Quality and Nutritional Evaluation of Regina Tomato, a Traditional Long-Storage Landrace of Puglia (Southern Italy)
Abstract
1. Introduction
2. Materials and Methods
2.1. Location and Cropping Details
2.2. Physical Analysis
2.3. Samples Preparation for Chemical Analysis
2.4. Isoprenoids Analysis
2.5. Ascorbic Acid Determination
2.6. Glucose and Fructose Assay, and Sweetness Index
2.7. Starch Determination
2.8. Inorganic Cation Contents
2.9. Statistical Analysis
3. Results and Discussion
3.1. Physical Traits at Harvest
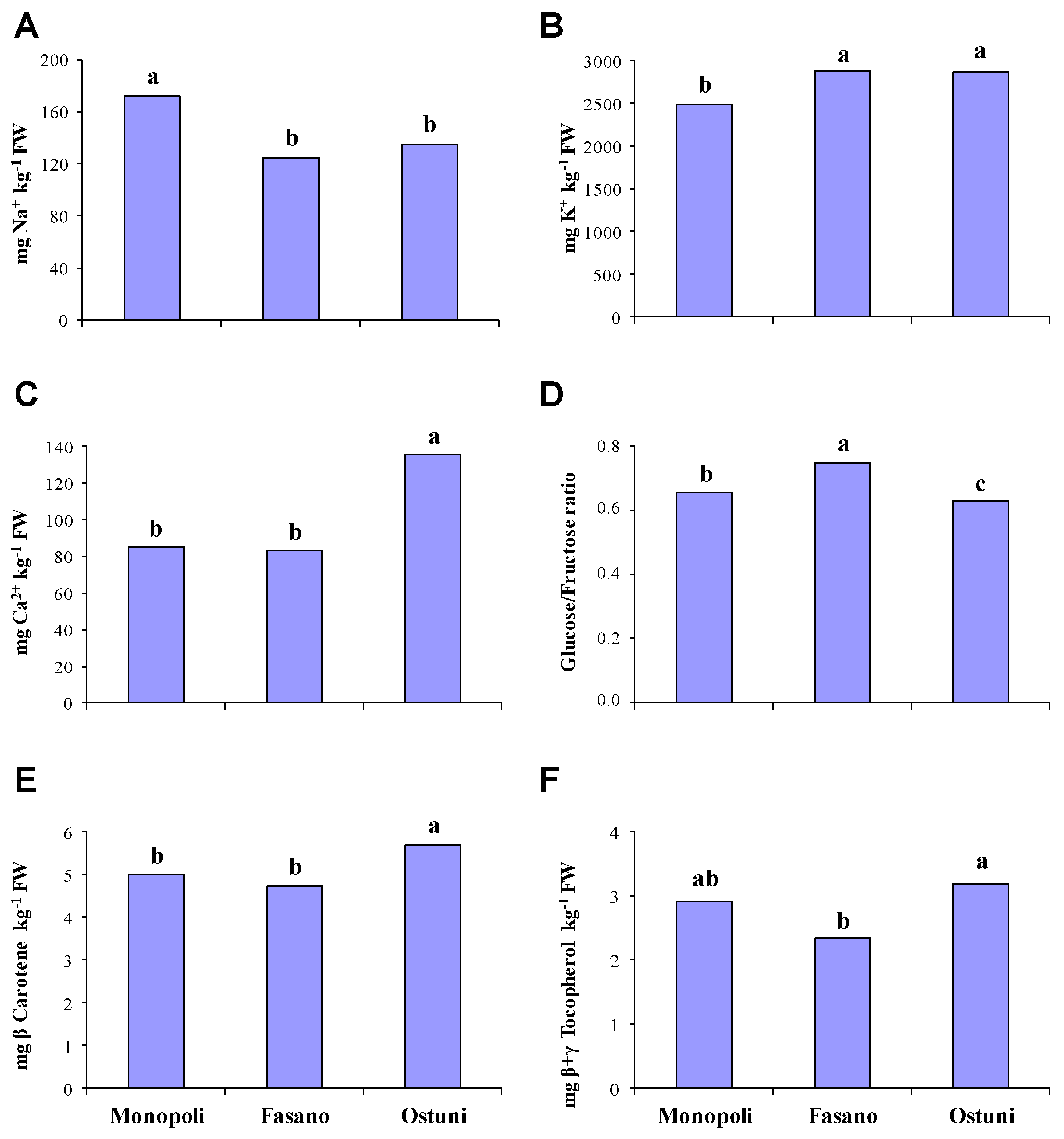
3.2. Dry Matter and Inorganic Cation Contents
3.3. Sugars, Starch Assay and Sweetness Index
3.4. Isoprenoids Content
3.5. Tocopherols and Ascorbic Acid Content
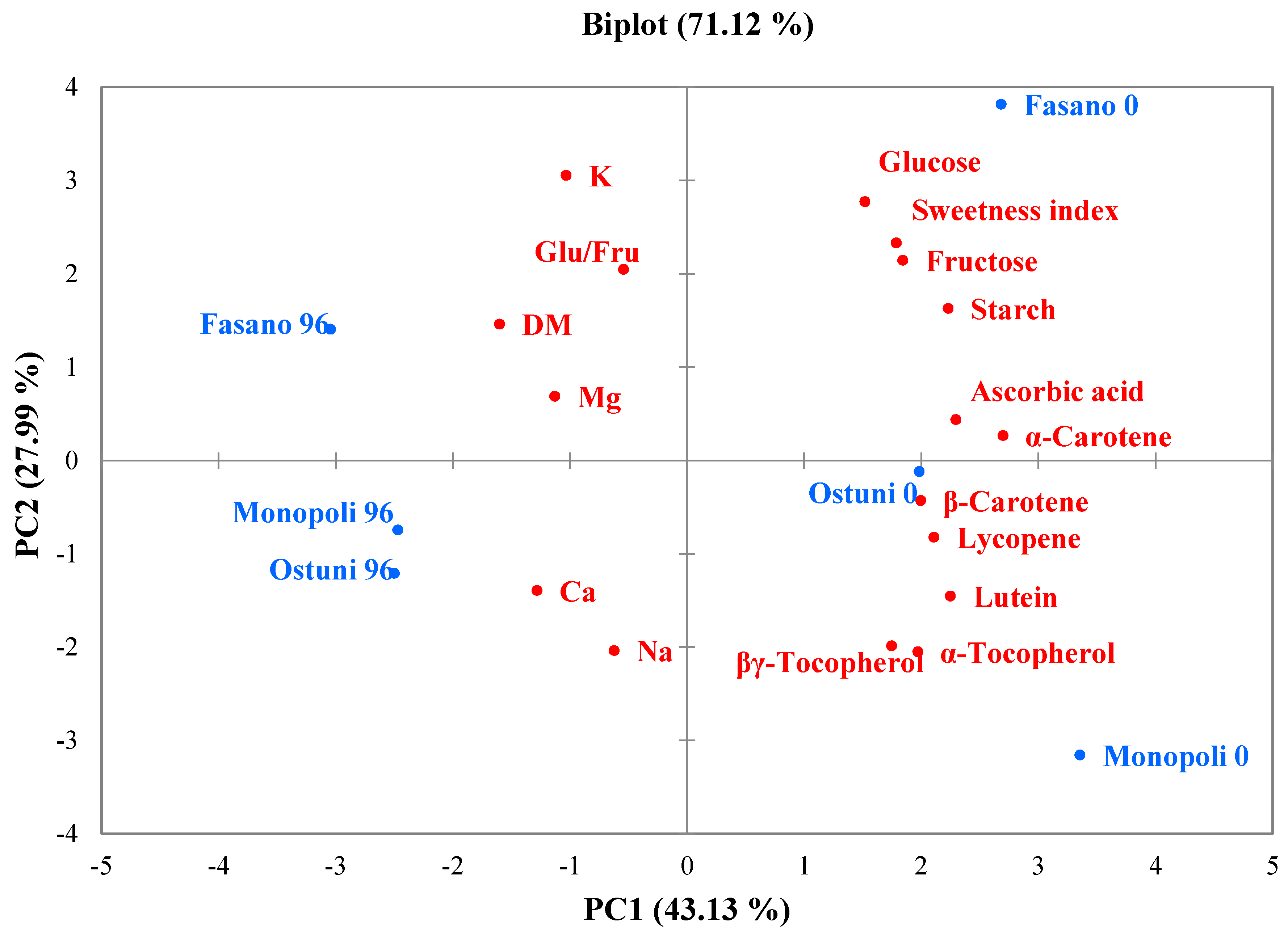
3.6. Principal Component Analysis
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Naranjo, R.D.D.P.; Otaiza, S.; Saragusti, A.C.; Baroni, V.; Carranza, A.D.V.; Peralta, I.E.; Valle, E.M.; Carrari, F.; Asis, R. Hydrophilic antioxidants from Andean tomato landraces assessed by their bioactivities in vitro and in vivo. Food Chem. 2016, 206, 146–155. [Google Scholar] [CrossRef] [PubMed]
- Baldina, S.; Picarella, M.E.; Troise, A.D.; Pucci, A.; Ruggieri, V.; Ferracane, R.; Barone, A.; Fogliano, V.; Mazzucato, A. Metabolite profiling of Italian tomato landraces with different fruit types. Front. Plant Sci. 2016, 7, 664. [Google Scholar] [CrossRef] [PubMed]
- Mazzucato, A.; Ficcadenti, N.; Caioni, M.; Mosconi, P.; Piccinini, E.; Reddy Sanampudi, V.R.; Sestili, S.; Ferrari, V. Genetic diversity and distinctiveness in tomato (Solanum lycopersicum L.) landraces: The Italian case study of “A pera Abruzzese”. Sci. Hortic. 2010, 125, 55–62. [Google Scholar] [CrossRef]
- Muratore, G.; Licciardello, F.; Maccarone, E. Evaluation of the chemical quality of a new type of small-sized tomato cultivar, the plum tomato (Lycopersicon lycopersicum). Ital. J. Food Sci. 2005, 17, 75–81. [Google Scholar]
- Renna, M.; Rinaldi, V.A.; Gonnella, M. The Mediterranean Diet between traditional foods and human health: The culinary example of Puglia (Southern Italy). Int. J. Gastron. Food Sci. 2015, 2, 63–71. [Google Scholar] [CrossRef]
- Elia, A.; Santamaria, P. Biodiversity in vegetable crops, a heritage to save: The case of Puglia region. Ital. J. Agron. 2013, 8, 4. [Google Scholar] [CrossRef]
- Slow Food Torre Canne Regina Tomato—Presìdi Slow Food—Slow Food Foundation. Available online: https://www.fondazioneslowfood.com/en/slow-food-presidia/torre-canne-regina-tomato/ (accessed on 3 May 2018).
- Siracusa, L.; Patanè, C.; Avola, G.; Ruberto, G. Polyphenols as chemotaxonomic markers in Italian “long-storage” tomato genotypes. J. Agric. Food Chem. 2011, 60, 309–314. [Google Scholar] [CrossRef] [PubMed]
- Patanè, C.; Pellegrino, A.; Di Silvestro, I. Effects of calcium carbonate application on physiology, yield and quality of field-grown tomatoes in a semi-arid Mediterranean climate. Crop Pasture Sci. 2018, 69, 411–418. [Google Scholar] [CrossRef]
- Siracusa, L.; Patanè, C.; Rizzo, V.; Cosentino, S.L.; Ruberto, G. Targeted secondary metabolic and physico-chemical traits analysis to assess genetic variability within a germplasm collection of “long storage” tomatoes. Food Chem. 2018, 244, 275–283. [Google Scholar] [CrossRef] [PubMed]
- Naranjo, R.D.D.P.; Otaiza, S.; Saragusti, A.C.; Baroni, V.; Carranza, A.V.; Peralta, I.E.; Valle, E.M.; Carrari, F.; Asis, R. Data on polyphenols and biological activity analyses of an Andean tomato collection and their relationships with tomato traits and geographical origin. Data Brief 2016, 7, 1258–1268. [Google Scholar] [CrossRef] [PubMed]
- Figàs, M.R.; Prohens, J.; Raigón, M.D.; Fita, A.; García-Martínez, M.D.; Casanova, C.; Borràs, D.; Plazas, M.; Andújar, I.; Soler, S. Characterization of composition traits related to organoleptic and functional quality for the differentiation, selection and enhancement of local varieties of tomato from different cultivar groups. Food Chem. 2015, 187, 517–524. [Google Scholar] [CrossRef] [PubMed]
- López Camelo, A.F.; Gómez, P.A. Comparison of color indexes for tomato ripening. Hortic. Bras. 2004, 22, 534–537. [Google Scholar] [CrossRef]
- Sadler, G.; Davis, J.; Dezman, D. Rapid extraction of lycopene and β-carotene from reconstituted tomato paste and pink grapefruit homogenates. J. Food Sci. 1990, 55, 1460–1461. [Google Scholar] [CrossRef]
- Perkins-Veazie, P.; Collins, J.K.; Pair, S.D.; Roberts, W. Lycopene content differs among red-fleshed watermelon cultivars. J. Sci. Food Agric. 2001, 81, 983–987. [Google Scholar] [CrossRef]
- Fraser, P.D.; Pinto, M.E.S.; Holloway, D.E.; Bramley, P.M. Application of high-performance liquid chromatography with photodiode array detection to the metabolic profiling of plant isoprenoids. Plant J. 2008, 24, 551–558. [Google Scholar] [CrossRef]
- Ferreira, I.C.F.R.; Aires, E.; Barreira, J.C.M.; Estevinho, L.M. Antioxidant activity of Portuguese honey samples: Different contributions of the entire honey and phenolic extract. Food Chem. 2009, 114, 1438–1443. [Google Scholar] [CrossRef]
- Caretto, S.; Parente, A.; Serio, F.; Santamaria, P. Influence of potassium and genotype on vitamin E content and reducing sugar of tomato fruits. HortScience 2008, 43, 2048–2051. [Google Scholar]
- Crespo, P.; Bordonaba, J.G.; Terry, L.A.; Carlen, C. Characterisation of major taste and health-related compounds of four strawberry genotypes grown at different Swiss production sites. Food Chem. 2010, 122, 16–24. [Google Scholar] [CrossRef]
- D’Imperio, M.; Renna, M.; Cardinali, A.; Buttaro, D.; Serio, F.; Santamaria, P. Calcium biofortification and bioaccessibility in soilless “baby leaf” vegetable production. Food Chem. 2016, 213, 149–156. [Google Scholar] [CrossRef] [PubMed]
- Serio, F.; Ayala, O.; Bonasia, A.; Santamaria, P. Antioxidant properties and health benefits of tomato. In Progress in Medicinal Plants—Search for Natural Drugs; Studium Press LLC: Houston, TX, USA, 2006; Volume 13, pp. 159–179. ISBN 9780976184959. [Google Scholar]
- Siracusa, L.; Avola, G.; Patanè, C.; Riggi, E.; Ruberto, G. Re-evaluation of traditional Mediterranean foods. The local landraces of “Cipolla di Giarratana” (Allium cepa L.) and long-storage tomato (Lycopersicon esculentum L.): Quality traits and polyphenol content. J. Sci. Food Agric. 2013, 93, 3512–3519. [Google Scholar] [CrossRef] [PubMed]
- Barbagallo, R.N.; Chisari, M.; Branca, F.; Spagna, G. Pectin methylesterase, polyphenol oxidase and physicochemical properties of typical long-storage cherry tomatoes cultivated under water stress regime. J. Sci. Food Agric. 2008, 88, 389–396. [Google Scholar] [CrossRef]
- Arias, R.; Lee, T.C.; Logendra, L.; Janes, H. Correlation of lycopene measured by HPLC with the L*, a*, b* color readings of a hydroponic tomato and the relationship of maturity with color and lycopene content. J. Agric. Food Chem. 2000, 48, 1697–1702. [Google Scholar] [CrossRef] [PubMed]
- Yahia, E.M.; Brecht, J. Tomatoes. In Crop Post-Harvest: Science and Technology: Perishables; Rees, D., Farrell, G.O.J., Eds.; Wiley-Blackwell: Oxford, UK, 2012; pp. 5–23. ISBN 0632057254. [Google Scholar]
- Elia, A.; Santamaria, P.; Colelli, G.; De Boni, A.; Ventrella, D.; Serio, F. Produzione, qualità e attitudine alla conservazione di popolazioni di pomodoro da serbo. Risultati preliminari. In Atti Giornate Scientifiche SOI; Società Orticola Italiana: Sesto Fiorentino, Italy, 1992; pp. 406–407. [Google Scholar]
- Serio, F.; De Gara, L.; Caretto, S.; Leo, L.; Santamaria, P. Influence of an increased NaCl concentration on yield and quality of cherry tomato grown in posidonia (Posidonia oceanica L. Delile). J. Sci. Food Agric. 2004, 84, 1885–1890. [Google Scholar] [CrossRef]
- Nielsen, F.H. Trace mineral deficiencies. In Handbook of Nutrition and Food; CRC Press, Taylor & Francis: Boca Raton, FL, USA, 2008; pp. 159–176. ISBN 9781420008890. [Google Scholar]
- Chapagain, B.P.; Wiesman, Z. Effect of potassium magnesium chloride in the fertigation solution as partial source of potassium on growth, yield and quality of greenhouse tomato. Sci. Hortic. 2004, 99, 279–288. [Google Scholar] [CrossRef]
- USDA Food Composition Databases Show Foods—Tomatoes, Red, Ripe, Raw, Year Round Average. Available online: https://ndb.nal.usda.gov/ndb/foods/show/302115?manu=&fgcd=&ds=SR&q=Tomatoes, red, ripe, raw, year round average (accessed on 3 May 2018).
- Elia, A.; Serio, F.; Parente, A.; Santamaria, P.; Ruiz Rodriguez, G. Electrical conductivity of nutrient solution, plant growth and fruit quality of soilless grown tomato. Acta Hortic. 2001, 503–508. [Google Scholar] [CrossRef]
- European Food Safety Authority Tolerable Upper Intake Levels for Vitamins and Minerals. Available online: http://www.efsa.europa.eu/sites/default/files/efsa_rep/blobserver_assets/ndatolerableuil.pdf (accessed on 3 May 2018).
- Kader, A.A. Flavor quality of fruits and vegetables. J. Sci. Food Agric. 2008, 88, 1863–1868. [Google Scholar] [CrossRef]
- Bornet, F.R.; Costagliola, D.; Rizkalla, S.W.; Blayo, A.; Fontvieille, A.M.; Haardt, M.J.; Letanoux, M.; Tchobroutsky, G.; Slama, G. Insulinemic and glycemic indexes of six starch-rich foods taken alone and in a mixed meal by type 2 diabetics. Am. J. Clin. Nutr. 1987, 45, 588–595. [Google Scholar] [CrossRef] [PubMed]
- Beckles, D.M. Factors affecting the postharvest soluble solids and sugar content of tomato (Solanum lycopersicum L.) fruit. Postharvest Biol. Technol. 2012, 63, 129–140. [Google Scholar] [CrossRef]
- George, B.; Kaur, C.; Khurdiya, D.S.; Kapoor, H.C. Antioxidants in tomato (Lycopersium esculentum) as a function of genotype. Food Chem. 2004, 84, 45–51. [Google Scholar] [CrossRef]
- Lenucci, M.S.; Caccioppola, A.; Durante, M.; Serrone, L.; De Caroli, M.; Piro, G.; Dalessandro, G. Carotenoid content during tomato (Solanum lycopersicum L.) fruit ripening in traditional and high-pigment cultivars. Ital. J. Food Sci. 2009, 21, 461–472. [Google Scholar]
- Ilahy, R.; Hdider, C.; Lenucci, M.S.; Tlili, I.; Dalessandro, G. Antioxidant activity and bioactive compound changes during fruit ripening of high-lycopene tomato cultivars. J. Food Compos. Anal. 2011, 24, 588–595. [Google Scholar] [CrossRef]
- Hart, D.J.; Scott, K.J. Development and evaluation of an HPLC method for the analysis of carotenoids in foods, and the measurement of the carotenoid content of vegetables and fruits commonly consumed in the UK. Food Chem. 1995, 54, 101–111. [Google Scholar] [CrossRef]
- Raiola, A.; Tenore, G.; Barone, A.; Frusciante, L.; Rigano, M. Vitamin E content and composition in tomato fruits: Beneficial roles and bio-fortification. Int. J. Mol. Sci. 2015, 16, 29250–29264. [Google Scholar] [CrossRef] [PubMed]
- Seybold, C.; Frohlich, K.; Bitsch, R.; Otto, K.B.V. Changes in contents of carotenoids and vitamin E during tomato processing. J. Agric. Food Chem. 2004, 5, 7005–7010. [Google Scholar] [CrossRef] [PubMed]
- Durante, M.; Montefusco, A.; Marrese, P.P.; Soccio, M.; Pastore, D.; Piro, G.; Mita, G.; Lenucci, M.S. Seeds of pomegranate, tomato and grapes: An underestimated source of natural bioactive molecules and antioxidants from agri-food by-products. J. Food Compos. Anal. 2017, 63, 65–72. [Google Scholar] [CrossRef]
- Soto-Zamora, G.; Yahia, E.M.; Brecht, J.K.; Gardea, A. Effects of postharvest hot air treatments on the quality and antioxidant levels in tomato fruit. LWT—Food Sci. Technol. 2005, 38, 657–663. [Google Scholar] [CrossRef]
- Harris, J.R. Ascorbic Acid: Biochemistry and Biomedical Cell Biology; Plenum Press: New York, NY, USA, 1996; ISBN 1461303257. [Google Scholar]
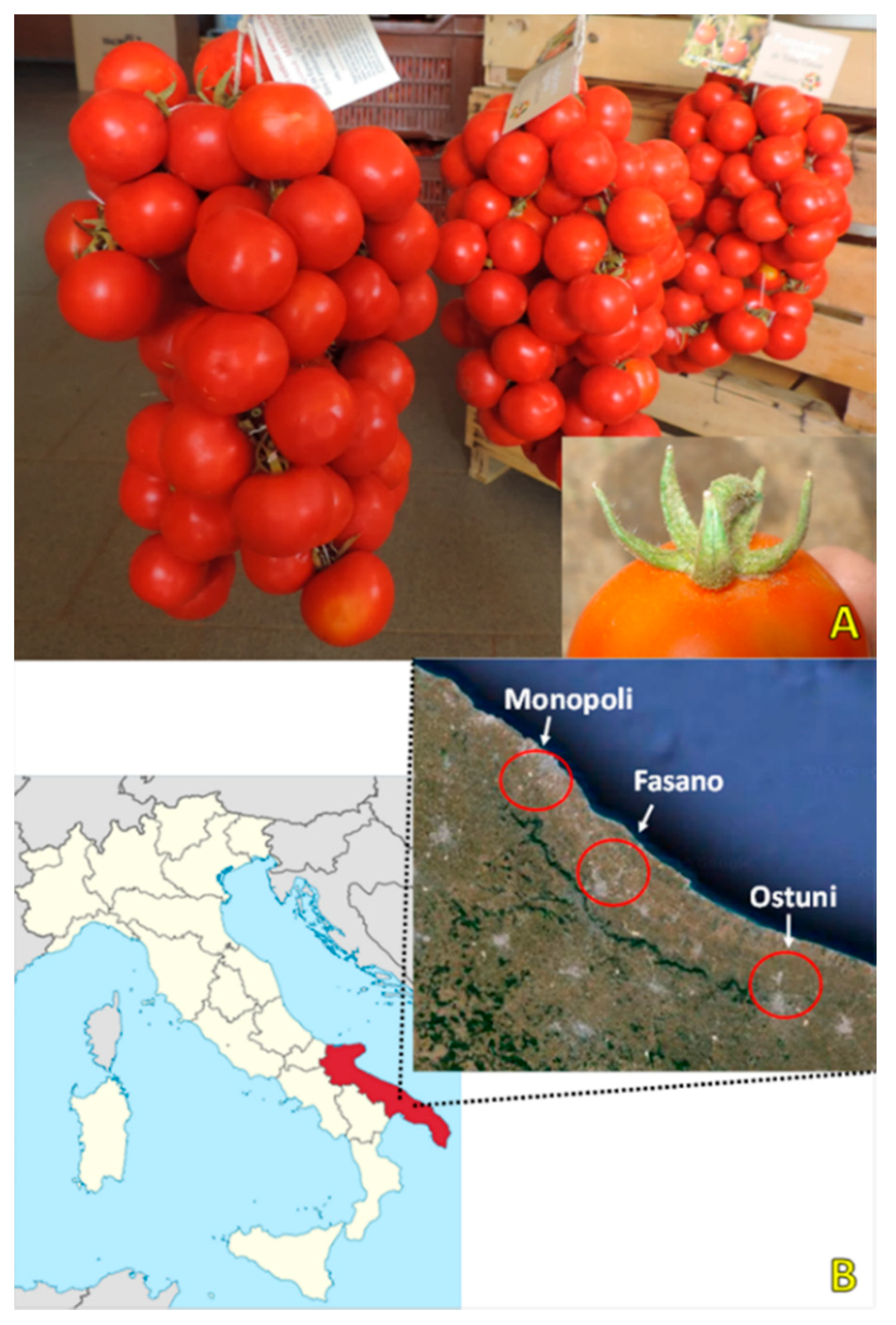
| Ecotype | Value | Fruit Weight (g) | Fruit Diameter (mm) | TSS (°Brix) | L* | a* | b* | h° | C | Colour Index | a*/b* | |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Pol | Equ | |||||||||||
| Monopoli | Mean | 21.5 | 30.0 | 34.8 | 7.0 b | 47.0 a | 32.2 | 41.0 a | 51.7 a | 52.3 a | 26.5 b | 0.8 b |
| SD | 3.7 | 2.1 | 1.9 | 0.3 | 2.7 | 3.0 | 4.4 | 5.2 | 2.3 | 4.5 | 0.2 | |
| Fasano | Mean | 20.1 | 29.4 | 34.0 | 6.4 c | 40.0 b | 32.3 | 30.3 b | 43.1 b | 44.4 b | 36.6 a | 1.1 b |
| SD | 3.3 | 1.5 | 2.1 | 0.1 | 2.8 | 4.3 | 4.6 | 1.9 | 6.1 | 3.2 | 0.1 | |
| Ostuni | Mean | 25.0 | 30.2 | 34.8 | 7.6 a | 38.1 b | 34.4 | 32.8 b | 43.6 b | 47.5 ab | 38.0 a | 1.1 b |
| SD | 6.3 | 2.6 | 4.4 | 0.3 | 1.6 | 2.8 | 2.6 | 1.5 | 3.6 | 1.9 | 0.1 | |
| Significance 1 | ns | ns | ns | ** | *** | ns | ** | ** | * | *** | ** | |
| Ecotype | Days of Storage | Value | Dry Matter | Na+ | K+ | Ca2+ | Mg2+ | Glucose | Fructose | Glu/Fru Ratio | Sweetness Index | Starch |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| g 100 g−1 FW | mg·kg−1 FW | g·kg−1 FW | mg·100 g−1 FW | |||||||||
| Monopoli | 0 | Mean | 6.24 d | 164.6 | 2546 | 83.3 | 105.1 b | 5.9 | 9.0 | 0.66 | 2.7 | 3.5 |
| SD | 0.18 | 11.4 | 14 | 22.7 | 3.7 | 1.6 | 2.3 | 0.02 | 0.7 | 0.3 | ||
| 96 | Mean | 6.80 c | 178.9 | 2448 | 86.6 | 108.8 ab | 5.7 | 8.6 | 0.66 | 2.6 | 1.9 | |
| SD | 0.18 | 26.9 | 114 | 17.7 | 14.2 | 0.5 | 0.7 | 0.01 | 0.2 | 0.1 | ||
| Fasano | 0 | Mean | 7.09 b | 117.7 | 2838 | 74.1 | 115.5 a | 9.3 | 13.0 | 0.71 | 3.9 | 4.3 |
| SD | 0.06 | 15.2 | 89 | 20.8 | 9.0 | 0.2 | 0.3 | 0.01 | 0.1 | 0.5 | ||
| 96 | Mean | 7.28 b | 132.0 | 2909 | 91.9 | 116.0 a | 6.3 | 8.0 | 0.79 | 2.5 | 2.9 | |
| SD | 0.07 | 5.0 | 92 | 0.9 | 0.4 | 0.5 | 0.7 | 0.01 | 0.2 | 0.1 | ||
| Ostuni | 0 | Mean | 7.07 b | 119.3 | 2759 | 108.2 | 100.5 b | 6.9 | 10.8 | 0.64 | 3.2 | 3.4 |
| SD | 0.06 | 6.4 | 173 | 12.4 | 10.1 | 1.1 | 1.5 | 0.01 | 0.4 | 0.5 | ||
| 96 | Mean | 7.66 a | 150.6 | 2966 | 163.6 | 118.3 a | 5.4 | 8.7 | 0.62 | 2.5 | 2.2 | |
| SD | 0.23 | 18.5 | 74 | 45.8 | 4.1 | 0.9 | 1.2 | 0.02 | 0.4 | 0.1 | ||
| Significance 1 | ||||||||||||
| Ecotype (E) | ** | * | ** | * | * | ns | ns | ** | ns | * | ||
| Day of storage (D) | ns | * | ns | ns | * | * | * | ns | * | ** | ||
| E × D | ** | ns | ns | ns | * | ns | ns | ns | ns | ns | ||
| Ecotype | Days of Storage | Value | Lutein | α-Carotene | β-Carotene | Lycopene | α Tocopherol | β + γ Tocopherol | Ascorbic Acid |
|---|---|---|---|---|---|---|---|---|---|
| mg·kg−1 FW | mg·g−1 FW | ||||||||
| Monopoli | 0 | Mean | 5.9 | 0.61 | 5.4 | 53.7 a | 25.2 a | 3.4 | 0.24 b |
| SD | 0.2 | 0.03 | 0.4 | 2.8 | 1.0 | 0.2 | 0.03 | ||
| 96 | Mean | 5.1 | 0.47 | 4.7 | 37.1 b | 19.0 b | 2.4 | 0.19 b | |
| SD | 0.3 | 0.10 | 0.4 | 1.5 | 2.0 | 0.4 | 0.01 | ||
| Fasano | 0 | Mean | 5.4 | 0.60 | 5.4 | 41.2 b | 20.1 b | 2.5 | 0.28 a |
| SD | 0.3 | 0.03 | 0.1 | 1.8 | 0.7 | 0.1 | 0.02 | ||
| 96 | Mean | 4.9 | 0.48 | 4.1 | 37.4 b | 19.3 b | 2.2 | 0.09 d | |
| SD | 0.4 | 0.08 | 0.1 | 2.3 | 0.5 | 0.1 | 0.01 | ||
| Ostuni | 0 | Mean | 5.2 | 0.58 | 6.2 | 41.3 b | 21.5 b | 3.5 | 0.20 b |
| SD | 0.4 | 0.03 | 0.3 | 0.4 | 1.1 | 0.6 | 0.02 | ||
| 96 | Mean | 5.1 | 0.49 | 5.2 | 27.3 c | 20.3 b | 2.8 | 0.14 c | |
| SD | 0.4 | 0.09 | 0.3 | 2.4 | 1.7 | 0.1 | 0.01 | ||
| Significance 1 | |||||||||
| Ecotype (E) | ns | ns | * | ** | * | * | * | ||
| Day of storage (D) | ns | * | ** | ** | ** | ** | *** | ||
| E × D | ns | ns | ns | * | ** | ns | ** | ||
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Renna, M.; Durante, M.; Gonnella, M.; Buttaro, D.; D’Imperio, M.; Mita, G.; Serio, F. Quality and Nutritional Evaluation of Regina Tomato, a Traditional Long-Storage Landrace of Puglia (Southern Italy). Agriculture 2018, 8, 83. https://doi.org/10.3390/agriculture8060083
Renna M, Durante M, Gonnella M, Buttaro D, D’Imperio M, Mita G, Serio F. Quality and Nutritional Evaluation of Regina Tomato, a Traditional Long-Storage Landrace of Puglia (Southern Italy). Agriculture. 2018; 8(6):83. https://doi.org/10.3390/agriculture8060083
Chicago/Turabian StyleRenna, Massimiliano, Miriana Durante, Maria Gonnella, Donato Buttaro, Massimiliano D’Imperio, Giovanni Mita, and Francesco Serio. 2018. "Quality and Nutritional Evaluation of Regina Tomato, a Traditional Long-Storage Landrace of Puglia (Southern Italy)" Agriculture 8, no. 6: 83. https://doi.org/10.3390/agriculture8060083
APA StyleRenna, M., Durante, M., Gonnella, M., Buttaro, D., D’Imperio, M., Mita, G., & Serio, F. (2018). Quality and Nutritional Evaluation of Regina Tomato, a Traditional Long-Storage Landrace of Puglia (Southern Italy). Agriculture, 8(6), 83. https://doi.org/10.3390/agriculture8060083








