Weed Management Programs in Grain Sorghum (Sorghum bicolor)
Abstract
:1. Introduction
2. Materials and Methods
3. Results
3.1. Weed Control
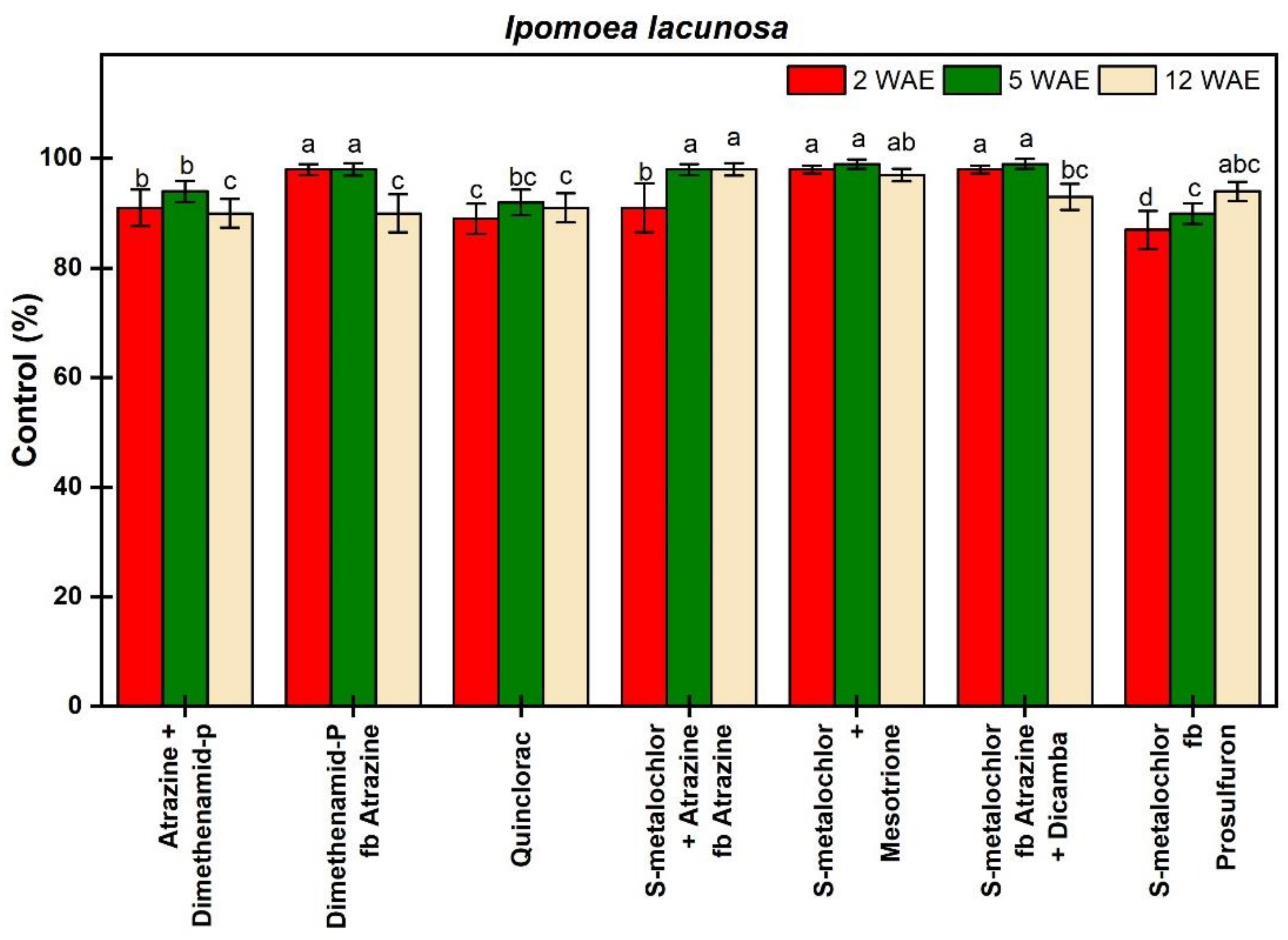
3.1.1. Ipomoea lacunosa
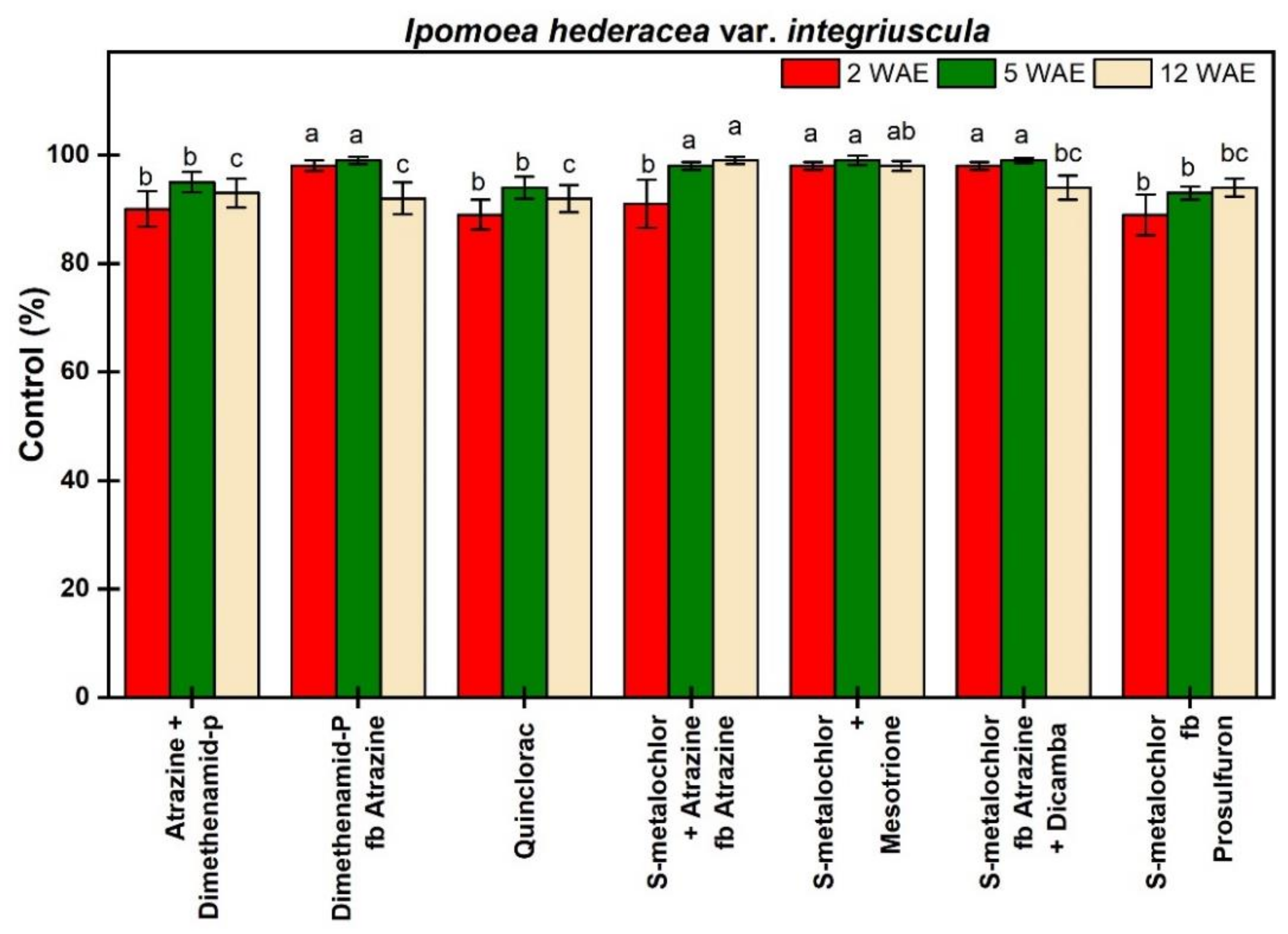
3.1.2. Ipomoea hederacea var. integriuscula
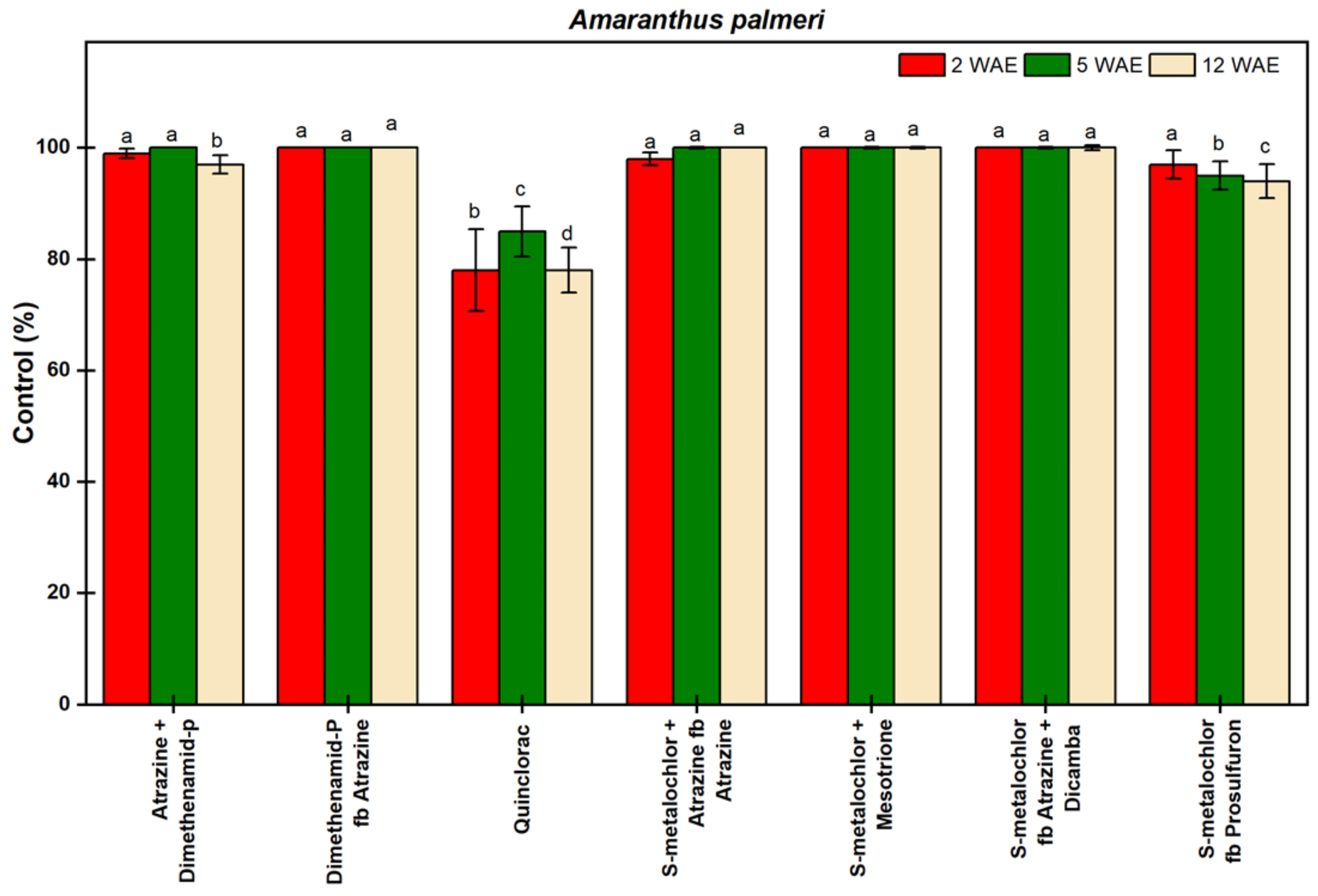
3.1.3. Amaranthus palmeri
3.1.4. Sesbania exaltata
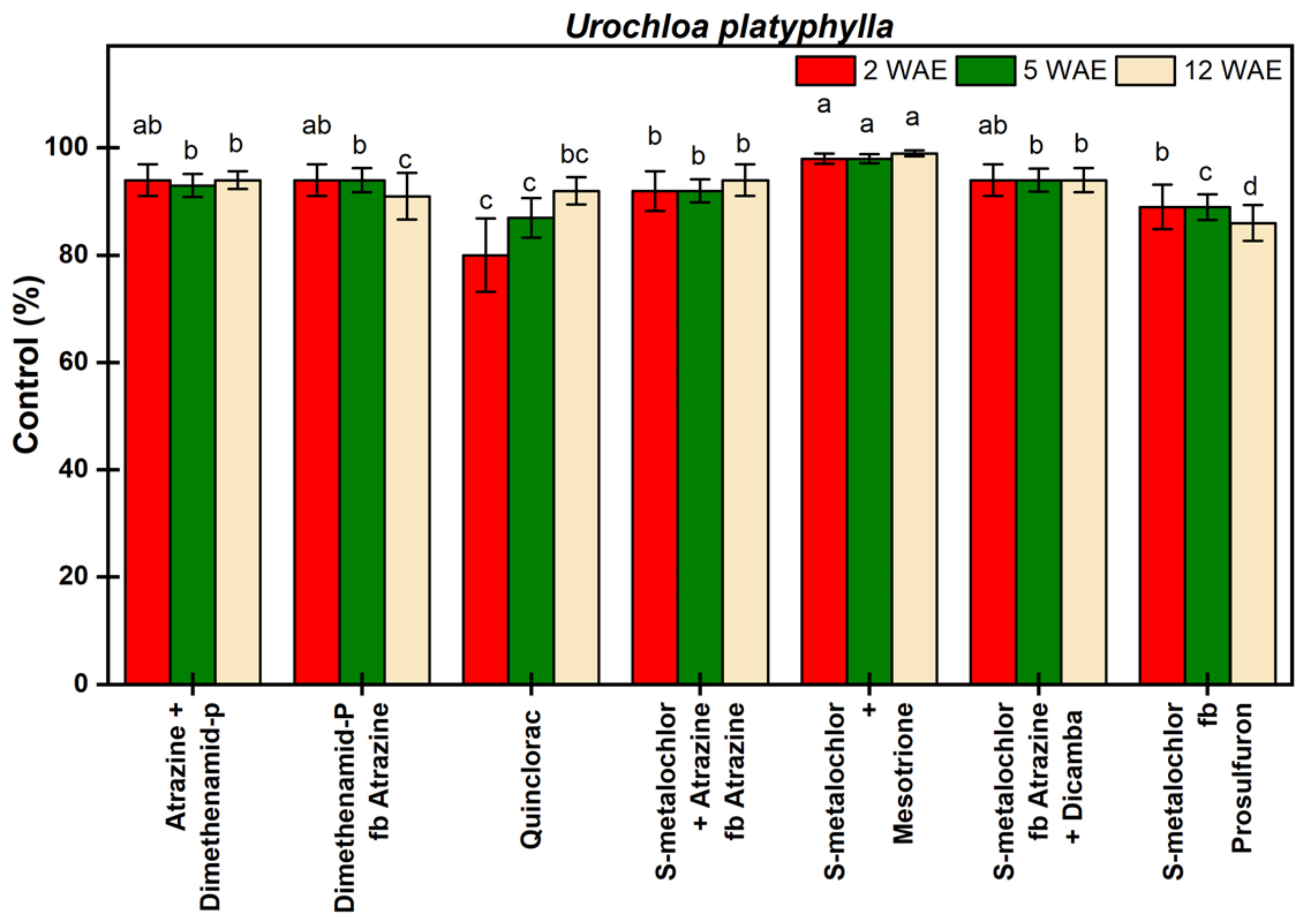
3.1.5. Urochloa platyphylla
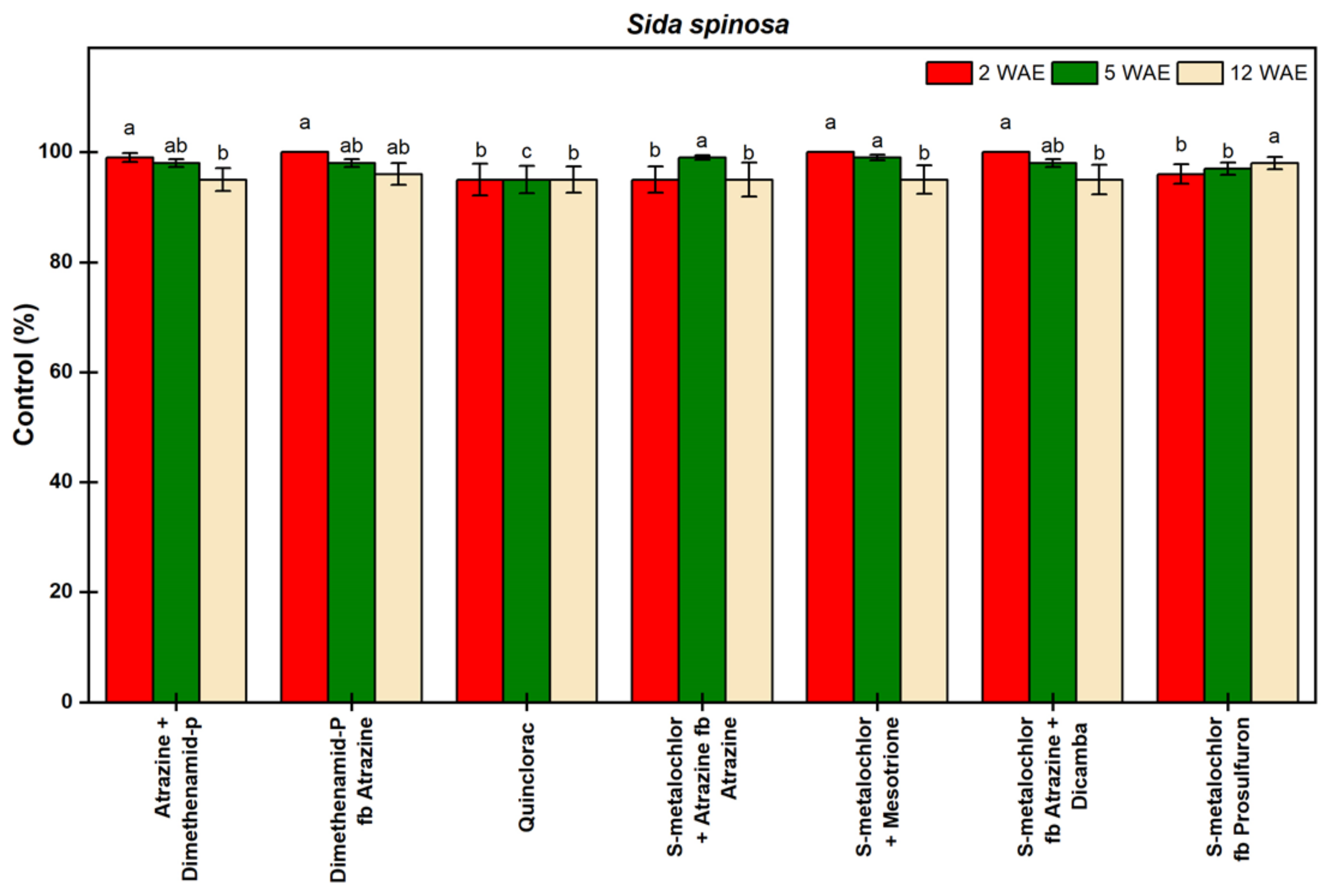
3.1.6. Sida spinosa
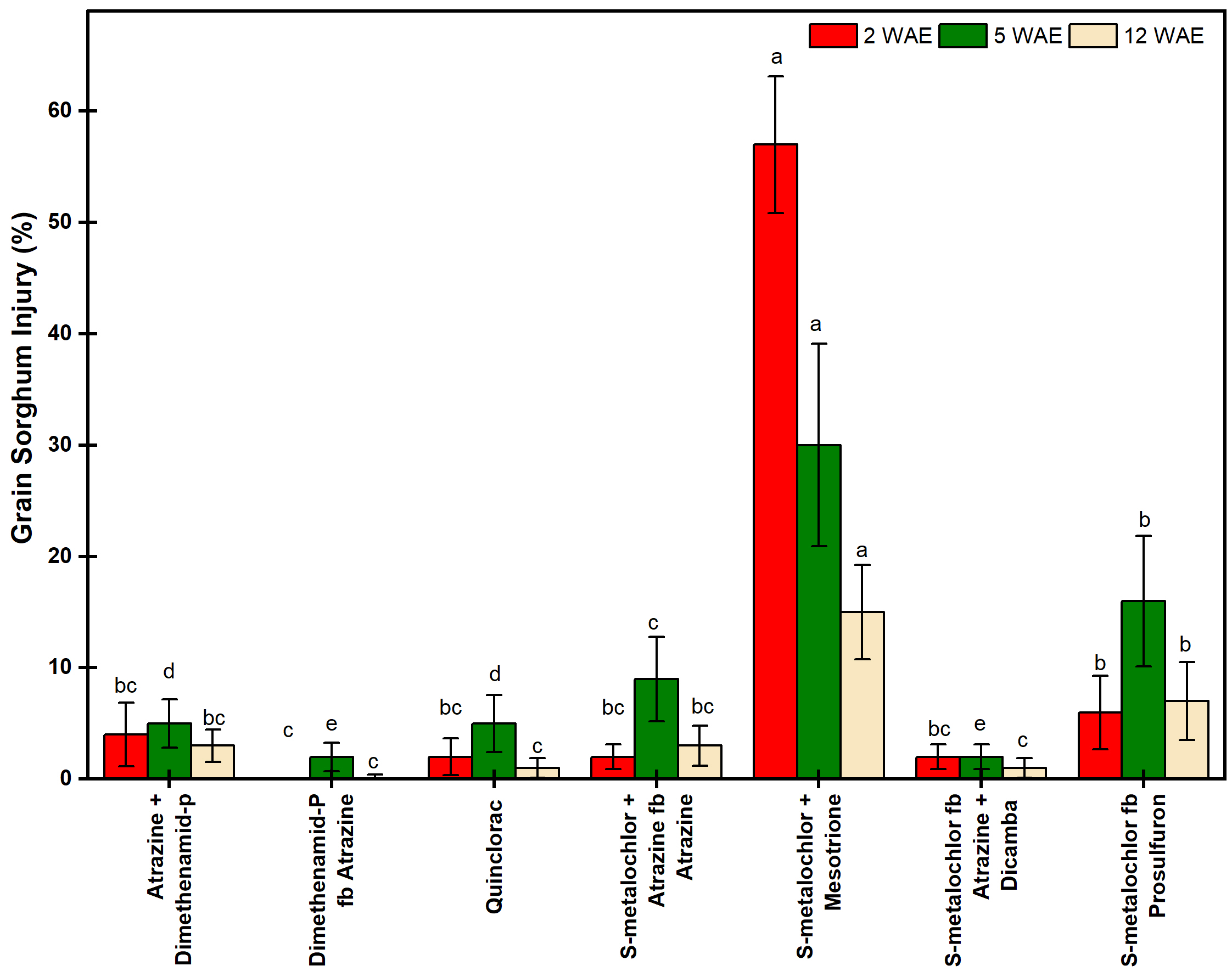
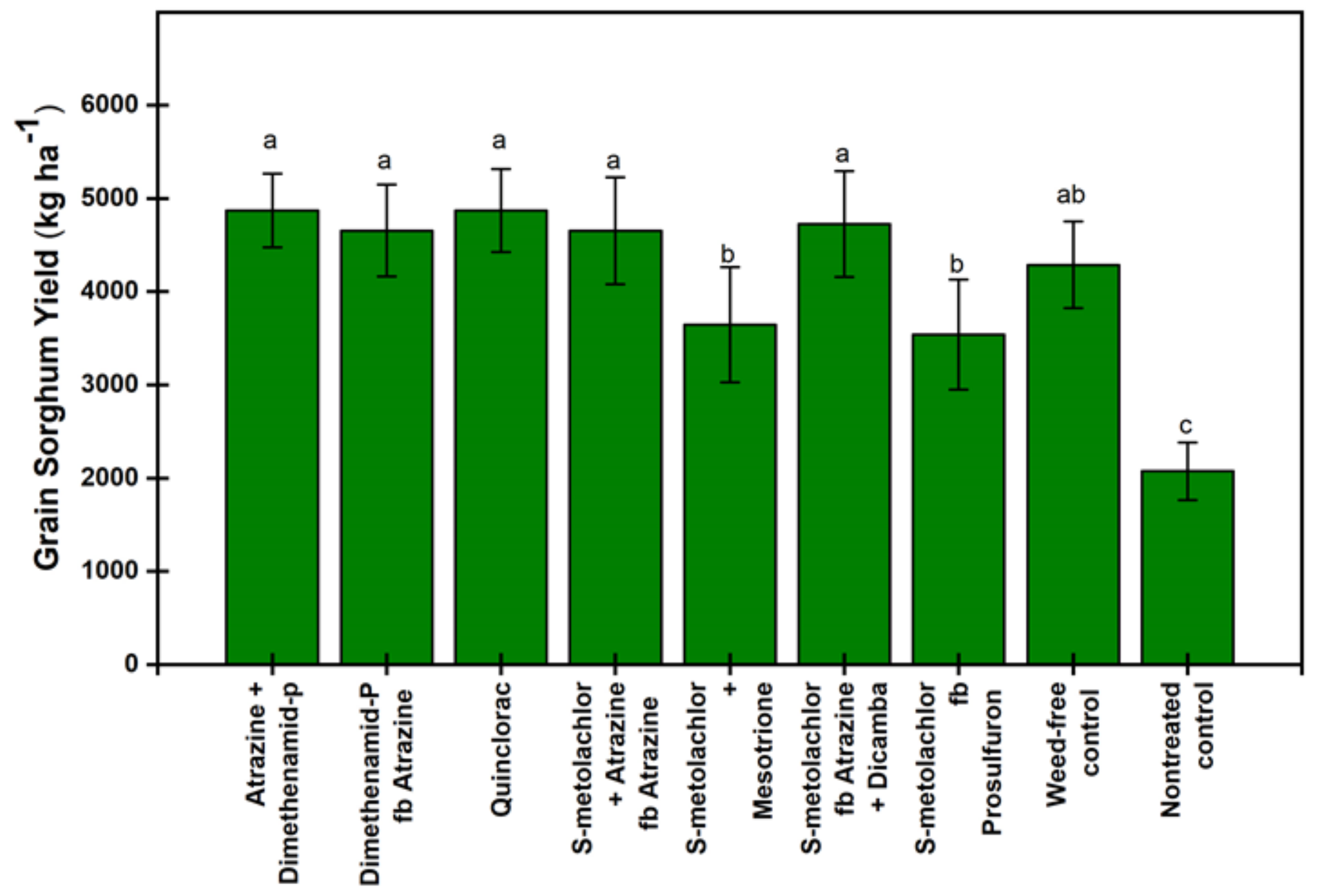
3.2. Grain Sorghum Injury and Yield
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Prasifka, J.; Heinz, K.; Sansone, C. Timing, magnitude, rates, and putative causes of predator movement between cotton and grain sorghum fields. Environ. Entomol. 2004, 33, 282–290. [Google Scholar] [CrossRef]
- Smith, K.; Scott, B. Weed Control in Grain Sorghum, Grain Sorghum Production Handbook; Espinoza, L., Kelley, J., Eds.; Cooperative Extension Service, University of Arkansas: Little Rock, AR, USA, 2010; pp. 47–49. [Google Scholar]
- Brown, D.W.; Al-Khatib, K.; Regehr, D.L.; Stahlman, P.W.; Loughin, T.M. Safening grain sorghum injury from metsulfuron with growth regulator herbicides. Weed Sci. 2004, 52, 319–325. [Google Scholar] [CrossRef]
- Abit, M.J.M.; Al-Khatib, K.; Regehr, D.L.; Tuinstra, M.R.; Claassen, M.M.; Geier, P.W.; Stahlman, P.W.; Gordon, B.W.; Currie, R.S. Differential response of grain sorghum hybrids to foliar-applied mesotrione. Weed Technol. 2009, 23, 28–33. [Google Scholar] [CrossRef]
- Grichar, W.J.; Besler, B.A.; Brewer, K.D. Weed control and grain sorghum (Sorghum bicolor) response to postemergence applications of atrazine, pendimethalin, and trifluralin. Weed Technol. 2005, 19, 999–1003. [Google Scholar] [CrossRef]
- Moore, J.W.; Murray, D.S.; Westerman, R.B. Palmer amaranth (Amaranthus palmeri) effects on the harvest and yield of grain sorghum (Sorghum bicolor). Weed Technol. 2004, 18, 23–29. [Google Scholar] [CrossRef]
- Martin, J. Early-Season Weed Management Strategies in Grain Sorghum. In Fact Sheet; University of Kentucky Cooperative Extension Service: Lexington, KY, USA, 2004. [Google Scholar]
- Rosales-Robles, E.; Sanchez-de-la-Cruz, R.; Salinas-Garcia, J.; Pecina-Quintero, V. Broadleaf weed management in grain sorghum with reduced rates of postemergence herbicides. Weed Technol. 2005, 19, 385–390. [Google Scholar] [CrossRef]
- O’sullivan, J.; Sikkema, P. Sweet corn (Zea mays) cultivar sensitivity to CGA 152005 postemergence. Weed Technol. 2001, 15, 204–207. [Google Scholar] [CrossRef]
- Vencill, W.K. Herbicide Handbook; Weed Science Society of America: Champaign, IL, USA, 2002. [Google Scholar]
- Wichert, R.; Townson, J.; Bartlett, D.; Foxon, G. Technical review of mesotrione, a new maize herbicide. In Proceedings of the Brighton Crop Protection Conference Weeds, Farnham, Surrey, UK, 1999; pp. 105–112. [Google Scholar]
- Duke, S.; Dayan, F.; Romagni, J.; Rimando, A. Natural products as sources of herbicides: Current status and future trends. Weed Res. 2000, 40, 99–111. [Google Scholar] [CrossRef]
- Armel, G.R.; Wilson, H.P.; Richardson, R.J.; Hines, T.E. Mesotrione combinations in no-till corn (Zea mays). Weed Technol. 2003, 17, 111–116. [Google Scholar] [CrossRef]
- Horky, K.; Martin, A. Evaluation of preemergence weed control programs in grain sorghum. In Weed Control in Specialty Crops; North Central Weed Science Society (NCWSS) Report v. 62: Lincoln, NE, USA, 2005; pp. 30–32. [Google Scholar]
- Miller, J.; Regehr, D. Grain sorghum tolerance to postemergence mesotrione applications. Proc. North Cent. Weed Sci. Soc. 2002, 57, 136–143. [Google Scholar]
- Kaczmarek, S. A study on Sorghum bicolor (L.) Moench response to split application of herbicides. J. Plant Prot. Res. 2017, 57, 152–157. [Google Scholar] [CrossRef]
- Osborne, B.T.; Shaw, D.R.; Ratliff, R.L. Soybean (Glycine max) cultivar tolerance to SAN 582H and metolachlor as influenced by soil moisture. Weed Sci. 1995, 43, 288–292. [Google Scholar] [CrossRef]
- Mueller, T.C.; Steckel, L.E. Efficacy and dissipation of pyroxasulfone and three chloroacetamides in a Tennessee field soil. Weed Sci. 2011, 59, 574–579. [Google Scholar] [CrossRef]
- Grichar, W.J.; Besler, B.A.; Brewer, K.D. Effect of row spacing and herbicide dose on weed control and grain sorghum yield. Crop Prot. 2004, 23, 263–267. [Google Scholar] [CrossRef]
- Horak, M.J.; Loughin, T.M. Growth analysis of four Amaranthus species. Weed Sci. 2000, 48, 347–355. [Google Scholar] [CrossRef]
- Sellers, B.A.; Smeda, R.J.; Johnson, W.G.; Kendig, J.A.; Ellersieck, M.R. Comparative growth of six Amaranthus species in Missouri. Weed Sci. 2003, 51, 329–333. [Google Scholar] [CrossRef]
- Keeley, P.E.; Carter, C.H.; Thullen, R.J. Influence of planting date on growth of Palmer amaranth (Amaranthus palmeri). Weed Sci. 1987, 35, 199–204. [Google Scholar] [CrossRef]
- Pannacci, E.; Bartolini, S. Evaluation of chemical weed control strategies in biomass sorghum. Plant Prot. Res. 2018, 58, 404–412. [Google Scholar]
- Riar, D.S.; Norsworthy, J.K.; Steckel, L.E.; Stephenson, D.O.; Eubank, T.W.; Bond, J.; Scott, R.C. Adoption of best management practices for herbicide-resistant weeds in midsouthern United States cotton, rice, and soybean. Weed Technol. 2013, 27, 788–797. [Google Scholar] [CrossRef]
| Year | Planting | Emergence | Harvest |
|---|---|---|---|
| 2005 | 17 May | 23 May | 23 September |
| 2004 | 26 May | 31 May | 21 September |
| 2003 | 27 May | 2 June | 24 September |
| Common Name | Trade Name (Formulation) | Manufacturer |
|---|---|---|
| Quinclorac | Facet (75 DF) | BASF Corporation, Research Triangle Park, NC 27,709 |
| Atrazine + Dimethenamid-p | Guardsman (5 L) | BASF Corporation, Research Triangle Park, NC 27,709 |
| S-metolachlor | Dual II Magnum (7.64 EC) | BASF Corporation, Research Triangle Park, NC 27,709 |
| Dimethenamid-P | Outlook (6 EC) | BASF Canada Inc., Mississauga, ON |
| Atrazine | AAtrex (4 L) | Syngenta Crop Protection, LLC, Greensboro, NC 27,419 |
| Mesotrione | Callisto (4 L) | Syngenta Crop Protection, LLC, Greensboro, NC 27,419 |
| Prosulfuron | Peak (57 DG) | Syngenta Crop Protection, LLC, Greensboro, NC 27,420 |
| Dicamba | Clarity (4 S) | BASF Corporation, Research Triangle Park, NC 27,709 |
| Herbicide Treatments | Application Rate | Application Timing |
|---|---|---|
| kg ai ha−1 | ||
| Quinclorac | 0.42 | Pre-emergence |
| Atrazine + Dimethenamid-p | 2.8 | Pre-emergence |
| S-metolachlor fb Atrazine + Dicamba | 1.40 fb 1.79 + 0.28 | Pre-emergence fb ‡ two-leaf stage of sorghum |
| Dimethenamid-P fb Atrazine + COC * | 0.78 fb 1.79 | Pre-emergence fb two-leaf stage of sorghum |
| S-metolachlor + Atrazine fb Atrazine + COC (standard) | 1.42 + 1.12 fb 1.12 | Pre-emergence fb four-leaf stage of sorghum |
| S-metolachlor + Mesotrione + COC | 1.42 + 0.14 | VE † |
| S-metolachlor fb Prosulfuron + COC | 1.42 + 0.03 | Pre-emergence fb two-leaf stage of sorghum |
| Common Name | Chemical Family | Group | Site of Action |
|---|---|---|---|
| Quinclorac | Quinoline carboxylic acid | 4 | Synthetic Auxin |
| S-metolachlor | Chloroacetamide | 15 | Mitosis Inhibitor |
| Dimethenamid-P | Chloroacetamide | 15 | Mitosis Inhibitor |
| Atrazine | Triazine | 5 | Inhibitor of photosynthesis at photosystem II site A |
| Mesotrione | Triketone | 27 | Inhibitor of 4-hydroxyphenyl-pyruvatedioxygenase (4-HPPD) |
| Prosulfuron | Sulfonylurea | 2 | Acetolactate Synthase (ALS) or Acetohydroxy Acid Synthase (AHAS) inhibitor |
| Dicamba | Benzoic acid | 4 | Synthetic Auxin |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bararpour, T.; Hale, R.R.; Kaur, G.; Singh, B.; Tseng, T.-M.P.; Wilkerson, T.H.; Willett, C.D. Weed Management Programs in Grain Sorghum (Sorghum bicolor). Agriculture 2019, 9, 182. https://doi.org/10.3390/agriculture9080182
Bararpour T, Hale RR, Kaur G, Singh B, Tseng T-MP, Wilkerson TH, Willett CD. Weed Management Programs in Grain Sorghum (Sorghum bicolor). Agriculture. 2019; 9(8):182. https://doi.org/10.3390/agriculture9080182
Chicago/Turabian StyleBararpour, Taghi, Ralph R. Hale, Gurpreet Kaur, Bhupinder Singh, Te-Ming P. Tseng, Tessie H. Wilkerson, and Cammy D. Willett. 2019. "Weed Management Programs in Grain Sorghum (Sorghum bicolor)" Agriculture 9, no. 8: 182. https://doi.org/10.3390/agriculture9080182
APA StyleBararpour, T., Hale, R. R., Kaur, G., Singh, B., Tseng, T.-M. P., Wilkerson, T. H., & Willett, C. D. (2019). Weed Management Programs in Grain Sorghum (Sorghum bicolor). Agriculture, 9(8), 182. https://doi.org/10.3390/agriculture9080182






