Automated Visual Identification of Foliage Chlorosis in Lettuce Grown in Aquaponic Systems
Abstract
1. Introduction
2. Related Work
3. Research Methodology
3.1. Data Preparation
3.2. Image Segmentation
3.2.1. HSV Color Space
3.2.2. Image Hue Thresholding
3.3. Foliage Color Detection Model Development
3.4. Ontology Model
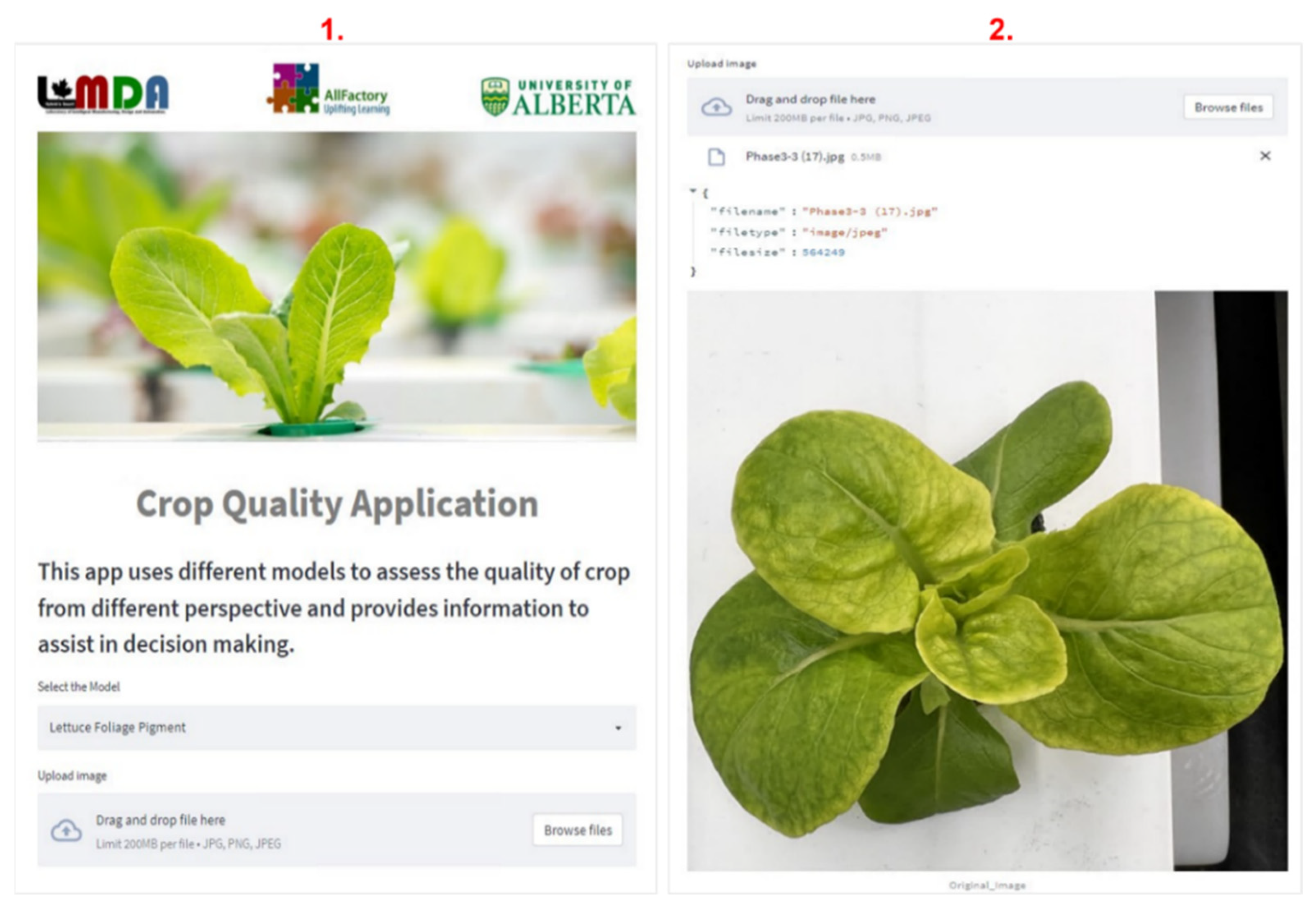
3.5. Cloud-Based Application
4. Results and Discussion
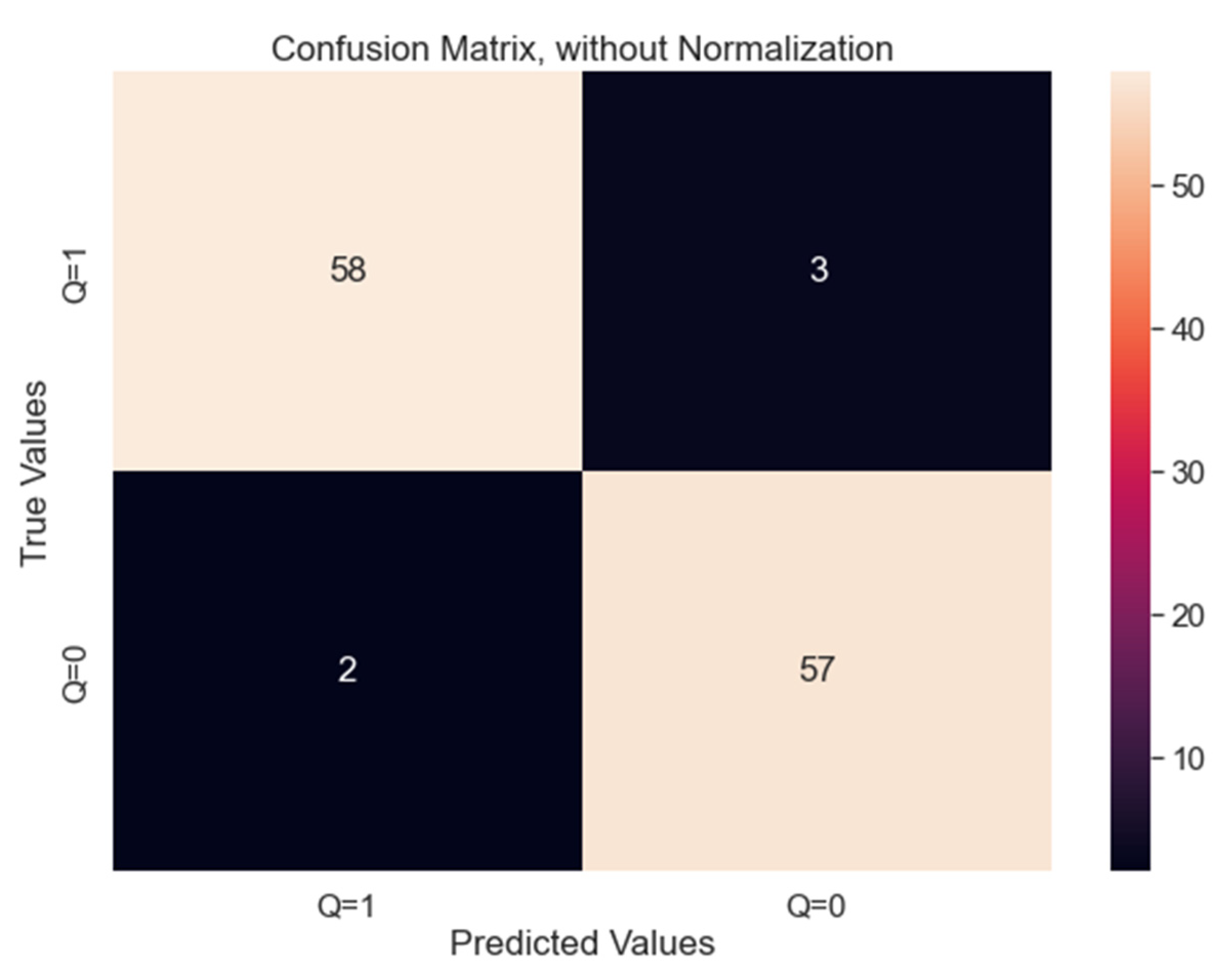
- True Positive (TP) = 58. Thus, 58 plants were healthy, and the model correctly classified them healthy as well.
- True Negative (TN) = 57. Thus, 57 plants were unhealthy, and the model correctly classified them unhealthy as well.
- False Positive (FP) = 3. Thus, 3 plants were unhealthy, but the model incorrectly classified them as healthy.
- False Negative (FN) = 2. Thus, 2 plants were healthy, but the model incorrectly classified them as unhealthy.
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Appendix A



References
- Abbasi, R.; Martinez, P.; Ahmad, R. An ontology model to support the automated design of aquaponic grow beds. Procedia CIRP 2021, 100, 55–60. [Google Scholar] [CrossRef]
- Reyes-Yanes, A.; Martinez, P.; Ahmad, R. Real-time growth rate and fresh weight estimation for little gem romaine lettuce in aquaponic grow beds. Comput. Electron. Agric. 2020, 179, 105827. [Google Scholar] [CrossRef]
- Lin, K.H.; Huang, M.Y.; Huang, W.D.; Hsu, M.H.; Yang, Z.W.; Yang, C.M. The effects of red, blue, and white light-emitting diodes on the growth, development, and edible quality of hydroponically grown lettuce (Lactuca sativa L. var. capitata). Sci. Hortic. 2013, 150, 86–91. [Google Scholar] [CrossRef]
- Haider, T.; Farid, M.S.; Mahmood, R.; Ilyas, A.; Khan, M.H.; Haider, S.T.A.; Chaudhry, M.H.; Gul, M. A Computer-Vision-Based Approach for Nitrogen Content Estimation in Plant Leaves. Agriculture 2021, 11, 766. [Google Scholar] [CrossRef]
- Taha, M.F.; Abdalla, A.; Elmasry, G.; Gouda, M.; Zhou, L.; Zhao, N.; Liang, N.; Niu, Z.; Hassanein, A.; Al-Rejaie, S.; et al. Using Deep Convolutional Neural Network for Image-Based Diagnosis of Nutrient Deficiencies in Plants Grown in Aquaponics. Chemosensors 2022, 10, 45. [Google Scholar] [CrossRef]
- Matysiak, B.; Ropelewska, E.; Wrzodak, A.; Kowalski, A.; Kaniszewski, S. Yield and Quality of Romaine Lettuce at Different Daily Light Integral in an Indoor Controlled Environment. Agronomy 2022, 12, 1026. [Google Scholar] [CrossRef]
- Abbasi, R.; Martinez, P.; Ahmad, R. The digitization of agricultural industry—A systematic literature review on agriculture 4.0. Smart Agric. Technol. 2022, 2, 100042. [Google Scholar] [CrossRef]
- Kowalczyk, K.; Sieczko, L.; Goltsev, V.; Kalaji, H.M.; Gajc-Wolska, J.; Gajewski, M.; Gontar, Ł.; Orliński, P.; Niedzińska, M.; Cetner, M.D. Relationship between chlorophyll fluorescence parameters and quality of the fresh and stored lettuce (Lactuca sativa L.). Sci. Hortic. 2018, 235, 70–77. [Google Scholar] [CrossRef]
- Song, J.; Huang, H.; Hao, Y.; Song, S.; Zhang, Y.; Su, W.; Liu, H. Nutritional quality, mineral and antioxidant content in lettuce affected by interaction of light intensity and nutrient solution concentration. Sci. Rep. 2020, 10, 2796. [Google Scholar] [CrossRef]
- Cook, S.E.; Bramley, R.G.V. Coping with variability in agricultural production -implications for soil testing and fertiliser management. Commun. Soil Sci. Plant Anal. 2000, 31, 1531–1551. [Google Scholar] [CrossRef]
- Kjeldahl, J. Neue Methode zur Bestimmung des Stickstoffs in organischen Körpern. Z. Anal. Chem. 1883, 22, 366–382. [Google Scholar] [CrossRef]
- Yang, W.H.; Peng, S.; Huang, J.; Sanico, A.L.; Buresh, R.J.; Witt, C. Using Leaf Color Charts to Estimate Leaf Nitrogen Status of Rice. Agron. J. 2003, 95, 212–217. [Google Scholar] [CrossRef]
- Markwell, J.; Osterman, J.C.; Mitchell, J.L. Calibration of the Minolta SPAD-502 leaf chlorophyll meter. Photosynth. Res. 1995, 46, 467–472. [Google Scholar] [CrossRef]
- Zheng, H.; Cheng, T.; Li, D.; Zhou, X.; Yao, X.; Tian, Y.; Cao, W.; Zhu, Y. Evaluation of RGB, Color-Infrared and Multispectral Images Acquired from Unmanned Aerial Systems for the Estimation of Nitrogen Accumulation in Rice. Remote Sens. 2018, 10, 824. [Google Scholar] [CrossRef]
- Tao, M.; Ma, X.; Huang, X.; Liu, C.; Deng, R.; Liang, K.; Qi, L. Smartphone-based detection of leaf color levels in rice plants. Comput. Electron. Agric. 2020, 173, 105431. [Google Scholar] [CrossRef]
- Burdescu, D.D.; Brezovan, M.; Ganea, E.; Stanescu, L. A new method for segmentation of images represented in a HSV color space. In Proceedings of the Advanced Concepts for Intelligent Vision Systems: 11th International Conference, ACIVS 2009, Bordeaux, France, 28 September–2 October 2009; Springer: Berlin/Heidelberg, Germany, 2009; pp. 606–617. [Google Scholar] [CrossRef]
- Abbasi, R.; Martinez, P.; Ahmad, R. An ontology model to represent aquaponics 4.0 system’s knowledge. Inf. Process. Agric. 2022, 9, 514–532. [Google Scholar] [CrossRef]
- Yang, R.; Wu, Z.; Fang, W.; Zhang, H.; Wang, W.; Fu, L.; Majeed, Y.; Li, R.; Cui, Y. Detection of abnormal hydroponic lettuce leaves based on image processing and machine learning. Inf. Process. Agric. 2021, 10, 1–10. [Google Scholar] [CrossRef]
- Maity, S.; Sarkar, S.; Vinaba Tapadar, A.; Dutta, A.; Biswas, S.; Nayek, S.; Saha, P. Fault Area Detection in Leaf Diseases Using K-Means Clustering. In Proceedings of the 2018 2nd International Conference on Trends in Electronics and Informatics (ICOEI), Tirunelveli, India, 11–12 May 2018; pp. 1538–1542. [Google Scholar] [CrossRef]
- Yang, W.; Wang, S.; Zhao, X.; Zhang, J.; Feng, J. Greenness identification based on HSV decision tree. Inf. Process. Agric. 2015, 2, 149–160. [Google Scholar] [CrossRef]
- Luna-Benoso, B.; Martínez-Perales, J.C.; Cortés-Galicia, J.; Flores-Carapia, R.; Silva-García, V.M. Detection of Diseases in Tomato Leaves by Color Analysis. Electronics 2021, 10, 1055. [Google Scholar] [CrossRef]
- Streamlit • The Fastest Way to Build and Share Data Apps [WWW Document], n.d. Available online: https://streamlit.io/ (accessed on 7 June 2022).
- Buslaev, A.; Iglovikov, V.I.; Khvedchenya, E.; Parinov, A.; Druzhinin, M.; Kalinin, A.A. Albumentations: Fast and flexible image augmentations. Information 2020, 11, 125. [Google Scholar] [CrossRef]
- Loresco, P.J.M.; Valenzuela, I.C.; Dadios, E.P. Color Space Analysis Using KNN for Lettuce Crop Stages Identification in Smart Farm Setup. In Proceedings of the TENCON 2018-2018 IEEE Region 10 Conference, Jeju, Republic of Korea, 28–31 October 2018; pp. 2040–2044. [Google Scholar] [CrossRef]
- Hasan, S.; Jahan, S.; Islam, M.I. Disease detection of apple leaf with combination of color segmentation and modified DWT. J. King Saud Univ.-Comput. Inf. Sci. 2022, 34, 7212–7224. [Google Scholar] [CrossRef]
- Abbasi, R.; Martinez, P.; Ahmad, R. Data acquisition and monitoring dashboard for IoT enabled aquaponics facility. In Proceedings of the 2022 10th International Conference on Control, Mechatronics and Automation (ICCMA), Luxembourg, 9–12 November 2022; IEEE: Piscataway, NJ, USA, 2022. [Google Scholar] [CrossRef]








| Class | N (Truth) | N (Classified) | Accuracy | Precision | Recall | F1-Score |
|---|---|---|---|---|---|---|
| Q = 1 | 60 | 61 | 0.95 | 0.95 | 0.97 | 0.96 |
| Q = 0 | 60 | 59 | 0.95 | 0.97 | 0.95 | 0.96 |
| Average | - | - | 0.95 | 0.96 | 0.96 | 0.96 |
| Methods | Techniques and Parameters Used | Average Accuracy | Average Precision | Average Recall | Average F1-Score |
|---|---|---|---|---|---|
| Yang et al. [18] | SVM (support vector machine) and a* (CIELAB color space), G (green from RGB color space), and H (hue from HSV color space) | 0.91 | 0.92 | 0.93 | 0.925 |
| Maity et al. [19] | Otsu’s method and k-means clustering technique | 0.92 | 0.93 | 0.93 | 0.93 |
| Yang et al. [20] | HSV (hue, saturation, and value) color space and decision tree method | 0.89 | 0.91 | 0.90 | 0.905 |
| Luna-Benoso et al. [21] | Otsu’s method, SVM, k-NN (k-nearest neighbor) and MLP (multi-layer perceptron) | 0.90 | 0.91 | 0.91 | 0.91 |
| Hasan et al. [25] | L*a*b* color histogram, k-NN, and random forest | 0.94 | 0.95 | 0.94 | 0.945 |
| - | Proposed model | 0.95 | 0.96 | 0.96 | 0.96 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Abbasi, R.; Martinez, P.; Ahmad, R. Automated Visual Identification of Foliage Chlorosis in Lettuce Grown in Aquaponic Systems. Agriculture 2023, 13, 615. https://doi.org/10.3390/agriculture13030615
Abbasi R, Martinez P, Ahmad R. Automated Visual Identification of Foliage Chlorosis in Lettuce Grown in Aquaponic Systems. Agriculture. 2023; 13(3):615. https://doi.org/10.3390/agriculture13030615
Chicago/Turabian StyleAbbasi, Rabiya, Pablo Martinez, and Rafiq Ahmad. 2023. "Automated Visual Identification of Foliage Chlorosis in Lettuce Grown in Aquaponic Systems" Agriculture 13, no. 3: 615. https://doi.org/10.3390/agriculture13030615
APA StyleAbbasi, R., Martinez, P., & Ahmad, R. (2023). Automated Visual Identification of Foliage Chlorosis in Lettuce Grown in Aquaponic Systems. Agriculture, 13(3), 615. https://doi.org/10.3390/agriculture13030615







