Abstract
Chlorpyrifos® (Thaion Agro Chemical CO., Ltd. Yannawa, Bangkok 10120, Thailand) (an almost water insoluble organophosphate insecticide) has been extensively used, resulting in the presence as a surface contaminant in foodstuffs, surface streams and soils. It is thus critically essential to develop methods to degrade or remove and eliminate this pollutant from environments. Chlorpyrifos® has very limited solubility and so it is primarily a contaminant of the surfaces of foodstuffs. We present the effect of magnetically treated water (MTW) to remove Chlorpyrifos® contaminating in Brassica chinensis Linn., a commonly eaten vegetable in Thailand and globally. Samples were washed with magnetically treated water (MTW) prior to detection of Chlorpyrifos® with GC-MS (Gas Chromatograph-Mass Spectroscopy). Chlorpyrifos® was removed by a factor of 413 times (−99.7%) by MTW compared to the un-washed sample, whereas the removal factor for tap water washing was only 9.6 (−89%). The MTW washed material easily passed safety criteria (Maximum Residue Load—MRL), but the tap water washed vegetables did not do so reliably. Although Chlorpyrifos® may be banned in many countries, the binding properties of replacement organophosphates are likely to be similar, and so our results should generalize to pesticides in the organophosphate chemical class that are not readily water soluble.
1. Introduction
The products of agriculture such as fruits and vegetables are an important component of human food in view of their health benefits and their nutritional worth. The demand for “healthy” fruits and vegetables is high. Therefore, clean and healthy food that is free of pathogens, agricultural chemicals and pesticides has become one of the leading interests and demands of consumers. The high demands for fruits and vegetables have led to the use of fertilizers and pesticides in agriculture to increase the yield, improve the quality, and extend the storage life of food crops. Destruction and wastage of agricultural products is causing starvation [1]. Crop losses due to pest infestation can be as high as 100% if they are not controlled [2]. To prevent losses from pests and diseases, pesticides have played an important role in industrialized agriculture. However, if not well-controlled, the use of pesticides may result in unsafe residues in agricultural produce, which in turn results in excessive pesticide exposure to farmers and consumers, causing health hazards [3]. In addition to pesticide contamination, the disposal of agricultural products may cause pollution during the recycling process. Insecticidal poisoning may be confused with microbial food poisoning [4]. Organophosphates (OP) are a pesticide group that is widely used as insecticides. Most organophosphorus insecticides possess a similar structure, containing three phosphoester linkages and are hence often termed phosphotriesters. This group of pesticides has been reported as presenting a health risk for acute neurotoxicity attributed to their ability to suppress acetylcholinesterase (AchE) [5], and in the case of poisoning in human beings, various clinical effects can occur. Chlorpyrifos® is one example of a pesticide in the organophosphate group that is widely used and is a widespread surface contaminant of foodstuffs, natural water and ecosystems.
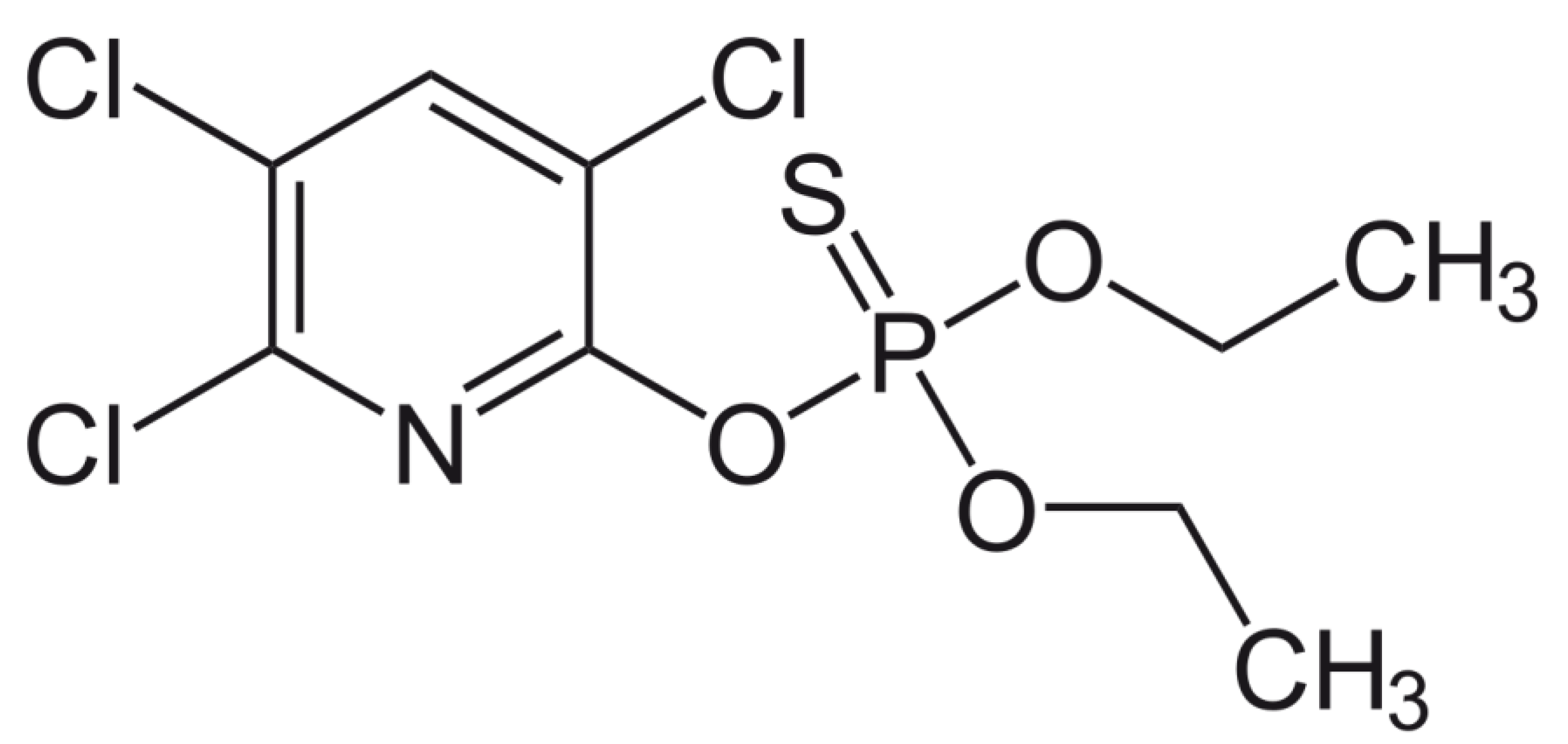
Chlorpyrifos®, [O,O-diethyl O-[3,5,6-trichloro-2-pyridyl) phosphorothioate] is one of the most extensively used broad-spectrum insecticides (see structure Figure 1). Its phosphorus is linked to a sulfur with a double bond (P=S). Its structure is representative of the class of organophosphate insecticides (phosphorothioates), all of which have a similar core chemical structure. The chlorination of the sidegroup of Chlorpyrifos® is performed to improve its chemical stability. Chlorpyrifos® has been widely used globally as an insecticide to control crop pests in agriculture and to reduce household insect pests. Its extensive use and associated serious soil and water contamination has earned increasing environmental concern, and it poses risks to human health. Chlorpyrifos® may cause either acute or chronic toxicity in consumers after consumption and handling of contaminated products [6]. Health risks may range from acute: headache, coughing, stomach pain, nausea, vomiting and diarrhea to chronic in the form of endocrine disruption, reproduction and immune system malfunction and development of some cancers [7]. Inevitably the use of pesticides will increase in the future.
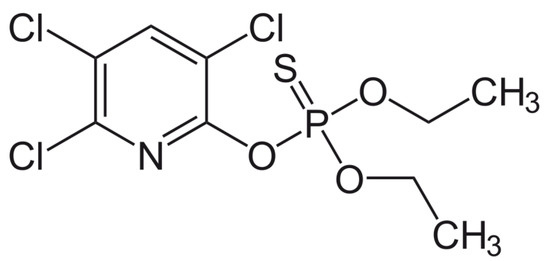
Figure 1.
Chlorpyrifos® structure. Its structure is representative of the class of organophosphate insecticides (phosphorothioates), but is a particularly insoluble organophosphate and has chlorine groups to limit and slow down biodegradation. It is a weak electrolyte.
Like many similar organophosphates, Chlorpyrifos® is only very sparingly soluble in water (≈1.4 mg L−1) and is usually used with a dispersant (detergent or emulsifier) [8,9,10]. Compared to Malathion® (solubility ≈ 150 mg L−1: [10]) it is practically insoluble in water. The target pests for such insecticides designed as surface contact insecticides are biting and chewing insects, not sap sucking pests. In contrast, Dimethoate® is a readily soluble organophosphate designed to kill sucking insects and is much more water soluble to allow it to penetrate plant xylem and phloem systems [10]. Dispersants are easily washed away, the consequence of which is that any Chlorpyrifos® that binds to the surface of a vegetable or other biological material becomes very difficult to remove. Washing with detergent to remove Chlorpyrifos®, although probably effective for some fruits, would not be an acceptable practice for vegetables [9].
The elimination of insecticide residue in agricultural products is very important in food safety. Currently, residue of pesticides on vegetables, including contamination with agricultural pesticides, is a major concern to both the producer and consumer, leading to development of methods to remove the pesticides before marketing or rules about how long before harvesting is it permissible to use a pesticide. The toxicity Chlorpyrifos® to human health has been reported [11] and importantly breakdown products are documented to have insidious effects [12]. For these reasons, detection methods for traces of harmful pesticide such as Chlorpyrifos® are needed usually by Gas Chromatograph-Mass Spectroscopy (GC-MS) [13,14], but such analyses are both labor intensive and expensive.
There are several methods to degrade applied pesticides. To remove insecticides from agricultural products, washing with water is widely used and recommended in the household and restaurant trade [9,13]. An extensive review explained that rinsing vegetables with tap water significantly decreased the level of several insecticides, including cypermethrin®, fenithrotion®, dimethoate® and trochlorfon® [15]. However, a study of the insecticide removal effects of tap water showed that water was also unable to remove residues of Bifenthrin®, a fluorinated synthetic pyrethroid-type insecticide, in agricultural product samples [9]. Similar to Chlorpyrifos®, it is almost insoluble and used with detergent carriers. Moreover, hand washing methods caused significant health risks not only from the amount of the insecticide Chlorpyrifos® remaining, but also the transfer of the compounds to the hands of those washing the vegetables [16]. Routine washing methods have been reported to remove Chlorpyrifos® by as little as only about 50% [17]. Other methods such as dipping in 2% salt solution, dipping in lemon juice, and dipping in acetic acid solution were mentioned by Rao et al. [17]. However, none of the previously mentioned domestic methods can decontaminate Chlorpyrifos® by much more than 50%. Later, various methods were developed to degrade such pesticides including gamma irradiation, and exposure to natural sunlight [18] or ozone [19]. Recently, microbial biodegradation was carried out using immobilized microorganism technology [5,20]. Microbial degradation is an environmentally friendly and low-cost approach of pesticide abatement that shows potential to remove Chlorpyrifos® residue [3,9], but microbial degradation can be slower than is sometimes thought [21]. However, the ability to remove Chlorpyrifos® by microbial action can generally only be carried out on a small scale, and the geometry of some food materials make it unsuitable.
Chlorpyrifos® has a reputation of breaking down rapidly in the environment and so it is logical to impose protocols for not using it close to harvesting, but this reputation for rapid breakdown might not be entirely justified: the chlorinated sidegroup (Figure 1) discourages biodegradation [21]. A more sinister problem is that the breakdown products of biodegradation might not be benign. Velmurugan et al. [12] found that microbial breakdown of organophosphates in the human gut induces glucose intolerance (diabetes) in humans. A reputation for rapid breakdown in the environment can actually be counter-productive because it encourages the careless use of pesticides thinking that they will “break down” quickly anyway. Thus, new pesticide removal methods are needed to improve the removal efficiency and apply the technique in field practice.
Based on the literature reviews carried out on magnetized water during the last few years, most researchers have worked on the influence of magnetic field treatments on the properties of water [5,22,23,24], especially surface tension [25] and the solubility of compounds that are typically not very soluble in water [26]. Various researchers have studied the effects of magnetized water on the morphology of precipitated calcium carbonate [27,28]. However, there is no report on the use of magnetized water on pesticide removal, many of which are polar compounds that are not very soluble in water. Considering the above, in this work we investigated the use of magnetically treated water (MTW) as a new innovation for the removal of pesticide residues in vegetables. MTW can be prepared easily by passing it through a magnetic field. Physically, when water is exposed to a magnetic field, there are changes in its properties such as raising its pH, oxygen solubility, and mineral solubility, increasing the total hardness, which has biological effects, particularly anything to do with calcium metabolism such as egg-laying in chickens [24]. Moreover, when water passes through a magnetic field, its conductivity increases [29] and the solubility of various other compounds also increase [25]. In addition, surface tension was found to be reduced and decreases hydrophobicity due to the clustering structure of the water molecules (micellization) and improves polarizing effects [25]. This has important consequences for its potential to remove surface contaminants. Even though the physical theory of the effect of MF on water is still a controversial issue, and the mechanism of magnetic field treatment is not unambiguous. However, MTW has been used mainly in industry and construction [22] owing to the changes of the physicochemical properties of water.
Some note should be made of the current status and usage of Chlorpyrifos®. The sale and distribution of Chlorpyrifos® has been banned by the Government of Thailand from 20 June 2020; however, organophosphate (phosphorothioate) insecticides are still used in Thailand (Azinphosmethyl®, Diazinon®, Dimethoate®, Dichlorvos®, Malathion®, Parathion®), as well as globally share a similar chemistry and most share similar limited solubility properties. Dimethoate® is something of an exception because it is highly water soluble to target sucking insects such as aphids and thrips.
There appears to be no report of the effects of MTW on pesticide removal from plant material, but since MTW can reduce surface tension [25], increases solubility of poorly soluble compounds (particularly calcium complexes) and removes the scaling of metallic surfaces [27], magnetically treated water (MTW) shows promise that it would be useful in the removal of adhering pesticide residues. The aim of the present study was to test the efficiency of (MTV) as a minimum technology method to remove Chlorpyrifos® contamination from vegetables and compare it to recommended washing with tap water. The binding properties of replacement organophosphate insecticides are likely to have very similar binding properties to those of Chlorpyrifos® found in the present study.
2. Materials and Methods
2.1. Magnetically Treated Water
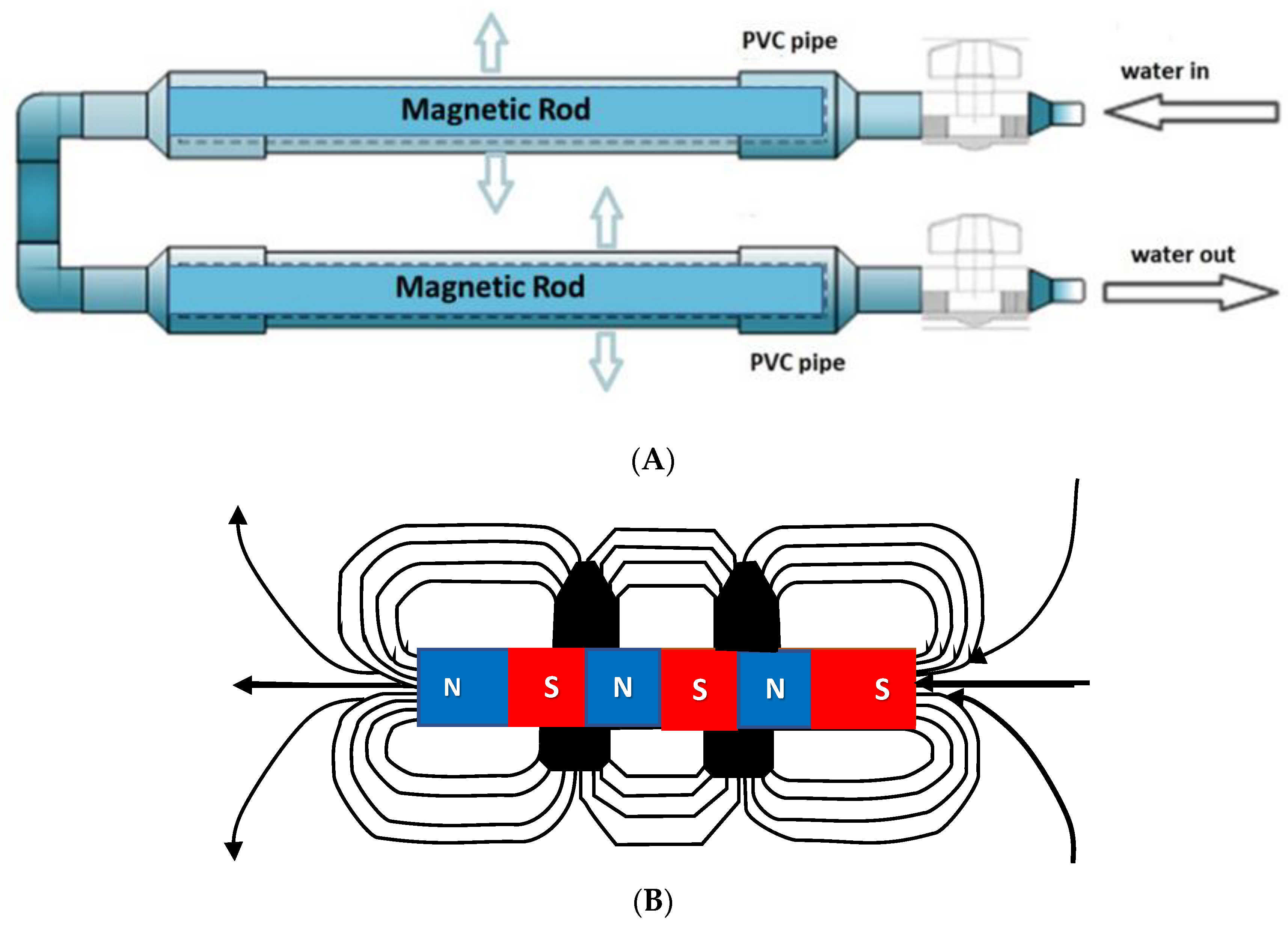
We sought to design a simple prototype device suitable for domestic and restaurant use. The magnetic equipment for the treatment of water used in the investigation was adapted from the apparatus described by Surendran et al. [29]. Permanent magnet rods having magnetic intensities of 800 mT were used for treating the water in a simple flow-thru pipe design (Figure 2A). The two permanent magnets were 300 mm in length and 25.4 mm in diameter. The magnets created a static uniform magnetic field exposed perpendicularly to the direction of water flow. The arrangement of their north and south poles and direction of the magnetic field generated are shown in Figure 2B. MTW retains its magnetized state for at least 24 h [29,30].
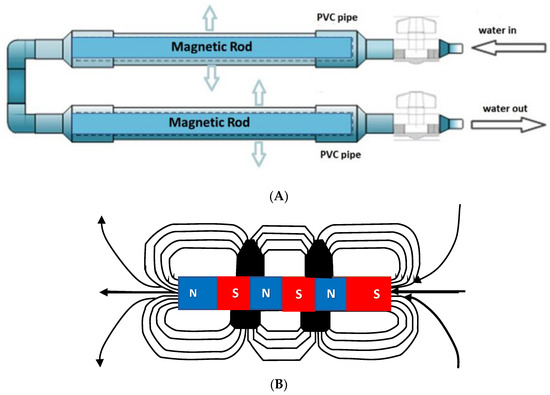
Figure 2.
(A) Developed magnetic device for creating MTW. (B) is a schematic line diagram of the magnetic device showing their north and south poles, the direction of magnetic field generated, and water flow through the rod magnets in the PVC pipe.
The permanent magnets were made of neodymium magnet (Nd2Fe14B), which is an advanced material for permanent magnet applications. Such rods are inexpensive and easily available commercially (MagnetDD, 11/8 Moo.5 Plai Bang, Bang Kruai District, Nonthaburi 11130, Thailand). Tap water (TW) was used to prepare MTW. The magnetic field strength was measured by telemeter (PHYWE, No. 13610-93, Germany) in µS cm−1 (100 µS cm−1 ≡ 1 mS m−1). The electrical conductivity (EC) of tap water (TW) was about 86 µS cm−1 and measured using a conductivity meter (Mettler Toledo MC, 126-2M, Switzerland). The pH was about 7.8 and measured using a pH meter (Eutech Instrument. pH 700, Singapore). Chloropyrifos® is a weak electrolyte and so it can be detected by a conductivity meter when it dissolves.
To ensure adequate exposure to the magnetic field, the experimental water was passed in a cycle continuously through the device for 24 h into a 50 L storage plastic tank. The magnetic treatment of the water was under dynamic conditions because there was a continuous flow of water by the way of pumping it with a 368 Watt (½ HP) centrifugal pump through the PVC (PolyVinylChloride) pipes at the rate of 2 L s−1 through the magnet array. After the treatment, the parameter conditions such as electrical conductivity (EC) were measured.
2.2. Sampling of Vegetables
The test vegetables, Brassica chinensis Linn., were planted and grown without application of pesticides. After a grow-out to saleable size (about 5 weeks) the trays of plants were allocated randomly to a control group and a Chlorpyrifos®-treated group. The controls were kept well away (5 m) from the sprayed group to avoid cross contamination. One group of Brassica chinensis was sprayed with commercial Chlorpyrifos® (40% w/v emulsifier concentration) (Thaion Agro Chemical CO., Ltd. Yannawa, Bangkok 10120, Thailand) with a concentration of 2% w/v [19] and sprayed as recommended by the manufacturer’s instructions as the experimental treatment. The emulsifier used in the commercial preparation is intellectual property (IP) and so its identity was not available to the investigators in this study. The pesticide-treated vegetables were left 1 day after spraying prior to being used experimentally. Each sample (1 kg) was collected in a clean transparent air tight polyethylene bag. To avoid cross contamination, each bag was properly labeled with a sample number. Samples sprayed with a range of concentrations of pesticides were subjected to washing experiments.
2.3. Washing Experiment
Brassica chinensis samples that were sprayed with Chlorpyrifos® were subjected to washing experiments. This was specifically to compare the effectiveness of household/restaurant treatments (tap water wash) [13] and MTW washing. To detect Chlorpyrifos® residue dissolved in washing solutions (it is a weak electrolyte), their electrical conductivities were recorded both before and after all washing. Washing by running water method was carried out for the sample (approximately 1 kg) under running tap water for 5 min. Then, the washing solution was discarded and the sample was again rinsed with running tap water. Washing with MTW, the Brassica chinensis samples were soaked in 5 L of MTW for 30 min. Then, the solution was discarded after its conductance was measured and the vegetables were rinsed again with a fresh lot of 5 L of MTW. All washed samples were kept on blotting paper to remove the excess water. Means ± SE (n = 3) were calculated and significant differences were identified by ANOVA and Student–Newman–Kuels (Tukey test) at p < 0.05.
2.4. Analytical Method
A standard protocol for analysis of organophosphates was followed using an Agilent 7890B Gas Chromatograph (GC) combined with a 7000 D triple quadrupole Mass Spectrometer (MS) (QQQ) operated in MRM mode (Agilent Technologies, Santa Clara, CA, USA). The analysis of pesticide residues was carried out using the pesticide multiresidue QuEChERS (Quick Easy Cheap Effective Rugged and Safe) method as described by the BS EN 15662 protocol [31,32] following previous work [13,14]. The QuEChERS protocol uses 2 mg L−1 stocks (2 ppm) from an official supplier of pesticide standards (traceable to the US Environmental Protection Agency Pesticide Repository, Fort Meade, MD, USA) [31,32]. The extraction Chlorpyrifos® procedure was performed by extracting 10 g of homogenized vegetable (Brassica oleracea L.), with a mixed solution of 4 g of anhydrous magnesium sulfate, 10 mL acetonitrile saturated, 1 g of anhydrous sodium acetate, and 1 g of sodium chloride. The extraction was followed by the process of the routine cleaning up procedure [13,14]. This was carried out by transferring the supernatant (1 mL) into another tube containing of 25 mg alumina N and 25 mg of primary–secondary amines (PSA). After shaking for 1 min and centrifugation for 5 min at 3000× g, the extract supernatant was then transferred to an autosampler vial for GC-MS analysis. The Scientific Laboratory and Equipment Center, Prince of Songkla University in Suratthani, 90110, Thailand followed their standard certified procedure for pesticide analysis by GC-MS. After calculation of Chloropyrifos® concentrations in the test material (ppm means ± SE, n = 3) significant differences were identified by ANOVA and Student–Newman–Kuels testing (Tukey) tests (p < 0.05).
3. Results
3.1. Analysis of Pesticide by Electric Conductivity
Prior to washing, the electric conductivities of all washing solutions were measured. The starting conductivity and pH of TW were about 90 μS cm−1 and pH 7.2, respectively. Exposing the TW to a magnetic field at a field intensity of 2000 G to become MTW increased the conductivity to about 470 μS cm−1 and reached a pH of about 8.4. In each set of washing protocols, the electric conductivity of the washing solution was recorded at every 5 min ranging from 10 min to 30 min. The obtained conductivities for all solutions over experimental washing times are illustrated in Table 1 A,B. Chlorphyrifos® is a weak electrolyte.

Table 1.
(A). Chloropyrifos® Removal by Washing: First Washing Conductivity (µS cm−1). (B). Chloropyrifos® Removal by Washing: Second Washing Conductivity (µS cm−1).
Table 1A shows the conductivity change over the investigating time of two washing solutions, TW (tap water) and MTW (magnetically treated water), at first washing. The control was tap water left standing and not used for washing. The tabulated values are a mean value ± standard errors (SE) from three replications. Table 1B shows the results of the second wash session, as a mean value ± SE from three replications.
The results of the first washing are shown in Table 1A. Conductivities of TW remained almost constant and behave closely to that of the control, whereas that of MTW increased with the washing time. The EC of MTW increased to reach a value 380 µS cm−1 within 25 min before remaining at a constant value until 30 min as found in previous experience of the conductivity of TW and MTW water [30]. The increase in the EC of the solutions was hypothesized to be because pesticide residues in the samples were dissolving and hence increasing the ionic conductivity of the solution. Therefore, after the first washing was accomplished, a second washing was performed on all the samples carried out as in the previous washing procedure. The results of the second washing are presented in Table 1B. The percent difference of the change in the EC in the washing solution is presented in Table 2. In the first washing (Table 1A), at the time of 20 min, the increase of EC was about 60% above the EC at zero time. In TW, EC continuously increased to about 20% at 25 min. In the second washing, no further change in EC in MTW was observed. This finding infers that no more residue dissolved in the washing solution. In TW, however, the EC and increase of less than 10% was still recorded. In the tap water washing treatment, the contaminant still remains in the samples and continues to only slowly dissolve into the washing solution.

Table 2.
First and Second Washing: Calculated % Change in Conductivity (Controls Table 1A,B).
Table 2 shows the percent difference of conductivity over two washing times in two washing solutions, TP (tap water) and MTW (magnetically treated water), calculated from the data in Table 1A,B. Each point is the mean ± SE value from three replications. Washing with tap water did not remove much Chlorpyrifos® and there was only a slight improvement upon the second washing. A single wash in MTW removed nearly all the mobilizable Chlorpyrifos®, but the second wash did not increase the total removal.
3.2. Analysis Pesticide by GC-MS
After samples were washed in washing solutions, the samples washed by TW and MTW were selected for analysis of pesticide residue by GC-MS. The GC–MS chromatogram of blank Brassica chinensis extract showed that there were three significant endogenous compounds eluted at retention times (RT) of 9.114, 12.117 and 14.090 min (Figure S1). These three endogenous peaks were observed in every sample of Brassica chinensis extract. This suggests that the observed two peaks in the extracts of Brassica chinensis are endogenous compounds that occurred naturally in Brassica chinensis. Chromatogram of the Chlorpyrifos® pesticide standard reference stock is illustrated in Figure S2. The authorized authentic stock was traceable to the US Environmental Protection Agency Pesticide Repository (Fort Meade, MD, USA) [31,32]. There was an abundant peak with RT of 24.776 min with an occupied area of 550,154,886.5. The GC-MS database identified the peak compound as Chlorpyrifos®. A representative chromatogram of a Brassica chinensis sample washed by TW after extraction is illustrated in Figure S3. This sample has similar endogenous peaks as had been found in Figure S2 with RT 24.888 min with a peak area of 57,022,224.82. In Figure S4, a chromatogram of an extracted sample from vegetables washed by MTW is presented. The peak at RT 24.894 min possessed an area peak of 1,332,817.43. Comparisons of the component RT, component area and percent removal factor are tabulated in Table 3. Calculated residues were compared to the Maximum Residue Load (MRL) criterion for Chlorpyrifos® (50 ppb—[10,33,34]). Significant differences between means in Table 3 were identified by ANOVA and Student–Newman–Kuels procedures (p < 0.05). Although simple washing removed statistically significant Chlopyrifos®, it was not sufficient to meet MRL guidelines. Sufficient Chlorophyrifos® to meet the MRL criteria was only achieved in the case of MTW-washed material.

Table 3.
Comparison of component retention time (RT), component area and percent match factor of extracted samples washed by TW and MTW comparing to control (no pesticide contamination). Values quoted are means ±standard errors, n = 3. The Maximum Residue Load (MRL) value for Chlorpyrifos® is 50 ppb [10,33,34]. Conclusions drawn in columns headed Final Concentration, Removal fraction and Pass/Fail MRL are based on ANOVA and Student–Newmann–Kuels test criterion (p < 0.05).
4. Discussion
4.1. Measurement of Pesticide Removal by Electrical Conductivity
Many insecticides designed to kill biting and chewing insects possess a low water solubility [9,10]. In the case of Chlorpyrifos®, the water solubility is about 1.40 mg L−1 [8,10]. This means it is hydrophobic, meaning that it tends to repel, or fail to mix with, water. Therefore, when this compound is immersed into water, only a few conducting ions are dissolved in water and consequently there is little increase in the ionic conductivity of water. Commercial preparations have “carriers” or detergents to increase its solubility. The change of electrical conductivity (EC) of all washing solutions are presented in Table 1A,B. No change of EC was recorded in control (no contaminating of pesticide), meaning that there were no extra ions dissolving in the water. In TW, the EC increase was only about 10% (Table 2) in washings from the pesticide-treated vegetables. This indicated few ions were released from the washed vegetables. This is not surprising given the very low solubility of Chlorpyrifos® [8]. However, when contaminated vegetables were washed with MTW for 30 min, there was a dramatic increase of EC of about 60% (Table 2), indicating that there was a release of conducting ions into the MTW washing solution. The results show that MTW enhances the water solubility of the pesticide. Magnetic fields are known to have positive effects on decreasing surface tension and increasing the solubility of polar compounds and improving polarizing effects [25]. Nevertheless, these characteristics have never been applied to mobilizing and removing pesticide residues from agricultural products. After the first washing procedure (Table 1A), all samples were rewashed with the same washing solutions. The change in EC of the wash medium was recorded (Table 1B). No further change in EC was observed for the MTW experiment, meaning no additional ions were dissolved or, in other words, no significant pesticide residue remained on the sample. In the tap water experiment, however, a further increase in electrical conductivity of about 10% was found to be consistent with some residue still remaining on the vegetables after the initial washing and dissolving very slowly with no indication of reaching completion even after a total wash-time of 60 min (Table 2). On the other hand, Table 1B and Table 2 show that washing with MTW removed most of the removable Chloropyrifos® in only one 30 min washing session with an approximate ½ time of only approximately 3 ± 0.26 min (n = 7, r = 0.9972, p < 0.001) based on a least squares fit to a simple exponential saturation model.
4.2. Analysis Pesticide by GC-MS
The residues on the vegetables were measured using GC-MS (Figures S1–S4). The chromatograms of the blank extraction of a Brassica chinensis sample is shown in Figure S1. The area peak of the certified standard Chlorpyrifos® of 550,154,886.5 found at RT of 24.776 min is presented in Figure S2. This peak is not apparent in Figure S1. The chromatograms of an extracted sample washed using TW is illustrated in Figure S3. This sample has endogenous peaks at RT 24.888 min with a peak area of 57,022,224.82. The peak area of the extracted sample washed by MTW was 1,332,817.43 at RT 24.894 min (Figure S4). Comparisons of area peaks of the chromatograms for all experiments including the removal capability of each washing solution are tabulated in Table 3. The results show that washing the vegetable using MTW can diminish Chlorpyrifos® by 412.77 times (−99.75%) the initial Chlorpyrifos® contamination level and effectively only a single washing was needed. Double washing the vegetable samples in tap water, however, reduces Chlorpyrifos® by only about 9.64 times (−89.6%).
Based on the safety regulation criteria known as Maximum Residue Load (MRL) [10,33,34], the maximum permissible contamination by Chlorpyrifos® is 50 ppm. Table 3 shows that the control plants easily passed this criterion, while the Chlorpyrifos® sprayed vegetables failed the criterion. The tap water-washed vegetables did not reliably pass the criterion, but the vegetables washed with magnetically treated water easily passed. Recently, Yang et al. [34] have looked at the issue of washing leafy vegetables, including lettuce, spinach and Brassica sp. They looked at many pesticides, but not an organophosphate. In general, they found that washing methods were not very effective and failed to reach satisfactory MRL load levels, but considered that in oriental use vegetables were typically cooked (destroying many pesticides), whereas in western cultures many are traditionally eaten raw. Wu et al. [35] have also recently confirmed previous work that most washing methods [9,13,15,16,17,18,19], including electrolysis, were very unsatisfactory for removing pesticides and found that Chlorophyrifos® was the most recalcitrant pesticide they tried.
4.3. Mechanism of Action
These results from both washing procedures are consistent with previous reports mentioning the enhancement of water solubility of salt solutions by MTW [25] and consequent increases in conductivity because of hydrogen bonding [36]. The well-documented positive effects on seed germination [30] could also be largely a surface-interaction effect involving calcium ions rather than calcium-signaling per se or biochemistry [24,32,37]. The release of larger ions from pesticide residues in MTW may be caused by the influence of magnetic fields causing dipole polarization of water molecules. Each water molecule is bound to the others with low energy intermolecular van der Waals forces, dipole–dipole interactions and hydrogen bonding [27,36,38,39] including in a biological system [40]. Magnetic fields may alter the deformation of hydrogen bonds and their partial connections, as well as the binding energy [41]. In addition, after exposure to magnetic fields, the migration of mobile protons H+ within the associative elements of water may distribute H2O molecules in temporary associative clusters with the general formula (H2O)n [41,42,43]. These effects may in combination alter the structure of water that leads to the observed changes in its density, surface tension, viscosity, pH value, and parameters of physical and chemical processes occurring in water under applied magnetic fields, including the dissolution and crystallization of dissolved inorganic salts and importantly micellization around compounds of limited solubility [25,39,41,42,43].
5. Conclusions
Even though the mechanism of magnetization of water and its interactions are not yet clearly described or understood, magnetized water has well-documented biological effects. The magnetization of water is a very complicated problem, although some theoretical models have been proposed. A better knowledge of its mechanism of action has value in extending the practical applications of magnetization of water. Our findings showed that MTW has high potential for the safe removal of Chlorpyrifos® as a representative non-water soluble organophosphate insecticide. It is a new method giving highly efficient removal compared to other previous methods to achieve a pass grade in Maximum Residue Load (MRL) regulations. Many other types of pesticides have been widely used in agriculture, particularly other polar compounds that are poorly water-soluble that need to be used with dispersants and detergents for application, but leave essentially water-insoluble residues on agricultural products that are very difficult to remove. This new method gives high removal efficiency and would be readily adaptable for food processing, restaurant and domestic use: it is a low-cost and simple to construct magnetic device.
Supplementary Materials
The following supporting information can be downloaded at: https://www.mdpi.com/article/10.3390/agriculture13030714/s1.
Author Contributions
Conceptualization, C.J.S. and R.J.R.; Methodology, C.J.S., N.J. and R.J.R.; Validation, C.J.S.; Formal analysis, C.J.S. and R.J.R.; Investigation, C.J.S. and N.J.; Resources, C.J.S.; Data curation, C.J.S.; Writing—original draft, C.J.S. and R.J.R.; Writing—review & editing, C.J.S. and R.J.R.; Supervision, C.J.S.; Project administration, C.J.S.; Funding acquisition, C.J.S. All authors have read and agreed to the published version of the manuscript.
Funding
This research was supported by National Science, Research and Innovation Fund (NSRF) and Prince of Songkla University (Grant No SIT6405010S).
Data Availability Statement
The full data sets are available upon request including the GC-MS data.
Acknowledgments
Ch. J. S. is grateful for a financial support from Prince of Songkla University, Suratthani Campus, Suratthani, Thailand. The authors thank the Office for access to their certified Chlorpyrifos® standard (Figure S2) traceable to the US Environmental Protection Agency Pesticide Repository (Fort Meade, MD, USA).
Conflicts of Interest
The authors declare no conflict of interest.
References
- Botwe, B.; Kelderman, P.; Drechsel, P.; Carboo, D.; Nartey, V.; Gijzen, H. Pesticide residues contamination of vegetables and their public health implications in Ghana. J. Environ. Issues Agric. Dev. Ctries. 2011, 3, 10–18. [Google Scholar]
- Rajabu, J.; Tarimo, M.; Hangali, T. Health effects, trends and knowledge on pesticide use in Tanzania. Int. J. Sci. Res. Innov. Technol. 2017, 4, 100–122. [Google Scholar]
- Foong, S.Y.; Ma, N.L.; Lam, S.S.; Peng, W.; Low, F.; Lee, B.H.K.; Alstrup, A.K.O.; Sonne, C. A recent global review of hazardous chlorpyrifos pesticide in fruit and vegetables: Prevalence, remediation and actions needed. J. Hazard. Mater. 2020, 400, 123006. [Google Scholar] [CrossRef] [PubMed]
- Mathur, A.; Joshi, A.; Harwani, D. Microbial Contamination of Raw Fruits and Vegetables. Internet J. Food Saf. 2014, 16, 26–28. [Google Scholar]
- Chen, S.; Liu, C.; Peng, C.; Liu, H.; Hu, M.; Zhong, G. Biodegradation of chlorpyrifos and its hydrolysis product 3,5,6-trichloro-2-pyridinol by a new fungal strain Cladosporium cladosporioides Hu-01. PLoS ONE 2012, 7, e47205. [Google Scholar] [CrossRef]
- Ntow, W.J.; Drechsel, P.; Botwe, B.O.; Kelderman, P.; Gijzen, H.J. The impact of agricultural runoff on the quality of two streams in vegetable farm areas in Ghana. J. Environ. Qual. 2008, 37, 696–703. [Google Scholar] [CrossRef]
- Toft, G.; Hagmar, L.; Giwercman, A.; Peter, J. Epidemiological evidence on reproductive effects of persistent organochlorines in humans. Reprod. Toxicol. 2004, 19, 5–26. [Google Scholar] [CrossRef]
- Krol, W.J.; Arsenault, T.L.; Pylypiw, H.M.; Incorvia Mattina, M.J. Reduction of pesticide residues on produce by rinsing. J. Agric. Food Chem. 2000, 48, 4666–4670. [Google Scholar] [CrossRef]
- Mostafalou, S.; Abdollahi, M. Pesticides and human chronic diseases: Evidences, mechanisms, and perspectives. Toxicol. Appl. Pharmacol. 2013, 268, 157–177. [Google Scholar] [CrossRef]
- Tomlin, C.D.S. The E-Pesticide Manual, Version 3.0; BCPC: London, UK, 2009. [Google Scholar]
- Sabarwal, A.; Kumar, K.; Singh, R.P. Hazardous effects of chemical pesticides on human health–Cancer and other associated disorders. Environ. Toxicol. Pharmacol. 2018, 63, 103–114. [Google Scholar]
- Velmurugan, G.; Ramprasath, T.; Swaminathan, K.; Mithieux, G.; Rajendhran, J.; Dhivakar, M.; Parthasarathy, A.; Babu, D.D.V.; Thumburaj, L.J.; Freddy, A.J. Gut microbial degradation of organophosphate insecticides-induces glucose intolerance via gluconeogenesis. Genome Biol. 2017, 18, 8. [Google Scholar] [CrossRef] [PubMed]
- Wanwimolruk, S.; Kanchanamayoon, O.; Phopin, K.; Prachayasittikul, V. Food safety in Thailand 2: Pesticide residues found in Chinese kale (Brassica oleracea), a commonly consumed vegetable in Asian countries. Sci. Total Environ. 2015, 532, 447–455. [Google Scholar] [CrossRef] [PubMed]
- Wanwimolruk, S.; Phopin, K.; Boonpangrak, S.; Prachayasittikul, V. Food safety in Thailand 4: Comparison of pesticide residues found in three commonly consumed vegetables purchased from local markets and supermarkets in Thailand. PeerJ 2016, 4, e2432. [Google Scholar] [CrossRef]
- Djordjevic, T.; Djurovic-Pejcev, R. Food processing as a means for pesticide residue dissipation. Pestic. Fitomed. 2016, 31, 89–105. [Google Scholar] [CrossRef]
- Fenske, R.A.; Lu, C. Determination of handwash removal efficiency: Incomplete removal of the pesticide chlorpyrifos from skin by standard handwash techniques. Am. Ind. Hyg. Assoc. J. 1994, 55, 425–432. [Google Scholar] [CrossRef]
- Rao, C.S.; Bhushan, V.S.; Reddy, A.H.; Darsi, R.M.A.; Ramesh, B. Risk Mitigation Methods for Removal of Pesticide Residues in Brinjal for Food Safety. Univers. J. Agric. Res. 2014, 2, 279–283. [Google Scholar] [CrossRef]
- Hossain, M.S.; Fakhruddin, A.N.M.; Chowdhury, M.A.Z.; Alam, M.K. Degradation of chlorpyrifos, an organophosphorus insecticide in aqueous solution with gamma irradiation and natural sunlight. J. Environ. Chem. Eng. 2013, 1, 270–274. [Google Scholar] [CrossRef]
- Khaled, A.O.; Fahad, B.; Abdullah, A. Ozone as a Safety Post-Harvest Treatment for Chlorpyrifos Removal from Vegetables and its Effects on Vegetable Quality. Int. J. Food Nutr. Sci. 2017, 4, 38–48. [Google Scholar]
- Ishag, A.E.S.A.; Abdelbagi, A.O.; Hammad, A.M.A.; Elsheikh, E.A.E.; Elsaid, O.E.; Hur, J.-H.; Laing, M.D. Biodegradation of Chlorpyrifos, Malathion, and Dimethoate by Three Strains of Bacteria Isolated from Pesticide-Polluted Soils in Sudan. J. Agric. Food Chem. 2016, 64, 8491–8498. [Google Scholar] [CrossRef]
- Amiri, M.C.; Dadkhah, A.A. On reduction in the surface tension of water due to magnetic treatment. Colloid Surf. A 2006, 278, 252–255. [Google Scholar] [CrossRef]
- Toledo, E.J.L.; Ramalho, T.C.; Magriotis, Z.M. Influence of magnetic field on physical–chemical properties of the liquid water: Insights from experimental and theoretical models. J. Mol. Struct. 2008, 888, 409–415. [Google Scholar] [CrossRef]
- Saksono, N.; Gozan, M.; Bismo, S.; Krisanti, E.; Widaningrum, R.; Song, S.K. Effects of magnetic field on calcium carbonate precipitation: Ionic and particle mechanisms. Korean J. Chem. Eng. 2008, 25, 1145–1150. [Google Scholar] [CrossRef]
- El-Sabrout, K.; Hanafy, M. Effect of magnetized water on productive traits of laying chickens. Prof. Anim. Sci. 2017, 33, 739–742. [Google Scholar] [CrossRef]
- Holysz, L.; Szczes, A.; Chibowski, E. Effects of a static magnetic field on water and electrolyte solutions. J. Colloid Interface Sci. 2007, 316, 996–1002. [Google Scholar] [CrossRef]
- Waleed, F.A. Overview of Role of Magnetizing Treated Water in Agricultural Sector Development. Adv. Agric. Technol. Plant Sci. 2019, 2, 180023. [Google Scholar]
- Wang, Y.; Wei, H.; Li, Z. Effect of magnetic field on the physical properties of water. Results Phys. 2018, 8, 262–267. [Google Scholar] [CrossRef]
- Lin, L.; Jiang, W.; Xu, X.; Xu, P. A critical review of the application of electromagnetic fields for scaling control in water systems: Mechanisms, characterization, and operation. Clean Water 2020, 3, 25. [Google Scholar] [CrossRef]
- Surendran, U.; Sandeep, O.; Joseph, E.J. The impacts of magnetic treatment of irrigation water on plant, water and soil characteristics. Agric. Water Manag. 2016, 178, 21–29. [Google Scholar]
- Sudsiri, C.J.; Nattawat, J.; Kongchana, P.; Ritchie, R.J. Effect of magnetically treated water on germination and seedling growth of oil palm (Elaeis guineensis). Seed Sci. Technol. 2016, 44, 267–280. [Google Scholar] [CrossRef]
- Arienzo, M.; Cataldo, D.; Ferrara, L. Pesticide residues in fresh-cut vegetables from integrated pest management by ultra performance liquid chromatography coupled to tandem mass spectrometry. BS EN 15662. Foods of Plant Origin—Determination of Pesticide Residues Using GC-MS and/or LC-MS/MS Following Acetonitrile Extraction/Partitioning and Clean-up by Dispersive SPE—QuEChERS-Method. 2008. Food Control 2013, 31.1, 108–115. [Google Scholar]
- Lehotay, S.J.; Ae Son, K.; Kwon, H.; Koesukwiwat, U.; Fu, W.; Mastovska, K.; Hoh, E.; Leepipatpiboon, N. Comparison of QuEChERS sample preparation methods for the analysis of pesticide residues in fruits and vegetables. J. Chromatogr. A 2010, 1217, 2548–2560. [Google Scholar] [CrossRef]
- FAO. Pesticide Residues in Food and Feed; Codex Alimentarius Commission, FAO: Rome, Italy, 2021; Available online: http://www.codexalimentarius.net/pestres/data/pesticides/index.html (accessed on 19 March 2022).
- Yang, S.-J.; Mun, S.; Kim, H.J.; Han, S.J.; Kim, D.W.; Cho, B.-S.; Kim, A.G.; Park, D.W. Effectiveness of Different Washing Strategies on Pesticide Residue Removal: The First Comparative Study on Leafy Vegetables. Foods 2022, 11, 2916. [Google Scholar] [CrossRef] [PubMed]
- Wu, Y.; An, Q.; Li, D.; Wu, J.; Pan, C. Comparison of different home/commercial washing strategies for ten typical pesticide residue removal effects in kumquat, spinach and cucumber. Int. J. Environ. Res. Public Heal. 2019, 16, 472. [Google Scholar] [CrossRef] [PubMed]
- Cai, R.; Yang, H.; He, J.; Zhu, W. The effects of magnetic fields on water molecular hydrogen bonds. J. Mol. Struct. 2009, 938, 15–19. [Google Scholar] [CrossRef]
- Yacout, M.; Hassan, A.; Khalel, M.S.; Shwerab, A.; Abdel-Gawad, E.I.; Abdel-kader, Y.I. Effect of Magnetic Water on the Performance of Lactating Goats. J. Dairy Vet. Anim. Res. 2015, 2, 159–170. [Google Scholar]
- Pang, X.F.; Deng, B. Investigation of changes in properties of water under the action of a magnetic field. Sci. China Ser. G Phys. Mech. Astro. 2008, 51, 1621–1632. [Google Scholar] [CrossRef]
- Wang, Y.; Zhang, B.; Gong, Z.; Gao, K.; Ou, Y.; Zhang, J. The effect of a static magnetic field on the hydrogen bonding in water using frictional experiments. J. Mol. Struct. 2013, 1052, 102–104. [Google Scholar] [CrossRef]
- Moon, J.-D.; Chung, H.-S. Acceleration of germination of tomato seed by applying AC electric and magnetic fields. J. Electrost. 2000, 48, 103–114. [Google Scholar] [CrossRef]
- Ignatov, I.; Mosin, O. Mathematical Theory and Modeling Structural Mathematical Models Describing Water Clusters. Math. Theory Model. 2013, 3, 72–88. [Google Scholar]
- Ahmed, H.I. Behavior of magnetic concrete incorporated with Egyptian nano alumina. Constr. Build. Mater. 2017, 150, 404–408. [Google Scholar] [CrossRef]
- Esmaeilnezhad, E.; Choi, H.J.; Schaffie, M.; Gholizadeh, M.; Ranjbar, M. Characteristics and applications of magnetized water as a green technology. J. Clean. Prod. 2017, 161, 908–921. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).