Effects of Weather on Sugarcane Aphid Infestation and Movement in Oklahoma
Abstract
1. Introduction
2. Methods and Data
2.1. Structural Model of SCA Movement
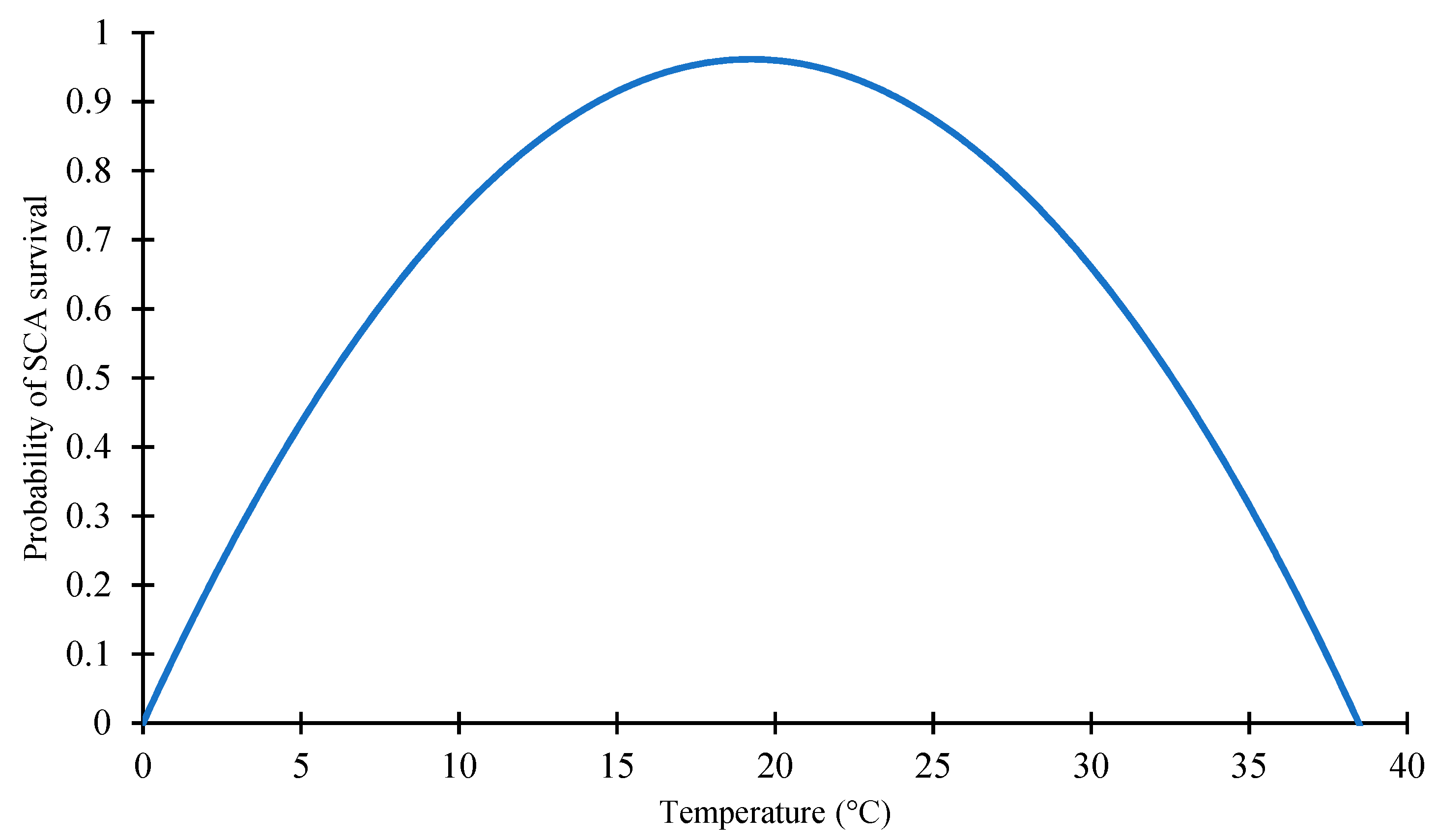
2.1.1. Effect of Temperature on SCA Survivability
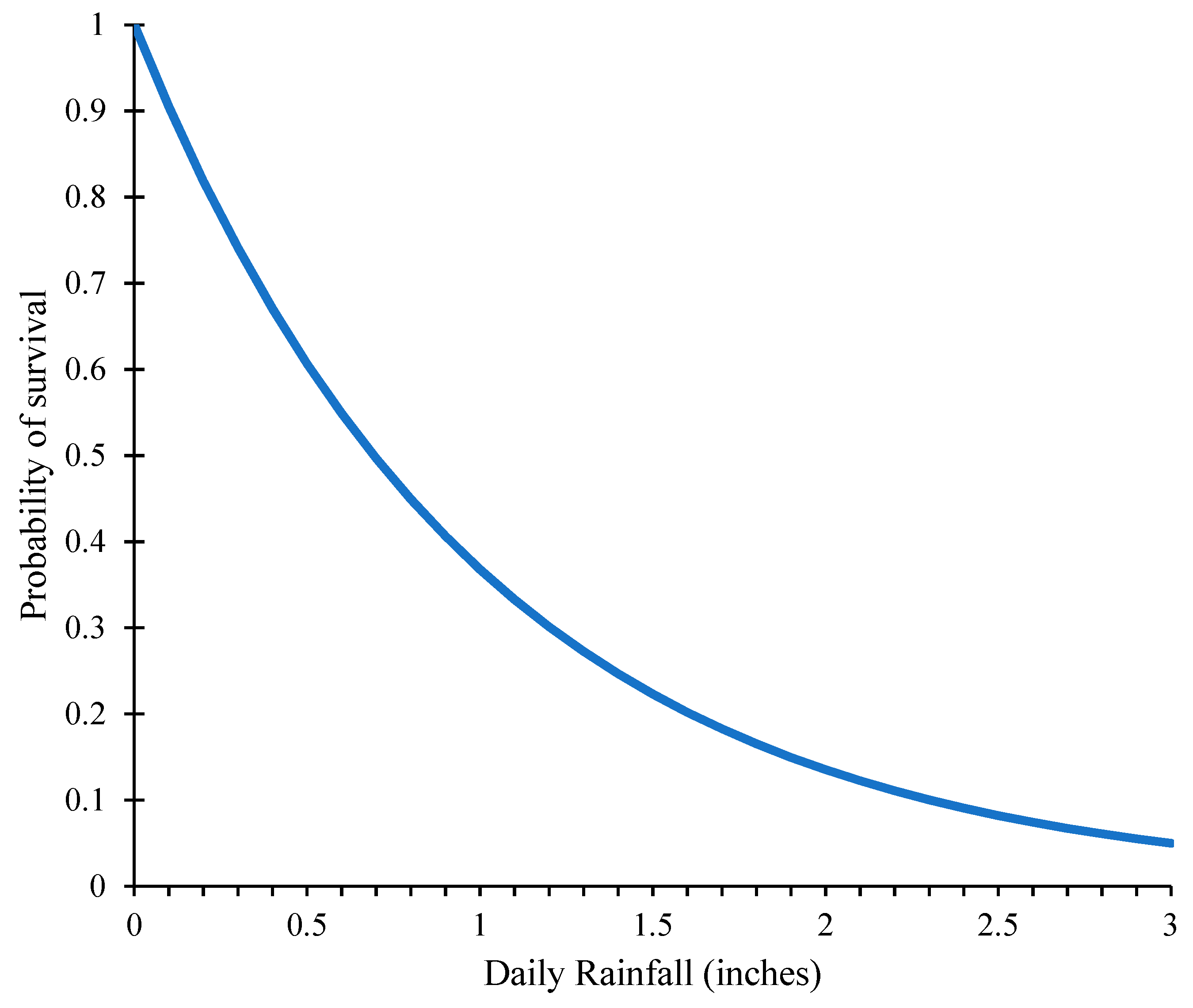
2.1.2. Effect of Rainfall on SCA Survivability
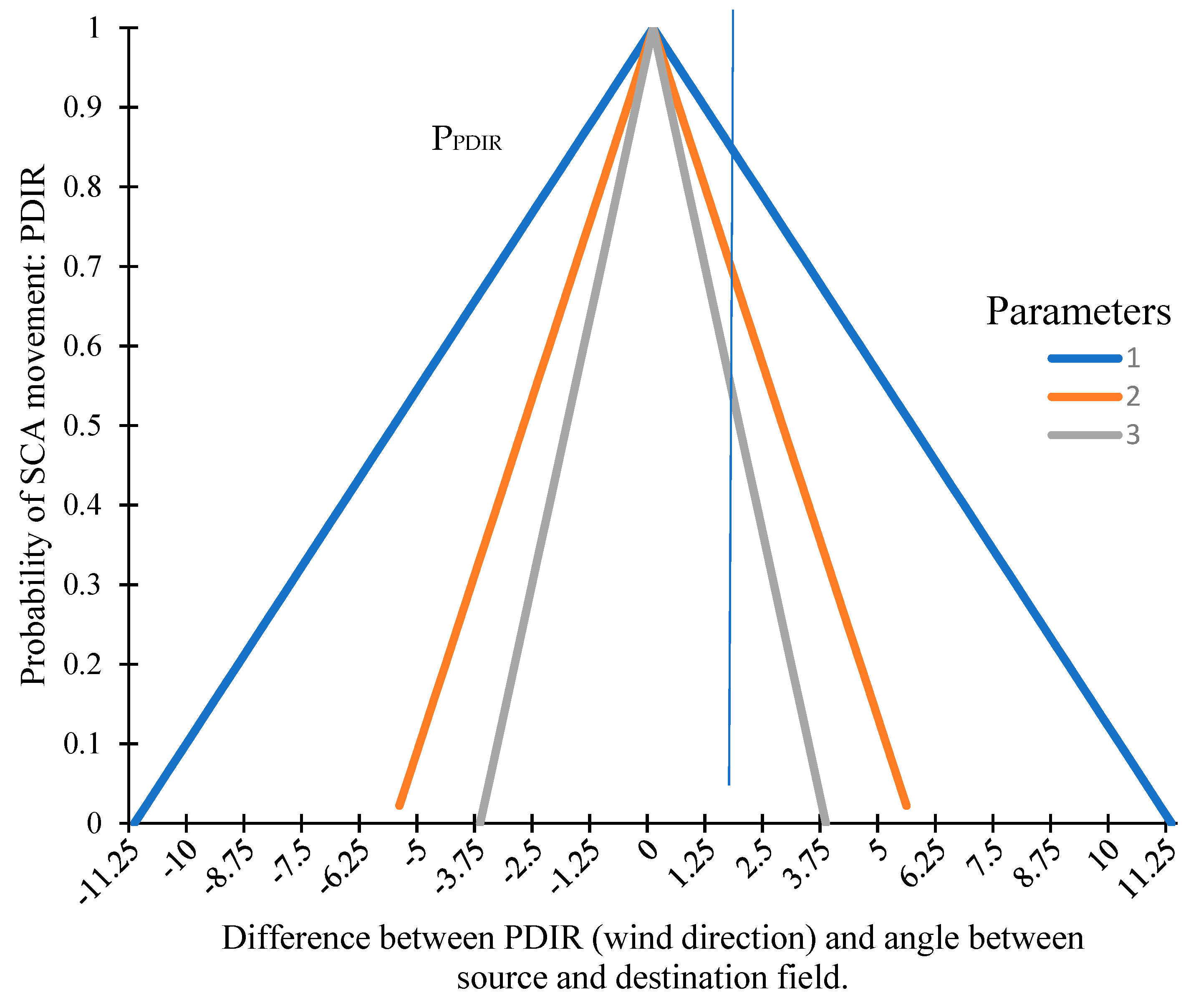
2.1.3. Effect of Wind on SCA Movement
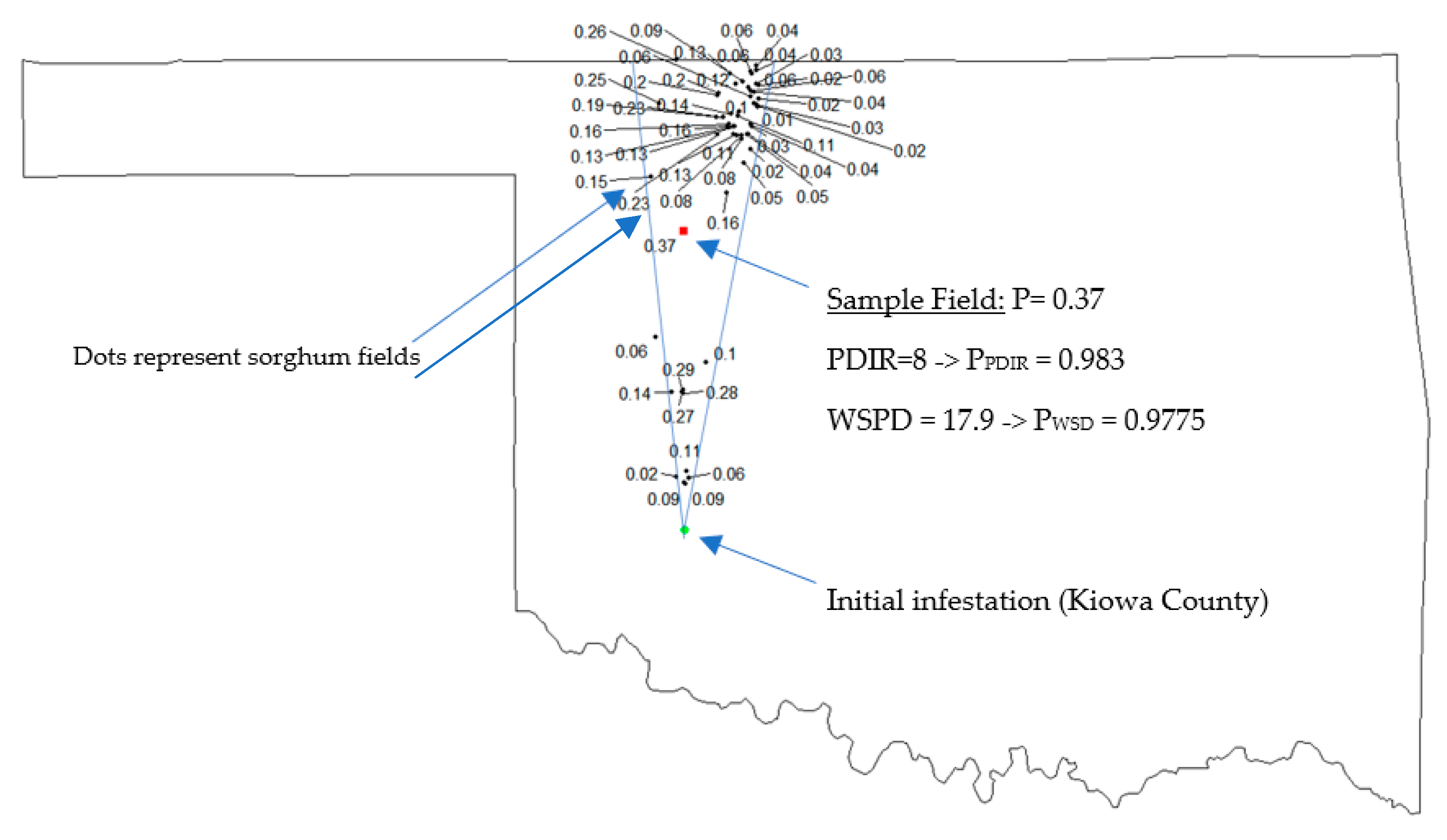
2.1.4. Joint Probability of SCA Movement
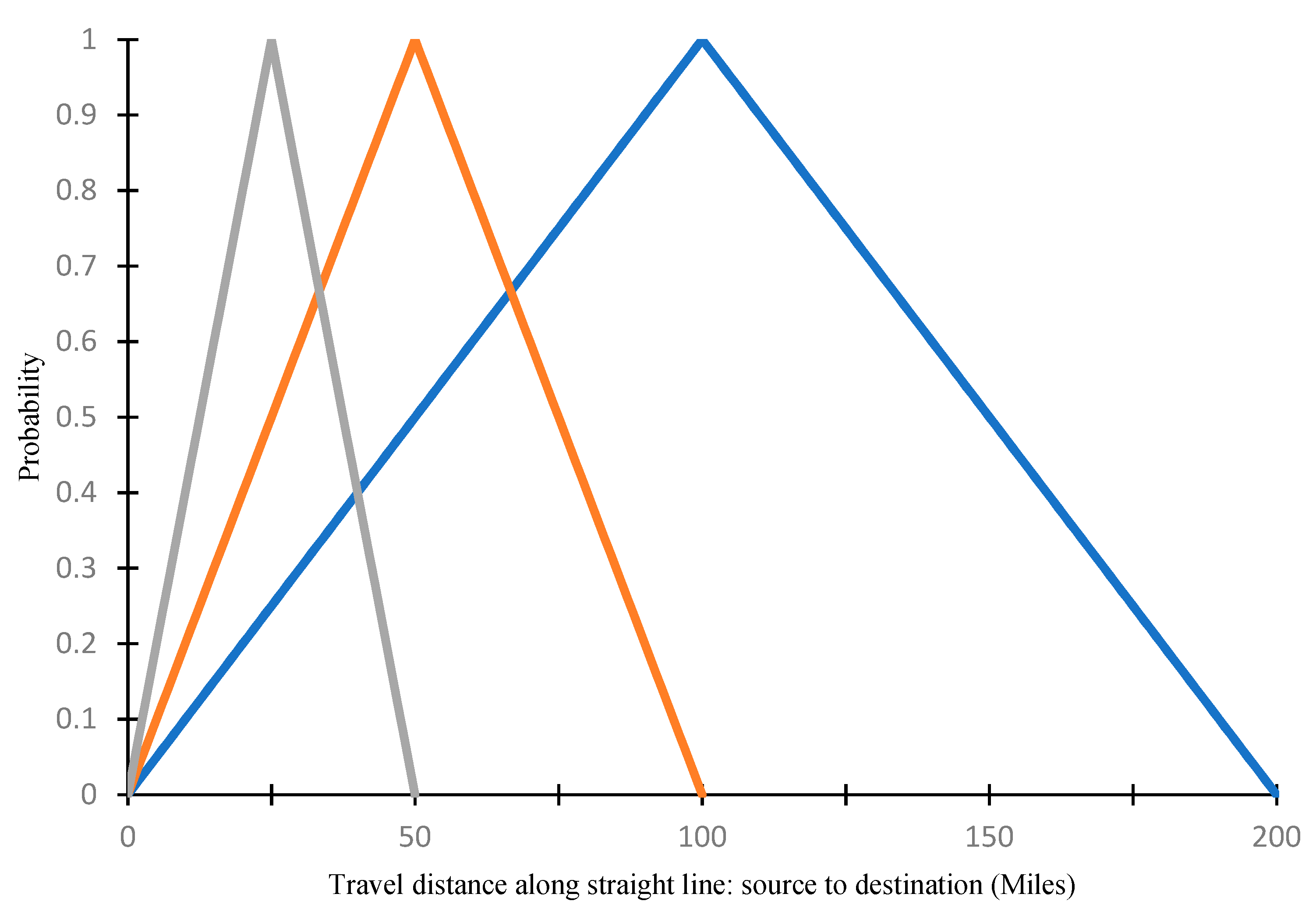
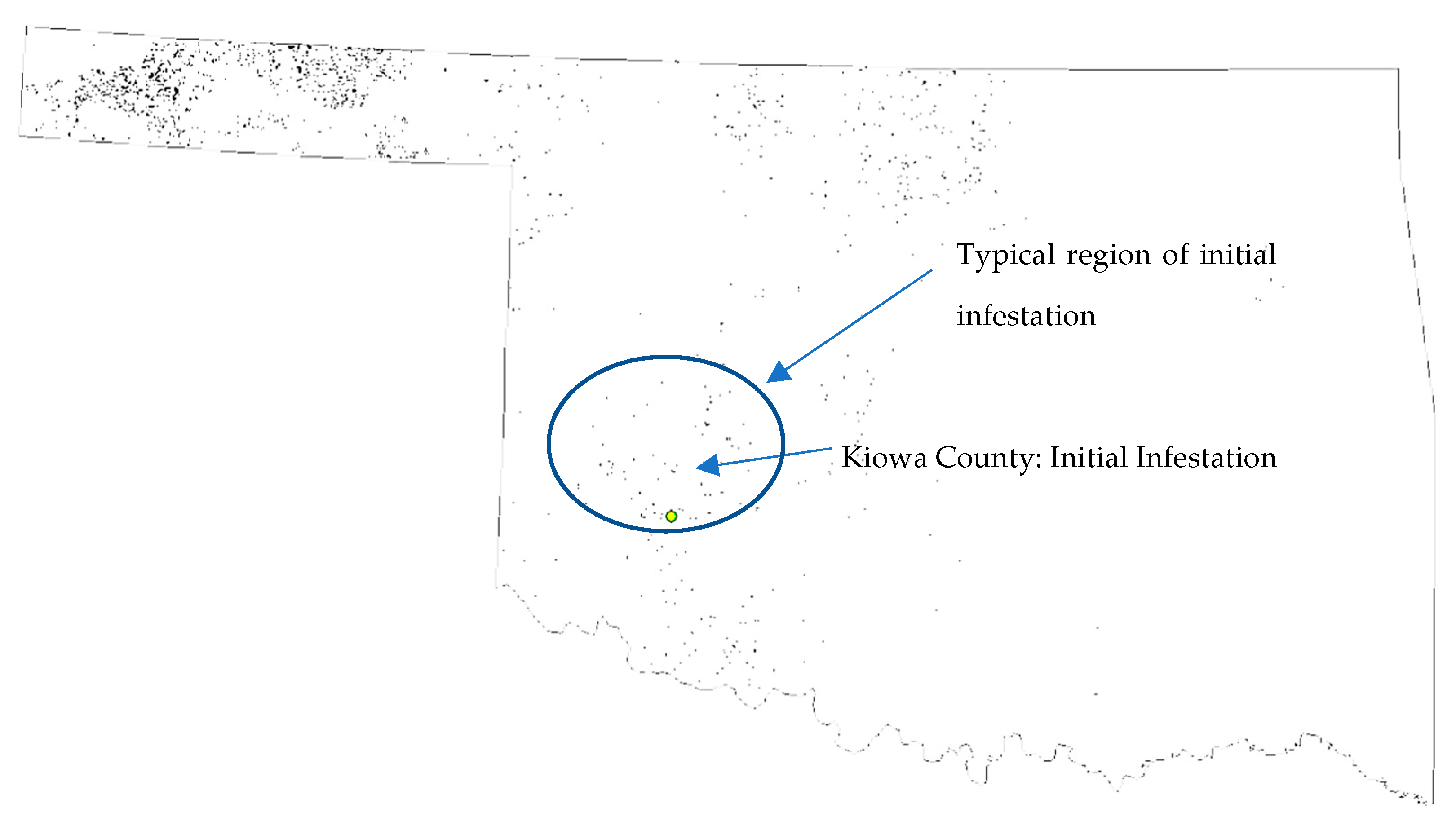
2.2. Explaining Spatial Patterns of SCA Movements over Time
2.3. Data
3. Results
3.1. Effect of Weather Variables on Predicted Cumulative Probability
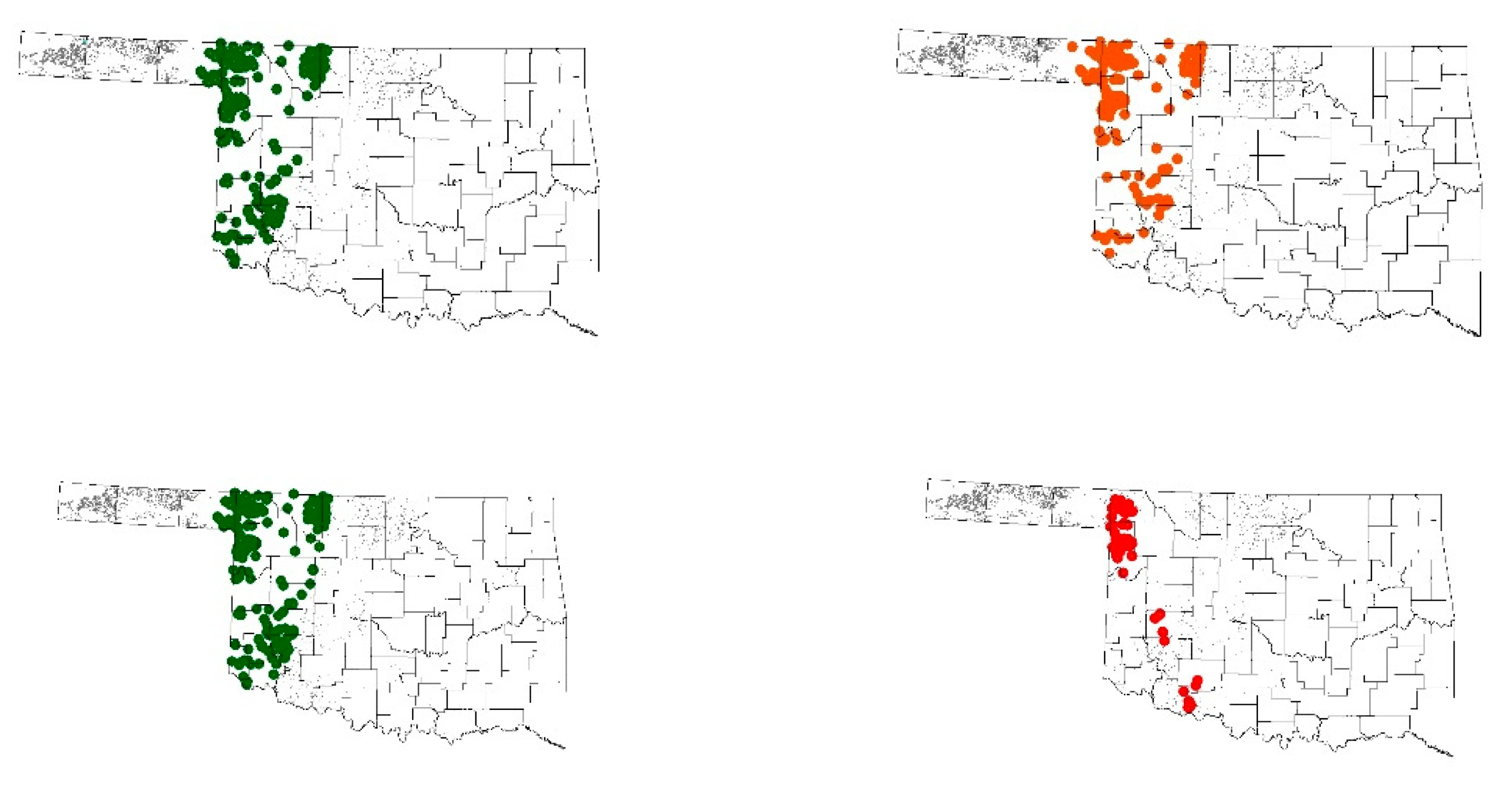
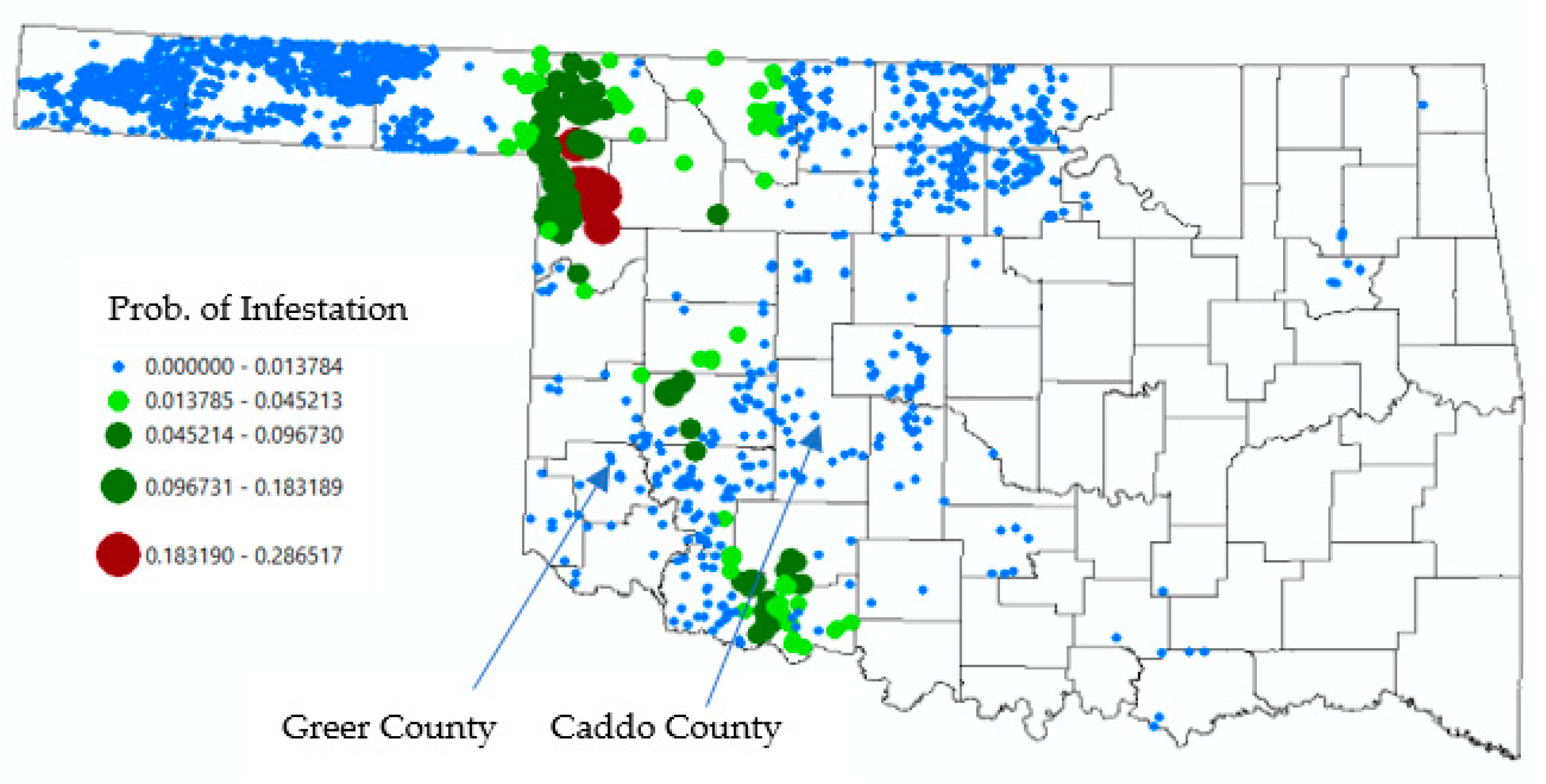
3.2. Discussion
3.3. Population Survivability
3.4. Weather Persistence and Improved Forecasting
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Aruna, C.; Suguna, M.; Visarada, K.; Deepika, C.; Ratnavathi, C.; Tonapi, V. Identification of sorghum genotypes suitable for specific end uses: Semolina recovery and popping. J. Cereal Sci. 2020, 93, 102955. [Google Scholar] [CrossRef]
- Foreign Agricultural Service (FAS). Available online: https://www.fas.usda.gov/data (accessed on 28 February 2023).
- Brewer, M.J.; Gordy, J.W.; Kerns, D.L.; Woolley, J.B.; Rooney, W.L.; Bowling, R.D. Sugarcane aphid population growth, plant injury, and natural enemies on selected grain sorghum hybrids in Texas and Louisiana. J. Econ. Entomol. 2017, 110, 2109–2118. [Google Scholar] [CrossRef]
- Gordy, J.W.; Brewer, M.J.; Bowling, R.D.; Buntin, G.D.; Seiter, N.J.; Kerns, D.L.; Reay-Jones, F.P.; Way, M. Development of economic thresholds for sugarcane aphid (Hemiptera: Aphididae) in susceptible grain sorghum hybrids. J. Econ. Entomol. 2019, 112, 1251–1259. [Google Scholar] [CrossRef] [PubMed]
- Bergtold, J.S.; Ramsey, S.; Maddy, L.; Williams, J.R. A review of economic considerations for cover crops as a conservation practice. Renew. Agric. Food Syst. 2017, 34, 62–76. [Google Scholar] [CrossRef]
- Smith, C.M.; Chuang, W.P. Plant resistance to aphid feeding: Behavioral, physiological, genetic and molecular cues regulate aphid host selection and feeding. Pest Manag. Sci. 2014, 70, 528–540. [Google Scholar] [CrossRef] [PubMed]
- Elliott, N.; Brewer, M.; Seiter, N.; Royer, T.; Bowling, R.; Backoulou, G.; Gordy, J.; Giles, K.; Lindenmayer, J.; McCornack, B. Sugarcane Aphid1 Spatial Distribution in Grain Sorghum Fields. Southwest. Entomol. 2017, 42, 27–35. [Google Scholar] [CrossRef]
- Szczepaniec, A. Assessment of a density-based action threshold for suppression of sugarcane aphids,(Hemiptera: Aphididae), in the Southern High Plains. J. Econ. Entomol. 2018, 111, 2201–2207. [Google Scholar]
- Ukoroije, R.B.; Otayor, R.A. Review on the bio-insecticidal properties of some plant secondary metabolites: Types, formulations, modes of action, advantages and limitations. Asian J. Res. Zool. 2020, 3, 27–60. [Google Scholar]
- Chen, C.; Harvey, J.A.; Biere, A.; Gols, R. Rain downpours affect survival and development of insect herbivores: The specter of climate change? Ecology 2019, 100, e02819. [Google Scholar] [CrossRef]
- Kobori, Y.; Amano, H. Effect of rainfall on a population of the diamondback moth, Plutella xylostella (Lepidoptera: Plutellidae). Appl. Entomol. Zool. 2003, 38, 249–253. [Google Scholar] [CrossRef]
- Huberty, A.F.; Denno, R.F. Plant water stress and its consequences for herbivorous insects: A new synthesis. Ecology 2004, 85, 1383–1398. [Google Scholar] [CrossRef]
- Ye, S.; Rogan, J.; Zhu, Z.; Hawbaker, T.J.; Hart, S.J.; Andrus, R.A.; Meddens, A.J.; Hicke, J.A.; Eastman, J.R.; Kulakowski, D. Detecting subtle change from dense Landsat time series: Case studies of mountain pine beetle and spruce beetle disturbance. Remote Sens. Environ. 2021, 263, 112560. [Google Scholar] [CrossRef]
- Gutierrez, J.; Barry-Ryan, C.; Bourke, P. The antimicrobial efficacy of plant essential oil combinations and interactions with food ingredients. Int. J. Food Microbiol. 2008, 124, 91–97. [Google Scholar] [CrossRef] [PubMed]
- Magstadt, S.; Gwenzi, D.; Madurapperuma, B. Can a Remote Sensing Approach with Hyperspectral Data Provide Early Detection and Mapping of Spatial Patterns of Black Bear Bark Stripping in Coast Redwoods? Forests 2021, 12, 378. [Google Scholar] [CrossRef]
- Meddens, A.J.; Hicke, J.A. Spatial and temporal patterns of Landsat-based detection of tree mortality caused by a mountain pine beetle outbreak in Colorado, USA. For. Ecol. Manag. 2014, 322, 78–88. [Google Scholar] [CrossRef]
- Leal-Sáenz, A.; Waring, K.M.; Sniezko, R.A.; Menon, M.; Hernández-Díaz, J.C.; López-Sánchez, C.A.; Martínez-Guerrero, J.H.; Mariscal-Lucero, S.D.R.; Silva-Cardoza, A.; Wehenkel, C. Differences in cone and seed morphology of Pinus strobiformis and Pinus ayacahuite. Southwest. Nat. 2021, 65, 9–18. [Google Scholar] [CrossRef]
- Robertson, C.; Wulder, M.; Nelson, T.; White, J. Risk rating for mountain pine beetle infestation of lodgepole pine forests over large areas with ordinal regression modelling. For. Ecol. Manag. 2008, 256, 900–912. [Google Scholar] [CrossRef]
- White, J.; Coops, N.; Hilker, T.; Wulder, M.; Carroll, A. Detecting mountain pine beetle red attack damage with EO-1 Hyperion moisture indices. Int. J. Remote Sens. 2007, 28, 2111–2121. [Google Scholar] [CrossRef]
- O’Neal, M.R.; Frankenberger, J.R.; Ess, D.R. Use of CERES-Maize to study effect of spatial precipitation variability on yield. Agric. Syst. 2002, 73, 205–225. [Google Scholar] [CrossRef]
- Kravchenko, A.; Robertson, G.; Thelen, K.; Harwood, R. Management, topographical, and weather effects on spatial variability of crop grain yields. Agron. J. 2005, 97, 514–523. [Google Scholar] [CrossRef]
- Stevens, S.C. Evidence for a Weather Persistence Effect on the Corn, Wheat and Soybean Growing Season Price Dynamics. 1990. Available online: https://ageconsearch.umn.edu/record/13907/ (accessed on 28 February 2023).
- Weatherhead, E.; Gearheard, S.; Barry, R.G. Changes in weather persistence: Insight from Inuit knowledge. Glob. Environ. Chang. 2010, 20, 523–528. [Google Scholar] [CrossRef]
- Duffy, C.; Fealy, R.; Fealy, R.M. An improved simulation model to describe the temperature-dependent population dynamics of the grain aphid, Sitobion avenae. Ecol. Model. 2017, 354, 140–171. [Google Scholar] [CrossRef]
- Ma, Z.S.; Bechinski, E.J. A survival-analysis-based simulation model for Russian wheat aphid population dynamics. Ecol. Model. 2008, 216, 323–332. [Google Scholar] [CrossRef]
- Acreman, S.; Dixon, A. The effects of temperature and host quality on the rate of increase of the grain aphid (Sitobion avenae) on wheat. Ann. Appl. Biol. 1989, 115, 3–9. [Google Scholar] [CrossRef]
- Angilletta, J.; Michael, J.; Dunham, A.E. The temperature-size rule in ectotherms: Simple evolutionary explanations may not be general. Am. Nat. 2003, 162, 332–342. [Google Scholar] [CrossRef]
- Asin, L.; Pons, X. Effect of high temperature on the growth and reproduction of corn aphids (Homoptera: Aphididae) and implications for their population dynamics on the northeastern Iberian peninsula. Environ. Entomol. 2001, 30, 1127–1134. [Google Scholar] [CrossRef]
- Auad, A.; Alves, S.; Carvalho, C.; Silva, D.; Resende, T.; Veríssimo, B. The impact of temperature on biological aspects and life table of Rhopalosiphum padi (Hemiptera: Aphididae) fed with signal grass. Fla. Entomol. 2009, 92, 569–577. [Google Scholar] [CrossRef]
- Bale, J.S.; Masters, G.J.; Hodkinson, I.D.; Awmack, C.; Bezemer, T.M.; Brown, V.K.; Butterfield, J.; Buse, A.; Coulson, J.C.; Farrar, J. Herbivory in global climate change research: Direct effects of rising temperature on insect herbivores. Glob. Chang. Biol. 2002, 8, 1–16. [Google Scholar] [CrossRef]
- Murúa, M.G.; Vera, M.A.; Michel, A.; Casmuz, A.S.; Fatoretto, J.; Gastaminza, G. Performance of field-collected Spodoptera frugiperda (Lepidoptera: Noctuidae) strains exposed to different transgenic and refuge maize hybrids in Argentina. J. Insect Sci. 2019, 19, 21. [Google Scholar] [CrossRef]
- Souza, M.; Davis, J. Detailed characterization of Melanaphis sacchari (Hemiptera: Aphididae) feeding behavior on different host plants. Environ. Entomol. 2020, 49, 683–691. [Google Scholar] [CrossRef] [PubMed]
- De Souza, M.A.; Armstrong, J.S.; Hoback, W.W.; Mulder, P.G.; Paudyal, S.; Foster, J.E.; Payton, M.E.; Akosa, J. Temperature dependent development of sugarcane aphids Melanaphis Sacchari, (Hemiptera: Aphididae) on three different host plants with estimates of the lower and upper threshold for fecundity. Curr. Trends Entomol. Zool. Stud. 2019. [Google Scholar] [CrossRef]
- Weisser, W.W.; Volkl, W.; Hassell, M.P. The importance of adverse weather conditions for behaviour and population ecology of an aphid parasitoid. J. Anim. Ecol. 1997, 1, 386–400. [Google Scholar] [CrossRef]
- Rodríguez-del-Bosque, L.A.; Silva-Serna, M.M.; Aranda-Lara, U. Effect of Natural and Simulated Rainfall and Wind on Melanaphis sacchari1 on Sorghum. Southwest. Entomol. 2020, 45, 357–364. [Google Scholar] [CrossRef]
- Balikai, R.; Lingappa, S. Bio-Ecology and Management of Sorghum Aphid: Melanaphis Sacchari; LAP LAMBERT Academic Publishing: Saarland, Germany, 2012. [Google Scholar]
- Patel, D.; Purohit, M. Influence of different weather parameters on aphid, Melanaphis sacchari infesting Kharif sorghum. Int. J. Plant Prot. 2013, 6, 484–486. [Google Scholar]
- Colares, F.; Michaud, J.; Bain, C.L.; Torres, J.B. Indigenous aphid predators show high levels of preadaptation to a novel prey, Melanaphis sacchari (Hemiptera: Aphididae). J. Econ. Entomol. 2015, 108, 2546–2555. [Google Scholar] [CrossRef]
- Mann, J.A.; Tatchell, G.M.; Dupuch, M.J.; Harrington, R.; Clark, S.J.; McCartney, H.A. Movement of apterous Sitobion avenae (Homoptera: Aphididae) in response to leaf disturbances caused by wind and rain. Ann. Appl. Biol. 1995, 126, 417–427. [Google Scholar] [CrossRef]
- Shao, X.; Zhang, Q.; Liu, Y.; Yang, X. Effects of wind speed on background herbivory of an insect herbivore. Écoscience 2020, 27, 71–76. [Google Scholar] [CrossRef]
- Leslie, P.H. On the use of matrices in certain population mathematics. Biometrika 1945, 33, 183–212. [Google Scholar] [CrossRef]
- Nordheim, E.V.; Hogg, D.B.; Chen, S.-Y. Leslie matrix models for insect populations with overlapping generations. In Estimation and Analysis of Insect Populations; Springer: Berlin/Heidelberg, Germany, 1989; pp. 289–298. [Google Scholar]
- Carter, N.; Aikman, D.; Dixon, A. An appraisal of Hughes’ time-specific life table analysis for determining aphid reproductive and mortality rates. J. Anim. Ecol. 1978, 47, 677–687. [Google Scholar] [CrossRef]
- Bannerman, J.A.; Roitberg, B.D. Impact of extreme and fluctuating temperatures on aphid–parasitoid dynamics. Oikos 2014, 123, 89–98. [Google Scholar] [CrossRef]
- Koralewski, T.E.; Wang, H.-H.; Grant, W.E.; Brewer, M.J.; Elliott, N.C. Evaluation of Areawide Forecasts of Wind-borne Crop Pests: Sugarcane Aphid (Hemiptera: Aphididae) Infestations of Sorghum in the Great Plains of North America. J. Econ. Entomol. 2022, 115, 863–868. [Google Scholar] [CrossRef] [PubMed]
- Wennergren, U.; Landin, J. Population growth and structure in a variable environment: I. Aphids and temperature variation. Oecologia 1993, 93, 394–405. [Google Scholar] [CrossRef] [PubMed]

| Year | TAVG (°F) | RAIN (Inches) | WSPD (Miles/Hours) | PDIR (16-Point) |
|---|---|---|---|---|
| 2013 | 79.80 | 0.13 | 14.80 | 7.29 |
| 2014 | 77.40 | 0.12 | 13.90 | 7.53 |
| 2015 | 78.50 | 0.03 | 11.90 | 7.65 |
| 2016 | 81.40 | 0.10 | 11.10 | 7.63 |
| 2017 | 77.00 | 0.08 | 10.50 | 6.43 |
| 2018 | 76.30 | 0.27 | 12.80 | 7.70 |
| 2019 | 73.30 | 0.13 | 9.78 | 8.32 |
| 2020 | 77.50 | 0.14 | 13.30 | 6.83 |
| Ave | 77.34 | 0.12 | 12.26 | 7.42 |
| Variable | Coeff. | Std. Err | Z | p > |z| | [95% CI] Lower Upper | |
|---|---|---|---|---|---|---|
| X | −0.0132 a | 0.001 | −10.22 | 0 | −0.0158216 | −0.010 |
| Y | 0.0094 a | 0.001 | 8.88 | 0 | 0.00739 | 0.0115 |
| X2 | −0.0003 a | 1.1 × 10−5 | −29.41 | 0 | −0.0003467 | −0.0003 |
| Y2 | −0.5 × 10−3 a | 8.1 × 10−6 | −5.81 | 0 | −0.0000632 | −3.1 × 10−5 |
| XY | −0.23 × 10−3 b | 1.2 × 10−5 | −2.00 | 0.046 | −0.0000467 | −4.20 × 10−7 |
| 2014 | −0.0484 | 0.0612 | −0.79 | 0.429 | −0.1684028 | 0.0715 |
| 2015 | −0.8925 a | 0.0981 | −9.09 | 0 | −1.084918 | −0.7001 |
| 2016 | −0.3168 a | 0.0778 | −4.07 | 0 | −0.4694808 | −0.1642 |
| 2017 | −0.2949 a | 0.0556 | −5.30 | 0 | −0.4040613 | −0.1857 |
| 2018 | 0.25337 a | 0.0434 | 5.83 | 0 | 0.168127 | 0.3386 |
| 2019 | −0.2769 a | 0.0511 | −5.41 | 0 | −0.3773219 | −0.1766 |
| 2020 | 0.2020 a | 0.0445 | 4.53 | 0 | 0.11464 | 0.2894 |
| a | −2.305402 a | 0.035933 | −64.16 | 0 | −2.375828 | −2.23498 |
| Var. | Coef. | Std. Err | z | p > |z| | [95% CI] Lower Upper | |
|---|---|---|---|---|---|---|
| X | 3.19 × 10−6 | 2.03 × 10−6 | −1.57 | 0.116 | −7.17 × 10−6 | 7.83 × 10−7 |
| Y | 1.85 × 10−5 | 1.77 × 10−6 | 10.49 | 0 | 1.51 × 10−5 | 0.000022 |
| 2014 | −0.00036 | 0.000435 | −0.82 | 0.415 | −0.0012079 | 0.0004979 |
| 2015 | −0.00292 | 0.000218 | −13.4 | 0 | −0.0033454 | −0.00249 |
| 2016 | −.001765 | 0.000344 | −5.13 | 0 | −0.0024399 | −0.00109 |
| 2017 | −0.00168 | 0.000282 | −5.95 | 0 | −0.0022324 | −0.00113 |
| 2018 | 0.002551 | 0.000486 | 5.25 | 0 | 0.001598 | 0.0035047 |
| 2019 | −0.00161 | 0.000273 | −5.88 | 0 | −0.0021415 | −0.00107 |
| 2020 | 0.001927 | 0.000462 | 4.17 | 0 | 0.001022 | 0.0028326 |
| Test Statistic | TAVG | RAIN | PDIR | WSPD |
|---|---|---|---|---|
| Serial correlation | ||||
| BSJK (LM) | 4.3 b | 3.3098 c | 2.6254 | 17.333 a |
| Breusch–Godfrey (LM) | 212.34 a | 89.813 a | 235.96 a | 345.01 a |
| Spatial dependence | ||||
| BSJK (LM) | 104.84 a | 146.37 a | 60.99 a | 130.65 a |
| Pesaran CS Dependence (Z) | 108.14 a | 97.8 a | 64.473 a | 105.26 a |
| Joint Serial–Spatial correlation | ||||
| BSJK (LM) | 894.68 a | 579.17 a | 1042.4 a | 1569.4 a |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lee, S.; Vitale, J.; Lambert, D.; Vitale, P.; Elliot, N.; Giles, K. Effects of Weather on Sugarcane Aphid Infestation and Movement in Oklahoma. Agriculture 2023, 13, 613. https://doi.org/10.3390/agriculture13030613
Lee S, Vitale J, Lambert D, Vitale P, Elliot N, Giles K. Effects of Weather on Sugarcane Aphid Infestation and Movement in Oklahoma. Agriculture. 2023; 13(3):613. https://doi.org/10.3390/agriculture13030613
Chicago/Turabian StyleLee, Seokil, Jeffrey Vitale, Dayton Lambert, Pilja Vitale, Norman Elliot, and Kristopher Giles. 2023. "Effects of Weather on Sugarcane Aphid Infestation and Movement in Oklahoma" Agriculture 13, no. 3: 613. https://doi.org/10.3390/agriculture13030613
APA StyleLee, S., Vitale, J., Lambert, D., Vitale, P., Elliot, N., & Giles, K. (2023). Effects of Weather on Sugarcane Aphid Infestation and Movement in Oklahoma. Agriculture, 13(3), 613. https://doi.org/10.3390/agriculture13030613







