Non-Destructive Appraisal of Macro- and Micronutrients in Persimmon Leaves Using Vis/NIR Hyperspectral Imaging
Abstract
1. Introduction
2. Materials and Methods
2.1. Field Experiment
2.2. Spectral Measurements of the Leaves
2.3. Ionomic Leaf Analysis
2.4. Data Analysis
3. Results and Discussion
3.1. Foliar Nutrient Concentration
3.2. Predictive Analyses
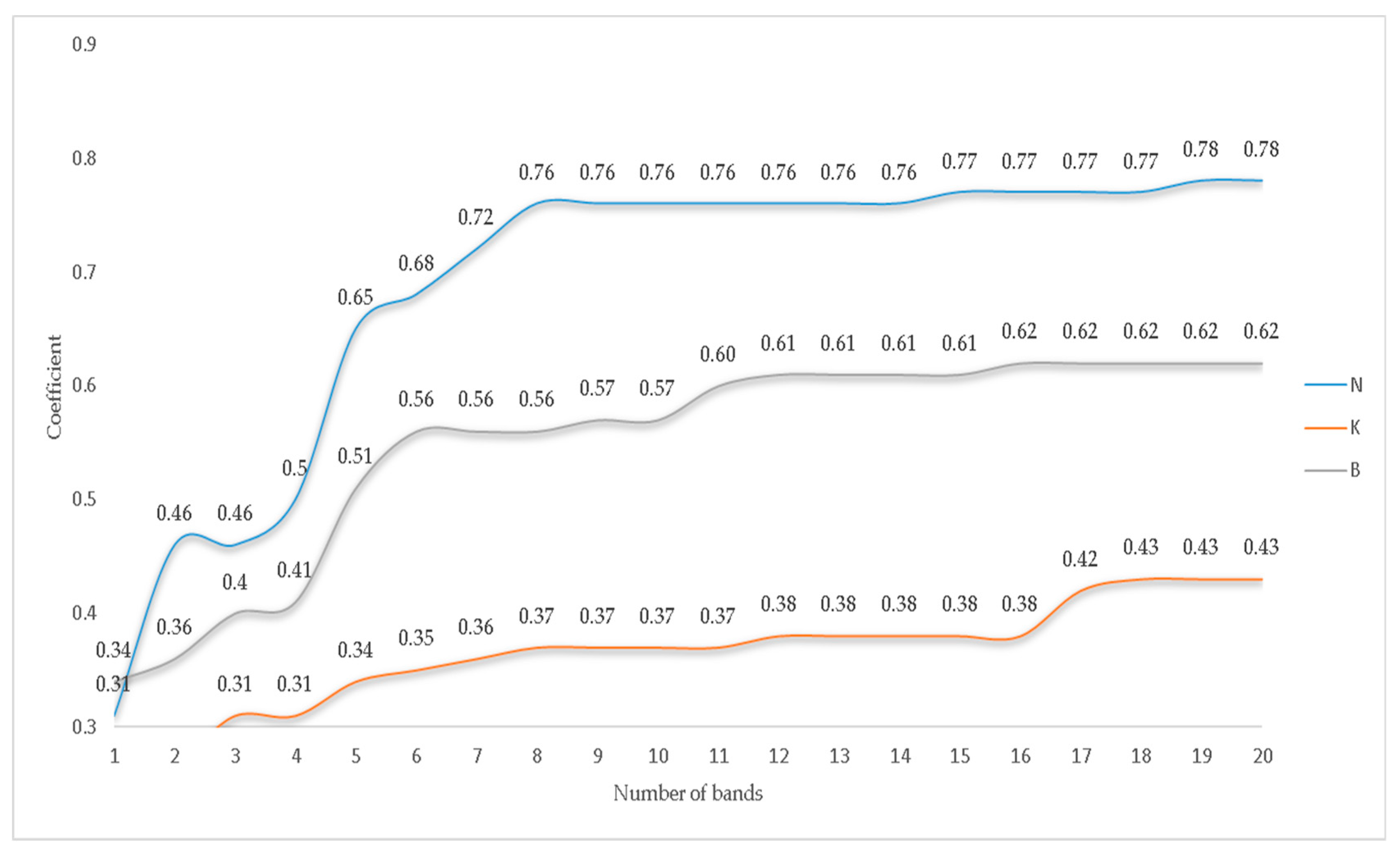
3.3. Selection of Optimal Wavelengths
3.4. Discussion
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Albiach, R.; Climent, C.; Canet, R.; Pomares, F. Soil Fertility and Nutritional State of Persimmon Rojo Brillante Plantations in the Ribera Alta (Valencia, Spain). Commun. Soil Sci. Plant Anal. 2012, 43, 2767–2776. [Google Scholar] [CrossRef]
- FAOSTAT. «FAOSTAT, 2017». Available online: https://www.fao.org/faostat/en/#home (accessed on 13 April 2023).
- Kirkby, E. Introduction, Definition and Classification of Nutrients; Elsevier Ltd.: Amsterdam, The Netherlands, 2011. [Google Scholar] [CrossRef]
- Hawkesford, M.; Horst, W.; Kichey, T.; Lambers, H.; Schjoerring, J.; Møller, I.S.; White, P. Functions of Macronutrients. In Marschner’s Mineral Nutrition of Higher Plants, 3rd ed.; Elsevier Inc.: Amsterdam, The Netherlands, 2011; pp. 135–189. [Google Scholar] [CrossRef]
- Broadley, M.; Brown, P.; Cakmak, I.; Rengel, Z.; Zhao, F. Function of Nutrients: Micronutrients. In Marschner’s Mineral Nutrition of Higher Plants, 3rd ed.; Elsevier Inc.: Amsterdam, The Netherlands, 2011; pp. 191–248. [Google Scholar] [CrossRef]
- Malagón, J.; Monzó, J.C. Diseño de la plantación de caqui y su manejo en los primeros años del cultivo. Agrícola Vergel Frutic. Hortic. Floric. 2015, 34, 61–67. [Google Scholar]
- Cubero, S.; Marco-noales, E.; Aleixos, N.; Barbé, S.; Blasco, J. Robhortic: A field robot to detect pests and diseases in horticultural crops by proximal sensing. Agriculture 2020, 10, 276. [Google Scholar] [CrossRef]
- Walsh, K.B.; Blasco, J.; Zude-Sasse, M.; Sun, X. Visible-NIR ‘point’ spectroscopy in postharvest fruit and vegetable assessment: The science behind three decades of commercial use. Postharvest Biol. Technol. 2020, 168, 111246. [Google Scholar] [CrossRef]
- Pandey, P.; Ge, Y.; Stoerger, V.; Schnable, J.C. High throughput in vivo analysis of plant leaf chemical properties using hyperspectral imaging. Front. Plant Sci. 2017, 8, 1348. [Google Scholar] [CrossRef]
- Lorente, D.; Aleixos, N.; Gómez-Sanchis, J.; Cubero, S.; García-Navarrete, O.L.; Blasco, J. Recent Advances and Applications of Hyperspectral Imaging for Fruit and Vegetable Quality Assessment. Food Bioprocess Technol. 2012, 5, 1121–1142. [Google Scholar] [CrossRef]
- Peron-Danaher, R.; Russell, B.; Cotrozzi, L.; Mohammadi, M.; Couture, J. Incorporating Multi-Scale, Spectrally Detected Nitrogen Concentrations into Assessing Nitrogen Use Efficiency for Winter Wheat Breeding Populations. Remote Sens. 2021, 13, 3991. [Google Scholar] [CrossRef]
- Gómez-Casero, M.T.; López-Granados, F.; Peña-Barragán, J.M.; Jurado-Expósito, M.; García-Torres, L.; Fernández-Escobar, R. Assessing Nitrogen and Potassium Deficiencies in Olive Orchards through Discriminant Analysis of Hyperspectral Data. J. Am. Soc. Hortic. Sci. 2007, 132, 611–618. [Google Scholar] [CrossRef]
- Jin, X.; Wang, L.; Zheng, W.; Zhang, X.D.; Liu, L.; Li, S.; Rao, Y.; Xuan, J. Predicting the nutrition deficiency of fresh pear leaves with a miniature near-infrared spectrometer in the laboratory. Meas. J. Int. Meas. Confed. 2022, 188, 110553. [Google Scholar] [CrossRef]
- Zhang, X.; Liu, F.; He, Y.; Gong, X. Detecting macronutrients content and distribution in oilseed rape leaves based on hyperspectral imaging. Biosyst. Eng. 2013, 115, 56–65. [Google Scholar] [CrossRef]
- Christensen, L.K.; Bennedsen, B.S.; Jørgensen, R.N.; Nielsen, H. Modelling nitrogen and phosphorus content at early growth stages in spring barley using hyperspectral line scanning. Biosyst. Eng. 2004, 88, 19–24. [Google Scholar] [CrossRef]
- Yanli, L.; Qiang, L.; Shaolan, H.; Shilai, Y.; Xuefeng, L.; Rangjin, X.; Yongqiang, Z.; Lie, D. Prediction of nitrogen and phosphorus contents incitrus leavesbased onhyperspectral imaging. Int. J. Agric. Biol. Eng. 2015, 8, 80–88. [Google Scholar]
- Li, D.; Wang, C.; Jiang, H.; Peng, Z.; Yang, J.; Su, Y.; Song, J.; Chen, S. Monitoring litchi canopy foliar phosphorus content using hyperspectral data. Comput. Electron. Agric. 2018, 154, 176–186. [Google Scholar] [CrossRef]
- Visconti, F.; de Paz, J.M. Non-destructive assessment of chloride in persimmon leaves using a miniature visible near-infrared spectrometer. Comput. Electron. Agric. 2019, 164, 104894. [Google Scholar] [CrossRef]
- Bremner, J.M. Inorganic forms of nitrogen. Agronomy 1965, 9, 1179–1237. [Google Scholar]
- Savitzky, A.; Golay, M.J.E. Smoothing and differentiation of data by simplified least squares procedures. Anal. Chem. 1964, 36, 1627–1639. [Google Scholar] [CrossRef]
- Barnes, R.J.; Dhanoa, M.S.; Lister, S.J. Standard Normal Variate Transformation and De-Trending of Near-Infrared Diffuse Reflectance Spectra. Appl. Spectrosc. 1989, 43, 772–777. [Google Scholar] [CrossRef]
- Li, Y.; Sun, Y.; Jiang, J.; Liu, J. Spectroscopic determination of leaf chlorophyll content and color for genetic selection on Sassafras tzumu. Plant Methods 2019, 15, 73. [Google Scholar] [CrossRef]
- Burnett, A.C.; Anderson, J.; Davidson, K.J.; Ely, K.S.; Lamour, J.; Li, Q.; Morrison, B.D.; Yang, D.; Rogers, A.; Serbin, S.P. A best-practice guide to predicting plant traits from leaf-level hyperspectral data using partial least squares regression. J. Exp. Bot. 2021, 72, 6175–6189. [Google Scholar] [CrossRef]
- Geladi, P.; Kowalski, B.R. Partial least-squares regression: A tutorial. Anal. Chim. Acta 1986, 185, 1–17. [Google Scholar] [CrossRef]
- Stone, M.; Brooks, R.J. Continuum Regression: Cross-Validated Sequentially Constructed Prediction Embracing Ordinary Least Squares, Partial Least Squares and Principal Components Regression. J. R. Stat. Soc. Ser. B (Methodol.) 1990, 52, 237–258. [Google Scholar] [CrossRef]
- Tang, G.; Huang, Y.; Tian, K.; Song, X.; Yan, H.; Hu, J.; Xiong, Y.; Min, S. A new spectral variable selection pattern using competitive adaptive reweighted sampling combined with successive projections algorithm. Analyst 2014, 139, 4894. [Google Scholar] [CrossRef] [PubMed]
- Chong, I.G.; Jun, C.H. Performance of some variable selection methods when multicollinearity is present. Chemom. Intell. Lab. Syst. 2005, 78, 103–112. [Google Scholar] [CrossRef]
- Sorochan Armstrong, M.D.; de la Mata, A.P.; Harynuk, J.J. Review of Variable Selection Methods for Discriminant-Type Problems in Chemometrics. Front. Anal. Sci. 2022, 2, 867938. [Google Scholar] [CrossRef]
- Stoessel, D.; Stellmann, J.P.; Willing, A.; Behrens, B.; Rosenkranz, S.C.; Hodecker, S.C.; Stürner, K.H.; Reinhardt, S.; Fleischer, S.; Deuschle, C.; et al. Metabolomic Profiles for Primary Progressive Multiple Sclerosis Stratification and Disease Course Monitoring. Front. Hum. Neurosci. 2018, 12, 226. [Google Scholar] [CrossRef]
- Viscarra Rossel, R.A. ParLeS: Software for chemometric analysis of spectroscopic data. Chemom. Intell. Lab. Syst. 2008, 90, 72–83. [Google Scholar] [CrossRef]
- Petisco, C.; García-Criado, B.; Vázquez de Aldana, B.R.; Zabalgogeazcoa, I.; Mediavilla, S.; García-Ciudad, A. Use of near-infrared reflectance spectroscopy in predicting nitrogen, phosphorus and calcium contents in heterogeneous woody plant species. Anal. Bioanal. Chem. 2005, 382, 458–465. [Google Scholar] [CrossRef]
- Ye, X.; Abe, S.; Zhang, S. Estimation and mapping of nitrogen content in apple trees at leaf and canopy levels using hyperspectral imaging. Precis. Agric. 2020, 21, 198–225. [Google Scholar] [CrossRef]
- Malmir, M.; Tahmasbian, I.; Xu, Z.; Farrar, M.B.; Bai, S.H. Prediction of macronutrients in plant leaves using chemometric analysis and wavelength selection. J. Soils Sediments 2020, 20, 249–259. [Google Scholar] [CrossRef]
- Abenina, M.I.A.; Maja, J.M.; Cutulle, M.; Melgar, J.C.; Liu, H. Prediction of Potassium in Peach Leaves Using Hyperspectral Imaging and Multivariate Analysis. AgriEngineering 2022, 4, 400–413. [Google Scholar] [CrossRef]
- de Oliveira, L.F.R.; Santana, R.C. Estimation of leaf nutrient concentration from hyperspectral reflectance in Eucalyptus using partial least squares regression. Sci. Agric. 2020, 77. [Google Scholar] [CrossRef]
- De Silva, A.L.; Trueman, S.J.; Kämper, W.; Wallace, H.M.; Nichols, J.; Hosseini Bai, S. Hyperspectral Imaging of Adaxial and Abaxial Leaf Surfaces as a Predictor of Macadamia Crop Nutrition. Plants 2023, 12, 558. [Google Scholar] [CrossRef] [PubMed]
- Cotrozzi, L.; Peron, R.; Tuinstra, M.R.; Mickelbart, M.V.; Couture, J.J. Spectral Phenotyping of Physiological and Anatomical Leaf Traits Related with Maize Water Status. Plant Physiol. 2020, 184, 1363–1377. [Google Scholar] [CrossRef]
- Cotrozzi, L.; Couture, J.J. Hyperspectral assessment of plant responses to multi-stress environments: Prospects for managing protected agrosystems. Plants People Planet 2020, 2, 244–258. [Google Scholar] [CrossRef]


| Treatment | N | K2O | P2O3 | CaO | MgO |
|---|---|---|---|---|---|
| T1 (N-0%) | 0 | 110 | 38.8 | 62.5 | 25.3 |
| T2 (N-33%) | 35 | 110 | 38.8 | 62.5 | 25.3 |
| T3 (N-50%) | 53 | 110 | 38.8 | 62.5 | 25.3 |
| T4 (K2O-0%) | 106 | 0 | 38.8 | 62.5 | 25.3 |
| T5 (K2O-50%) | 106 | 55 | 38.8 | 62.5 | 25.3 |
| Control (N and K2O-100%) | 106 | 110 | 38.8 | 62.5 | 25.3 |
| Nutrient | Average Cycle | Standard Deviation | Coefficient of Variation | July | Cycle | Range | Std Bias | Kurtosis |
|---|---|---|---|---|---|---|---|---|
| Min–Max | Min–Max | |||||||
| N | 1.74 | 0.39 | 0.22 | 1.62–2.30 | 1.06–2.82 | 1.75 | 2.48 | 1.09 |
| P | 0.11 | 0.06 | 0.55 | 0.13–0.30 | 0.04–0.30 | 0.26 | 8.87 | 5.6 |
| K | 1.94 | 0.53 | 0.28 | 1.08–2.30 | 0.83–2.96 | 2.13 | 0.71 | 2.53 |
| Ca | 2.7 | 1.41 | 0.52 | 0.30–2.11 | 0.30–6.52 | 6.22 | 0.81 | 0.17 |
| Mg | 0.5 | 0.2 | 0.39 | 0.17–0.58 | 0.17–0.95 | 0.78 | 0.68 | 1.26 |
| S | 0.18 | 0.04 | 0.21 | 0.20–0.25 | 0.11–0.29 | 0.18 | 1.94 | 1.41 |
| Na | 0.01 | 0.01 | 0.79 | 0.01–0.06 | 0.005–0.06 | 0.07 | 5.73 | 4.16 |
| Fe | 39.52 | 15.45 | 0.39 | 31.00–69.00 | 13.22–81.77 | 68.54 | 3.36 | 1.36 |
| Zn | 6.62 | 2.91 | 0.44 | 4.00–14.00 | 2.02–15.68 | 13.66 | 3.79 | 0.32 |
| Mn | 181.07 | 72.92 | 0.4 | 32.00–118.00 | 32.81–295.02 | 262.21 | 2.66 | 2.27 |
| B | 48.76 | 22.89 | 0.47 | 12.00–26.00 | 12.18–102.10 | 89.92 | 0.06 | 2.49 |
| Cu | 3.48 | 1.53 | 0.44 | 4.00–9.00 | 1.33–8.97 | 7.64 | 5.07 | 1.80 |
| Nutrient | Pre-Treatment | LV | Calibration | Cross-Validation | Test | |||
|---|---|---|---|---|---|---|---|---|
| RMSEC | R2 | RMSECV | R2 | RMSEP | R2 | |||
| N | MC + 1D | 6 | 0.14 | 0.84 | 0.14 | 0.83 | 0.16 | 0.80 |
| P | Raw spectra | 5 | 0.01 | 0.69 | 0.01 | 0.68 | 0.01 | 0.62 |
| K | Mean centre | 10 | 0.88 | 0.68 | 0.90 | 0.64 | 0.83 | 0.67 |
| Ca | Raw spectra | 12 | 0.68 | 0.65 | 0.73 | 0.60 | 0.77 | 0.54 |
| Mg | Raw spectra | 11 | 0.11 | 0.65 | 0.11 | 0.60 | 0.12 | 0.58 |
| S | Raw spectra | 12 | 0.03 | 0.45 | 0.03 | 0.37 | 0.03 | 0.37 |
| Na | Raw spectra | 3 | 0.01 | 0.28 | 0.01 | 0.20 | 0.01 | 0.27 |
| Fe | Raw spectra | 12 | 9.43 | 0.44 | 9.91 | 0.38 | 10.32 | 0.34 |
| Zn | Raw spectra | 12 | 2.58 | 0.50 | 2.82 | 0.40 | 3.37 | 0.20 |
| Mn | MC + SNV | 9 | 48.81 | 0.24 | 51.45 | 0.16 | 49.85 | 0.22 |
| B | MC | 11 | 8.69 | 0.72 | 9.34 | 0.70 | 9.59 | 0.69 |
| Cu | MC + 1D | 6 | 0.84 | 0.31 | 0.89 | 0.23 | 0.93 | 0.24 |
| Nutrient | Wavelengths (nm) | Pre-Treatment | * LV | Calibration | Cross-Validation | Test | |||
|---|---|---|---|---|---|---|---|---|---|
| RMSEC | R2 | RMSECV | R2 | RMSEP | R2 | ||||
| N | 620, 720, 760, 780, 880, 940, 950, 960 | MC | 3 | 0.16 | 0.79 | 0.17 | 0.77 | 0.18 | 0.76 |
| B | 730, 700, 670, 720, 740, 660, 850, 760, 570, 800, 910 | MC | 5 | 10.12 | 0.62 | 10.27 | 0.62 | 10.42 | 0.61 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Acosta, M.; Rodríguez-Carretero, I.; Blasco, J.; de Paz, J.M.; Quiñones, A. Non-Destructive Appraisal of Macro- and Micronutrients in Persimmon Leaves Using Vis/NIR Hyperspectral Imaging. Agriculture 2023, 13, 916. https://doi.org/10.3390/agriculture13040916
Acosta M, Rodríguez-Carretero I, Blasco J, de Paz JM, Quiñones A. Non-Destructive Appraisal of Macro- and Micronutrients in Persimmon Leaves Using Vis/NIR Hyperspectral Imaging. Agriculture. 2023; 13(4):916. https://doi.org/10.3390/agriculture13040916
Chicago/Turabian StyleAcosta, Maylin, Isabel Rodríguez-Carretero, José Blasco, José Miguel de Paz, and Ana Quiñones. 2023. "Non-Destructive Appraisal of Macro- and Micronutrients in Persimmon Leaves Using Vis/NIR Hyperspectral Imaging" Agriculture 13, no. 4: 916. https://doi.org/10.3390/agriculture13040916
APA StyleAcosta, M., Rodríguez-Carretero, I., Blasco, J., de Paz, J. M., & Quiñones, A. (2023). Non-Destructive Appraisal of Macro- and Micronutrients in Persimmon Leaves Using Vis/NIR Hyperspectral Imaging. Agriculture, 13(4), 916. https://doi.org/10.3390/agriculture13040916








