Effect of High Pressure Processing on the Microbial Inactivation in Fruit Preparations and Other Vegetable Based Beverages
Abstract
1. Introduction
2. High Pressure Processing in the Food Industry
3. Fruit Preparations
4. Effects on Microorganisms and Viruses
5. Conclusions
Acknowledgments
Conflicts of Interest
References
- Patrignani, F.; Siroli, L.; Serrazanetti, D.I.; Gardini, F.; Lanciotti, R. Innovative strategies based on the use of essential oils and their components to improve safety, shelf-life and quality of minimally processed fruits and vegetables. Trends Food Sci. Tech. 2015, 46, 311–319. [Google Scholar] [CrossRef]
- Tumpanuvatr, T.; Jittanit, W. The temperature prediction of some botanical beverages, concentrated juices and purees of orange and pineapple during ohmic heating. J. Food Eng. 2012, 113, 226–233. [Google Scholar] [CrossRef]
- Mohideen, F.W.; Solval, K.M.; Li, J.; Zhang, J.; Chouljenko, A.; Chotiko, A.; Prudente, A.D.; Bankston, J.D.; Sathivel, S. Effect of continuous ultra-sonication on microbial counts and physico-chemical properties of blueberry (Vacciniumcorymbosum) juice. LWT-Food Sci. Technol. 2015, 60, 563–570. [Google Scholar] [CrossRef]
- Zhao, L.; Wang, S.; Liu, F.; Dong, P.; Huang, W.; Xiong, L.; Liao, X. Comparing the effects of high hydrostatic pressure and thermal pasteurization combined with nisin on the quality of cucumber juice drinks. Innov. Food Sci. Emerg. Technol. 2013, 17, 27–36. [Google Scholar] [CrossRef]
- Ferrario, M.; Alzamora, S.M.; Guerrero, S. Study of the inactivation of spoilage microorganisms in apple juice by pulsed light and ultrasound. Food Microbiol. 2015, 46, 635–642. [Google Scholar] [CrossRef] [PubMed]
- Abadias, M.; Alegre, I.; Usall, J.; Torres, R.; Vinas, I. Evaluation of alternative sanitizers to chlorine disinfection for reducing foodborne pathogens in fresh-cut apple. Postharvest Biol. Technol. 2011, 59, 289–297. [Google Scholar] [CrossRef]
- Drissner, D.; Zuercher, U. Safety of food and beverages: Fruits and vegetables. In Encyclopedia of Food Safety; Motarjemi, Y., Ed.; Academic Press: San Diego, CA, USA, 2014; pp. 253–259. [Google Scholar]
- Lerasle, M.; Duranton, F.; Simonin, H.; Federighi, M. Traitements par hautespressionshydrostatiques des denréesalimentaires: État de l’art. Rev. Méd. Vét. 2012, 163, 595–614. [Google Scholar]
- Yordanov, D.G.; Angelova, G.V. High Pressure Processing for Foods Preserving. Biotech. Biotechnol. Equip. 2010, 24, 1940–1945. [Google Scholar] [CrossRef]
- Ledward, D.A. High Pressure Processing of Foods; Nottingham University Press: Nottingham, UK, 1995; p. 1. [Google Scholar]
- Olsson, S. High Pressure Processing of Foods; Nottingham University Press: Nottingham, UK, 1995; p. 167. [Google Scholar]
- Farkas, D.; Hoover, D. High Pressure Processing. J. Food Sci. 2000, 65, 47–64. [Google Scholar] [CrossRef]
- Augustin, M.A.; Riley, M.; Stockmann, R.; Bennett, L.; Kahl, A.; Lockett, T.; Osmond, M.; Sanguansri, P.; Stonehouse, W.; Zajac, I.; et al. Role of food processing in food and nutrition security. Trends Food Sci. Technol. 2016, 56, 115–125. [Google Scholar] [CrossRef]
- Téllez-Luis, S.J.; Ramírez, J.A.; Pérez-Lamela, C.; Vázquez, M.; Simal-Gandara, J. Application of high hydrostatique pressure in the food preservation. CYTA J. Food 2001, 3, 66–80. [Google Scholar] [CrossRef][Green Version]
- Santhirasegaram, V.; Razali, Z.; Somasundram, C. Safety improvement of fruit juices by novel thermal and nonthermal processing. In Food Hygiene and Toxicology in Ready-to-Eat Foods; Elsevier: Amsterdam, The Netherlands, 2016; pp. 209–223. [Google Scholar]
- Borda, D.; Bleoanca, I.; Turtoi, M. Advancements in high pressure processing & applications in vegetal origin foods and food safety indicators. AUDJG Food Technol. 2013, 37, 18–34. [Google Scholar]
- Grauwet, T.; Rauh, C.; Van der Planken, I.; Vervoort, L.; Hendrickx, M.; Delgado, A.; Van Loey, A. Potential and limitations of methods for temperature mapping in high pressure thermal processing. Trends Food Sci. Technol. 2012, 23, 97–110. [Google Scholar] [CrossRef]
- Council of the European Union. Directive 2001/112/EC of 20 December 2001, Relating to Fruit Juices and Certain Similar Products Intended for Human Consumption. Available online: http://eur-lex.europa.eu/legal-content/EN/TXT/?uri=celex%3A32001L0112 (accessed on 29 August 2017).
- Council of the European Union. Directive 2001/113/EC of 20 December 2001, Relating to Fruit Jams, Jellies and Marmalades and Sweetened Chestnut Purée Intended for Human Consumption. Available online: http://eur-lex.europa.eu/legal-content/EN/ALL/?uri=CELEX%3A32001L0113 (accessed on 29 August 2017).
- Tonello-Samson, C. Advances in High Pressure Processing: Case Studies. In Proceedings of the IFT Annual Meeting & Food Expo, New Orleans, LA, USA, 21–24 June 2014. [Google Scholar]
- British Soft Drink Association. Fruit Juice Technical Guidance. 2016. Available online: http://www.britishsoftdrinks.com/write/MediaUploads/Publications/BSDA_-_FRUIT_JUICE_GUIDANCE_May_2016.pdf (accessed on 3 July 2017).
- Fernandes, L.; Casal, S.; Pereira, J.A.; Ramalhosa, E.; Saraiva, J.A. Effect of High Hydrostatic Pressure (HHP) Treatment on Edible Flowers’ Properties. Food Bioprocess Technol. 2017, 10, 799–807. [Google Scholar] [CrossRef]
- Gattegno, I. Hautespresions: Une progression en douceur. RIA 2017, 788, 48–50. [Google Scholar]
- Council of the European Union. Regulation (EC) No 852/2004 of the European Parliament and of the Council of 29 April, on the Hygiene of Foodstuffs. Available online: http://eur-lex.europa.eu/legal-content/EN/TXT/?uri=CELEX%3A32004R0852 (accessed on 29 August 2017).
- Council of the European Union. Regulation (EC) No 2073/2005 of the Commission of 15 November 2005 on Microbiological Criteria for Foodstuffs. Available online: http://eur-lex.europa.eu/legal-content/EN/ALL/?uri=CELEX%3A32005R2073 (accessed on 29 August 2017).
- Council of the European Union. Regulation (EC) No 1441/2007 of the Commission of 5 December 2007, Amending Regulation (EC) No 2073/2005 on Microbiological Criteria for Foodstuffs. Available online: http://eur-lex.europa.eu/legal-content/EN/TXT/?uri=CELEX:32007R1441 (accessed on 29 August 2017).
- FDA. Guidance for Industry: Juice HACCP Hazards and Controls Guidance First Edition. 3 March 2004. U.S. Department of Health and Human Services, Food and Drug Administration, Center for Food Safety and Applied Nutrition. Available online: https://www.fda.gov/food/guidanceregulation/guidancedocumentsregulatoryinformation/juice/ucm072557.htm (accessed on 3 July 2017).
- Timmermans, R.A.H.; Mastwijk, H.C.; Knol, J.J.; Quataert, M.C.J.; Vervoort, L.; Van der Plancken, I.; Hendrickx, M.E.; Matser, A.M. Comparing equivalent thermal, high pressure and pulsed electric field processes for mild pasteurization of orange juice. Part I: Impact on overall quality attributes. Innov. Food Sci. Emerg. Technol. 2011, 12, 235–243. [Google Scholar] [CrossRef]
- Lou, F.; Neetoo, H.; Chen, H.; Li, J. High hydrostatic pressure processing: A promising nonthermal technology to inactivate viruses in high-risk foods. Annu. Rev. Food Sci. Technol. 2015, 6, 389–409. [Google Scholar] [CrossRef] [PubMed]
- Sido, R.F.; Huang, R.; Liu, C.; Chen, H. High hydrostatic pressure inactivation of murine norovirus and human noroviruses on green onions and in salsa. Int. J. Food Microbiol. 2017, 242, 1–6. [Google Scholar] [CrossRef] [PubMed]
- Huang, R.; Li, X.; Huang, Y.; Chen, H. Strategies to enhance high pressure inactivation of murine norovirus in strawberry puree and on strawberries. Int. J. Food Microbiol. 2014, 185, 1–6. [Google Scholar] [CrossRef] [PubMed]
- Pan, H.; Buenconsejo, M.; Reineke, K.F.; Shieh, Y.C. Effect of process temperature on virus inactivation during high hydrostatic pressure processing of contaminated fruit puree and juice. J. Food Prot. 2016, 79, 1517–1526. [Google Scholar] [CrossRef] [PubMed]
- Lima Tribst, A.A.; de Souza Sant’Ana, A.; de Massaguer, P.R. Review: Microbiological quality and safety of fruit juices-past, present and future perspectives. Crit. Rev. Microbiol. 2009, 35, 310–319. [Google Scholar] [CrossRef] [PubMed]
- Corbo, M.R.; Bevilacqua, A.; Campaniello, D.; D’Amato, D.; Speranza, B.; Sinigaglia, M. Prolonging microbial shelf life of foods through the use of natural compounds and non-thermal approaches—A review. Int. J. Food Sci. Tech. 2009, 44, 223–241. [Google Scholar] [CrossRef]
- Torres, J.A.; Velazquez, G. Hydrostatic pressure processing of foods. In Food Processing Operations Modeling: Design and Analysis, 2nd ed.; CRC Press: Boca Raton, FL, USA, 2008; pp. 173–212. [Google Scholar]
- Terefe, N.S.; Buckow, R.; Versteeg, C. Quality-Related Enzymes in Fruit and Vegetable Products: Effects of Novel Food Processing Technologies, Part 1: High-Pressure Processing. Crit. Rev. Food Sci. Nutr. 2014, 54, 24–63. [Google Scholar] [CrossRef] [PubMed]
- Georget, E.; Sevenich, R.; Reineke, K.; Mathys, A.; Heinz, V.; Callanan, M.; Rauhc, C.; Knorr, D. Inactivation of microorganisms by high isostatic pressure processing in complex matrices: A review. Innov. Food Sci. Emerg. Technol. 2015, 27, 1–14. [Google Scholar] [CrossRef]
- Pinela, J.; Ferreira, I.C.F.R. Nonthermal physical technologies to decontaminate and extend the shelf-life of fruits and vegetables: Trends aiming at quality and safety. Crit. Rev. Food Sci. Nutr. 2017, 57, 2095–2111. [Google Scholar] [CrossRef] [PubMed]
- Fonberg-Broczek, M.; Windyga, B.; Szczawiński, J.; Szczawińska, M.; Pietrzak, D.; Prestamo, G. High pressure processing for food safety. ACTA Biochim. Pol. Engl. Edit. 2005, 52, 721–724. [Google Scholar]
- Shahbaz, H.M.; Yoo, S.; Seo, B.; Ghafoor, K.; Un Kim, Y.; Lee, D.U.; Park, J. Combination of TiO2-UVPhotocatalysis and High Hydrostatic Pressure to Inactivate Bacterial Pathogens and Yeast in Commercial Apple Juice. Food Bioprocess Technol. 2016, 9, 182–190. [Google Scholar] [CrossRef]
- Bayindirli, A.; Alpas, H.; Bozoglu, F.; Hizal, M. Efficiency of high pressure treatment on inactivation of pathogenic microorganisms and enzymes in apple, orange, apricot and sour cherry juices. Food Control 2006, 17, 52–58. [Google Scholar] [CrossRef]
- Shigehisa, T.; Ohmori, T.; Saito, A.; Taji, S.; Hayashi, R. Effects of high hydrostatic pressure on characteristics of pork slurries and inactivation of microorganisms associated with meat and meat products. Int. J. Food Microbiol. 1991, 12, 207–216. [Google Scholar] [CrossRef]
- Butz, P.; Trangott, V.; Ludwig, H.; Ries, J.; Weber, H. The high pressure inactivation of bacteria and bacterial spores. Die Pharm. 1990, 52, 487–491. [Google Scholar]
- Heinz, V.; Knorr, D. High pressure germination and inactivation kinetics of bacterial spores. In High Pressure Food Science, Bioscience and Chemistry; The Royal Society of Chemistry: Cambridge, UK, 1998; pp. 436–441. [Google Scholar]
- Basak, S.; Ramaswamy, H.S.; Piette, J.P.G. High pressure destruction kinetics of Leuconostocmesenteroides and Saccharomyces cerevisiae in single strength and concentrated orange juice. Innov. Food Sci. Emerg. Technol. 2002, 3, 223–231. [Google Scholar] [CrossRef]
- Rajkovic, A.; Smigic, N.; Devlieghere, F. Contemporary strategies in combating microbial contamination in food chain. Int. J. Food Microbiol. 2010, 141, S29–S42. [Google Scholar] [CrossRef] [PubMed]
- Goh, E.L.C.; Hocking, A.D.; Stewart, C.M.; Buckle, K.A.; Fleet, G.H. Baroprotective effect of increased solute concentrations on yeast and moulds during high pressure processing. Innov. Food Sci. Emerg. Technol. 2007, 8, 535–542. [Google Scholar] [CrossRef]
- Landl, A.; Abadias, M.; Sárraga, C.; Viñas, I.; Picouet, P.A. Effect of high pressure processing on the quality of acidified Granny Smith apple purée product. Innov. Food Sci. Emerg. Technol. 2010, 11, 557–564. [Google Scholar] [CrossRef]
- Hartyáni, P.; Dalmadi, I.; Knorr, D. Electronic nose investigation of Alicyclobacillusacidoterrestris inoculated apple and orange juice treated by high hydrostatic pressure. Food Control 2013, 32, 262–269. [Google Scholar] [CrossRef]
- Lee, S.Y.; Chung, H.J.; Kang, D.H. Combined treatment of high pressure and heat on killing spores of Alicyclobacillusacidoterrestris in apple juice concentrate. J. Food Prot. 2006, 69, 1056–1060. [Google Scholar] [CrossRef] [PubMed]
- Préstamo, G.; Sanz, P.D.; Fonberg-Broczek, M.; Arroyo, G. High pressure response of fruit jams contaminated with Listeria monocytogenes. Lett. Appl. Microbiol. 1999, 28, 313–316. [Google Scholar] [CrossRef] [PubMed]
- Li, R.; Wang, Y.; Wang, S.; Liao, X. A Comparative Study of Changes in Microbiological Quality and Physicochemical Properties of N2-Infused and N2-Degassed Banana Smoothies after High Pressure Processing. Food Bioprocess Technol. 2015, 8, 333–342. [Google Scholar] [CrossRef]
- Muñoz, M.; De Ancos, B.; Sánchez-Moreno, C.; Cano, M.P. Evaluation of chemical and physical (high-pressure and temperature) treatments to improve the safety of minimally processed mung bean sprouts during refrigerated storage. J. Food Prot. 2008, 69, 2395–2402. [Google Scholar] [CrossRef]
- Li, L.; Feng, L.; Yi, J.Y.; Hua, C.; Chen, F.; Liao, X.; Wang, Z.; Hu, X. High hydrostatic pressure inactivation of total aerobic bacteria, lactic acid bacteria, yeasts in sour Chinese cabbage. Int. J. Food Microbiol. 2010, 142, 180–184. [Google Scholar] [CrossRef] [PubMed]
- Moussa-Ayoub, T.E.; Jäger, H.; Knorr, D.; El-Samahy, S.K.; Kroh, L.W.; Rohn, S. Impact of pulsed electric fields, high hydrostatic pressure, and thermal pasteurization on selected characteristics of Opuntiadillenii cactus juice. LWT Food Sci. Technol. 2017, 79, 534–542. [Google Scholar] [CrossRef]
- Mukhopadhyay, S.; Sokorai, K.; Ukukub, D.; Fan, X.; Junej, V. Effect of high hydrostatic pressure processing on the background microbial loads and quality of cantaloupe puree. Food Res. Int. 2017, 91, 55–62. [Google Scholar] [CrossRef] [PubMed]
- Andrés, V.; Villanueva, M.J.; Tenorio, M.D. Influence of high pressure processing on microbial shelf life, sensory profile, soluble sugars, organic acids, and mineral content of milk-and soy-smoothies. LWT-Food Sci. Technol. 2016, 65, 98–105. [Google Scholar] [CrossRef]
- Lavinas, F.C.; Miguel, M.A.L.; Lopes, M.L.M.; Valentemesquita, V.L. Effect of High Hydrostatic Pressure on Cashew Apple (Anacardium occidentale L.) Juice Preservation. J. Food Sci. 2008, 73, M273–M277. [Google Scholar] [CrossRef] [PubMed]
- Pérez Pulido, R.; Toledo, J.; Grande, M.J.; Gálvez, A.; Lucas, R. Analysis of the effect of high hydrostatic pressure treatment and enterocin AS-48 addition on the bacterial communities of cherimoya pulp. Int. J. Food Microbiol. 2015, 196, 62–69. [Google Scholar] [CrossRef] [PubMed]
- Duong, T.; Balaban, M.; Perera, C.; Bi, X. Microbial and Sensory Effects of Combined High Hydrostatic Pressure and Dense Phase Carbon Dioxide Process on Feijoa Puree. J. Food Sci. 2015, 80, E2478–E2485. [Google Scholar] [CrossRef] [PubMed]
- Mert, M.; Buzrul, S.; Alpas, H. Effects of high hydrostatic pressure on microflora and some quality attributes of grape juice. High Press. Res. 2013, 33, 55–63. [Google Scholar] [CrossRef]
- Chai, C.; Lee, J.; Lee, Y.; Na, S.; Park, J. A combination of TiO2-UV photocatalysis and high hydrostatic pressure to inactivate Bacillus cereus in freshly squeezed Angelica keiskei juice. LWT-Food Sci. Technol. 2014, 55, 104–109. [Google Scholar] [CrossRef]
- Fernández-Sestelo, A.B.; Sendra de Saa, R.; Pérez Lamela, C.; Torrado-Agrasar, A.; Rúa, M.L.; Pastrana-Castro, L. Overall quality properties in pressurized kiwi purée: Microbial, physicochemical, nutritive and sensory test during refrigerated storage. Innov. Food Sci. Emerg. Technol. 2013, 20, 64–72. [Google Scholar] [CrossRef]
- Buzrul, S.; Alpas, H.; Largeteau, A.; Demazeau, G. Inactivation of Escherichia coli and Listeria innocua in kiwifruit and pineapple juices by high hydrostatic pressure. Int. J. Food Microbiol. 2008, 124, 275–278. [Google Scholar] [CrossRef] [PubMed]
- Carreño, J.M.; Gurrea, M.C.; Sampedro, F.; Carbonell, J.V. Effect of high hydrostatic pressure and high-pressure homogenisation on Lactobacillus plantarum inactivation kinetics and quality parameters of mandarin juice. Eur. Food Res. Technol. 2001, 232, 265–274. [Google Scholar] [CrossRef]
- Liu, F.; Li, R.; Wang, Y.; Bi, X.; Liao, X. Effects of high hydrostatic pressure and high-temperature short-time on mango nectars: Changes in microorganisms, acid invertase, 5-hydroxymethylfurfural, sugars, viscosity, and cloud. Innov. Food Sci. Emerg. Technol. 2014, 22, 22–30. [Google Scholar] [CrossRef]
- Liu, F.; Wang, Y.; Bi, X.; Guo, X.; Fu, S.; Liao, X. Comparison of Microbial Inactivation and Rheological Characteristics of Mango Pulp after High Hydrostatic Pressure Treatment and High Temperature Short Time Treatment. Food Bioprocess Technol. 2013, 6, 2675–2684. [Google Scholar] [CrossRef]
- Delgado-Adamez, J.; Franco, M.N.; Sánchez, J.; De Miguel, C.; Ramírez, M.R.; Martín-Vertedor, D. Comparative effect of high pressure processing and traditional thermal treatment on the physicochemical, microbiology, and sensory analysis of olive jam. Grasas y Aceites 2013, 64, 432–441. [Google Scholar] [CrossRef]
- Bull, M.K.; Zerdin, K.; Howe, E.; Goicoechea, D.; Paramanandhan, P.; Stockman, R.; Sellahewa, J.; Szabo, E.A.; Johnson, R.L.; Stewart, C.M. The effect of high pressure processing on the microbial, physical and chemical properties of Valencia and Navel orange juice. Innov. Food Sci. Emerg. Technol. 2004, 5, 135–149. [Google Scholar] [CrossRef]
- Katsaros, G.I.; Tsevdou, M.; Panagiotou, T.; Taoukis, P.S. Kinetic study of high pressure microbial and enzyme inactivation and selection of pasteurisation conditions for Valencia orange juice. Int. J. Food Sci. Technol. 2010, 45, 1119–1129. [Google Scholar] [CrossRef]
- Linton, M.; Mcclements, J.M.J.; Patterson, M.F. Inactivation of Escherichia coli O157:H7 in orange juice using a combination of high pressure and mild heat. J. Food Prot. 1999, 62, 277–279. [Google Scholar] [CrossRef] [PubMed]
- Polydera, A.C.; Stoforos, N.G.; Taoukis, P.S. Quality degradation kinetics of pasteurised and high pressure processed fresh Navel orange juice: Nutritional parameters and shelf life. Innov. Food Sci. Emerg. Technol. 2005, 6, 1–9. [Google Scholar] [CrossRef]
- Syed, Q.A.; Buffa, M.; Guamis, B.; Saldo, J. Effect of Compression and Decompression Rates of High Hydrostatic Pressure on Inactivation of Staphylococcus aureus in Different Matrices. Food Bioprocess Technol. 2014, 7, 1202–1207. [Google Scholar] [CrossRef]
- Yoo, S.; Ghafoor, K.; Un Kim, J.; Kim, S.; Jung, B.; Lee, D.U.; Park, J. Inactivation of Escherichia coli O157:H7 on orange fruit surfaces and in juice using Photocatalysis and High Hydrostatic Pressure. J. Food Prot. 2015, 78, 1098–1105. [Google Scholar] [CrossRef] [PubMed]
- Jordan, S.L.; Pascual, C.; Bracey, E.; Mackey, B.M. Inactivation and injury of pressure-resistant strains of Escherichia coli O157 and Listeria monocytogenes in fruit juices. J. Appl. Microbiol. 2001, 91, 463–469. [Google Scholar] [CrossRef] [PubMed]
- Erkmen, O.; Dogan, C. Effects of ultra high hydrostatic pressure on Listeria monocytogenes and natural flora in broth, milk and fruit juices. Int. J. Food Sci. Technol. 2004, 39, 91–97. [Google Scholar] [CrossRef]
- Guerrero-Beltrán, J.; Barbosa-Cánovas, G.V.; Welti-Chanes, J. High hydrostatic pressure effect on Saccharomyces cerevisiae, Escherichia coli and Listeria innocua in pear nectar. J. Food Qual. 2011, 34, 371–378. [Google Scholar] [CrossRef]
- Peñas, E.; Frias, J.; Gomez, R.; Vidal-Valverde, C. High hydrostatic pressure can improve the microbial quality of sauerkraut during storage. Food Control 2010, 21, 524–528. [Google Scholar] [CrossRef]
- Marszałek, K.; Mitek, M.; Skąpska, S. The effect of thermal pasteurization and high pressure processing at cold and mild temperatures on the chemical composition, microbial and enzyme activity in strawberry purée. Innov. Food Sci. Emerg. Technol. 2015, 27, 48–56. [Google Scholar] [CrossRef]
- Hsu, K.C.; Tan, F.J.; Chi, H.Y. Evaluation of microbial inactivation and physicochemical properties of pressurized tomato juice during refrigerated storage. LWT-Food Sci. Technol. 2008, 41, 367–375. [Google Scholar] [CrossRef]
- Krebbers, B.; Matser, A.M.; Hoogerwerf, S.W.; Moezelaar, R.; Tomassen, M.M.M.; van den Berg, R.W. Combined high-pressure and thermal treatments for processing of tomato puree: Evaluation of microbial inactivation and quality parameters. Innov. Food Sci. Emerg. Technol. 2003, 4, 377–385. [Google Scholar] [CrossRef]
- Aganovic, K.; Smetana, S.; Grauwet, T.; Toepfl, S.; Mathys, A.; Van Loey, A.; Heinz, V. Pilot scale thermal and alternative pasteurization of tomato and watermelon juice: An energy comparison and life cycle assessment. J. Clean. Prod. 2017, 141, 514–525. [Google Scholar] [CrossRef]
- Hurtado, A.; Guàrdia, M.D.; Picouet, P.; Jofré, A.; Ros, J.M.; Bañón, S. Stabilization of red fruit-based smoothies by high-pressure processing. Part A. Effects on microbial growth, enzyme activity, antioxidant capacity and physical stability. J. Sci. Food Agric. 2017, 97, 770–776. [Google Scholar] [CrossRef] [PubMed]
- Scolari, G.; Zacconi, C.; Busconi, M.; Lambri, M. Effect of the combined treatments of high hydrostatic pressure and temperature on Zygosaccharomycesbailii and Listeria monocytogenes in smoothies. Food Control 2015, 47, 166–174. [Google Scholar] [CrossRef]
- Araud, E.; DiCaprio, E.; Yang, Z.; Li, X.; Lou, F.; Hughes, J.H.; Chen, H.; Li, J. High-pressure inactivation of rotaviruses: Role of treatment temperature and strain diversity in virus inactivation. Appl. Environ. Microbiol. 2015, 81, 6669–6678. [Google Scholar] [CrossRef] [PubMed]
- Considine, K.M.; Kelly, A.L.; Fitzgerald, G.F.; Hill, C.; Sleator, R.D. High-pressure processing—Effects on microbial food safety and food quality. FEMS Microbiol. Lett. 2008, 281, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Peleg, M.; Cole, M. Reinterpretation of Microbial Survival Curves. Crit. Rev. Food Sci. Nutr. 1998, 38, 353–380. [Google Scholar] [CrossRef] [PubMed]
- Torres, J.A.; Pérez Lamela, C. Pressure processing of foods: Microbial inactivation and chemical changes in pressure-assisted thermal processing (PATP)-Part 2. AgroFOOD Ind. Hi-Tech 2008, 19, 34–36. [Google Scholar]
- Mújica-Paz, H.; Valdez-Fragoso, A.; Samson, C.T.; Welti-Chanes, J.; Torres, A. High-Pressure Processing Technologies for the Pasteurization and Sterilization of Foods. Food Bioprocess Technol. 2011, 4, 969–985. [Google Scholar] [CrossRef]
- Wang, Y.; Yi, J.; Yi, J.; Dong, P.; Hu, X.; Liao, X. Influence of Pressurization Rate and Mode on Inactivation of Natural Microorganisms in Purple Sweet Potato Nectar by High Hydrostatic Pressure. Food Bioprocess Technol. 2013, 6, 1570–1579. [Google Scholar] [CrossRef]
- Zwietering, M.H.; Jongenburger, I.; Rombouts, F.M.; Van’t Riet, K. Modeling of the Bacterial Growth Curve. Appl. Environ. Microbiol. 1990, 56, 1875–1881. [Google Scholar] [PubMed]
- Badhuri, S.; Smith, P.W.; Palumbo, S.A.; Turner-Jones, C.; Smith, J.; Marmer, B.; Buchanan, R.; Zaika, L.; Williams, A. Thermal destruction of Listeria monocytogenes in liver sausage slurry. Food Microbiol. 1991, 8, 75–78. [Google Scholar] [CrossRef]
- Linton, R.H.; Carter, W.H.; Pierson, M.D.; Hackney, C.R. Use of a Modified Gompertz Equation to Model Nonlinear Survival Curves for Listeria monocytogenes Scott A. J. Food Prot. 1995, 58, 946–954. [Google Scholar] [CrossRef]
- Buzrul, S.; Alpas, H. Modeling the synergistic effect of high pressure and heat on inactivation kinetics of Listeria innocua: A preliminary study. FEMS Microbiol. Lett. 2004, 238, 29–36. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Serment-Moreno, V.; Franco-Vega, A.; Escobedo-Avellaneda, Z.; Fuentes, C.; Torres, J.A.; Dibildox-Alvarado, E.; Welti-Chanes, J. The Logistic-Exponential Weibull Model as a Tool to Predict Natural Microflora Inactivation of Agave Mapsiaga Aguamiel (Agave Sap) by High Pressure Treatments. J. Food. Process. Preserv. 2017, 41, e12816. [Google Scholar] [CrossRef]
- Serment-Moreno, V.; Fuentes, C.; Torres, J.A.; Guerrero-Beltrán, J.A.; Welti-Chanes, J. A Gompertz Model Approach to Microbial Inactivation Kinetics by High-Pressure Processing Incorporating the Initial Counts, Microbial Quantification Limit, and Come-Up Time Effects. Food Bioprocess Technol. 2017, 10, 1495–1508. [Google Scholar] [CrossRef]
- Serment-Moreno, V.; Torres, J.A.; Fuentes, C.; Ríos-Alejandro, J.G.; Barbosa-Cánovas, G.; Welti-Chanes, J. Limitations of the log-logistic model for the analysis of sigmoidal microbial inactivation data for high pressure processing (HPP). Food Bioprocess Technol. 2016, 9, 901–916. [Google Scholar] [CrossRef]
- Cole, M.B.; Davies, K.W.; Munro, G.; Holyaok, C.D.; Kilsby, D.C. A vitalistic model to describe the thermal inactivation of Listeria monocytogenes. J. Ind. Microbiol. Biotechnol. 1993, 12, 232–239. [Google Scholar]


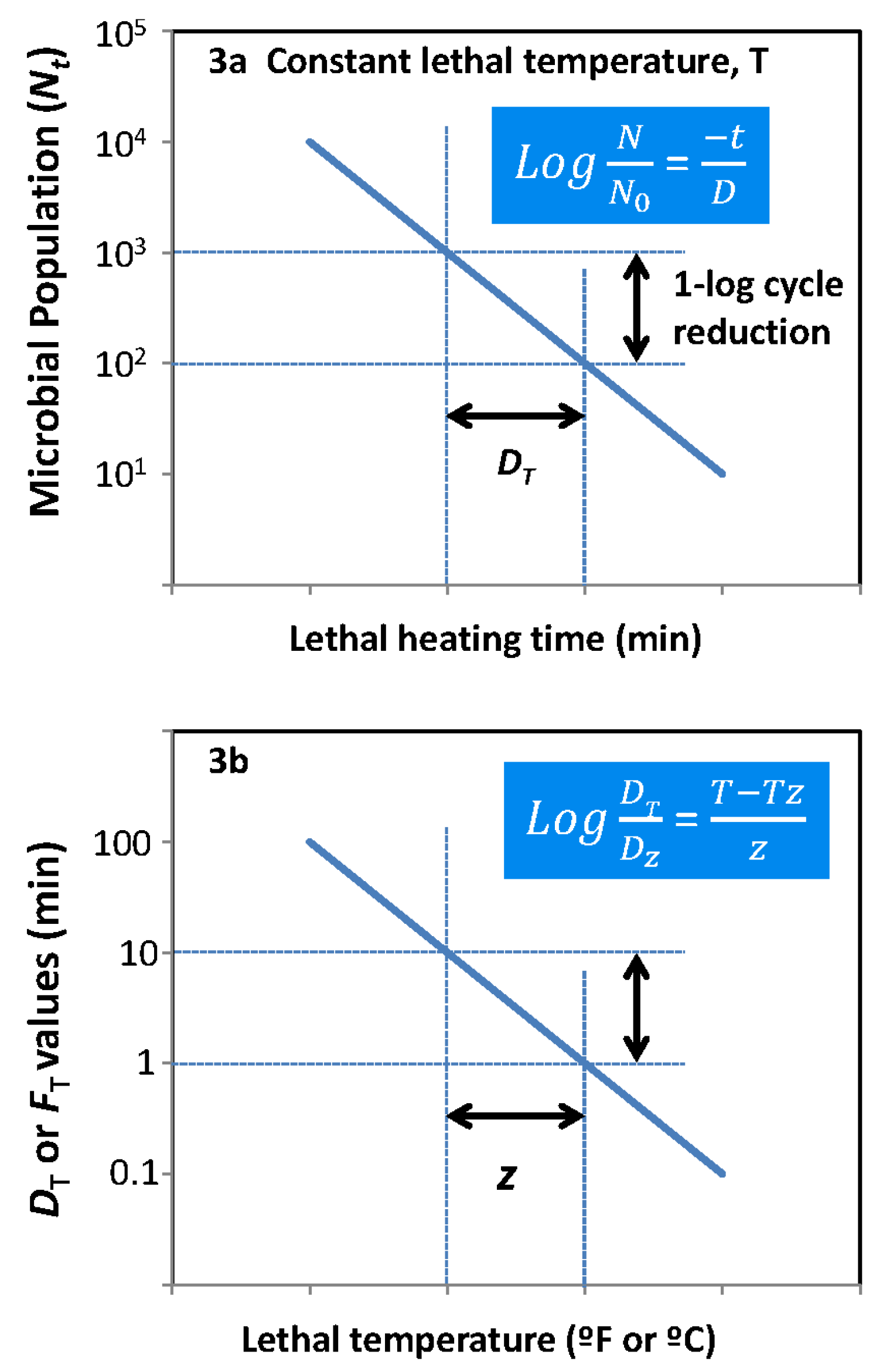
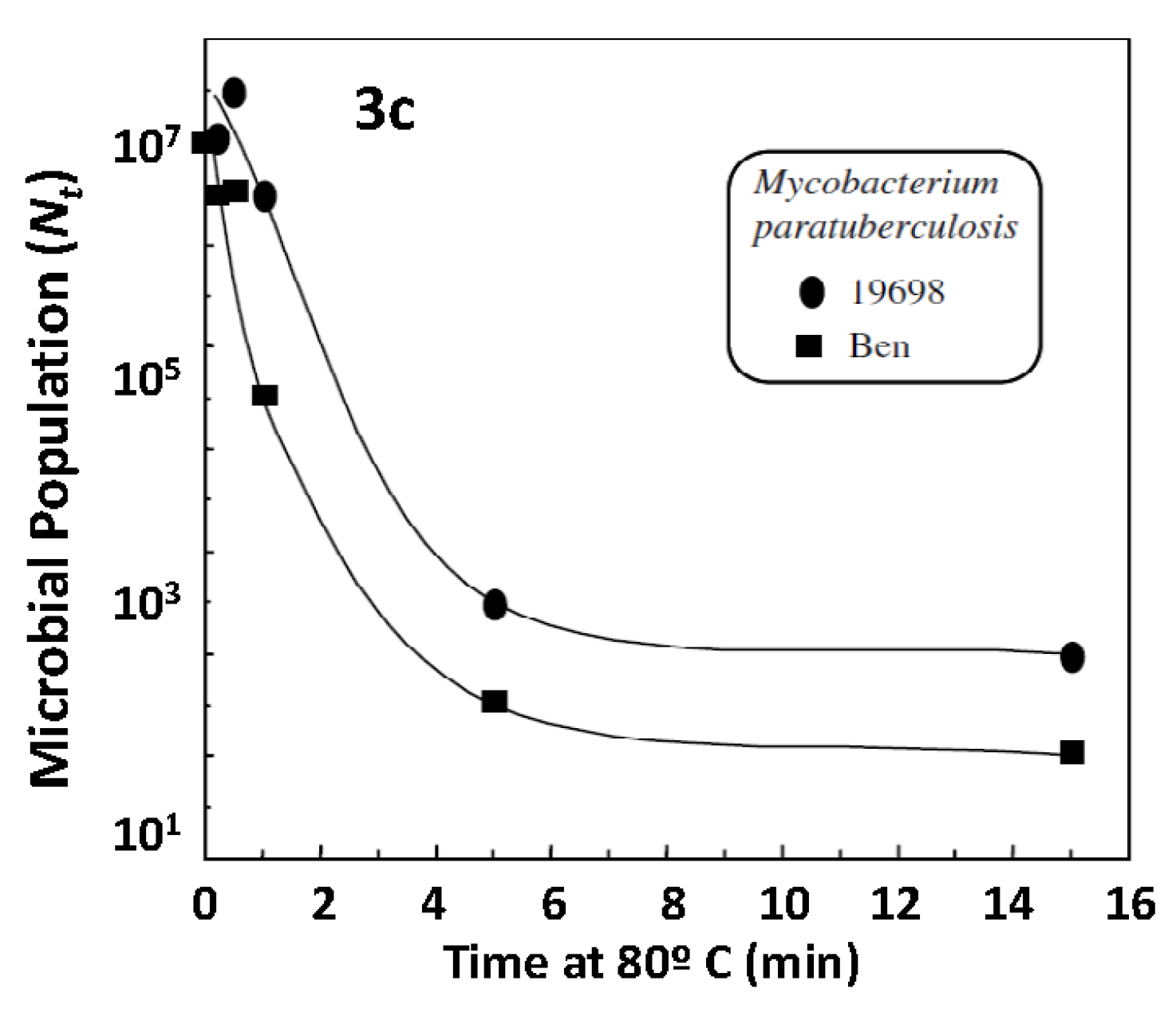
| Brand | Product | Web Pages |
|---|---|---|
| Coldpress | Juices | http://www.cold-press.com/ |
| Créaline | Purees, Soups | http://www.crealine.fr/ |
| Evolution | Juices, Smoothies | www.evolutionfresh.com/ |
| Fresh nutribits | Juices | http://www.nutribits.com |
| Fruity line | Juices, Smoothies | http://fruity-line.nl/en/ |
| Hoogsteger | Juices | http://www.hoogesteger.nl/ |
| In Fruit | Juices, Smoothies | http://www.infruit.fr/ |
| Invo | Coconut water | https://www.invococonutwater.com/ |
| La fruitière du Val Evel | Purees | http://www.lafruitiere.com/red-fruits.html |
| Presha fruits | Juices | http://www.preshafruit.com.au/cold-pressed |
| Press & Reset | Juices | http://www.pressandreset.com/ |
| Romantics | Juices, Smoothies | https://www.shopromantics.es/ |
| Teresa’s juicery | Juices, Purees | https://teresasjuicery.com/ |
| The juicy group | Juices, Smoothies | http://www.juicygroup.be/ |
| Ulti daregal | Juices | http://www.daregal.fr/en/home/ |
| Category of Foodstuff | Microorganisms/Toxins, Metabolites | Sampling Plan 1 | Limits 2,3 | Reference Analysis Method | Application of Criterion | ||
|---|---|---|---|---|---|---|---|
| n | c | m | M | ||||
| 1.19 Fruits and vegetable precut (ready to be consumed) | Salmonella | 5 | 0 | Absence in 25 g | EN/ISO 6579 | Product placed on the market during their shelf life. | |
| 1.20 Fruits juice and vegetable unpasteurized | Salmonella | 5 | 0 | Absence in 25 g | EN/ISO 6579 | Product placed on the market during their shelf life. | |
| 2.5.1 Fruits and vegetable precut (ready to be consumed) | E.coli | 5 | 2 | 100 cfu/g | 1000 cfu/g | ISO 16649-1 or 2 | Improvement of production hygiene and selection of raw materials. |
| 2.5.2 Fruits juice and vegetable unpasteurized (ready to be consumed) | E.coli | 5 | 2 | 100 cfu/g | 1000 cfu/g | ISO 16649-1 or 2 | Improvement of production hygiene and selection of raw materials. |
| Fruit/Vegetable | Product Type | Reference | HPP Conditions | Microbial Inactivation Criterion | Log Reductions * | Shelf Life (Storage Ta) | |
|---|---|---|---|---|---|---|---|
| Author, Year | No. | ||||||
| Apple | Puree | Landl, 2010 | [48] | 400-600 MPa 15 min 20 °C | Aerobic mesophilic bacteria Yeasts/Molds | 3.3 3.2 | 14–21 days 4 °C |
| Apple | Juice | Shahbaz, 2016 | [40] | 500 MPa 1 min 25 °C | L. monocytogenes S. aureus E. coli S. tiphimurium S. cerevisiae | 4.8 2.4 5.0 7.0 5.8 | NP |
| Apple Orange | Juices | Hartyáni, 2013 | [49] | 200–600 MPa 10 min 20–60 °C | Alicyclobacillus acidoterrestris | 2.2 2.0 | 14–28 days 4 °C |
| Apple | Juice concentrate | Lee, 2006 | [50] | 207–621 MPa 5–10 min 22–90 °C | Spores of Alicyclobacillus acidoterrestris | 2–5 | NS |
| Apple/Plum | Jams | Prestamo, 1999 | [51] | 100–400 MPa 5–15 min 5–20 °C | L. monocytogenes | 1–9 | NS |
| Apple/Apricot | Juices | Bayindirli, 2006 | [41] | 350 MPa 5 min 30 °C | S. aureus E. coli S. enteriditis | 6.8/6.9 7.1/7.3 7.8/8.0 | NP |
| Sour Cherry/Orange | S. aureus E. coli S. enteriditis | 7.4/7.3 7.7/7.4 8.5/8.5 | |||||
| Banana | Smoothie N2-degassed | Li, 2015 | [52] | 550 MPa 2–10 min 20 °C | Total aerobic bacteria Yeasts/Molds | 2 2.5 | >15 days 4 °C |
| Mung Bean Sprout | Juices | Muñoz, 2008 | [53] | 150–400 MPa 2 min 20–40 °C | Aerobic mesophilic bacteria Fecal coliforms | 1.7–5.7 up to 7.8 | >13 days 4 °C |
| Sour Chinese Cabbage | Fresh | Li, 2010 | [54] | 400–600 MPa 10–30 min rt | Total aerobic bacteria Lactic acid bacteria Yeasts | 2.7–4.5 2.4–7 1.5–4.2 | 60 days 4 °C |
| Cactus | Juice | Moussa, 2017 | [55] | 600 MPa 10 min 15 °C | Viable microbial cells Yeast/Molds Acid tolerant microorganims | 3 | NP |
| Cantaloupe | Puree | Mukhopadhyay, 2017 | [56] | 300–500 MPa 5 min 8 °C, 15 °C | Total aerobic bacteria | 1–3.3 | 10 days 4 °C |
| Carrot, Melon or Papaya | Soy Smoothie | Andrés, 2016 | [57] | 550–650 MPa 3 min 20 °C | NP | NP | 45 days 4 °C |
| Cashew apple | Juice | Lavinas, 2008 | [58] | 250–400 MPa 3–7 min 25 °C | Aerobic mesophilic bacteria Yeasts/Filamentous fungi E. coli | 1.8–5.9 NP 6.4–6.5 | 56 days 4 °C |
| Cherimoya | Pulp | Perez, 2015 | [59] | 600 MPa 8 min 20 °C | Viable microbial cells | 5 | 7–15 days |
| Cucumber | Juice | Zhao, 2013 | [4] | 500 Mpa 2 min | Viable microbial cells Yeasts/Molds | 2 3–4 | 50 days 4 °C |
| Feijoa | Puree | Duong, 2015 | [60] | 200–400 MPa 6 min 25 °C | E. coli B. subtilis S. cerevisiae | 2.5 2.5 6.5 | NS |
| Red Grape White Grape | Juices | Mert, 2013 | [61] | 150–250 MPa 5–15 min, 20–40 °C | Viable microbial cells | 5 7 | 90 days 20 °C (dark storage) |
| Keiskei | Juice | Chai, 2014 | [62] | 550 MPa 1.5 min rt | Coliform bacteria Yeasts/Molds Pseudomonas B. cereus | 6.0 4.8 5.3 2.3 | 6 days 4 °C |
| Kiwi | Puree | Fernández-Sestelo, 2013 | [63] | 500 MPa 3 min rt (20–25 °C) | Viable microbial cells | 3.4 | 21 days 4 °C |
| Kiwi/Pineapple | Juices | Buzrul, 2008 | [64] | 350 Mpa 5 min 10–20 °C | E. coli L. innocua | ~5.5/~2.5 ~5.5/~3.5 | 21 days 4 °C–37 °C |
| Mandarin | Juice | Carreño, 2011 | [65] | 150–450 MPa 0,1–1 min 15–45 °C | L. plantarum | 0–1 | NS |
| Mango | Nectar | Liu, 2014 | [66] | 600 MPa 1 min 20 °C | Total aerobic bacteria Yeasts/Molds | 5.2 3.1 | NS |
| Mango | Pulp | Liu, 2013 | [67] | 300-600 MPa 1–20 min ºC NP | Total aerobic bacteria Yeasts/Molds | 3.8 1.5–2.0 | NS |
| Olive | Jam | Delgado-Adamez, 2013 | [68] | 450/600 MPa 5 min 10 °C | Aerobic mesophilic bacteria Yeasts/Molds E. coli | ND | >6 months |
| Orange | Juice | Bull, 2004 | [69] | 600 MPa 1 min | Total aerobic bacteria Yeasts/Molds | 4.5 3.5 | 56 days 10 °C |
| Orange | Juice | Katsaros, 2010 | [70] | 100–500 MPa 1–30 min 20–40 °C | L. plantarum L. brevis | 0.2–4.5 0.1–4.0 | NS |
| Orange | Juice | Linton, 2015 | [71] | 400–550 MPa 5 min 20–30 °C | E. coli | 5–6 | NS |
| Orange | Juice | Polydera, 2004 | [72] | 600 Mpa 4 min 40 °C | NS | NS | 187–147 days 0 °C |
| Orange | Juice | Syed, 2014 | [73] | 700 Mpa 5 min 4 °C | S. aureus | 6.2–6.6 | 15 days 4 °C |
| Orange | Juice | Timmermans, 2011 | [28] | 600 Mpa 1 min 17 °C | Viable microbial cells Yeasts/Molds | 3–8 3–5 | 58 days 4 °C |
| Orange | Juice | Yoo, 2015 | [74] | 400 Mpa 1 min | E. coli | 2.4 | NP |
| Orange, Apple or Tomato | Juices | Jordan, 2001 | [75] | 500 MPa 5 min rt (20 °C) | E. coli | ~5 | NP |
| Orange/Peach | Juices | Erkmen, 2004 | [76] | 200–700 MPa 1–90 min 25 °C | Total aerobic bacteria L. monocytogenes | 7.8/7.5 7.8/7.5 | NS |
| Pear | Nectar | Guerrero, 2011 | [77] | 103–241 MPa 2 s–15 min 25 °C | E. coli L. innocua S. cerevisiae | 0.1–1.5 0.1–0.2 0.1–0.4 | NS |
| Sauerkraut | Fermented cabbage | Peñas, 2010 | [78] | 300 MPa 10 min 40 °C | Total coliforms Aerobic mesophilic bacteria Lactic acid bacteria Faecal coliforms Yeasts/Molds | <1 4.2 4.2 <1 <1 | 60 days 4 °C |
| Soy | Smoothies | Andrés, 2016 | [57] | 450–650 MPa 3 min 20 °C | Aerobic mesophilic bacteria | 3 | 30 days 4 °C |
| Strawberry | Puree | Marszalek, 2015 | [79] | 300–500 MPa 1–15 min 50 °C | Viable microbial cells Yeasts Molds | 1.5–2.4 2.6–3.6 0.5–3.8 | NS |
| Tomato | Juice | Hsu, 2008 | [80] | 300–500 MPa 10 min 25 °C | Viable microbial cells Yeasts/Molds Enterobacteria Lactic acid bacteria | 0.9–4.1 3.7/3.6 2.1 4.2 | 28 days 4 °C |
| Tomato | Puree | Krebbers, 2003 | [81] | 700 MPa, 0.5–2 min 20–90 °C | B. stearothermophilus | 2.7–6.1 | 56 days 4 °C |
| Tomato/Water melon | Juice | Aganovic, 2017 | [82] | 600 Mpa 5 min | Collected from literature | 5 | NP |
| Red fruits with orange, banana + lime | Smoothies | Hurtado, 2017 | [83] | 350 MPa 7 min <25 °C | Aerobic mesophilic bacteria Psychrotrophic bacteria Yeasts/Molds Enterobacteria | 1.8 2.5 1.8 2.4 | 28 days 4 °C |
| Wild berries with grapes, apples + orange | Smoothies | Scolari, 2015 | [84] | 100–300 MPa 5 min 5–45 °C | L. monocytogenes | 2–6.3 | NS |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Daher, D.; Le Gourrierec, S.; Pérez-Lamela, C. Effect of High Pressure Processing on the Microbial Inactivation in Fruit Preparations and Other Vegetable Based Beverages. Agriculture 2017, 7, 72. https://doi.org/10.3390/agriculture7090072
Daher D, Le Gourrierec S, Pérez-Lamela C. Effect of High Pressure Processing on the Microbial Inactivation in Fruit Preparations and Other Vegetable Based Beverages. Agriculture. 2017; 7(9):72. https://doi.org/10.3390/agriculture7090072
Chicago/Turabian StyleDaher, Dahlia, Soléne Le Gourrierec, and Concepción Pérez-Lamela. 2017. "Effect of High Pressure Processing on the Microbial Inactivation in Fruit Preparations and Other Vegetable Based Beverages" Agriculture 7, no. 9: 72. https://doi.org/10.3390/agriculture7090072
APA StyleDaher, D., Le Gourrierec, S., & Pérez-Lamela, C. (2017). Effect of High Pressure Processing on the Microbial Inactivation in Fruit Preparations and Other Vegetable Based Beverages. Agriculture, 7(9), 72. https://doi.org/10.3390/agriculture7090072






