Abstract
The studies of seasonal phytoplankton dynamics, its growth rate, and microzooplankton grazing were conducted on two stations in the western Black Sea coastal waters near Sevastopol from January 2021 to December 2022. The phytoplankton species composition has remained relatively the same during recent years compared to the end of the last century and the beginning of the 2000s. However, significant changes have occurred in the ratio between different species of diatoms, and the proportion of dinoflagellates was increased, especially in the autumn. Large diatoms and dinoflagellates play a crucial role in forming the phytoplankton biomass seasonal peaks. The first central maximum was observed in July, and the second smaller one was in September–November. Whereas two decades ago, the small diatoms generated three peaks annually: in February, May, and September–October. The maximum values of the phytoplankton growth rate and the rate of its consumption by microzooplankton decreased 2–3 times compared to the beginning of the 2000s. The relative share of primary production consumed by microzooplankton annually averages 35%, two times lower than before.
1. Introduction
Phytoplankton is a primary component of any aquatic ecosystem, and variability determines the development and dynamics of all subsequent trophic levels. Phytoplankton is responsible for approximately half of the global primary production [1] and for 90% of primary production in the ocean [2]. The main share of the Black Sea phytoplankton primary production is formed in the coastal areas, and the greatest values of this parameter are observed in the bays [3]. The total annual value of primary production depends significantly on the seasonal dynamics of phytoplankton biomass. In the Black Sea coastal waters near Sevastopol during the period between 1960 and 2007, the seasonal phytoplankton community biomass was usually characterized by the presence of three peaks: the first one in February, the second at the end of May at the beginning of June, and the third in September–October. The first peak was formed by the small diatom Skeletonema costatum (Greville) Cleve. Two others were conditioned by the bloom of small diatom species, Chaetoceros socialis H.S.Lauder and Chaetoceros curvisetus Cleve [4,5]. In addition, coccolithophore Emiliania huxleyi (Lohmann) W.W.Hay & H.P. Mohler regularly reached the bloom level from late May to early June, corresponding to the blooming period of the small diatoms [4]. However, in 2010, when in all seasons the water temperature was 1–3 °C higher than previously, during a significant part of the year, dinoflagellates dominated and created the summer maximum of phytoplankton biomass. Only in February, when a small biomass peak was observed, S. costatum predominated [5]. In 2014, large diatom species created two biomass maxima at the end of summer and late autumn. In the summer peak, Pseudosolenia calcar-avis (Schultze) B.G.Sundström prevailed; in the autumn maximum, Cerataulina pelagica (Cleve) Hendey dominated. However, during the year bloom of S. costatum, genus Chaetoceros representatives and coccolithophore E. huxleyi were not also observed [6]. The results of our research in the Black Sea coastal waters near Sevastopol in 2020 indicated that the seasonal phytoplankton biomass dynamic at different stations is characterized by one or two peaks in the summer and autumn periods [7]. They were conditioned by the intensive growth of the large diatom species P. calcar-avis and Proboscia alata (Brightwell) Sundström. Thus, since 2010, the number of seasonal peaks of phytoplankton biomass has decreased from three to two and, in some cases, to one.
We consider that the revealed changes in Black Sea coastal waters phytoplankton could be partially caused by interannual variability in the phytoplankton community. However, the two factors probably, play a key role. The first one is an increase in surface water temperature, and the second is rapidly growing anthropogenic pollution of coastal marine areas. In the Black Sea coastal areas near Sevastopol, a positive temperature trend has been detected since the beginning of the current century [8,9]. In addition, as a result of the steep increase in the population of Sevastopol during recent years, anthropogenic pollution of the coastal marine area has increased significantly [9,10]. Probably, these two factors affected not only phytoplankton but also trophic relationships between phytoplankton and its main consumer—microzooplankton [7,9]. In order to understand whether the changes in phytoplankton identified in recent years are sustainable, it is necessary to continue regular studies.
Our paper aims to study the features of seasonal variability of the phytoplankton chlorophyll concentration, its biomass, dominant species, growth rate, as well as microzooplankton grazing under conditions of the combined influence of the climate and anthropogenic pollution in the coastal waters of the Black Sea near Sevastopol during 2021–2022. This goal’s accomplishment will assist in identifying the main strategies for adapting phytoplankton to current environmental conditions and establishing patterns of coastal ecosystem transformation in these conditions.
2. Materials and Methods
2.1. Study Area and Processing
The results were obtained within a period of studies conducted during 2021–2022 in the Black Sea coastal waters near Sevastopol at two stations (Figure 1). The first station was located at the exit from the Quarantine Bay (St. 1), and the second was in Sevastopol Bay (St. 2). The total depth at the stations was 14–18 m.
Figure 1.
Map of the sampling stations: 1—the exit from the Quarantine Bay (St.1); 2—Sevastopol Bay (St. 2).
In the second decade of each month, seawater samples (10 L) were taken in a 0–1 m layer using Niskin bottles. For the nano- and microphytoplankton analysis, 2 L volume samples were concentrated to 50–70 mL by the soft filtration method, using track membranes (1 µm pore size) in the closed inverse filtering funnel. Phytoplankton samples were fixed with neutralized 40% formaldehyde (final concentration in the sample—2%).
The algae cells count and their linear dimensions were measured in a 0.1 mL Naujotte counting chamber in three replications under a light microscope Carl Zeiss Primo Star. We used the equations to calculate the carbon biomass of phytoplankton by cell volumes [11]. Phytoplankton species identification was carried out using the manual [12]. The diatom Skeletonema costatum (Greville) Cleve, which is found in the studied waters in mass quantities, was identified using scanning electron microscopy. For this purpose, a Hitachi SU3500 scanning electron microscope (Japan) was used.
The growth rate of phytoplankton and microzooplankton grazing was determined by the dilution method [13]. Surface seawater was collected and pre-screened with 200 μm nylon netting for dilution experiments at each station. Particle-free seawater was obtained by filtering the seawater through a filter with a pore size of 0.22 μm. Four dilutions of initial seawater samples were used with dilution factor (DF) equal to 0.2, 0.4, 0.8, and 1.0 in two replications. After preparation, the samples were poured into 1 L polycarbonate bottles without supplementary addition of nutrients and exposed in situ during the day. After the daily exposition, initial samples were filtered through Whatman GF/F fiberglass filters (25 mm in diameter). Then the filters were placed into 90% acetone for the chlorophyll a extraction. Its concentration was measured using the Trilogy Laboratory Fluorometer (USA) and calculated according to the equation presented in the work [14]. The daily net phytoplankton growth rate, , for each dilution was evaluated by the Equation:
where and are the initial and final concentrations of chlorophyll a. Based on the obtained values for all four dilutions and the corresponding values of DF the linear regression equations were calculated, which were used for the estimation of true phytoplankton growth rate (µ, day−1) and microzooplankton grazing rate (g, day−1):
Parameters μ and g used in Equation (2) showed a standard error that usually ranged from 5 to 15%.
The nutrient concentrations in the water were determined using the methods described in detail [15]. For example, the Morphey–Riley method was used with ascorbic acid as a reducing agent to determine phosphorus. Determination of nitrite was carried out by the method of Bendschneider and Robinson with sulfonamide in a solution of 1.2 N hydrochloric acid and N-(1-naphthyl)ethylenediamine. The determination of nitrate is based on reducing nitrate to nitrite using copper-plated cadmium. Disodium EDTA salt (Trilon B, Lenreaktiv, St. Petersburg, Russia) was used as a complexing agent. The method for determining silicate in seawater is the Koroleff method, which is based on the colorimetry of a blue silicon-molybdenum complex. Ammonium nitrogen was determined by the Grasshoff–Johanssen method, which is based on the reaction of ammonium in an alkaline solution with an excess of 1,3-dichloroisocyanuric acid (trione) resulting in mono chloramine. The latter in phenolic and nitroprusside solution yields indophenol blue. Dissolved organic matter content in water was conducted using the permanganate oxidizability method [16]. This method involves heating the sample in a boiling water bath with a known amount of potassium permanganate and concentrated sulphuric acid for a fixed time (10 min). Part of the permanganate reduces by oxidizable material in the sample. The consumed permanganate was determined by adding an excess of oxalate solution, followed by titration with permanganate.
2.2. Data Analysis
The average value of the parameters, standard deviation (SD) and standard error (SE) was calculated using MS Excel 2010. The Student’s criterion (t-criterion) was used to assess the significance of the differences between the average values. The normality of the value distribution in the statistical sampling was evaluated using the Sigma Plot version 12.5. The Grafer version 7.0 was used for plotting graphs, and the Surfer version 8.0 was used for plotting maps.
3. Results
3.1. Research Conducted during 2021
At St.1, one peak of phytoplankton biomass (510 mg C·m−3) was observed during the year. It was received in the summer period in July (Figure 2A) at a water temperature of 22 °C. At the same time, the maximum concentration of chlorophyll (2.50 mg·m−3) was revealed. Sufficiently high concentrations of nitrates (4.50 µM) and phosphates (0.30 µM) in the previous period in June (Figure 3A,B) contributed to the formation of the summer phytoplankton maximum, in which the relative diatoms proportion in the total biomass of algae was only 15% (Figure 2A). At that time, the base of phytoplankton biomass was formed by dinoflagellates, among which species Prorocentrum cordatum (Ostenfeld) J.D. Dodge, Prorocentrum micans Ehrenberg, Prorocentrum compressum (Bailey) T.H.Abé ex J.D. Dodge, Gymnodinium simplex (Lohmann) Kofoid & Swezy and Gymnodinium variabile E.C. Herdman dominated (Table 1). A sufficiently high specific growth rate of phytoplankton (0.90 day−1), a shallow rate of microzooplankton grazing (0.09 day−1), and an insignificant relative share of primary production consumed by microzooplankton, which amounted to only 10% (Figure 2B), contributed to the formation of the summer peak of phytoplankton biomass.
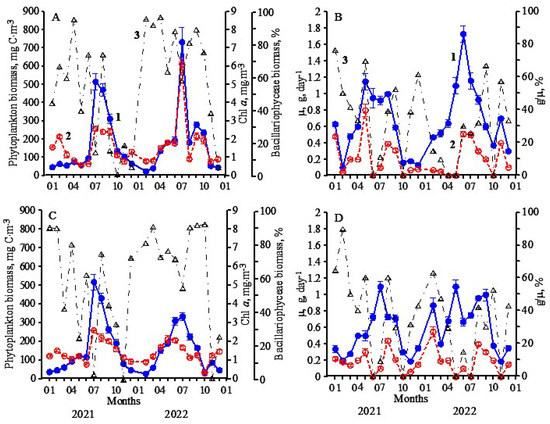
Figure 2.
Seasonal dynamics of phytoplankton, its growth rate and microzooplankton grazing: (A,C)—biomass (1–blue), chlorophyll a concentration (2–red), the relative biomass of diatoms (3–black); (B,D)—growth rate, µ (1), microzooplankton grazing, g (2), ratio g/µ (3) in Station 1 (A,B) and Station 2 (C,D).
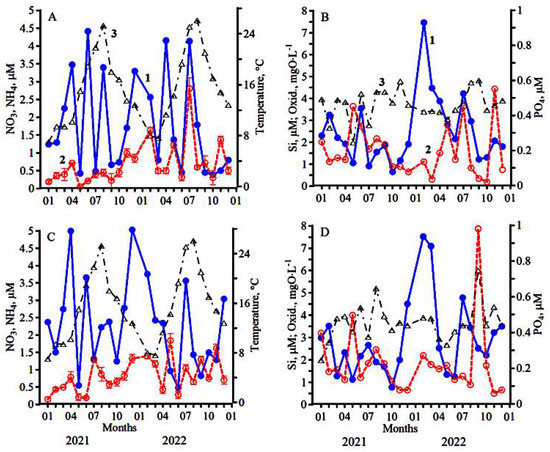
Figure 3.
Seasonal dynamics of nutrient concentrations and the permanganate oxidizability (Oxid.): (A,C)—nitrates (1), ammonium (2), temperature (3); (B,D)—silicates (1), phosphates (2), Oxid. (3) in Station 1 (A,B) and Station 2 (C,D).

Table 1.
Dominant phytoplankton species at Station 1 and Station 2 in 2021.
The low values of phytoplankton biomass (50–70 mg C·m−3) were observed during January–May and in December at a water temperature of 8–15 °C and different concentrations of nutrients. In January–May, from 40 to 95% of phytoplankton biomass consisted of various species of diatoms, and in December, dinoflagellates P. micans, P. cordatum and Procentrum lima (Ehrenberg) F.Stein dominated (Table 1).
One of the phytoplankton species involved in forming the chlorophyll peak in February was the diatom S. costatum, which did not reach the bloom level. However, its species identification was clarified for the first time using light and scanning electron microscopy. The light microscopy showed that in the algal species in question, lenticular-shaped cells with a diameter of 10–12 μm are linked in long chains through a marginal annulus of straight outgrowths. In addition, there is one lamellar chromatophore adjacent to the valve (Figure 4A). The scanning electron microscopy images show that the intercalary outgrowths are flattened tubes with an outer pore at the base and a longitudinal seam running from the pore to the end of the outgrowth (Figure 4B,C). Connections of the intercalary outgrowths of two adjacent cells can be of two types. One is an outgrowth connected to only one outgrowth of the opposite valve forming a straight line. The other one is the lateral extensions of the outgrowth tip connected to extensions of adjacent outgrowths of the opposite valve. In both cases, a solid line is visible at the level of the outgrowth junctions (Figure 4B,C). Terminal outgrowths end with a claw-like protrusion. At some distance from the center valve, there is a rimoportula with an external process (Figure 4D). Comparing our data with the previously taken light and electron microscopy images of representatives of the genus Skeletonema [17,18,19] allows us to conclude that the species under consideration is S. costatum.
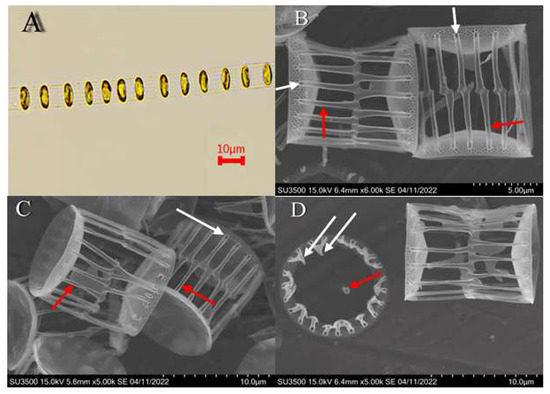
Figure 4.
Skeletonema costatum: (A)—light microscopy, fragment of chain; (B–D)—SEM: (B,C)—valves of two cells connected by fultoportula processes: intercalary fultoportula processes (red arrow), external pore (white arrow); (D)—rimoportula (red arrow), claw-like protrusion (white arrow).
At St. 2, located in Sevastopol Bay, similar changes were observed in the phytoplankton species composition (Table 1), biomass, chlorophyll a concentration, the phytoplankton growth rate, microzooplankton grazing and the share of primary production, consumed by the microzooplankton (Figure 2C,D).
At both stations during a year, the biomass of coccolithophore E. huxleyi was extremely insignificant, and its abundance did not exceed 100 thousand cells· L−1, which is an order of magnitude lower than the bloom level. Similar results were obtained for small diatoms of the genus Chaetoceros.
3.2. Research Conducted during 2022
During most of 2022, the primary biomass of phytoplankton was created by diatoms (Figure 2A,C), which probably was caused by the increased silicon concentration in the water compared to 2021 (Figure 3B,D).
At St.1, two peaks of phytoplankton biomass were observed during the year. The first peak of this parameter (750 mg C·m−3) and a peak of chlorophyll a concentration (6.20 mg·m−3) were observed in July (Figure 2A) at a water temperature of 25.1 °C. High concentrations of nitrates (4.00 µM), silicates (4.10 µM) and phosphates (0.45 µM) in this period (Figure 3A,B), as well as the highest value of the phytoplankton growth rate (1.75 day−1) and its relatively weak consumption by the microzooplankton (0.50 day−1), contributed to forming the summer algae peak. In this period, a significant share of phytoplankton biomass (60%) was formed by large diatoms and, first of all, P. calcar-avis and P. alata (Table 2). The rest of the biomass was created by dinoflagellates.

Table 2.
Dominant species in the phytoplankton of station 1 and station 2 in 2022.
The second peak of biomass (280 mg C·m−3) and chlorophyll a concentration (2.20 mg·m−3) was detected in September (Figure 2B). During this period, the biomass of diatoms was 90% (Figure 2A), and among the species, both the large and small algae were presented (Table 2). However, the relative share of primary production consumed by microzooplankton varied from 0 to 68%. In the summer biomass maximum in July, it was 30%, while during the autumn peak, it increased to 50% (Figure 2B). The low phytoplankton biomass (20–50 mg C·m−3) and the chlorophyll concentration (0.20–0.50 mg·m−3) were seen from February to March and November to December at a water temperature of 8.0–12.5 °C.
At St. 2, the first maximum of phytoplankton biomass (330 mg C·m−3) was observed, as well as at St. 1, in July (Figure 2C). The specific growth rate of phytoplankton was 0.75 day−1, and microzooplankton grazing was extremely low (0.03 day−1), contributing to the biomass peak formation. During this period, the relative diatoms’ share in the total phytoplankton biomass was equal to 55% (Figure 2C). Large species were dominated, while the rest of the biomass was created mainly by dinoflagellates (Table 2). The second weak peak of biomass (85 mg C·m−3) and chlorophyll a concentration (1.20 mg·m−3) was revealed in November. It was formed by various dinoflagellate species (98%).
The relative coccolithophores biomass was no higher than 2–4% at both stations during the year. Coccolithophore E. huxleyi dominated this algae group; however, its abundance was not exceeded 150 thousand cells·L−1, which is approximately seven times lower than the bloom level for this species.
The permanganate oxidizability value at St. 1 changed from 3.06 to 4.82 mg O·L−1 (Figure 3B), and at St. 2, it changed from 2.67 to 6.01 mg O·L−1 (Figure 3D).
The summarization of all hydrobiological and hydrochemical data obtained at two stations of the coastal waters near Sevastopol in 2021–2022 indicated that the main phytoplankton biomass is created within this area in the summer, composing an average of 343 mg C·m−3 (Table 3). The lowest values of algae biomass (on average 45 mg C·m−3) were observed in winter. Intermediate values of phytoplankton biomass were obtained in spring and autumn. In the autumn period, the relative biomass of diatoms decreased to an average of 42%. This value is significantly lower than in spring (p = 0.045). In autumn, the average content of nitrates in the water, necessary for intensive diatoms development, is lower than in spring (at p = 0.014). The silicon deficiency relative to nitrogen (Si/N = 0.71) in the autumn period also contributed to the decrease of the diatom role. The average value of the permanganate oxidizability in autumn was highest, on average 4.14 mg O ·L−1, and significantly differed from the average values of this indicator obtained for winter and spring (p = 0.016).

Table 3.
Hydrobiological and hydrochemical parameters in the different seasons of 2021–2022.
4. Discussion
The analysis of satellite information [20] and data from direct determinations of species composition and phytoplankton biomass [5,21] shows that significant changes have occurred in the state of the primary producers of the Black Sea pelagic ecosystem over the past two decades. These changes probably arose due to increased surface layer temperature observed in the Black Sea and on the global scale [22,23]. For example, in 80–90 of the last century, during a year, the maximum biomass of phytoplankton in the Black Sea deep-water area was observed from late February till early March and was formed by small-celled diatoms of the genus Pseudo-nitzschia [24]. However, currently, it occurs in September due to the dominance of the large-cell diatom species P. calcar-avis and P. alata [20]. Changes have also occurred in the western shelf of the Black Sea, where phytoplankton blooms have become rarer, and their average annual biomass has decreased [25].
The ecosystem of the Black Sea coastal waters, in addition to climate change, is affected by growing anthropogenic pollution. In the last few years, the concentrations of nitrates, ammonium and silicates in the Sevastopol bays have reached their highest values, as we marked in 2020 [7] and 2021–2022. The main reason is the increased inflow of nutrients into the sea with riverine discharge, urban wastewater, and terrestrial runoff [10,26]. Water pollution with organic matter has also increased, as seen in the high permanganate oxidizability values. Until 2004, this parameter was 1–3 mg O·L−1 in the water areas under consideration [27], whereas in 2021–2022, according to our data, it was much higher, and in some cases, it reached 5.19–6.01 mg O·L−1. A year earlier, during the warm period, the value of permanganate oxidizability occasionally increased to 6.01–6.52 mg O·L−1 [7]. Along with traditional pollutants (oil, heavy metals, different industrial products) discharged into the sea, various artificial polymers enter the marine environment. Pollution in the Sevastopol bays has increased significantly due to the rapid growth of the population of Sevastopol in the last decade [10].
The increased anthropogenic pollution of the marine waters we studied probably contributes significantly to the algae species composition. Experimental studies carried out by us on marine microalgae cultures showed that the algae placed in water samples from bays near Sevastopol in other months often experienced either a toxic impact from complex water pollution or growth stimulation [28,29]. The growth inhibition was observed typically in the small diatom Phaeodactylum tricornutum Bohlin and coccolithophore E. huxleyi. At the same time, the growth of the dinoflagellates Prorocentrum pussilum (Schiller) Dodge & Bibby, Prorocentrum cordatum (Ostenfeld) J.D.Dodge and Gyrodinium fissum (Levander) Kofoid & Swezy was more intense in the water from the bays than in control. Probably, this was one of the reasons for the decrease in the abundance of small diatoms and E. huxleyi in the Black Sea bays. On the other hand, the large diatom species P. calcar-avis., P. alata, L. flabellate and H. hyaline are probably also highly resistant to pollution due to the low values of their specific surface area (0.20–0.30 µm−1).
We assume that both anthropogenic pollution and rising water temperatures had a significant impact on the changes in phytoplankton that were identified as early as 2010 [5]. According to the data obtained in the last decade, it can be concluded that the seasonal dynamics of phytoplankton biomass in coastal waters near Sevastopol have changed significantly, and the number of seasonal peaks has decreased. These peaks were formed, most often, by dinoflagellates and large diatoms [6,7]. Among diatoms, there is presently no single small species that would cause algal blooms. However, previously small diatom species S. costatum and species of the genus Chaetoceros regularly caused blooms in winter, spring, and autumn [4]. One of the main reasons for these changes in phytoplankton probably is associated with the weakening of the water masses’ dynamic activity in winter-spring and autumn due to warming. This phenomenon could cause a decrease in the number of resting spores of small planktonic algae entering the photosynthesis zone from the lower layers of the sea. For example, in the early 2000s, in February, while S. costatum bloomed, the surface water temperature was about 7 °C, which caused the high dynamic activity of water masses [4]. In 2020, it reached 8.2–8.4 °C during the same period [8]; in 2021, it rose to 9.2–9.4 °C, and in 2022 it was 8.2 °C, which reduced the water mixing process. At that time, the S. costatum abundance was two orders of magnitude lower than the level typical for a bloom of this species.
The high summer water temperature, which has increased by several degrees in recent years, directly negatively affects diatoms. This phenomenon is probably conditioned by the fact that the maximum growth temperature, which, when exceeded, causes the degradation of their representatives, particularly S. costatum and C. curvisetus, and is 23–25 °C. For dinoflagellates, this parameter is 4–6 °C higher. However, some diatom species isolated from the Black Sea plankton, for instance, Thalassiosira weissflogii (Grunow) G.Fryxell & Hasle and Cylindrotheca closterium (Ehrenberg) Reimann & J.C. Lewin do not differ from dinoflagellates by their temperature resistance [30]. It is perhaps due to this reason that the previously dominating diatoms, for example, representatives of the genus Chaetoceros, were found in small amounts in the researched bays during the summer period. At the same time, other species, such as P. calcar-avis and P. alata, began to prevail, which may indicate a high degree of their temperature resistance.
Light is an essential factor regulating the growth of algae. In the Black Sea, during most of the year, this factor is optimal for surface water phytoplankton [5,6]. However, in summer, algae, including large diatoms, can be subjected to photoinhibition, as the intensity of solar radiation there reaches 1000 µmol quantum·m−2·s−1 [5,31]. In this context, the ability of large cells to resist photoinhibition due to chloroplast packing effects has particular importance [31,32,33]. This effect occurs through the formation of chloroplast layers, which in a large cell leads to internal self-shading and prevents photodamage to the pigment complex that absorbs light [31,34,35]. At the same time, dinoflagellates, making vertical migrations, can avoid high light intensities and, in case of nutrient deficiency, switch to heterotrophic nutrition, including phagotrophy [36,37].
One of the fundamental abiotic factors that regulate the seasonal course of phytoplankton biomass and its species composition is the nutrients and ratio between their primary forms [38,39]. Some authors believe that relatively high concentrations of nitrates, silicates, and phosphates are necessary for the intensive growth of diatoms, and the molar ratios between the mineral forms of nitrogen and phosphorus, as well as the ratios between silicon and nitrogen, should be close to the modified Redfield ratio: N:P:Si = 16:1:15 [40,41]. However, in the waters we studied during spring, when diatoms dominated most frequently, the nutrient concentrations and their molar ratios varied widely (Table 3). At the same time, the ratio N:P:Si averaged 19:1:27. Similar results were obtained for nutrients in the spring period for the northeastern part of the Black Sea, where small species of diatoms, such as Pseudo-nitzschia pseudodelicatissima (Hasle) Hasle and Chaetoceros curvisetus Cleve dominated [39]. On the other hand, in the coastal sea waters near Sevastopol, large species diatoms P. calcar-avis and P. alata dominated during the summer period with an average value of N:P:Si = 16:1:11 and at sufficiently high concentrations of nutrients (Table 3).
Large diatoms have several adaptive capabilities that ensure their dominance in the summer, even with nutrient deficiencies. These algae, having a sizeable intracellular vacuole, can store nutrients that ensure their growth without external sources of nitrates, phosphates, and silicates. As we demonstrated, the P. calcar-avis culture, in the condition of nutrients absence in the water, can perform five cell divisions using the nutritious substance contained in a large cellular vacuole, which occupies approximately 80% of the cell volume [42]. An insignificant cytoplasm volume in the studied species increases the efficiency of nutrient transport from the external environment into the cell, and the cytoplasm movement along the cell membrane enhances the intracellular transport of nutrients [43,44,45]. Under conditions of nutrient deficiency in stratified waters during the warm season, P. calcar-avis can derive nutrients from the depths due to periodic weakening of the temperature stratification of the water column resulting from increased wind activity and storms [20,46]. A potential additional nitrogen source for the species we study can be atmospheric nitrogen, the ability to fix which has been demonstrated for cyanobacteria Richelia intracellularis Schmidt—endosymbiont of some diatom species, including Rhizosolenia genus [47]. Additionally, large diatom cells can regulate their buoyancy, permitting them to migrate into waters with a higher nutrient concentration and absorb them [48,49,50].
Among the biotic factors contributing to the predominance of large algae, the main one is the grazing of phytoplankton by micro- and mesozooplankton [51,52,53]. With a sufficiently large cell volume and a silicon shell, large diatoms are practically not consumed by microzooplankton [5,54]. At the same time, the preferred food source for it is small algae [36,37,55,56]. The nutritional value of large diatoms for zooplankton is lower than that of small ones due to the lower carbon content in their cells [11,57]. In addition, according to one of the hypotheses, the presence of a certain lag time before the appearance of a predator of adequate size allows large diatoms to increase their biomass [58,59]. It is also likely that large diatoms are more resistant to virus attacks [60,61].
It is considered that microzooplankton is the main consumer of primary phytoplankton production in the sea. It accounts for an average of 65% of the annual primary production consumption [5,62,63]. However, in the Black Sea coastal waters near Sevastopol in 2014 [7] and 2020 [9], as well as in 2021–2022, there was a significant decrease in the primary production share consumed by microzooplankton. The main reason for such a decrease, in our opinion, could be a deterioration of the nutrition quality for microzooplankton. Water pollution is probably the second most important factor. It was found that during the warm period of 2020, as the content of dissolved organic substances in the waters of Sevastopol bays increased, microzooplankton predation decreased [9]. Due to the decrease in microzooplankton grazing, the transfer of matter and energy from phytoplankton to higher trophic levels through protozoa has decreased in recent years to an average of 35%.
5. Conclusions
In the studied coastal waters of the Black Sea during the last years, significant changes in the phytoplankton biomass annual dynamics and its dominant species composition have taken place. At the end of the last century and in the early 2000s, three biomass maximums during the year were regularly observed due to the intensive development of small species of diatoms S. costatum, C. socialis, and C. curvisetus. Presently, the share of these species in total phytoplankton biomass is insignificant. According to our proposal, this is mainly due to the influence of increased water temperature and growing anthropogenic pollution. At the same time, interannual variability of phytoplankton parameters cannot be ruled out.
In recent years, one or two maximums of phytoplankton biomass have been recorded. The first major maximum is formed in July by dinoflagellates or large species of diatoms, such as P. calcar-avis and P. alata. The second minor maximum was observed in the autumn period and is due to the intensive development of various species of either diatoms or dinoflagellates. Possible mechanisms of adaptation of large diatoms and dinoflagellates to modern environmental conditions are discussed. These species are able to adapt to strong light, high temperatures, nutrient deficiencies in the water, and pollution.
Currently, as a result of the phytoplankton species composition restructuring, the maximum values of the phytoplankton growth rate and microzooplankton grazing have significantly decreased compared to the respective values in the early 2000s. Reduced nutrition quality for microzooplankton and the suppressive effects of water pollutants on them caused a twofold decrease in the relative share of primary production consumed by microzooplankton. This suggests that the flow of matter and energy from phytoplankton through microzooplankton to the highest trophic levels has significantly decreased.
Author Contributions
Conceptualization and methodology, L.S.; field investigation, L.S. and N.K.; data—formal analysis, L.S. and N.K.; writing—original draft and text, L.S. and T.G. All authors have read and agreed to the published version of the manuscript.
Funding
This research was written within the framework of the state assignment of A.O. Kovalevsky Institute of Biology of the Southern Seas of RAS No. 121041400077-1 (“Functional, metabolic and toxicological aspects of the existence of aquatic organizations and their populations in habitats with different physical and chemical regime”) and on the state assignment of the Institute of Natural and Technical Systems, project reg. no. 121122300077-8 (“Analysis of the mechanism of integrated biological indicators of aquatic ecosystems condition application in the regions of recreational and tourism specialization”).
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
All data used in this study are available upon request from the corresponding author.
Acknowledgments
The authors are grateful to Irina Babich, Olga Alatartseva and Natalia Minina for their help in conducting a field study and obtaining data on chlorophyll concentration, phytoplankton biomass and species composition of algae.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Field, C.B.; Behrenfeld, M.J.; Randerson, J.T.; Falkowski, P.G. Primary Production of the Biosphere: Integrating Terrestrial and Oceanic Components. Science 1998, 281, 237–240. [Google Scholar] [CrossRef] [PubMed]
- Duarte, C.M.; Cebrián, J. The fate of marine autotrophic production. Limnol. Oceanogr. 1996, 41, 1758–1766. [Google Scholar] [CrossRef]
- Bologa, A.S.; Frangopol, P.T.; Vedernikov, V.I.; Stelmakh, L.V.; Yunev, O.A.; Yilmaz, A.; Oguz, T. Distribution of Planktonic Primary Production in the Black Sea. In Environmental Degradation of the Black Sea: Challenges and Remedies; Besiktepe, S.T., Unluata, U., Bologa, A.S., Eds.; Kluwer Academic Publishers: Dordrecht, The Netherlands, 1998; pp. 131–145. [Google Scholar]
- Senicheva, M.I. Species Diversity, Seasonal and Interannual Variability of Microalgae in Plankton off the Coast of Crimea. In the Black Sea microalgae: Problems of Biodiversity Preservation and Biotechnological Usage; Tokarev, Y.N., Finenko, Z.Z., Shadrin, N.V., Eds.; ECOSI-Gidrofizika: Sevastopol, Russia, 2008; pp. 5–17. [Google Scholar]
- Stelmakh, L.V.; Georgieva, E.Y. Microzooplankton: The trophic role and involvement in the phytoplankton loss and bloom formation in the Black Sea. Turk. J. Fish. Aquat. Sci. 2014, 14, 955–964. [Google Scholar] [CrossRef]
- Finenko, Z.Z.; Stelmakh, L.V.; Mansurova, I.M.; Georgieva, E.Y.; Tsilinsky, V.S. Seasonal dynamics of structural and functional indicators of phytoplankton community in Sevastopol Bay. Environ. Control. Syst. 2017, 9, 73–82. [Google Scholar]
- Stelmakh, L.; Kovrigina, N. Phytoplankton Growth Rate and Microzooplankton Grazing under Conditions of Climatic Changes and Anthropogenic Pollution in the Coastal Waters of the Black Sea (Sevastopol Region). Water 2021, 13, 3230. [Google Scholar] [CrossRef]
- Repetin, L.N. Spatial and temporal variability of the temperature regime of the Black Sea coastal zone. Ecol. Saf. Coast. Shelf Zones Integr. Use Shelf Resour. 2012, 26, 99–116. [Google Scholar]
- Stelmakh, L.; Kovrigina, N.; Gorbunova, T. Phytoplankton adaptation strategies under the influence of climatic changes and anthropogenic pressure on the Black Sea coastal ecosystems on the example Sevastopol Bay. Ecol. Montenegrina 2020, 37, 34–42. [Google Scholar] [CrossRef]
- Orekhova, N.A.; Varenik, A.V. Current Hydrochemical Regime of the Sevastopol Bay. Phys. Oceanogr. 2018, 25, 124–135. [Google Scholar] [CrossRef]
- Menden-Deuer, S.; Lessard, E.J. Carbon to volume relationships for dinoflagellates, diatoms, and other protist plankton. Limnol. Oceanogr. 2000, 45, 569–579. [Google Scholar] [CrossRef]
- Tomas, C.R. Identifying Marine Diatoms and Dinoflagellates; Academic Press: New York, NY, USA, 1997; p. 858. [Google Scholar]
- Landry, M.R.; Hassett, R.P. Estimating the Grazing Impact of Marine Micro-Zooplankton. Mar. Biol. 1982, 67, 283–288. [Google Scholar] [CrossRef]
- JGOFS Protocols. Protocols for the Joint Global Ocean Flux Study (JGOFS) Core Measurement; Manual and Guides; UNESCO: Paris, France, 1994; p. 170. [Google Scholar] [CrossRef]
- Grasshoff, K.; Kremling, K.; Ehrhardt, M. Methods of Seawater Analysis; Verlag Chemie: Weinheim, Germany, 1976; p. 317. [Google Scholar]
- Skopintsev, B.A. Formation of the Modern Chemical Composition of the Black Sea Waters; Gidrometeoizdat: Leningrad, Russia, 1975; p. 33. [Google Scholar]
- Sarno, D.; Kooistra, W.H.C.F.; Balzano, S.; Hargraves, P.E.; Zignone, A. Diversity in the genus Skeletonema (Bacillariophyceae): III. Phylogenetic position and morphological variability of Skeletonema costatum and Skeletonema grevillei, with the description of Skeletonema ardens sp. Nov. J. Phycol. 2007, 43, 156–170. [Google Scholar] [CrossRef]
- Gu, H.; Zhang, X.; Sun, J.; Luo, Z. Diversity and Seasonal Occurrence of Skeletonema(Bacillariophyta) Species in Xiamen Harbour and Surrounding Seas, China. Cryptogam. Algologie. 2012, 33, 245–263. [Google Scholar] [CrossRef]
- Shevchenko, O.G.; Ponomareva, A.A.; Shulgina, M.A.; Orlova, T.Y. Phytoplankton in the Coastal Waters of Russky Island, Peter the Great Bay, Sea of Japan. Bot. Pac. A J. Plant Sci. Conserv. 2019, 8, 133–141. [Google Scholar] [CrossRef]
- Yunev, O.A.; Carstensen, J.; Stelmakh, L.V.; Belokopytov, V.N.; Suslin, V.V. Reconsideration of the phytoplankton seasonality in the open Black Sea. Limnol. Oceanogr. Lett. 2020, 6, 51–59. [Google Scholar] [CrossRef]
- Mikaelyan, A.S.; Kubryakov, A.A.; Silkin, V.A.; Pautova, L.A.; Chasovnikov, V.K. Regional climate and patterns of phytoplankton annual succession in the open waters of the Black Sea. Deep Sea Res. Part I 2018, 142, 44–57. [Google Scholar] [CrossRef]
- Oguz, T.; Gilbert, D. Abrupt transitions of the top-down controlled Black Sea pelagic ecosystem during 1960–2000: Evidence for regime-shifts under strong fishery exploitation and nutrient enrichment modulated by climate-induced variations. Deep Sea Res. Part I 2007, 54, 220–242. [Google Scholar] [CrossRef]
- Hã Der, D.-P.; Gao, K. Interactions of anthropogenic stress factors on marine phytoplankton. Front. Environ. Sci. 2015, 3, 1–14. [Google Scholar] [CrossRef]
- Finenko, Z.Z.; Krupatkina, D.K. Primary production in the Black Sea in the winter-spring period. Oceanology 1993, 33, 94–104. [Google Scholar]
- Yunev, O.; Carstensen, J.; Stelmakh, L.; Belokopytov, V.; Suslin, V. Temporal changes of phytoplankton biomass in the western Black Sea shelf waters: Evaluation by satellite data (1998–2018). Estuarine. Coast. Shelf Sci. 2022, 271, 107865. [Google Scholar] [CrossRef]
- Sovga, E.E.; Mezenceva, I.V.; Hmara, T.V.; Slepchuk, K.A. On the prospects and possibilities of assessing the self-cleaning ability of the water area of Sevastopol Bay. Environ. Saf. Coast. Shelf Zones Integr. Use Shelf Resour. 2014, 28, 153–164. (In Russian) [Google Scholar]
- Kuftarkova, E.A.; Kovrigina, N.P.; Rodionova, N.Y.; Bobko, N.I. Hydrochemical Characteristics of the Coastal Waters of the Crimean Peninsula. In Mariculture of Mussels in the Black Sea; Ivanov, V.A., Ed.; ECOSI-Hydrophysica: Sevastopol, Russia, 2007; pp. 74–93. [Google Scholar]
- Stelmakh, L.; Kovrigina, N.; Gorbunova, T. Response of marine microalgae Phaeodactylum tricornutum, Prorocentrum cordatum and Gyrodinium fissum to complex pollution of Sevastopol bays (Black Sea). Ecol. Montenegrina 2021, 48, 109–116. [Google Scholar] [CrossRef]
- Stelmakh, L.V.; Mansurova, I.M. Functional state of cultures of marine microalgae as an indicator of the level of pollution of the waters of the Sevastopol Bay. Monit. Syst. Environ. 2021, 4, 83–90. [Google Scholar] [CrossRef]
- Akimov, A.I.; Solomonova, E.S. Characteristics of Growth and Fluorescence of Certain Types of Algae during Acclimation to Different Temperatures under Culture Conditions. Oceanology 2019, 59, 316–326. [Google Scholar] [CrossRef]
- Silkin, V.; Fedorov, A.; Flynn, K.J.; Paramonov, L.; Pautova, L. Protoplasmic streaming of chloroplasts enables rapid photoacclimation in large diatoms. J. Plankton Res. 2021, 43, 831–845. [Google Scholar] [CrossRef]
- Finkel, Z.V. Light absorption and size scaling of light-limited metabolism in marine diatoms. Limnol. Oceanogr. 2001, 46, 86–94. [Google Scholar] [CrossRef]
- Finkel, Z.V.; Irwin, A.J. Modeling Size-dependent Photosynthesis: Light Absorption and the Allometric Rule. J. Theor. Biol. 2000, 204, 361–369. [Google Scholar] [CrossRef]
- Raven, J.A. The cost of photoinhibition. Physiol. Plant. 2011, 142, 87–104. [Google Scholar] [CrossRef]
- Bouman, H.A.; Platt, T.; Doblin, M.; Figueiras, F.G.; Gudmundsson, K.; Gudfinnsson, H.G.; Huang, B.; Hickman, A. Photosynthesis–irradiance parameters of marine phytoplankton: Synthesis of a global data set. Earth Syst. Sci. Data 2018, 10, 251–266. [Google Scholar] [CrossRef]
- Sherr, E.B.; Sherr, B.F. Heterotrophic dinoflagellates: A significant part of microzooplankton biomass and major grazers of diatoms in the sea. Mar. Ecol. Progr. Ser. 2007, 352, 187–197. [Google Scholar] [CrossRef]
- Putland, J.N.; Iverson, R.L. Microzooplankton: Major herbivores in an estuarine planktonic food web. Mar. Ecol. Prog. Ser. 2007, 345, 63–73. [Google Scholar] [CrossRef]
- Goldenberg, S.U.; Taucher, J.; Fernandez-Méndez, M.; Ludwig, A.; Aristegui, J.; Baumann, M.; Ortiz, J.; Stuhr, A.; Riebesell, U. Nutrient composition (Si: N) as driver of plankton communities during artificial upwelling. Front. Mar. Sci. 2022, 9, 1015188. [Google Scholar] [CrossRef]
- Silkin, V.A.; Pautova, L.A.; Giordano, M.; Chasovnikov, V.K.; Vostokov, S.V.; Podymov, O.I.; Pakhomova, S.V.; Moskalenko, L.V. Drivers of phytoplankton blooms in the northeastern Black Sea. Mar. Pollut. Bull. 2018, 138, 274–284. [Google Scholar] [CrossRef] [PubMed]
- Brzezinski, M.A. The Si:C:N ratio of marine diatoms: Interspecific variability and the effect of some environmental variables. J. Phycol. 1985, 21, 347–357. [Google Scholar] [CrossRef]
- Choudhury, A.K.; Bhadury, P. Relationship between N:P: Si ratio and phytoplankton community composition in a tropical estuarine mangrove ecosystem. Biogeosciences Discuss. 2015, 12, 2307–2355. [Google Scholar] [CrossRef]
- Stelmakh, L.V. Features of the Structural and Functional Characteristics of the Diatom Alga Pseudosolenia calcar-avis. Inland Water Biol. 2022, 15, 315–323. [Google Scholar] [CrossRef]
- Raven, J.A. The role of vacuoles. New Phytol. 1987, 106, 357–422. [Google Scholar] [CrossRef]
- Richardson, T.L.; Ciotti, A.M.; Cullen, J.J. Physiological and optical properties of rhizosolenia formosa (bacillariophyceae) in the context of open-ocean vertical migration. J. Phycol. 1996, 32, 741–757. [Google Scholar] [CrossRef]
- Baek, S.H.; Shimode, S.; Han, M.-S.; Kikuchi, T. Growth of dinoflagellates, Ceratium furca and Ceratium fusus in Sagami Bay, Japan: The role of nutrients. Harmful Algae 2008, 7, 729–739. [Google Scholar] [CrossRef]
- Barton, A.D.; Ward, B.A.; Williams, R.G.; Follows, M.J. The impact of fine-scale turbulence on phytoplankton community structure. Limnol. Oceanogr. Fluids Environ. 2014, 4, 34–49. [Google Scholar] [CrossRef]
- Anderson, E.E.; Wilson, C.; Knap, A.H.; Villareal, T.A. Summer diatom blooms in the eastern North Pacific gyre investigated with a long-endurance autonomous surface vehicle. PeerJ 2018, 6, e5387. [Google Scholar] [CrossRef]
- Villareal, T.A.; Woods, S.; Moore, J.K.; Culver-Rymsza, K. Vertical migration of Rhizosolenia mats and their significance to NO3 −fluxes in the central North Pacific gyre. J. Plankton Res. 1996, 18, 1103–1121. [Google Scholar] [CrossRef]
- Gemmell, B.J.; Oh, G.; Buskey, E.J.; Villareal, T.A. Dynamic sinking behaviour in marine phytoplankton: Rapid changes in buoyancy may aid in nutrient uptake. Proc. R. Soc. B. 2016, 283, 20161126. [Google Scholar] [CrossRef] [PubMed]
- Flynn, K.J.; Skibinski, D.O.F.; Lindemann, C. Effects of growth rate, cell size, motion, and elemental stoichiometry on nutrient transport kinetics. PLoS Comput. Biol. 2018, 14, e1006118. [Google Scholar] [CrossRef] [PubMed]
- Kiørboe, T.A. Mechanistic Approach to Plankton Ecology; Princeton University Press: Princeton, NJ, USA, 2008; p. 224. [Google Scholar]
- Kiørboe, T. How zooplankton feed: Mechanisms, traits and tradeoffs. Biol. Rev. Cam. Phil. Soc. 2011, 86, 311–339. [Google Scholar] [CrossRef]
- Ward, B.A.; Dutkiewicz, S.; Jahn, O.; Follows, M.J. A size-structured food-web model for the global ocean. Limnol. Oceanogr. 2012, 57, 1877–1891. [Google Scholar] [CrossRef]
- Wirtz, K.W. Who is eating whom? Morphology and feeding type determine the size relation between planktonic predators and their ideal prey. Mar. Ecol. Prog. Ser. 2012, 445, 1–12. [Google Scholar] [CrossRef]
- Wirtz, K.W.; Sommer, U. Mechanistic origins of variability in phytoplankton dynamics. Part II: Analysis of mesocosm blooms under climate change scenarios. Mar. Biol. 2013, 160, 2503–2516. [Google Scholar] [CrossRef]
- Saito, H.; Ota, T.; Suzuki, K.; Nishioka, J.; Tsuda, A. Role of heterotrophic dinoflagellate Gyrodinium sp. in the fate of an iron induced diatom bloom. Geophys. Res. Lett. 2006, 33, 1–4. [Google Scholar] [CrossRef]
- McBeain, K.H.; Halsey, K.A. Altering phytoplankton growth rates changes their value as food for microzooplankton grazers. Aquat. Microb. Ecol. 2018, 82, 19–29. [Google Scholar] [CrossRef]
- Behrenfeld, M.J.; Halsey, K.H.; Boss, E.; Karp-Boss, L.; Milligan, A.J.; Peers, G. Thoughts on the evolution and ecological niche of diatoms. Ecol. Monogr. 2021, 91, e01457. [Google Scholar] [CrossRef]
- Behrenfeld, M.J.; Boss, E.; Halsey, K.H. Phytoplankton community structuring and succession in a competition-neutral resource landscape. ISME Commun. 2021, 1, 12. [Google Scholar] [CrossRef] [PubMed]
- Hamm, C.E.; Merkel, R.; Springer, O.; Jurkojc, P.; Maier, C.; Prechtel, K.; Smetacek, V. Architecture and material properties of diatom shells provide effective mechanical protection. Nature 2003, 421, 841–843. [Google Scholar] [CrossRef] [PubMed]
- Flynn, K.J.; Kimmance, S.A.; Clark, D.R.; Mitra, A.; Polimene, L.; Wilson, W.H. Modelling the Effects of Traits and Abiotic Factors on Viral Lysis in Phytoplankton. Front. Mar. Sci. 2021, 8, 667184. [Google Scholar] [CrossRef]
- Schmoker, C.; Hernandes-Leon, S.; Calbet, A. Microzooplankton grazing in the oceans: Impacts, data variability, knowledge gaps and future directions. J. Plankton Res. 2013, 35, 691–706. [Google Scholar] [CrossRef]
- Landry, M.R.; Karen, E.; Selph, M.D.; Décima, M.; Gutiérrez-Rodríguez, A.; Stukel, M.R.; Taylor, A.G.; Pasulka, A.L. Phytoplankton production and grazing balances in the Costa Rica Dome. J. Plankton Res. 2015, 38, 366–379. [Google Scholar] [CrossRef] [PubMed]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).