Green Tea as an Agricultural Based Health Promoting Food: The Past Five to Ten Years
Abstract
1. Introduction
| Country | Quantities (tons) |
|---|---|
| China | 1,217,039 |
| India | 963,313 |
| Kenya | 344,596 |
| Sri Lanka | 304,036 |
| Turkey | 209,535 |
| Viet Nam | 167,531 |
| Indonesia | 154,328 |
| Iran | 108,751 |
| Japan | 92,233 |
| Argentina | 76,071 |
| Thailand | 59,203 |
| Bangladesh | 58,763 |
| Malawi | 46,155 |
| Uganda | 41,543 |
| United Republic of Tanzania | 31,850 |
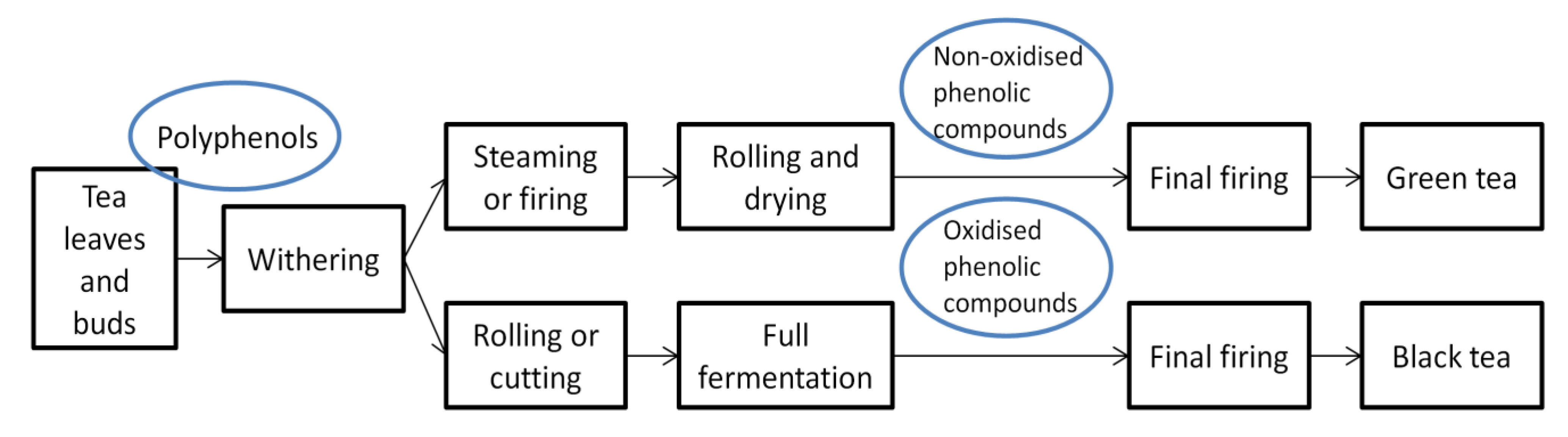
2. Composition of Green Tea
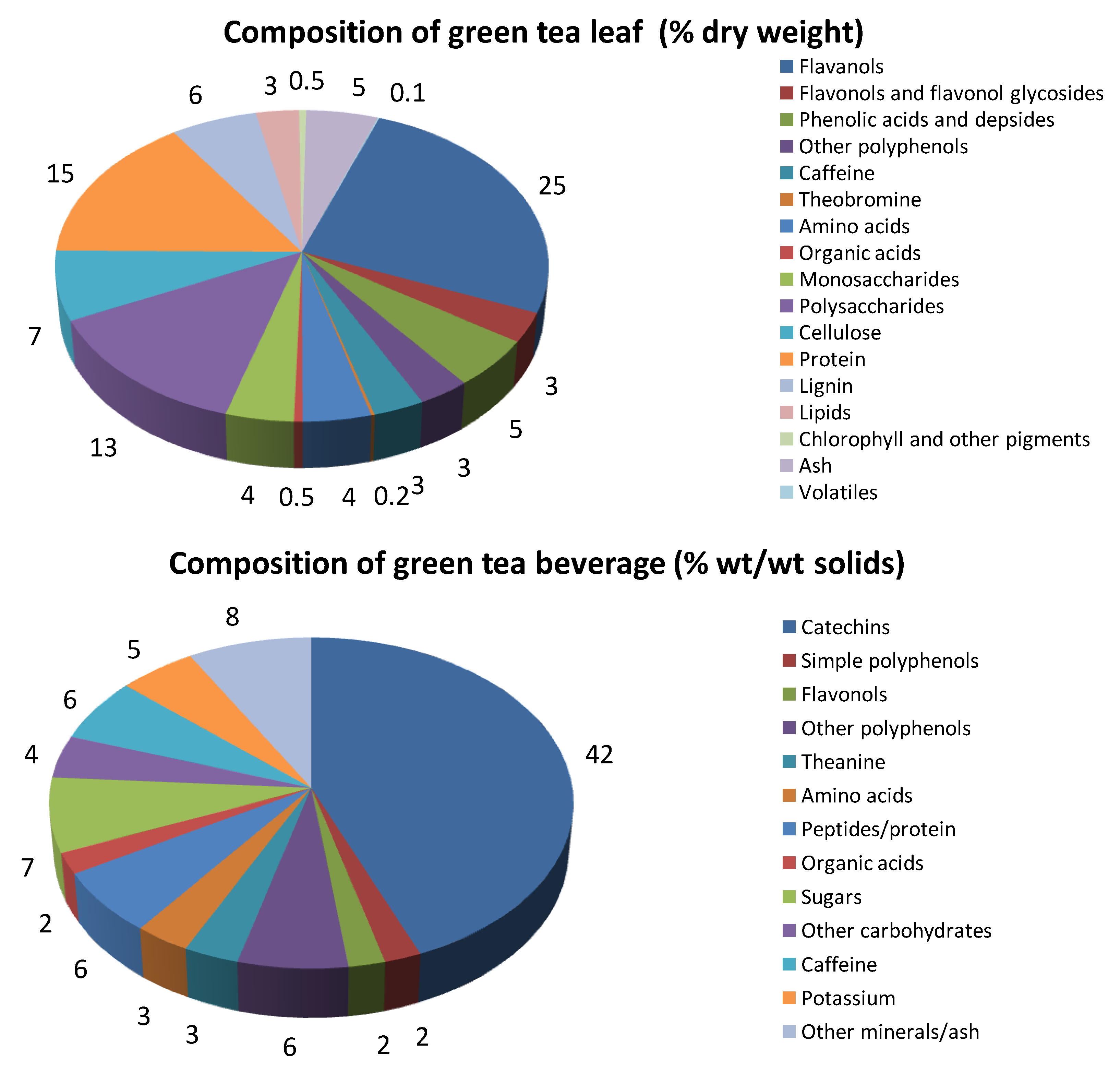
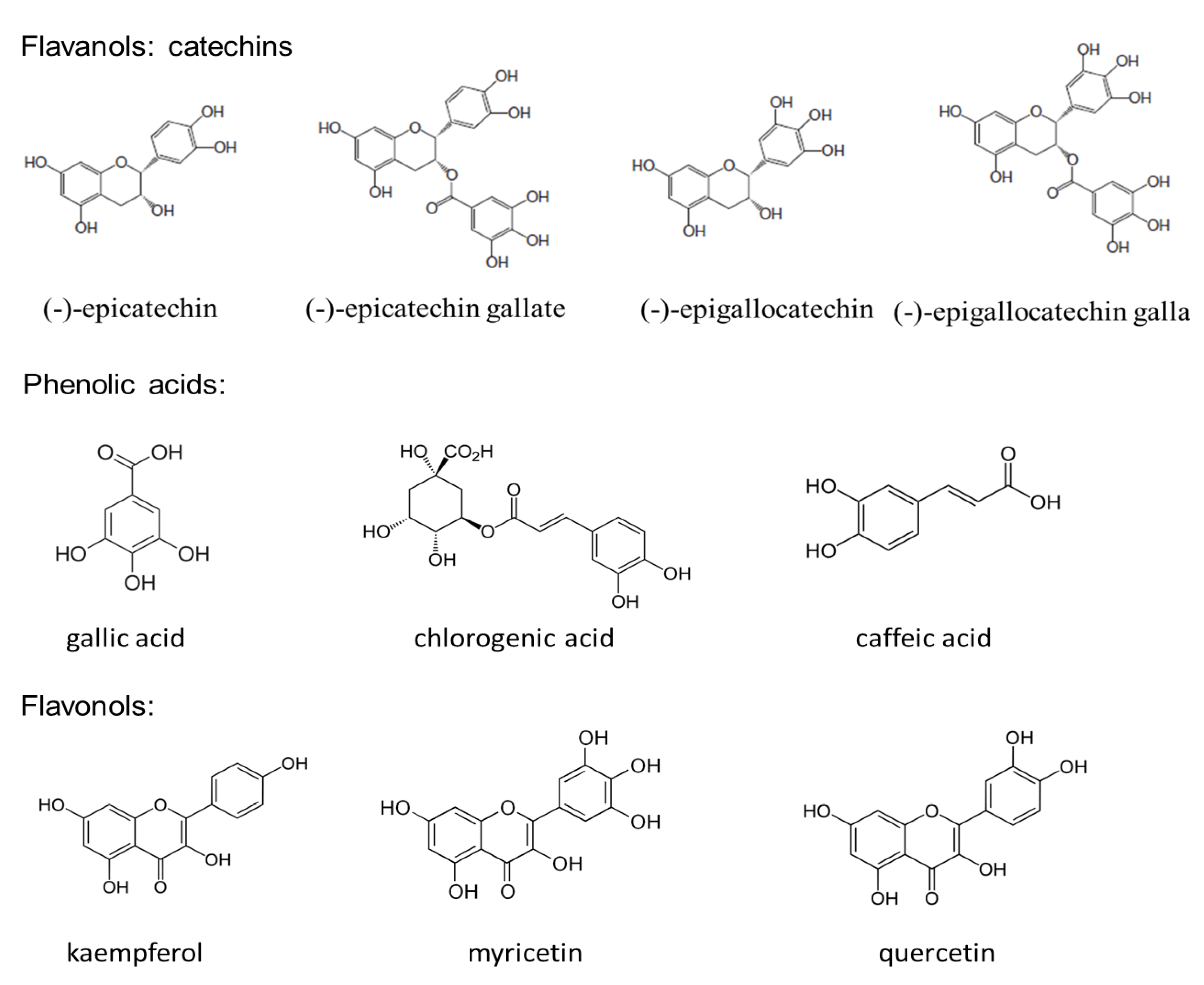
3. Oxidative Stress
4. Inflammation
| References | Model | Treatment | Dosage | Main outcomes |
|---|---|---|---|---|
| Schroeder et al. [16] | Rat cerebellar granule neurons cultures | EGCG | 5, 10, or 20 μM | 90%–95% of 3H-EGCG accumulated in mitochondria. EGCG treatments resulted in reduced levels of free radicals and protected neuron from apoptosis induced by oxidative stress. |
| Guo et al. [17] | 6-hydroxydopamine (6-OHDA)-treated rat model of Parkinson’s Disease | GTP (98% purity) containing 50% EGCG, 22% ECG, 18% EGC and 10% EC | 150, 450 mg/kg/day-fed | GTP treatment dose-dependently decreased ROS and NO level, lipid peroxidation, nitrite/nitrate ratio, and expression of inducible nitric oxide synthase and protein-bound 3-nitro-tyrosine. |
| Quesada et al. [18] | Human HepG2 cells | EGCG | 10 μM | EGCG binded to zinc cations with high affinity and thus reduced the accumulation of zinc. |
| Wei & Meng [19] | Myocytes from thehearts of adult rats | EGCG | 0–60 μg/mL | Formation of OH−, O2−, and H2O2 was decreased significantly as EGCG concentration increased compared to lead treated group. |
| Maeta et al. [21] | Budding yeast S. cerevisiae and fission yeast S. pombe | EGCG and GTE | 0.05% EGCG | H2O2 produced by EGCG and GTE induced the expression of nuclear localization of oxidative-stress-responsive transcription factors (Yap1 and Msn2 in S. cerevisiae, and Pap1 in S. pombe). |
| 0.1% GTE | ||||
| Elbling et al. [22] | Human keratinocytes | EGCG | 0–100 μM | Low-dose H2O2 and EGCG treatment enhanced the clearance of ROS and reduced DNA damage and cytotoxicity in keratinocyte-derived HaCat cell. |
5. Cardiovascular Health
| References | Model | Treatment | Dosage | Main Outcomes |
|---|---|---|---|---|
| Dona et al. [26] | Neutrophils and C57 black mice | EG CG | 1–100 μM | EGCG down-modulated ROS activity, and inhibited apoptosis and chemotaxis in vitro model. EGCG also inhibited neutrophil recruitment, angiogenesis and pulmonary fibrosis. |
| Shen et al. [27] | 3-month-old female rats | GTP (98.5% purity) | 0.5% in water | The development of fibrosis in vessels and myocardium in rat decreased significantly with supplementation via down-regulation of TNF-α mRNA expression. |
| Wong et al. [29] | Human Jurkat leukemic CD4+ T cell line and eight-week old Balb/c male mice | EGCGGT beverage | 2, 10, 50 μM | EGCG induced expression of Foxp3 and increased T-reg frequencies and numbers both in vitro and in vivo. |
| 2%, w/v | ||||
| Tsai et al. [30] | 1 2-weeks-old female NZB/W F1 mice | EGCG | 120 mg/kg body weight | T-reg activity increased in EGCG treated mice. |
| Wu et al. [31] | C57BL mice spleen cells | EGCG | 2.5–10 μM | EGCG targeted the pathway which is related to IL-2/IL-2 receptor signaling and inhibited T cell proliferation. |
| Ranjith-Kumar et al. [32] | Human HEK293T cells | EGCG | 0–10 μM | EGCG inhibited RIG-I by binding to RIG-I and suppressing ATPase activity of recombinant RIG-I. |
| Noh and Park [33] | Human keratinocytes | EGCG | 100 μmol/L | EGCG down regulated MIF mRNA expression and MIF induced cytokines and chemokines. |
| Ripley et al. [34] | Monocytes of human PBMCs and SOC1-deficient C57BL/6 mice | GTE and EGCG | 0–200 μM | EGCG inhibited cytokine signaling 1 gene expression via inducing the SOC1 expression and un-regulating STAT5 transcription factor. |
6. Obesity and Diabetes
7. Cancer
| References | Model | Treatment | Dosage | Main outcomes |
|---|---|---|---|---|
| Ohga et al. [57] | Human cell lines A375SM and HSC3 | EGCG | 100 μM | EGCG selectively targeted tumor-associated endothelial cells and endothelial progenitor cells, thereby suppressing their migration. |
| Kato et al. [58] | Human cell lines HSC3, HSC4, SCC9, SCC25 and HeLa | EGCG | 5, 10, 20, 50 μM | EGCG treatment partially reversed the hypermethylation status of the RECK gene. |
| Siddiqui et al. [59] | LNCaP and 22Rν1 cells | EGCG | 0, 10, 20, 40, 60 μM | EGCG competitively binding to AR and interrupting the gene expression regulated by AR. |
| Yuasa et al. [61] | Tissue of gastric cancer patients | Brewed GT | 0–10 cups per day | Methylation of CDX2 and BMP-2 were inversely correlated with GT intake |
| Thangapazham et al. [62] | MDA-MB-231 cell line and athymic nude mice model | EGCG and GTP | 0–200 μg/ml | EGCG and GTP down regulated cancer related gene expression and inhibited proliferation of cancer cell. |
| Henning et al. [63] | Male SCID mice | Brewed GT | Tumor volume, oxidative DNA and protein damage in tumor tissue were all significantly reduced with GT consumption. | |
| Bettuzzi et al. [64] | Men with high-grade prostate intraepithelialneoplasia | GT catechins | 600 mg/day | One tumor was diagnosed in treatment group whereas nine cancers were detected among the placebo group. Treated group received higher score of international prostate symptom. |
| Kuriyama et al. [54] | Japanese adults | Brewed GT | <1, 1–2, 3–4, >5 cups per day | GT consumption did not significantly associate with cancer mortality. |
8. Gastrointestinal Health
9. Bioavailability
10. Conclusions
References
- Khokhar, S.; Magnusdottir, S.G.M. Total phenol, catechin, and caffeine contents of teas commonly consumed in the United Kingdom. J. Agric. Food Chem. 2002, 50, 565–570. [Google Scholar] [CrossRef]
- Moxham, R. Tea: Addiction, Exploitation, and Empire; Carroll & Graf Publishers: New York, NY, USA, 2003. [Google Scholar]
- Pettigrew, J. The Tea Companion: A Connoisseur’s Guide, 1st ed; Running Press Book Publishers: Philadelphia, PA, USA, 2004. [Google Scholar]
- Li, X. Study on the Competitiveness of Chinese Tea in the Global Market.
- FAO Web site. Available online: http://faostat3.fao.org/home/index.html (accessed on 18 September 2012).
- Singh, B.N.; Shankar, S.; Srivastava, R.K. Green tea catechin, epigallocatechin-3-gallate (EGCG): Mechanisms, perspectives and clinical applications. Biochem. Pharmacol. 2011, 82, 1807–1821. [Google Scholar]
- Butt, M.S.; Sultan, M.T. Green tea: Nature’s defense against malignancies. Crit. Rev. Food Sci. Nutr. 2009, 49, 463–473. [Google Scholar] [CrossRef]
- Cabrera, C.; Artacho, R.; Giménez, R. Beneficial effects of green tea—A review. J. Am. Coll. Nutr. 2006, 25, 79–99. [Google Scholar]
- Balentine, D.A.; Wiseman, S.A.; Bouwens, L.C.M. The chemistry of tea flavonoids. Crit. Rev. Food Sci. Nutr. 1997, 37, 693–704. [Google Scholar]
- Lin, L.Z.; Chen, P.; Harnly, J.M. New phenolic components and chromatographic profiles of green and fermented teas. J. Agric. Food Chem. 2008, 56, 8130–8140. [Google Scholar] [CrossRef]
- Hatanaka, A. The biogeneration of green odour by green leaves. Phytochemistry 1993, 34, 1201–1218. [Google Scholar]
- Wang, Y.; Wei, X.; Jin, Z. Structure analysis of a neutral polysaccharide isolated from green tea. Food Res. Int. 2009, 42, 739–745. [Google Scholar] [CrossRef]
- Chen, H.; Zhang, M.; Qu, Z.; Xie, B. Antioxidant activities of different fractions of polysaccharide conjugates from green tea ( Camellia sinensis). Food Chem. 2007, 106, 559–563. [Google Scholar]
- Circu, M.L.; Aw, T.Y. Reactive oxygen species, cellular redox systems, and apoptosis. Free Radic. Biol. Med. 2010, 48, 749–762. [Google Scholar] [CrossRef]
- Kowaltowski, A.J.; Souza-Pinto, N.C.; Castilho, R.F.; Vercesi, A.E. Mitochondria and reactive oxygen species. Free Radic. Biol. Med. 2009, 47, 333–343. [Google Scholar] [CrossRef]
- Schroeder, E.K.; Kelsey, N.A.; Doyle, J.; Breed, E.; Bouchard, R.J.; Loucks, F.A.; Linseman, D.A. Green tea epigallocatechin 3-gallate accumulates in mitochondria and displays a selective antiapoptotic effect against inducers of mitochondrial oxidative stress in neurons. Antioxid. Redox Signal. 2009, 11, 469–480. [Google Scholar] [CrossRef]
- Guo, S.; Yan, J.; Yang, T.; Yang, X.; Bezard, E.; Zhao, B. Protective effects of green tea polyphenols in the 6-OHDA rat model of Parkinson’s Disease through inhibition of ROS-NO pathway. Biol. Psychiatry 2007, 62, 1353–1362. [Google Scholar] [CrossRef]
- Quesada, I.M.; Bustos, M.; Blay, M.; Pujadas, J.; Ardèvol, A.; Salvadó, M.J.; Bladé, C.; Arola, L.; Fernández-Larrea, J. Dietary catechins and procyanidins modulate zinc homeostasis in human HepG2 cells. J. Nutr. Biochem. 2011, 22, 153–163. [Google Scholar] [CrossRef]
- Wei, H.; Meng, Z. Protective effects of epigallocatechin-3-gallate against lead-induced oxidative damage. Hum. Exp. Toxicol. 2010, 30, 1521–1528. [Google Scholar]
- Lambert, J.D.; Elias, R.J. The antioxidant and pro-oxidant activities of green tea polyphenols: A role in cancer prevention. Arch. Biochem. Biophys. 2010, 501, 65–72. [Google Scholar] [CrossRef]
- Maeta, K.; Nomura, W.; Takatsume, Y.; Izawa, S.; Inoue, Y. Green tea polyphenols function as prooxidants to activate oxidative-stress-responsive transcription factors in yeasts. Appl. Environ. Microbiol. 2007, 73, 572–580. [Google Scholar] [CrossRef]
- Elbling, L.; Herbacek, I.; Weiss, R.-M.; Jantschitsch, C.; Micksche, M.; Gerner, C.; Pangratz, H.; Berger, W. Hydrogen peroxide mediates EGCG-induced antioxidant protection in human keratinocytes. Free Radic. Biol. Med. 2010, 49, 1444–1452. [Google Scholar]
- Lin, W.; Karin, M. A cytokine-mediated link between innate immunity, inflammation, and cancer. J. Clin. Investig. 2007, 117, 1175–1183. [Google Scholar] [CrossRef]
- Lumeng, C.N.; Saltiel, A.R. Inflammatory links between obesity and metabolic disease. J. Clin. Investig. 2011, 121, 2111–2117. [Google Scholar]
- Smith, G.R.; Missailidis, S. Cancer, inflammation and the AT1 and AT2 receptors. J. Inflamm. 2004, 1, 1–12. [Google Scholar] [CrossRef] [Green Version]
- Dona, M.; Dell’Aica, L.; Calabrese, F.; Benelli, R.; Morini, M.; Albini, A.; Garbisa, S. Neutrophil restraint by green tea: Inhibition of inflammation, associated angiogenesis, and pulmonary fibrosis. J. Immunol. 2003, 170, 4335–4341. [Google Scholar]
- Shen, C.-L.; Samathanam, C.; Tatum, O.L.; Graham, S.; Tubb, C.; Cao, J.J.; Dunn, D.M.; Wang, J.-S. Green tea polyphenols avert chronic inflammation-induced myocardial fibrosis of female rats. Inflamm. Res. 2011, 60, 665–672. [Google Scholar] [CrossRef]
- Askenasya, N.; Kaminitza, A.; Yarkoni, S. Mechanisms of T regulatory cell function. Autoimmun. Rev. 2008, 7, 370–375. [Google Scholar]
- Wong, C.P.; Nguyena, L.P.; Nohb, S.K.; Braya, T.B.; Brunob, R.S.; Ho, E. Induction of regulatory T cells by green tea polyphenol EGCG. Immunol. Lett. 2011, 139, 7–13. [Google Scholar] [CrossRef]
- Tsai, P.-Y.; Ka, S.-M.; Chang, J.-M.; Chen, H.-C.; Shui, H.-A.; Li, C.-Y.; Hua, K.-F.; Chang, W.-L.; Huang, J.-J.; Yang, S.-S.; et al. Epigallocatechin-3-gallate prevents lupus nephritis development in mice via enhancing the Nrf2 antioxidant pathway and inhibiting NLRP3 inflammasome activation. Free Radic. Biol. Med. 2011, 51, 744–754. [Google Scholar] [CrossRef]
- Wu, D.; Guo, Z.; Ren, Z.; Guo, W.; Meydani, S.N. Green tea EGCG suppresses T cell proliferation through impairment of IL-2/IL-2 receptor signaling. Free Radic. Biol. Med. 2009, 47, 636–643. [Google Scholar] [CrossRef]
- Ranjith-Kumar, C.T.; Lai, Y.; Sarisky, R.T.; Kao, C.C. Green tea catechin, epigallocatechingallate, suppresses signaling by the dsRNA innate immune receptor RIG-I. PLoSOne 2010, 5, e12878. [Google Scholar]
- Noh, S.U.; Park, Y.M. The effect of green tea polyphenols on macrophage migration inhibitory factor-associated steroid resistance. Br. Assoc. Dermatol. 2012, 166, 653–657. [Google Scholar]
- Ripley, B.J.M.; Fujimoto, M.; Serada, S.; Ohkawara, T.; Nishikawa, T.; Terabe, F.; Naka, T. Green tea polyphenolepigallocatechingallate inhibits cell signaling by inducing SOCS1 gene expression. Int. Immunol. 2010, 22, 359–366. [Google Scholar]
- Deka, A.; Vita, J.A. Tea cardiovascular disease. Pharmacol. Res. 2012, 64, 136–145. [Google Scholar] [CrossRef]
- Del Rio, D.; Rodriguez-Mateos, A.; Spencer, J.P.E.; Tognolini, M.; Borges, G.; Crozier, A. Dietary polyphenolics in human health: Structures, bioavailability, and evidence of protective effects against chronic diseases. Antioxid. Redox Signal. 2012. [Google Scholar] [CrossRef]
- Qin, B.; Polansky, M.M.; Harry, D.; Anderson, R.A. Green tea polyphenols improve cardiac muscle mRNA and protein levels of signal pathways related to insulin and lipid metabolism and inflammation in insulin resistant rats. Mol. Nutr. Food Res. 2010, 54, S14–S23. [Google Scholar] [CrossRef]
- Lorenz, M.; Urban, J.; Engelhardt, U.; Baumann, G.; Stangl, K.; Stangl, V. Green and black tea are equally potent stimuli of NO production and vasodilation: New insights into tea ingredients involved. Basic Res. Cardiol. 2009, 104, 100–110. [Google Scholar] [CrossRef]
- El Bedoui, J.; Oak, M.H.; Anglard, P.; Schini-Kerth, V.B. Catechins prevent vascular smooth muscle cell invasion by inhibiting MT1-MMP activity and MMP-2 expression. Cardiovasc. Res. 2005, 67, 317–325. [Google Scholar] [CrossRef]
- Bursill, C.A.; Roach, P.D. Modulation of cholesterol metabolism by the green tea polyphenol (−)-epigallocatechingallate in cultured human liver (HepG2) cells. J. Agric. Food Chem. 2006, 54, 1621–1626. [Google Scholar] [CrossRef]
- Bursill, C.A.; Abbey, M.; Roach, P.D. A green tea extract lowers plasma cholesterol by inhibiting cholesterol synthesis and upregulating the LDL receptor in the cholesterol-fed rabbit. Atherosclerosis 2007, 193, 86–93. [Google Scholar] [CrossRef]
- Alexopoulos, N.; Vlachopoulos, C.; Aznaouridis, K.; Baou, K.; Vasiliadou, C.; Pietri, P.; Xaplanteris, P.; Stefanadi, E.; Stefanadis, C. The acute effect of green tea consumption on endothelial function in healthy individuals. Eur. J. Cardiovasc. Prev. Rehabil. 2008, 15, 300–305. [Google Scholar]
- Zheng, X.-X.; Xu, Y.-L.; Li, H.-S.; Liu, X.-X.; Hui, J.; Huang, X.-H. Green tea intake lowers fasting serum total and LDL cholesterol in adults: A meta-analysis of 14 randomized controlled trials. Am. J. Clin. Nutr. 2011, 94, 601–610. [Google Scholar] [CrossRef]
- Sae-Tan, S.; Grove, K.A.; Lambert, J.D. Weight control and prevention of metabolic syndrome by green tea. Pharmacol. Res. 2012, 64, 146–154. [Google Scholar]
- Bose, M.; Lambert, J.D.; Ju, J.; Reuhl, K.R.; Shapses, S.A.; Yang, C.S. The major green tea polyphenol, (−)-Epigallocatechin-3-Gallate, inhibits obesity, metabolic syndrome, and fatty liver disease in high-fat-fed mice. J. Nutr. 2008, 138, 1677–1683. [Google Scholar]
- Sae-Tan, S.; Grove, K.A.; Kennettb, M.J.; Lambert, J.D. (-)-Epigallocatechin-3-gallate increases the expression of genes related to fat oxidation in the skeletal muscle of high fat-fed mice. Food Funct. 2011, 2, 111–116. [Google Scholar] [CrossRef]
- Lee, M.-S.; Kim, C.-T.; Kim, I.-H.; Kim, Y. Inhibitory effects of green tea catechin on the lipid accumulation in 3T3-L1 adipocytes. Phytother. Res. 2009, 23, 1088–1091. [Google Scholar]
- Richard, D.; Kefi, K.; Barbe, U.; Poli, A.; Bausero, P.; Visioli, F. Weight and plasma lipid control by decaffeinated green tea. Pharmocol. Res. 2009, 59, 351–354. [Google Scholar] [CrossRef]
- Cao, H.; Hininger-Favier, I.; Kelly, M.A.; Benaraba, R.; Dawson, H. D.; Coves, S.; Roussel, A.M.; Anderson, R.A. Green tea polyphenol extract regulates the expression of genes involved in glucose uptake and insulin signaling in rats fed a high fructose diet. J. Agric. Food Chem. 2007, 55, 6372–6378. [Google Scholar] [CrossRef]
- Hininger-Favier, I.; Benaraba, R.; Coves, S.; Anderson, R.A.; Roussel, A.M. Green tea extract decreases oxidative stress and improves insulin sensitivity in an animal model of insulin resistance, the fructose-fed rat. J. Am. Coll. Nutr. 2009, 28, 355–361. [Google Scholar]
- Ma, J.; Li, Z.; Xing, S.; Ho, W.T.; Fu, X.; Zhao, Z.J. Tea contains potent inhibitors of tyrosine phosphatase PTP1B. Biochem. Biophys. Res. Commun. 2011, 407, 98–102. [Google Scholar] [CrossRef]
- Oba, S.; Nagata, C.; Nakamura, K.; Fujii, K.; Kawachi, T.; Takatsuka, N.; Shimizu, H. Consumption of coffee, green tea, oolong tea, black tea, chocolate snacks and the caffeine content in relation to risk of diabetes in Japanese men and women. Br. J. Nutr. 2010, 103, 453–459. [Google Scholar] [CrossRef]
- Hsu, C.-H.; Tsai, T.-H.; Kao, Y.-H.; Hwang, K.-C.; Tseng, T.-Y.; Chou, P. Effect of green tea extract on obese women: A randomized, double-blind, placebo-controlled clinical trial. Clin.Nutr. 2008, 27, 363–370. [Google Scholar] [CrossRef]
- Kuriyama, S.; Shimazu, T.; Ohmori, K.; Kikuchi, N.; Nakaya, N.; Nishino, Y.; Tsubono, Y.; Tsuji, I. Green tea consumption and mortality due to cardiovascular disease, cancer, and all causes in Japan: The Ohsaki study. J. Am. Med. Assoc. 2006, 296, 1255–1265. [Google Scholar]
- Basu, A.; Sanchez, K.; Leyva, M.J.; Wu, M.; Betts, N.M.; Aston, C.E.; Lyons, T.J. Green tea supplementation affects body weight, lipids, and lipid peroxidation in obese subjects with metabolic syndrome. J. Am. Coll. Nutr. 2010, 29, 31–40. [Google Scholar]
- Khan, N.; Mukhtar, H. Multi-targeted therapy of cancer by green tea polyphenols. Cancer Lett. 2008, 269, 269–280. [Google Scholar]
- Ohga, N.; Hida, K.; Hida, Y.; Muraki, C.; Tsuchiya, K.; Matsuda, K.; Ohiro, Y.; Totsuka, Y.; Shindoh, M. Inhibitory effects of epigallocatechin-3 gallate, a polyphenol in green tea, on tumor-associated endothelial cells and endothelial progenitor cells. Cancer Sci. 2009, 100, 1963–1970. [Google Scholar] [CrossRef]
- Kato, K.; Long, N.K.; Makita, H.; Toida, M.; Yamashita, T.; Hatakeyama, D.; Hara, A.; Mori, H.; Shibata, T. Effects of green tea polyphenol on methylation status of RECK gene and cancer cell invasion in oral squamous cell carcinoma cells. Br. J. Cancer 2008, 99, 647–654. [Google Scholar] [CrossRef]
- Siddiqui, I.A.; Asim, M.; Hafeez, B.B.; Adhami, V.M.; Tarapore, R.S.; Mukhtar, H. Green tea polyphenol EGCG blunts androgen receptor function in prostate cancer. FASEB J. 2011, 25, 1198–1207. [Google Scholar] [CrossRef]
- Lee, Y.H.; Kwak, J.; Choi, H.K.; Choi, K.C.; Kim, S.; Lee, J.; Jun, W.; Park, H.J.; Yoon, H.G. EGCG suppresses prostate cancer cell growth modulating acetylation of androgen receptor by anti-histone acetyltransferase activity. Int. J. Mol. Med. 2012. [Google Scholar] [CrossRef]
- Yuasa, Y.; Nagasaki, H.; Akiyama, Y.; Hashimoto, Y.; Takizawa, T.; Kojima, K.; Nakachi, K. DNA methylation status is inversely correlated with green tea intake and physical activity in gastric cancer patients. Int. J. Cancer 2009, 124, 2677–2682. [Google Scholar] [CrossRef]
- Thangapazham, R.L.; Singh, A.K.; Sharma, A.; Warren, J.; Gaddipati, J.P.; Maheshwari, R.K. Green tea polyphenols and its constituent epigallocatechingallate inhibits proliferation of human breast cancer cells in vitro and in vivo. Cancer Lett. 2007, 245, 232–241. [Google Scholar] [CrossRef]
- Henning, S.M.; Wang, P.; Said, J.; Magyar, C.; Castor, B.; Doan, N.; Tosity, C.; Moro, A.; Gao, K.; Li, L.; et al. Polyphenols in brewed green tea inhibit prostate tumor xenograft growth by localizing to the tumor and decreasing oxidative stress and angiogenesis. J. Nutr. Biochem. 2012, 23, 1537–1542. [Google Scholar]
- Bettuzzi, S.; Brausi, M.; Rizzi, F.; Castagnetti, G.; Peracchia, G.; Corti, A. Chemoprevention of human prostate cancer by oral administration of green tea catechins in volunteers with high-grade prostate intraepithelial neoplasia: A preliminary report from a one-year proof-of-principle study. Cancer Res. 2006, 66, 1234–1240. [Google Scholar] [CrossRef]
- Yang, C.S.; Wang, H.; Li, G.X.; Yang, Z.; Guan, F.; Jin, H. Cancer prevention by tea: Evidence from laboratory studies. Pharmacol. Res. 2012, 64, 113–122. [Google Scholar]
- Yuan, J.-M.; Sun, C.; Butler, L.M. Tea and cancer prevention: Epidemiological studies. Pharmacol. Res. 2012, 64, 123–135. [Google Scholar]
- Chow, H.-H.S.; Hakim, I.A. Pharmacokinetic and chemoprevention studies on tea in humans. Pharmacol. Res. 2012, 64, 105–112. [Google Scholar]
- Flint, H.J.; Duncan, S.H.; Scott, K.P.; Louis, P. Interactions and competition within the microbial community of the human colon: Links between diet and health. Environ. Microbiol. 2007, 9, 1101–1111. [Google Scholar] [CrossRef]
- Cani, P.D.; Possemiers, S.; van de Wiele, T.; Guiot, Y.; Everard, A.; Rottier, O.; Geurts, L.; Naslain, D.; Neyrinck, A.; Lambert, D.M.; et al. Changes in gut microbiota control inflammation in obese mice through mechanism involving GLP-2-driven improvement of gut permeability. Gut 2009, 58, 1091–1103. [Google Scholar] [CrossRef]
- Corr, S.C.; Li, Y.; Riedel, C.U.; O’Toole, P.W.; Hill, C.; Gahan, C.G.M. Bacteriocin production as a mechanism for the anti-infective activity of Lactobacillus salivarius UCC118. Proc. Natl. Acad. Sci. USA 2007, 104, 7617–7621. [Google Scholar]
- Lee, Y.K.; Menezes, J.S.; Umesaki, Y.; Mazmanian, S.K. Proinflammatory T-cell responses to gut microbiota promote experimental autoimmune encephalomyelitis. Proc. Natl. Acad. Sci. USA 2011, 108, 4615–4622. [Google Scholar]
- Dunne, C.; Murphy, L.; Flynn, S.; O’Mahony, L.; O’Halloran, S.; Feeney, M.; Morrissey, D.; Thornton, G.; Fitzgerald, G.; Daly, C.; et al. Probiotics: from myth to reality. Demonstration of functionality in animal models of disease and in human clinical trials. Antonie van Leeuwenhoek. 1999, 76, 279–292. [Google Scholar]
- Crittenden, R.; Playne, M.J. Modifying the human intestinal microbiota with prebiotics. In Gastrointestinal Microbiology; Ouwehand, A.C., Vaughan, E.E., Eds.; Taylor & Francis Inc.: New York, NY, USA, 2006; pp. 285–314. [Google Scholar]
- Khoruts, A.; Dicksved, J.; Jansson, J.K.; Sadowsky, M.J. Changes in the composition of the human fecal microbiome after bacteriotherapy for recurrent Clostridium difficile-associated diarrhea. J. Clin. Gastroenterol. 2010, 44, 354–360. [Google Scholar]
- Lee, H.C.; Jenner, A.M.; Low, C.S.; Lee, Y.K. Effect of tea phenolics and their aromatic fecal bacterial metabolites on intestinal microbiota. Res. Microbiol. 2006, 157, 876–884. [Google Scholar]
- Su, P.; Henriksson, A.; Nilsson, C.; Mitchell, H. Synergistic effect of green tea extract and probiotics on the pathogenic bacteria, Staphylococcus aureus and Streptococcus pyogenes. World J. Microbiol. Biotechnol. 2008, 24, 1837–1841. [Google Scholar] [CrossRef]
- Sutherland, J.; Miles, M.; Hedderley, D.; Li, J.; Devoy, S.; Sutton, K.; Lauren, D. In vitro effects of food extracts on selected probiotic and pathogenic bacteria. Int. J. Food Sci. Nutr. 2009, 60, 717–727. [Google Scholar] [CrossRef]
- Molan, A.L.; Flanagan, J.; Wei, W.; Moughan, P.J. Selenium-containing green tea has higher antioxidant and prebiotic activities than regular green tea. Food Chem. 2009, 114, 829–835. [Google Scholar]
- Green, R.J.; Murphy, A.S.; Schulz, B.; Watkins, B.A.; Ferruzzi, M.G. Common tea formulations modulate in vitro digestive recovery of green tea catechins. Mol. Nutr. Food Res. 2007, 51, 1152–1162. [Google Scholar] [CrossRef]
- Stalmach, A.; Troufflard, S.; Serafini, M.; Crozier, A. Absorption, metabolism and excretion of Choladi green tea flavan-3-ols by humans. Mol. Nutr. Food Res. 2009, 53, S44–S53. [Google Scholar]
- Roowi, S.; Stalmach, A.; Mullen, W.; Lean, W.E.J.; Edwards, C.A.; Crozier, A. Green tea flavan-3-ols: Colonic degradation and urinary excretion of catabolites by humans. J. Agric. Food Chem. 2010, 58, 1296–1304. [Google Scholar]
- Calani, L.; Del Rio, D.; Luisa Callegari, M.; Morelli, L.; Brighenti, F. Updated bioavailability and 48 h excretion profile of flavan-3-ols from green tea in humans. Int. J. Food Sci. Nutr. 2012, 63, 513–521. [Google Scholar]
- Peters, C.M.; Green, R.J.; Janle, E.M.; Ferruzzi, M.G. Formulation with ascorbic acid and sucrose modulates catechin bioavailability from green tea. Food Res. Int. 2010, 43, 95–102. [Google Scholar] [CrossRef]
- Henning, S.M.; Niu, Y.; Lee, N.H.; Thames, G.D.; Minutti, R.R.; Wang, H.; Go, V.L.W.; Heber, D. Bioavailability and antioxidant activity of tea flavanols after consumption of green tea, black tea, or a green tea extract supplement. Am. J. Clin. Nutr. 2004, 80, 1558–1564. [Google Scholar]
- Chow, H.-H.S.; Hakim, I.A.; Vining, D.R.; Crowell, J.A.; Ranger-Moore, J.; Chew, W.M.; Celaya, C.A.; Rodney, S.R.; Hara, Y.; Alberts, D.A. Effects of dosing condition on the oral bioavailability of green tea catechins after single-dose administration of polyphenon E in healthy individuals. Clin. Cancer Res. 2005, 11, 4627–4633. [Google Scholar] [CrossRef]
- Archivio, M.D.; Filesi, C.; Rosaria, V.; Scazzocchio, B.; Masella, R. Bioavailability of the polyphenols: Status and controversies. Int. J. Mol. Sci. 2010, 11, 1321–1342. [Google Scholar] [CrossRef]
- Sang, S.; Lambert, J.D.; Ho, C.-T.; Yang, C.S. The chemistry and biotransformation of tea constituents. Pharmacol. Res. 2011, 64, 87–99. [Google Scholar] [CrossRef]
- Hollman, P.C.H. Absorption, bioavailability and metabolism of flavonoids. Pharm. Biol. 2004, 42, 74–83. [Google Scholar]
© 2012 by the authors; licensee MDPI, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Shi, Q.-Y.; Schlegel, V. Green Tea as an Agricultural Based Health Promoting Food: The Past Five to Ten Years. Agriculture 2012, 2, 393-413. https://doi.org/10.3390/agriculture2040393
Shi Q-Y, Schlegel V. Green Tea as an Agricultural Based Health Promoting Food: The Past Five to Ten Years. Agriculture. 2012; 2(4):393-413. https://doi.org/10.3390/agriculture2040393
Chicago/Turabian StyleShi, Qin-Yin, and Vicki Schlegel. 2012. "Green Tea as an Agricultural Based Health Promoting Food: The Past Five to Ten Years" Agriculture 2, no. 4: 393-413. https://doi.org/10.3390/agriculture2040393
APA StyleShi, Q.-Y., & Schlegel, V. (2012). Green Tea as an Agricultural Based Health Promoting Food: The Past Five to Ten Years. Agriculture, 2(4), 393-413. https://doi.org/10.3390/agriculture2040393




