A Field Study on the Prevalence of and Risk Factors for Endoparasites in Beef Suckler Cow Herds in Germany
Abstract
:1. Introduction
2. Materials and Methods
2.1. Farm Selection and Animals
2.2. Sample Collection
2.3. Statistical Analysis
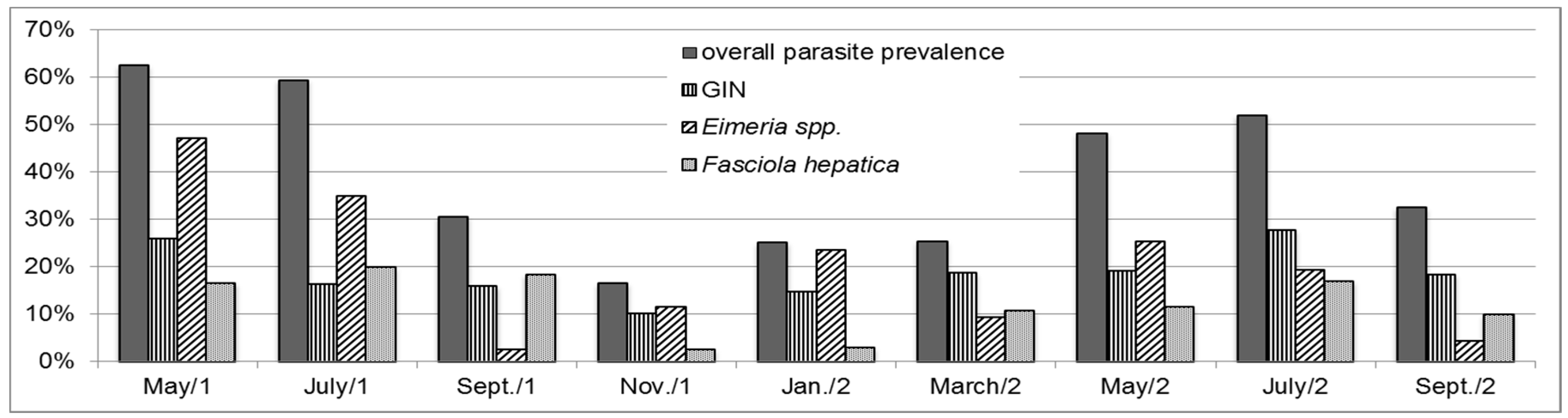
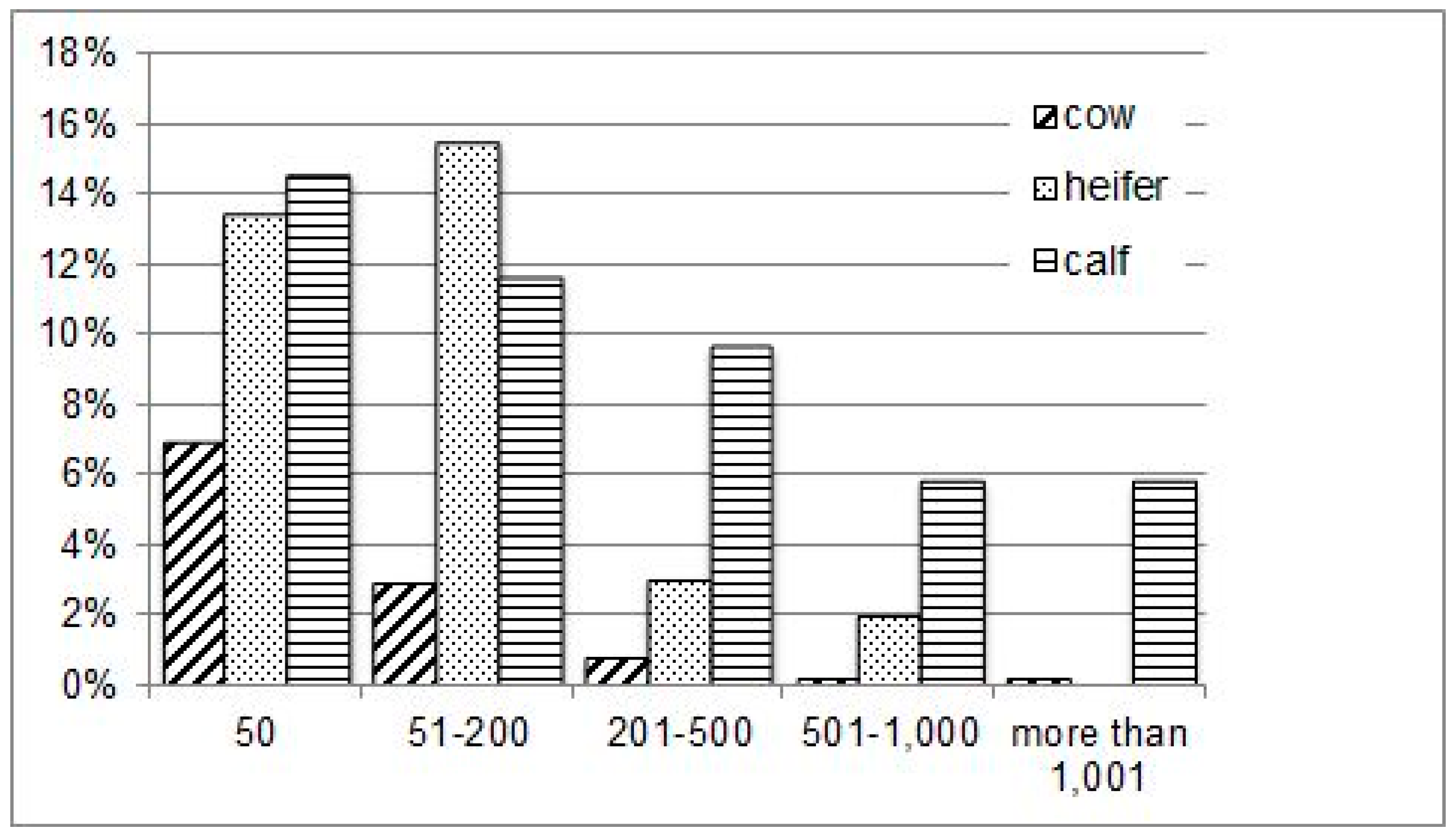
3. Results
3.1. General Parasitological Status
3.2. Eimeria spp.
3.3. Gastrointestinal Nematodes (GIN)
3.4. Fasciola Hepatica
3.5. Other Parasites
3.6. Deworming Strategies and Management
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- DE-Statis, Statistisches Bundesamt. Available online: https://www.destatis.de/DE/ZahlenFakten/Wirtschaftsbereiche/LandForstwirtschaftFischerei/TiereundtierischeErzeugung/Tabellen/BetriebeRinderBestand.html (accessed on 5 August 2018).
- Conner, S.; Oppenheim, D. Demand for pasture-raised livestock products in Michigan: Results of consumer surveys and experimental auctions. J. Food Distr. Res. 2008, 39, 45–50. [Google Scholar]
- Fernqvist, F.; Ekelund, L. Credence and the effect on consumer liking of food—A review. Food Qual. Prefer. 2014, 32, 340–353. [Google Scholar] [CrossRef]
- Gemma, L.; Charlton, S.; Rutter, M. The behaviour of housed dairy cattle with and without pasture access—A review. Appl. Anim. Behav. Sci. 2017, 192, 2–9. [Google Scholar]
- Demeler, J.; Van Zeveren, A.M.J.; Kleinschmidt, N.; Vercruysse, J.; Höglund, J.; Koopmann, R.; Cabaret, J.; Claerbout, E.; Areskog, M.; von Samson-Himmelstjerna, G. Monitoring the efficacy of ivermectin and albendazole against gastro intestinal nematodes of cattle in Northern Europe. Vet. Parasitol. 2009, 160, 109–115. [Google Scholar] [CrossRef] [PubMed]
- Hallmann, C.A.; Sorg, M.; Jongejans, E.; Siepel, H.; Hofland, N.; Schwan, H.; Stenmans, W.; Müller, A.; Sumser, H.; Hörren, T.; et al. More than 75 percent decline over 27 years in total flying insect biomass in protected areas. PLoS ONE 2017, 12, e0185809. [Google Scholar] [CrossRef] [PubMed]
- Strong, L.; Wall, R.; Woolford, A.; Djeddour, D. The effect of feacally excreted ivermectin and fenbendazol on the insect colonization of cattle dung following the oral administration of sustained-release boluses. Vet. Parasitol. 1995, 62, 253–266. [Google Scholar] [CrossRef]
- Beynon, S.A.; Peck, M.; Mann, D.J.; Lewis, O.T. Consequences of alternative and conventional endoparasite control in cattle for dung—Associated invertebrates and ecosystem functioning. Agric. Ecosyst. Environ. 2012, 162, 36–44. [Google Scholar] [CrossRef]
- Voison, A. Grass Productivity; Philosophical Library Inc.: New York, NY, USA, 1959. [Google Scholar]
- Vercruysse, J.; Dorny, P. Integrated control of nematode infection in cattle. A reality? A need? A future? Int. J. Parasitol. 1999, 29, 165–175. [Google Scholar] [CrossRef]
- Fahrenkrog, J.; Koopmann, R.; von Samson-Himmelsterna, G.; Demeler, J. Targeted Selective Treatment (TST) bei Rindern zur Reduzierung der Resistenzbildung bei Endoparasiten. In Landbauforschung Sonderheft; Johann Heinrich von Thünen-Institut: Braunschweig, Germany, 2011; Volume 346, pp. 71–76. [Google Scholar]
- Ploeger, H.W.; Van Doorn, D.C.K.; Nijsse, E.R.; Eysker, M. Decision trees on the web—A parasite compendium. Trends Parasitol. 2008, 24, 203–204. [Google Scholar] [CrossRef] [PubMed]
- Shaw, D.J.; Vercruysse, J.; Claerebout, E.; Dorny, P. Gastrointestinal nematode infections of first-grazing season calves in Western Europe: General patterns and the effect of chemoprophylaxis. Vet. Parasitol. 1998, 75, 115–131. [Google Scholar] [CrossRef]
- Kemper, N.; Henze, C. Effects of pasture’s re-wetting on endoparasites in cattle in northern Germany. Vet. Parasitol. 2009, 161, 302–306. [Google Scholar] [CrossRef] [PubMed]
- Tander, F. Untersuchungen zum Vorkommen und zur Epidemiologie von Endoparasitosen bei Kühen in verschiedenen Haltungssystemen. Ph.D. Thesis, Ludwig-Maximiliam-University, Munich, Germany, 2004. [Google Scholar]
- Schmäschke, R. Die Koproskopische Diagnostik von Endoparasiten in der Veterinärmedizin; Schlütersche: Berlin, Germany, 2014. [Google Scholar]
- Deinhofer, G. Weidemanagement: Möglichkeiten in der Parasitenregulation bei Rind und Kleinen Wiederkäuern. In Proceedings of the Parasitologische Fachtagung für Biologische Landwirtschaft, Raumberg-Gumpenstein, Austria, 22 March 2009; pp. 9–13. [Google Scholar]
- Hawkins, J.A. Economic benefits of parasite control in cattle. Vet. Parasitol. 1993, 46, 159–173. [Google Scholar] [CrossRef]
- Rommel, M. Protozoen. In Veterinärmedizinische Parasitologie, 4th ed.; Boch, J., Supperer, R., Eds.; Verlag Paul Parey: Berlin/Hamburg, Germany, 1992; pp. 109–174. [Google Scholar]
- Heile, C. Orientierende Untersuchungen zur Parasitenkontrolle bei Mutterkuhherden auf unterschiedlichen Standorten in Brandenburg. Ph.D. Thesis, Free University, Berlin, Germany, 1999. [Google Scholar]
- Bürger, H.-J. Helminthen. In Veterinärmedizinische Parasitologie, 4th ed.; Boch, J., Supperer, R., Eds.; Verlag Paul Parey: Berlin/Hamburg, Germany, 1992; pp. 174–322. [Google Scholar]
- Armour, J. The epidemiology of helminth disease in farm animals. Vet. Parasitol. 1980, 6, 7–46. [Google Scholar] [CrossRef]
- Connan, R.J. Effect of lactation on the immune response to gastro-intestinal nematodes. Vet. Rec. 1976, 99, 476–477. [Google Scholar] [CrossRef] [PubMed]
- Schwörling, J. Erprobung des “Targeted Selective Treatment“ mit Levamisol zur Endoparasitenbekämpfung bei Lämmern. Ph.D. Thesis, University of Veterinary Medicine, Hannover, Germany, 2016. [Google Scholar]
- Precision Grazing. Available online: http://www.precisiongrazing.com/technograzingtm/ (accessed on 5 August 2018).
- Knubben-Schweizer, G.; Scheuerle, M.; Pfister, K. Die bekämpfung des großen leberegels beim rind. Tierarztl Prax Großtiere 2011, 3, 179–185. [Google Scholar]
- Rommel, M.; Schnieder, T. Neue Anthelminthika und neue Behandlungssysteme zur Behandlung von Weideparasitosen der Rinder. Angew. Parasitol. 1989, 30, 101–109. [Google Scholar] [PubMed]
- Christensen, N.; Nansen, P.; Frandsen, F. The influence of temperature on the infectivity of Fasciola hepatica miracidia to Lymnea truncatula. J. Parasitol. 1976, 62, 698–701. [Google Scholar] [CrossRef] [PubMed]
- Ploeger, H.W.; Borgsteede, F.H.M.; Sol, J.; Mirck, M.H.; Huyben, M.W.; Kooyman, F.N.J.; Eysker, M. Cross-sectional serological survey on gastrointestinal and loung nematode infections in first and second-year replacement stock in the Netherlands: Relation with management practise and use of anthelmintics. Vet. Parasitol. 2000, 36, 57–70. [Google Scholar] [CrossRef]
- Schnieder, T.; Bellmer, A.; Tenter, A.M. Seroepidemiologigal study on Dictyocaulus viviparus infections of first grazing cattle in Northern Germany. Vet. Parasitol. 1993, 47, 289–300. [Google Scholar] [CrossRef]
- Holst, C.R. Untersuchungen zum Einfluss nematophager Pilze auf das Nematoden-Infektionsrisiko bei Schafen und Ziegen. Ph.D. Thesis, University of Veterinary Medicine, Hannover, Germany, 2005. [Google Scholar]
- Hull, J. Ducks and the control of liver fluke in sheep. Vet. Rec. 2017, 181, 270–271. [Google Scholar] [CrossRef] [PubMed]
- Albers, G.A.A.; Gray, G.D. Breeding for worm resistance: A perspective. Int. J. Parasitol. 1987, 12, 559–566. [Google Scholar] [CrossRef]
| Parameter | Farm A | Farm B | Farm C | Farm D | Farm E |
|---|---|---|---|---|---|
| Region | Central Germany | Southern Germany | Southern Germany | German Baltic coastal region | Central Germany |
| Absolute altitude (m) | 111 | 450 | 456 | 65 | 400 |
| Average temperature (°C) 1 | 11.5 | 12.3 | 12.3 | 11.7 | 12.5 |
| Average rainfall (l/m2/month) 1 | 59.4 | 48.6 | 48.6 | 45.2 | 65.7 |
| Production | organic | organic | organic | conventional | conventional |
| Herd size (adult cows) | 182 | 48 | 71 | 411 | 77 |
| Pasture size (hectare) | 96 | 27 | 20 | 250 | 93 |
| Pasture category | wet | dry | dry | wet | wet |
| Grazing interval | 2–12 weeks | no rotation | no rotation | 3–6 weeks | 3–6 weeks |
| Housing in winter season | barn | barn | barn | barn | weather protection |
| Number of sampled calves | 21 | 25 | 17 | 17 | 23 |
| Number of sampled heifers | 25 | 12 | 13 | 30 | 17 |
| Number of sampled cows | 126 | 53 | 64 | 115 | 118 |
| Not classified | 5 | 0 | 1 | 20 | 6 |
| Total number of sampled animals | 177 | 90 | 95 | 182 | 164 |
| Age Status | OR | 95% CI | p-Value | |
|---|---|---|---|---|
| General parasitological status | calf | 2.5 | 1.4–4.2 | <0.001 |
| heifer | 2.0 | 1.1–3.5 | ||
| adult | 1.0 | - | ||
| Eimeria spp. | calf | 4.5 | 2.5–7.9 | <0.0001 |
| heifer | 3.0 | 1.7–5.6 | ||
| adult | 1.0 | - | ||
| GIN | calf | 2.1 | 1.3–3.6 | <0.01 |
| heifer | 1.9 | 1.1–3.2 | ||
| adult | 1.0 | - | ||
| Fasciola hepatica | calf | 0.2 | 0.1–0.5 | <0.001 |
| heifer | 0.3 | 0.1–0.7 | ||
| adult | 1.0 | - |
| Occurrence of Parasite Species in % | Farm A 1 (n = 177) | Farm B 1 (n = 90) | Farm C 1 (n = 95) | Farm D (n = 182) | Farm E (n = 164) |
|---|---|---|---|---|---|
| Capillaria spp. | 0.00 | 0.00 | 1.05 | 0.55 | 0.61 |
| Dicrocoelium spp. | 0.00 | 0.00 | 2.11 | 0.55 | 0.61 |
| Dictyocaulus viviparus | 0.00 | 2.22 | 2.11 | 0.55 | 17.68 |
| Eimeria spp. | 22.60 | 18.89 | 16.84 | 23.08 | 17.07 |
| Fasciola hepatica | 12.43 | 0.00 | 1.05 | 9.34 | 32.32 |
| GIN | 21.02 | 20.00 | 15.79 | 19.78 | 15.85 |
| Moniezia spp. | 3.95 | 4.44 | 2.11 | 1.65 | 2.44 |
| Nematodirus spp. | 0.00 | 1.11 | 0.00 | 0.00 | 0.00 |
| Calicophoron spp. | 1.02 | 0.00 | 0.00 | 0.55 | 4.88 |
| Protostrongylus spp. | 0.00 | 0.00 | 0.00 | 0.00 | 0.00 |
| Strongyloides spp. | 0.65 | 0.00 | 1.05 | 1.65 | 1.83 |
| Trichuris spp. | 0.00 | 0.00 | 0.00 | 0.55 | 0.00 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gillandt, K.; Stracke, J.; Hohnholz, T.; Waßmuth, R.; Kemper, N. A Field Study on the Prevalence of and Risk Factors for Endoparasites in Beef Suckler Cow Herds in Germany. Agriculture 2018, 8, 132. https://doi.org/10.3390/agriculture8090132
Gillandt K, Stracke J, Hohnholz T, Waßmuth R, Kemper N. A Field Study on the Prevalence of and Risk Factors for Endoparasites in Beef Suckler Cow Herds in Germany. Agriculture. 2018; 8(9):132. https://doi.org/10.3390/agriculture8090132
Chicago/Turabian StyleGillandt, Kathia, Jenny Stracke, Tatiana Hohnholz, Ralf Waßmuth, and Nicole Kemper. 2018. "A Field Study on the Prevalence of and Risk Factors for Endoparasites in Beef Suckler Cow Herds in Germany" Agriculture 8, no. 9: 132. https://doi.org/10.3390/agriculture8090132
APA StyleGillandt, K., Stracke, J., Hohnholz, T., Waßmuth, R., & Kemper, N. (2018). A Field Study on the Prevalence of and Risk Factors for Endoparasites in Beef Suckler Cow Herds in Germany. Agriculture, 8(9), 132. https://doi.org/10.3390/agriculture8090132






