1. Introduction
Muscadines (
Vitis rotundifolia Michx., syn.
Muscandinia rotundifolia (Michx.) Small) are a native American fruit crop and the first American grape to be cultivated [
1]. Muscadines were historically, and are currently, cultivated throughout the southeastern U.S. (defined herein as the U.S. states of Alabama, Florida, Georgia, Arkansas, Louisiana, Mississippi, North Carolina, South Carolina, and Tennessee) and, to lesser extents, in the U.S. states of Texas, Virginia, and Maryland. Georgia and North Carolina have historically contained the greatest muscadine acreages [
1] and, while recent surveys are not available, this is still believed to be the case today. The most recent, peer reviewed overview of the U.S. muscadine industry was published almost three decades ago [
1]. Along with production guides [
2], that review has served as a good resource to document the size and scale of the muscadine industry and to support applied muscadine culture. Updated, online resources are currently available to support judicious cultivar selection for both juice and fresh market [
3]. An updated regional muscadine production guide is currently being edited and is set for a late 2019 release [
4].
Muscadines, though planted across substantial acreage throughout the southeastern U.S. [
4,
5], remain a small-market, lesser known crop relative to the more widely known bunch grapes used for wine production. However, muscadines remain highly resilient and adaptable relative to
Vitis vinifera and interspecific hybrid bunch grapes cultivated for wine production due to their relative pest tolerance [
6,
7]. For example, muscadines require fewer pesticide inputs relative to bunch winegrapes [
8,
9]. Further, manual labor inputs, such as canopy management, is imperative for successful bunch winegrape production [
10,
11] but is less necessary for muscadine production [
2]. The inverse relationship between the ease of muscadine cultivation and their popularity is speculated to be a function of their regionally limited production range and historically limited consumption when compared to European bunch grapes. For example, muscadines remain a regional product and have been cultivated for consumption since the 19th century, while European winegrapes and their wines are global products that have been cultivated for consumption since centuries BC. Muscadine cultivation began several decades prior to the introduction of European grapes (
Vitis vinifera) to the southeastern U.S., which commenced in the 1980s. Yet, as alluded to above, the growth and proliferation of the muscadine industry has not paralleled that of the wine industry, which is centered around production of European and hybrid bunch winegrapes.
This review is descriptive and aims to educate the reader about the muscadine grape, a lesser known grape type that is anticipated to be of value to future consumers of locally produced food crops with native resiliency and heritage. In this sense, our review is educational and perhaps promotional. The fact that muscadine has been limited by regional production over a relatively short history may not justify it to remain a niche food crop with marginal production and marketability. Given the “native American food crop focus” of this special issue, we sought to provide an overview of muscadine’s history, taxonomy, and botany and discuss current and future production and marketing opportunities that highlight muscadine as a resilient, native food crop of the southeastern U.S.
2. Historical Perspectives
When New World grapes were introduced to the Old World, phylloxera (
Daktulosphaira vitifoliae Fitch) emerged as a major pest of European wine grapes (
V. vinifera L.) [
12]. Grape phylloxera is a common and important pest, which nearly eliminated
V. vinifera production when Europeans took infested native North American grapes to Europe. The use of rootstocks with native American genetics in their pedigree has become common practice to control grape phylloxera damage to
V. vinifera scions. However, muscadine can be planted on their own roots and grape phylloxera are not of concern.
V. vinifera production in the southeastern U.S. is extremely limited due to Pierce’s disease —caused by the bacterium
Xylella fastidiosa [
12]—in addition to unfavorable climatic conditions of humidity and highly variable precipitation patterns throughout the growing season. Although highly prevalent in its native range, Pierce’s disease does not limit the production of muscadine. Due to
V. vinifera’s susceptibility to
Xylella and grape phylloxera, muscadine became the primary fruit used for wine production in the southeastern U.S. [
13]; this is still the case today [
14] despite the continued and successful use of grapevine rootstocks with native genetics to overcome grape phylloxera damage to purely
V. vinifera scions.
Muscadine is ethnobotanically important; indigenous cultures of the southeastern U.S. used muscadine fruit as a food source and the vines for cordage [
15]. European colonists that first explored the Cape Fear estuaries of the New World Carolinas noticed that grapevines were in abundance [
13]. A vast majority of the fruits from the grapevines were dark in color and in local vernacular were referred to as “bullis”, “bull grape”, and “bullet grape” [
16,
17]. Early muscadine cultivars consisted of chance selections obtained from the wild. The relative ease by which vines could be propagated by layering allowed superior wild plants to easily be cloned [
18]. Initial selections from the wild were made based on traits important to any grape cultivar including berry size, fruit quality, vine vigor, and productivity. However, a unique concern to muscadine is the persistence of the berry on the vine. Unlike
V. vinifera berries, muscadine berries generally abscise from the pedicel at maturity which complicates their harvest. Muscadine berries ripen unevenly and thus require recurring, periodic harvests to pick ripe berries before they fall from the vine. Initial selections were made from vines in which the fruit were persistently attached to the cluster. This allowed harvest to be delayed until berries were more fully ripened and had a better flavor, they could also be cluster picked and shipped to market without shattering.
While most native vines produce dark purple berries, rare bronze colored (non-pigmented mutation) grapes had superior winemaking potential and were predominantly selected for cultivation [
19]. The first bronze cultivars were propagated from the wild and may have been from several sources gathered from various locations [
13]. The outcome of early cultivation was the proliferation of bronze colored cultivars for fruit production. “Scuppernong” became the common vernacular to refer to these early, bronze selections [
13]. Eventually, a bronze cultivar called ‘Scuppernong’ would emerge that was commonly cultivated for wine, juice, preserves, and, to lesser extents, fresh consumption.
Early muscadine cultivation used an overhead arbor trellis system, minimal pruning, and few inputs [
20]. Because the plant material was native, it was well adapted to the environment and common pests thereof. Muscadines were grown throughout the south and were part of the landscape of rural homes into the 20th century [
19]. Native muscadines are dioecious and fruiting vineyards consisted of female plants that needed male plants as pollinizers. These were provided via wild male muscadines in the nearby woodlands or by interspersed male vines within the vineyard [
15,
18]. As demand increased for muscadine wines, production was increased by adding to existing arbors or constructing new arbors [
21]. In 1843, the largest recorded ‘Scuppernong’ planting covered 2.4 ha and produced an estimated 50 barrels (6 kl) of wine in North Carolina [
21,
22]. Until national prohibition, about 60% of muscadine production was for wine, and “Virginia Dare” muscadine wine was the most popular wine in the United States [
23].
By the early 1900s, agricultural reports on muscadine from land grant universities in the southeastern U.S. and the United States Department of Agriculture (USDA) were disseminating information gathered from applied research into muscadine cultural practices, trellising systems, cultivar evaluations, and pests [
24,
25,
26]. Muscadines were considered to be well-suited for production in the southeastern Coastal Plain and suggestions for improving yield through pruning, fertilization, and breeding were popular topics within the reports [
24,
25,
26]. The grape breeding program in North Carolina began in 1908 by the North Carolina Experiment Station in cooperation with the USDA. The primary goal of this program was the production of hermaphroditic cultivars. Early breeding work determined that self-fertile seedlings could be produced at a very low rate from crosses between male and female vines, and the resulting hermaphrodites were used to develop new hermaphroditic cultivars [
17]. Such breeding efforts led to the release of several hermaphroditic cultivars in 1946. Hermaphroditism improved vineyard yield due to several factors including: replacement of male vines with hermaphroditic vines that produce a crop, improved pollination levels in hermaphroditic vines leading to increased crop set, and a larger cluster size in hermaphroditic vines. Hermaphroditic cultivars were quickly adapted for the majority of the processing market due to their increased productivity, but female cultivars predominated in the fresh-market due to their superior, consumer-preferred fruit quality. The University of Georgia began breeding muscadines in 1909 [
13] and was first to release new muscadine cultivars from controlled crosses [
27]. Early releases were strongly selected for non-shattering berries with sweet tender pulp, improved ripening, and harvestability. The majority of cultivars being grown at the time were for home use or local markets as fresh fruit, preserves, or jellies.
4. Botany
Subgenus
Muscadinia species have genetic and morphological differences compared to subgenus
Vitis species. The most distinct genetic difference is the number of somatic chromosomes:
Muscadinia species have 40 (2 ×= 2n = 40), while
Vitis species have 38 (2 ×= 2n = 38) [
41]. Linkage map analysis indicates an overall high degree of collinearity between
V. vinifera physical map and muscadine maps [
42]. Linkage analysis indicates that
V. vinifera chromosome 7 is split into two independently segregating linkage groups in muscadine [
42]. The muscadine berry color locus mapped to an interval aligning to
V. vinifera chromosome 4, while the MYB transcription factor genes controlling fruit color in
V. vinifera map to chromosome 2, suggesting that a mutation in a different gene in the anthocyanin biosynthesis pathway determines berry color in muscadine [
42]. While the muscadine sex locus mapped to a region on chromosome 2 associated with the sex locus of
V. vinifera subsp.
sylvestris [
42], genetic control of the sex expression in some backgrounds of muscadine differ from
V. vinifera [
43].
Morphologically, muscadine grapes (
V. rotundifolia) have smaller clusters, unbranched tendrils, formation of an abscission zone between the fruit and rachis, smooth and thin bark, prominent lenticels, continuous pith, and berries with thick skins and a unique fruity aroma. In contrast, other
Vitis species (other than
V. rotundifolia) have bigger clusters, branched tendrils, no abscission zone between the berry and rachis, striated bark on young wood, thick and rough bark that peels in strips on old wood, less-dense wood than
Muscadinia, and pith interrupted by diaphragms at nodes [
44].
Muscadinia and
Vitis are graft incompatible [
1]. Even though
Vitis species can hybridize freely,
Vitis ×
Muscadinia hybrids are rare and normally sterile as a result of having 39 chromosomes.
Muscadine possess three distinct floral types: perfect hermaphrodite, staminate (male), and imperfect hermaphrodite (female) [
45] (
Figure 1). The staminate flowers have only filaments and anthers; the imperfect hermaphrodite flowers, though perfect flowers, have stunted filaments and anthers with sterile pollen; and perfect hermaphrodite are self-fertile and only rarely observed in the wild [
46]. Each flower type has floral petals that form a cap (calyptra) that is shed at bloom, exposing the nectary; however, there remains uncertainty as to how or what is transferring muscadine pollen. Insects were reported to be the dominate mode of muscadine pollen transport [
46]. Pollen from muscadine have a waxy surface, which is ideal for adhesion to protuberances such as fine hairs of insects (e.g., bees). Muscadine flowers are small (2 to 3 mm), odiferous, and have shallow nectaries representing small reward for larger pollinizing insects. Small mining bees (
Halectus stultus Cress.) were found to be efficient pollinators of muscadine [
46]. Green bee (
Agapostemon splendens Lep.), gray bee (
Magachile sp. Latreille) and small bumble bee (
Bombus impatiens Cress.) also contribute to muscadine pollination [
47].
Wind pollination has somewhat dubiously been suggested to occur in muscadine [
44]. Wind-pollinated species such as hops (
Humulus lupulus L.) and olive (
Olea europaea L.) have relatively long pistils, lengthy styles, and downy stigmas, which have evolved to catch wind-blown pollen, whereas muscadine stigmas are relatively small and smoothly rounded. By contrast to wind-pollinated plants, the pollen of muscadine is waxy and does not easily release from the anther and adheres strongly to hairs (e.g., camel’s-hair brush) [
46]; further, muscadine flowers develop deep within the grapevine canopy, usually under dense leaf cover and largely protected from wind. These are evolutionary characteristics that lend themselves more to insect than wind-dependent pollination.
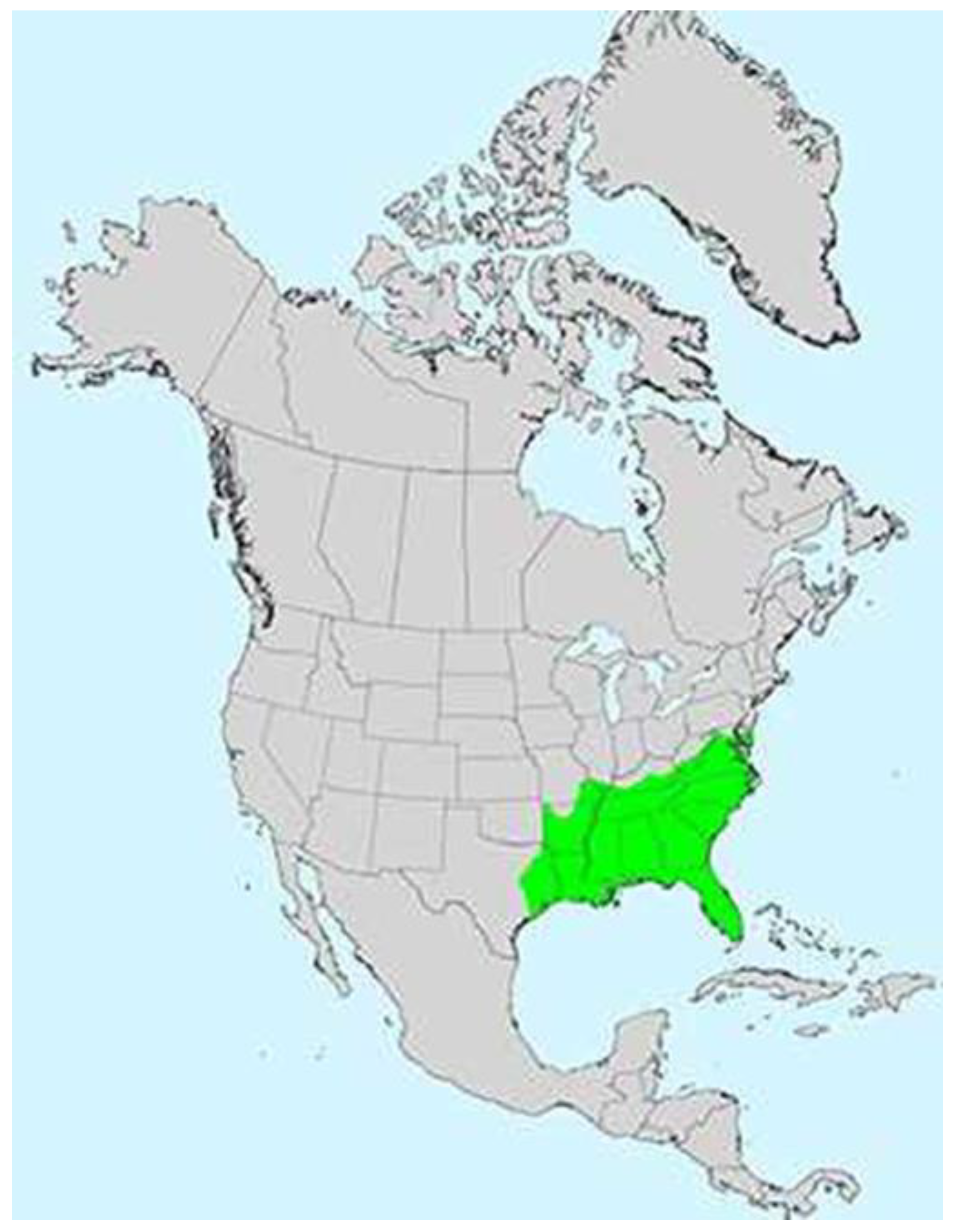
5. Production Range and Scale
Muscadine’s pest tolerance, perennial longevity, and adaptation to variable winter temperatures enable cultivation in its native, humid, subtropical environment. Muscadine range is predominantly composed of the coastal plains to the lower piedmont and mountain regions throughout the southeastern U.S. (
Figure 2) [
48] and is limited by cold temperatures where −12 °C (10 °F) causes damage [
2] and −18 °C (0 °F) results in mortality [
44]. Therefore, while newer, commercial plantings remain viable and common in the piedmont of North Carolina and piedmont and mountains of Georgia, cold injury is often reported in those locations, particularly in sensitive cultivars. Nonetheless, muscadine acreage continues to expand in non-traditional regions (e.g., outside of the U.S. coastal plains) in the southeastern U.S. [
49]. The northern range limit for muscadine is Virginia and Maryland wherein conditions are much more favorable for muscadine cultivation along the Atlantic Coast. Westward, muscadine ranges along the Gulf States through Texas. Muscadine habitat is in subtropical climate, where other native American grapes (e.g., V. labrusca L.) have not been widely used even though bunch grapes can be grown in piedmont areas within the same region [
12]. Therefore, some American bunch grapes do not possess the genetic potential to successfully evolve in the environment of the coastal plains [
50]. Contrastingly, muscadines resiliency in productivity and disease tolerance in the southeastern U.S. coastal plains is suggestive of evolution on sandy to sandy loam soils with extreme disease and pest pressure, and intense heat and humidity.
Starting in the 1960s, muscadine plantings increased, which followed the development of agricultural chemistries to control pests and improve production practices to aid in disease management, trellising, plant spacing, and pruning [
1,
6,
7,
51,
52,
53]. In the 1960s there was an estimated 450 ha of muscadine planted throughout the southeastern U.S., which included Alabama, Arkansas, North and South Carolina, Florida, Georgia, and Mississippi [
6]. Historically, muscadine had been grown in small plantings for home use and was sold locally as fresh fruit, preserves, juice, and wine [
6]. Growers in the southeastern U.S. envisioned muscadine as a commercial crop and continued to increase production throughout the 1970s. Muscadine production in North Carolina alone increased by 76% over an eight-year-period when it went from 240 ha in 1968 to about 1000 ha in 1976 [
1]. Plantings continued to expand throughout the region over the last few decades, though not at nearly that same rate as previous. Today, there is an estimated 1200 ha of muscadines planted in the Carolinas, Georgia, and Florida [
4] and likely several hundred more ha planted throughout other regional states. A regional survey of muscadine acreage and cultivar make up is well overdue; to the authors’ knowledge, the last widely available regional survey was published in 1991 [
1].
6. Diseases and Insects
Muscadine is tolerant to many diseases, particularly relative to bunch winegrapes that contain at least partial
V. vinifera genetics [
6,
8,
9,
10]. However, fungal diseases, particularly fruit rots, can still limit muscadine production. Fruit rots can degrade quality especially on bronze colored fruit, e.g., ‘Fry’ (fresh market) and ‘Carlos’ (wine and juice). Bitter rot (
Greeneria uvicola Berk. and Curt.), Ripe rot [
Colletotrichum gloeosporioides (Penz.) Penz. and Sacc. (teleomorph
Glomerella cingulate (Stonem.) Spauld. and Schrenk)], and Macrophoma rot [
Botryosphaeria dothidea (Moug. Ex. Fr.) Ces. and de Not] are three important diseases of muscadine that can be controlled with repeated azoxystrobin and ethyl mercaptan (captan) applications throughout the season [
8]. These chemistries also control leaf fungal diseases, e.g., black rot [Guignardia bidwellii (Ellis) Viala and Ravas] and angular leaf spot (Mycosphaerella angulate Jenkins) [
8]. Other, non-fungal organisms (e.g., bacterium, viruses, phytoplasmas, or nematodes) have not proven to limit muscadine production [
6,
44] as they have for bunch winegrapes [
9,
11]. Wood borne diseases and a host of other fungal diseases, namely downy mildew (
Plasmopara viticola) and
Botrytis bunch rot, largely affect bunch winegrapes but not muscadine [
6,
8,
9,
10]. Grape crown gall (caused by
Agrobacterium vitis) is almost ubiquitous throughout bunch winegrape vineyards in the eastern U.S. but has only recently been verified in muscadine vineyards nearly 2000 ft above sea level in northern Georgia [
54].
In an effort to use softer chemistries and reduce input costs, sulfur and copper products can be used as cover sprays to reduce fungal infection and minimize pathogen resistance to conventional fungicides. Some formulations of sulfur are organic certified and can be used as a fungicide. Efficacy of sulfur can be low, but resistance is nil, which is important due to weekly application needed for control [
8,
44]. Lime sulfur is not recommended due to phytotoxicity in muscadine [
44]. Copper is also organic certified and can be more effective than sulfur on some fungal pathogens. Due to lime sulfur being phytotoxic, caution should be taken when applying copper compounds that are basic (≥7 pH). Further research into organic certified fungicides is warranted.
The grape root borer (
Vitacea polistiformis) is a prominent insect pest in muscadines in the southeastern U.S. and can occasionally lead to die-back and yield loss [
8]. However, root borer dispersion is often unpredictable and not all vineyards suffer equally from root borer infestation. Generally, insect pressure is minimal and growers will apply insecticides when economic thresholds are surpassed, e.g., leaf or berry damage. As new regional pests have emerged, e.g., spotted winged drosophila (
Drosophila suzukii Matsumura) and the brown marmorated stink bug (
Halyomorpha halys Stål), muscadine has yet to be significantly affected [
8]. In contrast to the relatively few insect pests in muscadine [
8], there are a suite of insect pests that need managed in commercial bunch winegrape (hybrid and
V. vinifera) production, either due to their roles as vectors of systemic diseases like bacteria and viruses and/or due to their physical damage of both reproductive and vegetative vine tissues [
9,
10,
11]
7. Cultivars
The North Carolina State University/USDA muscadine breeding program focused on juice cultivars suitable for the wine industry. From their efforts, ‘Carlos’ and ‘Noble’ were released in the 1970s, which together account for an estimated 80% of wine being produced within the region. Both of these cultivars have a strong vine vigor, hermaphroditic flowers, relatively good cold tolerance (as observed in comparison to other muscadine cultivars) and are high-yielding (15 to 25 t ha
−1). Muscadine breeding from the University of Georgia program emphasized breeding for fresh-market cultivars, emphasizing a larger berry size, improved skin texture, and excellent flavor. Through repeated phenotypic selection, berry size was increased from around 5 g in the first releases to 15 g in the most recent releases [
55]. In the past, female cultivars played a dominant role in fresh-market plantings because berry size in female cultivars was typically larger than in hermaphroditic cultivars, perhaps due to gene linkage [
56]. However, recent releases from the program have been able to combine very large berry size with hermaphroditic flowers, and female selections are no longer considered for release in the program due to their lower and irregular yields [
57]. This development is allowing fresh-fruit growers to experience similar yield gains that juice growers experienced with the adoption of hermaphroditic cultivars. Other breeding programs include private endeavors [
58], the University of Arkansas, and Florida A&M University.
While fresh-market cultivars might be considered by many to be the backbone of historical commercial muscadine production, ‘Carlos’ and ‘Noble’ remain the most widely planted muscadine cultivars [
2,
4,
5]; both cultivars are highly productive and do not require absence of cosmetic defects as they are processed and not for the fresh market. Yet, commercial growers are using fresh-market cultivars for wine production [
59,
60], and this may be a good use for fresh-market cultivars that remain on the vine for extended periods and are no longer marketable for fresh consumption.
8. Production
Cultivated muscadines are vigorous plants only requiring significant labor inputs for young vine training and perennial dormant pruning [
2,
4]. Such management practices greatly improved yields and were facilitated by the transition of trellising practices from predominately overhead arbor systems to more scalable and reachable, single-wire trellises [
19,
44]. The single-wire trellis was later useful in the adaptation to mechanization, particularly for commercial producers of muscadine wine [
2,
20,
44]. Through advancements in mechanized farming, trellis systems, and pruning methods, vineyards are presently established to accommodate ease of harvest, improve yield, maximize spray penetration into canopies, and provide quick egress of freshly harvested fruit [
16,
44]. Trellising, training, and pruning remain important commercial practices [
18,
44]. There have been investigations in trellis types [4-arm renewal, Geneva double-curtain (GDC), and single wire] [
18,
44], of which presently the single, high wire trellis remains the most frequently used based on commercial production efficiency and economy. Compared to the GDC and 4-arm renewal, single-wire systems require less material and labor inputs, permit greater vine densities per unit land, are easier to maintain, and are adapted for mechanical harvest. Pruning represents the most labor-intensive input in muscadine vineyards. Some growers have adopted mechanical pruning to lower dormant pruning costs and increase its efficiency [
49]. Although the practice is not widely adopted, some growers will mechanically summer prune the shoots trailing to the ground to promote better canopy and fruit zone spray penetration and improve harvest efficiency. While muscadine production requires hand labor primarily for young vine training, pruning, and harvest (if fresh-market cultivars are grown), commercial bunch winegrape production requires hand labor inputs for most of the season [
10,
11] as pruning, shoot thinning, shoot positioning, fruit zone leaf removal, multiple canopy hedging passes, bird netting placement, and harvest all represent timely labor inputs that cannot currently be implemented with a tractor.
Muscadines have two traits that make them better adapted to a highly variable winter climate relative to other non-native fruit crops being grown in the southeastern U.S. First, muscadines require relatively few chill hours in order to successfully bloom. While little work has been done to determine the exact amount of chilling needed, muscadines are currently being commercially grown in a region in Florida which receives a modest 200 chill hours on average. Secondly, they also have a high heat-accumulation requirement, which ultimately equates to a late bud break and relatively low frost susceptibility—particularly relative to other non-native perennial fruit crops produced throughout the southeastern U.S. For example, while several perennial fruit crops have suffered from recent spring frost events in the southeastern U.S., muscadines were largely unaffected by those frost events. Muscadine will thus remain a viable option for perennial fruit crop production if trends of more moderate winter temperatures continue and result in early bud break (and frost injury), and the failure to meet chill hours, in other regionally produced perennial fruit crops.
9. A Future Prospectus for Increased Production, Market Awareness, and Consumption
In the early 21st century, muscadine remains regionally popular but has little demand outside of the southeastern U.S. As new technologies are developed in grape production, the future looks promising for muscadine cultivation if the market demand can be increased. The future acreage under commercial muscadine grape production in the southeastern U.S. has the potential to rise in the face of a variable climate which may become increasingly unfavorable to the culture of non-native grapes. Due to muscadine’s native resiliency and tolerance to most fungal, viral, and bacterial pests, they require minimal chemical inputs for successful production. For example, muscadine requires fewer labor and pesticide inputs when compared to bunch grapes [
8,
9,
10,
11]. Therefore, muscadine cultivation is extremely possible in the southeastern U.S. and can be a fiscally and environmentally sustainable, commercial production system. However, for muscadine production to increase, marketing will play a key role. There are two outlets for muscadine fruit: fresh market and processed products. Presently, processed fruit dominate the market as juice for wine. Because muscadine can be successfully grown in the coastal plain, there are many small plantings that produce fruit for homeowners and their neighbors. Future marketing should focus on increasing access into urban centers and areas outside of the growing region by targeting a new consumer base.
Muscadine could greatly benefit from heightened consumer acceptance and awareness, and thus marketability, of muscadines. Muscadines remain “niche” (appealing to small segment of population) in their market, perhaps due to social and cultural aspects that have made wine and winegrapes prominent, even in non-native, fungal disease-pervasive regions. It will be important for muscadines to gain a broader market footprint to encourage substantial muscadine industry growth. The muscadine industry is well positioned to take advantage of the “locally produced” and “organic” market segments, as muscadine are one of the very few crops that can meet both of these label requirements in the southeastern U.S.
Potential strategies to increase consumer demand are to highlight sustainability and nutraceutical benefits, and to continue the development of new cultivars with improved quality. Sustainability in agriculture relates to economically viable crops that promote environmentally sound cultural practices, stewardship of the land, and a benefit to the community. Muscadine can be successfully cultivated with minimal inputs thus permitting insect and arthropod communities to thrive. In muscadine vineyards, cover crops are managed in the row middles mitigating soil erosion. Muscadine are long-lived and in the southeastern U.S., there are multigenerational vineyards that have provided income sources benefiting local communities. These topics would make for good marketing points and areas for further research.
Recent work has shown that familiarity with muscadine products (wine, jams, jellies, health products, etc.) positively influences the purchasing of muscadine wine in the southern U.S. [
61]. A key aspect in the expansion of the muscadine market will therefore be the development of products that appeal to consumers outside of the region and to those with no prior experience with muscadines. While belief about muscadine’s health benefits was only slightly influential in consumer decision to purchase muscadine wine [
61], muscadine has tremendous nutritional benefits. Muscadine may be considered a super food due its high concentration of phenolic compounds [
62] and compounds shown to limit cancer growth [
62]. One possible route to increase marketability of muscadine products is to highlight the health benefits of muscadine consumption, akin to the recent trend for consumer acceptance of products made from “power fruits” such as acai berry, blueberry, and pomegranate. Recent research found muscadines contain a wide range of polyphenolic compounds with antioxidant antiplatelet properties, such as ellagic acid, anthocyanins and flavonols [
62,
63,
64]. Muscadine grape skin extract has been reported to inhibit prostate cancer cell growth and induce apoptosis without affecting normal prostate epithelial cells [
65,
66,
67,
68]. Water-soluble muscadine seed extract (pH 3.3–3.78) has been reported to produce strong antimicrobial inhibition to
Enterobacter sakazakii, which causes meningitis and necrotizing enterocolitis in infants [
69]. In the past two decades, there has been an increasing interest in developing nutraceutical food products and pharmaceuticals from muscadine tissues including skin, seeds and pulp.
Increasing the muscadine fresh fruit market will require a continued refinement of berry quality via traditional breeding efforts to compete with
V. vinifera table grapes. Since the majority of fresh-market sales are for immediate consumption rather than processing, improved berry texture is a paramount trait. Consumers are unlikely to be accepting of muscadine berries with the traditional tough skins and soft flesh. Newer releases combine a crisp pulp with a tender skin [
70,
71]. However, a balance must be sought as selections with a very tender skin are more susceptible to cracking and splitting after precipitation from rain and overhead irrigation [
72]. Seedlessness within muscadine would be an ideal trait that can improve consumer perception. Recently, stenospermocarpic seedlessness has been introgressed from
V. vinifera into muscadine [
58,
73]. The development of seedless muscadine cultivars, especially if combined with superior flesh, skin, and flavor attributes, has the potential to compete in the marketplace with other fruit and berry crops.
Without increased consumption of muscadine, new technologies will be adopted slowly. Growers may use soil and tissue sampling to gauge fertilizer applications. However, unlike V. vinifera winegrape viticulturalists, muscadine growers have yet to crop thin, monitor canopy light levels, leaf thin, or be concerned with vineyard terroir. Generally, the greatest concern is monitoring the amount of precipitation to guide spray programs and irrigation. The lack of technological advancements in muscadine production may partially be due to a lack of regional research efforts relative to blueberries, peaches, and bunch grapes, all which have received greater research attention in the southeastern U.S. Muscadine growers are indeed interested in technologically advancing vineyard management and inputs; however, support is needed for research by which marketing and increased consumption could help generate funding.
As with most horticultural crops, access to affordable labor is and will be a concern. Advances in harvest technology, sorting, packing, and transport could ease the burden through automated and machine assist technologies. Labor inputs into muscadine are primarily for dormant season pruning and, in the case of fresh-market plantings, berry harvest. Pruning labor can be minimized with the adoption of mechanized pruning, which has already been implemented in several large commercial muscadine vineyards. Mechanized harvest is generally used in berries destined for the juice market but is currently not possible in fresh-market plantings. Fresh fruit are hand harvested, sorted, placed into clamshell packaging, and shipped to market. Temporary harvest labor needed to accomplish these tasks can cost 30% to 40% of the gross revenue [
74]. Research to identify efficient use of labor and improve harvest mechanization could greatly enhance the profitability of muscadine production.