Nematicidal Effects of Four Terpenes Differ among Entomopathogenic Nematode Species
Abstract
1. Introduction
2. Materials and Methods
2.1. Survival Bioassays
2.1.1. Source and Preparation of Nematodes
2.1.2. Preparation of Terpene Solutions
2.2. Pathogenicity Bioassays
2.3. Statistical Analysis
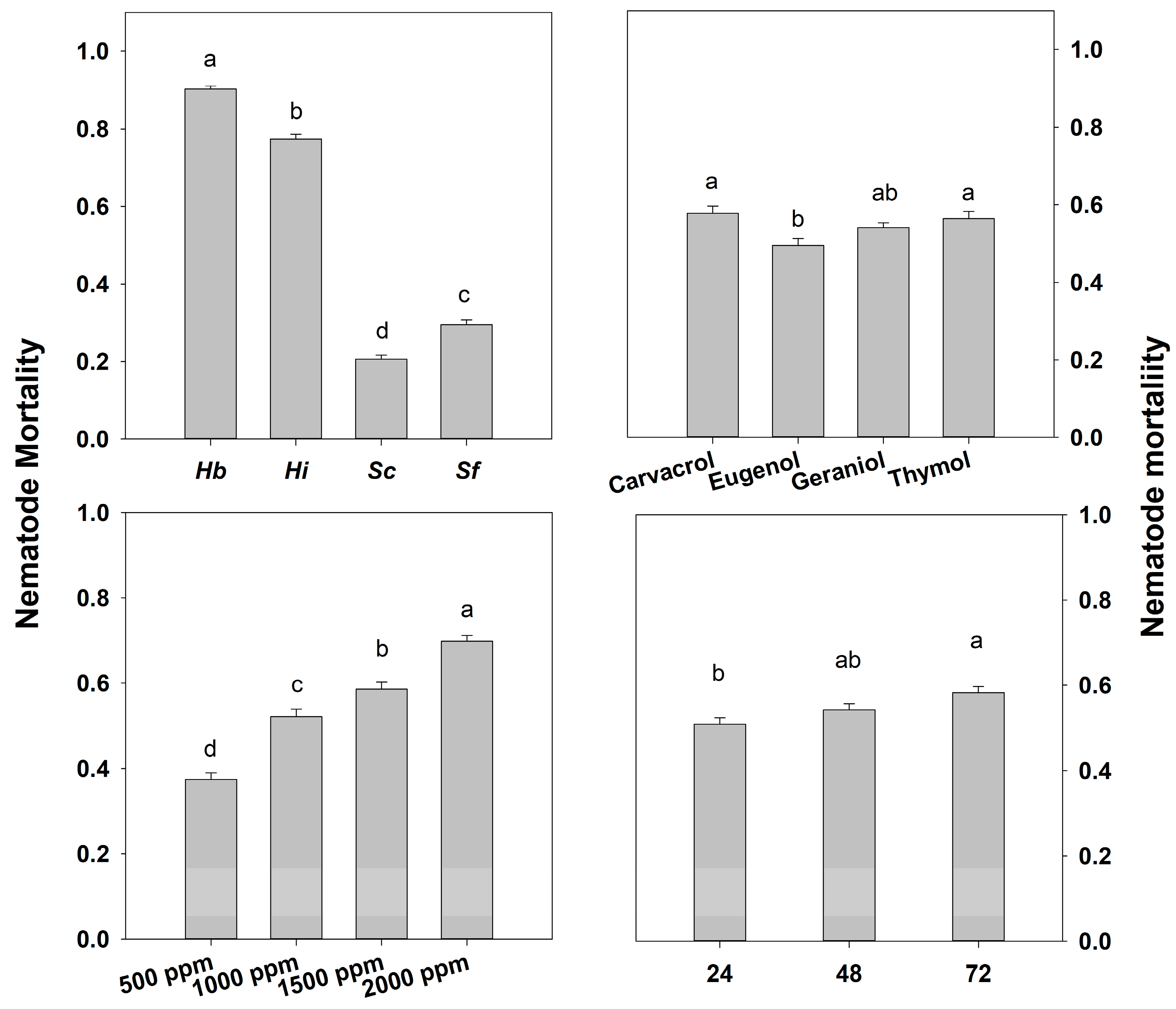
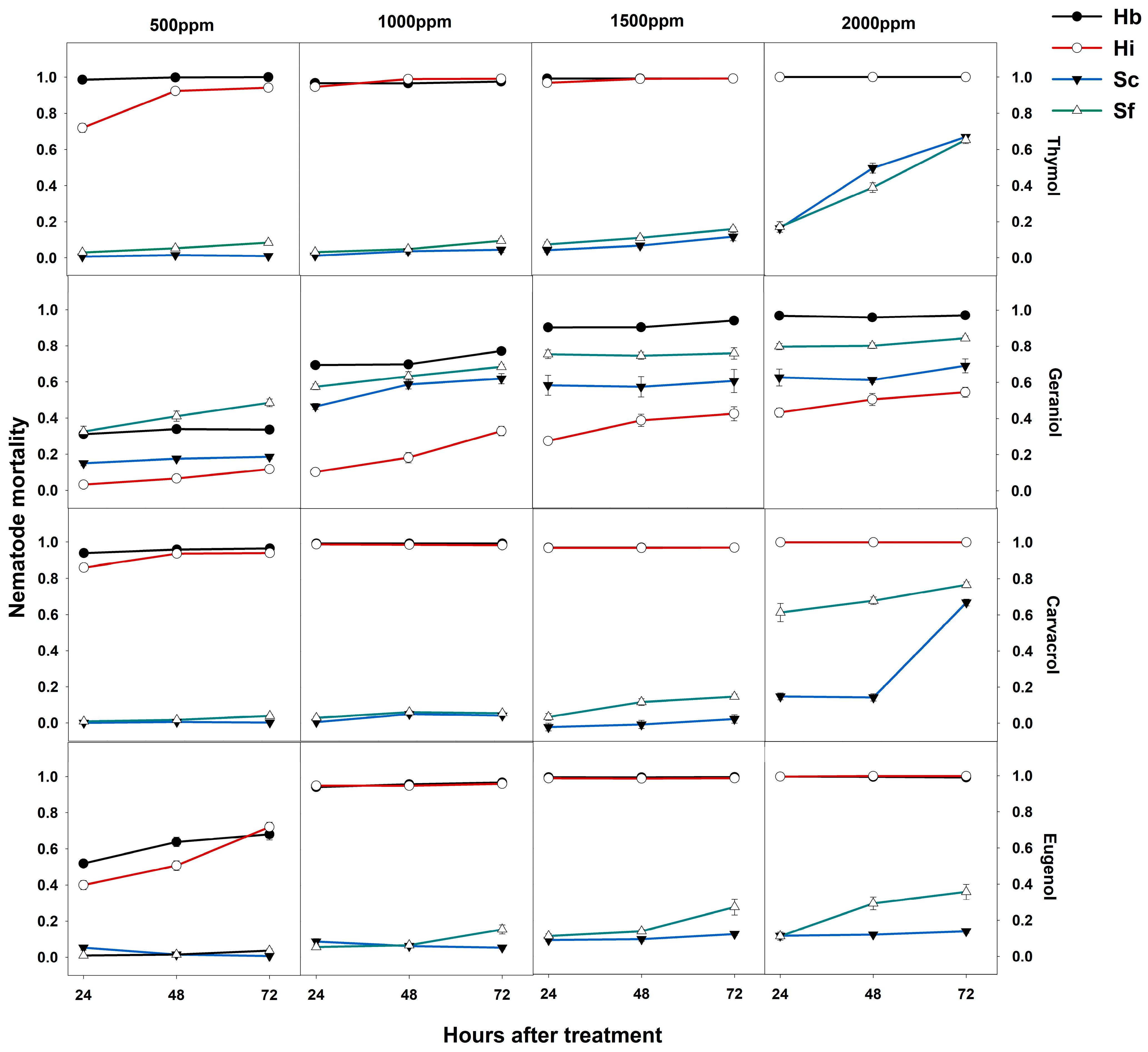
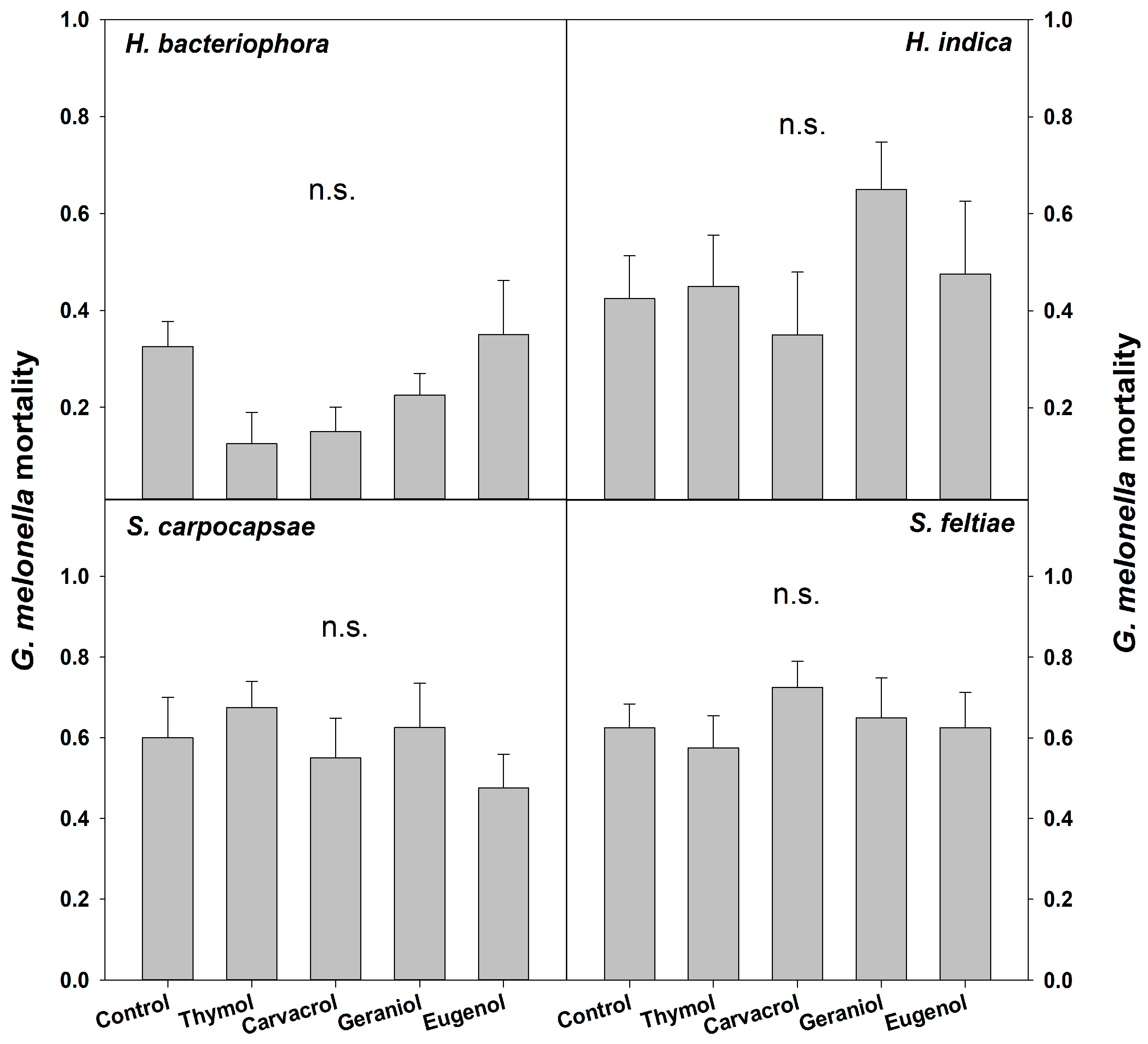
3. Results
4. Discussion
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Grewal, P.S.; Ehlers, R.U.; Shapiro-Ilan, D.I. Nematodes as biocontrol agent. In Nematodes as Biocontrol Agents; CABI: Wallingford, UK, 2005; pp. 1–505. [Google Scholar] [CrossRef]
- Kaya, H.K.; Gaugler, R. Entomopathogenic Nematodes. Annual Review of Entomology. Annu. Rev. Entomol. 1993, 38, 181–206. [Google Scholar] [CrossRef]
- Lacey, L.A.; Georgis, R. Entomopathogenic Nematodes for Control of Insect Pests Above and Below Ground with Comments on Commercial Production. J. Nematol. 2012, 44, 218. [Google Scholar]
- Poinar, G.O. Taxonomy and biology of steinernematidae and heterorhabditidae. Entomopathog. Nematodes Biol. Control. 1990, 54, 23–62. [Google Scholar] [CrossRef]
- Hazir, S.; Kaya, H.K.; Stock, S.P.; Keskin, N. Entomopathogenic nematodes (Steinernematidae and Heterorhabditidae) for biological control of soil pests. Turk. J. Biol. 2003, 27, 181–202. [Google Scholar]
- Lu, D.; Macchietto, M.; Chang, D.; Barros, M.M.; Baldwin, J.; Mortazavi, A.; Dillman, A.R. Activated entomopathogenic nematode infective juveniles release lethal venom proteins. PLoS Pathog. 2017, 13, e1006302. [Google Scholar] [CrossRef]
- Yüksel, E.; Özdemir, E.; Albayrak Delialioğlu, R.; Canhilal, R. Insecticidal activities of the local entomopathogenic nematodes and cell-free supernatants from their symbiotic bacteria against the larvae of fall webworm, Hyphantria cunea. Exp. Parasitol. 2022, 242, 108380. [Google Scholar] [CrossRef]
- Boncan, D.A.T.; Tsang, S.S.; Li, C.; Lee, I.H.; Lam, H.M.; Chan, T.F.; Hui, J.H. Terpenes and terpenoids in plants: Interactions with environment and insects. Int. J. Mol. Sci. 2020, 21, 7382. [Google Scholar] [CrossRef] [PubMed]
- Gershenzon, J.; Dudareva, N. The function of terpene natural products in the natural world. Nature Chemical Biology. Nat. Chem. Biol. 2007, 3, 408–414. [Google Scholar] [CrossRef] [PubMed]
- Singh, B.; Sharma, R.A. Plant terpenes: Defense responses, phylogenetic analysis, regulation and clinical applications. 3 Biotech 2015, 5, 129–151. [Google Scholar] [CrossRef]
- Lee, S.; Peterson, C.J.; Coats, J.R. Fumigation toxicity of monoterpenoids to several stored product insects. J. Stored Prod. Res. 2003, 39, 77–85. [Google Scholar] [CrossRef]
- Hammer, K.A.; Carson, C.F.; Riley, T.V. Antifungal activity of the components of Melaleuca alternifolia (tea tree) oil. J. Appl. Microbiol. 2003, 95, 853–860. [Google Scholar] [CrossRef] [PubMed]
- Friedman, M.; Henika, P.R.; Mandrell, R.E. Bactericidal Activities of Plant Essential Oils and Some of Their Isolated Constituents against Campylobacter jejuni, Escherichia coli, Listeria monocytogenes, and Salmonella enterica. J. Food Prot. 2002, 65, 1545–1560. [Google Scholar] [CrossRef] [PubMed]
- Frank, T.; Bieri, K.; Speiser, B. Feeding deterrent effect of carvone, a compound from caraway seeds, on the slug Arion lusitanicus. Ann. Appl. Biol. 2002, 141, 93–100. [Google Scholar] [CrossRef]
- Szczepanik, M.; Dams, I.; Wawrzeńczyk, C.; Wawrzeńczyk, W. Feeding Deterrent Activity of Terpenoid Lactones with the p-Menthane System Against the Colorado Potato Beetle (Coleoptera: Chrysomelidae). Environ. Entomol. 2005, 34, 1433–1440. [Google Scholar] [CrossRef]
- Vourc’h, G.; De Garine-Wichatitsky, M.; Labbé, A.; Rosolowski, D.; Martin, J.L.; Fritz, H. Monoterpene effect on feeding choice by deer. J. Chem. Ecol. 2002, 28, 2411–2427. [Google Scholar] [CrossRef] [PubMed]
- Seiber, J.N.; Coats, J.; Duke, S.O.; Gross, A.D. Biopesticides: State of the Art and Future Opportunities. J. Agric. Food Chem. 2014, 62, 11613–11619. [Google Scholar] [CrossRef]
- Isman, M.B. Plant essential oils for pest and disease management. Crop Prot. 2000, 19, 603–608. [Google Scholar] [CrossRef]
- Koul, O.; Walia, S.; Dhaliwal, G.S. Essential oils as green pesticides: Potential and constraints. Biopestic. Int. 2008, 4, 63–84. [Google Scholar]
- Greff, B.; Sáhó, A.; Lakatos, E.; Varga, L. Biocontrol Activity of Aromatic and Medicinal Plants and Their Bioactive Components against Soil-Borne Pathogens. Plants 2023, 12, 706. [Google Scholar] [CrossRef]
- Calvet, C.; Pinochet, J.; Camprubí, A.; Estaún, V.; Rodríguez-Kábana, R. Evaluation of natural chemical compounds against root-lesion and root-knot nematodes and side-effects on the infectivity of arbuscular mycorrhizal fungi. Eur. J. Plant Pathol. 2001, 107, 601–605. [Google Scholar] [CrossRef]
- Nasiou, E.; Giannakou, I.O. The potential use of carvacrol for the control of Meloidogyne javanica. Eur. J. Plant Pathol. 2017, 149, 415–424. [Google Scholar] [CrossRef]
- Nasiou, E.; Giannakou, I.O. Effect of geraniol, a plant-based alcohol monoterpene oil, against Meloidogyne javanica. Eur. J. Plant Pathol. 2018, 152, 701–710. [Google Scholar] [CrossRef]
- Nasiou, E.; Giannakou, I.O. The potential of eugenol as a nematicidal agent against Meloidogyne javanica (Treub) Chitwood. J. Nematol. 2020, 52, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Ntalli, N.G.; Ferrari, F.; Giannakou, I.; Menkissoglu-Spiroudi, U. Phytochemistry and Nematicidal Activity of the Essential Oils from 8 Greek Lamiaceae Aromatic Plants and 13 Terpene Components. J. Agric. Food Chem. 2010, 58, 7856–7863. [Google Scholar] [CrossRef] [PubMed]
- Stavropoulou, E.; Nasiou, E.; Skiada, P.; Giannakou, I.O. Effects of four terpenes on the mortality of Ditylenchus dipsaci (Kühn) Filipjev. Eur. J. Plant Pathol. 2021, 160, 137–146. [Google Scholar] [CrossRef]
- Al-Banna, L.; Aburjai, T.; Darwish, R.M. Effect of Plant Eztracts and Essential Oils on Root-Knot Nematode. Phytopathol. Mediterr. 2003, 42, 123–128. [Google Scholar]
- Ibrahim, K.; Traboulsi, F. Effect of essential oils and plant extracts on hatching, migration and mortality of Meloidogyne incognita. Phytopathol. Mediterr. 2006, 45, 238–246. [Google Scholar]
- Soler-Serratosa, A.; Kokalis-Burelle, N.; Rodríguez-Kábana, R.; Weaver, C.F.; King, P.S. Allelochemicals for control of plant-parasitic nematodes. 1. In vivo nematicidal efficacy of thymol and thymol/benzaldehyde combinations. Nematropica 1996, 26, 57–71. [Google Scholar]
- Dillman, A.R.; Guillermin, M.L.; Lee, J.H.; Kim, B.; Sternberg, P.W.; Hallem, E.A. Olfaction shapes host-parasite interactions in parasitic nematodes. Proc. Natl. Acad. Sci. USA 2012, 109, E2324–E2333. [Google Scholar] [CrossRef]
- Ali, J.G.; Alborn, H.T.; Stelinski, L.L. Subterranean herbivore-induced volatiles released by citrus roots upon feeding by Diaprepes abbreviatus recruit entomopathogenic nematodes. J. Chem. Ecol. 2010, 36, 361–368. [Google Scholar] [CrossRef]
- Van Tol, R.W.H.M.; Van Der Sommen, A.T.C.; Boff, M.I.C.; Van Bezooijen, J.; Sabelis, M.W.; Smits, P.H. Plants protect their roots by alerting the enemies of grubs. Ecol. Lett. 2001, 4, 292–294. [Google Scholar] [CrossRef]
- Rasmann, S.; Köllner, T.G.; Degenhardt, J.; Hiltpold, I.; Toepfer, S.; Kuhlmann, U.; Gershenzon, J.; Turlings, T.C. Recruitment of entomopathogenic nematodes by insect-damaged maize roots. Nature 2005, 434, 732–737. [Google Scholar] [CrossRef]
- Filgueiras, C.C.; Willett, D.S.; Moino, A.; Pareja, M.; El Borai, F.; Dickson, D.W.; Stelinski, L.L.; Duncan, L.W. Stimulation of the salicylic acid pathway aboveground recruits entomopathogenic nematodes belowground. PLoS ONE 2016, 11, e0154712. [Google Scholar] [CrossRef]
- Laznik, Ž.; Trdan, S. Attraction Behaviors of Entomopathogenic Nematodes (Steinernematidae and Heterorhabditidae) to Synthetic Volatiles Emitted by Insect Damaged Potato Tubers. J. Chem. Ecol. 2016, 42, 314–322. [Google Scholar] [CrossRef] [PubMed]
- Laznik, Ž.; Trdan, S. An investigation on the chemotactic responses of different entomopathogenic nematode strains to mechanically damaged maize root volatile compounds. Exp. Parasitol. 2013, 134, 349–355. [Google Scholar] [CrossRef]
- Jagodič, A.; Ipavec, N.; Trdan, S.; Laznik, Ž. Attraction behaviors: Are synthetic volatiles, typically emitted by insect-damaged Brassica nigra roots, navigation signals for entomopathogenic nematodes (Steinernema and Heterorhabditis)? BioControl 2017, 62, 515–524. [Google Scholar] [CrossRef]
- Laznik, Ž.; Košir, I.J.; Košmelj, K.; Murovec, J.; Jagodič, A.; Trdan, S.; Ačko, D.K.; Flajšman, M. Effect of Cannabis sativa L. root, leaf and inflorescence ethanol extracts on the chemotrophic response of entomopathogenic nematodes. Plant Soil 2020, 455, 367–379. [Google Scholar] [CrossRef]
- Kergunteuil, A.; Röder, G.; Rasmann, S. Environmental gradients and the evolution of tri-trophic interactions. Ecol. Lett. 2019, 22, 292–301. [Google Scholar] [CrossRef]
- Lewis, E.; Gaugler, R.; Harrison, R. Entomopathogenic nematode host finding: Response to host contact cues by cruise and ambush foragers. Parasitology 1992, 105, 309–315. [Google Scholar] [CrossRef]
- Zhang, X.; Li, L.; Kesner, L.; Robert, C.A.M. Chemical host-seeking cues of entomopathogenic nematodes. Curr. Opin. Insect Sci. 2021, 44, 72–81. [Google Scholar] [CrossRef]
- Abbott, W.S. A method of computing the effectiveness of an insecticide. J. Econ. Entomol. 1925, 18, 265–267. [Google Scholar] [CrossRef]
- Trailović, S.M.; Marjanović, D.S.; Nedeljković Trailović, J.; Robertson, A.P.; Martin, R.J. Interaction of carvacrol with the Ascaris suum nicotinic acetylcholine receptors and gamma-aminobutyric acid receptors, potential mechanism of antinematodal action. Parasitol. Res. 2015, 114, 3059–3068. [Google Scholar] [CrossRef]
- Katiki, L.M.; Barbieri, A.M.E.; Araujo, R.C.; Veríssimo, C.J.; Louvandini, H.; Ferreira, J.F.S. Synergistic interaction of ten essential oils against Haemonchus contortus in vitro. Vet. Parasitol. 2017, 243, 47–51. [Google Scholar] [CrossRef]
- Abdel Rasoul, M.A. Evaluation of nematicidal effects of monoterpenes against root-knot nematode, Meloidogyne incognita. J. Plant Prot. Pathol. 2013, 4, 445–456. [Google Scholar] [CrossRef]
- Choi, I.; Kim, J.; Shin, S.; Park, I. Nematicidal activity of monoterpenoids against the pine wood nematode (Bursaphelenchus xylophilus). Russ. J. Nematol. 2007, 15, 35. [Google Scholar]
- Blaxter, M.L.; De Ley, P.; Garey, J.R.; Liu, L.X.; Scheldeman, P.; Vierstraete, A.; Vanfleteren, J.R.; Mackey, L.Y.; Dorris, M.; Frisse, L.M.; et al. A molecular evolutionary framework for the phylum Nematoda. Nature 1998, 392, 71–75. [Google Scholar] [CrossRef]
- Poinar, G.O. Origins and phylogenetic relationships of the entomophilic rhabditids, Heoerorhabditis and Steïnernema. Fundam. Appl. Nematol. 1993, 16, 333–338. [Google Scholar]
- Gaugler, R. Biological control potential of neoaplectanid nematodes. J. Nematol. 1981, 13, 241. [Google Scholar]
- Strong, D.R. Populations of entomopathogenic nematodes in foodwebs. In Entomopathogenic Nematology; CABI: Wallingford, UK, 2002; pp. 225–240. [Google Scholar] [CrossRef]
- Willett, D.S.; Alborn, H.T.; Duncan, L.W.; Stelinski, L.L. Social Networks of Educated Nematodes. Sci. Rep. 2015, 5, 14388. [Google Scholar] [CrossRef]
- Koppenhöfer, A.M.; Fuzy, E.M. Early timing and new combinations to increase the efficacy of neonicotinoid-entomopathogenic nematode (Rhabditida: Heterorhabditidae) combinations against white grubs (Coleoptera: Scarabaeidae). Pest Manag. Sci. 2008, 64, 725–735. [Google Scholar] [CrossRef]
- Nishimatsu, T.; Jackson, A.J. Interaction of Insecticides, Entomopathogenic Nematodes, and Larvae of the Western Corn Rootworm (Coleoptera: Chrysomelidae). J. Econ. Entomol. 1998, 91, 410–418. [Google Scholar] [CrossRef] [PubMed]
- Shapiro-Ilan, D.I.; Cottrell, T.E.; Wood, B.W. Effects of combining microbial and chemical insecticides on mortality of the pecan weevil (Coleoptera: Curculionidae). J. Econ. Entomol. 2011, 104, 14–20. [Google Scholar] [CrossRef] [PubMed]
- Duncan, L.W.; Graham, J.H.; Dunn, D.C.; Zellers, J.; McCoy, C.W.; Nguyen, K. Incidence of endemic entomopathogenic nematodes following application of Steinernema riobrave for control of Diaprepes abbreviatus. J. Nematol. 2003, 35, 178–186. [Google Scholar] [PubMed]
- Duncan, L.W.; Graham, J.H.; Zellers, J.; Bright, D.; Dunn, D.C.; El-Borai, F.E.; Porazinska, D. Food web responses to augmenting the entomopathogenic nematodes in bare and animal manure-mulched soil. J. Nematol. 2007, 39, 176. [Google Scholar]
- Manrakhan, A.; Daneel, J.H.; Moore, S.D. The impact of naturally occurring entomopathogenic nematodes on false codling moth, Thaumatotibia leucotreta (Lepidoptera: Tortricidae), in citrus orchards. Biocontrol Sci. Technol. 2014, 24, 241–245. [Google Scholar] [CrossRef]
| Effect | Mortality | ||
|---|---|---|---|
| Source | DF | F | p |
| Between exposure intervals | |||
| Intercept | 1 | 92,096.90 | <0.0001 |
| Nematode species | 3 | 9280.10 | <0.0001 |
| Terpene | 3 | 100.3 | <0.0001 |
| Terpene dose | 3 | 1389.30 | <0.0001 |
| Nematode species × Terpene | 9 | 1388.90 | <0.0001 |
| Nematode species × Terpene dose | 9 | 54.7 | <0.0001 |
| Terpene × Terpene dose | 9 | 109.5 | <0.0001 |
| Nematode species × Terpene × Terpene dose | 27 | 50.5 | <0.0001 |
| Within exposure intervals | |||
| Exposure | 2 | 338.7 | <0.0001 |
| Exposure × Nematode species | 6 | 34.5 | <0.0001 |
| Exposure × Terpene | 6 | 6.8 | <0.0001 |
| Exposure × Terpene dose | 6 | 28.1 | <0.0001 |
| Exposure × Nematode species × Terpene | 18 | 17.3 | <0.0001 |
| Exposure × Nematode species × Terpene dose | 18 | 26.9 | <0.0001 |
| Exposure × Terpene × Terpene dose | 18 | 17.4 | <0.0001 |
| Exposure × Nematode species × Terpene × Terpene dose | 54 | 13.1 | <0.0001 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kotsinis, V.; Dritsoulas, A.; Ntinokas, D.; Giannakou, I.O. Nematicidal Effects of Four Terpenes Differ among Entomopathogenic Nematode Species. Agriculture 2023, 13, 1143. https://doi.org/10.3390/agriculture13061143
Kotsinis V, Dritsoulas A, Ntinokas D, Giannakou IO. Nematicidal Effects of Four Terpenes Differ among Entomopathogenic Nematode Species. Agriculture. 2023; 13(6):1143. https://doi.org/10.3390/agriculture13061143
Chicago/Turabian StyleKotsinis, Vasileios, Alexandros Dritsoulas, Dionysios Ntinokas, and Ioannis O. Giannakou. 2023. "Nematicidal Effects of Four Terpenes Differ among Entomopathogenic Nematode Species" Agriculture 13, no. 6: 1143. https://doi.org/10.3390/agriculture13061143
APA StyleKotsinis, V., Dritsoulas, A., Ntinokas, D., & Giannakou, I. O. (2023). Nematicidal Effects of Four Terpenes Differ among Entomopathogenic Nematode Species. Agriculture, 13(6), 1143. https://doi.org/10.3390/agriculture13061143







