Abstract
The practice of incorporating organic manures and fermented liquid biofertilizers of different origins and compositions can supply valuable nutrients to the soil. To ascertain the effect of fermented liquid organic nutrient formulations (FLONFs) on the rhizospheric microbial population, growth characteristics, nutritional status, and yield of brinjal, a field experiment was conducted in consecutive seasons in 2017 and 2018. Fermented liquid biofertilizers (panchagavya and jeevamrut) were prepared and applied along with organic manures to brinjal plants in ten treatment combinations in three replications. The treatment (T4) involving the combined use of organics and FLONFs contributed significantly to the soil dehydrogenase enzyme activity (4.9 mg TPF h−1 g−1 soil), phosphatase enzyme activity (25.5 mmoles PNP h−1 g−1 soil), and urease enzyme activity (0.27 mg NH4+ g−1 soil). The mean bacterial count, fungal count, and actinomycete levels were 203.3 × 108 cfu g−1, 4.34 × 103 cfu g−1, and 3.41 × 102 cfu g−1 soil, respectively, along with a mean soil microbial biomass of carbon value of 66.1 mg g−1 soil. The brinjal yield was maximal under treatment T4 with values of 389.2 q ha−1 and 153.7 q ha−1 for 2017 and 2018, respectively. As a result, for higher yields and soil longevity, the combination of organics and FLONFs can be advocated.
1. Introduction
Chemical fertilizers have caused degraded soil fertility in modern agriculture, making India’s soil acidic, threatening Indian agriculture’s long-term sustainability, and jeopardizing the farming community’s survival. The excessive input rate has resulted in serious human health and environmental threats, such as soil erosion, pesticide poisoning, water contamination, a dwindling groundwater table, water logging, and biodiversity impoverishment. To overcome the side effects of inorganic fertilizers on the soil, the application of organic manures is highly recommended. For example, the application of organics ameliorates the physicochemical properties, preserves the quality of the soil, and boosts the productivity of the crop. The soil quality includes the soil fertility and biological activity levels, which are inextricably linked because the mineralization of important organic elements occurs through the biomass [1]. India ranks 8th in the world in organic agricultural land area and first in total production per population. As per the National Program for Organic Production, the total area certified for organic agriculture measures 4.3 million ha (2020–2021), with 2.65 million ha being cultivable and 1.68 million ha being used for wild harvest collection. The maximum areas in Indian states certified as organic follow the order of Madhya Pradesh > Rajasthan > Maharashtra > Chhattisgarh > Himachal Pradesh > Jammu and Kashmir > Karnataka [2].
In addition to organic manures, traditional organic formulations have been embraced and used to enhance soil fertility and productivity. The application of fermented liquid bio-formulations such as panchagavya, jeevamrut, amritpani, humic acid, seaweed, and vermiwash promotes soil microorganisms, promotes plant growth, and increases root biomass. These formulations play a critical role in preserving and boosting the soil’s microflora population, which in turn increases the soil fertility and nutrient availability [3,4]. Additionally, organic manures (viz. compost, vermicompost, poultry manure, and farm yard manure) have the capacity to mobilize crop nutrients and improve crop productivity [5].
Furthermore, the addition of dung, jaggery, urine, gramme flour, and milk as constituents in organic inputs, which are rich in carbon and nitrogen, may be the reason for the higher total microbial counts in the fermented liquid organic nutrient formulations (FLONFs), which include panchgavya, matkakhad, jeevamrut, and beejamrit [4]. The high levels of microbial activity in jeevamrut multiply with its application in soil and serve as a stimulant to proliferate soil microbial activity [6]. The diversity of microorganisms, including lactic acid bacteria, actinomycetes, fungi, phosphobacteria, methylotrophs Azotobacter, Pseudomonas, and Azospirillum, are known to be present in panchagavya, jeevamruth, and beejamruth made from cow products [7]. The enzymatic activity is indicated by the presence of easily degradable organic compounds in organics and FLONFs, such as simple sugars [8,9].
Additionally, soil enzymes have a pivotal role in the maintenance of ecology, physicochemical properties, fertility, and organic matter decomposition in the soil. The soil’s enzymatic activity catalyzes the microorganisms’ biochemical activity, demonstrating the soil’s ability to allow the basic biochemical processes required for soil fertility. Organics improve the soil’s overall microbial population and enzymatic activity [3].
Soil microorganisms are thought to be critical in the maintenance of soil health, sustainability, quality, and productivity [10]. The rhizosphere’s diversity of microorganisms has a significant impact on plant growth [11]. They act as catalysts for the breakdown of organic matter, the cycling of nutrients (such as carbon, nitrogen (N), phosphorus (P), and other nutrients), and other chemical transformations [12]. Although chemical fertilizers and pesticides increase yields, their indiscriminate use and intensive cropping systems have resulted in soil health depletion, which has resulted in declining crop productivity [13].
In countries with a predominantly vegetarian population, such as India, vegetables are an essential part of the daily diet. Their significance as components of nutritional security for healthy food and better sources of dietary essential nutrients, minerals, and vitamins is growing, as is the public awareness of the negative health effects of indiscriminate agrochemical use in crop productivity. The latter has increased demand for vegetables that are cultivated organically because they are purported to be devoid of any lingering toxins that could be hazardous to one’s health [2]. Around the world, there is a growing awareness of, demand for, and market value for organic produce. One study reported the effects of Azotobacter sp. and Azospirillum sp. as liquid biofertilizers on the growth, quality, and yield of brinjal, where the maximum growth was observed in treatment that comprised 100% NPK along with Azotobacter and Azospirillum [14]. Consequently, a growing number of farmers are making the switch to organic farming. In this scenario, finding new organic manures that are affordable and environmentally benign is crucial. To the best of our knowledge, the information on the use of organic fertilizers for improving the nutritional status of brinjal is still lacking. It was hypothesized for the present experiment that the use of organics would strengthen the soil properties, microbial characteristics, and yield attributes of brinjal. Hence, in this study, the preliminary field evaluations were carried out to ascertain the effect of the fermented liquid organic nutrient formulations (FLONFs) (jeevamrut and panchagavya) on the rhizospheric microbial population, growth characteristics, nutritional status, and yield of Solanum melongena (L.) cultivations.
2. Materials and Methods
2.1. Geomorphology and Climate
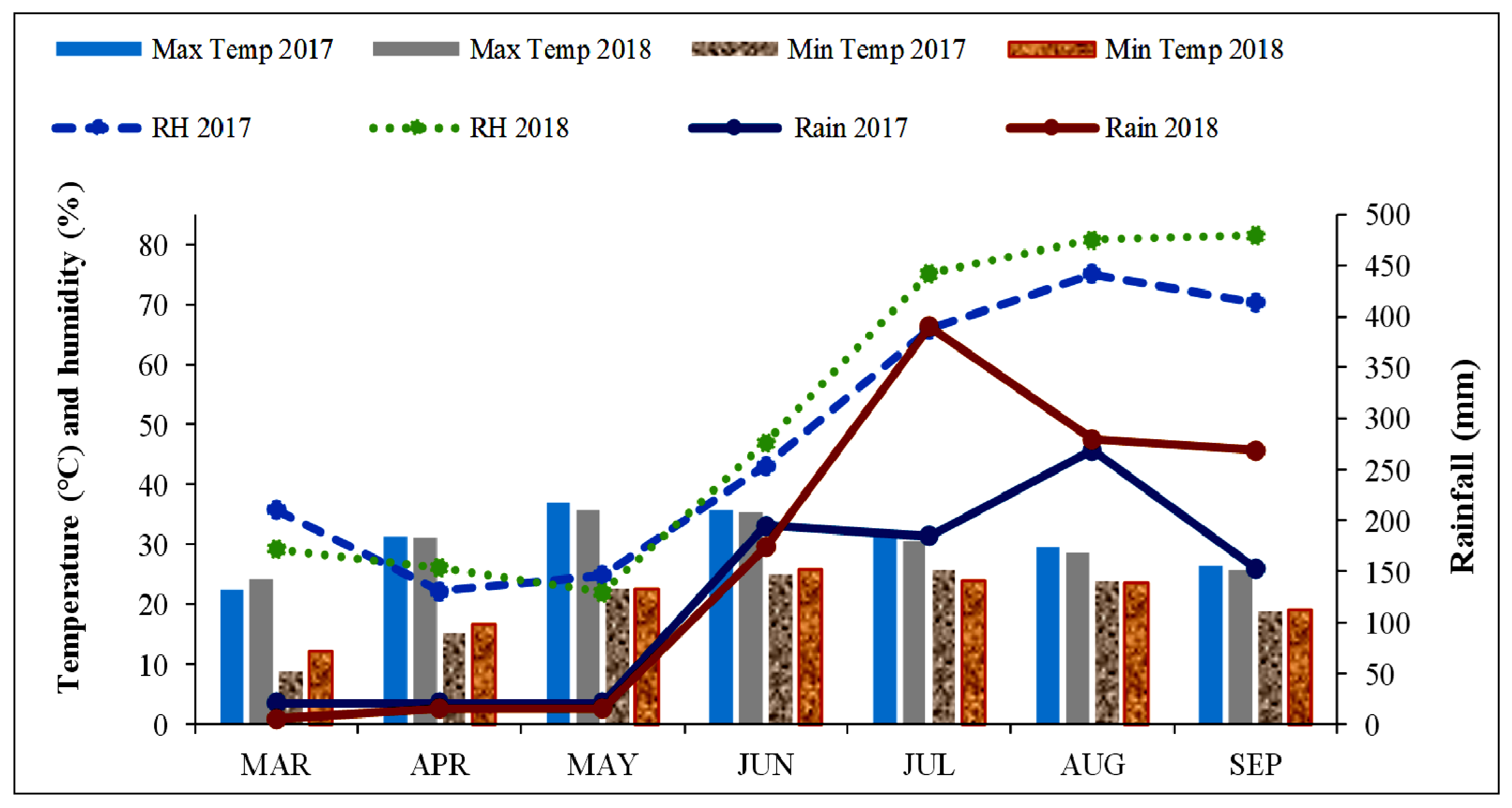
The field experiment was carried out in two consecutive growing seasons in 2017 and 2018 at the experimental farm of Dr. Y.S. Parmar University of Horticulture and Forestry Nauni, Solan (HP), which is located at 30°52′ latitude north and 77°11′ longitude east at an elevation of 1260 m above the mean sea level in Himachal Pradesh’s mid-hills zone. The normal rainfall in the area is approximately 1115 mm, with approximately 75% falling in the course of the monsoon season (mid-June–mid-September). The climatic conditions in both seasons of 2017 and 2018 are presented in Figure 1.
Figure 1.
Agrometeorological data during the crop period (2017 and 2018).
The maximum temperatures during both crop seasons varied from 22.4 to 36.9 °C and 24.3 to 35.8 °C, respectively. However, the minimum temperatures varied from 8.9 to 25.7 °C and 12.2 to 25.7 °C during 2017 and 2018, respectively. The average relative humidity in 2017 ranged from 22.3 to 75.1% and in the year 2018 it varied from 22 to 81.6%. The total rainfall rates during the crop season (March to September) were 865 mm and 1150 mm in 2017 and 2018, respectively. The rainfall from March to June decreased during 2018 in comparison to 2017, whereas the next three months (July–September) received excess rain with higher relative humidity, which damaged the crops and caused higher levels of disease incidence. According to the USDA Soil Taxonomy, the soils in the study area are classified as Inceptisol soils. The standard methods were followed to determine the pH, EC, and OC values, which were found to be 6.5, 0.14 dSm−1, and 2.05 g kg−1, respectively [15,16]. The method proposed by Subbiah and Asija, the Olsen extractable P method, and the method proposed by Merwin and Peech were used to estimate the availability rates of N, P, and K to be 274.4 Kg ha−1, 55.2 Kg ha−1, and 331 Kg ha−1 in the soil, respectively [17,18,19]. Additionally, the initial micronutrient levels of Zn, Cu, Fe, and Mn in the soil were 3.11 mg kg−1, 98.0 mg kg−1, 2.17 mg kg−1, and 10.32 mg kg−1, respectively [20], as calculated using the AAS instrument (Varian AA 240FS, LabX, 334 King Street, Midland, ON, Canada).
2.2. Experimental Design and Physicochemical Analysis
The experiment comprised 10 treatments in a randomized block design (RBD) with the three replications given in Table 1. For the RDN (recommended dose of nitrogen), the vermicompost (VC) and poultry manure (PM) were used to apply the nutrients at a nitrogen-equivalent ratio of 50:50. The seeds of the brinjal (variety “Pusa purple long”) were sown in a well-prepared raised nursery bed. The one-month-old seedlings were transplanted with a spacing of 60 cm × 45 cm in a plot measuring 4.84 m2 (2.20 m (length) × 2.20 m (breadth)) and three to four irrigation phases were applied in this process. Here, 62 kg of N (137 kg of urea), 62 kg of P205 (385 kg of single superphosphate), and 30 kg of K2O (50 kg of muriate of potash) per acre were applied at the time of transplanting. After two pickings, again 62 kg of N (137 kg of urea) per acre was applied.

Table 1.
Treatment details for the experimental field.
The recommended doses of nutrients for brinjal for nitrogen, phosphorus, and potassium were 50 kg ha−1, 60 kg ha−1, and 45 kg ha−1, respectively, with farmyard manure (FYM) at 100 q ha−1. The fermented liquid organic formulations 5% panchagavya and 5% jeevamrut were applied two times as a soil drench (250 mL plant−1) at a 15-day interval after transplanting. The organic liquid formulations’ nutrient status and microbial load levels are given in Table 2. Throughout the investigation, the experimental plants were subjected to the same recommended cultural practices.

Table 2.
Nutrient status and microbial load levels in panchagavya and jeevamrut.
2.3. Yield, Nutrient Content, and Uptake
The average yield of the fruit was calculated in quintals per hectare (q ha−1). The plant samples (including the fruits, leaves, stems, and roots) were procured, cleaned, and air-dried in the shade before being ground in an electric steel grinder and stored in paper bags for further chemical analyses. Furthermore, for the nutrient content, the plants were chemically analyzed for their nitrogen content using the micro-Kjeldhal method; for phosphorus using the vanado-molybdate-phosphoric yellow color method; for potassium using the flame photometer method [15]; and for micronutrients using an atomic absorption spectrophotometer (AAS) [20].
2.4. Physicochemical Properties
Before the experiment began, composite soil samples from soil depths of 0–15 cm were taken. After the experiment terminated, soil from furrow slices was collected to determine the impacts of various organic inputs on the pH using a potentiometric analysis with a 1:2 soil/water suspension, including the electrical conductivity (Wheatstone bridge circuit method; 1:2 soil/water suspension); organic carbon (rapid titration method); available nitrogen (N) from alkaline potassium permanganate; available phosphorus (P) from sodium bicarbonate extraction; available potassium (K) using the ammonium acetate method; and available copper (Cu), iron (Fe), zinc (Zn), and manganese (Mn) using the DTPA extractant method.
2.5. Microbiological Properties
The viable microbial count was recorded using the serial dilution standard spread plate technique [21] on nutrient agar (NA) medium, potato dextrose agar medium, and Kenknight and Munaier’s medium. The population was expressed as cfu g−1 soil (colony-forming units per gram of soil). The soil fumigation extraction method was used for microbial biomass-C determination [22]:
where K represents 0.25 ± 0.05 (factor showing the efficiency of the microbial biomass of carbon extraction), EC (F) represents the total amount of fumigated soil-extractable carbon, and EC (UF) represents the total amount of non-fumigated soil-extractable carbon. The TTC reduction method (2,3,5-triphenyl tetrazolium chloride) was used to estimate the dehydrogenase activity in liquid formulations [23], Tabatabai and Bremner’s procedure was used to estimate phosphatase activity [24], and the Hoffman method was used to estimate urease activity [25].
2.6. Statistical Analysis
The SPSS version 16.0 (SPSS Inc., Chicago, IL, USA) packages were used to analyze the data recorded during the investigations. A one-way analysis of variance (ANOVA) was performed on pooled data using the RBD (randomized block design), and the treatments were compared at the 0.05% level of significance. A Pearson correlation analysis was also performed in the present study.
3. Results
3.1. Yield and Uptake of Nutrients
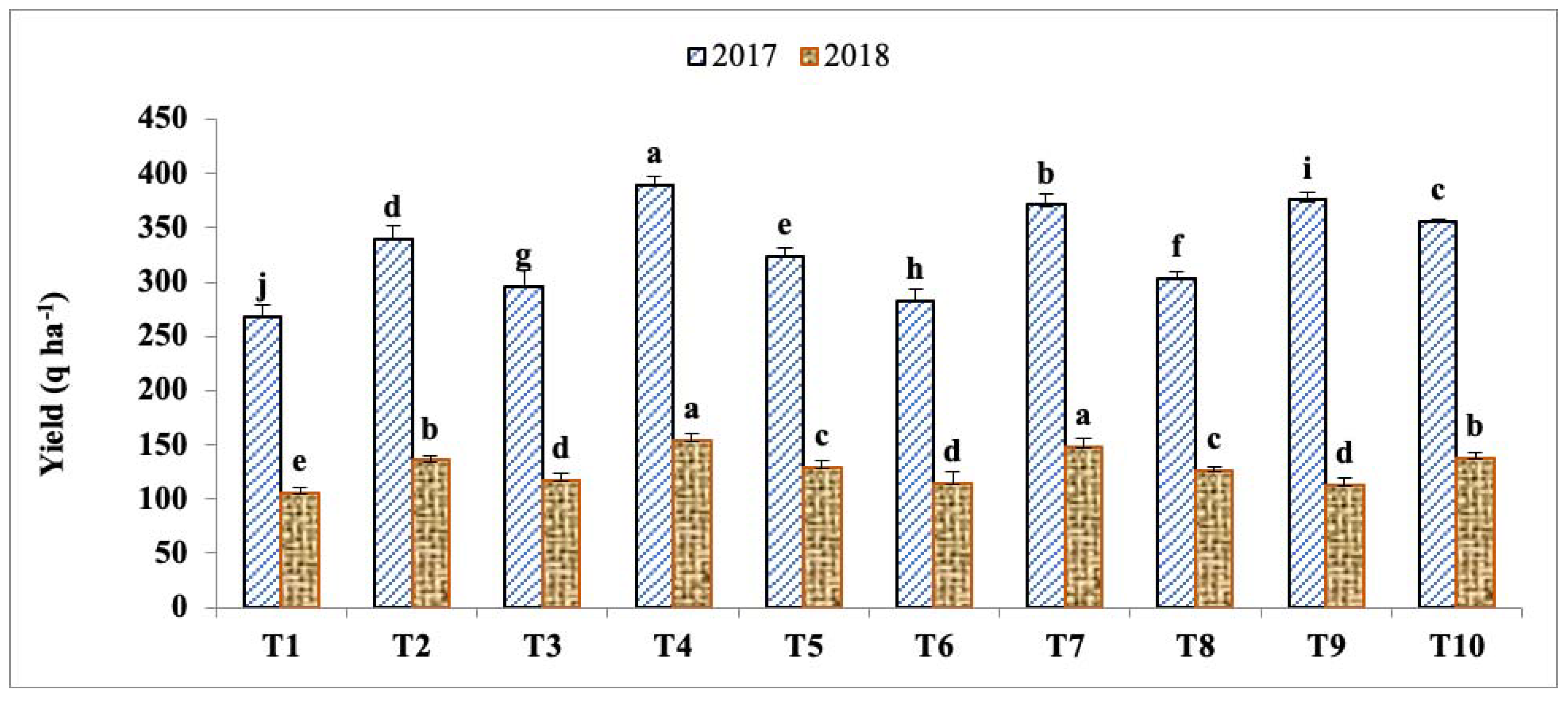
Significant difference were recorded in the highest brinjal yields (389.2 q ha−1 and 153.7 q ha−1) during 2017 and 2018, respectively, under treatment T4, and likewise for the lowest yields (268.6 q ha−1 and 105.6 q ha−1, respectively) under treatment T1 (Figure 2). During 2018, there was an infestation of fruit and shoot borer, as well as fungal diseases such as leaf blight, bacterial wilt, Phomopsis blight, and Phytophthora blight, leading to the early termination of crops and yield reductions.
Figure 2.
Effects of panchagavya, jeevamrut, and organic manures on the yield of brinjal. Treatment details are given in Table 1. Using Duncan’s multiple ranges test, the values with similar letters in superscript do not differ significantly at the 5% level.
The nutrient contents in the plants showed significant differences with FLONFs and organic amendments; the maximum nitrogen, phosphorus, and potassium contents were 2.36%, 0.69%, and 2.17%, respectively, under the treatment T4, while the minimum nitrogen, phosphorus, and potassium contents were 2.06%, 0.48%, and 1.96%, respectively, under treatment T1 (Table 3). Similarly, the highest micronutrient contents for Cu, Fe, Zn, and Mn (i.e., 2.69 mg kg−1, 243.3 mg kg−1, 2.9 mg kg−1, and 113.9 mg kg−1, respectively), were recorded under treatment T4 and the lowest Cu, Fe, Zn, and Mn contents (i.e., 1.5 mg kg−1, 203.0 mg kg−1, 2.2 mg kg−1, and 62.9 mg kg−1, respectively) were recorded under treatment T1 (Table 3).

Table 3.
Effects of panchagavya, jeevamrut, and organic manures on the macronutrient and micronutrient contents of plants.
The application of FLONFs and organic amendments resulted in significant differences in plant total nutrient uptake. The maximum total N, P, and K uptakes rates of 127.4 kg ha−1, 37.1 kg ha−1, and 117.3 kg ha−1, respectively, were recorded under treatment T4, while the minimum total N, P, and K uptake rates of 40.1 kg ha−1, 9.4 kg ha−1, and 38.3 kg ha−1, respectively, were recorded under treatment T1. Similarly, there were significant differences in micronutrient uptake by the plants. Treatment T1 had the highest total Cu, Fe, Zn, and Mn uptake rates of 146.5 g ha−1, 13,192.0 g ha−1, 153.4 g ha−1, and 6161.3 g ha−1, while treatment T1 had the lowest total Cu, Fe, Zn, and Mn uptake rates of 29.5 g ha−1, 3967.9 g ha−1, 42.5 g ha−1, and 1230.7 g ha−1, respectively (Table 4).

Table 4.
Effects of panchagavya, jeevamrut, and organic manures on nutrient uptake by plants.
3.2. Soil Physiochemical Properties and Nutrient Contents in the Soil
The application of fermented organic liquid manures (jeevamrut and panchagavya) and organic manures (VM and PM) had no significant effect on the pH and EC levels during both years of the investigation. The average pH values ranged from 6.37 to 6.53, whereas the EC values ranged from 0.13 to 0.16 d Sm−1. The results for the pH and EC were statistically non-significant. With FLONF application and organic manures, the mean highest organic carbon contents ranged from 24.5 g kg−1 to 20.8 g kg−1. There were significant differences in the average organic carbon contents; treatment T4 had the highest (24.5 g kg−1) content and treatment T9 had the lowest (20.8 g kg−1) content. The T4 treatment was statistically on par with the T7 and T10 treatments, as presented in Table 5.

Table 5.
Effects of panchagavya, jeevamrut, and organic manures on physicochemical properties and nutrient contents in the soil.
3.3. Available Macronutrients
The mean macronutrient values in the soil samples for available nitrogen ranged from 419.1 kg ha−1–317.8 kg ha−1; for available phosphorus from 86.5 kg ha−1–65.8 kg ha−1; and for available potassium from 478.2 kg ha−1–395.5 kg ha−1, as shown in Table 5. Differences in the average nitrogen, phosphorous, and potassium availability with the application of organic liquid manures and amendments were significant. The results of treatment T2 (398.0 kg ha−1) were statistically on par with treatment T3 (394.8 kg ha−1) for N as compared to the other treatments. The values for treatment T4 (86.5 kg ha−1 and 478.3 kg ha−1) were statistically on par with treatments T7 (84.1 kg ha−1) and T2 (473.0 kg ha−1) for P and K, respectively.
3.4. DTPA-Extractable Micronutrients
The mean DTPA-extractable micronutrient contents of Cu, Fe, Zn, and Mn of 4.50 mg kg−1, 19.9 mg kg−1, 2.95 mg kg−1, and 12.8 mg kg−1 were the highest under treatment T4 and were the lowest under treatments T1 Cu, Fe, Zn, and Mn (i.e., 3.16 mg kg−1, 15.6 mg kg−1, 2.26 mg kg−1, and 10.5 mg kg−1, respectively). The DTPA-extractable micronutrients had a significant effect due to the application of liquid inputs and organic manures, as shown in Table 5.
3.5. Soil Microbiological Properties
3.5.1. Soil Enzymes
The mean activity levels of the dehydrogenase enzyme in the soil were highest (4.9 mg TPF h−1 g−1 soil) in treatment T4 and lowest (2.9 mg TPF h−1 g−1 soil) in treatment T1, the phosphatase enzyme levels in the soil were highest (25.5 mmole PNP h−1 g−1 soil) in treatment T4 and lowest (17.6 mmole PNP h−1 g−1 soil) in treatment T1, and the urease enzyme levels in the soil were highest (0.27 μg NH4+ g−1 soil) in treatment T4 and lowest in treatment T1 (0.18 μg NH4+ g−1 soil). The differences in the average dehydrogenase, phosphatase, and urease enzymatic activity levels with the application of FLONFs and organic manures were significant (Table 6).

Table 6.
Effects of panchagavya, jeevamrut, and organic manures on the microbiological properties of the soil.
3.5.2. Viable Microbial Count
The mean viable bacterial count was the highest (203.3 × 108 cfu g−1 soil) for the population of bacteria under the T4 treatment, and the lowest (144.7 × 108 cfu g−1 soil) viable bacterial count was recorded under the T1 treatment, as per the data embodied in Table 5. The different organic treatments had significant impacts on the fungal population. The mean highest (4.34 × 103 cfu g−1 soil) fungal count was recorded under the T4 treatment and the lowest (3.15 × 103 cfu g−1 soil) fungal count was recorded with the T1 treatment. The mean highest (3.41 × 102 cfu g−1 soil) actinomycete count was recorded under the T4 treatment and the lowest (2.64 × 102 cfu g−1 soil) actinomycete count was recorded under the T1 treatment. The significant effects of the organic manure and liquid inputs on the viable microbial populations in the soil are shown in Table 5.
3.5.3. Soil Microbial Biomass-C
The mean highest soil microbial biomass-C content (66.1 μg g−1 soil), i.e., the microbial biomass of carbon, was registered under treatment T4, which was statistically on par with treatment T7 (65.2 μg g−1 soil), while the lowest (43.6 μg g−1 soil) microbial biomass-C content was registered under treatment T1 (Table 5). The differences in average soil microbial biomass-C content with the application of FLONFs and organic manures were significant.
3.5.4. Pearson Correlation Analysis of the Yields, Nutrient Content of the Plants, and Soil Properties
The results of the Pearson correlation analysis for the yields, plant nutrient contents, and soil properties after the application of organic liquid formulations and amendments are given in Table 7. The yield and N, P, and K contents in the plants had a positive correlation with all soil macronutrients, micronutrients, and microbiological properties except the soil pH and electrical conductivity. For the yield, the highest correlation was recorded with the phosphatase enzyme (r = 0.998). The nitrogen content in the plants had the highest correlation with the dehydrogenase enzyme (DHA) (r = 0.997). The phosphorus content in the plants had the highest correlation with fungi (r = 0.988), whereas potassium content had the highest correlation with urease (r = 0.997).

Table 7.
Correlation of crop yield and nutrient content values with soil properties.
3.5.5. Pearson Correlation Analysis of Microbial Properties and Nutrient Contents of Soil
The results of the Pearson correlation analysis for the soil microbial properties with the available macro- and micronutrient contents of soil are shown in Table 8. Organic carbon, dehydrogenase, and phosphatase had the highest positive correlation rates with DTPA-Fe (r = 0.952, r = 0.975, r = 0.978, respectively). Urease had the highest positive correlation with DTPA-Mn (r = 0.973). The bacterial count and fungi showed positive correlations with DTPA-Cu (r = 0.968, r = 0.974, respectively). Actinomycetes showed a positive correlation with DTPA-Zn (r = 0.965) and the microbial C-biomass showed a positive correlation with DTPA-Mn (r = 0.984).

Table 8.
Correlation of soil biological properties with nutrient contents in soil.
4. Discussion
4.1. Plant Development and Nutrient Uptake
The yield-determining components such as the fruit number are increased with the application of PM and inorganic fertilizer, which could be because of sufficient availability, which in turn improves the transmission of nutrients and photoassimilates, resulting in improved plant growth. Likewise, results were reported by [26] in snake gourd with the application of vermicompost, panchagavya, and 3G extract, and by Arahunashi [27] in tomato by applying FYM, poultry manure, and RDN. From the results, it was shown that the organic manures along with the panchagavya and jeevamrut had a significant influence on the fruit weight, diameter, and length, leading to a significant increment in the yield of brinjal. A significant increase in the brinjal yield was observed due to the ample supply of nutrients at various stages of crop growth, the presence of growth regulators in panchagvya, and the availability of photoassimilates. The results recorded showed that the influence of jeevamrut, panchagavya, and cow urine significantly varied the yield productivity of capsicum [28]. The substantial increase in nutrient intake could be attributable to the nutrients’ increased accessibility from indigenous sources and farmyard manure, vermicompost, and poultry manure, as well as liquid formulations of panchagavya and jeevamrut in brinjal and bioregulators such panchagavya, which offer additional macro- and micronutrients, along with growth regulators such as auxins and gibberellic acid, which assisted in producing more biomass and improved the nutrient recovery. The results showed that the spraying of panchagavya increased the uptake in black gram [29], and the application of farmyard manure, vermicompost, and jeevamrut also increased the uptake in sweet corn and black gram [30,31] (Table 2 and Table 3).
4.2. Soil Physicochemical Properties
Organic matter is essential to the quality and sustainability of the environment. In tropical and subtropical soils, intensive cultivation relying solely on inorganic fertilizers with little or without any organic fertilizer incorporation has resulted in lower carbon contents. The application of organic manures (VM) accounts for the cumulation of organic matter in soil in sweet corn, brinjal, and cucumber [32,33,34]. The higher increment in total SOC in soils amended with long-term PM may be a direct result of PM-C input [35,36]. The N mineralization from added organics and panchgavya was attributed to increases [37,38]. The increased P availability in soil following organic application is primarily due to the release of inorganic P from added organics, as well as the impedition of P adsorption by organic compounds secreted by the organics [37]. Additionally, an increase was observed in the availability of NPK with the combined application of FYM and RDN in wheat [39] and an increment in the available NPK content was observed with the application of vermicompost in okra and onion [40]. Because of the presence of higher amounts of microorganisms, when compared to other organic amendments, the soils that were enriched with panchgavya significantly allowed improved basal respiration [41]. Organic amendments such as farmyard manure not only add Zn to the soil but also promote biochemical reactions that dissolve inaccessible Zn. The Fe availability significantly increased probably due to increased concentrations of soil organic carbon and alterations in the soil microenvironment, which might have led to the release of plant-available Mn in a pearl millet–wheat cropping system [42]. The usage of VM, PM, and FYM in conjunction with PGPR increased the availability of micronutrients, possibly due to chelating agents being released during organic matter decomposition, which may have prevented micronutrient precipitation, oxidation, and leaching [12]. The effect of chicken manure on radishes significantly increased the soil-available micronutrients (Fe, Zn and Mn) [43] (Table 4). Microbes’ synergistic interactions improve soil productivity and function. Bio-enhancer preparations comprise efficient microorganisms that function continuously as renewable bio-inoculums to carry out mineralization processes and restore soil fertility. A microbial analysis of bio-enhancers [44] revealed that panchagavya had the maximum total bacteria content (6.25 × 109 cfu mL−1) and jeevamrut had the minimum content (3.24 × 109 cfu mL−1) of total bacteria, whereas jeevamrut had the highest fungal population (1.20 × 107 cfu mL−1) and panchagavya had the lowest (0.20 × 105 cfu mL−1). Additionally, these biostimulants function as reservoirs for significant labile pools of carbon and mineral nutrients, which release nutrients as the microorganisms perish [45]. Fermented organic liquid inputs had significant effects on microbial populations in maize, paddy, soybean, and field bean crops [46]. The microbial activity of the soil was enhanced as a result of the application of varied concentrations of panchagavya to the seeds of legumes and cereals, including pea, gramme, green gram, black gram, dry bean, lentil, moth bean, and soybean [41]. Jeevamrut contains a high microbial load that proliferates in the soil and functions as a tonic to boost microbial activity [6]. The FLONFs of panchagavya, jeevamruth, and beejamruth contain high concentrations of beneficial microorganisms such as actinomycetes, fungi, methylotrophs, lactic acid bacteria, Pseudomonas, Azospirillum, Azotobacter, and Phosphobacteria, as these are made from cow products [9]. The activity of enzymes in various organic inputs indicates the presence of easily biodegradable compounds such as simple sugars. Soil enzyme activity increases are indicators of soil fertility, as it is affected by the climate, conditioners, cultivation practices, crop type, and edaphic properties. Dehydrogenases are linked to soil microbiological activity because they are found in living cells, where they catalyze oxidoreductive processes, and also in soils [47]. The phosphatase activity in the soil is associated with the activity of enzymes, which is linked with soil colloids and humic substances and also with live and dead cells, plants, and microorganisms [47]. The application of FYM, beejamrut, jeevamrut, and panchagavya in chili resulted in a significant increment in dehydrogenase activity in the soil [48]. As a result of the cumulative effect of the amendments, the soil microbial biomass, a living component of the soil organic matter, acts as a labile reservoir for plant-available N, P, and S (sulfur), affecting the soil’s ability to supply nutrients to plants via soil organic matter turnover [49]. The application of organic manures (FYM) and biofertilizers (Azotobacter) resulted in a significant increase in soil microbial biomass-C due to the application of FYM, rock phosphate, and neem cake in a paddy field [50,51] (Table 5).
4.3. Correlation Analysis
Significant correlations of the yield and nutrient contents in plants with the available macronutrients (N, P, and K), DTPA-extractable micronutrients (Cu, Fe, Zn, and Mn), microbial properties (viable microbial count, enzymes (dehydrogenase, phosphatase, urease), and microbial biomass-C contents were noted in this study. Significant correlation were recorded between microbes, enzymes, and organic carbon with available soil nutrients. The microbes’ release enzymes present in the liquid formulations, further accentuating the mineralization of nutrients in the soil, which are proliferated. Additionally, the organic amendments help in the slow release of nutrients, which are used up by the plants, resulting in high yields and increases in nutrient contents [43,47,48,50] (Table 6 and Table 7).
5. Conclusions
This investigation sought to identify the best soil and crop management practices for promoting the slower microbial decomposition of organic matter, causing net carbon buildup in soils and potentially mitigating the negative effects of global warming and climate change. This study is a novel approach to determining the best possible combination of organics beneficial for increasing the nutritional status of brinjal. The results suggested that the different treatment combinations of poultry manure (0.81 t ha−1) + vermicompost (1.2 t ha−1) along with the drenching of panchagavya (5%) and jeevamrut (5%) were found to be the best options for obtaining higher yields and sustaining the longevity of the soil health. Increases in the available N, P, and K contents of 20%, 24%, and 14%, respectively, were recorded over the recommended doses of these nutrients. Significant increases in the DTPA-extractable micronutrient content, soil enzymatic activity, viable microbial count, and microbial biomass of carbon were recorded with the application of organic manures and liquid organic bio-formulations. The affirmative effects of the organic manures and liquid organic bio-formulations help in releasing nutrients slowly by primarily immobilizing organic compounds and the biomass, thereby improving the soil’s physicochemical properties, soil moisture holding capacity, and nutrient build-up, which are crucial to ameliorating the soil quality and health. Thus, this study suggests the use of organic manures and fermented liquid organic nutrient formulations to improve the nutritional status of brinjal. Additionally, an advanced concept for organic formulations was introduced in our experiments, which would be beneficial for achieving improved crop quality.
Author Contributions
Conceptualization, G.R., R.K., V.S., G.S. and S.C.; methodology, G.R., R.K. and V.S.; software, G.R.; validation, G.R., R.K., V.S. and S.S.D.; formal analysis, S.C. and G.S.; investigation, G.R., R.K., V.S., G.S. and S.C.; resources, S.S.D.; data curation, A.M.A., A.G., G.R. and R.K.; writing—original draft preparation, G.R., R.K. and V.S.; writing—review and editing, G.R., R.K., A.M.A., A.G., A.H. and V.S.; visualization, G.R., R.K. and V.S.; supervision, A.M.A., A.G. and S.S.D.; project administration, G.R., R.K. and V.S.; funding acquisition, A.M.A., A.G., G.R., R.K., V.S., G.S. and S.C. All authors have read and agreed to the published version of the manuscript.
Funding
The current work was funded by Department of Soil Science, Punjab Agricultural University, Ludhiana, Punjab 141027, India and also partially funded by the Princess Nourah bint Abdulrahman University Researchers Support Project number (PNURSP2023R65), Princess Nourah bint Abdulrahman University, Riyadh, Saudi Arabia.
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
All data are available in the manuscripts.
Acknowledgments
The authors also extend their gratitude to Department of Soil Science, Punjab Agricultural University, Ludhiana 141027, India and also the Princess Nourah bint Abdulrahman University Researchers Support Project number (PNURSP2023R65), Princess Nourah bint Abdulrahman University, Riyadh, Saudi Arabia, for supporting the current research.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Patel, D.P.; Das, A.; Kumar, M.; Munda, G.C.; Ngachan, S.V.; Ramkrushna, I.; Jayanta, L.; Pongla, N.; Buragohain, J.; Somireddy, U. Continuous application of organic amendments enhances soil health, produce quality and system productivity of vegetable- based cropping systems in subtropical eastern Himalayas. Exp. Agric. 2015, 51, 85–106. [Google Scholar] [CrossRef]
- Willer, H.; Trávníček, J.; Meier, C.; Schlatter, B. (Eds.) The World of Organic Agriculture. Statistics and Emerging Trends 2022; Research Institute of Organic Agriculture FiBL: Frick, Switzerland; IFOAM—Organics International: Bonn, Germany, 2022; Available online: www.organic-world.net/yearbook/yearbook-2022.html (accessed on 7 February 2023).
- Chatterjee, R.; Bandyopadhyay, S. Effects of organic, inorganic and biofertilizers on plant nutrient status and availability of major nutrients in tomato. Int. J. Bio-Resour. Stress Manag. 2014, 5, 93–97. [Google Scholar] [CrossRef]
- Sharma, S.K.; Jain, D.; Choudharya, R.; Jata, G.; Jain, P.; Bhojiya, A.A.; Jain, R.; Yadav, S.K. Microbiological and enzymatic properties of diverse jaivikkrishi inputs used in organic farming. Indian J. Tradit. Knowl. 2021, 20, 237–243. [Google Scholar]
- Joshi, H.N.; Varma, L.R.; More, S.G. Effects of organic nutrients in combination with biofertilizers on uptake N, P, K and yield of garden pea (Pisum sativum L.) CV. Pharma Innov. J. 2020, 9, 385–389. [Google Scholar]
- Palekar, S. Shoonyabandovaladanaisargikakrushi; Swamy Anand Agri Prakashana: Bangalore, India, 2006. [Google Scholar]
- Sreenivasa, M.N.; Nagaraj, M.; Naik, N.; Bhat, S.N. Beneficial traits of microbial isolates of organic liquid manures. In Proceedings of the First Asian PGPR Congress for Sustainable Agriculture, Hyderabad, India, 21–24 June 2009; ANGRAU: Hyderabad, India. [Google Scholar]
- Pane, C.; Spaccini, R.; Piccolo, A.; Scala, F.; Bonanomi, G. Compost amendments enhance peat suppressiveness to Pythium ultimum, Rhizoctoniasolani and Sclerotinia minor. Biol. Control 2011, 56, 115–124. [Google Scholar] [CrossRef]
- Bonilla, N.; Gutiérrez-Barranquero, J.A.; de Vicente, A.; Cazorla, F.M. Enhancing soil quality and plant health through suppressive organic amendments. Diversity 2012, 4, 475–491. [Google Scholar] [CrossRef]
- Zhao, J.; Ni, T.; Li, Y.; Xiong, W.; Ran, W.; Shen, B.; Zhang, R. Responses of bacterial communities in arable soils in a rice-wheat cropping system to different fertilizer regimes and sampling times. PLoS ONE 2014, 9, e85301. [Google Scholar] [CrossRef]
- Upadhyay, N.; Vishwakarma, K.; Singh, J.; Verma, R.K.; Prakash, V.; Jain, S.; Kumar, V.; Rani, R.; Tripathi, D.K.; Sharma, S. Plant-Microbe-Soil Interactions for Reclamation of Degraded Soils: Potential and Challenges In Phyto and Rhizo Remediation; Springer: Singapore, 2019; pp. 147–173. [Google Scholar]
- Murphy, D.V.; Stockdale, E.A.; Brookes, P.C.; Goulding, K.W. Impact of microorganisms on chemical transformations in soil. In Soil Biological Fertility; Springer: Berlin/Heidelberg, Germany, 2007; pp. 37–59. [Google Scholar]
- Khandare, R.N.; Chandra, R.; Pareek, N.; Raverkar, K.P. Carrier-based and liquid bioinoculants of Azotobacter and PSB saved chemical fertilizers in wheat (Triticumaestivum L.) and enhanced soil biological properties in Mollisols. J. Plant Nutr. 2020, 43, 36–50. [Google Scholar] [CrossRef]
- Sherpa, M.K.; Thombare, M.V.; Masih, H.; Lal, A.A.; Adhikari, A.; Thalai, R. Response of liquid biofertilizers on growth and yield of brinjal (Solanum melongena L.). J. Pharmacogn. Phytochem. 2019, 8, 1540–1544. [Google Scholar]
- Jackson, M.L. Soil Chemical Analysis; Prentice Hall of India Pvt. Ltd.: New Delhi, India, 1973. [Google Scholar]
- Walkey, A.; Black, T.A. An experimentation of vegetative method for determining soil organic matter and proposed modification of the chromic acid titration method. Soil Sci. 1934, 37, 38–39. [Google Scholar]
- Subbiah, B.V.; Asija, G.L. Rapid procedure for the estimation of the available nitrogen in soils. Curr. Sci. 1956, 25, 259–260. [Google Scholar]
- Olsen, S.R.; Cole, C.U.; Wattannable, F.; Sandean, D.A. Estimation of Available Phosphorous in Soil by Extraction with Sodium Bicarbonate; USDA Circulation: Washington, DC, USA, 1954; p. 939. [Google Scholar]
- Merwin, H.D.; Peech, M. Exchange ability of soil potassium in the sand, silt and clay fractions as influenced by the nature and complementary exchangeable cations. Soil Sci. Soc. Am. Proc. 1951, 15, 125–128. [Google Scholar] [CrossRef]
- Lindsay, W.H.; Norvell, W.A. Development of DTPA soil test for Zn, Fe, Mn and Cu. J. Soil Sci. Soc. Am. 1978, 42, 420–428. [Google Scholar] [CrossRef]
- Subba Rao, N.S. Soil Microorganisms and Plant Growth; Oxford and IBH publishing Company: New Delhi, India, 1999; pp. 1–333. [Google Scholar]
- Vance, E.D.; Brookes, P.C.; Jenkinson, D.S. An extraction method for measuring soil microbial biomass C. Soil Biol. Biochem. 1987, 19, 703–707. [Google Scholar] [CrossRef]
- Casida, L.E.; Klein, D.A.; Santoro, T. Soil dehydrogenase activity. Soil Sci. 1964, 98, 371–376. [Google Scholar] [CrossRef]
- Tatabai, B. Use of p-nitrophenyl phosphate for assay of soil phosphatase activity. Soil Biol. Biochem. 1969, 1, 301–317. [Google Scholar] [CrossRef]
- Hoffman, E. Methods of Enzymatic Analysis; Bergmeyer, H., Ed.; Academic Press: New York, NY, USA, 1965; pp. 219–221. [Google Scholar]
- Mohan, K.K.; Somasundharam, E.; Marimuthu, S. Influence of various organic inputs on growth and yield of snake gourd (Trichosanthes anguina L.). Int. J. Agric. Sci. 2016, 8, 3158–3161. [Google Scholar]
- Arahunashi, C.S. Influence of Organics on Growth, Yield and Quality of Tomato (Lycopersicum esculentum L. Mill.). Master’s Thesis, University of Agriculture Sciences, Dharwad, India, 2011. [Google Scholar]
- Boraiah, B.; Devakumar, N.; Shubha, S.; Palanna, K.B. Effect of panchagavya, jeevamrutha and cow urine on beneficial microorganisms and yield of capsicum (Capsicum annuum L. var. grossum). Int. J. Curr. Microbiol. 2017, 6, 3226–3234. [Google Scholar] [CrossRef]
- Choudhary, G.L.; Sharma, S.K.; Choudhary, S.; Singh, K.P.; Kaushik, M.K.; Bazaya, B.R. Effect of panchagavya on quality, nutrient content and nutrient uptake of organic blackgram [Vigna mungo (L.) Hepper]. J. Pharmacogn. Phytochem. 2017, 6, 1572–1575. [Google Scholar]
- Pawar, V.R.; Tambe, A.D.; Raut, S.A.; Udmale, K.B. Response of sweet corn (Zea mays var. Saccharata) cv. sugar 75 to different organic sources. Adv. Res. J. Crop Improv. 2012, 3, 22–25. [Google Scholar]
- Rao, T.K.; Reddy, S.D.; Naidu, J.G. The quality parameters of maize as affected by organic farming. Green Farming 2010, 1, 373–376. [Google Scholar]
- Muktamar, Z.; Adiprasetyo, T.; Yulia, S.; Sari, L.; Fahrurrozi, F.; Setyowati, N. Residual effect of vermicompost on sweet corn growth and selected chemical properties of soils from different organic farming practices. Int. J. Agric. Technol. 2018, 14, 1471–1482. [Google Scholar]
- Wani, K.A.; Rao, R.J. Effect of vermicompost on growth of brinjal plant (Solanum melongena) under field conditions. J. New Biol. Rep. 2012, 1, 25–28. [Google Scholar]
- Bhattarai, B.P.; Sapkota, B. Effect of organic nutrients management on yield of cucumber (Cucumis sativus) and its residual effect on soil. Int. J. Agric. Environ. Res. 2016, 2, 1768–1776. [Google Scholar]
- Antil, R.S.; Singh, M. Effects of organic manures and fertilizers on organic matter and nutrients status of the soil. Arch. Agron. Soil Sci. 2007, 53, 519–528. [Google Scholar] [CrossRef]
- Ewulo, B.; Ojeniyi, S.O. Effect of poultry manure on selected soil physical and chemical properties, growth, yield and nutrient status of tomato. Afr. J. Agric. Res. 2008, 3, 612–616. [Google Scholar]
- Aher, S.B.; Lakaria, B.L.; Kaleshananda, S.; Singh, A.B. Yield, nutrient uptake and economics of soybean–wheat cropping system under organic nutrient management in central. India. J. Plant Nutr. 2021, 45, 904–919. [Google Scholar] [CrossRef]
- Chen, D.; Yuan, L.; Liu, Y.; Ji, J.; Hou, H. Long-term application of manures plus chemical fertilizers sustained high rice yield and improved soil chemical and bacterial properties. Eur. J. Agron. 2017, 90, 34–42. [Google Scholar] [CrossRef]
- Kumar, S.; Dahiya, R.; Kumar, P.; Jhorar, B.S.; Phogat, V.K. Long-term effect of organic materials and fertilizers on soil properties in pearl millet-wheat cropping system. Indian J. Agric. Sci. 2012, 46, 161–166. [Google Scholar]
- Sharma, R.P.; Datt, N.; Chander, G. Effect of vermicompost, farmyard manure and chemical fertilizers on yield, nutrient uptake and soil fertility in okra (Abelmoschus esculentus)—Onion (Allium cepa) sequence in wet temperate zone of Himachal Pradesh. J. Indian Soc. Soil Sci. 2009, 57, 357–361. [Google Scholar]
- Jain, P.; Sharma, R.C.; Bhattacharya, P.; Banik, P. Effect of new organic supplement (panchagavya) on seed germination and soil quality. Environ. Monit. Assess. 2014, 186, 1999–2011. [Google Scholar] [CrossRef]
- Moharana, P.C.; Sharma, B.M.; Biswas, D.R. Changes in the soil properties and availability of micronutrients after six-year application of organic and chemical fertilizers using STCR-based targeted yield equations under pearl millet-wheat cropping system. J. Plant Nutr. 2017, 40, 165–176. [Google Scholar] [CrossRef]
- Zeid, H.A.; Wafaa, H.M.; Seoud, I.I.A.E.; Alhadad, W.A.A. Effect of organic materials and inorganic fertilizers on the growth, mineral composition and soil fertility of radish plants (Raphine’s sativus) grown in sandy soil. Middle East J. Agric. Res. 2015, 4, 77–87. [Google Scholar]
- Ram, R.A.; Singha, A.; Vaish, S. Microbial characterization of on-farm produced bio-enhancers used in organic farming. Indian J. Agric. Sci. 2018, 88, 35–40. [Google Scholar]
- Vaish, S.; Garg, N.; Ahmad, I.Z. Microbial basis of organic farming systems with special reference to biodynamic preparations. Indian J. Agric. Sci. 2020, 90, 1219–1225. [Google Scholar] [CrossRef]
- Aulakh, C.S.; Singh, H.; Walia, S.S.; Phutela, R.P.; Singh, G. Evaluation of microbial culture (jeevamrit) preparation and its effect on productivity of field crops. Indian J. Agron. 2013, 58, 182–186. [Google Scholar]
- Gałązka, A.; Gawryjołek, K.; Perzyński, A.; Gałązka, R.; Księżak, J. Changes in enzymatic activities and microbial communities in soil under long-term maize monoculture and crop rotation. Pol. J. Environ. Stud. 2017, 26, 39–46. [Google Scholar] [CrossRef]
- Chandrakala, M.; Hebsur, N.S.; Bidari, B.I.; Radder, B.M. Effect of FYM and fermented liquid manures on nutrients uptake by chilli (Capsicum annum L.) and soil nutrient status at harvest. Asian J. Hort. 2007, 4, 19–24. [Google Scholar]
- Logah, V.; Safo, E.Y.; Quansah, C.; Danso, I. Soil microbial biomass carbon, nitrogen and phosphorus dynamics under different amendments and cropping systems in the semi—Deciduous forest zone of Ghana. West Afr. J. Appl. Ecol. 2010, 17, 121–133. [Google Scholar]
- Tomar, A.S. Study the effect of integrated nutrient management on carbon pools (organic & microbial biomass carbon) in pearl millet- wheat cropping system. Int. J. Chem. Stud. 2017, 5, 2019–2022. [Google Scholar]
- Nakhro, N.; Dkhar, M.S. Impact of organic and inorganic fertilizers on microbial populations and biomass carbon in paddy field soil. J. Agron. 2010, 9, 102–110. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).