Grasslands and Croplands Have Different Microbial Biomass Carbon Levels per Unit of Soil Organic Carbon
Abstract
:1. Introduction
2. Materials and Methods
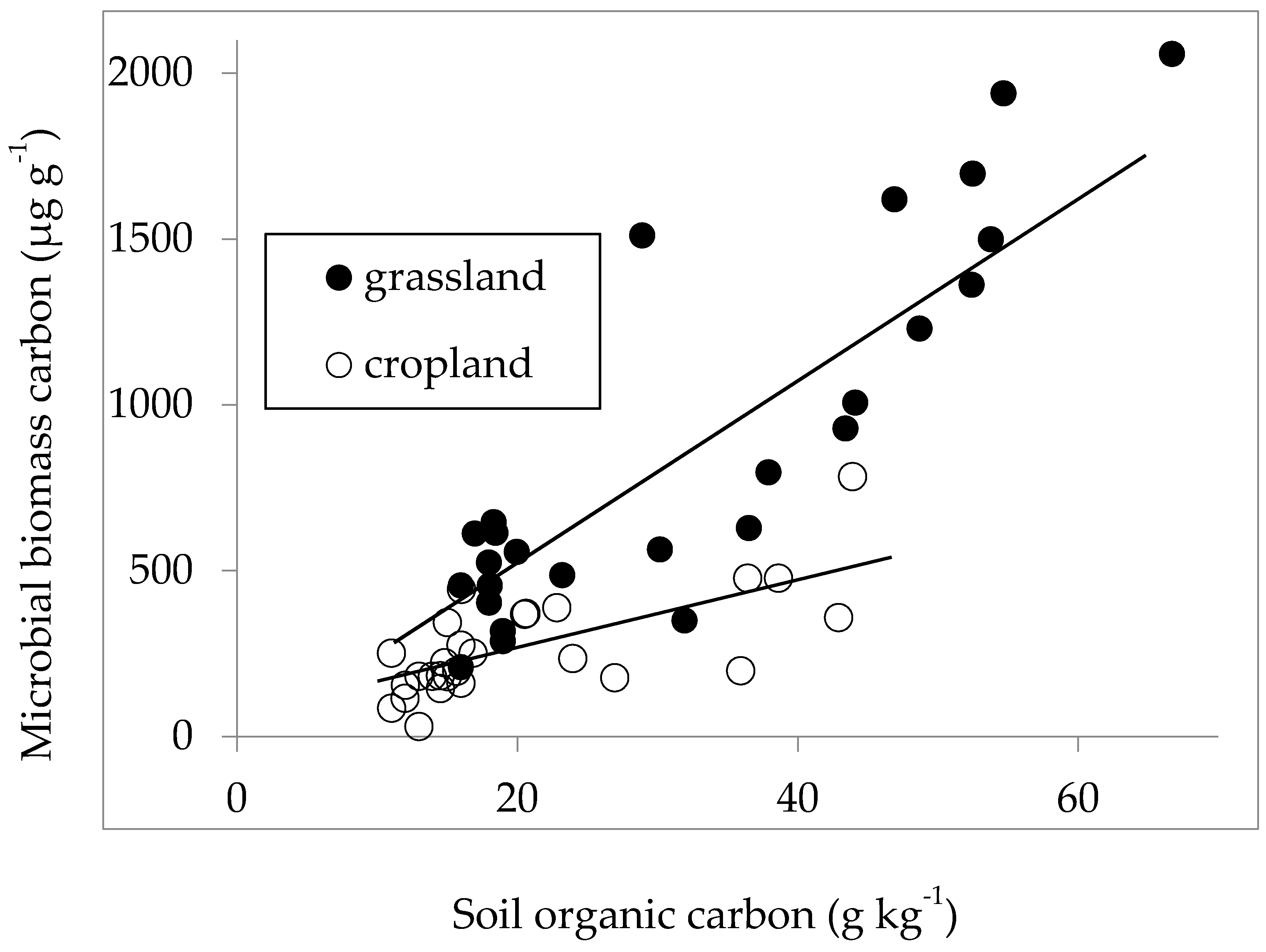
3. Results
4. Discussion
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Powlson, D.S. The soil microbial biomass: Before, beyond and back. In Beyond the Biomass; Ritz, K., Dighton, J., Giller, K.L., Eds.; Wiley-Sayce: London, UK, 1994; pp. 3–20. [Google Scholar]
- Powlson, D.S.; Brookes, P.C.; Christensen, B.T. Measurement of soil microbial biomass provides an early indication of changes due to straw incorporation. Soil Biol. Biochem. 1987, 19, 159–164. [Google Scholar] [CrossRef]
- Wander, M. Soil organic matter fractions and their relevance to soil function. In Soil Organic Matter in Sustainable Agriculture; Magdoff, F., Weil, R.R., Eds.; CRC Press: Boca Raton, FL, USA, 2004; pp. 67–102. [Google Scholar]
- Rovira, P.; Jorba, M.; Romanya, J. Active and passive organic matter fractions in Mediterranean forest soils. Biol. Fertil. Soils 2010, 46, 355–369. [Google Scholar] [CrossRef]
- Hassink, J. Density fractions of soil macroorganic matter and microbial biomass as predictors of C and N mineralization. Soil Biol. Biochem. 1995, 27, 1099–1108. [Google Scholar] [CrossRef]
- Doran, J.W.; Zeiss, M.R. Soil health and sustainability: Managing the biotic component of soil quality. Appl. Soil Ecol. 2000, 15, 3–11. [Google Scholar] [CrossRef]
- Anderson, T.-H. Microbial eco-physiological indicators to assess soil quality. Agric. Ecosyst. Environ. 2003, 98, 285–293. [Google Scholar] [CrossRef]
- Karlen, D.L.; Mausbach, M.J.; Doran, J.W.; Cline, R.G.; Harris, R.F.; Schuman, G.E. Soil quality: A concept, definition, and framework for evaluation (a guest editorial). Soil Sci. Soc. Am. J. 1997, 61, 4–10. [Google Scholar] [CrossRef]
- Zornoza, R.; Acosta, J.A.; Bastida, F.; Domínguez, S.G.; Toledo, D.M.; Faz, A. Identification of sensitive indicators to assess the interrelationship between soil quality, management practices and human health. Soil 2015, 1, 173. [Google Scholar] [CrossRef]
- Gonzalez-Quinones, V.; Stockdale, E.A.; Banning, N.C.; Hoyle, F.C.; Sawada, Y.; Wherrett, A.D.; Jones, D.L.; Murphy, D.V. Soil microbial biomass—Interpretation and consideration for soil monitoring. Soil Res. 2011, 49, 287–304. [Google Scholar] [CrossRef]
- Andrews, S.S.; Karlen, D.L.; Cambardella, C.A. The soil management assessment framework. Soil Sci. Soc. Am. J. 2004, 68, 1945–1962. [Google Scholar] [CrossRef]
- Bastida, F.; Zsolnay, A.; Hernandez, T.; Garcia, C. Past, present and future of soil quality indices: A biological perspective. Geoderma 2008, 147, 159–171. [Google Scholar] [CrossRef]
- Erkossa, T.; Itanna, F.; Stahr, K. Indexing soil quality: A new paradigm in soil science research. Soil Res. 2007, 45, 129–137. [Google Scholar] [CrossRef]
- Carter, M.R. The influence of tillage on the proportion of organic carbon and nitrogen in the microbial biomass of medium-textured soils in a humid climate. Biol. Fertil. Soils 1991, 11, 135–139. [Google Scholar] [CrossRef]
- Moore, J.M.; Klose, S.; Tabatabai, M.A. Soil microbial biomass carbon and nitrogen as affected by cropping systems. Biol. Fertil. Soils 2000, 31, 200–210. [Google Scholar] [CrossRef]
- Xu, X.; Thornton, P.E.; Post, W.M. A global analysis of soil microbial biomass carbon, nitrogen and phosphorus in terrestrial ecosystems. Glob. Ecol. Biogeogr. 2013, 22, 737–749. [Google Scholar] [CrossRef]
- Lovell, R.D.; Jarvis, S.C.; Bardgett, R.D. Soil microbial biomass and activity in long-term grassland: Effects of management changes. Soil Biol. Biochem. 1995, 27, 969–975. [Google Scholar] [CrossRef]
- Lovell, R.D.; Jarvis, S.C. Effect of cattle dung on soil microbial biomass C and N in a permanent pasture soil. Soil Biol. Biochem. 1996, 28, 291–299. [Google Scholar] [CrossRef]
- Wang, F.E.; Chen, Y.X.; Tian, G.M.; Kumar, S.; He, Y.F.; Fu, Q.L.; Lin, Q. Microbial biomass carbon, nitrogen and phosphorus in the soil profiles of different vegetation covers established for soil rehabilitation in a red soil region of southeastern China. Nutr. Cycl. Agroecosyst. 2004, 68, 181–189. [Google Scholar] [CrossRef]
- Kabiri, V.; Raiesi, F.; Ghazavi, M.A. Tillage effects on soil microbial biomass carbon, SOM mineralization and enzyme activity in a semi-arid calcixerepts. Agric. Ecosyst. Environ. 2016, 232, 73–84. [Google Scholar] [CrossRef]
- Franzluebbers, A.J.; Arshad, M.A. Soil organic matter pools during early adoption of conservation tillage in northwestern Canada. Soil Sci. Soc. Am. J. 1996, 60, 1422–1427. [Google Scholar] [CrossRef]
- Karlen, D.L.; Stott, D.E.; Cambardella, C.A.; Kremer, R.J.; King, K.W.; McCarty, G.W. Surface soil quality in five midwestern cropland conservation effects assessment project watersheds. J. Soil Water Conserv. 2014, 69, 393–401. [Google Scholar] [CrossRef]
- Vance, E.D.; Brookes, P.C.; Jenkinson, D.S. An extraction method for measuring soil microbial biomass C. Soil Biol. Biochem. 1987, 19, 703–707. [Google Scholar] [CrossRef]
- Jenkinson, D.S. Studies on the decomposition of plant material in soil. J. Soil Sci. 1966, 17, 280–302. [Google Scholar] [CrossRef]
- Jenkinson, D.S. Determination of microbial biomass carbon and nitrogen in soil. In Advances in Nutrient Cycling in Agricultural Ecosystems; Wilson, J.R., Ed.; CAB International: Wallingford, UK, 1988; pp. 368–386. [Google Scholar]
- Anderson, J.P.E.; Domsch, K.H. A physiological method for the quantitative measurement of microbial biomass in soils. Soil Biol. Biochem. 1978, 10, 215–221. [Google Scholar] [CrossRef]
- Islam, K.R.; Weil, R.R. Microwave irradiation of soil for routine measurement of microbial biomass carbon. Biol. Fertil. Soils 1998, 27, 408–416. [Google Scholar] [CrossRef]
- Turner, B.L.; Bristow, A.W.; Haygarth, P.M. Rapid estimation of microbial biomass in grassland soils by ultra-violet absorbance. Soil Biol. Biochem. 2001, 33, 913–919. [Google Scholar] [CrossRef]
- Gregorich, E.G.; Wen, G.; Voroney, R.P.; Kachanoski, R.G. Calibration of a rapid direct chloroform extraction method for measuring soil microbial biomass C. Soil Biol. Biochem. 1990, 22, 1009–1011. [Google Scholar] [CrossRef]
- Strecker, T.; Barnard, R.L.; Niclaus, P.A.; Scherer-Lorenzen, M.; Weigelt, A.; Scheu, S.; Eisenhauer, N. Effects of plant diversity, functional group composition, and fertilization on soil microbial properties in experimental grassland. PLoS ONE 2015, 10, e0125678. [Google Scholar] [CrossRef] [PubMed]
- Jenkinson, D.S.; Brookes, P.C.; Powlson, D.S. Measuring soil microbial biomass. Soil Biol. Biochem. 2004, 36, 5–7. [Google Scholar] [CrossRef]
- Johnson, C.K.; Wienhold, B.J.; Doran, J.W.; Drijber, R.A.; Wright, S.F. Linking microbial-scale findings to farm-scale outcomes in a dryland cropping system. Precis. Agric. 2004, 5, 311–328. [Google Scholar] [CrossRef]
- Seita, R.; Verma, S.L.; Marschner, P. Measuring microbial biomass carbon by direct extraction—Comparison with chloroform fumigation-extraction. Eur. J. Soil Biol. 2012, 53, 103–106. [Google Scholar]
- Nelson, D.W.; Sommers, L.E. Total carbon, organic carbon, and organic matter. In Methods of Soil Analysis. Part 3. Chemical Methods; Sparks, D.L., Ed.; Soil Science Society of America: Madison, WI, USA, 1996; pp. 961–1010. [Google Scholar]
- Iyyemperumal, K.; Israel, D.W.; Shi, W. Soil microbial biomass, activity and potential nitrogen mineralization in a pasture: Impact of stock camping activity. Soil Biol. Biochem. 2007, 39, 149–157. [Google Scholar] [CrossRef]
- Zar, J.H. Biostatistical Analysis, 5th ed.; Prentice Hall: Upper Saddle River, NJ, USA, 2010. [Google Scholar]
- Allison, V.J.; Miller, R.M.; Jastrow, J.D.; Matamala, R.; Zak, D.R. Changes in soil microbial community structure in a tallgrass prairie chronosequence. Soil Sci. Soc. Am. J. 2005, 69, 1412–1421. [Google Scholar] [CrossRef]
- Banerjee, M.R.; Burton, D.L.; McCaughey, W.P.; Grant, C.A. Influence of pasture management on soil biological quality. J. Range Manag. 2000, 53, 127–133. [Google Scholar] [CrossRef]
- Lovell, R.D.; Jarvis, S.C. Soil microbial biomass and activity in soil from different grassland management treatments stored under controlled conditions. Soil Biol. Biochem. 1998, 30, 2077–2085. [Google Scholar] [CrossRef]
- Corre, M.D.; Schnabel, R.R.; Stout, W.L. Spatial and seasonal variation of gross nitrogen transformations and microbial biomass in a Northeastern US grassland. Soil Biol. Biochem. 2002, 34, 445–457. [Google Scholar] [CrossRef]
- Jordan, D.; Kremer, R.J.; Bergfield, W.A.; Kim, K.Y.; Cacnio, V.N. Evaluation of microbial methods as potential indicators of soil quality in historical agricultural fields. Biol. Fertil. Soils 1995, 19, 297–302. [Google Scholar] [CrossRef]
- Friedel, J.K.; Scheller, E. Composition of hydrolysable amino acids in soil organic matter and soil microbial biomass. Soil Biol. Biochem. 2002, 34, 315–325. [Google Scholar] [CrossRef]
- Klose, S.; Tabatabai, M.A. Urease activity of microbial biomass in soils. Soil Biol. Biochem. 1999, 31, 205–211. [Google Scholar] [CrossRef]
- Balota, E.L.; Colozzi-Filho, A.; Andrade, D.S.; Dick, R.P. Microbial biomass in soils under different tillage and crop rotation systems. Biol. Fertil. Soils 2003, 38, 15–20. [Google Scholar] [CrossRef]
- Weil, R.R.; Islam, K.R.; Stine, M.A.; Gruver, M.A.; Samson-Liebig, S.E. Estimating active carbon for soil quality assessment: A simplified method for laboratory and field use. Am. J. Alter. Agric. 2003, 18, 3–17. [Google Scholar]
- Veum, K.S.; Goyne, K.W.; Kremer, R.J.; Miles, R.J.; Sudduth, K.A. Biological indicators of soil quality and soil organic matter characteristics in an agricultural management continuum. Biogeochemistry 2014, 117, 81–99. [Google Scholar] [CrossRef]
- Hurisso, T.T.; Culman, S.W.; Horwath, W.R.; Wade, J.; Cass, D.; Beniston, J.W.; Bowles, T.M.; Grandy, A.S.; Franzluebbers, A.J.; Schipanski, M.E.; et al. Comparison of permanganate-oxidizable carbon and mineralizable carbon for assessment of organic matter stabilization and mineralization. Soil Sci. Soc. Am. J. 2016, 80, 1352–1364. [Google Scholar] [CrossRef]
- Liu, M.-Y.; Chang, Q.-R.; Qi, Y.-B.; Liu, J.; Chen, T. Aggregation and soil organic carbon fractions under different land uses on the tableland of the Loess Plateau of China. Catena 2014, 115, 19–28. [Google Scholar] [CrossRef]
- Gregorich, E.G.; Beare, M.H.; McKim, U.F.; Skjemstad, J.O. Chemical and biological characteristics of physically uncomplexed organic matter. Soil Sci. Soc. Am. J. 2006, 70, 975–985. [Google Scholar] [CrossRef]
- Hsieh, Y.-P. Pool size and mean age of stable carbon in cropland. Soil Sci. Soc. Am. J. 1992, 56, 460–464. [Google Scholar] [CrossRef]
- Collins, H.P.; Elliott, E.T.; Paustian, K.; Bundy, L.G.; Dick, W.A.; Huggins, D.R.; Smucker, A.J.M.; Paul, E.A. Soil carbon pools and fluxes in long-term corn belt agroecosystems. Soil Biol. Biochem. 2000, 32, 157–168. [Google Scholar] [CrossRef]
- Burke, I.C.; Yonker, C.M.; Parton, W.J.; Cole, C.V.; Schimel, D.S.; Flach, K. Texture, climate, and cultivation effects on soil organic matter content in US grassland soils. Soil Sci. Soc. Am. J. 1989, 53, 800–805. [Google Scholar] [CrossRef]
- Brady, N.C.; Weil, R.R. The Nature and Properties of Soils, 13th ed.; Prentice Hall: Upper Saddle River, NJ, USA, 2002. [Google Scholar]
- Shi, X.P.; Li, X.G.; Long, R.J.; Singh, B.P.; Li, Z.T.; Li, F.M. Dynamics of soil organic carbon and nitrogen associated with physically separated fractions in a grassland-cultivation sequence in the Qinghai-Tibetan plateau. Biol. Fertil. Soils 2010, 46, 103–111. [Google Scholar] [CrossRef]
- Agbenin, J.O.; Adeniyi, T. The microbial biomass properties of a savanna soil under improved grass and legume pastures in northern Nigeria. Agric. Ecosyst. Environ. 2005, 109, 245–254. [Google Scholar] [CrossRef]
- Bailey, V.L.; Smith, J.L.; Bolton, H. Fungal-to-bacterial ratios in soils investigated for enhanced C sequestration. Soil Biol. Biochem. 2002, 34, 997–1007. [Google Scholar] [CrossRef]
- Mbuthia, L.W.; Acosta-Martínez, V.; Debryun, J.; Schaeffer, S.; Tyler, D.; Odoi, E.; Eash, N. Long term tillage, cover crop, and fertilization effects on microbial community structure, activity: Implications for soil quality. Soil Biol. Biochem. 2015, 89, 24–34. [Google Scholar] [CrossRef]
- Tiemann, L.K.; Grandy, A.S.; Atkinson, E.E.; Marin-Spiotta, E.; McDaniel, M.D. Crop rotational diversity enhances belowground communities and functions in an agroecosystem. Ecol. Lett. 2015, 18, 761–771. [Google Scholar] [CrossRef] [PubMed]
- Wardle, D.A. A comparative assessment of factors which influence microbial biomass carbon and nitrogen levels in soil. Biol. Rev. 1992, 67, 321–358. [Google Scholar] [CrossRef]
- Carter, M.R. Analysis of soil organic matter storage in agroecosystems. In Structure and Organic Matter Storage in Agricultural Soils; Carter, M.R., Stewart, B.A., Eds.; CRC Press: Boca Raton, FL, USA, 1996; pp. 3–11. [Google Scholar]
| System | Trials | Location | Field Use | Reference |
|---|---|---|---|---|
| Grassland | 9 | IL, USA | Restored prairie | [37] |
| Grassland | 8 | MB, Canada | Grazed pasture | [38] |
| Grassland | 4 | Devon, UK | Grazed pasture | [39] |
| Grassland | 3 | PA, USA | Native grassland | [40] |
| Grassland | 2 | MO, USA | Long-term Timothy | [41] |
| Grassland | 1 | MO, USA | Virgin prairie | [41] |
| Grassland | 1 | Lake Constance, Germany | Grassland | [42] |
| Cropland | 10 | IA, USA | Agricultural fields | [43] |
| Cropland | 6 | Parana, Brazil | Maize, wheat, cotton, soybean | [44] |
| Cropland | 5 | MO, USA | Maize, soybean | [41] |
| Cropland | 3 | Stuttgart, Germany | Winter wheat | [42] |
| Cropland | 2 | IL, USA | Maize, soybean | [37] |
| Cropland | 1 | Lovinghoeve, Netherlands | Arable | [42] |
| Source | DF | SS | MS | F | p |
|---|---|---|---|---|---|
| Regression | 1 | 6,998,352 | 6,998,352 | 84.4 | <0.001 |
| Residual | 26 | 2,155,244 | 82,894 | ||
| Total | 27 | 9,153,595 |
| Source | DF | SS | MS | F | p |
|---|---|---|---|---|---|
| Regression | 1 | 305,486 | 305,486 | 22.98 | <0.001 |
| Residual | 25 | 332,338 | 13294 | ||
| Total | 26 | 637,824 |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
McGonigle, T.P.; Turner, W.G. Grasslands and Croplands Have Different Microbial Biomass Carbon Levels per Unit of Soil Organic Carbon. Agriculture 2017, 7, 57. https://doi.org/10.3390/agriculture7070057
McGonigle TP, Turner WG. Grasslands and Croplands Have Different Microbial Biomass Carbon Levels per Unit of Soil Organic Carbon. Agriculture. 2017; 7(7):57. https://doi.org/10.3390/agriculture7070057
Chicago/Turabian StyleMcGonigle, Terence P., and William G. Turner. 2017. "Grasslands and Croplands Have Different Microbial Biomass Carbon Levels per Unit of Soil Organic Carbon" Agriculture 7, no. 7: 57. https://doi.org/10.3390/agriculture7070057
APA StyleMcGonigle, T. P., & Turner, W. G. (2017). Grasslands and Croplands Have Different Microbial Biomass Carbon Levels per Unit of Soil Organic Carbon. Agriculture, 7(7), 57. https://doi.org/10.3390/agriculture7070057





