2.1. Assay of Dominant Factors
Five separate experiments were conducted concurrently in controlled greenhouse conditions (Edinburg, TX) to examine the association of abiotic (moisture, micronutrients, phosphorus, nitrogen) and biotic (native microbial communities) conditions with nodulation in a common cover crop legume, cowpea (Vigna unguiculata). Each of these conditions can interact with any of the other factors, but exploring these interactions was beyond the scope of this study. In each experiment, the potential interaction of AMF as a participant in successful nodulation of cowpea was also examined. Although cowpea was not one of the three legume species that experienced nodulation failure in the original field trials, it is a commonly used warm season cover crop in this region and was planned for future plantings in this field.
For all experiments, Iron and Clay cowpea seeds (
Vigna unguiculata; Johnny’s Seeds, Winslow, ME) were soaked for 10 min in 55 °C water, then pregerminated for 3 days in petri dishes in the greenhouse at 30 °C. Pregerminated seeds were then transplanted into 15 cm diameter plastic pots with 1500 g of a 1:1 mixture of perlite and soil obtained from the field where nodulation failure occurred (Hilltop Gardens, Lyford, TX). The only thorough survey of native rhizobia in this region was conducted with
Phaseolus vulgaris and found diverse populations of
Rhizobium phaseoli Dangeard [
56]. Little is known about the native populations of
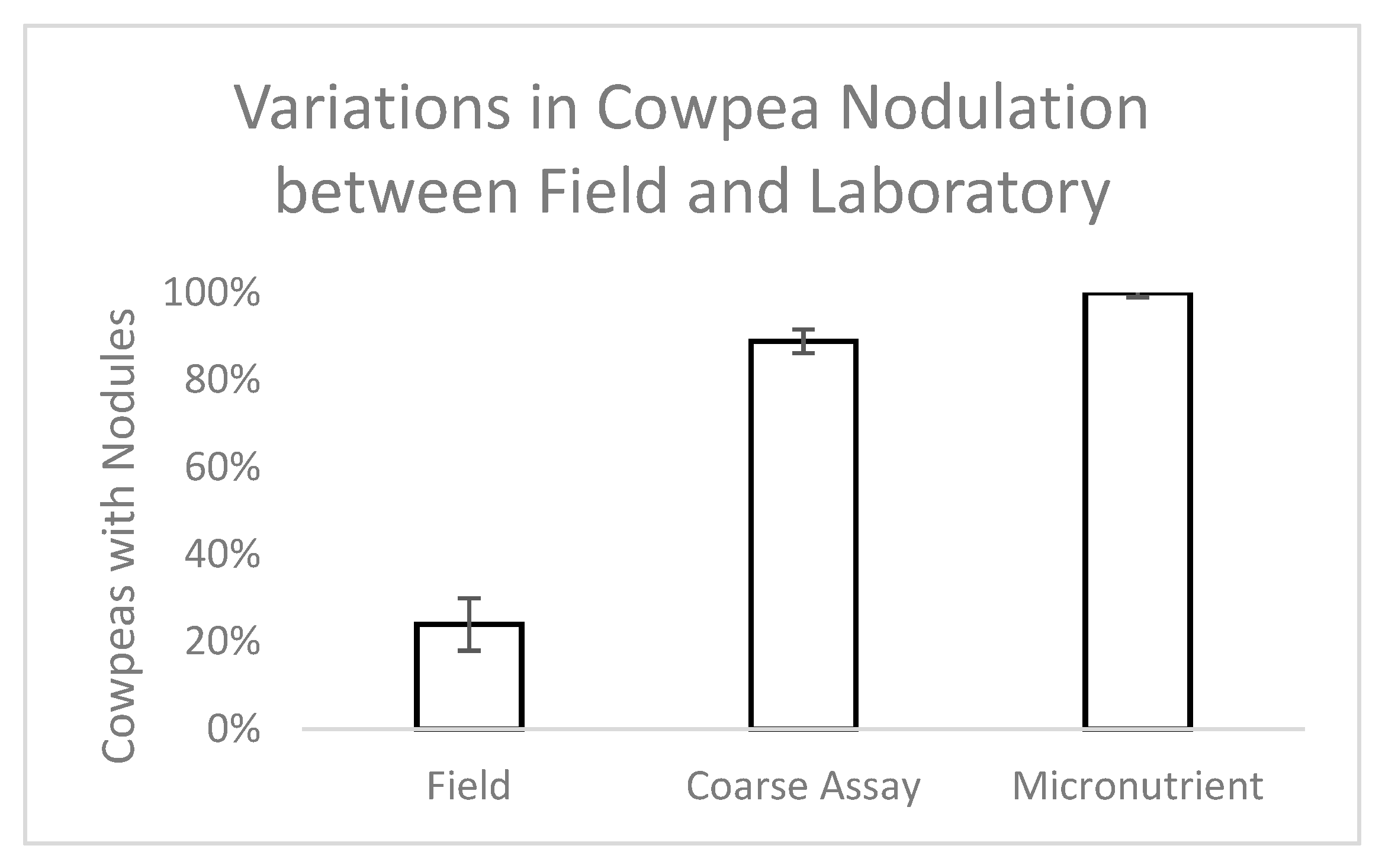
Bradyrhizobium spp. which nodulate cowpea, but uninoculated cowpea in native soil were included as a control in this study and did form nodules, suggesting the presence of a native rhizobial partner.
Native soil was a Willacy fine sandy loam with a pH of 8.1. Nutrient extractions were conducted by Texas Plant and Soil Lab (Edinburg, TX). Nitrogen and phosphorus values are the average of 25 samples analyzed by Mehlich III extraction. Micronutrient values are from a single soil sample using the hot water method for boron extraction and DTPA for cobalt, copper, manganese, molybdenum, and zinc. Soil pH was measured using a multimedia pH meter (Bluelab, Tauranga, New Zealand) and soil texture was determined using the USDA NRCS Web Soil Survey and confirmed by hydrometer [
63].
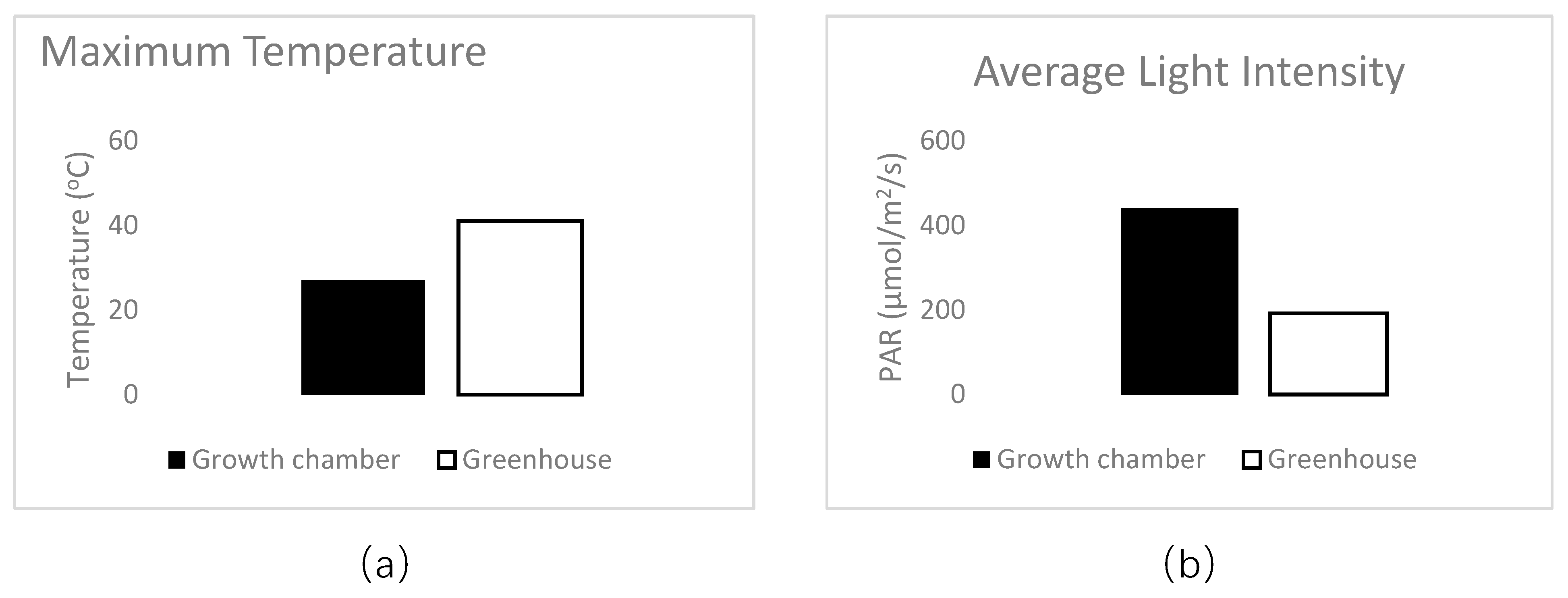
At transplant, 1 mL of Bradyrhizobium sp. (Vigna) inoculant solution (2 g inoculant/500 mL water; Verdesian N-Dure ®, Cary, NC) was applied to the seed radicle. This inoculant is sold for use with peanut, cowpea, lespedeza, and mung bean. On milliliter of mycorrhizal inoculant solution (1 g inoculant/500 mL water; Wildroot Organic, Austin, TX) was also applied at transplant to the cowpeas assigned to mycorrhizae (Myc+) treatments. In all experiments, cowpeas were grown for 75 days in greenhouse conditions. Daily temperature ranged between 28 °C and 6 °C on average, and relative humidity between 52% and 86%. Soil pH measurements were taken initially upon planting (mean −8.0 ± 0.1) and monthly during the experiment to check for pH changes from nutrient solutions, but none were detected. For all 30 treatments, eight replicate cowpeas were grown for each treatment and a subsample of five were randomly chosen for data collection.
Pots were watered based on daily moisture measurements using a moisture meter (ProCheck 5TE, Pullman WA). Except where otherwise indicated below, the pots were watered with 150 mL of tap water (or the designated nutrient solution,
Table 2 and
Table A1) whenever their soil moisture fell below a lower threshold of 5%. This amount of water raised the soil moisture to an upper target of 15%. Tap water was used instead of deionized water in order to better simulate field conditions since both rainwater and local irrigation water sources carry trace minerals [
64,
65]. However, in the absence of specific soil tests for nutrients of interest, exact treatment impacts cannot be determined. Levels listed in
Table 3 should be considered lower thresholds. These lower thresholds (µg element/g dry soil) were calculated using the concentration for each nutrient solution, the total volume of solution applied over the course of each experiment, and the dry mass of the soil.
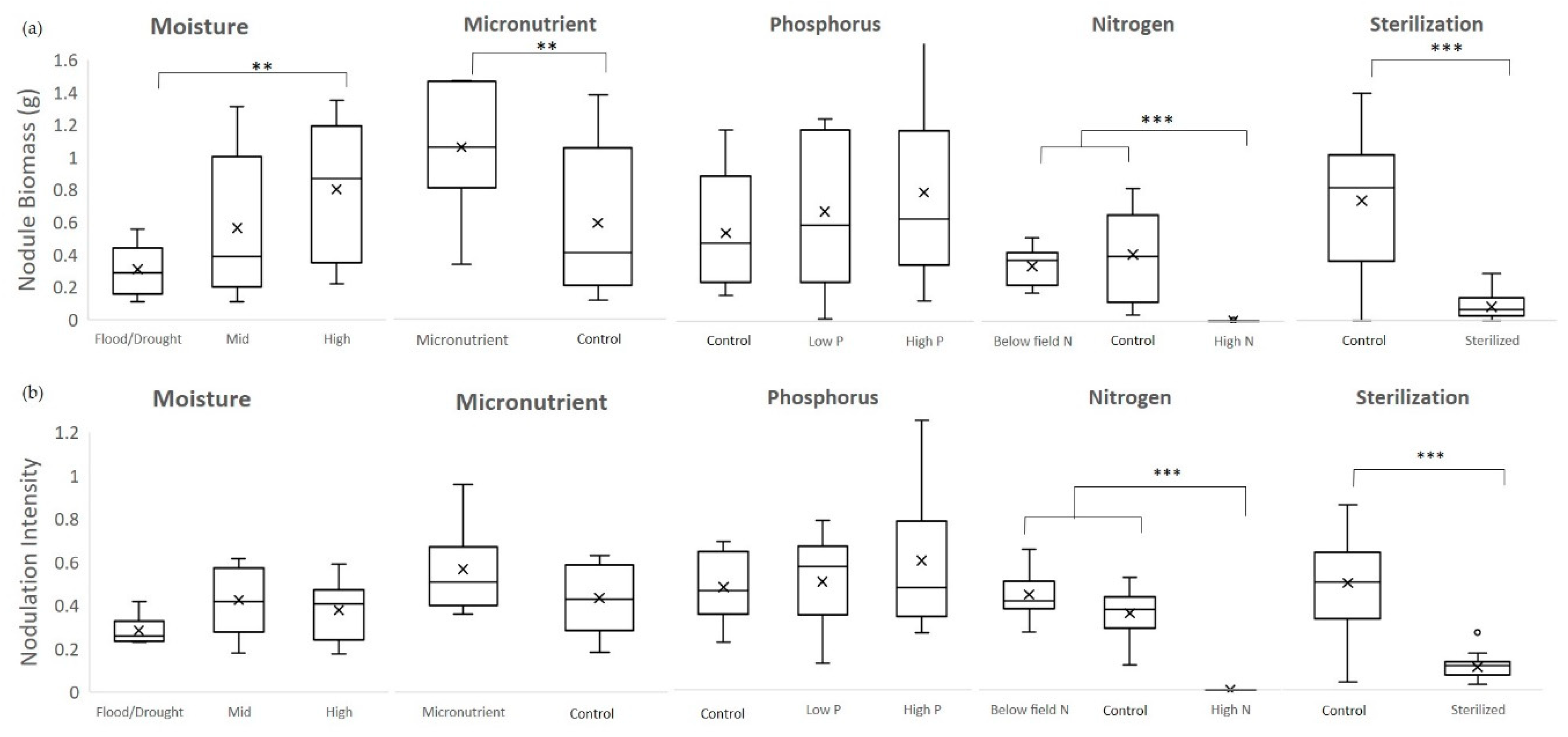
2.1.1. Moisture
Using a 3 × 2 factorial design, this experiment compared all combinations of three levels of moisture—high, mid, and cycle—and two levels of mycorrhizal inoculation—with (Myc+) or without (M–). Moisture levels were designated as high (soil moisture between 15%–25%), mid-range (soil moisture between 5%–15%) or saturation/drought cycle (between wilting point and field capacity). In the high moisture treatment, pots received 200 mL of tap water when they reached a lower threshold of 15% soil moisture which raised them to field saturation (around 25%). For the saturation/drought cycle, plants received 500 mL of tap water after 3 days below a threshold of 2.5% soil moisture. They were watered in two 250 mL increments to minimize leaching and runoff. The three-day wait was set based on the average time cowpeas took to wilt after reaching 2.5% soil moisture in a pre-trial assessment.
2.1.2. Micronutrients
Using a 2 × 2 factorial design, this experiment compared two levels of micronutrients—micronutrients added (Mi+) and field level (Control)—and two levels of mycorrhizal inoculation—Myc+ or Myc-. We compared impact of addition of copper (0.5 µM CuSO
4), cobalt (1.7 µM CoCl
2) boron (25 µM H
3BO
3), molybdenum (0.5 µM Na
2MoO
4), manganese (2 µM MnCl
2), and zinc (2 µM ZnSO
4) on root nodulation to a control with field level micronutrients (
Table 2). Micronutrient concentrations were based on a modified Hoagland’s solution [
62].
2.1.3. Phosphorus
Using a 3 × 2 factorial design, this experiment compared three levels of P—control, low, and high—and two levels of mycorrhizal inoculation (Myc+ or Myc-). The levels of P tested included field level P as a control, low P (0.1 mM KH
2PO
4, 1.9 mM KCl), and high P (2 mM KH
2PO
4). Since P (target nutrient) was supplied as KH
2PO
4, potassium levels (non-target) were also raised. To avoid confounding the impacts of P and K, low and field level treatments were supplemented with potassium chloride (KCl) to match the K levels applied to the high P treatment (
Table 2).
2.1.4. Nitrogen
Using a 3 × 2 factorial design, this experiment compared three levels of N—low, control, and high—and two levels of mycorrhizal inoculation (Myc+ or Myc-). We compared the impact on nodulation of field level N as a control (
Table 2) to N levels both higher (5 mM CaN
2O
6) and lower (1/2 field level). Higher N treatments had calcium nitrate added in solution while lower N was achieved through a 50/50 mix of field soil and sand. Since the high N treatment also received 5 mM Ca (non-target) in addition to 10 mM N (target), low and field level N treatments were supplemented with calcium chloride (5 mM CaCl
2) to match the calcium levels applied to the high N treatment (
Table 2). These adjustments were made to avoid confounding the impacts of Ca and N on nodulation.
2.1.5. Soil Sterilization
A sterilization experiment was included to isolate the effects of soil microbes impacting nodulation. For these treatments, soil media was steam sterilized in an autoclave at 121 °C for 30 min before planting. Using a 2 × 2 × 2 factorial design, this experiment compared all combinations of following three factors—sterilized (S+) or unsterilized (S-) soil, with (Myc+) or without (Myc-) mycorrhizal inoculation, with (R+) or without (R–) rhizobial inoculation. sterilization can affect soil pH as well as nutrient content and availability. Therefore, separate tests were conducted to determine the baseline pH and nutrient levels for the sterilized soil.
2.1.6. Data Collection
During the final week before termination (days 68–74), light-saturated photosynthesis measurements (Asat) were taken for three replicates from each treatment using a Portable Photosynthesis System (model LI-6400XT, LiCOR, Lincoln, NE, USA). Asat was recorded at 2000 umol m-2 s-1 PAR after the assimilation value had stabilized and the stomatal conductance value exceeded a threshold of 0.05 mol m-2 s-1. After 75 days, five replicates from each treatment were randomly chosen. Roots were cleaned and examined for nodules which were counted, weighed, and checked for internal color as an indicator of N fixation activity. Pink, red or brown nodules were counted as active while green, grey, tan, and any other color were considered inactive [
22]. Plants were then dried for at least 72 h at 70 °C and the dry biomass of root, stem, and leaf tissue for each plant were recorded.
2.1.7. Data Analysis
For the moisture, micronutrient, phosphorus, and nitrogen experiments, 2-way analyses of variance were conducted to compare the main effects of each factor and mycorrhizal inoculation and the interaction effect between that factor and mycorrhizae on nodule number, biomass, and activity and plant indicators including Asat, root, stem, leaf and total biomass, root to shoot ratio and nodule to plant biomass ratio. Multiple comparisons were performed using the Holm-Sidak method. When assumptions of normality and equal variance were violated, a Kruskal–Wallis 1-way ANOVA on ranks was employed, followed by Dunn’s method for multiple comparisons. For the sterilization experiment, three-way ANOVAs were conducted to compare the main effects of sterilization, mycorrhizal inoculation, and rhizobial inoculation and the interaction effects among the three (SYSTAT™, San Jose, CA).
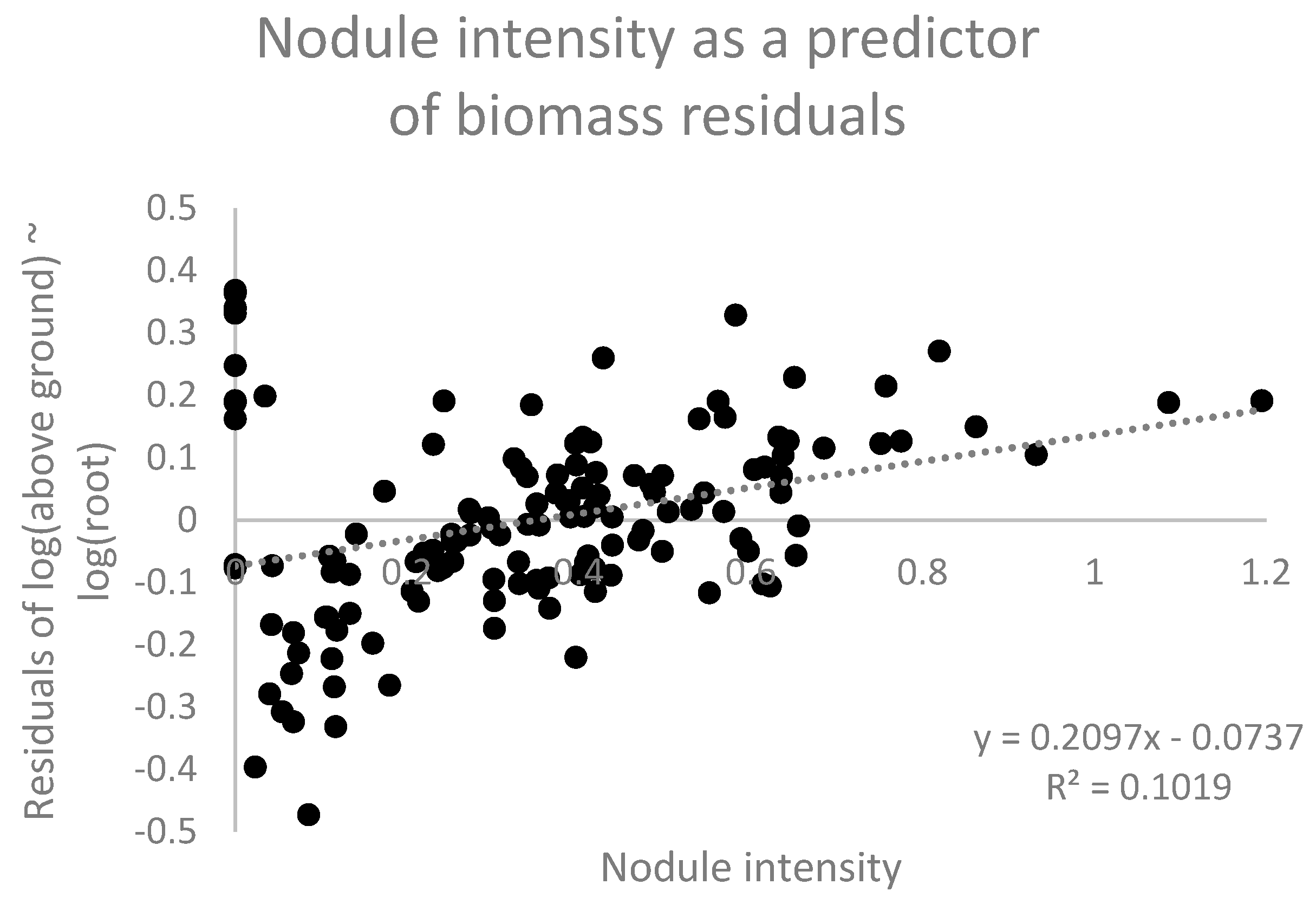
Nodulation intensity was calculated as the ratio of dry nodule biomass to dry root biomass, which is affected by both nodule number and mean nodule size. We tested whether nodulation intensity promotes enhanced leaf + stem development at a given plant size. To scale for the effects of plant size, we first regressed the aboveground biomass (stem and leaf) against root biomass and then investigated whether the residuals about this allometric relationship exhibited a positive relationship with nodulation intensity. Such a relationship would suggest that for plants of a given size, increased nodulation intensity promotes greater leaf development.
2.2. Micronutrient Experiment
In the second experiment of the series, developed based on the results of the first but following a modified protocol, Iron and Clay cowpea seeds (
Vigna unguiculata; Johnny’s Seeds, Winslow, ME) were surface sterilized through immersion in 2% hypochlorite solution for five minutes, followed by five rinses with sterile water. Two seeds were then planted into each 15 cm plastic pots with 1500 g of a 1:1 mixture of perlite and soil obtained from the field where nodulation failure occurred. Nutrient solutions and estimated treatment impact compared to field soil levels are in
Table 4.
At planting, a 1 mL solution of rhizobium inoculant solution (2 g inoculant/500 mL water; Verdesian Guard-N®, Cary, NC) was applied to the seed. Pots were thinned to one plant each after 7 days. Cowpeas were randomly assigned to one of 8 treatments that were watered with a nutrient solution of B, Co, Cu, Mn, Mo, or Zn individually, all 6 micronutrients together, or tap water (control). The plants were grown for 45 days in Percival Environmental Growth Chambers (Perry, Iowa) with 15 hours of light (PAR—440 µmol/m2/s) every 24 h. Light period temperatures were 27 °C, dark period temperatures were 24 °C, and relative humidity ranged between 45% and 70%. Pots were watered with 150 mL of the designated nutrient solution every three days for a total of 16 waterings (2.4 L solution/plant). Each treatment had 9 replicates for a total of 72 individuals.
Data were collected 45 days after seeding from 9 replicates for each treatment. Pre-termination measurements included spectral signatures (ASD Handheld 2, Malvern Panalytical, Longmont, CO) and chlorophyll content (SPAD 502 Chlorophyll Meter, Spectrum, Aurora, IL). After termination, roots were cleaned and examined for nodules which were counted, weighed, and checked for internal color as an indicator of N fixation activity. Plants were then dried for at least 72 h at 70 °C and the dry biomass of roots, stems, and leaves for each plant were recorded. One-way analyses of variance were used to examine differences among treatments (SYSTAT™, San Jose, CA).