legis. 2011, 4, 442–446. [Google Scholar]
- Cooper, W.T.; Lirman, D.; VanGroningen, M.P.; Parkinson, J.E.; Herlan, J.; McManus, J.W. Assessing techniques to enhance early post-settlement survival of corals in situ for reef restoration. Bull. Mar. Sci. 2014, 90, 651–664. [Google Scholar] [CrossRef]
- Tebben, J.; Guest, J.R.; Sin, T.M.; Steinberg, P.D.; Harder, T. Corals like it waxed: Paraffin-based antifouling technology enhances coral spat survival. PLoS ONE 2014, 9, e87545. [Google Scholar] [CrossRef] [PubMed]
- Toh, T.C.; Ng, C.S.L.; Peh, J.W.K.; Toh, K.B.; Chou, L.M. Augmenting the post-transplantation growth and survivorship of juvenile scleractinian corals via nutritional enhancement. PLoS ONE 2014, 9, e98529. [Google Scholar] [CrossRef] [PubMed]
- Horoszowski-Fridman, Y.B.; Brêthes, J.-C.; Rahmani, N.; Rinkevich, B. Marine silviculture: Incorporating ecosystem engineering properties into reef restoration acts. Ecol. Eng. 2015, 82, 201–213. [Google Scholar] [CrossRef]
- Chamberland, V.F.; Petersen, D.; Guest, J.R.; Petersen, U.; Brittsan, M.; Vermeij, M.J. New seeding approach reduces costs and time to outplant sexually propagated corals for reef restoration. Sci. Rep. 2017, 7, 18076. [Google Scholar] [CrossRef] [PubMed]
- Pollock, F.J.; Katz, S.M.; van de Water, J.A.; Davies, S.W.; Hein, M.; Torda, G.; Matz, M.V.; Beltran, V.H.; Buerger, P.; Puill-Stephan, E.; et al. Coral larvae for restoration and research: A large-scale method for rearing Acropora millepora larvae, inducing settlement, and establishing symbiosis. Peer J. 2017, 5, e3732. [Google Scholar] [CrossRef] [PubMed]
- Tagliafico, A.; Rangel, S.; Christidis, L.; Kelaher, B.P. A potential method for improving coral self-attachment. Restor. Ecol. 2018, 26, 1082–1090. [Google Scholar] [CrossRef]
- Tagliafico, A.; Rangel, S.; Kelaher, B.; Scheffers, S.; Christidis, L. A new technique to increase polyp production in stony coral aquaculture using waste fragments without polyps. Aquaculture 2018, 484, 303–308. [Google Scholar] [CrossRef]
- Zayasu, Y.; Suzuki, G. Comparisons of population density and genetic diversity in artificial and wild populations of an arborescent coral, Acropora yongei: Implications for the efficacy of “artificial spawning hotspots”. Restor. Ecol. 2019, 27, 440–446. [Google Scholar] [CrossRef]
- Levi, G.; Shaish, L.; Haim, A.; Rinkevich, B. Mid-water rope nursery—Testing design and performance of a novel reef restoration instrument. Ecol. Eng. 2010, 36, 560–569. [Google Scholar] [CrossRef]
- Horoszowski-Fridman, Y.B.; Izhaki, I.; Rinkevich, B. Engineering of coral reef larval supply through transplantation of nursery-farmed gravid colonies. J. Exp. Mar. Biol. Ecol. 2011, 399, 162–166. [Google Scholar] [CrossRef]
- Horoszowski-Fridman, Y.B.; Rinkevich, B. Restoring the animal forests: Harnessing silviculture biodiversity concepts for coral transplantation. In Marine Animal Forests: The Ecology of Benthic Biodiversity Hotspots; Rossi, S., Bramanti, L., Gori, A., Orejas, C., Eds.; Springer International Publishing: Cham, Switzerland, 2017; pp. 1313–1335. [Google Scholar]
- Sabater, M.G.; Yap, H.T. Growth and survival of coral transplants with and without electrochemical deposition of CaCO3. J. Exp. Mar. Biol. Ecol. 2002, 272, 131–146. [Google Scholar] [CrossRef]
- Borell, E.M.; Romatzki, S.B.C.; Ferse, S.C.A. Differential physiological responses of two congeneric scleractinian corals to mineral accretion and an electric field. Coral Reef. 2010, 29, 191–200. [Google Scholar] [CrossRef]
- Strömberg, S.M.; Lundälv, T.; Goreau, T.J. Suitability of mineral accretion as a rehabilitation method for cold-water coral reefs. J. Exp. Mar. Biol. Ecol. 2010, 395, 153–161. [Google Scholar] [CrossRef]
- Ng, C.S.L.; Toh, T.C.; Toh, K.B.; Guest, J.; Chou, L.M. Dietary habits of grazers influence their suitability as biological controls of fouling macroalgae in ex situ mariculture. Aquac. Res. 2014, 45, 1852–1860. [Google Scholar]
- Villanueva, R.D.; Baria, M.V.B.; dela Cruz, D.W. Effects of grazing by herbivorous gastropod (Trochus niloticus) on the survivorship of cultured coral Spat. Zool. Stud. 2013, 52, 44. [Google Scholar] [CrossRef]
- Forsman, Z.H.; Page, C.A.; Toonen, R.J.; Vaughan, D. Growing coral larger and faster: Micro-colony-fusion as a strategy for accelerating coral cover. Peer J. 2015, 3, e1313. [Google Scholar] [CrossRef] [PubMed]
- Frias-Torres, S.; Van de Geer, C. Testing animal-assisted cleaning prior to transplantation in coral reef restoration. Peer J. 2015, 3, e1287. [Google Scholar] [CrossRef] [PubMed]
- Chou, L.M. Rehabilitation engineering of Singapore reefs to cope with urbanization and climate change impacts. JCEA 2016, 10, 932–936. [Google Scholar]
- Montoya-Maya, P.H.; Smit, K.P.; Burt, A.J.; Frias-Torres, S. Large-scale coral reef restoration could assist natural recovery in Seychelles, Indian Ocean. J. Nat. Conserv. 2016, 16, 1–17. [Google Scholar] [CrossRef]
- Anthony, K.; Bay, L.K.; Costanza, R.; Firn, J.; Gunn, J.; Harrison, P.; Heyward, A.; Lundgren, P.; Mead, D.; Moore, T.; et al. New interventions are needed to save coral reefs. Nat. Ecol. Evol. 2017, 1, 1420–1422. [Google Scholar] [CrossRef] [PubMed]
- Linden, B.; Rinkevich, B. Elaborating of an eco-engineering approach for stock enhanced sexually derived coral colonies. J. Exp. Mar. Biol. Ecol. 2017, 486, 314–321. [Google Scholar] [CrossRef]
- Page, C.A.; Muller, E.M.; Vaughan, D.E. Microfragmenting for the successful restoration of slow growing massive corals. Ecol. Eng. 2018, 123, 86–94. [Google Scholar] [CrossRef]
- Rachmilovitz, E.N.; Rinkevich, B. Tiling the reef—Exploring the first step of an ecological engineering tool that may promote phase-shift reversals in coral reefs. Ecol. Eng. 2017, 105, 150–161. [Google Scholar] [CrossRef]
- Knoester, E.G.; Murk, A.J.; Osinga, R. Benefits of herbivorous fish outweigh costs of corallivory in coral nurseries placed close to a Kenyan patch reef. Mar. Ecol. Prog. Ser. 2019, 611, 143–155. [Google Scholar] [CrossRef]
- Hoegh-Guldberg, O.; Hughes, L.; McIntyre, S.; Lindenmayer, D.B.; Parmesan, C.; Possingham, H.P.; Thomas, C.D. Assisted colonization and rapid climate change. Science 2008, 321, 345–346. [Google Scholar] [CrossRef] [PubMed]
- Coles, S.L.; Riegl, B.M. Thermal tolerances of reef corals in the Gulf: A review of the potential for increasing coral survival and adaptation to climate change through assisted translocation. Mar. Pollut. Bull. 2013, 72, 323–332. [Google Scholar] [CrossRef] [PubMed]
- Riegl, B.M.; Purkis, S.J.; Al-Cibahy, A.S.; Abdel-Moati, M.A.; Hoegh-Guldberg, O. Present limits to heat-adaptability in corals and population-level responses to climate extremes. PLoS ONE 2011, 6, e24802. [Google Scholar] [CrossRef]
- Baker, A.C. Ecosystems—Reef corals bleach to survive change. Nature 2001, 411, 765–766. [Google Scholar] [CrossRef]
- Schopmeyer, S.A.; Lirman, D.; Bartels, E.; Byrne, J.; Gilliam, D.S.; Hunt, J.; Johnson, M.E.; Larson, E.A.; Maxwell, K.; Nedimyer, K.; et al. In situ coral nurseries serve as genetic repositories for coral reef restoration after an extreme cold-water event. Restor. Ecol. 2012, 20, 696–703. [Google Scholar] [CrossRef]
- Van Oppen, M.J.; Oliver, J.K.; Putnam, H.M.; Gates, R.D. Building coral reef resilience through assisted evolution. Proc. Natl. Acad. Sci. USA 2015, 112, 2307–2313. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bay, R.A.; Palumbi, S.R. Multilocus adaptation associated with heat resistance in reef-building corals. Curr. Biol. 2014, 24, 2952–2956. [Google Scholar] [CrossRef] [PubMed]
- Chakravarti, L.J.; van Oppen, M.J.H. Experimental evolution in coral photosymbionts as a tool to increase thermal tolerance. Front. Mar. Sci. 2018, 5, 227. [Google Scholar] [CrossRef]
- Chan, W.Y.; Peplow, L.M.; Menéndez, P.; Hoffmann, A.A.; van Oppen, M.J. Interspecific hybridization may provide novel opportunities for coral reef restoration. Front. Mar. Sci. 2018, 5, 160. [Google Scholar] [CrossRef]
- Quigley, K.M.; Bay, L.K.; Willis, B.L. Leveraging new knowledge of Symbiodinium community regulation in corals for conservation and reef restoration. Mar. Ecol. Prog. Ser. 2018, 600, 245–253. [Google Scholar] [CrossRef]
- Reshef, L.; Koren, O.; Loya, Y.; Zilber-Rosenberg, I.; Rosenberg, E. The coral probiotic hypothesis. Environ. Microbiol. 2006, 8, 2068–2073. [Google Scholar] [CrossRef] [PubMed]
- Peixoto, R.S.; Rosado, P.M.; Leite, D.C.; Rosado, A.S. Beneficial microorganisms for corals (BMC): Proposed mechanisms for coral health and resilience. Front. Microbiol. 2017, 8, 341. [Google Scholar] [CrossRef] [PubMed]
- Epstein, H.E.; Smith, H.A.; Torda, G.; van Oppen, M.J. Microbiome engineering: Enhancing climate resilience in corals. Front. Ecol. Environ. 2019, 17, 100–108. [Google Scholar] [CrossRef]
- Rosado, P.M.; Leite, D.C.; Duarte, G.A.; Chaloub, R.M.; Jospin, G.; da Rocha, U.N.; Saraiva, J.P.; Dini-Andreote, F.; Eisen, J.A.; Bourne, D.G.; et al. Marine probiotics: Increasing coral resistance to bleaching through microbiome manipulation. ISME J. 2019, 13, 921–936. [Google Scholar] [CrossRef]
- Potts, D.C. Natural-selection in experimental populations of reef-building corals (Scleractinia). Evolution 1984, 38, 1059–1078. [Google Scholar] [CrossRef]
- Putnam, H.M.; Gates, R.D. Preconditioning in the reef-building coral Pocillopora damicornis and the potential for trans-generational acclimatization in coral larvae under future climate change conditions. J. Exp. Biol. 2015, 8, 2365–2372. [Google Scholar] [CrossRef] [PubMed]
- Putnam, H.M.; Davidson, J.M.; Gates, R.D. Ocean acidification influences host DNA methylation and phenotypic plasticity in environmentally susceptible corals. Evol. Appl. 2016, 9, 1165–1178. [Google Scholar] [CrossRef] [PubMed]
- McClanahan, T.R. Changes in coral sensitivity to thermal anomalies. Mar. Ecol. Prog. Ser. 2017, 570, 71–85. [Google Scholar] [CrossRef] [Green Version]
- Dixon, G.B.; Bay, L.K.; Matz, M.V. Bimodal signatures of germline methylation are linked with gene expression plasticity in the coral Acropora millepora. BMC Genomics 2014, 15, 1109. [Google Scholar]
- Palumbi, S.R.; Barshis, D.J.; Traylor-Knowles, N.; Bay, R.A. Mechanisms of reef coral resistance to future climate change. Science 2014, 344, 895–898. [Google Scholar] [CrossRef] [PubMed]
- Dimond, J.L.; Gamblewood, S.K.; Roberts, S.B. Genetic and epigenetic insight into morphospecies in a reef coral. Mol. Ecol. 2017, 26, 5031–5042. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Liew, Y.J.; Zoccola, D.; Li, Y.; Tambutté, E.; Venn, A.A.; Michell, C.T.; Cui, G.; Deutekom, E.S.; Kaandorp, J.A.; Voolstra, C.R.; et al. Epigenome-associated phenotypic acclimatization to ocean acidification in a reef-building coral. Sci. Adv. 2018, 4, eaar8028. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Raymundo, L.J.; Maypa, A.P. Getting bigger faster: Mediation of size-specific mortality via fusion in juvenile coral transplants. Ecol. Appl. 2004, 14, 281–295. [Google Scholar] [CrossRef]
- Rinkevich, B. Coral chimerism as an evolutionary rescue mechanism to mitigate global climate change impacts. Glob. Change Biol. 2019, 25, 1198–1206. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Amar, K.O.; Chadwick, N.E.; Rinkevich, B. Coral kin aggregations exhibit mixed allogeneic reactions and enhanced fitness during early ontogeny. BMC Evol. Biol. 2008, 8, 126. [Google Scholar] [CrossRef] [PubMed]
- Puill-Stephan, E.; van Oppen, M.J.H.; Pichavant-Rafini, K.; Willis, B.L. High potential for formation and persistence of chimeras following aggregated larval settlement in the broadcast spawning coral, Acropora millepora. Proc. R. Soc. Lond. Ser. B Biol. Sci. 2012, 279, 699–708. [Google Scholar] [CrossRef] [PubMed]
- Mizrahi, D.; Navarrete, S.A.; Flores, A.A.V. Groups travel further: Pelagic metamorphosis and polyp clustering allow higher dispersal potential in sun coral propagules. Coral Reef. 2014, 33, 443–448. [Google Scholar] [CrossRef]
- Toh, T.C.; Chou, L.M. Aggregated settlement of Pocillopora damicornis planulae on injury sites may facilitate coral wound healing. Bull. Mar. Sci. 2013, 89, 583–584. [Google Scholar] [CrossRef]
- Rinkevich, B.; Shaish, L.; Douek, J.; Ben-Shlomo, R. Venturing in coral larval chimerism: A compact functional domain with fostered genotypic diversity. Sci. Rep. 2016, 6, 19493. [Google Scholar] [CrossRef] [PubMed]
- Jiang, L.; Lei, X.M.; Liu, S.; Huang, H. Fused embryos and pre-metamorphic conjoined larvae in a broadcast spawning reef coral. F1000 Res. 2015, 4, 44. [Google Scholar] [CrossRef]
- Mitsch, W. When will ecologists learn engineering and engineers learn ecology? Ecol. Eng. 2014, 65, 9–14. [Google Scholar] [CrossRef]
- Wild, C.; Hoegh-Guldberg, O.; Naumann, M.S.; Colombo-Pallotta, M.F.; Ateweberhan, M.; Fitt, W.K.; Iglesias-Prieto, R.; Palmer, C.; Bythell, J.C.; Ortiz, J.C.; et al. Climate change impedes scleractinian corals as primary reef ecosystem engineers. Mar. Freshw. Res. 2011, 62, 205–215. [Google Scholar] [CrossRef] [Green Version]
- Aronson, J.; Clewell, A.; Moreno-Mateos, D. Ecological restoration and ecological engineering: Complementary or indivisible? Ecol. Eng. 2016, 91, 392–395. [Google Scholar] [CrossRef] [Green Version]
- Mitsch, W.J.; Jørgensen, S.E. Ecological Engineering and Ecosystem Restoration; John Wiley & Sons: Hoboken, NJ, USA, 2004. [Google Scholar]
- Poore, A.G.; Graham, S.E.; Byrne, M.; Dworjanyn, S.A. Effects of ocean warming and lowered pH on algal growth and palatability to a grazing gastropod. Mar. Biol. 2016, 163, 99. [Google Scholar] [CrossRef]
- Amar, K.O.; Rinkevich, B. A floating mid-water coral nursery as larval dispersion hub: Testing an idea. Mar. Biol. 2007, 151, 713–718. [Google Scholar] [CrossRef]
- Shaish, L.; Levy, G.; Katzir, G.; Rinkevich, B. Coral reef restoration (Bolinao, Philippines) in the face of frequent natural catastrophes. Restor. Ecol. 2010, 18, 285–299. [Google Scholar] [CrossRef]
- Precht, W.F.; Aronson, R.B. Climate flickers and range shifts of reef corals. Front. Ecol. Environ. 2004, 2, 307–314. [Google Scholar] [CrossRef]
- Greenstein, B.J.; Pandolfi, J.M. Escaping the heat: Range shifts of reef coral taxa in coastal Western Australia. Glob. Change Biol. 2008, 14, 513–528. [Google Scholar] [CrossRef]
- Yamano, H.; Sugihara, K.; Nomura, K. Rapid poleward range expansion of tropical reef corals in response to rising sea surface temperatures. Geophys. Res. Lett. 2011, 38, L04601. [Google Scholar] [CrossRef]
- Denis, V.; Mezaki, T.; Tanaka, K.; Kuo, C.Y.; De Palmas, S.; Keshavmurthy, S.; Chen, C.A. Coverage, diversity, and functionality of a high-latitude coral community (Tatsukushi, Shikoku Island, Japan). PLoS ONE 2013, 8, e54330. [Google Scholar] [CrossRef] [PubMed]
- Baird, A.H.; Sommer, B.; Madin, J.S. Pole-ward range expansion of Acropora spp. along the east coast of Australia. Coral Reef. 2012, 31, 1063. [Google Scholar] [CrossRef]
- Tuckett, C.A.; de Bettignies, T.; Fromont, J.; Wernberg, T. Expansion of corals on temperate reefs: Direct and indirect effects of marine heatwaves. Coral Reef. 2017, 36, 947–956. [Google Scholar] [CrossRef]
- Muir, P.R.; Wallace, C.C.; Done, T.; Aguirre, J.D. Limited scope for latitudinal extension of reef corals. Science 2015, 348, 1135–1138. [Google Scholar] [CrossRef] [PubMed]
- Collyer, M.L.; Heilveil, J.S.; Stockwell, C.A. Contemporary evolutionary divergence for a protected species following assisted colonization. PLoS ONE 2011, 6, e22310. [Google Scholar] [CrossRef]
- Chauvenet, A.L.M.; Ewen, J.G.; Armstrong, D.P.; Blackburn, T.M.; Pettorelli, N. Maximizing the success of assisted colonizations. Anim. Conserv. 2013, 16, 161–169. [Google Scholar] [CrossRef]
- Wadgymar, S.M.; Cumming, M.N.; Weis, A.E. The success of assisted colonization and assisted gene flow depends on phenology. Glob. Change Biol. 2015, 21, 3786–3799. [Google Scholar] [CrossRef] [PubMed]
- Kreyling, J.; Bittner, T.; Jaeschke, A.; Jentsch, A.; Jonas Steinbauer, M.; Thiel, D.; Beierkuhnlein, C. Assisted colonization: A question of focal units and recipient localities. Restor. Ecol. 2011, 19, 433–440. [Google Scholar] [CrossRef]
- Jokiel, P.L. Long distance dispersal of reef corals by rafting. Coral Reef. 1984, 3, 113–116. [Google Scholar] [CrossRef]
- Van Oppen, M.J.H.; Puill-Stephan, E.; Lundgren, P.; De’ath, G.; Bay, L.K. First-generation fitness consequences of interpopulational hybridisation in a Great Barrier Reef coral and its implications for assisted migration management. Coral Reef. 2014, 33, 607–611. [Google Scholar] [CrossRef]
- Filbee-Dexter, K.; Smajdor, A. Ethics of assisted evolution in marine conservation. Front. Mar. Sci. 2019, 6, 20. [Google Scholar] [CrossRef]
- Baums, I.B. A restoration genetics guide for coral reef conservation. Mol. Ecol. 2008, 17, 2796–2811. [Google Scholar] [CrossRef]
- Raimundo, R.L.; Guimaraes, P.R., Jr.; Evans, D.M. Adaptive networks for restoration ecology. Trends Ecol. Evol. 2018, 33, 664–675. [Google Scholar] [CrossRef]
- Levin, R.A.; Voolstra, C.R.; Agrawal, S.; Steinberg, P.D.; Suggett, D.J.; van Oppen, M.J.H. Engineering strategies to decode and enhance the genomes of coral symbionts. Front. Microbiol. 2017, 8, 1220. [Google Scholar] [CrossRef]
- National Academies of Sciences, Engineering, and Medicine. A Research Review of Interventions to Increase the Persistence and Resilience of Coral Reefs; The National Academies Press: Washington, DC, USA, 2019. [Google Scholar]
- Webster, N.S.; Reusch, T.B. Microbial contributions to the persistence of coral reefs. ISME J. 2017, 11, 2167–2174. [Google Scholar] [CrossRef]
- Bošković, A.; Rando, O.J. Transgenerational epigenetic inheritance. Annu. Rev. Genet. 2018, 52, 21–41. [Google Scholar] [CrossRef]
- Cropley, J.E.; Suter, C.M.; Beckman, K.B.; Martin, D.I. Germ-line epigenetic modification of the murine Avy allele by nutritional supplementation. Proc. Natl. Acad. Sci. USA 2006, 103, 17308–17312. [Google Scholar] [CrossRef] [PubMed]
- Cooney, C.A.; Dave, A.A.; Wolff, G.L. Maternal methyl supplements in mice affect epigenetic variation and DNA methylation of offspring. J. Nutr. 2002, 132, S2393–S2400. [Google Scholar] [CrossRef] [PubMed]
- Champagne, F.A.; Weaver, I.C.; Diorio, J.; Dymov, S.; Szyf, M.; Meaney, M.J. Maternal care associated with methylation of the estrogen receptor-α1b promoter and estrogen receptor-α expression in the medial preoptic area of female offspring. Endocrinology 2006, 147, 2909–2915. [Google Scholar] [CrossRef] [PubMed]
- Bongiorni, L.; Giovanelli, D.; Rinkevich, B.; Pusceddu, A.; Chou, L.M.; Danovaro, R. First step in the restoration of a highly degraded coral reef (Singapore) by in situ coral intensive farming. Aquaculture 2011, 322, 191–200. [Google Scholar] [CrossRef]
- Rinkevich, B. Will two walk together, except they have agreed? J. Evol. Biol. 2004, 17, 1178–1179. [Google Scholar] [CrossRef]
- Rinkevich, B. Quo vadis chimerism? Chimerism 2011, 2, 1–5. [Google Scholar] [CrossRef] [PubMed]
- Frank, U.; Oren, U.; Loya, Y.; Rinkevich, B. Alloimmune maturation in the coral Stylophora pistillata is achieved through three distinctive stages, four months post metamorphosis. Proc. R. Soc. Lond. Ser. B Biol. Sci. 1997, 264, 99–104. [Google Scholar] [CrossRef]
- Barki, Y.; Gateño, D.; Graur, D.; Rinkevich, B. Soft-coral natural chimerism: A window in ontogeny allows the creation of entities comprised of incongruous parts. Mar. Ecol. Prog. Ser. 2002, 23, 91–99. [Google Scholar] [CrossRef]
- SER (Society for Ecological). The SER International Primer on Ecological Restoration; Society for Ecological Restoration International: Tucson, AZ, USA, 2006. [Google Scholar]
- Hobbs, R.J. Looking for the silver lining: Making the most of failure. Restor. Ecol. 2009, 17, 1–3. [Google Scholar]
- Prober, S.M.; Doerr, V.A.; Broadhurst, L.M.; Williams, K.J.; Dickson, F. Shifting the conservation paradigm: A synthesis of options for renovating nature under climate change. Ecol. Monog. 2019, 89, e01333. [Google Scholar] [CrossRef]
- Higgs, E.; Falk, D.A.; Guerrini, A.; Hall, M.; Harris, J.; Hobbs, R.J.; Jackson, S.T.; Rhemtulla, J.M.; Throop, W. The changing role of history in restoration ecology. Front. Ecol. Environ. 2014, 12, 499–506. [Google Scholar] [CrossRef]
- O’Dea, R.E.; Noble, D.W.; Johnson, S.L.; Hesselson, D.; Nakagawa, S. The role of non-genetic inheritance in evolutionary rescue: Epigenetic buffering, heritable bet hedging and epigenetic traps. Environ. Epigenet. 2016, 2, dvv014. [Google Scholar] [CrossRef] [PubMed]
- Williams, S.L.; Sur, C.; Janetski, N.; Hollarsmith, J.A.; Rapi, S.; Barron, L.; Heatwole, S.J.; Yusuf, A.M.; Yusuf, S.; Jompa, J.; et al. Large-scale coral reef rehabilitation after blast fishing in Indonesia. Rest. Ecol. 2019, 27, 447–456. [Google Scholar] [CrossRef]
- Foo, S.A.; Asner, G.P. Scaling up coral reef restoration using remote sensing technology. Front. Mar. Sci. 2019, 6, 79. [Google Scholar] [CrossRef]

| Avenue | Types of Coral Adaptation | Citations |
|---|---|---|
| Improved gardening methods | Development of various nursery types, adapted for a wide range of needs, improving coral self-attachment; using coral fragments without polyps; clustering of transplants improves outcomes; choosing favorable/improved substrates/coating, caging for recently settled spat—to enhance early post-settlement survival; spat feeding in ex situ nurseries for enhanced growth/survival; improved nursery maintenance by using environmentally friendly antifouling; increasing stocks of larvae from brooding coral species; improving seeding approaches; techniques for improved survival of coral propagules. | [25,26,27,28,29,30,31,32,33,34,35,36,37,38,39,40,41,42,43,44] |
| Ecological engineering | Use of herbivorous fish/invertebrates for improved nursery maintenance; animal-assisted cleaning; engineering of larval supply through transplantation of nursery-farmed gravid colonies; transplantation of ecological engineering species; development of larval hubs and ‘artificial spawning hotspots’; tiling the reef; nubbin fusions for enlarged colonies; micro-fragmentation; serially positioning nurseries to create new mid-water coral biological corridors through stepping stone mechanisms; using dietary habits of grazers as biological controls of fouling macroalgae; large scale restoration acts; enhanced calcification/survival rates via seawater electrolysis. | [1,25,26,27,28,32,39,45,46,47,48,49,50,51,52,53,54,55,56,57,58,59,60,61] |
| Assisted migration/colonization | Moving species outside their historic ranges may mitigate loss of biodiversity in the face of global climate change. | [62,63,64] |
| Assisted genetics/evolution | Enhanced coral adaptation, manipulating of algal symbionts to increase coral resistance to bleaching; using temperature tolerant genotypes; applying interspecific and intraspecific hybridization; using coral nurseries as genetic repositories. | [57,64,65,66,67,68,69,70,71] |
| Assisted microbiome | Adaptation by changing bacterial communities living in tissues, mucus layers and substrates to settle at the shortest timeframe of days/weeks; coral “microbial-therapy” and microbiome inoculation; improved nutrient cycles; contributing to coral host tolerance of thermal stress. | [72,73,74,75] |
| Epigenetics | Creation of novel alleles and traits that can better withstand environmental changes; developing resistance towards adverse conditions. | [46,76,77,78,79,80,81,82,83] |
| Chimerism | Enhanced growth and survival of spat/small colonies; countering the erosion of genetic and phenotypic diversity; high flexibility of chimeric entities on somatic constituents following changes in environmental conditions; the chimera synergistically presents the best-fitting combination of genetic components to environmental challenges; facilitating the healing of exposed coral skeletons | [84,85,86,87,88,89,90,91] |
© 2019 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rinkevich, B. The Active Reef Restoration Toolbox is a Vehicle for Coral Resilience and Adaptation in a Changing World. J. Mar. Sci. Eng. 2019, 7, 201. https://doi.org/10.3390/jmse7070201
Rinkevich B. The Active Reef Restoration Toolbox is a Vehicle for Coral Resilience and Adaptation in a Changing World. Journal of Marine Science and Engineering. 2019; 7(7):201. https://doi.org/10.3390/jmse7070201
Chicago/Turabian StyleRinkevich, Baruch. 2019. "The Active Reef Restoration Toolbox is a Vehicle for Coral Resilience and Adaptation in a Changing World" Journal of Marine Science and Engineering 7, no. 7: 201. https://doi.org/10.3390/jmse7070201
APA StyleRinkevich, B. (2019). The Active Reef Restoration Toolbox is a Vehicle for Coral Resilience and Adaptation in a Changing World. Journal of Marine Science and Engineering, 7(7), 201. https://doi.org/10.3390/jmse7070201





