Microalgae-Based PUFAs for Food and Feed: Current Applications, Future Possibilities, and Constraints
Abstract
:1. Introduction
2. Current Market for Microalgae-Derived Products, with a Special Focus on PUFAs
3. Microalgae-Based Genetic Engineering Technology for PUFA Production: Most Relevant Patents
4. Main Bottlenecks for PUFA-Derived Microalgae for Large-Scale Production and Possible Solutions
5. Concluding Remarks
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- FAO. The State of World Fisheries and Aquaculture 2020; FAO: Rome, Italy, 2020. [Google Scholar]
- Briggs, M.A.; Petersen, K.S.; Kris-Etherton, P.M. Saturated Fatty Acids and Cardiovascular Disease: Replacements for Saturated Fat to Reduce Cardiovascular Risk. Healthcare 2017, 5, 29. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Dessì, M.; Noce, A.; Bertucci, P.; Manca di Villahermosa, S.; Zenobi, R.; Castagnola, V.; Addessi, E.; Di Daniele, N. Atherosclerosis, dyslipidemia, and inflammation: The significant role of polyunsaturated Fatty acids. ISRN Inflamm. 2013, 2013, 191823. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gu, Z.; Shan, K.; Chen, H.; Chen, Y.Q. n-3 Polyunsaturated Fatty Acids and Their Role in Cancer Chemoprevention. Curr. Pharmacol. Rep. 2015, 1, 283–294. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Shepherd, C.J.; Jackson, A.J. Global fishmeal and fish-oil supply: Inputs, outputs and markets. J. Fish Biol. 2013, 83, 1046–1066. [Google Scholar] [CrossRef]
- Blasio, M.; Balzano, S. Fatty Acids Derivatives From Eukaryotic Microalgae, Pathways and Potential Applications. Front. Microbiol. 2021, 12, 718933. [Google Scholar] [CrossRef]
- Garcia, J.L.; de Vicente, M.; Galan, B. Microalgae, old sustainable food and fashion nutraceuticals. Microb. Biotechnol. 2017, 10, 1017–1024. [Google Scholar] [CrossRef] [Green Version]
- Torres-Tiji, Y.; Fields, F.J.; Mayfield, S.P. Microalgae as a future food source. Biotechnol. Adv. 2020, 41, 107536. [Google Scholar] [CrossRef]
- Richardson, J.W.; Johnson, M.D.; Outlaw, J.L. Economic comparison of open pond raceways to photo bio-reactors for profitable production of algae for transportation fuels in the Southwest. Algal Res. 2012, 1, 93–100. [Google Scholar] [CrossRef]
- Shen, Y.; Yuan, W.; Pei, Z.J.; Wu, Q.; Mao, E. Microalgae Mass Production Methods. Trans. ASABE 2009, 52, 1275–1287. [Google Scholar] [CrossRef]
- Li, T.; Chen, Z.; Wu, J.; Wu, H.; Yang, B.; Dai, L.; Wu, H.; Xiang, W. The potential productivity of the microalga, Nannochloropsis oceanica SCS-1981, in a solar powered outdoor open pond as an aquaculture feed. Algal Res. 2020, 46, 101793. [Google Scholar] [CrossRef]
- Salama, E.-S.; Kurade, M.B.; Abou-Shanab, R.A.I.; El-Dalatony, M.M.; Yang, I.-S.; Min, B.; Jeon, B.-H. Recent progress in microalgal biomass production coupled with wastewater treatment for biofuel generation. Renew. Sustain. Energy Rev. 2017, 79, 1189–1211. [Google Scholar] [CrossRef]
- Benvenuti, G.; Ruiz, J.; Lamers, P.P.; Bosma, R.; Wijffels, R.H.; Barbosa, M.J. Towards microalgal triglycerides in the commodity markets. Biotechnol. Biofuels 2017, 10, 188. [Google Scholar] [CrossRef] [PubMed]
- Dineshbabu, G.; Goswami, G.; Kumar, R.; Sinha, A.; Das, D. Microalgae-nutritious, sustainable aqua- and animal feed source. J. Funct. Foods 2019, 62, 103545. [Google Scholar] [CrossRef]
- Draaisma, R.B.; Wijffels, R.H.; Slegers, P.M.; Brentner, L.B.; Roy, A.; Barbosa, M.J. Food commodities from microalgae. Curr. Opin. Biotechnol. 2013, 24, 169–177. [Google Scholar] [CrossRef] [PubMed]
- Bortoleto, G.G.; de Miranda, H.L.; de Campos, R.H. Biodiesel from Microalgae: Third Generation Biofuel. In Plant-Based Genetic Tools for Biodiesel Production; Bentham Science Publishers: Sharja, United Arab Emirates, 2017; pp. 169–198. [Google Scholar]
- Gong, Y.M.; Jiang, M.L. Biodiesel production with microalgae as feedstock: From strains to biodiesel. Biotechnol. Lett. 2011, 33, 1269–1284. [Google Scholar] [CrossRef] [PubMed]
- Rahman, K.M. Food and High Value Products from Microalgae: Market Opportunities and Challenges. In Microalgae Biotechnology for Food, Health and High Value Products; Alam, M.A., Xu, J.-L., Wang, Z., Eds.; Springer: Singapore, 2020; pp. 3–27. [Google Scholar]
- Angulo, L.D.M.; Merida, L.G.R. Current status of microalgae producers companies for food and food supplements in Latin America. Rev. Venez. Cienc. Tecnol. Aliment. 2017, 8, 130–147. [Google Scholar]
- Farag, M.R.; Alagawany, M.; Abd El-Hack, M.E.; Dhama, K. Nutritional and Healthical Aspects of Spirulina (Arthrospira) for Poultry, Animals and Human. Int. J. Pharmacol. 2016, 12, 36–51. [Google Scholar] [CrossRef] [Green Version]
- Wachda; Hadiyanto, H.; Harjanto, G.D.; Huzain, M.L.; Aji, R.W. Iop. Production of antioxidant C-phycocyanin using extraction process of Spirulina platensis in large scale industry. In Proceedings of the 2nd International Conference on Food Science and Engineering (ICFSE), Surakarta, Indonesia, 25–26 September 2018. [Google Scholar]
- Sui, Y.X.; Vlaeminck, S.E. Effects of salinity, pH and growth phase on the protein productivity by Dunaliella salina. J. Chem. Technol. Biotechnol. 2019, 94, 1032–1040. [Google Scholar] [CrossRef]
- Borowitzka, M.A. Commercial production of microalgae: Ponds, tanks, tubes and fermenters. J. Biotechnol. 1999, 70, 313–321. [Google Scholar] [CrossRef]
- Rasheed, R.; Saadaoui, I.; Bounnit, T.; Cherif, M.; Al Ghazal, G.; Al Jabri, H. Sustainable Food Production and Nutraceutical Applications from Qatar Desert Chlorella sp. (Chlorophyceae). Animals 2020, 10, 1413. [Google Scholar] [CrossRef]
- Ratledge, C. Fatty acid biosynthesis in microorganisms being used for Single Cell Oil production. Biochimie 2004, 86, 807–815. [Google Scholar] [CrossRef] [PubMed]
- Oliver, L.; Dietrich, T.; Maranon, I.; Villaran, M.C.; Barrio, R.J. Producing Omega-3 Polyunsaturated Fatty Acids: A Review of Sustainable Sources and Future Trends for the EPA and DHA Market. Resources 2020, 9, 148. [Google Scholar] [CrossRef]
- World Health Organization. Report of the Joint FAO/WHO Expert Consultation on the Risks and Benefits of Fish Consumption. Rome, 25–29 January 2010; World Health Organization: Geneva, Switzerland, 2011.
- Jaspars, M.; De Pascale, D.; Andersen, J.H.; Reyes, F.; Crawford, A.D.; Ianora, A. The marine biodiscovery pipeline and ocean medicines of tomorrow. J. Mar. Biol. Assoc. UK 2016, 96, 151–158. [Google Scholar] [CrossRef] [Green Version]
- Ashour, M.; Elshobary, M.E.; El-Shenody, R.; Kamil, A.W.; Abomohra, A. Evaluation of a native oleaginous marine microalga Nannochloropsis oceanica for dual use in biodiesel production and aquaculture feed. Biomass Bioenergy 2019, 120, 439–447. [Google Scholar] [CrossRef]
- Santin, A.; Russo, M.T.; Ferrante, M.I.; Balzano, S.; Orefice, I.; Sardo, A. Highly Valuable Polyunsaturated Fatty Acids from Microalgae: Strategies to Improve Their Yields and Their Potential Exploitation in Aquaculture. Molecules 2021, 26, 7697. [Google Scholar] [CrossRef]
- Traller, J.C.; Cokus, S.J.; Lopez, D.A.; Gaidarenko, O.; Smith, S.R.; McCrow, J.P.; Gallaher, S.D.; Podell, S.; Thompson, M.; Cook, O.; et al. Genome and methylome of the oleaginous diatom Cyclotella cryptica reveal genetic flexibility toward a high lipid phenotype. Biotechnol. Biofuels 2016, 9, 258. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Richter, D.J.; Berney, C.; Strassert, J.F.H.; Poh, Y.-P.; Herman, E.K.; Muñoz-Gómez, S.A.; Wideman, J.G.; Burki, F.; de Vargas, C. EukProt: A database of genome-scale predicted proteins across the diversity of eukaryotes. BioRxiv 2020, 1–13. [Google Scholar] [CrossRef]
- Keeling, P.J. The Number, Speed, and Impact of Plastid Endosymbioses in Eukaryotic Evolution. Annu. Rev. Plant Biol. 2013, 64, 583–607. [Google Scholar] [CrossRef] [Green Version]
- Johnson, L.K.; Alexander, H.; Brown, C.T. Re-assembly, quality evaluation, and annotation of 678 microbial eukaryotic reference transcriptomes. GigaScience 2018, 8, giy158. [Google Scholar] [CrossRef] [Green Version]
- Sreenikethanam, A.; Raj, S.; Rajesh Banu, J.; Gugulothu, P.; Bajhaiya, A.K. Genetic Engineering of Microalgae for Secondary Metabolite Production: Recent Developments, Challenges, and Future Prospects. Front. Bioeng. Biotechnol. 2022, 10, 836056. [Google Scholar] [CrossRef]
- Davis, D.; Rudenko George, N.; Somanchi, A.; Casolari, J.; Franklin, S.; Ewing, A. Novel Ketoacyl Acp Synthase Genes and Uses Thereof. U.S. Patent 2018/0230442 A1, 16 August 2018. [Google Scholar]
- Jiang, P.; Yang, G.; Jin, Z.; Huaxin, C. Method for Increasing Lipid Content of Diatom Through Metabolism Gene Engineering. CN Patent 105219649 A, 6 January 2016. [Google Scholar]
- Guiheneuf, F.; Leu, S.; Zarka, A.; Khozin-Goldberg, I.; Boussiba, S. Acyl-Coa: Diacylglycerol Acyltransferase 1-Like Gene (Ptdgat1) And Uses Thereof. U.S. Patent 2014/0196177 A1, 10 July 2014. [Google Scholar]
- Deng, X.; Fei, X.; Gu, B.; Luo, Q. Chlamydomonas reinhardtii Lipid Metabolism Gene CrDGAT2-5, Encoding Protein Thereof, and Application Thereof. CN Patent 102321642 A, 18 January 2012. [Google Scholar]
- Xin, Y.; Xu, J. Gene with Triglyceride (TAG) Synthesis Function and Construction Method and Application Thereof. CN Patent 110305883 A, 8 October 2019. [Google Scholar]
- Yang Ji, W.; Kang Nam, K.; Jeong Byeong, R.; Park Min, S.; Lee Bong, S.; Choi Gang, G.; Jeon Seung, J.; Kwon So, H. Method for Increasing Lipid Productivity and Growth of Microalgae by Using an Overexpression of bHLH Transcription Factors. KR Patent 20160142024 A, 12 December 2016. [Google Scholar]
- Huang, Y.; Song, Y.; Hu, Z.; Jia, B.I.N.; Wu, M.I.N.; Lin, B. Chlamydomonas reinhardtii Acyl Coenzyme A Synthetase Anti-sense RNA Expression Vector and Structuring Method and Application Thereof. CN Patent 105647957 A, 8 June 2016. [Google Scholar]
- Ozaki, T. Lipid Production Method Using Acyl-Acp Thioesterase. JP Patent 2016007154 A, 18 January 2016. [Google Scholar]
- Jiang, M.; Xia, W.; Gong, Y.; Liang, Z.; Zhang, Y.; Bing, G.; Zhang, Y. Isochrysis sphaerica Delta 4-Fatty Acid Desaturase Gene and Cloning Method Thereof. CN Patent 102559710 B, 30 October 2013. [Google Scholar]
- Benedetti, M.; Vecchi, V.; Barera, S.; Dall’Osto, L. Biomass from microalgae: The potential of domestication towards sustainable biofactories. Microb. Cell Factories 2018, 17, 173. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hu, Z.; Guo, X.; Chen, Y.; Yin, W. CeDGAT1 (Chlorella ellipsoidea Diacylglycerol Acyltransferase 1) Gene and Application Thereof. CN Patent 103397007 A, 20 November 2013. [Google Scholar]
- Hu, Z.; Fan, C.; Sun, P.; Chen, Y. Chlorella ellipsoidea NF-YA Gene and Application Thereof. CN Patent 107936098 A, 20 April 2018. [Google Scholar]
- Hu, Z.; Fan, C.; Chen, Y. Chlorella ellipsoidea NF-YB Gene and Application Thereof. CN Patent 108003226 A, 8 May 2018. [Google Scholar]
- Hu, Z.; Fan, C.; Chen, Y. Chlorella ellipsoidea NF-YC Gene and Application Thereof. CN Patent 108101973 A, 1 June 2018. [Google Scholar]
- Jeong Won, J.; Ahn Joon, W.; Hwangbo, K.; Park Youn, I.; Liu Jang, R. Method For Increasing Total Fatty Acid Content And Useful Fatty Acid Ratio In Transgenic Microalgae Overexpressing Fab2 Gene. KR Patent 20140005001 A, 14 January 2014. [Google Scholar]
- Yang, J.-W.; Shin, S.-E.; Jeong, B.-R.; Kang Nam, K.; Choi Gang, G.; Jun Seung, J.; Kwak, M.-S. Recombinant Vector For Increasing Biomass And Lipid Productivity Of Microalgae And Use Thereof. WO Patent 2015/105233 A1, 16 July 2015. [Google Scholar]
- Chang Yong, K.; Koh Hyun, G.; Jeong Byeong, R.; Kang Nam, K.; Jeon Seung, J.; Shin Won, S. Recombinant Vector for Targeting Chloroplast of Microalgae and Uses Thereof. KR Patent 101855739 B1, 9 May 2018. [Google Scholar]
- Huang, Y.; Xie, X.; Jia, B.; Hu, Z. Recombinant Expression Vector of Chlamydomonas reinhardtii Dof (DNA Binding with One Finger) Gene as Well as Construction Method and Application of Recombinant Expression Vector. CN Patent 105755034 A, 13 July 2016. [Google Scholar]
- Suetsugu, M.; Ogawa, A. Gene For Improving Fatty Acid Productivity. JP Patent 2017127278 A, 27 July 2017. [Google Scholar]
- Hu, Z.; Zhang, D.; Li, S.; Fan, C.; Chen, Y. Cem GPDH (Glycerol-3-Phosphate Dehydrogenase) Gene and Application Thereof. CN Patent 104357415 A, 18 February 2015. [Google Scholar]
- Trimbur, D.; Im, C.-S.; Dillon, H.; Day, A.; Franklin, S.; Coragliotti, A. Recombinant Microalgal Cell And Method Of Producing Lipids Using Said Cell. EP Patent 3546588 A2, 2 October 2019. [Google Scholar]
- Xu, Y. New Delta12 Desaturase Gene Derived from Pavlova viridis. CN Patent 104388442 A, 4 March 2015. [Google Scholar]
- Xu, Y. Delta 8 Desaturase New Gene Derived from Pavlova viridis. CN Patent 104293769 A, 21 January 2015. [Google Scholar]
- Fan, J.; Li, Y.; Xu, H.U.I.; Run, C.; Fang, L.E.I.; Li, S. Method for Increasing Content of Fatty Acid and Oil and Fat in Microalgae by Using Type III NAD Kinase. CN Patent 105316358 A, 10 February 2016. [Google Scholar]
- Oh, H.M.; Lee, H.G.; Woo, L.J.; Kim, H.S.; Ha, S.J. DGTS Neutral Lipid-Rich Microalgae Transformants through Membrane Lipid DGTS Modulation and Uses Thereof. KR Patent 101893522 B1, 31 August 2018. [Google Scholar]
- Huang, Y.; Yin, J.; Jia, B. High-Oil-Yield Engineering Algae Constructed by Polygene Co-Silencing Technology as Well as Preparation Method and Application of High-Oil-Yield Engineering Algae. CN Patent 110564623 A, 13 December 2019. [Google Scholar]
- Zaki, M.A.; Ashour, M.; Heneash, A.M.M.; Mabrouk, M.M.; Alprol, A.E.; Khairy, H.M.; Nour, A.M.; Mansour, A.T.; Hassanien, H.A.; Gaber, A.; et al. Potential Applications of Native Cyanobacterium Isolate (Arthrospira platensis NIOF17/003) for Biodiesel Production and Utilization of Its Byproduct in Marine Rotifer (Brachionus plicatilis) Production. Sustainability 2021, 13, 1769. [Google Scholar] [CrossRef]
- Morales-Sanchez, D.; Schulze, P.S.C.; Kiron, V.; Wijffels, R.H. Production of carbohydrates, lipids and polyunsaturated fatty acids (PUFA) by the polar marine microalga Chlamydomonas malina RCC2488. Algal Res.-Biomass Biofuels Bioprod. 2020, 50, 102016. [Google Scholar] [CrossRef]
- Hang, L.T.; Mori, K.; Tanaka, Y.; Morikawa, M.; Toyama, T. Enhanced lipid productivity of Chlamydomonas reinhardtii with combination of NaCl and CaCl2 stresses. Bioprocess Biosyst. Eng. 2020, 43, 971–980. [Google Scholar] [CrossRef] [PubMed]
- Work, V.H.; Radakovits, R.; Jinkerson, R.E.; Meuser, J.E.; Elliott, L.G.; Vinyard, D.J.; Laurens, L.M.L.; Dismukes, G.C.; Posewitz, M.C. Increased Lipid Accumulation in the Chlamydomonas reinhardtii sta7-10 Starchless Isoamylase Mutant and Increased Carbohydrate Synthesis in Complemented Strains. Eukaryot. Cell 2010, 9, 1251–1261. [Google Scholar] [CrossRef] [Green Version]
- Nascimento, I.A.; Marques, S.S.I.; Cabanelas, I.T.D.; Pereira, S.A.; Druzian, J.I.; de Souza, C.O.; Vich, D.V.; de Carvalho, G.C.; Nascimento, M.A. Screening Microalgae Strains for Biodiesel Production: Lipid Productivity and Estimation of Fuel Quality Based on Fatty Acids Profiles as Selective Criteria. Bioenergy Res. 2013, 6, 1–13. [Google Scholar] [CrossRef]
- Chen, C.Y.; Kao, A.L.; Tsai, Z.C.; Chow, T.J.; Chang, H.Y.; Zhao, X.Q.; Chen, P.T.; Su, H.Y.; Chang, J.S. Expression of type 2 diacylglycerol acyltransferse gene DGTT1 from Chlamydomonas reinhardtii enhances lipid production in Scenedesmus obliquus. Biotechnol. J. 2016, 11, 336–344. [Google Scholar] [CrossRef]
- Mehta, P.; Rani, R.; Gupta, R.; Mathur, A.S.; Puri, S.K. Biomass and lipid production of a novel freshwater thermo-tolerant mutant strain of Chlorella pyrenoidosa NCIM 2738 in seawater salinity recycled medium. Algal Res.-Biomass Biofuels Bioprod. 2018, 36, 88–95. [Google Scholar] [CrossRef]
- Zhang, X.; Rung, J.F.; Chen, H.; He, C.L.; Wang, Q. Current status and outlook in the application of microalgae in biodiesel production and environmental protection. Front. Energy Res. 2014, 2, 32. [Google Scholar] [CrossRef] [Green Version]
- Xue, J.; Wang, L.; Zhang, L.; Balamurugan, S.; Li, D.W.; Zeng, H.; Yang, W.D.; Liu, J.S.; Li, H.Y. The pivotal role of malic enzyme in enhancing oil accumulation in green microalga Chlorella pyrenoidosa. Microb. Cell Factories 2016, 15, 120. [Google Scholar] [CrossRef] [Green Version]
- Mata, T.M.; Martins, A.A.; Caetano, N.S. Microalgae for biodiesel production and other applications: A review. Renew. Sustain. Energy Rev. 2010, 14, 217–232. [Google Scholar] [CrossRef] [Green Version]
- Yuan, Y.; Xuyang, L.; Quanyu, Z. Enhancing growth and lipid productivity in Dunaliella salina under high light intensity and nitrogen limited conditions. Bioresour. Technol. Rep. 2019, 7, 100211. [Google Scholar] [CrossRef]
- Talebi, A.F.; Tohidfar, M.; Bagheri, A.; Lyon, S.R.; Salehi-Ashtiani, K.; Tabatabaei, M. Manipulation of carbon flux into fatty acid biosynthesis pathway in Dunaliella salina using AccD and ME genes to enhance lipid content and to improve produced biodiesel quality. Biofuel Res. J. 2014, 1, 91–97. [Google Scholar] [CrossRef]
- Guerra, I.; Pereira, H.; Costa, M.; Silva, J.T.; Santos, T.; Varela, J.; Mateus, M.; Silva, J. Operation Regimes: A Comparison Based on Nannochloropsis oceanica Biomass and Lipid Productivity. Energies 2021, 14, 1542. [Google Scholar] [CrossRef]
- Han, X.; Song, X.; Li, F.; Lu, Y. Improving lipid productivity by engineering a control-knob gene in the oleaginous microalga Nannochloropsis oceanica. Metab. Eng. Commun. 2020, 11, e00142. [Google Scholar] [CrossRef]
- Patel, A.; Matsakas, L.; Hrůzová, K.; Rova, U.; Christakopoulos, P. Biosynthesis of Nutraceutical Fatty Acids by the Oleaginous Marine Microalgae Phaeodactylum tricornutum Utilizing Hydrolysates from Organosolv-Pretreated Birch and Spruce Biomass. Mar. Drugs 2019, 17, 119. [Google Scholar] [CrossRef] [Green Version]
- Daboussi, F.; Leduc, S.; Marechal, A.; Dubois, G.; Guyot, V.; Perez-Michaut, C.; Amato, A.; Falciatore, A.; Juillerat, A.; Beurdeley, M.; et al. Genome engineering empowers the diatom Phaeodactylum tricornutum for biotechnology. Nat. Commun. 2014, 5, 3831. [Google Scholar] [CrossRef]
- Byreddy, A.R.; Gupta, A.; Barrow, C.J.; Puri, M. Comparison of Cell Disruption Methods for Improving Lipid Extraction from Thraustochytrid Strains. Mar. Drugs 2015, 13, 5111–5127. [Google Scholar] [CrossRef] [Green Version]
- Li, Z.; Meng, T.; Ling, X.; Li, J.; Zheng, C.; Shi, Y.; Chen, Z.; Li, Z.; Li, Q.; Lu, Y.; et al. Overexpression of Malonyl-CoA: ACP Transacylase in Schizochytrium sp. to Improve Polyunsaturated Fatty Acid Production. J. Agric. Food Chem. 2018, 66, 5382–5391. [Google Scholar] [CrossRef]
- da Silva, T.L.; Moniz, P.; Silva, C.; Reis, A. The Dark Side of Microalgae Biotechnology: A Heterotrophic Biorefinery Platform Directed to omega-3 Rich Lipid Production. Microorganisms 2019, 7, 670. [Google Scholar] [CrossRef] [Green Version]
- Kreft, J.; Moe, E.; Garcia, N.; Ross, A.; Seames, W. Comparative scoping study report for the extraction of microalgae oils from two subspecies of Chlorella vulgaris. Clean Energy 2020, 4, 233–246. [Google Scholar] [CrossRef]
- Singh, G.; Patidar, S.K. Microalgae harvesting techniques: A review. J. Environ. Manag. 2018, 217, 499–508. [Google Scholar] [CrossRef] [PubMed]
- Grima, E.M.; Belarbi, E.H.; Fernandez, F.G.A.; Medina, A.R.; Chisti, Y. Recovery of microalgal biomass and metabolites: Process options and economics. Biotechnol. Adv. 2003, 20, 491–515. [Google Scholar] [CrossRef]
- Verma, N.M.; Mehrotra, S.; Shukla, A.; Mishra, B.N. Prospective of biodiesel production utilizing microalgae as the cell factories: A comprehensive discussion. Afr. J. Biotechnol. 2010, 9, 1402–1411. [Google Scholar]
- Soto-Sierra, L.; Stoykova, P.; Nikolov, Z.L. Extraction and fractionation of microalgae-based protein products. Algal Res.-Biomass Biofuels Bioprod. 2018, 36, 175–192. [Google Scholar] [CrossRef]
- Ahmad, N.; Mehmood, M.A.; Malik, S. Recombinant Protein Production in Microalgae: Emerging Trends. Protein Pept. Lett. 2020, 27, 105–110. [Google Scholar] [CrossRef]
- de Carvalho Silvello, M.A.; Severo Gonçalves, I.; Patrícia Held Azambuja, S.; Silva Costa, S.; Garcia Pereira Silva, P.; Oliveira Santos, L.; Goldbeck, R. Microalgae-based carbohydrates: A green innovative source of bioenergy. Bioresour Technol. 2022, 344, 126304. [Google Scholar] [CrossRef]
- Gouda, M.; Tadda, M.A.; Zhao, Y.; Farmanullah, F.; Chu, B.; Li, X.; He, Y. Microalgae Bioactive Carbohydrates as a Novel Sustainable and Eco-Friendly Source of Prebiotics: Emerging Health Functionality and Recent Technologies for Extraction and Detection. Front. Nutr. 2022, 9, 806692. [Google Scholar] [CrossRef]
- Sardo, A.; Orefice, I.; Balzano, S.; Barra, L.; Romano, G. Mini-Review: Potential of Diatom-Derived Silica for Biomedical Applications. Appl. Sci. 2021, 11, 4533. [Google Scholar] [CrossRef]
- Ruggiero, I.; Terracciano, M.; Martucci, N.M.; De Stefano, L.; Migliaccio, N.; Tatè, R.; Rendina, I.; Arcari, P.; Lamberti, A.; Rea, I. Diatomite silica nanoparticles for drug delivery. Nanoscale Res. Lett. 2014, 9, 329. [Google Scholar] [CrossRef] [Green Version]
- Dalgıç, A.; Atila, D.; Karataş, A.Y.; Tezcaner, A.; Keskin, D. Diatom shell incorporated PHBV/PCL-pullulan co-electrospun scaffold for bone tissue engineering. Mater. Sci. Eng. C Mater. Biol. Appl. 2019, 100, 735–746. [Google Scholar] [CrossRef] [PubMed]
- Abdelhamid, M.A.A.; Pack, S.P. Biomimetic and bioinspired silicifications: Recent advances for biomaterial design and applications. Acta Biomater. 2021, 120, 38–56. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.Y.; Khoiroh, I.; Vo, D.-V.N.; Senthil Kumar, P.; Show, P.L. Techniques of lipid extraction from microalgae for biofuel production: A review. Environ. Chem. Lett. 2021, 19, 231–251. [Google Scholar] [CrossRef]
- Lee, J.-Y.; Yoo, C.; Jun, S.-Y.; Ahn, C.-Y.; Oh, H.-M. Comparison of several methods for effective lipid extraction from microalgae. Bioresour. Technol. 2010, 101, S75–S77. [Google Scholar] [CrossRef]
- Rokicka, M.; Zieliński, M.; Dudek, M.; Dębowski, M. Effects of Ultrasonic and Microwave Pretreatment on Lipid Extraction of Microalgae and Methane Production from the Residual Extracted Biomass. BioEnergy Res. 2021, 14, 752–760. [Google Scholar] [CrossRef]
- Rahman, M.M.; Hosano, N.; Hosano, H. Recovering Microalgal Bioresources: A Review of Cell Disruption Methods and Extraction Technologies. Molecules 2022, 27, 2786. [Google Scholar] [CrossRef]
- Michalak, I.; Dmytryk, A.; Wieczorek, P.P.; Rój, E.; Łęska, B.; Górka, B.; Messyasz, B.; Lipok, J.; Mikulewicz, M.; Wilk, R.; et al. Supercritical Algal Extracts: A Source of Biologically Active Compounds from Nature. J. Chem. 2015, 2015, 597140. [Google Scholar] [CrossRef]
- Molino, A.; Mehariya, S.; Di Sanzo, G.; Larocca, V.; Martino, M.; Leone, G.P.; Marino, T.; Chianese, S.; Balducchi, R.; Musmarra, D. Recent developments in supercritical fluid extraction of bioactive compounds from microalgae: Role of key parameters, technological achievements and challenges. J. CO2 Util. 2020, 36, 196–209. [Google Scholar] [CrossRef]
- Daintree, L.S.; Kordikowski, A.; York, P. Separation processes for organic molecules using SCF technologies. Adv. Drug Deliv. Rev. 2008, 60, 351–372. [Google Scholar] [CrossRef]
- Spicer, A.; Molnar, A. Gene Editing of Microalgae: Scientific Progress and Regulatory Challenges in Europe. Biology 2018, 7, 21. [Google Scholar] [CrossRef] [Green Version]
- EU 1829/2003/EC; E.U. Regulation (EC) No. 1829/2003 of the European Parliament and of the Council on Genetically Modified Food and Feed. Official Journal L 268; European Union: Brussels, Belgium, 18 October 2003; pp. 1–23.
- EU 1830/2003/EC; E.U. Regulation (EC) No. 1830/2003 of the European Parliament and of the Council Concerning the Traceability and Labelling of Genetically Modified Organisms and the Traceability of Food and FEED products Produced from Genetically Modified Organisms and Amending Directive 2001/18/EC. Official Journal L 268; European Union: Brussels, Belgium, 18 October 2003; pp. 24–28.
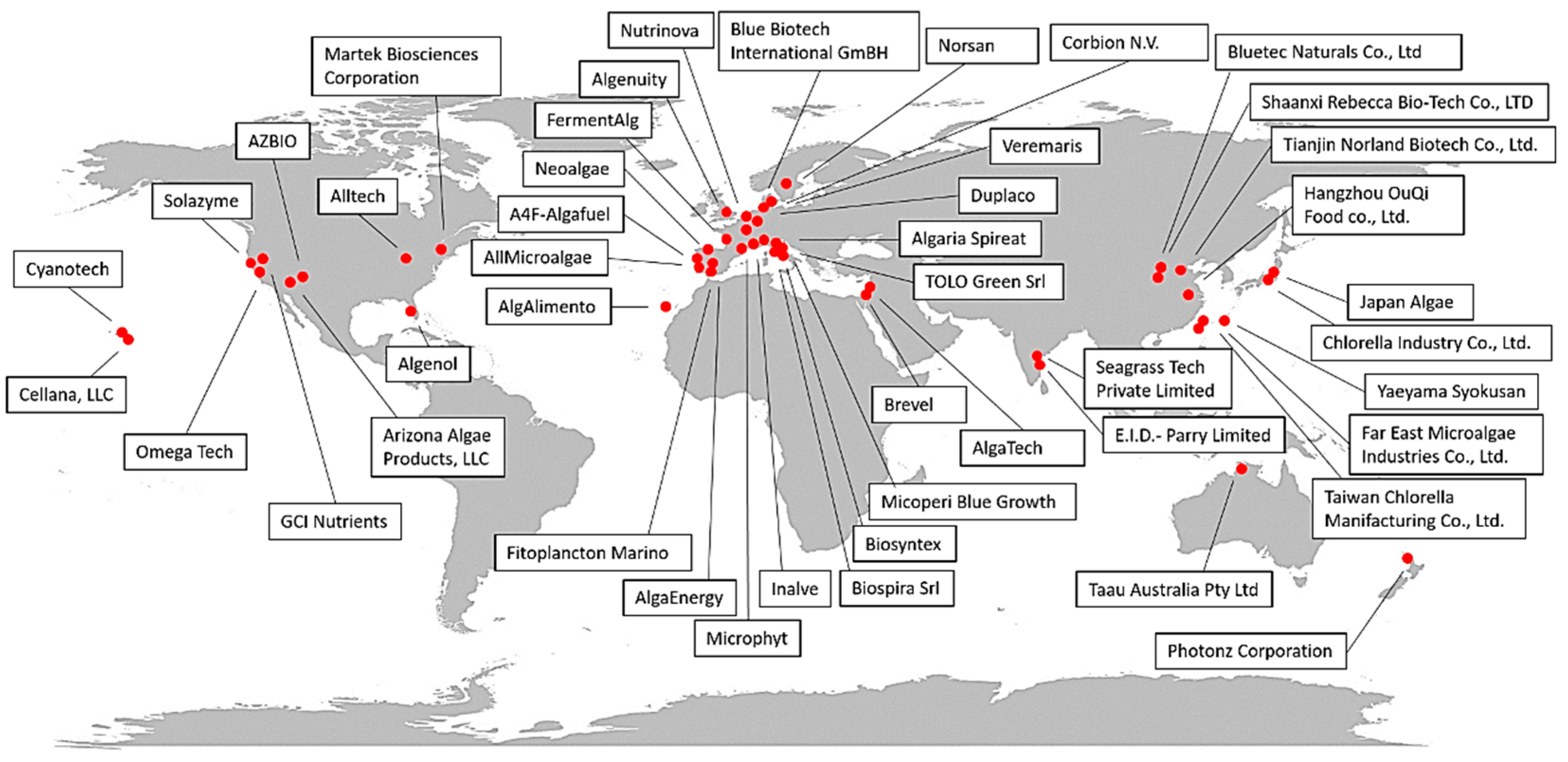
| Company | Country | Species/Category | Product(s) | Sector | Type of Plant/Growth Condition | Website or Reference(s) |
|---|---|---|---|---|---|---|
| Cyanotech | Hawaii | Arthrospira sp. (Spirulina) | Hawaiian BioAstin Hawaiian spirulina | Dietary supplements for human consumption | Open ponds | https://www.cyanotech.com/our-purpose/ |
| Cellana, LLC | Hawaii | Marine microalgae, Staurosira sp. | ReNew™ (ω-3 rich oils) | Human and animal food Whole algae enriched with EPA and DHA (as animal feed) | Open ponds | http://cellana.com/ |
| Alltech | United States | Schizochytrium sp. | All-G-RichTM | Dehydrated whole algae for poultry nutrition rich in DHA, biofuels | n.f. | https://www.alltech.com/ |
| TerraVia Holdings, Inc. (formerly Solazyme) | United States | Chlorella sp. | Golden Chlorella, AlgaVia (algal powder, food ingredient line recognized as GRAS), AlgaWise (food oils), AlgaPrime DHA (aquaculture) | Dietary supplements (high-value oils and whole-algae ingredients) for human and animal (aquaculture) consumption | Heterotrophic growth in stainless-steel containers | https://www.solazyme.com/ |
| Algenol | United States | Arthrospira sp. (spirulina) (other strains available) | Whole algae or protein isolates, phycocyanin | Food and food colorants | Open ponds | https://www.algenol.com/ |
| Omega Tech | United States | Schizochytrium sp. | DHA Gold (oil) | Food supplements | n.f. | [25] |
| Martek Biosciences Corporation | United States | Crypthecodinium cohnii | ω-3, ω-6, ARA | Food, beverages, dietary supplements, and early-life nutrition | n.f. | https://www.dsm.com/corporate/home.html https://www.linkedin.com/company/martek-biosciences/ |
| GCI Nutrients | United States | Chlorella, Arthrospira sp. (Spirulina) | DHA3Sure™ DHA Algae 35% oil complex, organic Chlorella, organic broken-cell Chlorella, | Nutrients | n.f. | https://gcinutrients.com/ |
| AZBIO | United States | Marine microalgae | AlgaeBio Omega-3 Origins™ | EPA and DHA for food and feed | Autotrophic growth | https://www.azbio.org/tag/algae-biosciences |
| Arizona Algae Products, LLC | United States | Marine microalgae | Protein+omega3 powder, EPA extract | Food, dietary supplements, and wellness products | Photobioreactors | https://www.azalgae.com/ |
| Taau Australia Pty Ltd. | Australia | Arthrospira sp. (spirulina) | Tabs and powder | Human consumption | Open ponds | https://www.taau.com.au/company.html |
| Photonz Corporation | New Zealand | Marine microalgae | EPA | Aquaculture (fish oil replacement purposes), pharmaceuticals | Fermentation processes | https://pureadvantage.org/photonz-corporation/ |
| Blue Biotech International GmBH | Germany | Nannochloropsis, Haematococcus | Algal concentrate, frozen paste, freeze-dried cells, phycocyanin, astaxanthin, microalgae powder | Hatchery, feed | Photobioreactors | https://www.bluebiotech.de/com/index.html |
| Nutrinova | Germany | Ulkenia sp. | DHActiveTM | Aquaculture | Fermentation processes (80 m3) | [25] |
| Corbion N.V. | Netherlands | Schizochytrium sp. | AlgaPrimeTM | Aquaculture, pet, and livestock industries | Fermentation processes | http://www.corbion.com |
| Veramaris | Netherlands | Marine microalgae | Veramaris® Pets (algal oil rich in EPA and DHA; GRAS product) | Dog food | n.f. | https://pets.veramaris.com/ |
| Duplaco | Netherlands | Chlorella | Powder, food ingredients, and supplements | Food | Photobioreactors and fermenters | https://duplaco.com/ |
| Neoalgae | Spain | Spirulina, Chlorella | Powders and capsules | Food | Photobioreactors | https://neoalgae.es/?lang=en |
| AlgaEnergy | Spain | Arthrospira sp. (spirulina) | Dietary supplements, feed for aquaculture, biostimulants for agriculture | Food and feed | Outdoor open ponds and outdoor photobioreactors | https://www.algaenergy.it/ |
| AlgAlimento | Spain | Tetraselmis sp., Dunaliella salina, Arthrospira sp. | Powder for antioxidant food supplements and cosmetics | Food | Outdoor open ponds | http://www.algalimento.com/ |
| Fitoplancton marino | Spain | Tetraselmis chuii | Cosmetics, food supplements, feed as Easyreefs® and Easyalgae® | Food, feed, and cosmetics | Open ponds and photobioreactors | http://www.fitoplanctonmarino.com/index.html |
| Microphyt | France | Arthrospira sp. (spirulina), Haematococcus pluvialis | Powders and pastes, dietary supplements | Food, feed, and cosmetics | Photobioreactors (indoor and outdoor systems) | http://www.microphyt.eu/ |
| Inalve | France | Biofilm-forming microalgae | Powders and pastes for food and feed supplements (e.g., FEALTM) | Food and feed | Photobioreactors | https://www.inalve.com/ |
| FermentAlg | France | Schizochytrium sp. | Powder and pastes for food supplements (e.g., DHA-ORIGIN TM) | Food | Fermentation process | https://www.fermentalg.com/ |
| Algenuity | United Kingdom | Chlorella vulgaris | Food ingredients | Food | Photobioreactors | https://www.algenuity.com/ |
| AllMicroalgae | Portugal | Chlorella vulgaris, Tetraselmis chui, Nannochloropsis oceanica, Scenedesmus obliquus | Powders and pastes | Food and feed | Photobioreactors (outdoor systems) | https://www.allmicroalgae.com/en/ |
| A4F-Algafuel | Portugal | Dunaliella salina (pilot), Lobosphaera incisa, Prorocentrum casubicum, Raphidonema sp., | Food colorants, ω-6 food additives for children, and dietary supplements | Food and feed | Open ponds and photobioreactors | https://a4f.pt/pt |
| Norsan | Norway, Germany | Schizochytrium sp. | ω-3 oil | Food supplements | n.f. | https://www.norsan-omega.com/ |
| Algaria Spireat | Italy | Arthrospira sp. (spirulina) | Snack and dietary supplements | Food supplements | n.f. | https://spireat.it/ |
| Biospira Srl | Italy | Arthrospira sp. (spirulina) | Capsules, powders, flakes | Food supplements | Monitored tanks that isolate the algae from the external environment | https://www.biospira.it/ |
| Biosyntex | Italy | Arthrospira sp. (spirulina), Haematococcus pluvialis | Food ingredients, beverages, nutraceuticals, cosmetics, feed for aquaculture, and biostimulants for agriculture | Mainly food, feed, and biostimulants | Indoor and outdoor photobioreactors | www.biosyntex.com/ |
| Micoperi Blue Growth | Italy | Phaeodactylum tricornutum, Arthrospira platensis, Euglena gracilis | Capsules, powders | Food supplements | Photobioreactors and indoor ponds | http://www.micoperibg.eu/?page_id=101 |
| TOLO Green Srl | Italy | Arthrospira sp. (spirulina) | Food and feed supplements, biostimulants | Food, feed, and agriculture | Open ponds and monitored tanks isolated from the external environment | https://www.tologreen.it/ |
| Alghitaly | Italy | Arthrospira sp. (spirulina) | Powders and pastes | Food and feed supplements | Outdoor photobioreactors | https://www.alghitaly.it/ |
| Brevel | Israel | n.f. | Natural salmon feed | Feed | Indoor photobioreactors | https://brevel.co.il/ |
| Algatech | Israel | Nannochloropsis sp., Porphyridium cruentum | Food supplements | Food | Outdoor photobioreactors | https://www.algatech.com/ |
| Chlorella Industry Co., Ltd. | Japan | Chlorella | Tablets, extracts for food ingredients | Food and feed | Open ponds | https://www.chlorella.co.jp/ |
| Yaeyama Syokusan | Japan | Chlorella | Powder, tablets, food, and feed supplements | Food and feed | Open ponds | https://www.yaeyamachlorella.com/ |
| Japan Algae | Japan | Arthrospira sp. (spirulina) | Powder and tablets for food ingredients and supplements | Food | Open ponds | http://www.sp100.com/ |
| Taiwan Chlorella Manifacturing Co., Ltd. | Taiwan | Chlorella sorokiniana | Tablets and food supplements | Food | Open ponds | http://www.taiwanchlorella.com/ |
| Far East Microalgae Industries Co., Ltd. | Taiwan | Chlorella, Arthrospira sp. (spirulina) | Powder, dietary supplements, skin care systems, aquaculture feeds | Food, feed, and cosmetics | Open ponds | http://www.femico.com.tw/ |
| Bluetec Naturals Co., Ltd. | China | Chlorella, Arthrospira sp. (spirulina) | DHA-rich powder and oil | Food supplement | Open ponds monitored and isolated from the external environment | https://www.bestphycocyanin.com/ |
| Tianjin Norland Biotech Co., Ltd. | China | Chlorella, Spirulina, Haematococcus pluvialis | Powder, oils, and tablets | Food | Open ponds | http://www.norlandbiotech.com/ |
| Hangzhou OuQi Food co., Ltd. | China | Spirulina, Chlorella, Dunaliella salina | Organic Spirulina, organic Chlorella, broken Chlorella, organic Dunaliella | Food ingredients and supplements | Indoor and outdoor systems | http://www.onlygreen.cn/webEn/LM_about%20us |
| Shaanxi Rebecca Bio-Tech Co., LTD | China | Dunaliella salina, Haematococcus pluvialis | Powder, extracts | Natural food | Fermentation process | http://it.rebeccabio.com/ |
| Seagrass Tech Private Limited | India | Dunaliella salina, Chlorella salina, Arthrospira subsalsa | SeaCarotene®, SeaLipro®, SeaProtein® | Food | n.f. | https://seagrasstech.com/company-overview/ |
| E.I.D.-Parry Limited | India | Arthrospira sp. (spirulina) | Organic Chlorella, spirulina, Phycocyanin, natural astaxanthin | Nutraceuticals | n.f. | https://www.eidparry.com/ |
| Transformation | Gene | Species | Results | Patent | Submission Date | Reference |
|---|---|---|---|---|---|---|
| Overexpression | KASI-KASIV-FATA-FATB | Chlorella or Prototheca spp. | +C8-C16 FAs | US 20180230442 A1 | July 2014 | [36] |
| Overexpression | FAB2 | C. reinhardtii | +FAs | KR20140005001 A | July 2012 | [50] |
| Overexpression | GPAT | P. tricornutum | +TAGs | CN105219649 A | October 2015 | [37] |
| Overexpression | DGAT1 | P. tricornutum | +TAGs | US2014196177 A1 | November 2010 | [38] |
| Overexpression | DGAT | N. oceanica | +TAGs, +PUFAs | CN110305883 A | March 2018 | [40] |
| Overexpression | DGAT2-5 | C. reinhardtii | +biomass, +neutral lipids | CN102321642 A | September 2011 | [39] |
| Overexpression | GAPDH | microalgae | +biomass, +lipids | WO2015105233 A1 | January 2014 | [51] |
| Overexpression | CAO | C. reinhardtii | +biomass, +lipids | KR101855739 B1 | June 2017 | [52] |
| Overexpression | bHLH2 | N. salina | +lipids | KR20160142024 A | June 2015 | [41] |
| Overexpression | DOF | C. reinhardtii | +lipids | CN105755034 A | March 2016 | [53] |
| Heterologous expression | 8 nucleotide sequences | from C. bastropiensis to bacteria and mi-croalgae | +FAs | JP2017127278 A | January 2016 | [54] |
| Heterologous expression | Acyl-ACP TE | from E. siliculosus to bacteria and microalgae | +lipids | JP2016007154 A | June 2014 | [43] |
| Heterologous expression | DGAT1 | from C. ellipsoidea to yeasts, plants or microalgae | +FAs | CN103397007 A | July 2013 | [46] |
| Heterologous expression | GPDH | from C. ellipsoidea to yeasts, plants or microalgae | +FAs | CN104357415 B | October 2014 | [55] |
| Heterologous expression | Sucrose invertase | bacteria, yeasts and microalgae | +lipids | EP3546588 A3 | June 2007 | [56] |
| Heterologous expression | Δ12-DES | from P. viridis | +LA, +EPA | CN104388442 A | November 2014 | [57] |
| Heterologous expression | Δ8-DES | from P. viridis | +DHA, +EPA | CN104293769 A | October 2014 | [58] |
| Heterologous expression | Δ4-DES | from E. sphaerica to yeast P. pastoris | function confirmed | CN102559710 B | June 2011 | [44] |
| Heterologous expression | NADK3 | from plant A. thaliana to C. pyrenoidesa | +FAs | CN105316358 A | October 2015 | [59] |
| Heterologous expression | NF-YA | from C. ellipsoidea to plant A. thaliana | +44.9% weight, +22.4% FAs | CN107936098 A | December 2017 | [47] |
| Heterologous expression | NF-YB | from C. ellipsoidea to plant A. thaliana | +51% weight, +11.2% FAs | CN108003226 A | December 2017 | [48] |
| Heterologous expression | NF-YC | from C. ellipsoidea to plant A. thaliana | +44.9% weight, +15.4% FAs | CN108101973 A | December 2017 | [49] |
| Silencing | ACS1-ACS2 | C. reinhardtii | +fatty acid excretion | CN105647957 A | January 2016 | [42] |
| Silencing | BTA1 | Chlamydomonas sp. | −glycolipids, 2.5-3X TAGs | KR101893522 B1 | April 2017 | [60] |
| Polygene co-silencing | Carbon metabolism genes | C. reinhardtii | +biomass, +lipids | CN110564623 A | September 2019 | [61] |
| Species | WT Biomass | WT Lipid Productivity | Reference | Number of GMO Strains | GMO Lipid Yields | Reference |
|---|---|---|---|---|---|---|
| Arthrospira platensis | 0.14 g/L/day | 14.37 mg/L/day | [62] | - | - | |
| Chlamydomonas malina and Chlamydomonas reinhardtii | 0.53 g/L/day (C. malina); 0.014 g/L/day (C. reinhardtii) | Total lipid 161.3 (C. malina); 10.9 mg/L/day (C. reinhardtii) PUFAs 85.4 mg/L/day (C. malina) | [63,64] | 26 (of which 4 are patented) | Total lipids ~3.2X more than C. reinhardtii WT (~118.3 μg/mL culture) | [65] |
| Scenedesmus obliquus | 0.16 g/L/day | 26.77 mg/L/day | [66] | 4 | Biomass ~17% higher than S. obliquus WT (~234.3 mg/L/day) and lipid productivity ~2.2X more than WT (~42.4 mg/L/day) | [67] |
| Chlorella vulgaris Chlorella pyrenoidosa | 0.73 mg/L/day (C. vulgaris) and 0.34 g/L/day (C. pyrenoidosa) | 204.91 mg/L/day (C. vulgaris) and 66 mg/L/day (C. pyrenoidosa) | [68,69] | 6 (of which 2 are patented) | Neutral lipid content ~3.2X and total PUFAs > 34% higher than C. pyrenoidosa WT | [70] |
| Tetraselmis sp. | 0.30 g/L/day | 43.4 mg/L/day | [71] | - | - | |
| Dunaliella salina | 1.03 g/L/day | 0.24 mg/L/day | [72] | 3 | Oil content ~13% higher than D. salina WT (~25%) | [73] |
| Nannochloropsis oceanica | 0.427 g/L/day | 39.6 mg/L/day | [74] | 11 (of which 2 are patented) | Lipid production 110.6% higher than N. oceanica WT (~1.15 g/L) and TAG 148.6% higher than WT (~0.80 g/L) | [75] |
| Phaeodactylum tricornutum | 0.254 g/L/day | 99.23 mg/L/day | [76] | 35 (of which 2 are patented) | TAG ~45X more than P. tricornutum WT | [77] |
| Schizochytrium sp. S31 | 0.81 g/L/day | 100.74 mg/L/day | [78] | 3 | Total lipid yield ~39.6% higher than Schizochytrium sp. WT (~110.5 g/L) | [79] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Santin, A.; Balzano, S.; Russo, M.T.; Palma Esposito, F.; Ferrante, M.I.; Blasio, M.; Cavalletti, E.; Sardo, A. Microalgae-Based PUFAs for Food and Feed: Current Applications, Future Possibilities, and Constraints. J. Mar. Sci. Eng. 2022, 10, 844. https://doi.org/10.3390/jmse10070844
Santin A, Balzano S, Russo MT, Palma Esposito F, Ferrante MI, Blasio M, Cavalletti E, Sardo A. Microalgae-Based PUFAs for Food and Feed: Current Applications, Future Possibilities, and Constraints. Journal of Marine Science and Engineering. 2022; 10(7):844. https://doi.org/10.3390/jmse10070844
Chicago/Turabian StyleSantin, Anna, Sergio Balzano, Monia Teresa Russo, Fortunato Palma Esposito, Maria Immacolata Ferrante, Martina Blasio, Elena Cavalletti, and Angela Sardo. 2022. "Microalgae-Based PUFAs for Food and Feed: Current Applications, Future Possibilities, and Constraints" Journal of Marine Science and Engineering 10, no. 7: 844. https://doi.org/10.3390/jmse10070844
APA StyleSantin, A., Balzano, S., Russo, M. T., Palma Esposito, F., Ferrante, M. I., Blasio, M., Cavalletti, E., & Sardo, A. (2022). Microalgae-Based PUFAs for Food and Feed: Current Applications, Future Possibilities, and Constraints. Journal of Marine Science and Engineering, 10(7), 844. https://doi.org/10.3390/jmse10070844








